Effect of Nano-SnS and Nano-MoS2 on the Corrosion Protection Performance of the Polyvinylbutyral and Zinc-Rich Polyvinylbutyral Coatings
Abstract
1. Introduction
2. Experimental
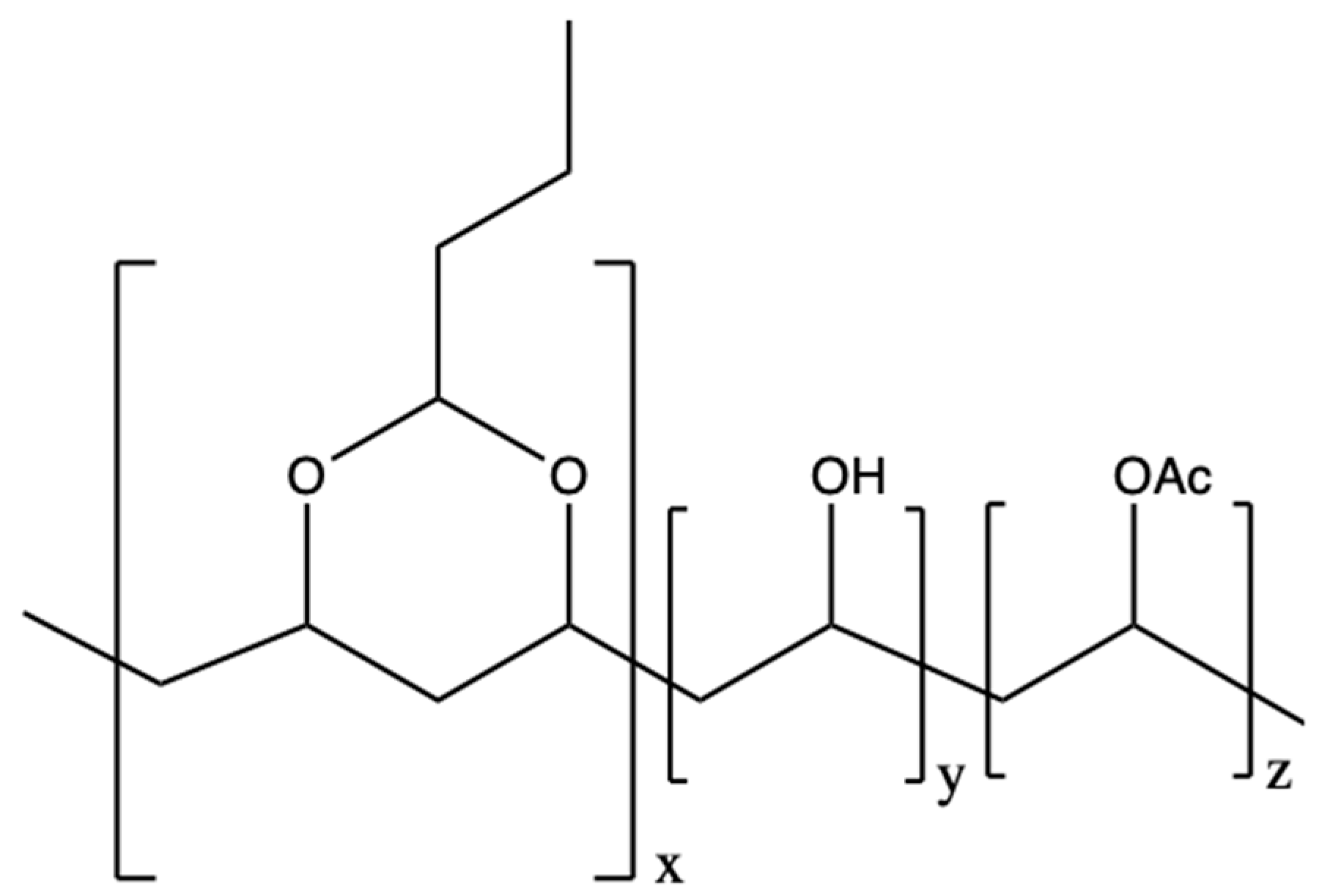
2.1. Material and Sample Preparation
2.2. Weight Loss Measurements
2.3. Electrochemical Experiments
2.4. Scanning Electron Microscope (SEM) Observations
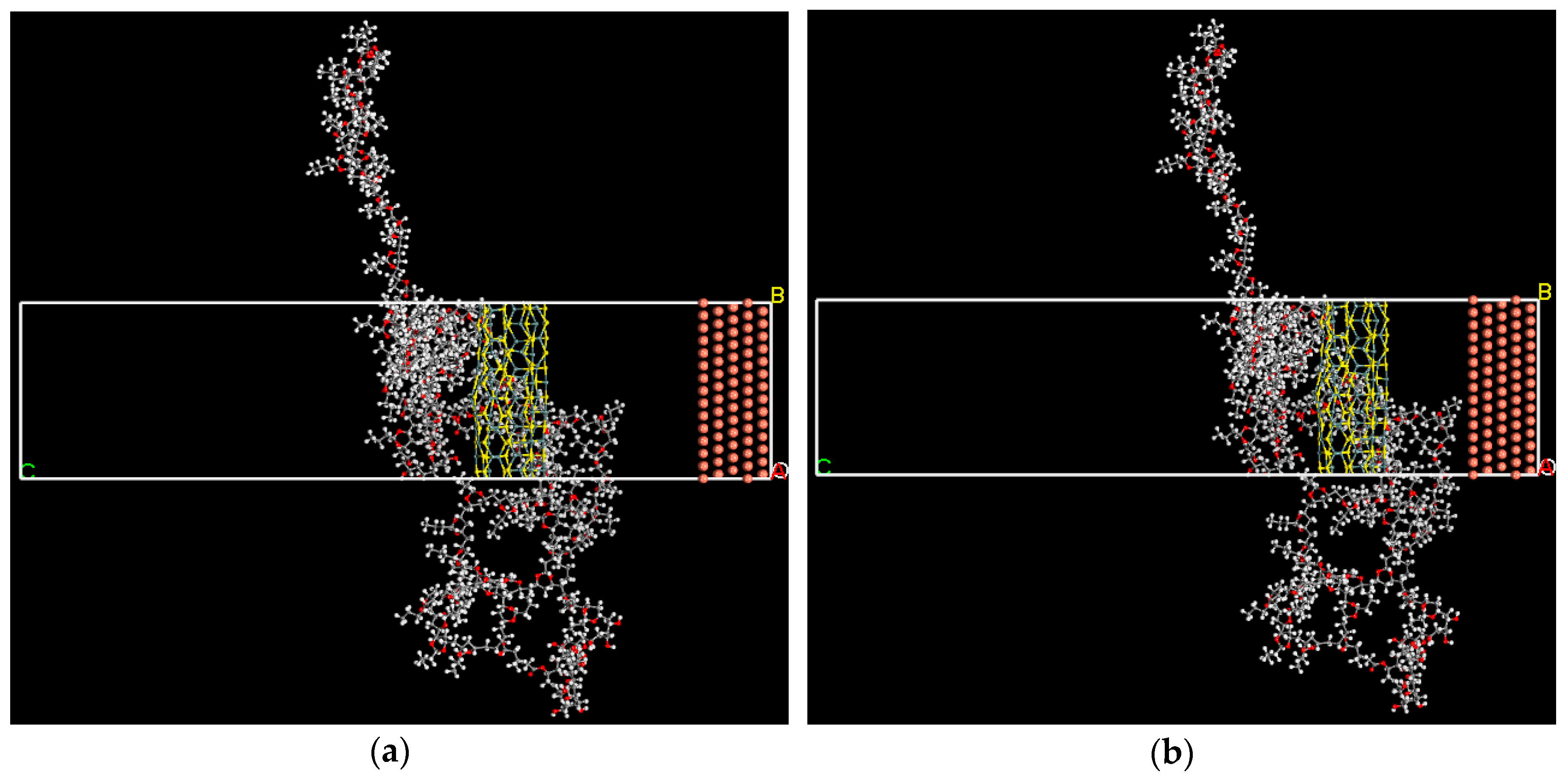
2.5. Theoretical Study
3. Results and Discussion
3.1. Weight Loss Measurements
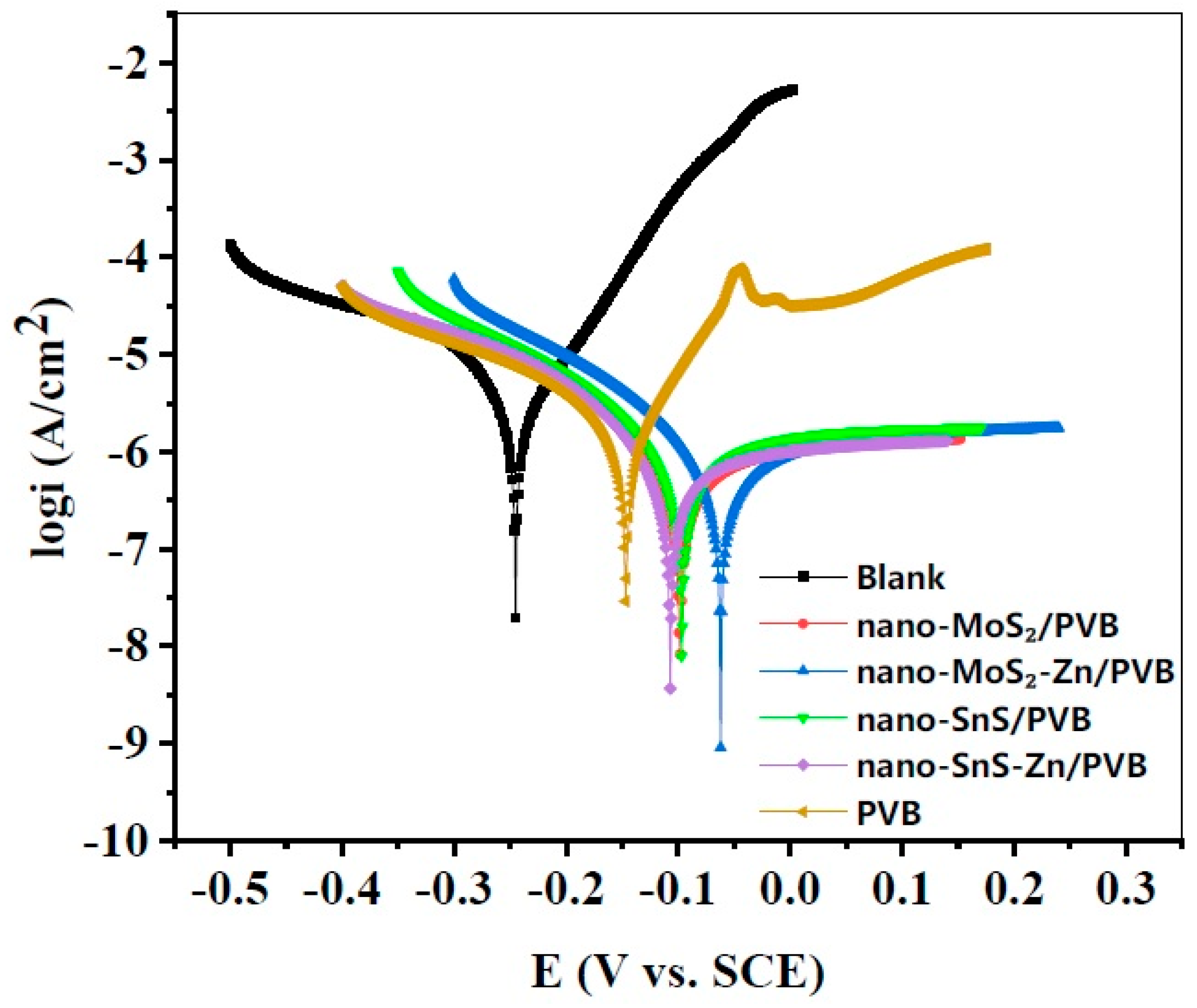
3.2. Polarization Curve
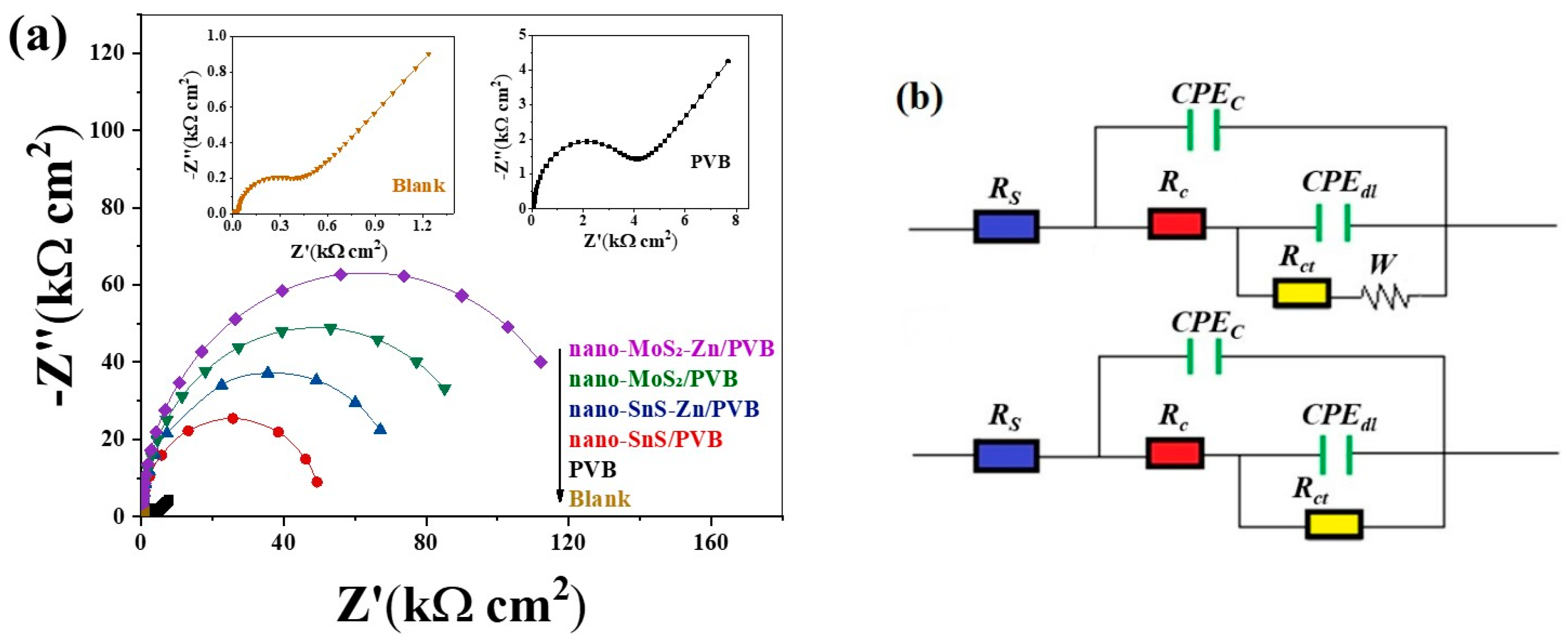
3.3. Electrochemical Impedance Spectroscopy (EIS)
3.4. SEM Analyses
3.5. Corrosion Mechanism Analysis
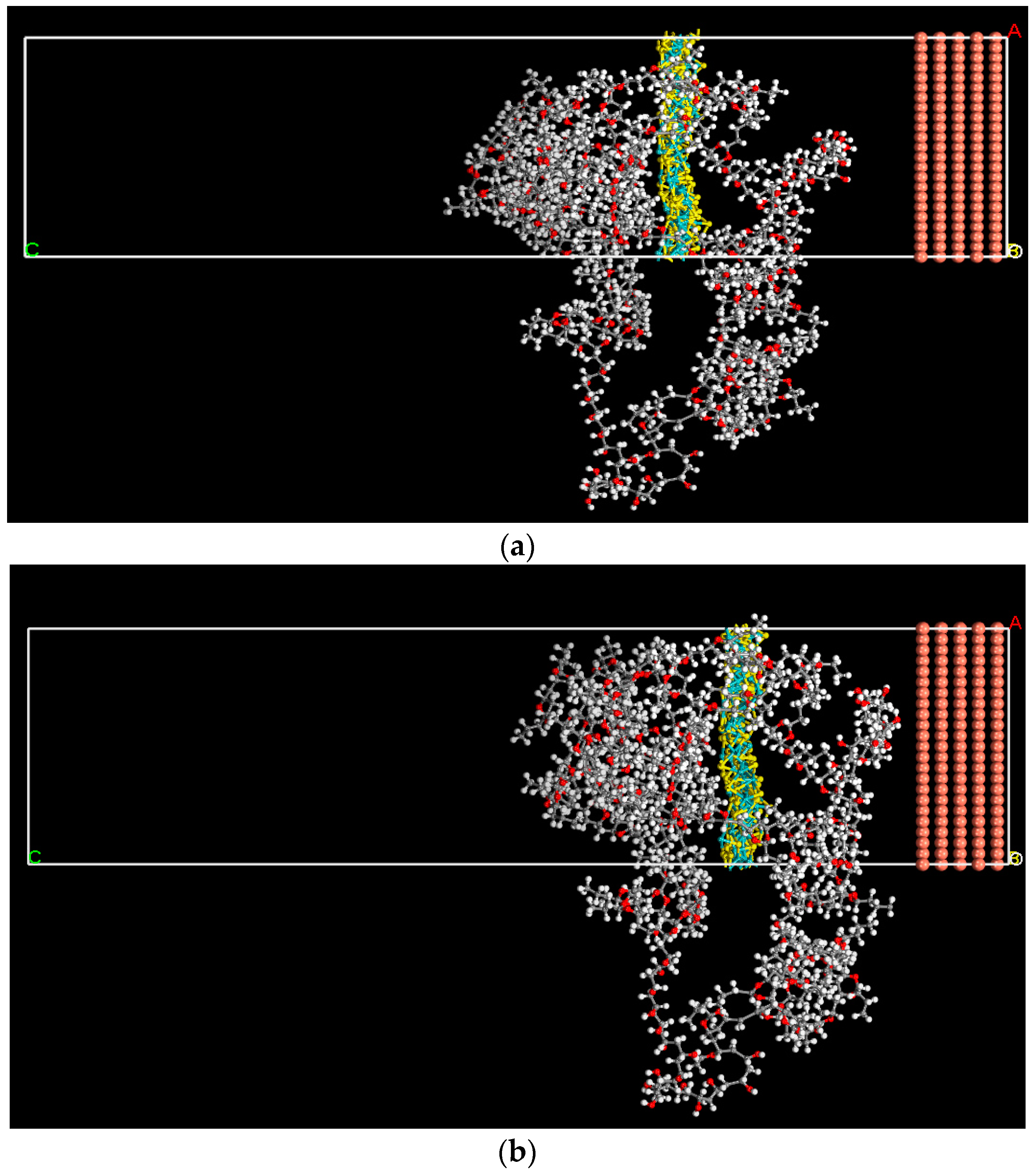
3.6. Molecular Dynamics Simulation
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Qiang, Y.; Zhang, S.; Xu, S.; Yin, L. The effect of 5-nitroindazole as an inhibitor for the corrosion of copper in a 3.0% NaCl solution. RSC Adv. 2015, 5, 63866–63873. [Google Scholar] [CrossRef]
- Al Kharafi, F.M.; Ghayad, I.M.; Abdallah, R.M. Corrosion inhibition of copper in seawater by 4-amino-4h-1,2,4-triazole-3-thiol. Corrosion 2013, 69, 58–66. [Google Scholar] [CrossRef]
- Amin, M.A. Weight loss, polarization, electrochemical impedance spectroscopy, SEM and EDX studies of the corrosion inhibition of copper in aerated NaCl solutions. J. Appl. Electrochem. 2006, 36, 215–226. [Google Scholar] [CrossRef]
