Influence of Solvent Evaporation Technique Parameters on Diameter of Submicron Lamivudine-Poly-ε-Caprolactone Conjugate Particles
Abstract
:1. Introduction
1.1. Size-Dependent Carrier Internalization
1.2. Particles Uptake by Macrophages
1.3. Solvent Evaporation Technique
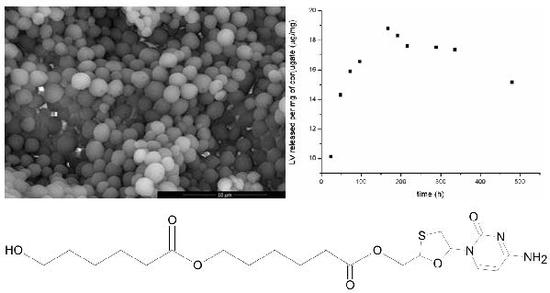
1.4. Evaluation of Lamivudine Conjugate-Based Particles
2. Materials and Methods
2.1. Materials
2.2. Conjugate Synthesis
2.3. Conjugate Degradation
2.4. Particle Preparation
2.5. Power Consumption Measurement
2.6. Spectroscopic Analysis
2.7. Gel permation Chromatography
2.8. High Performance Liquid Chromatography
2.9. Particle Size and Zeta Potential Evaluation
3. Results
3.1. Conjugate Synthesis
3.2. Drug Content
3.3. Particle Preparation
4. Discussion
4.1. Conjugate Synthesis
4.2. Drug Content
4.3. Particle Formation and Morphology
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Shang, L.; Nienhaus, K.; Nienhaus, G.U. Engineered nanoparticles interacting with cells: Size matters. J. Nanobiotechnol. 2014, 12, 5. [Google Scholar] [CrossRef] [PubMed]
- Rosales, C.; Uribe-Querol, E. Phagocytosis: A Fundamental Process in Immunity. BioMed Res. Int. 2017, 2017, 1–18. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, J.; Byrne, J.D.; Napier, M.E.; DeSimone, J.M. More Effective Nanomedicines through Particle Design. Small 2011, 7, 1919–1931. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhao, J.; Stenzel, M.H. Entry of nanoparticles into cells: The importance of nanoparticle properties. Polym. Chem. 2018, 9, 259–272. [Google Scholar] [CrossRef]
- Fröhlich, E. The role of surface charge in cellular uptake and cytotoxicity of medical nanoparticles. Int. J. Nanomed. 2012, 7, 5577–5591. [Google Scholar] [CrossRef] [PubMed]
- Xin, Y.; Yin, M.; Zhao, L.; Meng, F.; Luo, L. Recent progress on nanoparticle-based drug delivery systems for cancer therapy. Cancer Boil. Med. 2017, 14, 228–241. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hoshyar, N.; Gray, S.; Han, H.; Bao, G. The effect of nanoparticle size on in vivo pharmacokinetics and cellular interaction. Nanomedicine 2016, 11, 673–692. [Google Scholar] [CrossRef]
- Langille, S.E. Particulate matter in injectable drug products. PDA J. Pharm. Sci. Technol. 2013, 67, 186–200. [Google Scholar] [CrossRef] [PubMed]
- Decuzzi, P.; Godin, B.; Tanaka, T.; Lee, S.Y.; Chiappini, C.; Liu, X.; Ferrari, M. Size and shape effects in the biodistribution of intravascularly injected particles. J. Control. Release 2010, 141, 320–327. [Google Scholar] [CrossRef]
- Kaga, S.; Truong, N.P.; Esser, L.; Senyschyn, D.; Sanyal, A.; Sanyal, R.; Quinn, J.F.; Davis, T.P.; Kaminskas, L.M.; Whittaker, M.R. Influence of Size and Shape on the Biodistribution of Nanoparticles Prepared by Polymerization-Induced Self-Assembly. Biomacromolecules 2017, 18, 3963–3970. [Google Scholar] [CrossRef]
