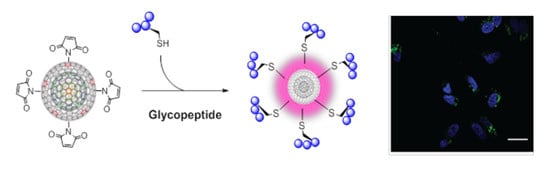
Carbon Nano-Onions as Non-Cytotoxic Carriers for Cellular Uptake of Glycopeptides and Proteins
Abstract
1. Introduction
2. Materials and Methods
2.1. Synthesis and Functionalization of Carbon Nano-Onions (CNOs)
2.1.1. p-CNOs
2.1.2. ox-CNOs
2.1.3. PEG-CNOs
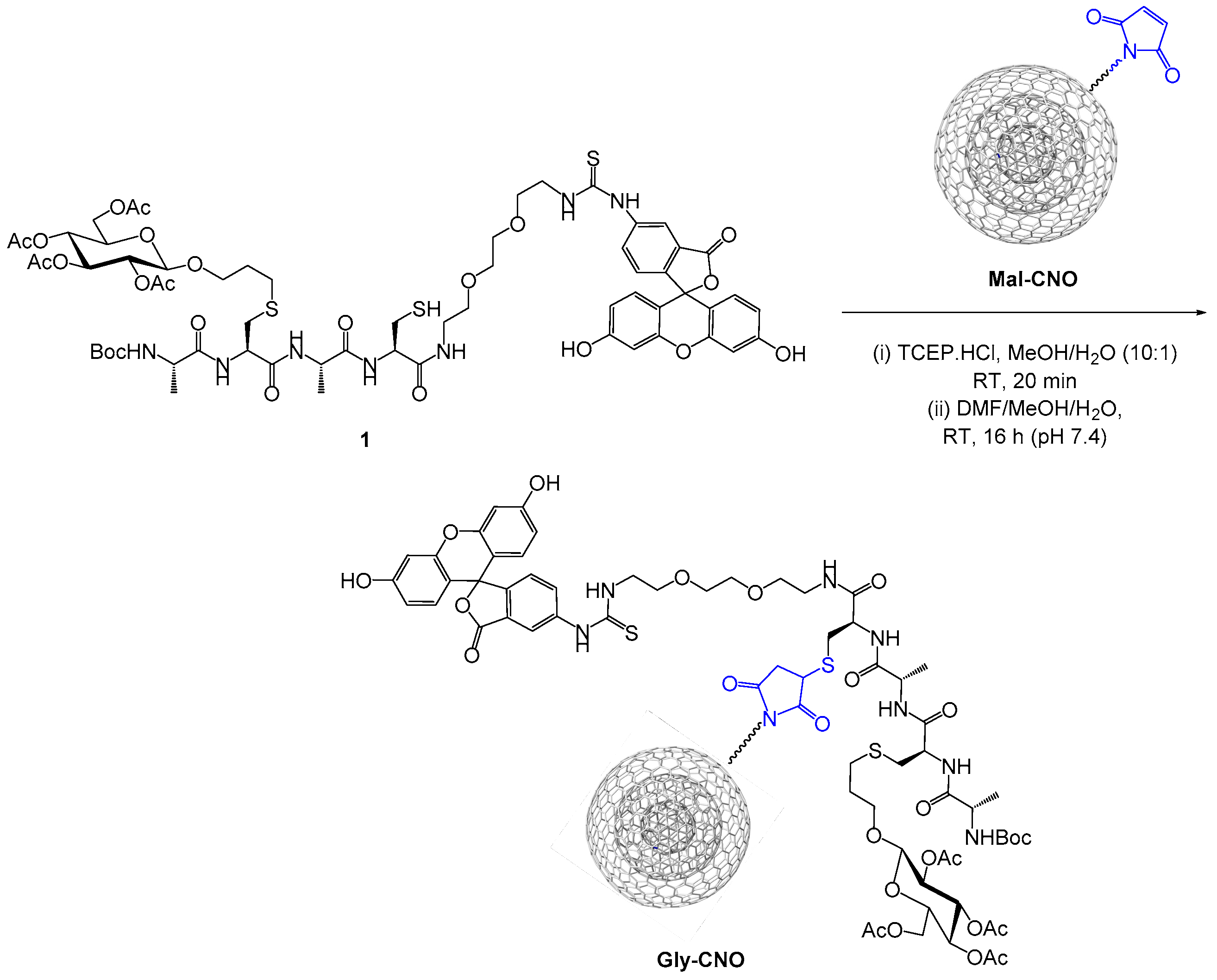
2.1.4. Maleimido Decorated CNOs (Mal-CNOs)
2.1.5. Glycopeptide-CNO Conjugates (Gly-CNOs)
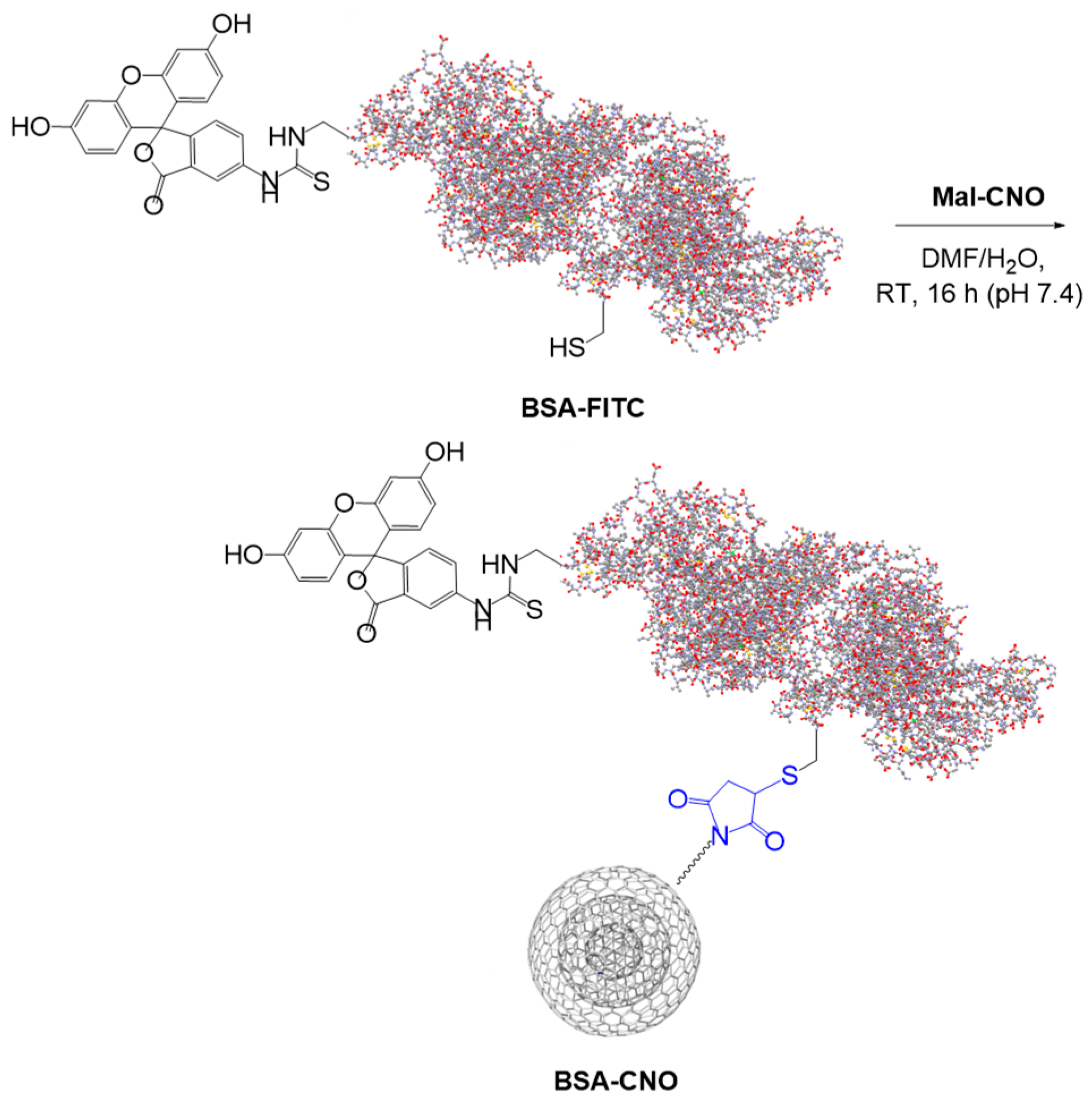
2.1.6. Bovine Serum Albumin-CNO Conjugates (BSA-CNOs)
2.1.7. Transmission Electron Microscopy (TEM) Analysis of CNOs
2.2. Biological Studies
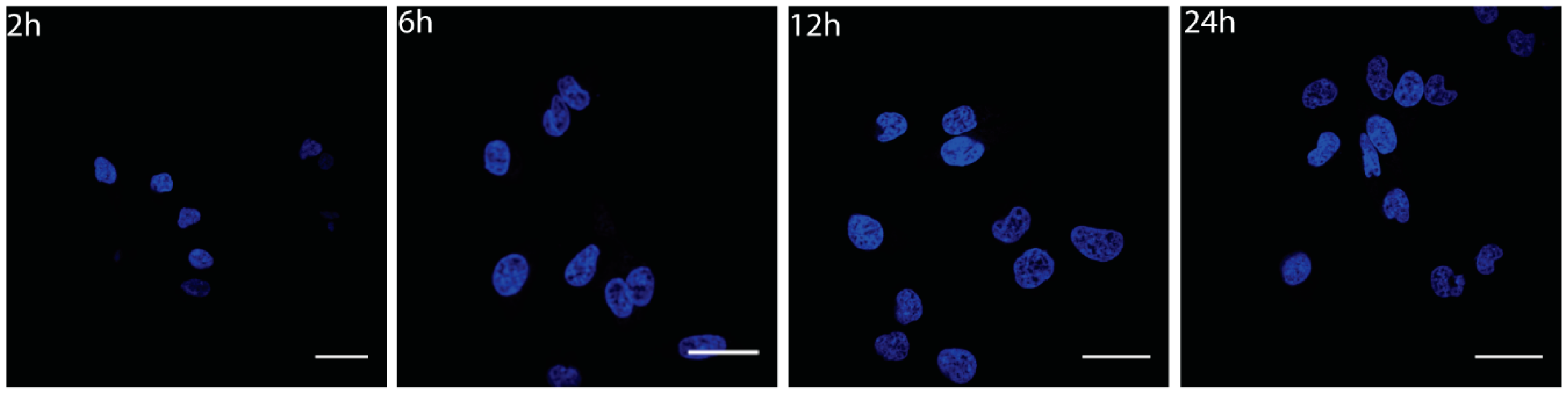
2.2.1. Cell Culture
2.2.2. Cytotoxicity Analysis
2.2.3. Cellular Uptake Mechanism
3. Results
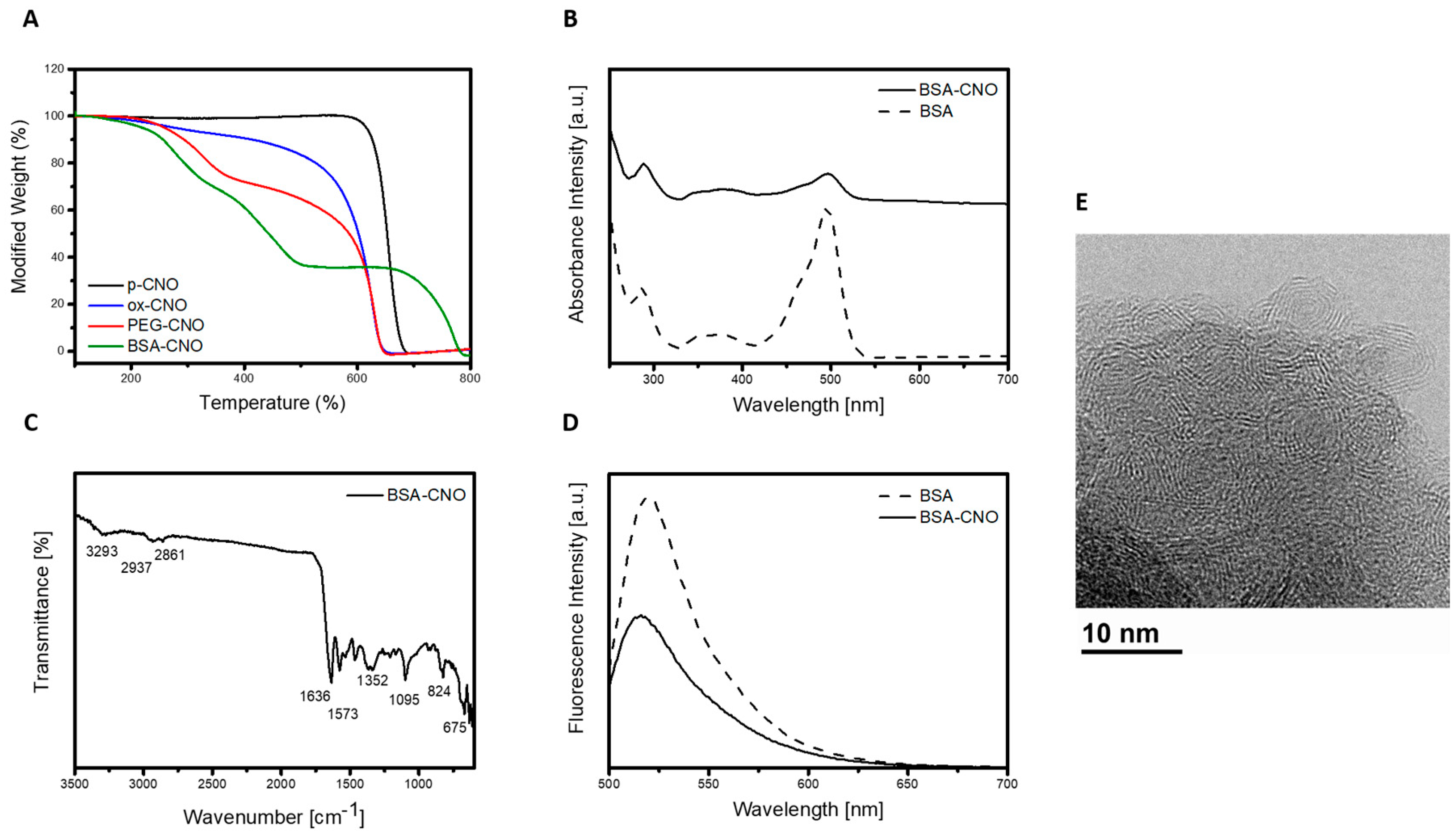
3.1. Synthesis of Gly-CNOs and BSA-CNOs
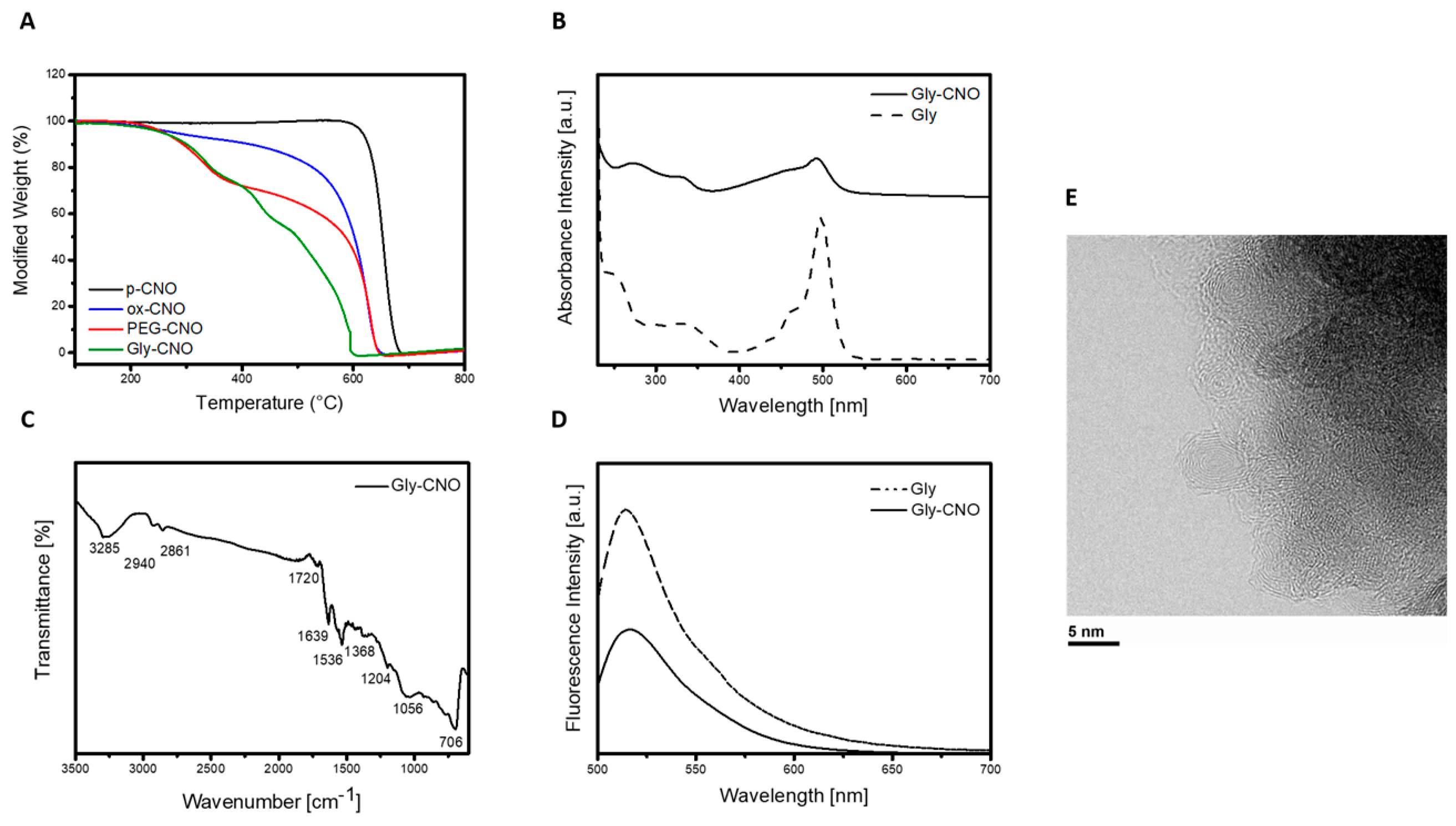
3.2. Characterization of Gly-CNOs and BSA-CNOs
