Wearable Laser Doppler Flowmetry Sensor: A Feasibility Study with Smoker and Non-Smoker Volunteers
Abstract
1. Introduction
2. Materials and Methods
2.1. Wearable Laser Doppler Flowmetry Monitor
2.2. Study Design
2.3. Testing Procedure
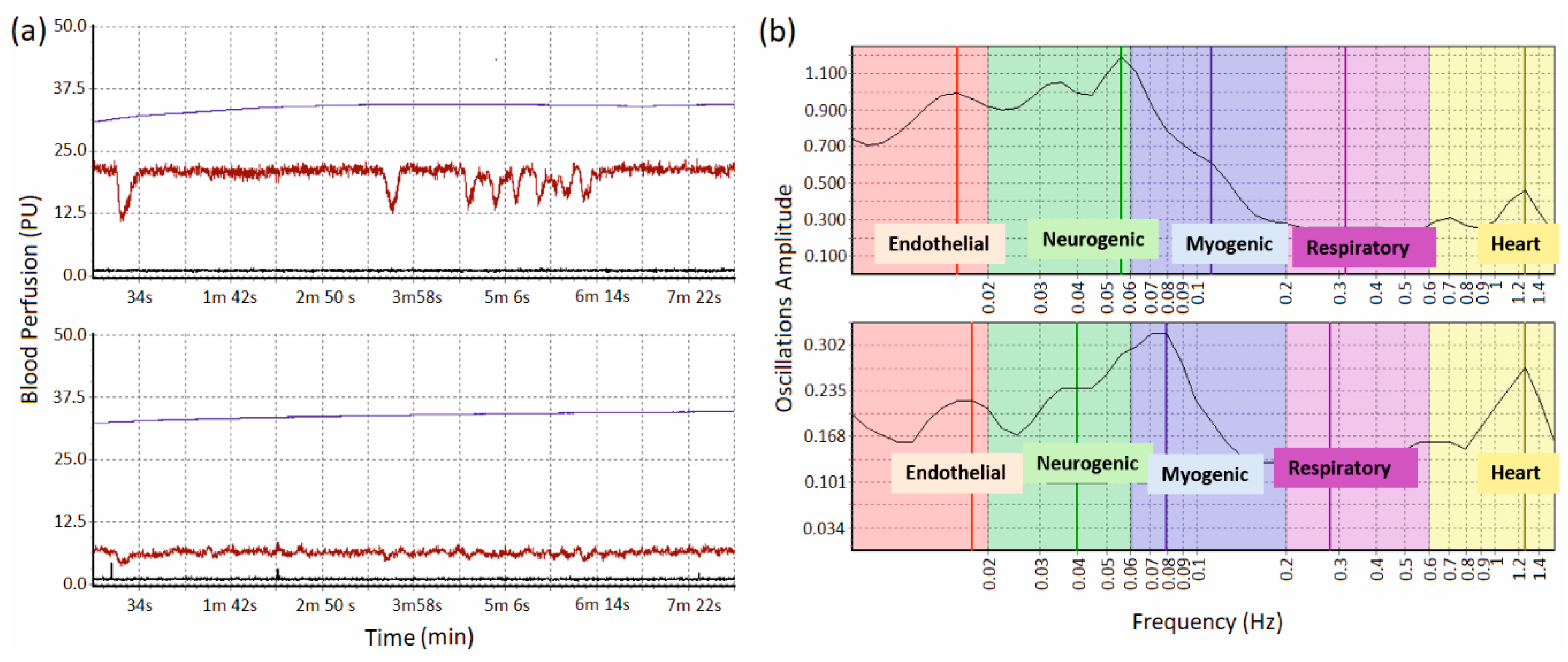
2.4. Data Proccesing and Analysis
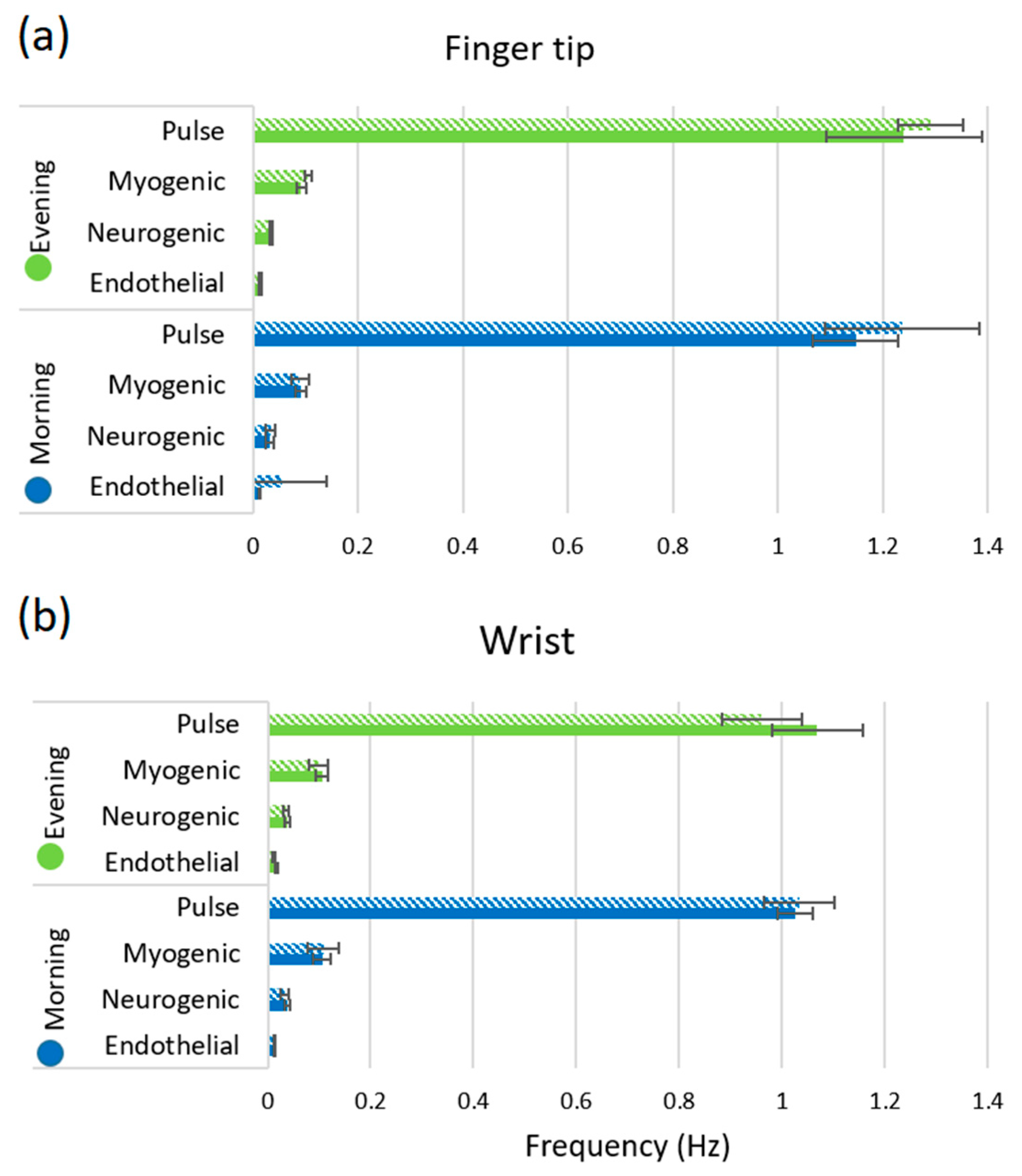
3. Results
4. Discussion
5. Study Limitations and Future Directions
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Tobacco Control Local Policy Statement—Cancer Research UK. Available online: https://www.cancerresearchuk.org/sites/default/files/tobacco_control_local_policy_statement.pdf (accessed on 29 October 2020).