- Braga, T.T.; Agudelo, J.S.H.; Câmara, N.O.S. Macrophages during the Fibrotic Process: M2 as Friend and Foe. Front. Immunol. 2015, 6, 602. [Google Scholar] [CrossRef] [PubMed]
- Peterson, K.R.; Cottam, M.A.; Kennedy, A.J.; Hasty, A.H. Macrophage-Targeted Therapeutics for Metabolic Disease. Trends Pharmacol. Sci. 2018, 39, 536–546. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.H.; Loo, C.Y.; Traini, D.; Young, P.M. Nano-and micro-based inhaled drug delivery systems for targeting alveolar macrophages. Expert Opin. Drug Deliv. 2015, 12, 1009–1026. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Tatematsu, K.; Somiya, M.; Iijima, M.; Kuroda, S.I. Development of a macrophage-targeting and phagocytosis-inducing bio-nanocapsule-based nanocarrier for drug delivery. Acta Biomater. 2018, 73, 412–423. [Google Scholar] [CrossRef] [PubMed]
- Ostrowski, P.P.; Grinstein, S.; Freeman, S.A. Review Diffusion Barriers, Mechanical Forces, and the Biophysics of Phagocytosis. Dev. Cell 2016, 38, 135–146. [Google Scholar] [CrossRef] [PubMed]
- Singh, C.; Purohit, S.; Pandey, B.L.; Singh, M. Solvent Evaporation Technique of Microencapsulation: A Systemic Review. Int. J. Adv. Pharm. Anal. 2014, 4, 96–104. [Google Scholar]
- Vysloužil, J.; Doležel, P.; Kejdušová, M.; Mašková, E.; Mašek, J.; Lukáč, R.; Košťál, V.; Vetchý, D.; Dvořáčková, K. Influence of different formulations and process parameters during the preparation of drug-loaded PLGA microspheres evaluated by multivariate data analysis. Acta Pharm. 2014, 64, 403–417. [Google Scholar] [CrossRef] [Green Version]
- Tiwari, S.; Verma, P. Microencapsulation technique by solvent evaporation method (Study of effect of process variables). Int. J. Pharm. Life Sci. 2011, 2, 998–1005. [Google Scholar]
- Iqbal, M.; Zafar, N.; Fessi, H.; Elaissari, A. Double emulsion solvent evaporation techniques used for drug encapsulation. Int. J. Pharm. 2015, 496, 173–190. [Google Scholar] [CrossRef] [PubMed]
- Bao, W.; Zhou, J.; Luo, J.; Wu, D. PLGA microspheres with high drug loading and high encapsulation efficiency prepared by a novel solvent evaporation technique. J. Microencapsul. 2006, 23, 471–479. [Google Scholar] [CrossRef]
- Božič, M.; Elschner, T.; Tkaučič, D.; Bračič, M.; Hribernik, S.; Stana Kleinschek, K.; Kargl, R. Effect of different surface active polysaccharide derivatives on the formation of ethyl cellulose particles by the emulsion-solvent evaporation method. Cellulose 2018, 25, 6901–6922. [Google Scholar] [CrossRef] [Green Version]
- De Hert, S.C.; Rodgers, T.; Rodgers, T. Continuous, recycle and batch emulsification kinetics using a high-shear mixer. Chem. Eng. Sci. 2017, 167, 265–277. [Google Scholar] [CrossRef]
- Nowacek, A.S.; Miller, R.L.; McMillan, J.; Kanmogne, G.; Kanmogne, M.; Mosley, R.L.; Ma, Z.; Graham, S.; Chaubal, M.; Werling, J.; et al. NanoART synthesis, characterization, uptake, release and toxicology for human monocyte—Macrophage drug delivery. Nanomedicine (Lond.) 2009, 4, 903–917. [Google Scholar] [CrossRef] [PubMed]
