Influence of Individual Ions on Silica Nanoparticles Interaction with Berea Sandstone Minerals
Abstract
:1. Introduction
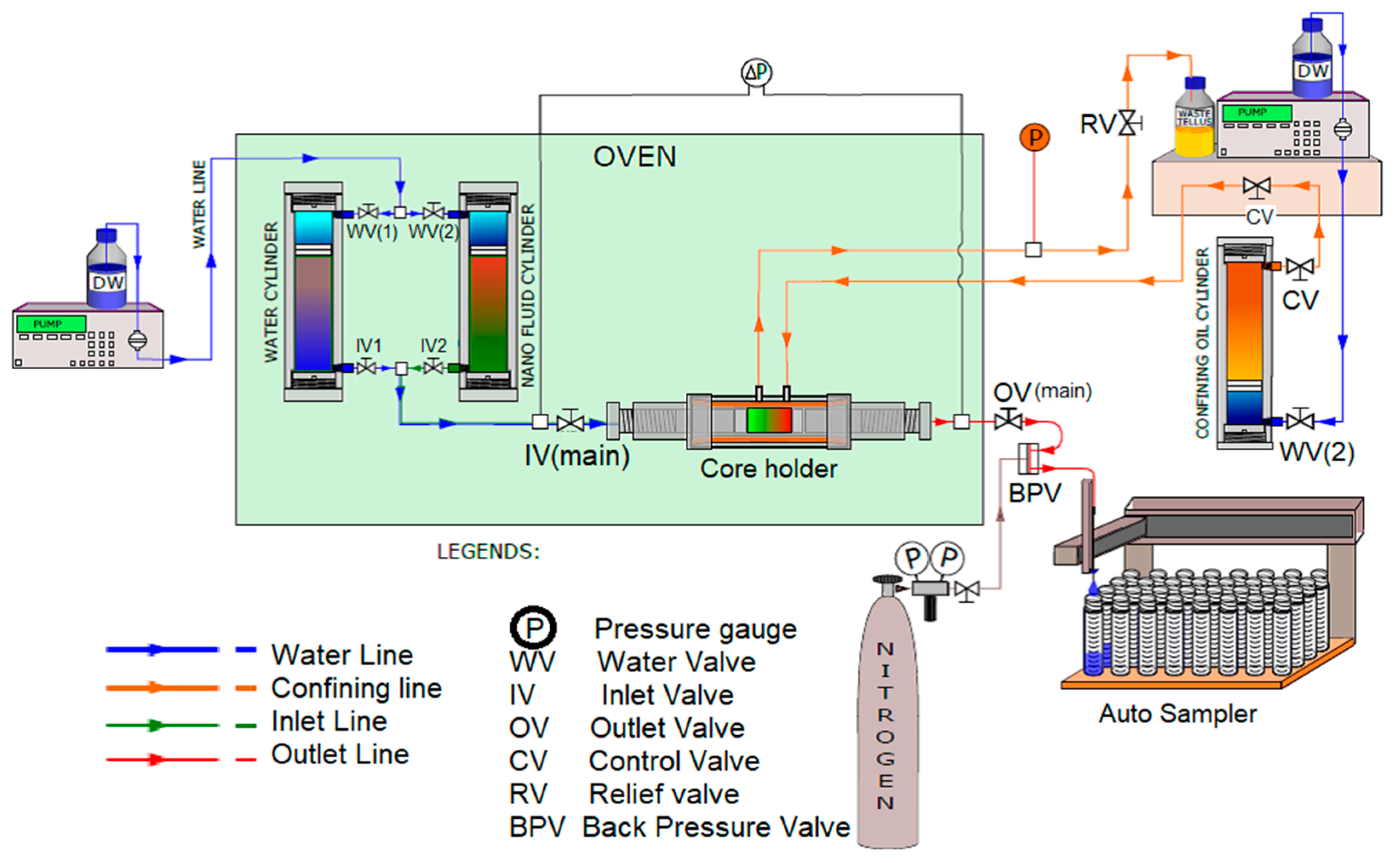
2. Materials and Methods
2.1. Nanoparticle Slug Injection
2.2. Electric Double Layer Interaction
3. Results and Discussions
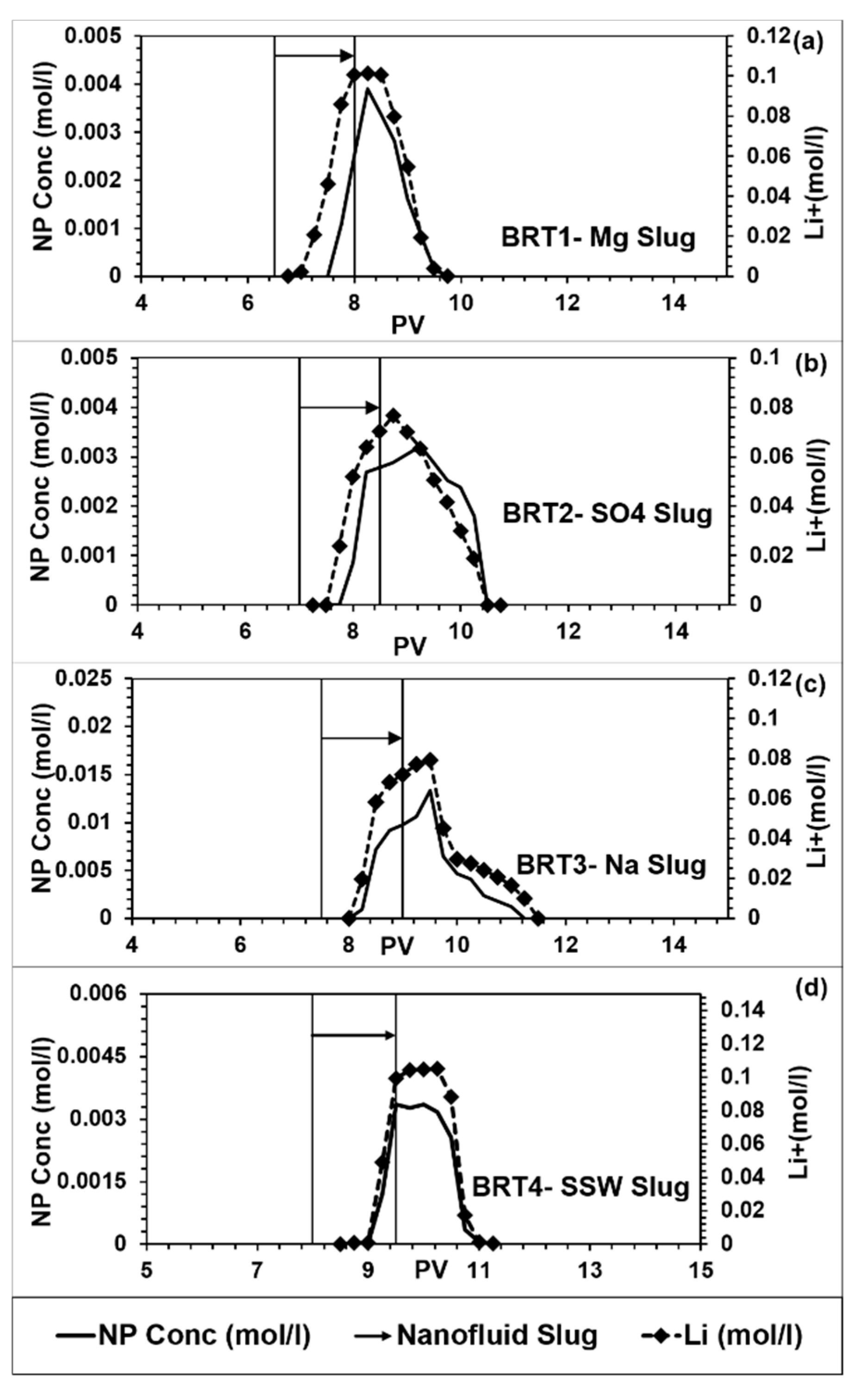
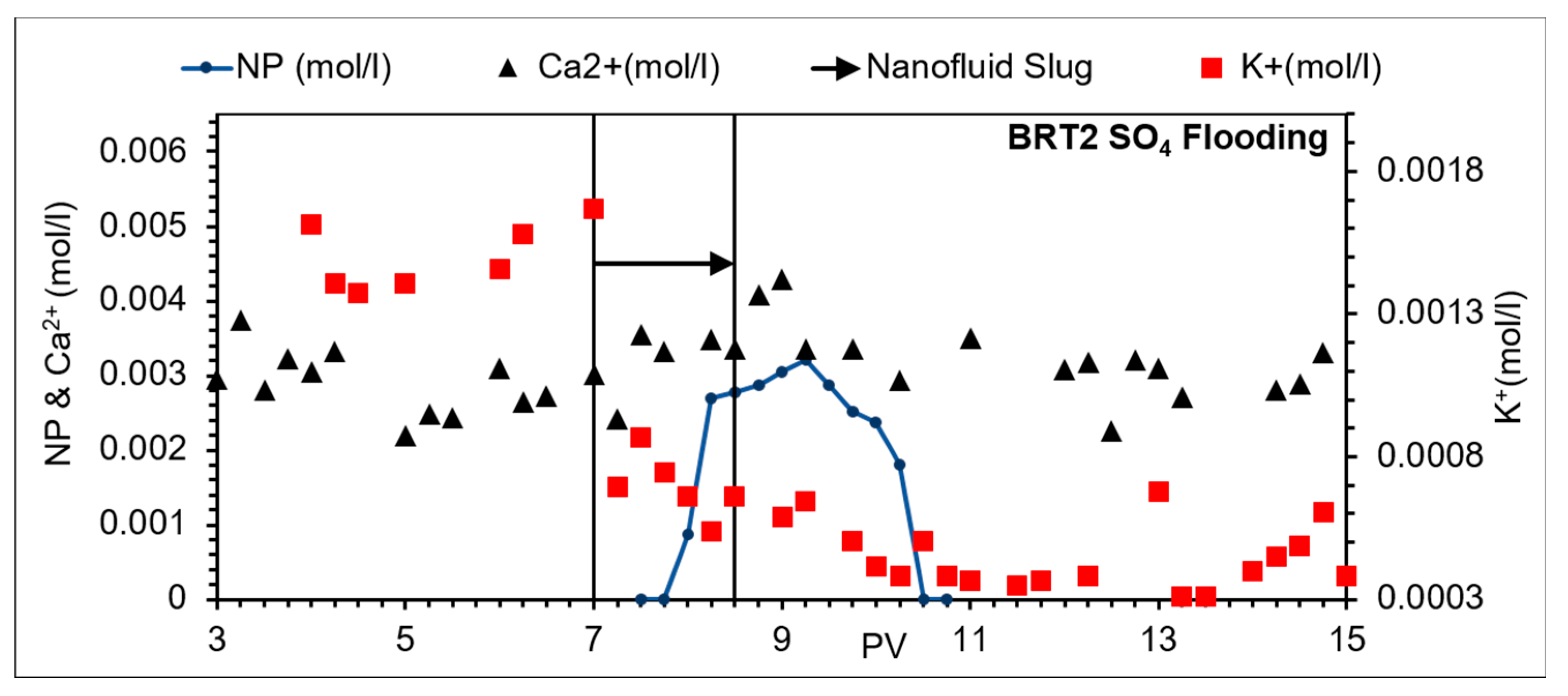
3.1. Nanoparticle and Tracer Profiles
