Study of the Thermal Annealing on Structural and Morphological Properties of High-Porosity A-WO3 Films Synthesized by HFCVD
Abstract
:1. Introduction
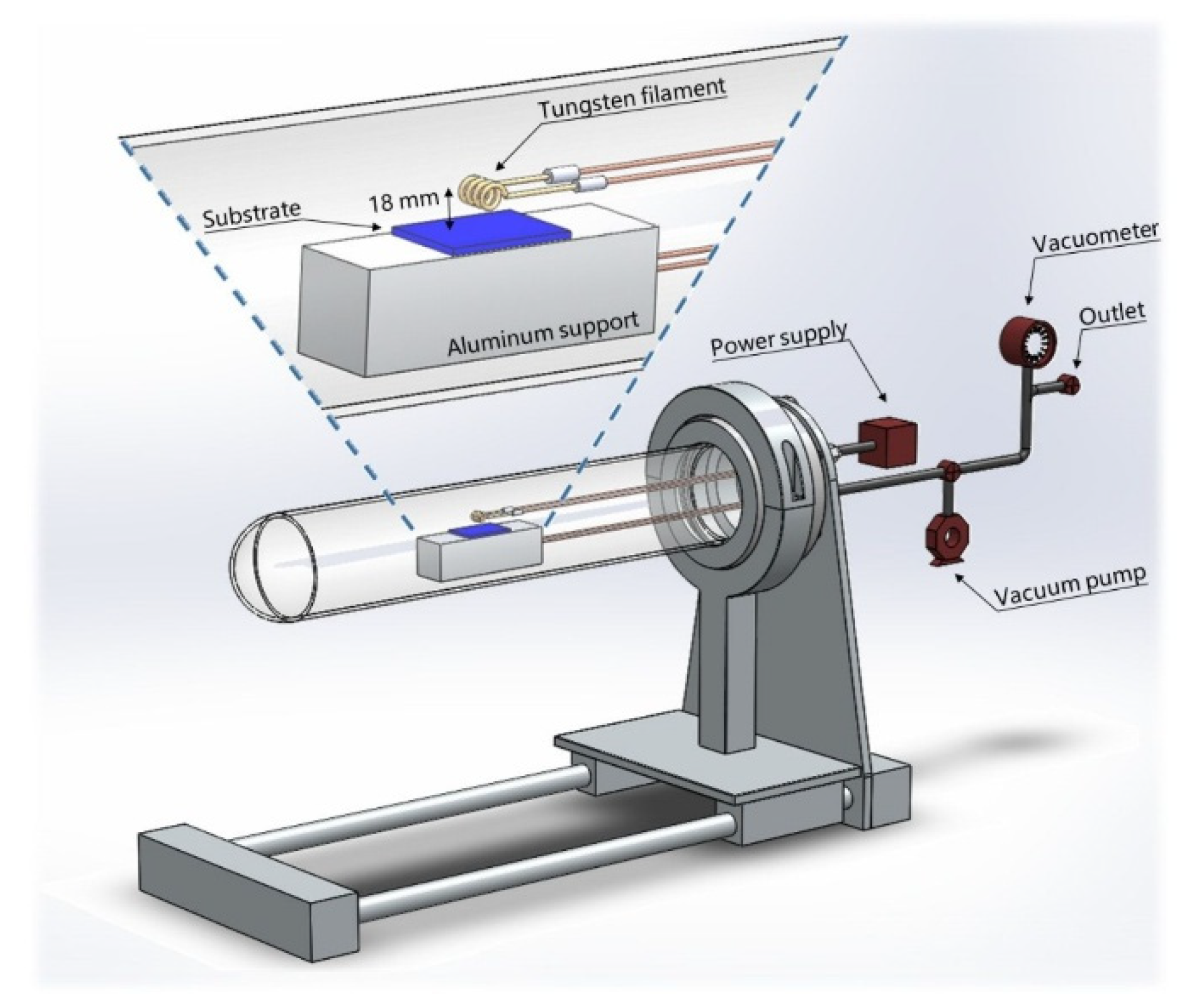
2. Materials and Methods
3. Results and Discussion
3.1. Morphology
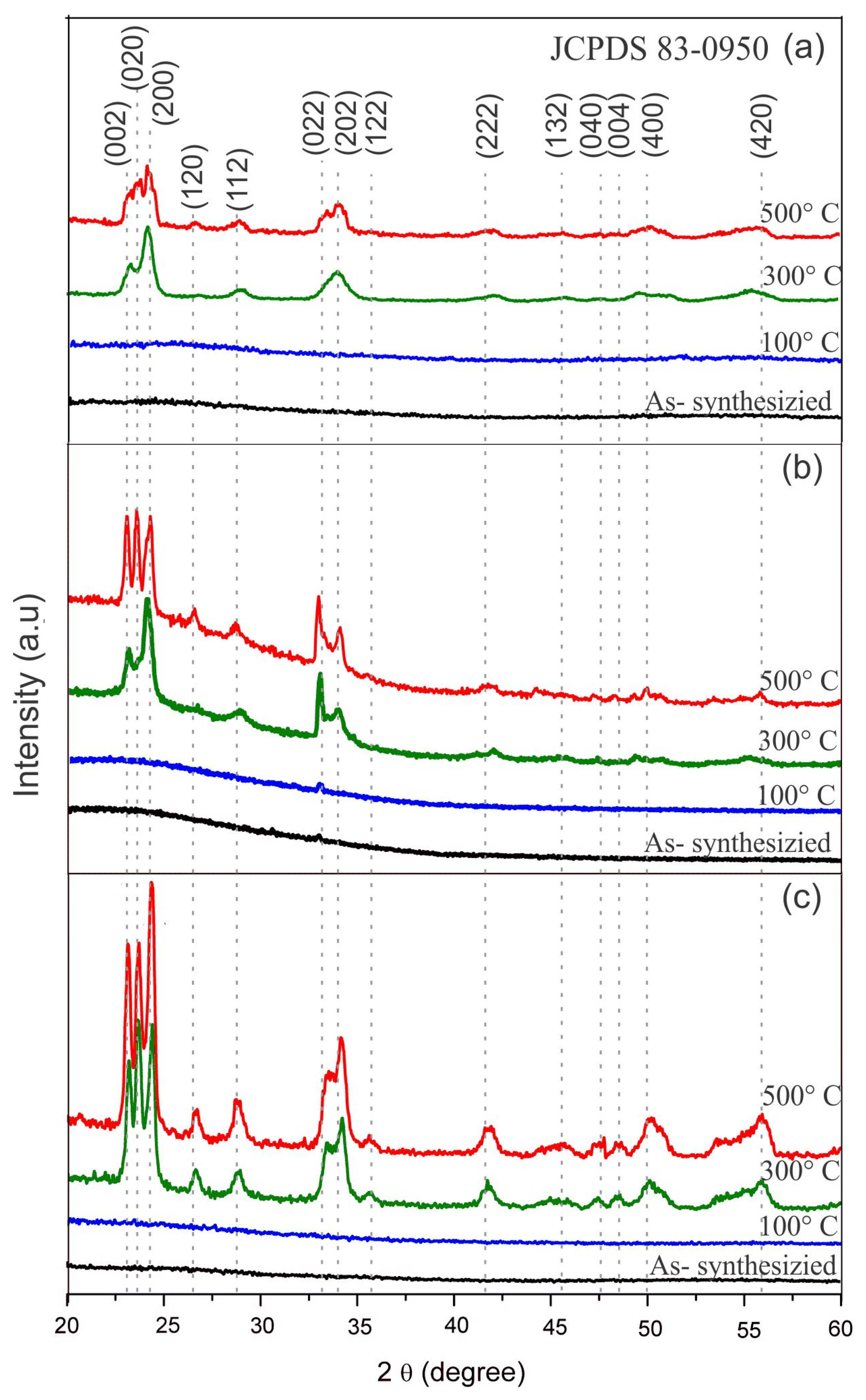
3.2. Crystalline Structure
3.3. Specific Surface Area
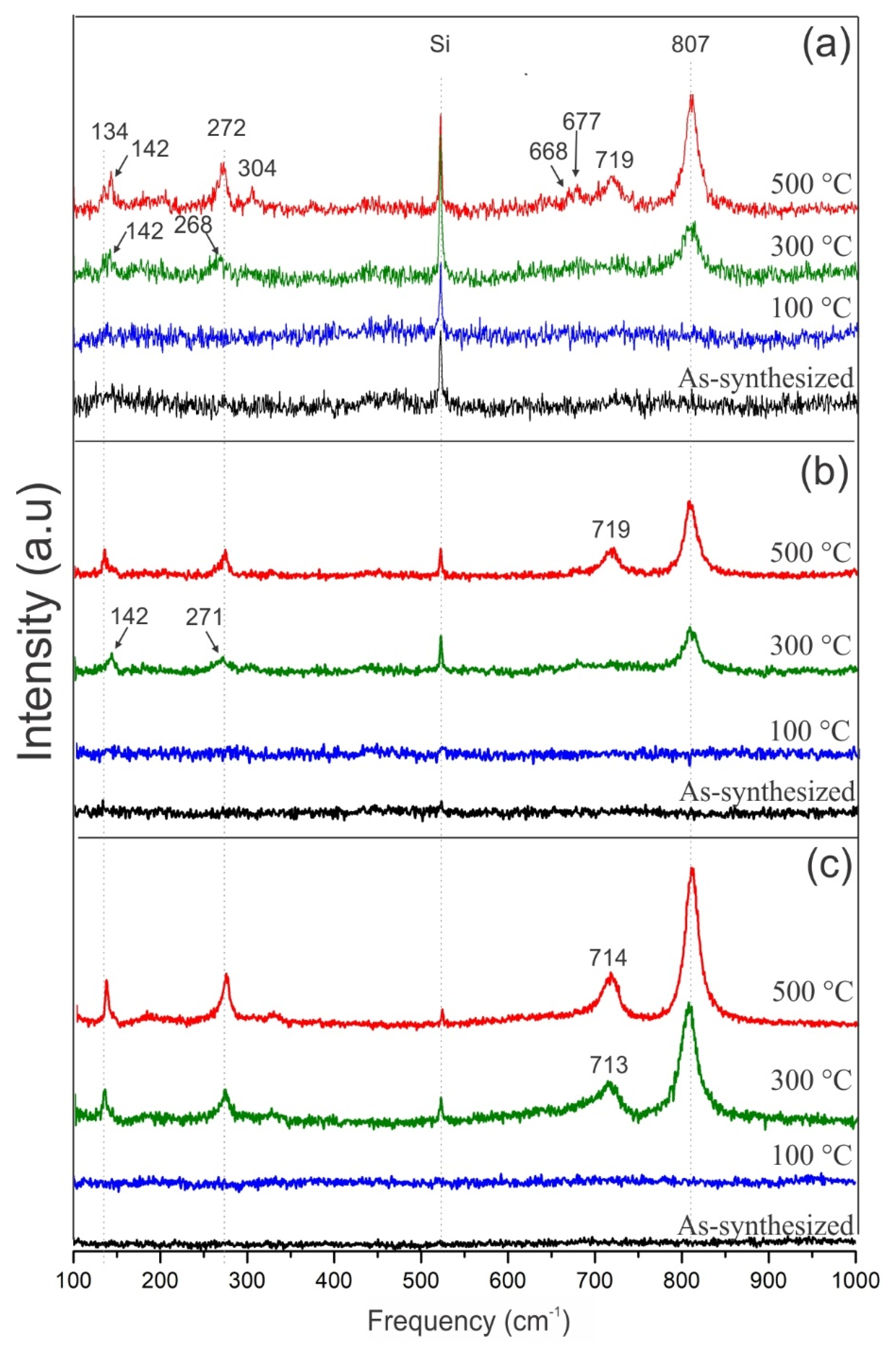
3.4. Micro-Raman Analysis
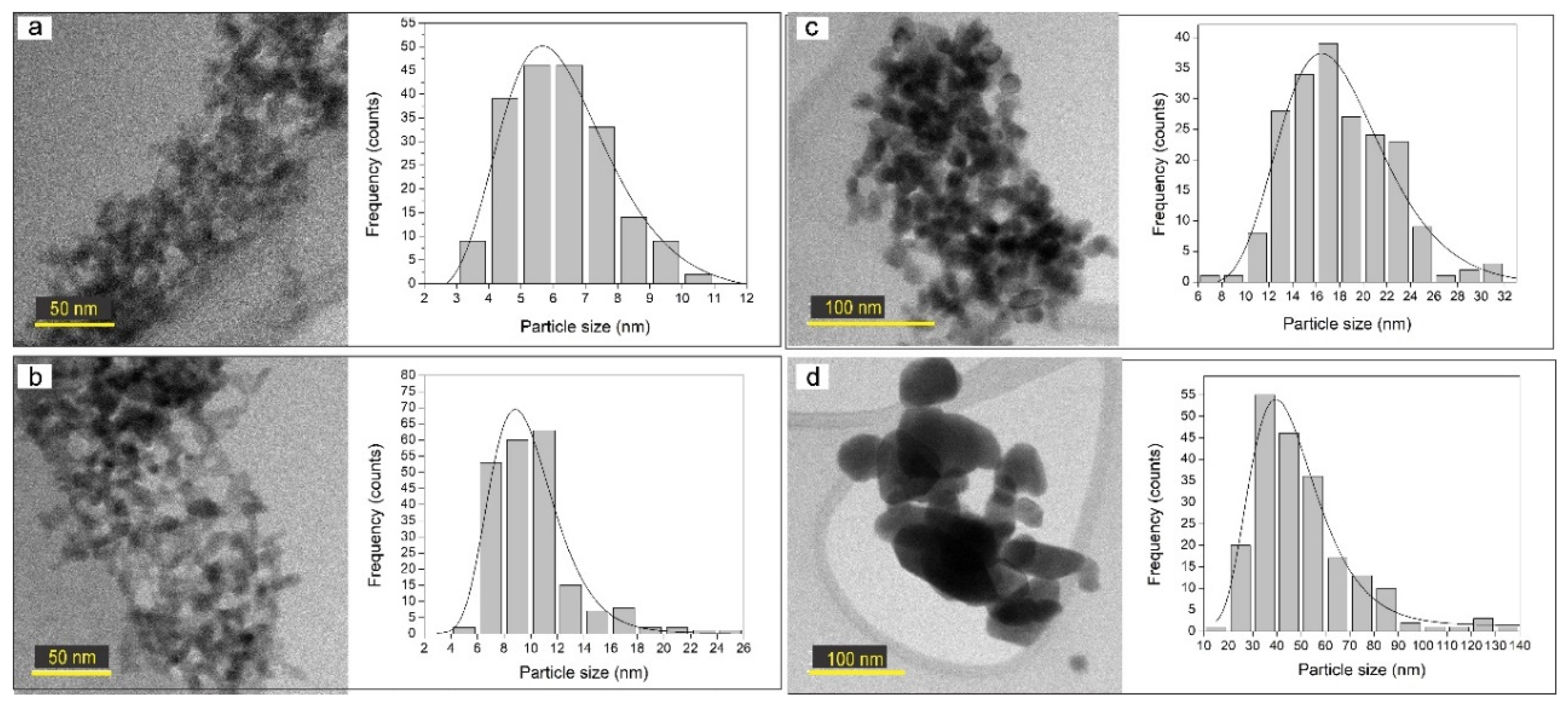
3.5. Transmition Electron Microscopy (TEM) Analysis
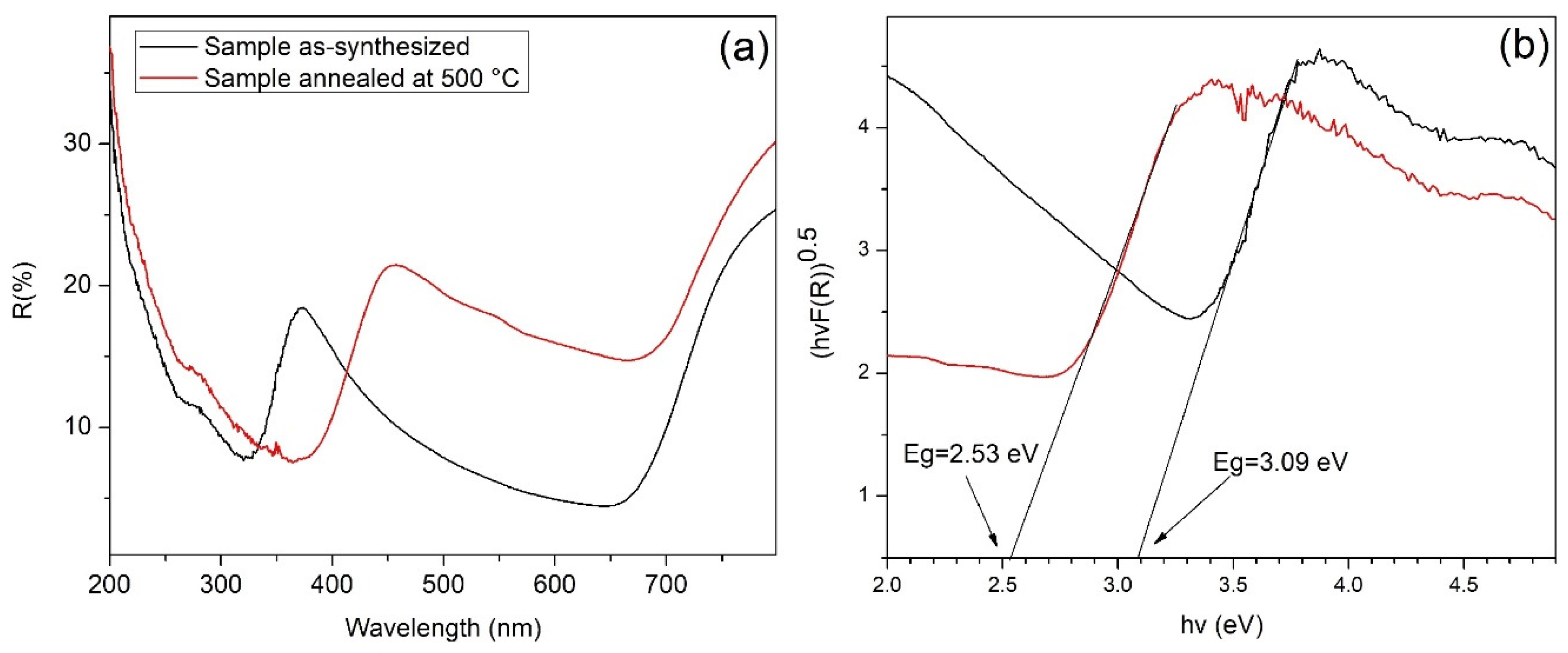
3.6. UV–Vis Diffuse Reflectance and Bandgap
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Reyes, L.F.; Saukko, S.; Hoel, A.; Lantto, V.; Granqvist, C.G. Structure engineering of WO3 nanoparticles for porous film applications by advanced reactive gas deposition. J. Eur. Ceram. Soc. 2004, 24, 1415–1419. [Google Scholar] [CrossRef]
- Takács, M.; Pap, A.E. Gas Sensitivity of Sol-gel Prepared Mesoporous WO3 thin Film. Proc. Procedia Eng. 2016, 168, 289–292. [Google Scholar] [CrossRef]
- Djaoued, Y.; Balaji, S.; Brüning, R. Electrochromic Devices Based on Porous Tungsten Oxide Thin Films. J. Nanomater. 2012, 2012, 7. [Google Scholar] [CrossRef]
- Zhu, T.; Chong, M.N.; Chan, E.S. Nanostructured tungsten trioxide thin films synthesized for photoelectrocatalytic water oxidation: A review. ChemSusChem 2014, 7, 2974–2997. [Google Scholar] [CrossRef] [PubMed]
- Kikuchi, T.; Kawashima, J.; Natsui, S.; Suzuki, R.O. Fabrication of porous tungsten oxide via anodizing in an ammonium nitrate/ethylene glycol/water mixture for visible light-driven photocatalyst. Appl. Surf. Sci. 2017, 422, 130–137. [Google Scholar] [CrossRef]
- Wang, X.; Chen, F.; Yang, M.; Guo, L.; Xie, N.; Kou, X.; Song, Y.; Wang, Q.; Sun, Y.; Lu, G. Dispersed WO3 nanoparticles with porous nanostructure for ultrafast toluene sensing. Sens. Actuators B Chem. 2019, 289, 195–206. [Google Scholar] [CrossRef]
- Jain, R.; Lei, Y.; Maric, R. Ultra-low NO2 detection by gamma WO3 synthesized by Reactive Spray Deposition Technology. Sens. Actuators B Chem. 2016, 236, 163–172. [Google Scholar] [CrossRef]