- Akhtar, R.; Yousaf, M.; Zahoor, A.F.; Naqvi, S.A.R.; Abbas, N. Synthesis of lamivudine (3TC) and its derivatives. Phosphorus Sulfur Silicon Relat. Elem. 2017, 192, 989–1001. [Google Scholar] [CrossRef]
- Chimalakonda, K.C.; Agarwal, H.K.; Kumar, A.; Parang, K.; Mehvar, R. Synthesis, Analysis, in Vitro Characterization, and in Vivo Disposition of a Lamivudine–Dextran Conjugate for Selective Antiviral Delivery to the Liver. Bioconj. Chem. 2007, 18, 2097–2108. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Du, Y.Z.; Yuan, H.; Zhang, X.G.; Miao, J.; Cui, F.D.; Hu, F.Q. Synthesis of Lamivudine stearate and antiviral activity of stearic acid-g-chitosan oligosaccharide polymeric micelles delivery system. Eur. J. Pharm. Sci. 2010, 41, 498–507. [Google Scholar] [CrossRef] [PubMed]
- Vasilyeva, S.V.; Shtil, A.A.; Petrova, A.S.; Balakhnin, S.M.; Achigecheva, P.Y.; Stetsenko, D.A.; Silnikov, V.N. Conjugates of phosphorylated zalcitabine and lamivudine with SiO2 nanoparticles: Synthesis by CuAAC click chemistry and preliminary assessment of anti-HIV and antiproliferative activity. Bioor. Med. Chem. 2017, 25, 1696–1702. [Google Scholar] [CrossRef]
- Rajca, A.; Li, Q.; Date, A.; Belshan, M.A.; Destache, C.J. Thermosensitive vaginal gel containing PLGA-NRTI conjugated nanoparticles for HIV prophylaxis. TechConnect Briefs 2013, 3, 293–296. [Google Scholar]
- Prakash, K.; Raju, P.N.; Shanta, K.K.; Lakshmi, M.N. Preparation and characterization of lamivudine microcapsules using various cellulose polymers. Trop. J. Pharm. Res. 2011, 6, 841–847. [Google Scholar] [CrossRef]
- Farago, P.V.; Raffin, R.P.; Pohlmann, A.R. Physicochemical Characterization of a Hydrophilic Model Drug-Loaded PHBV Microparticles Obtained by the Double Emulsion / Solvent Evaporation Technique. J. Braz. Chem. Soc. 2008, 19, 1298–1305. [Google Scholar] [CrossRef]
- Li, W.G.; Wang, H.Q.; Shi, Y. Anti-hepatitis B effect of lamivudine loaded HPMC microspheres in DHBV infected ducklings: Optimization by 32 full factorial design. Biomed. Res. 2017, 28, 447–452. [Google Scholar]
- Nuyken, O.; Pask, S.D. Ring-Opening Polymerization—An Introductory Review. Polymers 2013, 5, 361–403. [Google Scholar] [CrossRef]
- Urbaniak, T.; Machová, D.; Janoušková, O.; Musiał, W. Microparticles of Lamivudine—Poly-ε-Caprolactone Conjugate for Drug Delivery via Internalization by Macrophages. Molecules 2019, 24, 723. [Google Scholar] [CrossRef] [PubMed]
- Oosterhuis, N.M.G.; Kossen, N.W.F. Power input measurements in a production scale bioreactor. Biotechnol. Lett. 1981, 3, 645–650. [Google Scholar] [CrossRef]
- Save, M.; Soum, A. Controlled ring-opening polymerization of lactones and lactide initiated by lanthanum isopropoxide, 2a: Mechanistic studies. Macromol. Chem. Phys. 2002, 203, 2591–2603. [Google Scholar] [CrossRef]
- United States Pharmacopeia and National Formulary (USP41–NF36); United States Pharmacopeial Convention: Rockville, MD, USA, 2016.