- Ran, M.; Zheng, W.; Wang, H. Fabrication of superhydrophobic surfaces for corrosion protection: A review. Mater. Sci. Technol. 2019, 35, 313–326. [Google Scholar] [CrossRef]
- Heakal, F.E.-T.; Fekry, A.M.; Fatayerji, M.Z. Influence of halides on the dissolution and passivation behavior of AZ91D magnesium alloy in aqueous solutions. Electrochim. Acta 2009, 54, 1545–1557. [Google Scholar] [CrossRef]
- Zeino, A.; Abdulazeez, I.; Khaled, M.; Jawich, M.W.; Obot, I.B. Mechanistic study of polyaspartic acid (PASP) as eco-friendly corrosion inhibitor on mild steel in 3% NaCl aerated solution. J. Mol. Liq. 2018, 250, 50–62. [Google Scholar] [CrossRef]
- Qiang, Y.; Zhang, S.; Xu, S.; Li, W. Experimental and theoretical studies on the corrosion inhibition of copper by two indazole derivatives in 3.0% NaCl solution. J. Colloid Interface Sci. 2016, 472, 52–59. [Google Scholar] [CrossRef]
- Xu, S.; Gao, X.; Sun, J.; Hu, M.; Wang, D.; Jiang, D.; Zhou, F.; Weng, L.; Liu, W. Comparative study of moisture corrosion to WS2 and WS2/Cu multilayer films. Surf. Coat. Technol. 2014, 247, 30–38. [Google Scholar] [CrossRef]
- Sun, W.; Wang, L.; Wu, T.; Pan, Y.; Liu, G. Inhibited corrosion-promotion activity of graphene encapsulated in nanosized silicon oxide. J. Mater. Chem. A 2015, 3, 16843–16848. [Google Scholar] [CrossRef]
- Yu, Y.-H.; Lin, Y.-Y.; Lin, C.-H.; Chan, C.-C.; Huang, Y.-C. High-performance polystyrene/graphene-based nanocomposites with excellent anti-corrosion properties. Polym. Chem. 2014, 5, 535–550. [Google Scholar] [CrossRef]
