Functionalized Multiwalled CNTs in Classical and Nonclassical CaCO3 Crystallization
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Oxidation and Functionalization of MWCNTs
2.3. Classical Crystallization of CaCO3
2.4. Nonclassical Crystallization of CaCO3
2.5. Spectroscopic, SEM, and TEM Analyses of MWCNTs and CaCO3 Crystals
2.6. Zeta Potential of Oxidized and Functionalized MWCNTs
2.7. Back Titration of COOH Groups of MWCNTs
3. Results and Discussion
3.1. Characterization of MWCNTs
3.2. Gas Diffusion (GD) as Classical Crystallization
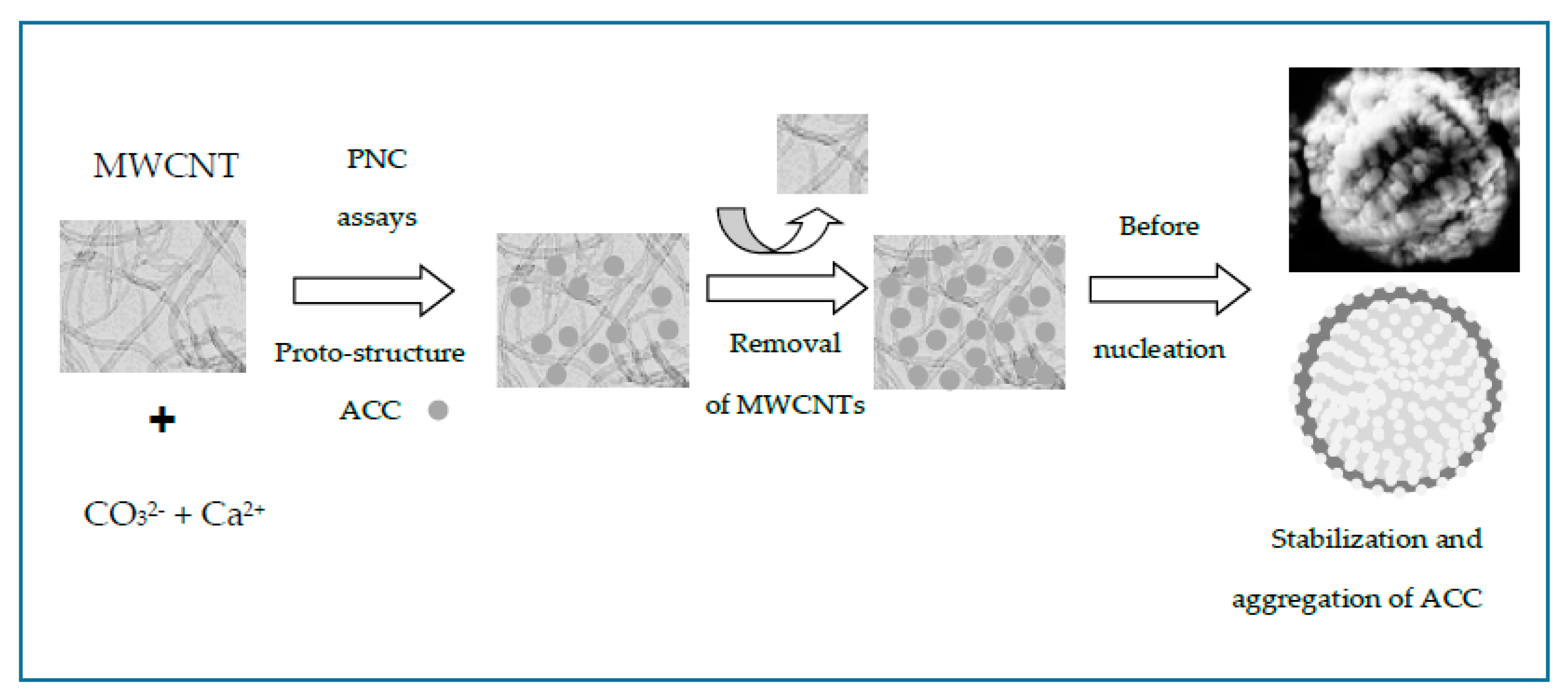
3.3. Prenucleation (PNC) as Nonclassical Crystallization
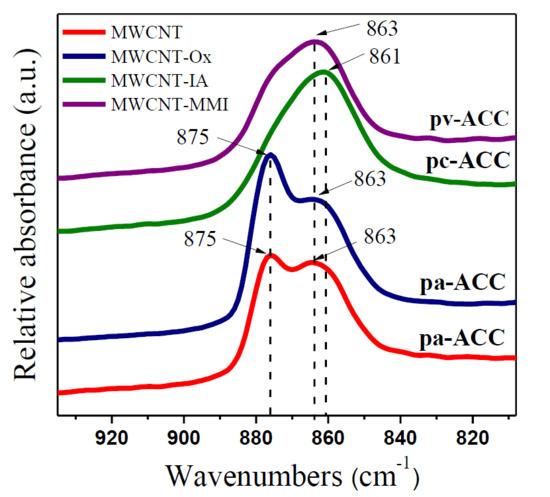
3.4. Stabilization of ACC Proto-Structures
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Schnorr, J.M.; Swager, T.M. Emerging applications of carbon nanotubes. Chem. Mater. 2011, 23, 646–657. [Google Scholar] [CrossRef]
- Van Bommel, K.J.C.; Friggeri, A.; Shinkai, S. Organic templates for the generation of inorganic materials. Angew. Chem. Int. Ed. 2003, 42, 980–999. [Google Scholar] [CrossRef]
- Declet, A.; Reyes, E.; Suarez, O. Calcium carbonate precipitation: A review of the carbonate crystallization process and applications in bioinspired composites. Rev. Adv. Mater. Sci. 2006, 44, 87–107. [Google Scholar]