- Jyothi, N.V.N.; Prasanna, P.M.; Sakarkar, S.N.; Prabha, K.S.; Ramaiah, P.S.; Srawan, G.Y. Microencapsulation techniques, factors influencing encapsulation efficiency. J. Microencapsul. 2010, 27, 187–197. [Google Scholar] [CrossRef]
- Prakash, A.; Teotia, A.K.; Farooqi, J.A.; Singh, G.N. Forced degradation study of abacavir sulfate under the frame of genotoxic impurity. Indian J. Chem. Sect. B Org. Med. Chem. 2016, 55B, 213–219. [Google Scholar]
- Deshmukh, R.; Wagh, P.; Naik, J. Solvent evaporation and spray drying technique for micro- and nanospheres/particles preparation: A review. Dry. Technol. 2016, 34, 1758–1772. [Google Scholar] [CrossRef]
- Vankova, N.; Tcholakova, S.; Denkov, N.D.; Ivanov, I.B.; Vulchev, V.D.; Danner, T. Emulsification in turbulent flow: 1. Mean and maximum drop diameters in inertial and viscous regimes. J. Colloid Interface Sci. 2007, 312, 363–380. [Google Scholar]
- Pooja, D.; Tunki, L.; Kulhari, H.; Reddy, B.B. Data in Brief Optimization of solid lipid nanoparticles prepared by a single emulsi fi cation-solvent evaporation method. Data Br. 2016, 6, 15–19. [Google Scholar] [CrossRef]
- Tham, C.; Hamid, Z.A.A.; Ismail, H.; Ahmad, Z. Poly (Vinyl Alcohol) in Fabrication of PLA Micro- and Nanoparticles Using Emulsion and Solvent Evaporation Technique. Adv. Mater. Res. 2014, 1024, 296–299. [Google Scholar] [CrossRef]
- Honary, S.; Zahir, F. Effect of Zeta Potential on the Properties of nano-drug delivery systems-a review (Part 2). Trop. J. Pharm. al Res. 2013, 12, 265–273. [Google Scholar]
- Mishra, A.; Panola, R.; Rana, A.C. Microemulsions: As drug delivry system. J. Sci. Innov. Res. 2014, 3, 467–474. [Google Scholar]
- Eccleston, G.M. Structure and rheology of cetomacrogol creams: The influence of alcohol chain length and homologue composition. J. Pharm. Pharmacol. 1977, 29, 157–162. [Google Scholar] [CrossRef] [PubMed]
- Saunders, G.M.; Barry, B.W. Rheology of Systems Containing Cetomacrogol lO00-Cetostearyl Alcohol. J. Colloid Interface Sci. 1972, 38, 616–625. [Google Scholar]
- Taghipour, B.; Yakhchali, M.; Haririan, I.; Tamaddon, A.M.; Samani, S.M. The effects of technical and compositional variables on the size and release profile of bovine serum albumin from PLGA based particulate systems. Res. Pharm. Sci. 2014, 9, 407–420. [Google Scholar] [PubMed]
- Mahmood, T.; Akhtar, N.; Manickam, S. Interfacial film stabilized W/O/W nano multiple emulsions loaded with green tea and lotus extracts: Systematic characterization of physicochemical properties and shelf-storage stability. J. Nanobiotechnol. 2014, 12, 20. [Google Scholar] [CrossRef] [PubMed]
- Feczko, T.; Tóth, J.; Gyenis, J. Comparison of the preparation of PLGA–BSA nano- and microparticles by PVA, poloxamer and PVP. Colloids Surf. A Physicochem. Eng. Asp. 2008, 319, 188–195. [Google Scholar] [CrossRef]
- Maia, J.L.; Santana, M.H.A. The Effect of Some Processing Conditions on the Characteristics of Biodegradable Microspheres Obtained By an Emulsion Solvent Evaporation Process. Braz. J. Chem. Eng. 2004, 21, 1–12. [Google Scholar] [CrossRef]
- Sharma, N.; Madan, P.; Lin, S. Effect of process and formulation variables on the preparation of parenteral paclitaxel-loaded biodegradable polymeric nanoparticles: A co-surfactant study. Asian J. Pharm. Sci. 2016, 11, 404–416. [Google Scholar] [CrossRef] [Green Version]
- Zhao, H.; Gagnon, J.; Häfeli, U.O. Process and formulation variables in the preparation of injectable and biodegradable magnetic microspheres. Biomagn. Res. Technol. 2007, 5, 2. [Google Scholar] [CrossRef] [PubMed]
- Tcholakova, S.; Lesov, I.; Golemanov, K.; Denkov, N.D. Drop size in concentrated emulsions, obtained by rotor-stator homogenization. World Congr. Emuls. 2010, 2010, 3–9. [Google Scholar]
- Pacek, A.; Baker, M.; Utomo, A.T. Characterisation of Flow Pattern in a Rotor Stator High Shear Mixer. Proc. Eur. Congr. Chem. Eng. 2007, 1, 16–20. [Google Scholar]
- Dapčević Hadnadev, T.; Dokić, P.; Krstonošić, V.; Hadnadev, M. Influence of oil phase concentration on droplet size distribution and stability of oil-in-water emulsions. Eur. J. Lipid Sci. Technol. 2013, 115, 313–321. [Google Scholar] [CrossRef]






 ) in preparations ST1-ST3, the bars represent SD.