- Cui, C.; Lim, A.T.O.; Huang, J. A cautionary note on graphene anti-corrosion coatings. Nat. Nanotechnol. 2017, 12, 834–835. [Google Scholar] [CrossRef] [PubMed]
- Ali, S.; Wang, F.; Zafar, S.; Iqbal, T. Hydrothermal synthesis, characterization and raman vibrations of chalcogenide SnS nanorods. In Proceedings of the 5th Annual International Conference on Material Science and Engineering, Xiamen, China, 20–22 Ocotber 2017; Aleksandrova, M., Szewczyk, R., Eds.; IOP Publishing: Bristol, UK, 2018; Volume 275. [Google Scholar]
- Gonzalez-Flores, V.E.; Mohan, R.N.; Ballinas-Morales, R.; Nair, M.T.S.; Nair, P.K. Thin film solar cells of chemically deposited SnS of cubic and orthorhombic structures. Thin Solid Film. 2019, 672, 62–65. [Google Scholar] [CrossRef]
- Cabrera-German, D.; Garcia-Valenzuela, J.A.; Cota-Leal, M.; Martinez-Gil, M.; Aceves, R.; Sotelo-Lerma, M. Detailed characterization of good-quality SnS thin films obtained by chemical solution deposition at different reaction temperatures. Mater. Sci. Semicond. Process. 2019, 89, 131–142. [Google Scholar] [CrossRef]
- Ye, J.; Qi, L.; Liu, B.; Xu, C. Facile preparation of hexagonal tin sulfide nanoplates anchored on graphene nanosheets for highly efficient sodium storage. J. Colloid Interface Sci. 2018, 513, 188–197. [Google Scholar] [CrossRef] [PubMed]
- Reddy, N.K.; Devika, M.; Gopal, E.S.R. Review on tin (II) sulfide (SnS) material: Synthesis, properties, and applications. Crit. Rev. Solid State Mater. Sci. 2015, 40, 359–398. [Google Scholar] [CrossRef]