- Anderson, R.E.; Barron, A.R. Effect of carbon nanomaterials on calcium carbonate crystallization. Main Group Chem. 2005, 4, 279–289. [Google Scholar] [CrossRef]
- Li, W.; Gao, C. Efficiently stabilized spherical vaterite CaCO3 crystals by carbon nanotubes in biomimetic mineralization. Langmuir 2007, 23, 4575–4582. [Google Scholar] [CrossRef] [PubMed]
- Tasis, D.; Pispas, S.; Bouropoulos, N. Growth of calcium carbonate on non-covalently modified carbon nanotubes. Mater. Lett. 2007, 61, 5044–5046. [Google Scholar] [CrossRef]
- Ford, W.E.; Yasuda, A.; Wessels, J.M. Microcrystalline composite particles of carbon nanotubes and calcium carbonate. Langmuir 2008, 24, 3479–3485. [Google Scholar] [CrossRef]
- Qi, C.; Lin, J.; Fu, L.H.; Huang, P. Calcium-based biomaterials for diagnosis, treatment, and theranostics. Chem. Soc. Rev. 2018, 47, 357–403. [Google Scholar] [CrossRef]
- Ren, D.; Feng, Q.; Bourrat, X. Effects of additives and templates on calcium carbonate mineralization in vitro. Micron 2011, 42, 228–245. [Google Scholar] [CrossRef]
- Gebauer, D.; Cölfen, H.; Verch, A.; Antonietti, M. The multiple roles of additives in CaCO3 crystallization: A quantitative case study. Adv. Mater. 2009, 21, 435–439. [Google Scholar] [CrossRef]