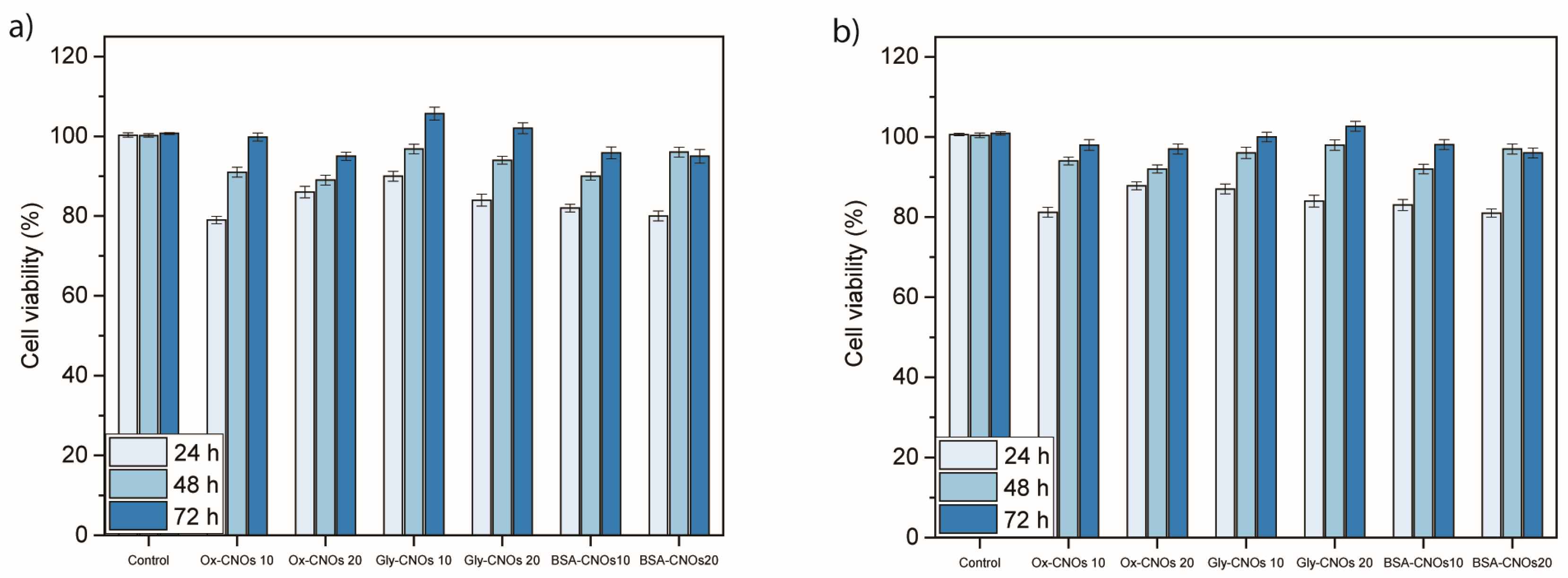
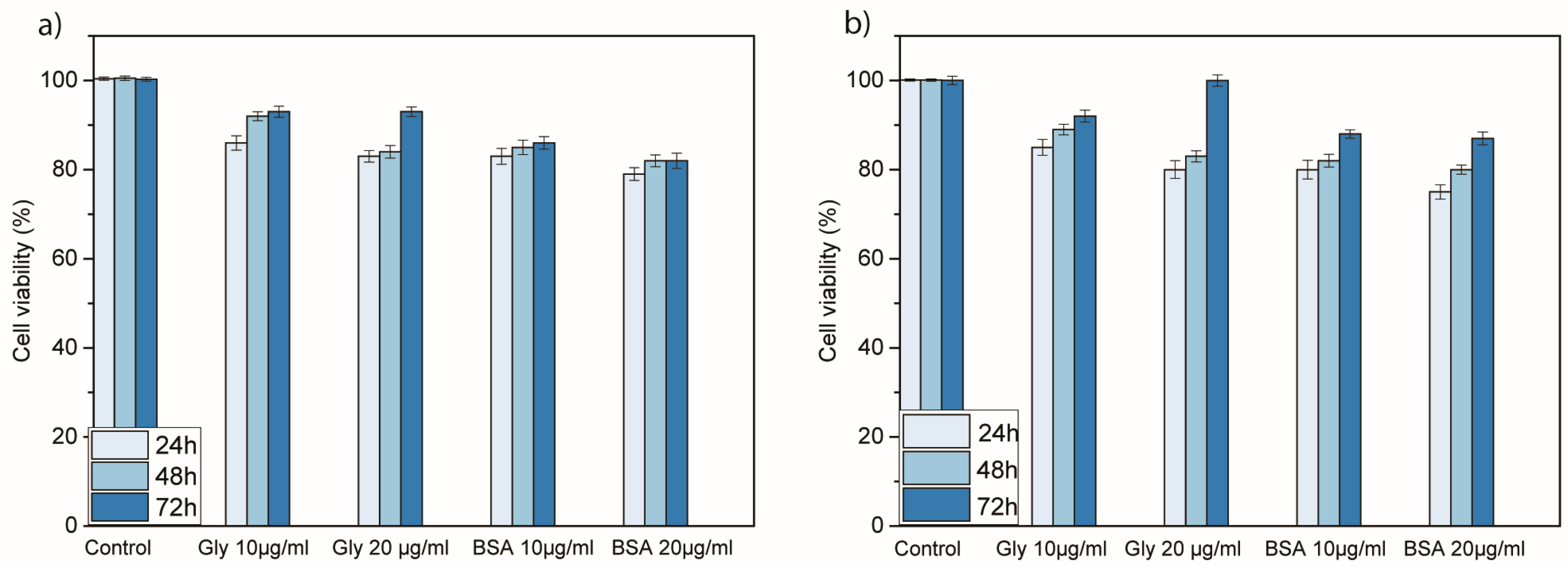
3.3. Cytotoxicity Analysis of Nanoconjugates
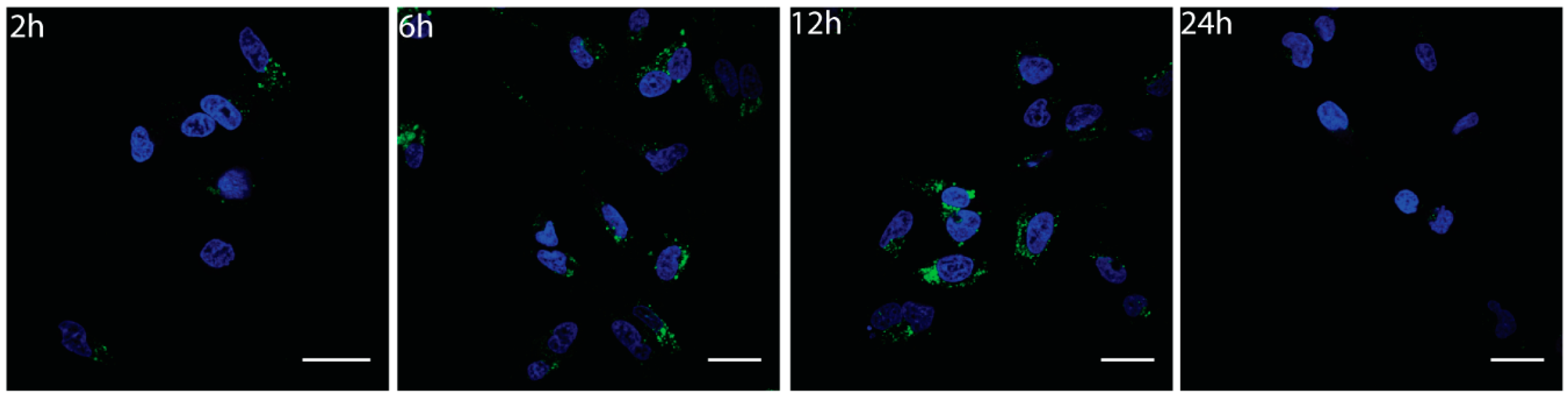
3.4. Cellular Uptake of Nanoconjugates
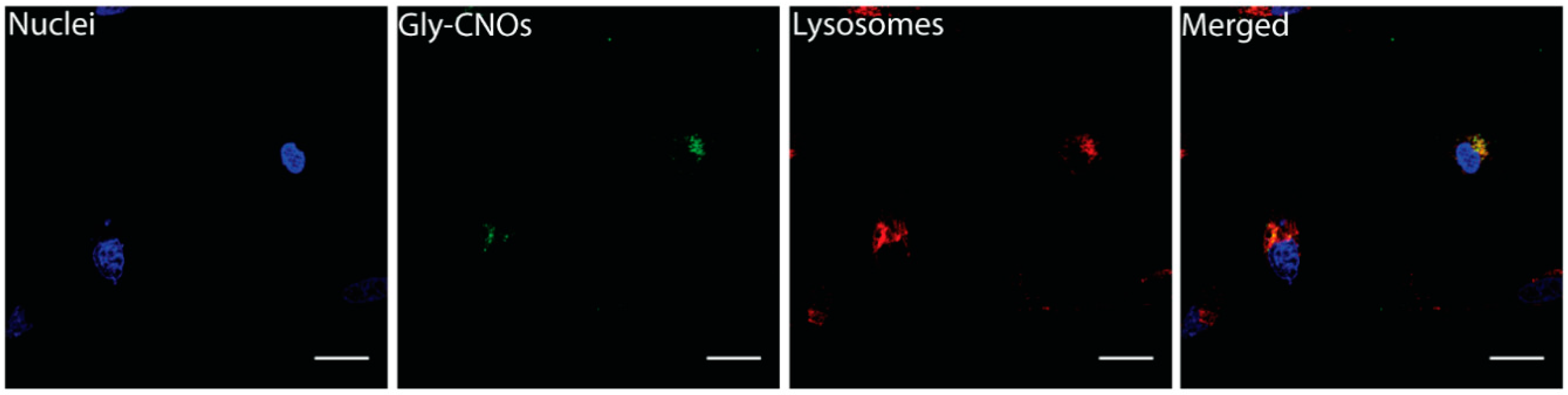
3.4.1. Cellular Internalization and Localization of Gly-CNOs
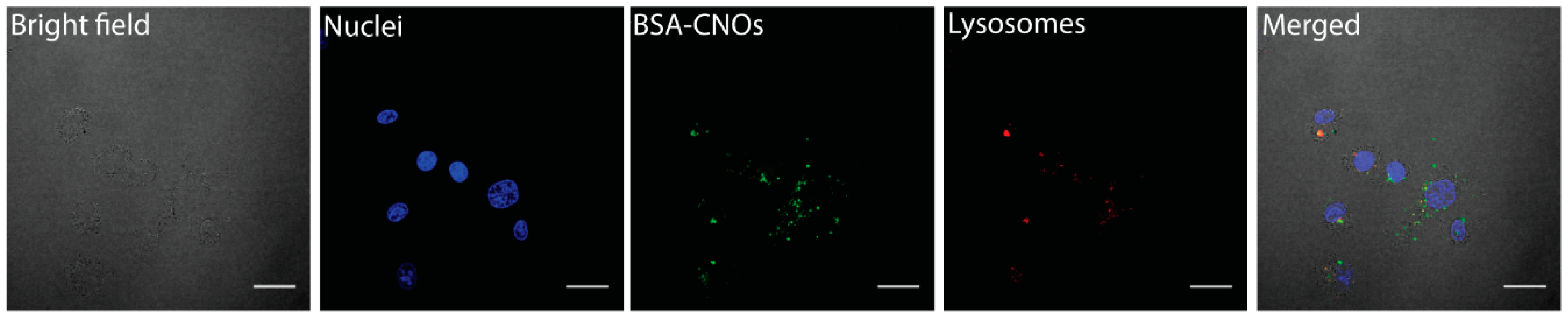
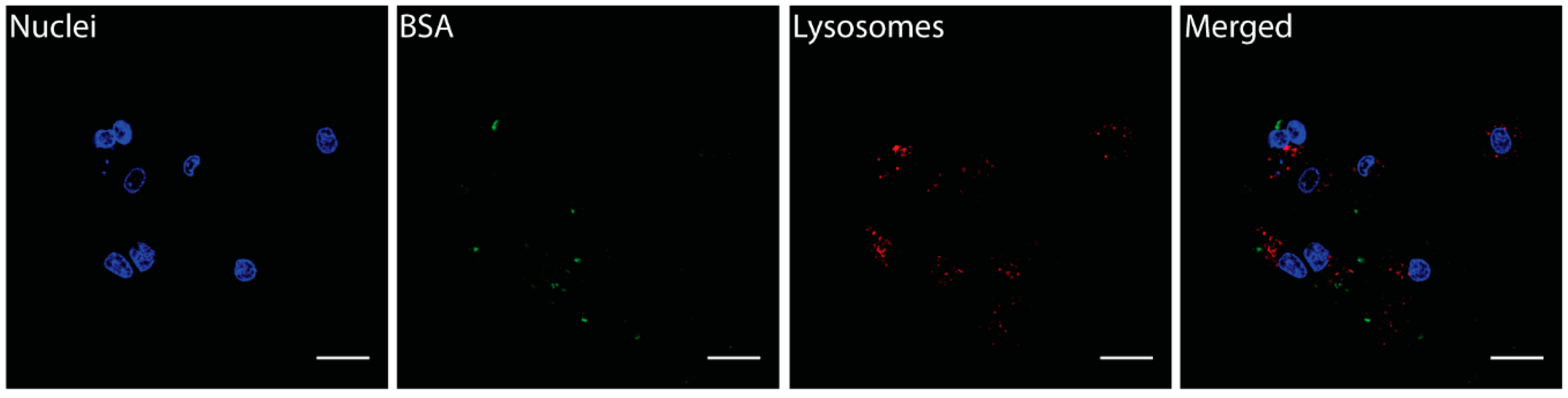
3.4.2. Cellular Internalization and Localization of BSA-CNOs
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bartelmess, J.; Quinn, S.J.; Giordani, S. Carbon nanomaterials: Multi-functional agents for biomedical fluorescence and Raman imaging. Chem. Soc. Rev. 2015, 44, 4672–4698. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, S.F.; Bisker, G.; Bakh, N.A.; Gibbs, S.L.; Landry, M.P.; Strano, M.S. Protein functionalized carbon nanomaterials for biomedical applications. Carbon 2015, 95, 767–779. [Google Scholar] [CrossRef]