) in preparations ST1-ST3, the bars represent SD.
 ) in preparations ST1-ST3, the bars represent SD.
) in preparations ST1-ST3, the bars represent SD.






| Batch | Homogenization Rate (rpm) | Homogenization Time (min) | Surfactant Concentration (w/w%) | o/w Phase Ratio | Surfactant Type | Polymer Concentration (w/v%) |
|---|---|---|---|---|---|---|
| ST1 | 25,000 | 5 | 0.5 | 1:10 | PGHE | 0.17 |
| ST2 | 25,000 | 5 | 0.5 | 1:10 | Polysorbate 80 | 0.17 |
| ST3 | 25,000 | 5 | 0.5 | 1:10 | PVA | 0.17 |
| SC1 | 25,000 | 5 | 0.1 | 1:10 | PVA | 0.17 |
| SC2 | 25,000 | 5 | 0.2 | 1:10 | PVA | 0.17 |
| SC3 | 25,000 | 5 | 0.5 | 1:10 | PVA | 0.17 |
| SC4 | 25,000 | 5 | 1 | 1:10 | PVA | 0.17 |
| SC5 | 25 000 | 5 | 1.5 | 1:10 | PVA | 0.17 |
| SC6 | 25,000 | 5 | 2 | 1:10 | PVA | 0.17 |
| HS1 | 10,000 | 5 | 1 | 1:10 | PVA | 0.17 |
| HS2 | 15,000 | 5 | 1 | 1:10 | PVA | 0.17 |
| HS3 | 20,000 | 5 | 1 | 1:10 | PVA | 0.17 |
| HS4 | 25,000 | 5 | 1 | 1:10 | PVA | 0.17 |
| HS5 | 30,000 | 5 | 1 | 1:10 | PVA | 0.17 |
| HS6 | 35,000 | 5 | 1 | 1:10 | PVA | 0.17 |
| HT1 | 25,000 | 1 | 0.5 | 1:10 | PVA | 0.17 |
| HT2 | 25,000 | 3 | 0.5 | 1:10 | PVA | 0.17 |
| HT3 | 25,000 | 5 | 0.5 | 1:10 | PVA | 0.17 |
| HT4 | 25,000 | 7 | 0.5 | 1:10 | PVA | 0.17 |
| HT5 | 25,000 | 9 | 0.5 | 1:10 | PVA | 0.17 |
| HT6 | 25,000 | 11 | 0.5 | 1:10 | PVA | 0.17 |
| PR1 | 25,000 | 5 | 0.5 | 1:2 | PVA | 0.17 |
| PR2 | 25,000 | 5 | 0.5 | 1:4 | PVA | 0.17 |
| PR3 | 25,000 | 5 | 0.5 | 1:6 | PVA | 0.17 |
| PR4 | 25,000 | 5 | 0.5 | 1:10 | PVA | 0.17 |
| PR5 | 25,000 | 5 | 0.5 | 1:20 | PVA | 0.17 |
| PR6 | 25,000 | 5 | 0.5 | 1:40 | PVA | 0.17 |
| CM1 | 25,000 | 5 | 0.5 | 1:10 | PVA | 0.02 |
| CM2 | 25,000 | 5 | 0.5 | 1:10 | PVA | 0.03 |
| CM3 | 25,000 | 5 | 0.5 | 1:10 | PVA | 0.10 |
| CM4 | 25,000 | 5 | 0.5 | 1:10 | PVA | 0.17 |
| CM5 | 25,000 | 5 | 0.5 | 1:10 | PVA | 0.23 |
| CM6 | 25,000 | 5 | 0.5 | 1:10 | PVA | 0.33 |
| Batch | Hydrodynamic Diameter ± RSD (nm) | PDI ± RSD | Zeta Potential ± RSD (mV) |
|---|---|---|---|
| ST1 | 605 ± 14.05% | 0.84 ± 23.03% | −6.89 ± 2.51% |
| ST2 | 454 ± 2.65% | 0.46 ± 2.22% | −17.77 ± 15.52% |
| ST3 | 332 ± 1.63% | 0.29 ± 8.74% | −8.57 ± 4.00% |
| SC1 | 785 ± 2.22% | 0.26 ± 9.84% | −15.2 ± 1.14% |