- Branca, F.; Lartey, A.; Oenema, S.; Aguayo, V.; Stordalen, G.A.; Richardson, R.; Arvelo, M.; Afshin, A. Transforming the food system to fight non-communicable diseases. BMJ 2019, 364, l296. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S. New approaches in smoking cessation. Indian Heart J. 2008, 60, B34–B37. [Google Scholar] [PubMed]
- Blackburn, H.; Brozek, J.; Taylor, H.L. Common circulatory measurements in smokers and nonsmokers. Circulation 1960, 22, 1112–1124. [Google Scholar] [CrossRef]
- Benowitz, N.L.; Burbank, A.D. Cardiovascular toxicity of nicotine: Implications for electronic cigarette use. Trends Cardiovasc. Med. 2016, 26, 515–523. [Google Scholar] [CrossRef]
- Guyton, A.C.; Hall, J.E. Textbook of Medical Physiology, 11th ed.; Elsevier Saunders: Philadelphia, PA, USA, 2006; p. 1116. [Google Scholar]
- Tayade, M.C.; Kulkarni, N.B. A comparative study of resting heart rate in smokers and nonsmokers. Int. J. Cur. Res. Rev. 2012, 4, 59–62. [Google Scholar]
- Joseph, P.; Leong, D.; McKee, M.; Anand, S.S.; Schwalm, J.-D.; Teo, K.; Mente, A.; Yusuf, S. Reducing the global burden of cardiovascular disease, part 1: The epidemiology and risk factors. Circ. Res. 2017, 121, 677–694. [Google Scholar] [CrossRef]
- Ezzati, M.; Lopez, A.D.; Rodgers, A.; Hoorn, S.V.; Murray, C.J. the Comparative Risk Assessment Collaborating Group. Selected major risk factors and global and regional burden of disease. Lancet 2002, 360, 1347–1360. [Google Scholar] [CrossRef]
- Loktionova, Y.I.; Zherebtsov, E.A.; Zharkikh, E.V.; Kozlov, I.O.; Zherebtsova, A.I.; Sidorov, V.V.; Sokolovski, S.G.; Rafailov, I.E.; Dunaev, A.V.; Rafailov, E.U. Studies of age-related changes in blood perfusion coherence using wearable blood perfusion sensor system. Proc. SPIE 2019, 11075, 1107507. [Google Scholar]
- Zherebtsov, E.A.; Zharkikh, E.V.; Kozlov, I.O.; Zherebtsova, A.I.; Loktionova, Y.I.; Chichkov, N.B.; Rafailov, I.E.; Sidorov, V.V.; Sokolovski, S.G.; Dunaev, A.V.; et al. Novel wearable VCSEL-based sensors for multipoint measurements of blood perfusion. Proc. SPIE 2019, 10877, 1087708. [Google Scholar]
- Fredriksson, I.; Fors, C.; Johansson, J. Laser Doppler Flowmetry-a Theoretical Framework Department of Biomedical Engineering; Linköping University: Linköping, Sweden, 2007; pp. 1–22. [Google Scholar]
- Dunaev, A.V.; Sidorov, V.V.; Krupatkin, A.I.; Rafailov, I.E.; Palmer, S.G.; Stewart, N.A.; Sokolovski, S.G.; Rafailov, E.U. Investigating tissue respiration and skin microhaemocirculation under adaptive changes and the synchronization of blood flow and oxygen saturation rhythms. Physiol. Meas. 2014, 35, 607. [Google Scholar] [CrossRef]
- Fushimi, H.; Kubo, M.; Inoue, T.; Yamada, Y.; Matsuyama, Y.; Kameyama, M. Peripheral vascular reactions to smoking—Profound vasoconstriction by atherosclerosis. Diabetes Res. Clin. Pract. 1998, 42, 29–34. [Google Scholar] [CrossRef]
- Uehara, K.; Sone, R.; Yamazaki, F. Cigarette smoking following a prolonged mental task exaggerates vasoconstriction in glabrous skin in habitual smokers. J. UOEH 2010, 32, 303–316. [Google Scholar] [CrossRef] [PubMed]
- Meekin, T.; Wilson, R.; Scott, D.; Ide, M.; Palmer, R. Laser Doppler flowmeter measurement of relative gingival and forehead skin blood flow in light and heavy smokers during and after smoking. J. Clin. Periodontol. 2000, 27, 236–242. [Google Scholar] [CrossRef] [PubMed]
- Palmer, R.; Scott, D.; Meekin, T.; Poston, R.; Odell, E.; Wilson, R. Potential mechanisms of susceptibility to periodontitis in tobacco smokers. J. Periodontal Res. 1999, 34, 363–369. [Google Scholar] [CrossRef] [PubMed]
- Tur, E.; Yosipovitch, G.; Oren-Vulfs, S. Chronic and acute effects of cigarette smoking on skin blood flow. Angiology 1992, 43, 328–335. [Google Scholar] [CrossRef]
- Newson, T.P.; Obeid, A.; Wolton, R.S.; Boggett, D.; Rolfe, P. Laser Doppler velocimetry: The problem of fibre movement artefact. J. Biomed. Eng. 1987, 9, 169–172. [Google Scholar] [CrossRef]