- Li, Z.; Hui, S.; Yang, J.; Hua, Y. A facile strategy for the fabrication of superamphiphobic MoS2 film on steel substrates with excellent anti-corrosion property. Mater. Lett. 2018, 229, 336–339. [Google Scholar] [CrossRef]
- Thangasamy, P.; Partheeban, T.; Sudanthiramoorthy, S.; Sathish, M. Enhanced superhydrophobic performance of BN-MoS2 heterostructure prepared via a rapid, one-pot supercritical fluid processing. Langmuir 2017, 33, 6159–6166. [Google Scholar] [CrossRef]
- Rheem, Y.; Han, Y.; Lee, K.H.; Choi, S.-M.; Myung, N.V. Synthesis of hierarchical MoO2/MoS2 nanofibers for electrocatalytic hydrogen evolution. Nanotechnology 2017, 28, 105605. [Google Scholar] [CrossRef]
- Zhou, Y.; Liu, Y.; Zhao, W.; Xie, F.; Xu, R.; Li, B.; Zhou, X.; Shen, H. Growth of vertically aligned MoS2 nanosheets on a Ti substrate through a self-supported bonding interface for high-performance lithium-ion batteries: A general approach. J. Mater. Chem. A 2016, 4, 5932–5941. [Google Scholar] [CrossRef]
- Gao, X.; Wang, X.; Ouyang, X.; Wen, C. Flexible superhydrophobic and superoleophilic MoS2 sponge for highly efficient oil-water separation. Sci. Rep. 2016, 6, 27207. [Google Scholar] [CrossRef]
- Shi, J.; Zhang, X.; Ma, D.; Zhu, J.; Zhang, Y.; Guo, Z.; Yao, Y.; Ji, Q.; Song, X.; Zhang, Y.; et al. Substrate facet effect on the growth of mono layer MoS2 on Au foils. ACS Nano 2015, 9, 4017–4025. [Google Scholar] [CrossRef] [PubMed]