| SC2 | 666 ± 1.11% | 0.3 ± 3.61% | −8.43 ± 1.96% |
| SC3 | 490 ± 3.17% | 0.35 ± 7.83% | −6.23 ± 4.36% |
| SC4 | 282 ± 1.04% | 0.36 ± 4.33% | −2.26 ± 7.12% |
| SC5 | 272 ± 1.59% | 0.21 ± 16.04% | −1.02 ± 10.01% |
| SC6 | 300 ± 1.33% | 0.22 ± 1.39% | −0.85 ± 9.88% |
| HS1 | 2344 ± 1.41% | 0.33 ± 9.24% | - |
| HS2 | 1041 ± 3.92% | 0.38 ± 9.49% | - |
| HS3 | 725 ± 4.31% | 0.29 ± 35.96% | - |
| HS4 | 552 ± 2.18% | 0.28 ± 2.78% | - |
| HS5 | 505 ± 1.94% | 0.22 ± 19.21% | - |
| HS6 | 412 ± 4.04% | 0.32 ± 12.31% | - |
| HT1 | 639 ± 0.54% | 0.34 ± 2.08% | - |
| HT2 | 422 ± 1.43% | 0.46 ± 1.95% | - |
| HT3 | 377 ± 2.22% | 0.34 ± 16.01% | - |
| HT4 | 310 ± 2.16% | 0.26 ± 4.89% | - |
| HT5 | 226 ± 1.58% | 0.24 ± 1.86% | - |
| HT6 | 235 ± 1.86% | 0.19 ± 4.38% | - |
| PR1 | 354 ± 2% | 0.24 ± 7.27% | - |
| PR2 | 361 ± 1.54% | 0.27 ± 4.26% | - |
| PR3 | 308 ± 0.74% | 0.25 ± 3.8% | - |
| PR4 | 351 ± 2.49% | 0.42 ± 6.02% | - |
| PR5 | 537 ± 1.58% | 0.58 ± 3.1% | - |
| PR6 | 351 ± 0.36% | 0.54 ± 8.66% | - |
| PM1 | 257 ± 1.89% | 0.35 ± 12.08% | - |
| PM2 | 294 ± 0.86% | 0.29 ± 1.09% | - |
| PM3 | 296 ± 0.26% | 0.27 ± 3.49% | - |
| PM4 | 307 ± 0.61% | 0.27 ± 3.01% | - |
| PM5 | 322 ± 2.35% | 0.36 ± 18.08% | - |
| PM6 | 293 ± 1.36% | 0.27 ± 13.94% | - |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Urbaniak, T.; Musiał, W. Influence of Solvent Evaporation Technique Parameters on Diameter of Submicron Lamivudine-Poly-ε-Caprolactone Conjugate Particles. Nanomaterials 2019, 9, 1240. https://doi.org/10.3390/nano9091240
Urbaniak T, Musiał W. Influence of Solvent Evaporation Technique Parameters on Diameter of Submicron Lamivudine-Poly-ε-Caprolactone Conjugate Particles. Nanomaterials. 2019; 9(9):1240. https://doi.org/10.3390/nano9091240
Chicago/Turabian StyleUrbaniak, Tomasz, and Witold Musiał. 2019. "Influence of Solvent Evaporation Technique Parameters on Diameter of Submicron Lamivudine-Poly-ε-Caprolactone Conjugate Particles" Nanomaterials 9, no. 9: 1240. https://doi.org/10.3390/nano9091240
APA StyleUrbaniak, T., & Musiał, W. (2019). Influence of Solvent Evaporation Technique Parameters on Diameter of Submicron Lamivudine-Poly-ε-Caprolactone Conjugate Particles. Nanomaterials, 9(9), 1240. https://doi.org/10.3390/nano9091240