- Thongpan, W.; Louloudakis, D.; Pooseekheaw, P.; Kumpika, T.; Kantarak, E.; Panthawan, A.; Tuantranont, A.; Thongsuwan, W.; Singjai, P. Electrochromic properties of tungsten oxide films prepared by sparking method using external electric field. Thin Solid Films 2019, 682, 135–141. [Google Scholar] [CrossRef]
- Shen, Y.; Yamazaki, T.; Liu, Z.; Meng, D.; Kikuta, T.; Nakatani, N. Influence of effective surface area on gas sensing properties of WO3 sputtered thin films. Thin Solid Films 2009, 517, 2069–2072. [Google Scholar] [CrossRef]
- Zhou, X.; Cheng, X.; Zhu, Y.; Elzatahry, A.A.; Alghamdi, A.; Deng, Y.; Zhao, D. Ordered porous metal oxide semiconductors for gas sensing. Chin. Chem. Lett. 2018, 29, 405–416. [Google Scholar] [CrossRef]
- Lakhtakia, A.; Martín-Palma, R.J. Vapor Deposition Techniques. In Engineered Biomimicry, 1st ed.; Elsevier: Amsterdam, The Netherlands, 2013; pp. 383–398. [Google Scholar]
- Schropp, R.E.I. Industrialization of Hot Wire Chemical Vapor Deposition for thin film applications. Proc. Thin Solid Films 2015, 595, 272–283. [Google Scholar] [CrossRef] [Green Version]
- Schropp, R.E.I. Frontiers in HWCVD. Thin Solid Films 2009, 517, 3415–3419. [Google Scholar] [CrossRef]
- Choy, K.L. Chemical vapour deposition of coatings. Prog. Mater. Sci. 2003, 48, 57–170. [Google Scholar] [CrossRef]
- Buch, V.R.; Chawla, A.K.; Rawal, S.K. Review on electrochromic property for WO3 thin films using different deposition techniques. Mater. Today Proc. 2016, 3, 1429–1437. [Google Scholar] [CrossRef]
- Madhuri, K.V.; Bujji Babu, M. Influence of substrate temperature on growth and Electrochromic properties of WO3 thin films. Optik (Stuttg). 2018, 174, 470–480. [Google Scholar] [CrossRef]
- Shendage, S.S.; Patil, V.L.; Vanalakar, S.A.; Patil, S.P.; Harale, N.S.; Bhosale, J.L.; Kim, J.H.; Patil, P.S. Sensitive and selective NO2 gas sensor based on WO3 nanoplates. Sens. Actuators B Chem. 2017, 240, 426–433. [Google Scholar] [CrossRef]