- Cölfen, H.; Antonietti, M. Crystal design of calcium carbonate microparticles using double-hydrophilic block copolymers. Langmuir 1998, 14, 582–589. [Google Scholar] [CrossRef]
- Zhang, Q.; Liu, S.J.; Yu, S.H. Recent advances in oriented attachment growth and synthesis of functional materials: Concept, evidence, mechanism, and future. J. Mater. Chem. 2009, 19, 191–207. [Google Scholar] [CrossRef]
- Farhadi-Khouzani, M.; Chevrier, D.M.; Zhang, P.; Hedin, N.; Gebauer, D. Water as the key to proto-aragonite amorphous CaCO3. Angew. Chem. Int. Ed. 2016, 55, 8117–8120. [Google Scholar] [CrossRef] [PubMed]
- Nebel, H.; Neumann, M.; Mayer, C.; Epple, M. On the structure of amorphous calcium carbonate—A detailed study by solid-state NMR spectroscopy. Inorg. Chem. 2008, 47, 7874–7879. [Google Scholar] [CrossRef] [PubMed]
- Aizenberg, J.; Lambert, G.; Weiner, S.; Addadi, L. Factors involved in the formation of amorphous and crystalline calcium carbonate: A study of an ascidian skeleton. J. Am. Chem. Soc. 2002, 124, 32–39. [Google Scholar] [CrossRef]
- Chu, H.; Wei, L.; Cui, R.; Wang, J.; Li, Y. Carbon nanotubes combined with inorganic nanomaterials: Preparations and applications. Coord. Chem. Rev. 2010, 254, 1117–1134. [Google Scholar] [CrossRef]