- Hu, L.; Zhang, S.; Li, W.; Hou, B. Electrochemical and thermodynamic investigation of diniconazole and triadimefon as corrosion inhibitors for copper in synthetic seawater. Corros. Sci. 2010, 52, 2891–2896. [Google Scholar] [CrossRef]
- Tang, Y.; Zhang, X.; Choi, P.; Xu, Z.; Liu, Q. Contributions of van der Waals interactions and hydrophobic attraction to molecular adhesions on a hydrophobic MoS2 surface in water. Langmuir 2018, 34, 14196–14203. [Google Scholar] [CrossRef] [PubMed]
- Kozbial, A.; Gong, X.; Liu, H.; Li, L. Understanding the intrinsic water wettability of molybdenum disulfide (MoS2). Langmuir 2015, 31, 8429–8435. [Google Scholar] [CrossRef]
- Chen, C.; He, Y.; Xiao, G.; Xia, Y.; Li, H.; He, Z. Two-dimensional hybrid materials: MoS2-RGO nanocomposites enhanced the barrier properties of epoxy coating. Appl. Surf. Sci. 2018, 444, 511–521. [Google Scholar] [CrossRef]
- Taheri, N.N.; Ramezanzadeh, B.; Mandavian, M.; Bahlakeh, G. In-situ synthesis of Zn doped polyaniline on graphene oxide for inhibition of mild steel corrosion in 3.5 wt.% chloride solution. J. Ind. Eng. Chem. 2018, 63, 322–339. [Google Scholar] [CrossRef]
- Ding, R.; Zheng, Y.; Yu, H.; Li, W.; Wang, X.; Gui, T. Study of water permeation dynamics and anti-corrosion mechanism of graphene/zinc coatings. J. Alloys Compd. 2018, 748, 481–495. [Google Scholar] [CrossRef]