- Chi, X.; Liu, C.; Liu, L.; Li, Y.; Wang, Z.; Bo, X.; Liu, L.; Su, C. Tungsten trioxide nanotubes with high sensitive and selective properties to acetone. Sens. Actuators B Chem. 2014, 194, 33–37. [Google Scholar] [CrossRef]
- Granqvist, C.G. Electrochromics for smart windows: Oxide-based thin films and devices. Thin Solid Films 2014, 564, 1–38. [Google Scholar] [CrossRef]
- Righettoni, M.; Tricoli, A.; Pratsinis, S.E. Si:WO3 sensors for highly selective detection of acetone for easy diagnosis of diabetes by breath analysis. Anal. Chem. 2010, 82, 3581–3587. [Google Scholar] [CrossRef]
- Liu, H.; Guo, W.; Li, Y.; He, S.; He, C. Photocatalytic degradation of sixteen organic dyes by TiO2/WO3-coated magnetic nanoparticles under simulated visible light and solar light. J. Environ. Chem. Eng. 2018, 6, 59–67. [Google Scholar] [CrossRef]
- Vidmar, T.; Topič, M.; Dzik, P.; Opara Krašovec, U. Inkjet printing of sol-gel derived tungsten oxide inks. Sol. Energy Mater. Sol. Cells 2014, 125, 87–95. [Google Scholar] [CrossRef]
- Feng, P.X.; Wang, X.P.; Zhang, H.X.; Yang, B.Q.; Wang, Z.B.; González-Berríos, A.; Morell, G.; Weiner, B. Study of the structural evolutions of crystalline tungsten oxide films prepared using hot-filament CVD. J. Phys. D. Appl. Phys. 2007, 40, 5239. [Google Scholar] [CrossRef]
- Deshpande, R.; Lee, S.H.; Mahan, A.H.; Parilla, P.A.; Jones, K.M.; Norman, A.G.; To, B.; Blackburn, J.L.; Mitra, S.; Dillon, A.C. Optimization of crystalline tungsten oxide nanoparticles for improved electrochromic applications. Solid State Ion. 2007, 178, 895–900. [Google Scholar] [CrossRef]
- Lou, J.; Ye, B.J.; Weng, H.M.; Du, H.J.; Wang, Z.B.; Wang, X.P. The influence of filament temperature and oxygen concentration on tungsten oxide nanostructures by hot filament metal oxide deposition. J. Phys. D Appl. Phys. 2008, 41, 155410. [Google Scholar] [CrossRef]
- Pal, S.; Jacob, C. The influence of substrate temperature variation on tungsten oxide thin film growth in an HFCVD system. Appl. Surf. Sci. 2007, 253, 3317–3325. [Google Scholar] [CrossRef]
- Jafari, A.; Ghoranneviss, M.; Salar Elahi, A. HFCVD Application for Growth of Monoclinic Tungsten Trioxide Crystal Nano-walls. J. Inorg. Organomet. Polym. Mater. 2016, 26, 254–258. [Google Scholar] [CrossRef]