- Ivanov, V.K.; Fedorov, P.P.; Baranchikov, A.Y.; Osiko, V.V. Oriented attachment of particles: 100 years of investigations of non-classical crystal growth. Russ. Chem. Rev. 2014, 83, 1204–1222. [Google Scholar] [CrossRef]
- Yazdani-Pedram, M.; Vega, H.; Quijada, R. Melt functionalization of polypropylene with methyl esters of itaconic acid. Polymer 2001, 42, 4751–4758. [Google Scholar] [CrossRef]
- Avilés, F.; Cauich-Rodríguez, J.V.; Moo-Tah, L.; May-Pat, A.; Vargas-Coronado, R. Evaluation of mild acid oxidation treatments for MWCNT functionalization. Carbon 2009, 47, 2970–2975. [Google Scholar] [CrossRef]
- Xu, A.W.; Antonietti, M.; Yu, S.H.; Cölfen, H. Polymer mediated mineralization and self-similar mesoscale-organized calcium carbonate with unusual superstructures. Adv. Mater. 2008, 20, 1333–1338. [Google Scholar] [CrossRef]
- Neira-Carrillo, A.; Vásquez-Quitral, P.; Yazdani-Pedram, M.; Arias, J.L. Synthesis and characterization of sulfonated polymethylsiloxane polymer as template for crystal growth of CaCO3. Colloid Polym. Sci. 2009, 287, 385–393. [Google Scholar] [CrossRef]
- Neira-Carrillo, A.; Pai, K.R.; Fernández, M.S.; Carreño, E.; Vásquez-Quitral, P.; Arias, J.L. Crystal growth of CaCO3 induced by monomethylitaconate grafted polymethylsiloxane. Eur. Polym. J. 2010, 46, 1184–1193. [Google Scholar] [CrossRef]