- Zhang, X.; Hao, H.; Shi, Y.; Cui, J. The mechanical properties of Polyvinyl Butyral (PVB) at high strain rates. Constr. Build. Mater. 2015, 93, 404–415. [Google Scholar] [CrossRef]
- Rappé, A.K.; Casewit, C.J.; Colwell, K.; Goddard, W.A., III; Skiff, W.M. UFF, a full periodic table force field for molecular mechanics and molecular dynamics simulations. J. Am. Chem. Soc. 1992, 114, 10024–10035. [Google Scholar] [CrossRef]
- Tian, H.; Li, W.; Cao, K.; Hou, B. Potent inhibition of copper corrosion in neutral chloride media by novel non-toxic thiadiazole derivatives. Corros. Sci. 2013, 73, 281–291. [Google Scholar] [CrossRef]
- Hu, C.; Li, Y.; Zhang, N.; Ding, Y. Synthesis and characterization of a poly(o-anisidine)-SiC composite and its application for corrosion protection of steel. RSC Adv. 2017, 7, 11732–11742. [Google Scholar] [CrossRef]
- Tian, H.; Li, W.; Liu, A.; Gao, X.; Han, P.; Ding, R.; Yang, C.; Wang, D. Controlled delivery of multi-substituted triazole by metal-organic framework for efficient inhibition of mild steel corrosion in neutral chloride solution. Corros. Sci. 2018, 131, 1–16. [Google Scholar] [CrossRef]
- Gonzalez-Rodriguez, J.G.; Porcayo-Calderon, I.; Vazquez-Velez, E.; de la Escalera, L.M.M.; Canto, I.; Martinez, L. Use of a palm oil-based imidazoline as corrsion inhibitor for copper in 3.5% NaCl solution. Int. J. Electrochem. Sci. 2016, 11, 8132–8144. [Google Scholar] [CrossRef]