- Chacón, C.; Rodríguez-Pérez, M.; Oskam, G.; Rodríguez-Gattorno, G. Synthesis and characterization of WO3 polymorphs: Monoclinic, orthorhombic and hexagonal structures. J. Mater. Sci. Mater. Electron. 2015, 26, 5526–5531. [Google Scholar] [CrossRef]
- Migas, D.B.; Shaposhnikov, V.L.; Rodin, V.N.; Borisenko, V.E. Tungsten oxides. I. Effects of oxygen vacancies and doping on electronic and optical properties of different phases of WO3. J. Appl. Phys. 2010, 108, 093713. [Google Scholar] [CrossRef]
- Woodward, P.M.; Sleight, A.W.; Vogt, T. Ferroelectric Tungsten Trioxide. J. Solid State Chem. 1997, 131, 9–17. [Google Scholar] [CrossRef]
- Diehl, R.; Brandt, G.; Salje, E. The crystal structure of triclinic WO3. Acta Cryst. B 1978, 34, 1105–1111. [Google Scholar] [CrossRef]
- Tanisaki, S. Crystal Structure of Monoclinic Tungsten Trioxide at Room Temperature. J. Phys. Soc. Jpn. 1960, 15, 573–581. [Google Scholar] [CrossRef]
- Salje, E. The orthorhombic phase of WO3. Acta Cryst. B 1977, 33, 574–577. [Google Scholar] [CrossRef]
- Kehl, W.L.; Hay, R.G.; Wahl, D. The structure of tetragonal tungsten trioxide. J. Appl. Phys. 1952, 23, 212–215. [Google Scholar] [CrossRef]
- Zheng, H.; Ou, J.Z.; Strano, M.S.; Kaner, R.B.; Mitchell, A.; Kalantar-Zadeh, K. Nanostructured tungsten oxide—Properties, synthesis, and applications. Adv. Funct. Mater. 2011, 21, 2175–2196. [Google Scholar] [CrossRef]
- Boulova, M.; Lucazeau, G. Crystallite Nanosize Effect on the Structural Transitions of WO3 Studied by Raman Spectroscopy. J. Solid State Chem. 2002, 167, 425–434. [Google Scholar] [CrossRef]
- Migas, D.B.; Shaposhnikov, V.L.; Borisenko, V.E. Tungsten oxides. II. the metallic nature of Magnéli phases. J. Appl. Phys. 2010, 108, 093714. [Google Scholar] [CrossRef]
- Al Mohammad, A.; Gillet, M. Phase transformations in WO3 thin films during annealing. Thin Solid Films 2002, 408, 302–309. [Google Scholar] [CrossRef]