- Gebauer, D.; Gunawidjaja, P.N.; Ko, J.Y.; Bacsik, Z.; Aziz, B.; Liu, L.; Hu, Y.; Bergström, L.; Tai, C.W.; Sham, T.K.; et al. Proto-calcite and proto-vaterite in amorphous calcium carbonates. Angew. Chem. Int. Ed. 2010, 49, 8889–8891. [Google Scholar] [CrossRef]
- Rieger, J.; Thieme, J.; Schmidt, C. Study of precipitation reactions by X-ray microscopy: CaCO3 precipitation and the effect of polycarboxylates. Langmuir 2000, 16, 8300–8305. [Google Scholar] [CrossRef]
- Neira-Carrillo, A.; Acevedo, D.F.; Peralta, D.O.; Barbero, C.; Cölfen, H.; Arias, J.L. Influence of conducting polymers based on carboxylated polyaniline on in vitro CaCO3 crystallization. Langmuir 2008, 24, 12496–12507. [Google Scholar] [CrossRef] [PubMed]
- Beato, C.; Fernández, M.S.; Fermani, S.; Reggi, M.; Neira-Carrillo, A.; Rao, A.; Falini, G.; Arias, J.L. Calcium carbonate crystallization in tailored constrained environments. CrystEngComm 2015, 17, 5953–5961. [Google Scholar] [CrossRef]
- Neira-Carrillo, A.; Gentsch, R.; Börner, H.G.; Acevedo, D.F.; Barbero, C.A.; Cölfen, H. Templated CaCO3 crystallization by submicrometer and nanosized fibers. Langmuir 2016, 32, 8951–8959. [Google Scholar] [CrossRef] [PubMed]