- Liu, Z.; Robinson, J.T.; Tabakman, S.M.; Yang, K.; Dai, H. Carbon materials for drug delivery & cancer therapy. Mater. Today 2011, 14, 316–323. [Google Scholar]
- Zhang, R.; Olin, H. Carbon nanomaterials as drug carriers: Real time drug release investigation. Mater. Sci. Eng. C 2012, 32, 1247–1252. [Google Scholar] [CrossRef]
- Amora, M.; Giordani, S. Carbon Nanomaterials for Nanomedicine. In Smart Nanoparticles for Biomedicine; Elsevier: Amsterdam, The Netherlands, 2018; pp. 103–113. [Google Scholar]
- Bartelmess, J.; Giordani, S. Carbon nano-onions (multi-layer fullerenes): Chemistry and applications. Beilstein J. Nanotechnol. 2014, 5, 1980–1998. [Google Scholar] [CrossRef] [PubMed]
- Bartelmess, J.; Baldrighi, M.; Nardone, V.; Parisini, E.; Buck, D.; Echegoyen, L.; Giordani, S. Synthesis and Characterization of Far-Red/NIR-Fluorescent BODIPY Dyes, Solid-State Fluorescence, and Application as Fluorescent Tags Attached to Carbon Nano-onions. Chem. A Eur. J. 2015, 21, 9727–9732. [Google Scholar] [CrossRef] [PubMed]