- Thummavichai, K.; Xia, Y.; Zhu, Y. Recent progress in chromogenic research of tungsten oxides towards energy-related applications. Prog. Mater. Sci. 2017, 88, 281–324. [Google Scholar] [CrossRef]
- Denardo, B. Temperature of a lightbulb filament. Phys. Teach. 2002, 40, 101–105. [Google Scholar] [CrossRef]
- Goiz, O.; Chávez, F.; Zaca-Morán, P.; Ortega-Mendoza, J.G.; Pérez-Sánchez, G.F.; Morales, N.; Felipe, C.; Peña-Sierra, R. Tungsten nanostructured thin films obtained via HFCVD. Proc. SPIE Nanostruct. Thin Films IV 2011, 8104, 810415. [Google Scholar]
- Saleem, M.; Al-Kuhaili, M.F.; Durrani, S.M.A.; Hendi, A.H.Y.; Bakhtiari, I.A.; Ali, S. Influence of hydrogen annealing on the optoelectronic properties of WO3 thin films. Int. J. Hydrog. Energy 2015, 40, 12343–12351. [Google Scholar] [CrossRef]
- Chávez, F.; Felipe, C.; Lima, E.; Lara, V.; Ángeles-Chávez, C.; Hernandez, M.A. Preparation of self-organized porous tungsten oxide using HFCVD technique. Mater. Chem. Phys. 2010, 120, 36–41. [Google Scholar] [CrossRef]
- Chen, H.L.; Lu, Y.M.; Hwang, W.S. Effect of film thickness on structural and electrical properties of sputter-deposited nickle oxide films. Mater. Trans. 2005, 46, 872–879. [Google Scholar] [CrossRef]
- Fardindoost, S.; Iraji zad, A.; Rahimi, F.; Ghasempour, R. Pd doped WO 3 films prepared by sol-gel process for hydrogen sensing. Int. J. Hydrog. Energy 2010, 35, 854–860. [Google Scholar] [CrossRef]
- Yang, B.; Barnes, P.R.F.; Bertram, W.; Luca, V. Strong photoresponse of nanostructured tungsten trioxide films prepared via a sol-gel route. J. Mater. Chem. 2007, 26, 2722–2729. [Google Scholar] [CrossRef]
- Altanany, S.M.; Gondal, M.A.; Baig, U. Synthesis and characterization of CuO/WO3 nanocomposite using hybrid method: Simple precipitation and pulsed laser ablation in liquids technique. AIP Conf. Proc. 2018, 1976, 020014. [Google Scholar]
- Madhukar, P.; Shanmukhi Jyothi, D.; Jayababu, N.; Ramana Reddy, M.V. Influence of annealing temperature on structural and dielectric properties of e-beam evaporated WO3 thin films. Proc. Mater. Today Proc. 2016, 3, 4199–4204. [Google Scholar] [CrossRef]