- Cölfen, H.; Qi, L. A systematic examination of the morphogenesis of calcium carbonate in the presence of a double-hydrophilic block copolymer. Chem. Eur. J. 2001, 7, 106–116. [Google Scholar] [CrossRef]
- Gebauer, D.; Völkel, A.; Cölfen, H. Stable prenucleation calcium carbonate clusters. Science 2008, 322, 1819–1822. [Google Scholar] [CrossRef]
- Rao, A.; Vásquez-Quitral, P.; Fernández, M.S.; Berg, J.K.; Sánchez, M.; Drechsler, M.; Neira-Carrillo, A.; Arias, J.L.; Gebauer, D.; Cölfen, H. pH-dependent schemes of calcium carbonate formation in the presence of alginates. Cryst. Growth Des. 2016, 16, 1349–1359. [Google Scholar] [CrossRef]
- Avaro, J.T.; Ruiz-Agudo, C.; Landwehr, E.; Hauser, K.; Gebauer, D. Impurity-free amorphous calcium carbonate, a preferential material for pharmaceutical and medical applications. Eur. J. Mineral. 2019, 31, 231–236. [Google Scholar] [CrossRef]
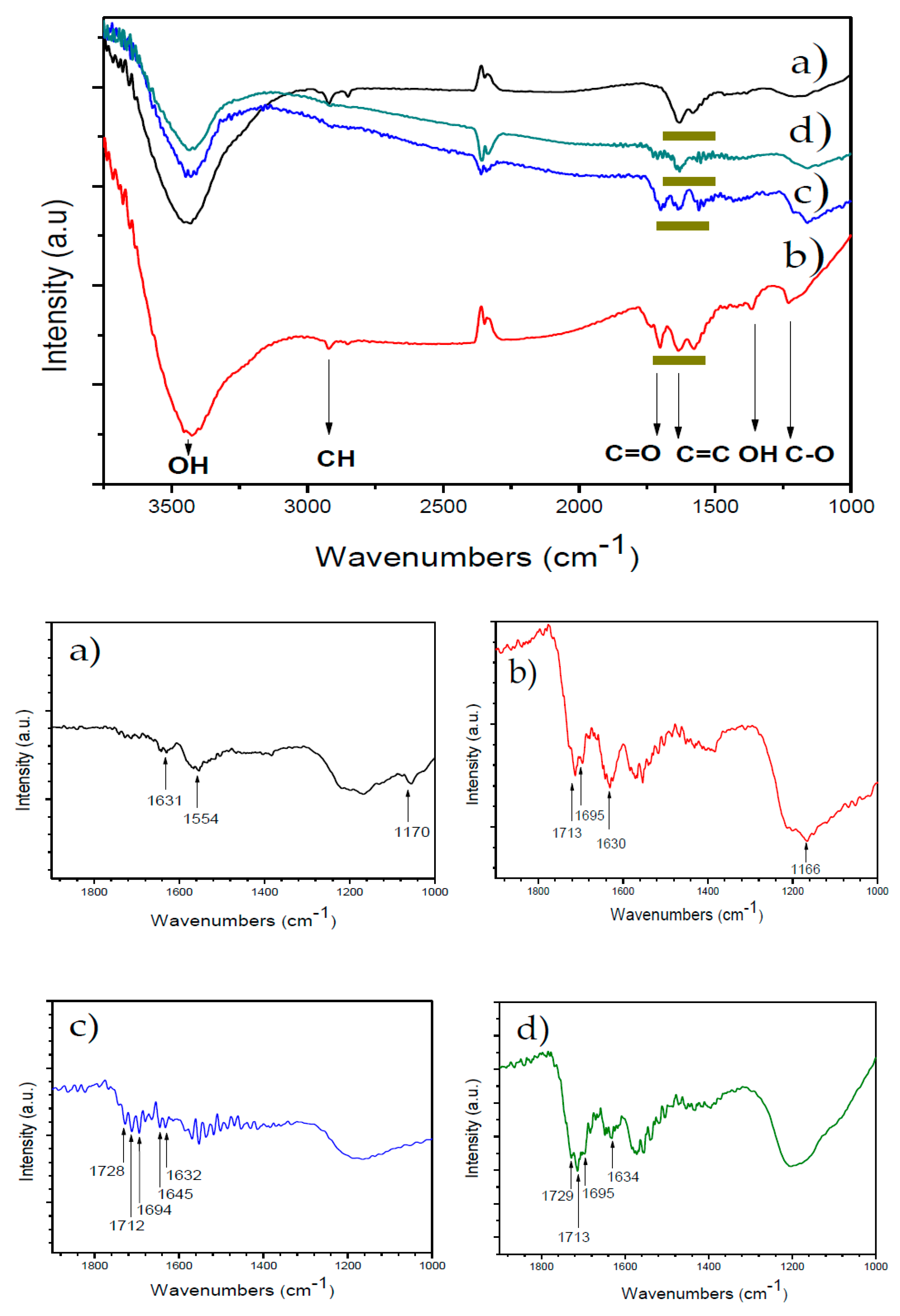

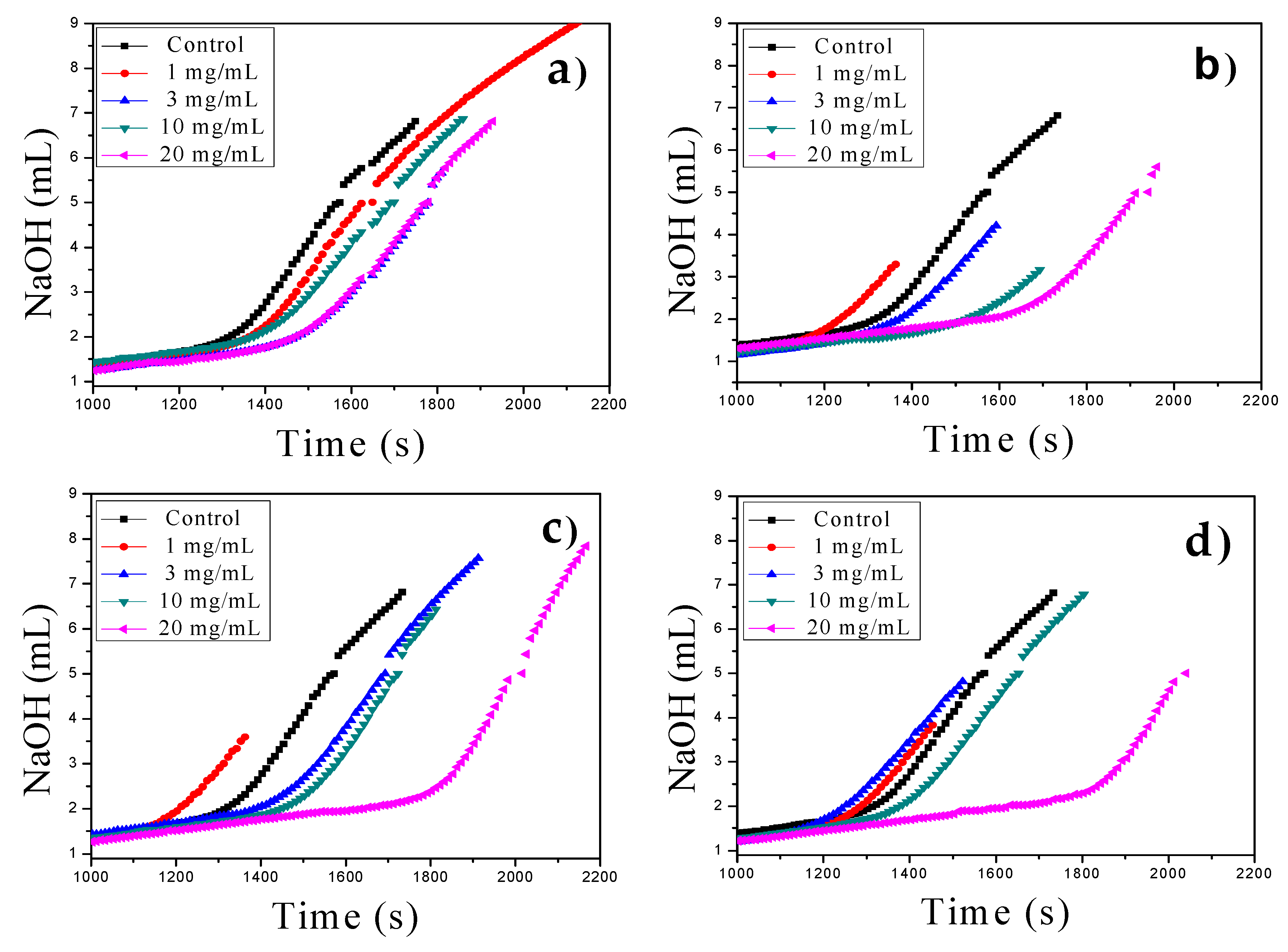
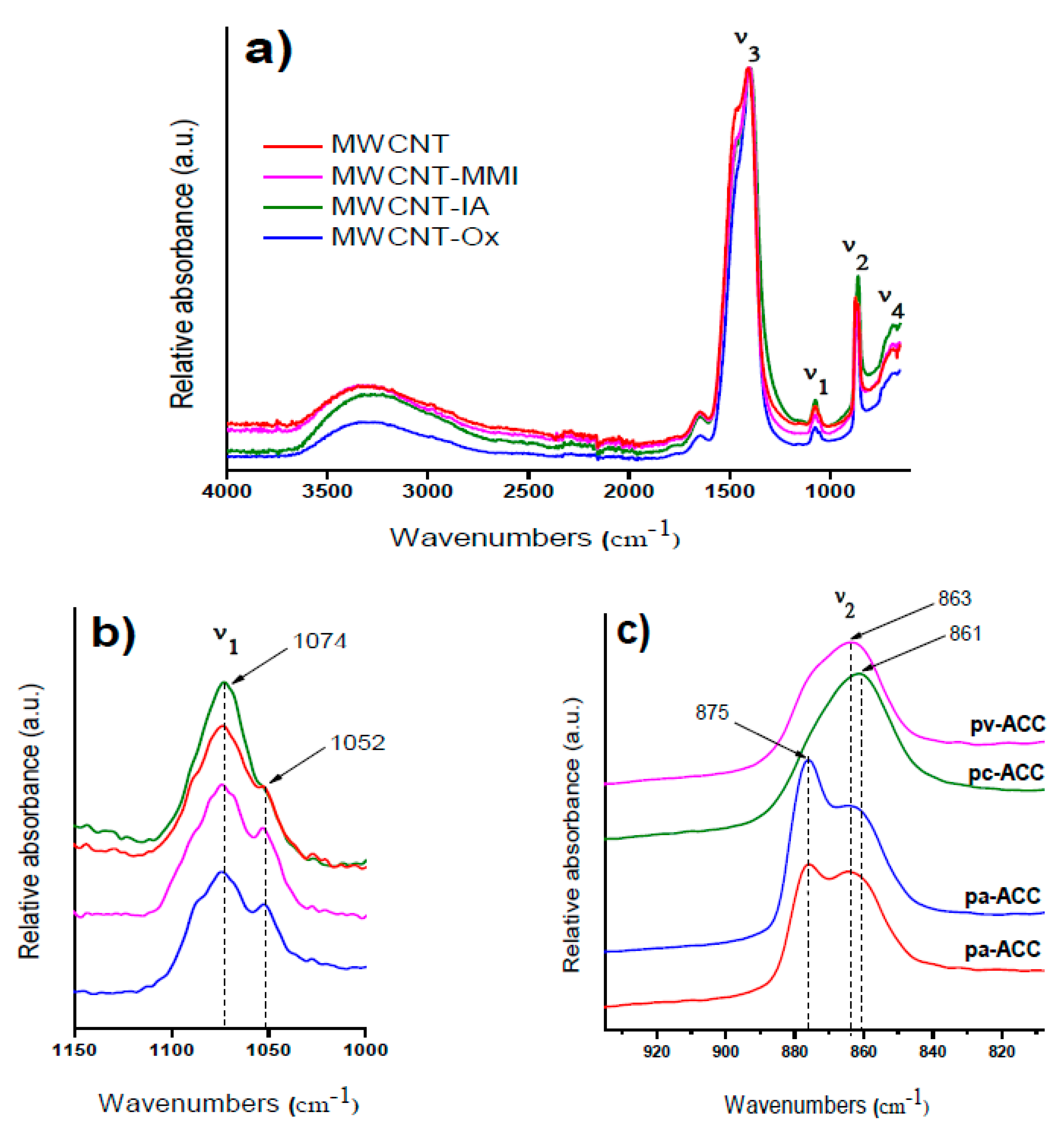


| pH | MWCNT | MWCNT-Ox | MWCNT-IA | MWCNT-MMI |
|---|---|---|---|---|
| 4.0 | +9.2 ± 3.5 | −24.4 ± 10.5 | −22.9 ± 4.1 | −22.3 ± 3.0 |
| 7.0 | −51.4 ± 0.9 | −54.7 ± 1.2 | −51.0 ± 3.1 | −59.5 ± 1.0 |