- Frasconi, M.; Marotta, R.; Markey, L.; Flavin, K.; Spampinato, V.; Ceccone, G.; Echegoyen, L.; Scanlan, E.M.; Giordani, S. Multi-functionalized carbon nano-onions as imaging probes for cancer cells. Chem. A Eur. J. 2015, 21, 19071–19080. [Google Scholar] [CrossRef] [PubMed]
- Amora, M.; Camisasca, A.; Lettieri, S.; Giordani, S. Toxicity Assessment of Carbon Nanomaterials in Zebrafish during Development. Nanomaterials 2017, 7, 414. [Google Scholar]
- Amora, M.; Rodio, M.; Bartelmess, J.; Sancataldo, G.; Brescia, R.; Cella Zanacchi, F.; Diaspro, A.; Giordani, S. Biocompatibility and biodistribution of functionalized carbon nano-onions (f-CNOs) in a vertebrate model. Sci. Rep. 2016, 6, 33923. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.; Flavin, K.; Kopf, I.; Radics, G.; Hearnden, C.H.; McManus, G.J.; Moran, B.; Villalta-Cerdas, A.; Echegoyen, L.A.; Giordani, S.; et al. Functionalization of carbon nanoparticles modulates inflammatory cell recruitment and NLRP3 inflammasome activation. Small 2013, 9, 4194–4206. [Google Scholar] [CrossRef] [PubMed]
- Giordani, S.; Bartelmess, J.; Frasconi, M.; Biondi, I.; Cheung, S.; Grossi, M.; Wu, D.; Echegoyen, L.; O’Shea, D.F. NIR fluorescence labelled carbon nano-onions: Synthesis, analysis and cellular imaging. J. Mater. Chem. B 2014, 2, 7459–7463. [Google Scholar] [CrossRef]
- Bartelmess, J.; De Luca, E.; Signorelli, A.; Baldrighi, M.; Becce, M.; Brescia, R.; Nardone, V.; Parisini, E.; Echegoyen, L.; Pompa, P.P.; et al. Boron dipyrromethene (BODIPY) functionalized carbon nano-onions for high resolution cellular imaging. Nanoscale 2014, 6, 13761–13769. [Google Scholar] [CrossRef] [PubMed]