- Sahai, A.; Goswami, N. Structural and vibrational properties of ZnO nanoparticles synthesized by the chemical precipitation method. Phys. E Low-Dimens. Syst. Nanostruct. 2014, 58, 130–137. [Google Scholar] [CrossRef]
- Chung, Y.K.; Kim, M.H.; Um, W.S.; Lee, H.S.; Song, J.K.; Choi, S.C.; Yi, K.M.; Lee, M.J.; Chung, K.W. Gas sensing properties of WO3 thick film for NO2 gas dependent on process condition. Sens. Actuators B Chem. 1999, 60, 49–56. [Google Scholar] [CrossRef]
- Ram, J.; Singh, R.G.; Gupta, R.; Kumar, V.; Singh, F.; Kumar, R. Effect of Annealing on the Surface Morphology, Optical and and Structural Properties of Nanodimensional Tungsten Oxide Prepared by Coprecipitation Technique. J. Electron. Mater. 2019, 48, 1174–1183. [Google Scholar] [CrossRef]
- Lee, D.-S.; Han, S.-D.; Huh, J.-S.; Lee, D.-D. Nitrogen oxides-sensing characteristics of WO3-based nanocrystalline thick film gas sensor. Sens. Actuators B Chem. 1999, 60, 57–63. [Google Scholar] [CrossRef]
- Besozzi, E.; Dellasega, D.; Russo, V.; Conti, C.; Passoni, M.; Beghi, M.G. Thermomechanical properties of amorphous metallic tungsten-oxygen and tungsten-oxide coatings. Mater. Des. 2019, 165, 107565. [Google Scholar] [CrossRef]
- Trasferetti, B.C.; Rouxinol, F.P.; Gelamo, R.V.; Bica de Moraes, M.A.; Davanzo, C.U.; De Faria, D.L. Berreman Effect in Amorphous and Crystalline WO3 Thin Films. J. Phys. Chem. B 2004, 108, 12333–12338. [Google Scholar] [CrossRef]
- Daniel, M.F.; Desbat, B.; Lassegues, J.C.; Gerand, B.; Figlarz, M. Infrared and Raman study of WO3 tungsten trioxides and WO3, xH2O tungsten trioxide tydrates. J. Solid State Chem. 1987, 67, 235–247. [Google Scholar] [CrossRef]
- Manciu, F.S.; Enriquez, J.L.; Durrer, W.G.; Yun, Y.; Ramana, C.V.; Gullapalli, S.K. Spectroscopic analysis of tungsten oxide thin films. J. Mater. Res. 2010, 25, 2401–2406. [Google Scholar] [CrossRef]
- Di Fonzo, F.; Bailini, A.; Russo, V.; Baserga, A.; Cattaneo, D.; Beghi, M.G.; Ossi, P.M.; Casari, C.S.; Li Bassi, A.; Bottani, C.E. Synthesis and characterization of tungsten and tungsten oxide nanostructured films. Catal. Today 2006, 116, 69–73. [Google Scholar] [CrossRef]