- Rajan, V.; Varghese, B.; van Leeuwen, T.G.; Steenbergen, W. Review of methodological developments in laser Doppler flowmetry. Lasers Med. Sci. 2009, 24, 269–283. [Google Scholar] [CrossRef]
- Dremin, V.; Zherebtsov, E.; Sidorov, V.; Krupatkin, A.; Makovik, I.; Zherebtsova, A.; Zharkikh, E.; Potapova, E.; Dunaev, A.; Doronin, A.; et al. Multimodal optical measurement for study of lower limb tissue viability in patients with diabetes mellitus. J. Biomed. Opt. 2017, 22, 085003. [Google Scholar] [CrossRef]
- Makovik, I.N.; Dunaev, A.V.; Dremin, V.V.; Krupatkin, A.I.; Sidorov, V.V.; Khakhicheva, L.S.; Muradyan, V.F.; Pilipenko, O.V.; Rafailov, I.E.; Litvinova, K.S. Detection of angiospastic disorders in the microcirculatory bed using laser diagnostics technologies. J. Innov. Opt. Heal. Sci. 2017, 11, 1750016. [Google Scholar] [CrossRef]
- Goltsov, A.; Anisimova, A.V.; Zakharkina, M.; Krupatkin, A.I.; Sidorov, V.V.; Sokolovski, S.G.; Rafailov, E. Bifurcation in blood oscillatory rhythms for patients with ischemic stroke: A small scale clinical trial using laser doppler flowmetry and computational modeling of vasomotion. Front. Physiol. 2017, 8, 160. [Google Scholar] [CrossRef]
- Tankanag, A.; Chemeris, N. Application of the adaptive wavelet transform for analysis of blood flow oscillations in the human skin. Phys. Med. Biol. 2008, 53, 5967. [Google Scholar] [CrossRef] [PubMed]
- Stefanovska, A.; Bracic, M.; Kvernmo, H.D. Wavelet analysis of oscillations in the peripheral blood circulation measured by laser Doppler technique. IEEE Trans. Biomed. Eng. 1999, 46, 1230–1239. [Google Scholar] [CrossRef] [PubMed]
- Dremin, V.; Kozlov, I.; Volkov, M.; Margaryants, N.; Potemkin, A.; Zherebtsov, E.; Dunaev, A.; Gurov, I. Dynamic evaluation of blood flow microcirculation by combined use of the laser Doppler flowmetry and high-speed videocapillaroscopy methods. J. Biophotonics 2019, 12, e201800317. [Google Scholar] [CrossRef] [PubMed]
- Krasnikov, G.V.; Tyurina, M.Y.; Tankanag, A.V.; Piskunova, G.M.; Chemeris, N.K. Analysis of heart rate variability and skin blood flow oscillations under deep controlled breathing. Respir. Physiol. Neurobiol. 2013, 185, 562–570. [Google Scholar] [CrossRef] [PubMed]
- Trembach, N.; Zabolotskikh, I. Breath-holding test in evaluation of peripheral chemoreflex sensitivity in healthy subjects. Respir. Physiol. Neurobiol. 2017, 235, 79–82. [Google Scholar] [CrossRef]
- Charan, J.; Biswas, T. How to calculate sample size for different study designs in medical research? Indian J. Psychol. Med. 2013, 35, 121–126. [Google Scholar] [CrossRef]
- Endothelial Dysfunction. Available online: https://www.diabetes.co.uk/diabetes-complications/endothelial-dysfunction.html (accessed on 29 October 2020).
- U.S. Department of Health and Human Services. How Tobacco Smoke Causes Disease: The Biology and Behavioral Basis for Smoking-Attributable Disease: A Report of the Surgeon General. Available online: https://www.ncbi.nlm.nih.gov/books/NBK53017/ (accessed on 29 October 2020).



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Saha, M.; Dremin, V.; Rafailov, I.; Dunaev, A.; Sokolovski, S.; Rafailov, E. Wearable Laser Doppler Flowmetry Sensor: A Feasibility Study with Smoker and Non-Smoker Volunteers. Biosensors 2020, 10, 201. https://doi.org/10.3390/bios10120201
Saha M, Dremin V, Rafailov I, Dunaev A, Sokolovski S, Rafailov E. Wearable Laser Doppler Flowmetry Sensor: A Feasibility Study with Smoker and Non-Smoker Volunteers. Biosensors. 2020; 10(12):201. https://doi.org/10.3390/bios10120201
Chicago/Turabian StyleSaha, Mou, Viktor Dremin, Ilya Rafailov, Andrey Dunaev, Sergei Sokolovski, and Edik Rafailov. 2020. "Wearable Laser Doppler Flowmetry Sensor: A Feasibility Study with Smoker and Non-Smoker Volunteers" Biosensors 10, no. 12: 201. https://doi.org/10.3390/bios10120201
APA StyleSaha, M., Dremin, V., Rafailov, I., Dunaev, A., Sokolovski, S., & Rafailov, E. (2020). Wearable Laser Doppler Flowmetry Sensor: A Feasibility Study with Smoker and Non-Smoker Volunteers. Biosensors, 10(12), 201. https://doi.org/10.3390/bios10120201