- Lettieri, S.; d’Amora, M.; Camisasca, A.; Diaspro, A.; Giordani, S. Carbon nano-onions as fluorescent on/off modulated nanoprobes for diagnostics. Beilstein J. Nanotechnol. 2017, 8, 1878–1888. [Google Scholar] [CrossRef] [PubMed]
- Lettieri, S.; Camisasca, A.; d’Amora, M.; Diaspro, A.; Uchida, T.; Nakajima, Y.; Yanagisawa, K.; Maekawa, T.; Giordani, S. Far-red fluorescent carbon nano-onions as a biocompatible platform for cellular imaging. RSC Adv. 2017, 7, 45676–45681. [Google Scholar] [CrossRef]
- Gaillard, C.; Cellot, G.; Li, S.; Toma, F.M.; Dumortier, H.; Spalluto, G.; Cacciari, B.; Prato, M.; Ballerini, L.; Bianco, A. Carbon Nanotubes Carrying Cell-Adhesion Peptides do not Interfere with Neuronal Functionality. Adv. Mater. 2009, 21, 2903–2908. [Google Scholar] [CrossRef]
- Bartolome, J.P.; Echegoyen, L.; Fragoso, A. Reactive Carbon Nano-Onion Modified Glassy Carbon Surfaces as DNA Sensors for Human Papillomavirus Oncogene Detection with Enhanced Sensitivity. Anal. Chem. 2015, 87, 6744–6751. [Google Scholar] [CrossRef] [PubMed]