- Thummavichai, K.; Wang, N.; Xu, F.; Rance, G.; Xia, Y.; Zhu, Y. In situ investigations of the phase change behaviour of tungsten oxide nanostructures. R. Soc. Open Sci. 2018, 5, 171932. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, Y.T.; Hsueh, S.H.; Chen, U.S.; Huang, M.W.; Shih, H.C. Rapid synthesis of bundled tungsten oxide nanowires by microwave plasma-enhanced chemical vapor deposition and their optical properties. J. Electrochem. Soc. 2010, 157, k183–k186. [Google Scholar] [CrossRef]
- Souza-Filho, A.G.; Freire, V.N.; Sasaki, J.M.; Mendes Filho, J.; Julião, J.F.; Gomes, U.U. Coexistence of triclinic and monoclinic phases in WO3 ceramics. J. Raman Spectrosc. 2000, 31, 451–454. [Google Scholar] [CrossRef]
- van den Aker, K.; Kong, S.; Schmitz, J.; Wolters, R. The Dependence of the Stress of Chemical Vapor Deposited Tungsten Films on Deposition Parameters. J. Electrochem. Soc. 1994, 141, 843–848. [Google Scholar]
- Bekri, M.; Shaalan, N.M.; Ahmed, A.S. Thermal evaporated WO3 nanoparticles film under different evaporation pressures for NO2 sensing. Dig. J. Nanomater. Biostruct. 2015, 10, 603–613. [Google Scholar]
- Kubelka, P.; Munk, F. Ein Beitrag zur Optik der Farbanstriche. Zeitschrift für Tech. Phys. 1931, 12, 593–601. [Google Scholar]
- Kwong, W.L.; Savvides, N.; Sorrell, C.C. Electrodeposited nanostructured WO 3 thin films for photoelectrochemical applications. Electrochim. Acta 2012, 75, 371–380. [Google Scholar] [CrossRef]
- Vasilopoulou, M.; Kostis, I.; Vourdas, N.; Papadimitropoulos, G.; Douvas, A.; Boukos, N.; Kennou, S.; Davazoglou, D. Influence of the oxygen substoichiometry and of the hydrogen incorporation on the electronic band structure of amorphous tungsten oxide films. J. Phys. Chem. C 2014, 118, 12632–12641. [Google Scholar] [CrossRef]
- Vemuri, R.S.; Engelhard, M.H.; Ramana, C.V. Correlation between surface chemistry, density, and band gap in nanocrystalline WO 3 thin films. ACS Appl. Mater. Interfaces 2012, 4, 1371–1377. [Google Scholar] [CrossRef] [PubMed]