- Shi, J.M.; He, C.L.; Li, G.P.; Chen, H.Z.; Fu, X.Y.; Li, R.; Ma, G.F.; Wang, J.M. Corrosion behaviorsof pure copper and Cu-Ni-Zn alloy in NaCl solution and artificialsaltwater. In Proceedings of the 2nd International Conference on New Material and Chemical Industry, Sanya, China, 18–20 November 2017; Xin, S., Ed.; IOP Publishing: Bristol, UK, 2018; Volume 292. [Google Scholar]
- Velazquez-Torres, N.; Martinez, H.; Porcayo-Calderon, I.; Vazquez-Velez, E.; Florez, O.; Campillo, B.; Gonzalez-Rodriguez, J.G.; Martinez-Gomez, L. Effect of plasma pre-oxidation on the Cu corrosion inhibition in 3.5% NaCl by an environmentally friendly amide. Int. J. Electrochem. Sci. 2018, 13, 8915–8930. [Google Scholar] [CrossRef]
- Wang, D.; Xiang, B.; Liang, Y.; Song, S.; Liu, C. Corrosion control of copper in 3.5 wt.% NaCl Solution by domperidone: Experimental and theoretical study. Corros. Sci. 2014, 85, 77–86. [Google Scholar] [CrossRef]
- Li, S.; Ma, Y.; Liu, Y.; Xin, G.; Wang, M.; Zhang, Z.; Liu, Z. Electrochemical sensor based on a three dimensional nanostructured MoS2 nanosphere-PANI/reduced graphene oxide composite for simultaneous detection of ascorbic acid, dopamine, and uric acid. RSC Adv. 2019, 9, 2997–3003. [Google Scholar] [CrossRef]
- Sherif, E.M.; Park, S.-M. Inhibition of copper corrosion in acidic pickling solutions by N-phenyl-1,4-phenylenediamine. Electrochim. Acta 2006, 51, 4665–4673. [Google Scholar] [CrossRef]
- Kim, H.; Lee, H.; Lim, H.-R.; Cho, H.-B.; Choa, Y.-H. Electrically conductive and anti-corrosive coating on copper foil assisted by polymer-nanocomposites embedded with graphene. Appl. Surf. Sci. 2019, 476, 123–127. [Google Scholar] [CrossRef]