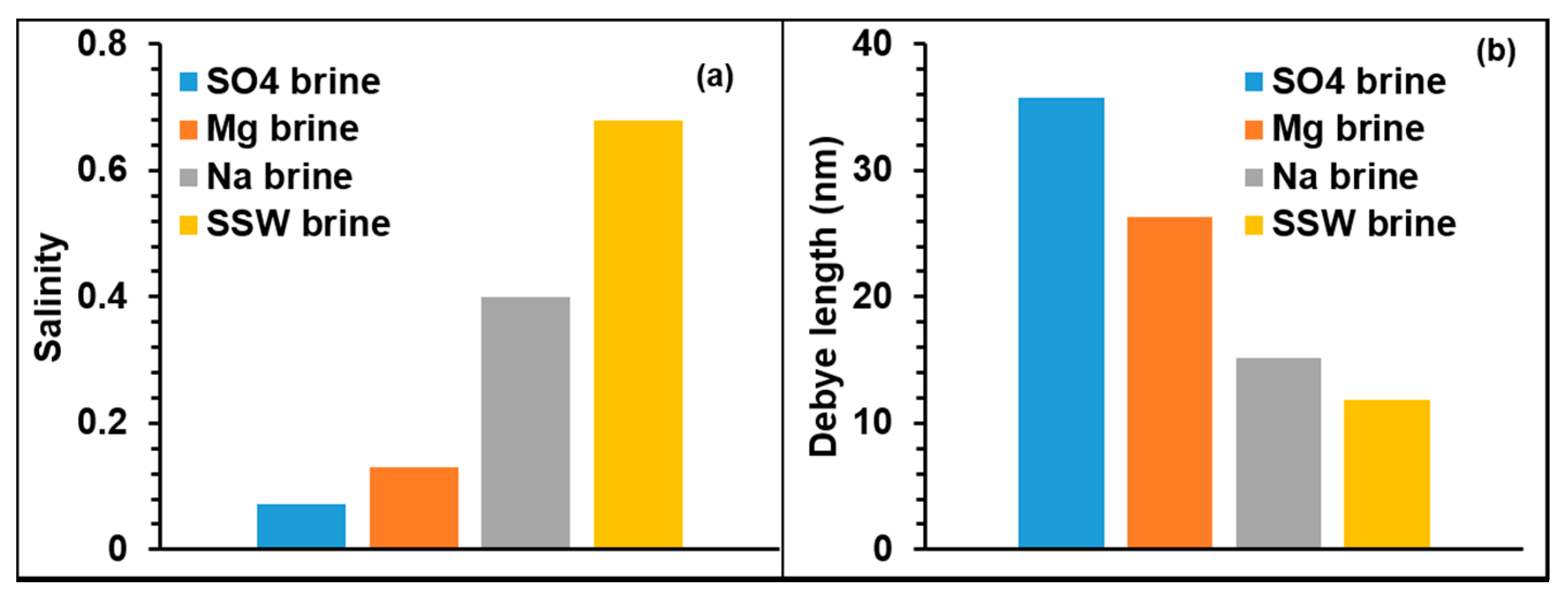
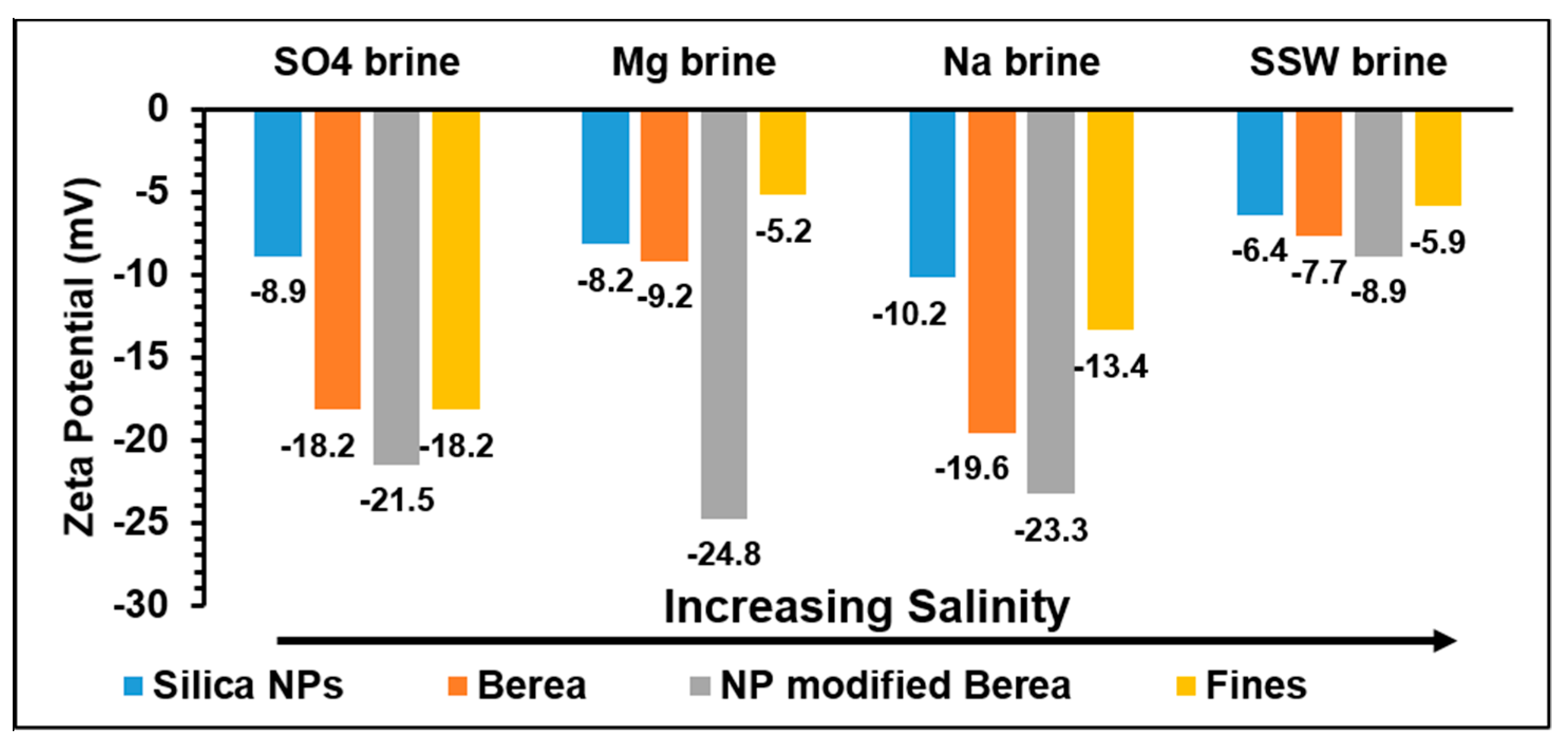
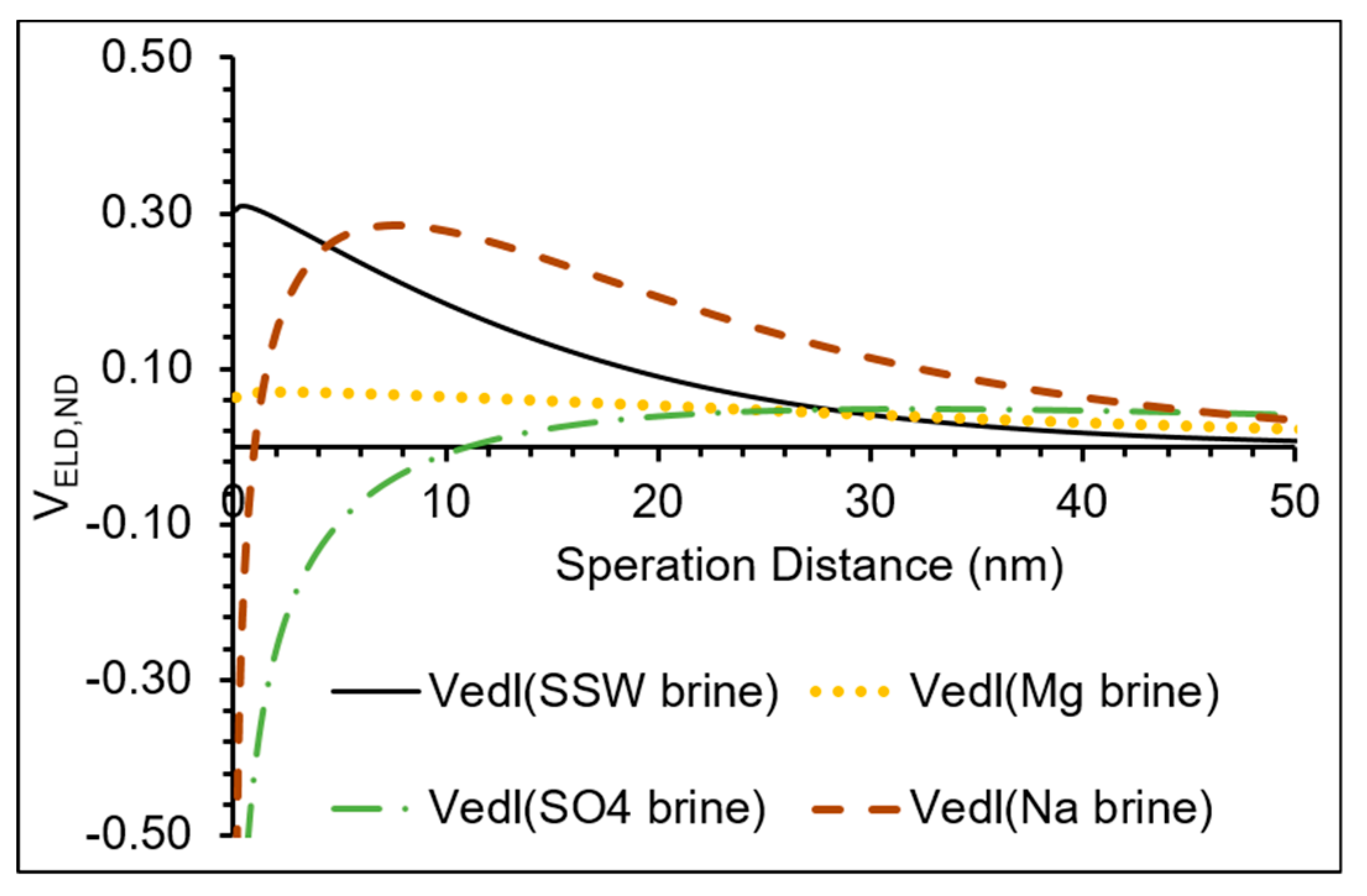
3.2. Double Layer Interaction
3.3. NPs/Mineral Interaction and Possible Mechanism(s)
3.3.1. MgCl2 Brine
3.3.2. Na2SO4 Brine
3.3.3. NaCl Brine
4. Summary and Conclusions
- All the used single salt brines showed reduced mineral dissolution, however to different degrees. This is related to the degree of adsorption on the minerals, where the highest were about 82% in the cases of Mg and SO4 single brines, compared to about 34% in the case of Na single brine. It is demonstrated that specific ions rather than brine ionic strength/salinity are the major influencing factor on adsorption of silica NPs on Berea mineral surface.
- The possible mechanism for the influence of Mg2+ on the adsorption is due to renewing and increasing of the contacted surface area at the mineral surfaces by a possible exchange reaction between Ca2+ and Mg2+. The mechanism in the case of SO42− ions, is due to their adsorption on kaolinite and release of OH−, which is followed by neutralization of the resulted negative charge by participation of neighboring Al ions resulting in ring formation, as suggested in the literature. This may also be supported by the increasing trends of the pH and the zeta potentials of the cations/minerals which are more negative than that in the case of SO4 brine. Another possible supporting point is that in the case of SO4 brine, the double layer interaction is less repulsive than those of the other brines.