- Joraid, A.A.; Alamri, S.N. Effect of annealing on structural and optical properties of WO3 thin films prepared by electron-beam coating. Phys. B Condens. Matter 2007, 391, 199–205. [Google Scholar] [CrossRef]



| Sample | Deposition Time (Min) | Annealing Temperature (10 Min) |
|---|---|---|
| s5m | 5 |
|
| s10m | 10 |
|
| s15m | 15 |
|
| Sample | Temperature of Annealing | Crystallite Size | Specific Surface Area m2/gr |
|---|---|---|---|
| s5m | 300 °C | 12.88 nm | 69.97 |
| 500 °C | 18.34 nm | 45.78 | |
| s10m | 300 °C | 13.0 nm | 64.59 |
| 500 °C | 28.70 nm | 29.25 | |
| s15m | 300 °C | 23.3 nm | 36.04 |
| 500 °C | 25.6 nm | 32.80 |
| Synthesis Method | Temperature of Annealing | Crystallite Size | Specific Surface Area m2/gr | Reference |
|---|---|---|---|---|
| Ball-milled | 600 °C | 59.2 nm | 3.16 | [50] |
| Solvothermal | 500 °C | 47–61 nm | 11.57–18.92 | [6] |
| Coprecipitation | 500 °C | 66 nm | -- | [51] |
| Sparking | 500 °C | 62 nm | -- | [8] |
| Sputtering | 600 °C | -- | 19.1 | [9] |
| Sol-coprecipitation | 600 °C | 11 nm | 4.7 | [52] |
| Reactive Spray Deposition | 500 °C | 20–30 nm | 46 | [7] |
| HFCVD | 500 °C | 28.7 nm | 29.25 | This work |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cruz-Leal, M.; Goiz, O.; Chávez, F.; Pérez-Sánchez, G.F.; Hernández-Como, N.; Santes, V.; Felipe, C. Study of the Thermal Annealing on Structural and Morphological Properties of High-Porosity A-WO3 Films Synthesized by HFCVD. Nanomaterials 2019, 9, 1298. https://doi.org/10.3390/nano9091298
Cruz-Leal M, Goiz O, Chávez F, Pérez-Sánchez GF, Hernández-Como N, Santes V, Felipe C. Study of the Thermal Annealing on Structural and Morphological Properties of High-Porosity A-WO3 Films Synthesized by HFCVD. Nanomaterials. 2019; 9(9):1298. https://doi.org/10.3390/nano9091298
Chicago/Turabian StyleCruz-Leal, M., O. Goiz, F. Chávez, G. F. Pérez-Sánchez, N. Hernández-Como, V. Santes, and C. Felipe. 2019. "Study of the Thermal Annealing on Structural and Morphological Properties of High-Porosity A-WO3 Films Synthesized by HFCVD" Nanomaterials 9, no. 9: 1298. https://doi.org/10.3390/nano9091298