- Sun, T.; Zhang, Y.S.; Pang, B.; Hyun, D.C.; Yang, M.; Xia, Y. Engineered nanoparticles for drug delivery in cancer therapy. Angew. Chem. Int. Ed. Engl. 2014, 53, 12320–12364. [Google Scholar] [CrossRef] [PubMed]
- Markey, L.; Giordani, S.; Scanlan, E.M. Native Chemical Ligation, Thiol–Ene Click: A Methodology for the Synthesis of Functionalized Peptides. J. Org. Chem. 2013, 78, 4270–4277. [Google Scholar] [CrossRef] [PubMed]
- Hanaor, D.; Michelazzi, M.; Leonelli, C.; Sorrell, C.C. The effects of carboxylic acids on the aqueous dispersion and electrophoretic deposition of ZrO2. J. Eur. Ceram. Soc. 2012, 32, 235–244. [Google Scholar] [CrossRef]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
d’Amora, M.; Maffeis, V.; Brescia, R.; Barnes, D.; Scanlan, E.; Giordani, S. Carbon Nano-Onions as Non-Cytotoxic Carriers for Cellular Uptake of Glycopeptides and Proteins. Nanomaterials 2019, 9, 1069. https://doi.org/10.3390/nano9081069
d’Amora M, Maffeis V, Brescia R, Barnes D, Scanlan E, Giordani S. Carbon Nano-Onions as Non-Cytotoxic Carriers for Cellular Uptake of Glycopeptides and Proteins. Nanomaterials. 2019; 9(8):1069. https://doi.org/10.3390/nano9081069
Chicago/Turabian Styled’Amora, Marta, Viviana Maffeis, Rosaria Brescia, Danielle Barnes, Eoin Scanlan, and Silvia Giordani. 2019. "Carbon Nano-Onions as Non-Cytotoxic Carriers for Cellular Uptake of Glycopeptides and Proteins" Nanomaterials 9, no. 8: 1069. https://doi.org/10.3390/nano9081069
APA Styled’Amora, M., Maffeis, V., Brescia, R., Barnes, D., Scanlan, E., & Giordani, S. (2019). Carbon Nano-Onions as Non-Cytotoxic Carriers for Cellular Uptake of Glycopeptides and Proteins. Nanomaterials, 9(8), 1069. https://doi.org/10.3390/nano9081069