- Clay minerals have exchangeable cations so for the addition of sodium brine, an exchange between Na+ with H+ releases hydrogen ions. This process changes the surface charge distribution; hence affecting the adsorption of materials such as NPs. This is supported by that in the case of Na brine, the lowest adsorption of NPs (~34%) occurred. The two other observations that may support the exchange mechanism are the domination of the repulsive double layer (Figure 5) and that Na brine has the highest negative zeta potential, for NPs/Berea in presence of NaCl as stabilizing fluid.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Amirian, E.; Dejam, M.; Chen, Z. Performance forecasting for polymer flooding in heavy oil reservoirs. Fuel 2018, 216, 83–100. [Google Scholar] [CrossRef]
- Saboorian-Jooybari, H.; Dejam, M.; Chen, Z. Heavy oil polymer flooding from laboratory core floods to pilot tests and field applications: Half-century studies. J. Pet. Sci. Eng. 2016, 142, 85–100. [Google Scholar] [CrossRef]
- Mashayekhizadeh, V.; Kord, S.; Dejam, M. Eor potential within Iran. Spec. Top. Rev. Porous Media Int. J. 2014, 5, 325–354. [Google Scholar] [CrossRef]
- Ayatollahi, S.; Zerafat, M.M. Nanotechnology-assisted eor techniques: New solutions to old challenges. In Proceedings of the SPE International Oilfield Nanotechnology Conference and Exhibition, Noordwijk, The Netherlands, 12–14 June 2012. [Google Scholar]
- Al-Anssari, S.; Wang, S.; Barifcani, A.; Lebedev, M.; Iglauer, S. Effect of temperature and sio 2 nanoparticle size on wettability alteration of oil-wet calcite. Fuel 2017, 206, 34–42. [Google Scholar] [CrossRef]
- Li, S.; Torsæter, O. Experimental investigation of the influence of nanoparticles adsorption and transport on wettability alteration for oil wet berea sandstone. In Proceedings of the SPE Middle East Oil & Gas Show and Conference, Manama, Bahrain, 8–11 March 2015. [Google Scholar]
- Abhishek, R.; Kumar, G.S.; Sapru, R. Wettability alteration in carbonate reservoirs using nanofluids. Pet. Sci. Technol. 2015, 33, 794–801. [Google Scholar] [CrossRef]
- Alramadan, H.A. Experimental Evaluation of Surface Treated Nanoparticles and Their Effect on Wettability Alteration of Carbonate Surfaces and Oil-Brine Interfacial Tension; The University of Texas at Austin: Austin, TX, USA, 2016. [Google Scholar]
- Olayiwola, S.O.; Dejam, M. Mathematical modelling of surface tension of nanoparticles in electrolyte solutions. Chem. Eng. Sci. 2019, 197, 345–356. [Google Scholar] [CrossRef]
- Wasan, D.T.; Nikolov, A.D. Spreading of nanofluids on solids. Nature 2003, 423, 156–159. [Google Scholar] [CrossRef]
- Zhang, H.; Nikolov, A.; Wasan, D. Enhanced oil recovery (eor) using nanoparticle dispersions: Underlying mechanism and imbibition experiments. Energy Fuels 2014, 28, 3002–3009. [Google Scholar] [CrossRef]
- Abhishek, R.; Hamouda, A.A.; Abdulhameed, F.M. Adsorption kinetics and enhanced oil recovery by silica nanoparticles in sandstone. Pet. Sci. Technol. 2019, 37, 1363–1369. [Google Scholar] [CrossRef]
- Yuan, B.; Ghanbarnezhad Moghanloo, R.; Pattamasingh, P. Applying method of characteristics to study utilization of nanoparticles to reduce fines migration in deepwater reservoirs. In Proceedings of the SPE European Formation Damage Conference and Exhibition, Budapest, Hungary, 3–5 June 2015. [Google Scholar]
- Arab, D.; Pourafshary, P. Nanoparticles-assisted surface charge modification of the porous medium to treat colloidal particles migration induced by low salinity water flooding. Colloids Surf. A Physicochem. Eng. Asp. 2013, 436, 803–814. [Google Scholar] [CrossRef]
- Yuan, B.; Moghanloo, R.G.; Wang, W. Using nanofluids to control fines migration for oil recovery: Nanofluids co-injection or nanofluids pre-flush?—A comprehensive answer. Fuel 2018, 215, 474–483. [Google Scholar] [CrossRef]
- Abhishek, R.; Hamouda, A.A. Effect of various silica nanofluids: Reduction of fines migrations and surface modification of berea sandstone. Appl. Sci. 2017, 7, 1216. [Google Scholar]