| 9.0 | −46.3 ± 0.7 | −64.6 ± 1.5 | −50.1 ± 4.1 | −53.6 ± 3.8 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Neira-Carrillo, A.; Vásquez-Quitral, P.; Sánchez, M.; Farhadi-Khouzani, M.; Aguilar-Bolados, H.; Yazdani-Pedram, M.; Cölfen, H. Functionalized Multiwalled CNTs in Classical and Nonclassical CaCO3 Crystallization. Nanomaterials 2019, 9, 1169. https://doi.org/10.3390/nano9081169
Neira-Carrillo A, Vásquez-Quitral P, Sánchez M, Farhadi-Khouzani M, Aguilar-Bolados H, Yazdani-Pedram M, Cölfen H. Functionalized Multiwalled CNTs in Classical and Nonclassical CaCO3 Crystallization. Nanomaterials. 2019; 9(8):1169. https://doi.org/10.3390/nano9081169
Chicago/Turabian StyleNeira-Carrillo, Andrónico, Patricio Vásquez-Quitral, Marianela Sánchez, Masoud Farhadi-Khouzani, Héctor Aguilar-Bolados, Mehrdad Yazdani-Pedram, and Helmut Cölfen. 2019. "Functionalized Multiwalled CNTs in Classical and Nonclassical CaCO3 Crystallization" Nanomaterials 9, no. 8: 1169. https://doi.org/10.3390/nano9081169
APA StyleNeira-Carrillo, A., Vásquez-Quitral, P., Sánchez, M., Farhadi-Khouzani, M., Aguilar-Bolados, H., Yazdani-Pedram, M., & Cölfen, H. (2019). Functionalized Multiwalled CNTs in Classical and Nonclassical CaCO3 Crystallization. Nanomaterials, 9(8), 1169. https://doi.org/10.3390/nano9081169