- Abhishek, R.; Hamouda, A.A.; Ayoub, A. Effect of silica nanoparticles on fluid/rock interactions during low salinity water flooding of chalk reservoirs. Appl. Sci. 2018, 8, 1093. [Google Scholar] [CrossRef]
- Abhishek, R.; Hamouda, A.A.; Murzin, I. Adsorption of silica nanoparticles and its synergistic effect on fluid/rock interactions during low salinity flooding in sandstones. Colloids Surf. A Physicochem. Eng. Asp. 2018, 555, 397–406. [Google Scholar] [CrossRef]
- Olayiwola, S.O.; Dejam, M. A comprehensive review on interaction of nanoparticles with low salinity water and surfactant for enhanced oil recovery in sandstone and carbonate reservoirs. Fuel 2019, 241, 1045–1057. [Google Scholar]
- Zahid, A.; Shapiro, A.A.; Skauge, A. Experimental studies of low salinity water flooding carbonate: A new promising approach. In Proceedings of the SPE EOR Conference at Oil and Gas West Asia, Muscat, Oman, 16–18 April 2012. [Google Scholar]
- Morrow, N.; Buckley, J. Improved oil recovery by low-salinity waterflooding. J. Pet. Technol. 2011, 63, 106–112. [Google Scholar] [CrossRef]
- Mahani, H.; Keya, A.L.; Berg, S.; Bartels, W.B.; Nasralla, R.; Rossen, W.R. Insights into the mechanism of wettability alteration by low-salinity flooding (lsf) in carbonates. Energy Fuels 2015, 29, 1352–1367. [Google Scholar] [CrossRef]
- Jackson, M.; Vinogradov, J.; Hamon, G.; Chamerois, M. Evidence, mechanisms and improved understanding of controlled salinity waterflooding part 1: Sandstones. Fuel 2016, 185, 772–793. [Google Scholar] [CrossRef] [Green Version]
- Rostami, P.; Mehraban, M.F.; Sharifi, M.; Dejam, M.; Ayatollahi, S. Effect of water salinity on oil/brine interfacial behaviour during low salinity waterflooding: A mechanistic study. Petroleum 2019, in press. [Google Scholar] [CrossRef]
- Khilar, K.C.; Fogler, H.S. The existence of a critical salt concentration for particle release. J. Colloid Interface Sci. 1984, 101, 214–224. [Google Scholar] [CrossRef] [Green Version]
- Kia, S.; Fogler, H.; Reed, M. Effect of ph on colloidally induced fines migration. J. Colloid Interface Sci. 1987, 118, 158–168. [Google Scholar] [CrossRef]
- Rosenbrand, E.; Kjøller, C.; Riis, J.F.; Kets, F.; Fabricius, I.L. Different effects of temperature and salinity on permeability reduction by fines migration in berea sandstone. Geothermics 2015, 53, 225–235. [Google Scholar] [CrossRef]
- Bhattacharya, S.; Paitaridis, J.; Pedler, A.; Badalyan, A.; Yang, Y.; Carageorgos, T.; Bedrikovetsky, P.; Warren, D.; Lemon, N. Fines mobilisation by low-salinity water injection: 3-point-pressure tests. In Proceedings of the SPE International Conference and Exhibition on Formation Damage Control, Lafayette, LA, USA, 24–26 February 2016. [Google Scholar]
- Mainwaring, J. Losal: Bp’s Low-Salinity Enhanced Oil Recovery Technology. Available online: www.rigzone.com (accessed on 24 September 2015).
- Li, S.; Hadia, N.J.; Lau, H.C.; Torsæter, O.; Stubbs, L.P.; Ng, Q.H. Silica nanoparticles suspension for enhanced oil recovery: Stability behavior and flow visualization. In Proceedings of the SPE Europec featured at 80th EAGE Conference and Exhibition, Copenhagen, Denmark, 11–14 June 2018. [Google Scholar]
- Hendraningrat, L.; Li, S.; Torsæter, O. A coreflood investigation of nanofluid enhanced oil recovery. J. Pet. Sci. Eng. 2013, 111, 128–138. [Google Scholar] [CrossRef]
- Torsater, O.; Li, S.; Hendraningrat, L. A coreflood investigation of nanofluid enhanced oil recovery in low-medium permeability berea sandstone. In Proceedings of the SPE International Symposium on Oilfield Chemistry, The Woodlands, TX, USA, 8–10 April 2013. [Google Scholar]
- Hendraningrat, L.; Torsæter, O. Effects of the initial rock wettability on silica-based nanofluid-enhanced oil recovery processes at reservoir temperatures. Energy Fuels 2014, 28, 6228–6241. [Google Scholar] [CrossRef]
- Monfared, A.D.; Ghazanfari, M.; Jamialahmadi, M.; Helalizadeh, A. Adsorption of silica nanoparticles onto calcite: Equilibrium, kinetic, thermodynamic and dlvo analysis. Chem. Eng. J. 2015, 281, 334–344. [Google Scholar] [CrossRef]
- Abhishek, R. Interaction of Silica Nanoparticles with Chalk and Sandstone Minerals: Adsorption, Fluid/Rock Interactions in the Absence and Presence of Hydrocarbons. Ph.D. Thesis, University of Stavanger, Stavanger, Norway, 2019. [Google Scholar]
- Dunphy Guzman, K.A.; Finnegan, M.P.; Banfield, J.F. Influence of surface potential on aggregation and transport of titania nanoparticles. Environ. Sci. Technol. 2006, 40, 7688–7693. [Google Scholar] [CrossRef]
- Zhang, T.; Murphy, M.J.; Yu, H.; Bagaria, H.G.; Yoon, K.Y.; Nielson, B.M.; Bielawski, C.W.; Johnston, K.P.; Huh, C.; Bryant, S.L. Investigation of nanoparticle adsorption during transport in porous media. SPE J. 2015, 20, 667–677. [Google Scholar] [CrossRef]
- Hamouda, A.A.; Valderhaug, O.M. Investigating enhanced oil recovery from sandstone by low-salinity water and fluid/rock interaction. Energy Fuels 2014, 28, 898–908. [Google Scholar] [CrossRef]
- Hamouda, A.A.; Maevskiy, E. Oil recovery mechanism (s) by low salinity brines and their interaction with chalk. Energy Fuels 2014, 28, 6860–6868. [Google Scholar] [CrossRef]
- Kingston, F.; Posner, A.; Quirk, J. Anion adsorption by goethite and gibbsite: I. The role of the proton in determining adsorption envelopes. J. Soil Sci. 1972, 23, 177–192. [Google Scholar] [CrossRef]
- Rao, S.M.; Sridharan, A. Mechanism of sulfate adsorption by kaolinite. Clays Clay Miner. 1984, 32, 414–418. [Google Scholar] [CrossRef]
- Rajan, S. Sulfate adsorbed on hydrous alumina, ligands displaced, and changes in surface charge 1. Soil Sci. Soc. Am. J. 1978, 42, 39–44. [Google Scholar] [CrossRef]
- Reid, V.; Longman, G.F.; Heinerth, E. Tenside. Anionic detergents by two-phase titration (II). Tenside 1968, 5, 90–96. [Google Scholar]
- Hanna, H.; Somasundaran, P. Equilibration of kaolinite in aqueous inorganic and surfactant solutions. J. Colloid Interface Sci. 1979, 70, 181–191. [Google Scholar] [CrossRef]



| Ion | SSW (mol/L) | Na Brine (mol/L) | Mg Brine (mol/L) | SO4 Brine (mol/L) |
|---|---|---|---|---|
| HCO3− | 0.002 | 0 | 0 | 0 |
| Cl− | 0.525 | 0.400068 | 0.089033 | 0 |
| SO42− | 0.0240 | 0 | 0 | 0.096 |
| Mg2+ | 0.045 | 0 | 0.178066 | 0 |
| Ca2+ | 0.013 | 0 | 0 | 0 |
| Na+ | 0.450 | 0.400068 | 0 | 0.048 |
| K+ | 0.010 | 0 | 0 | 0 |
| Ionic strength | 0.68 | 0.4 | 0.13 | 0.072 |
| Name | Initial Saturating Fluid | Pre-flush Fluid | NP Slug | Post-Flush Fluid |
|---|---|---|---|---|
| BRT 1 | SSW | Mg Brine | 1 (g/L) NP in Mg Brine with 0.1 M LiCl tracer | Mg Brine |
| OBRT 2 | SSW | SO4 Brine | 1 (g/L) NP in SO4 Brine with 0.1 M LiCl tracer | SO4 Brine |
| BRT 3 | SSW | Na Brine | 1 (g/L) NP in Na Brine with 0.1 M LiCl tracer | Na Brine |
| BRT 4 | SSW | SSW | 1 (g/L) NP in SSW with 0.1 M LiCl tracer | SSW |
| Experiment | % Nanoparticles Retained in the Core |
|---|---|
| BRT1 SSW Slug | 81.7 |
| BRT2 MG Slug | 81.3 |
| BRT3 SO4 Slug | 68.2 |
| BRT4 Na Slug | 33.9 |
| Dispersing Fluid | Average Hydrodynamic Radius of the NPs (nm) |
|---|---|
| SSW | 28.2 |
| Na Brine | 19.6 |
| Mg Brine | 19.4 |
| SO4 Brine | 19.5 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hamouda, A.A.; Abhishek, R. Influence of Individual Ions on Silica Nanoparticles Interaction with Berea Sandstone Minerals. Nanomaterials 2019, 9, 1267. https://doi.org/10.3390/nano9091267
Hamouda AA, Abhishek R. Influence of Individual Ions on Silica Nanoparticles Interaction with Berea Sandstone Minerals. Nanomaterials. 2019; 9(9):1267. https://doi.org/10.3390/nano9091267
Chicago/Turabian StyleHamouda, Aly A., and Rockey Abhishek. 2019. "Influence of Individual Ions on Silica Nanoparticles Interaction with Berea Sandstone Minerals" Nanomaterials 9, no. 9: 1267. https://doi.org/10.3390/nano9091267