- Ramezanzadeh, B.; Bahlakeh, G.; Moghadam, M.M.; Miraftab, R. Impact of size-controlled p-phenylenediamine (PPDA)-functionalized graphene oxide nanosheets on the GO-PPDA/Epoxy anti-corrosion, interfacial interactions and mechanical properties enhancement: Experimental and quantum mechanics investigations. Chem. Eng. J. 2018, 335, 737–755. [Google Scholar] [CrossRef]
- Kumar, N.; Manik, G. Molecular dynamics simulations of polyvinyl acetate-perfluorooctane based anti-stain coatings. Polymer 2016, 100, 194–205. [Google Scholar] [CrossRef]
- Hong, M.-S.; Park, Y.; Kim, J.G.; Kim, K. Effect of incorporating MoS2 in organic coatings on the corrosion resistance of 316L stainless steel in a 3.5% NaCl solution. Coatings 2019, 9, 45. [Google Scholar] [CrossRef]
- Kiran, N.U.; Dey, S.; Singh, B.P.; Besra, L. Graphene coating on copper by electrophoretic deposition for corrosion prevention. Coatings 2017, 7, 241. [Google Scholar] [CrossRef]

| Material | Manufacturer | Label |
|---|---|---|
| Polyvinylbutyral | MACKLIN | P815775 |
| Tin sulfide | 6Carbon Tech. Shenzhen | SC-CRYSTAL-SNS |
| Molybdenum disulfide | HANLANE | MoS2-50 |
| Zinc | HANLANE | Zn-50 |
| Coating | ω (mg m−2 h−1) | ηw (%) |
|---|---|---|
| Blank | 59.52 | - |
| PVB | 41.67 | 30 |
| nano-SnS | 23.81 | 60 |
| nano-SnS-Zn/PVB | 15.48 | 74 |
| nano-MoS2/PVB | 20.24 | 66 |
| nano-MoS2-Zn/PVB | 14.29 | 76 |
| Coating | Ecorr (mV per SCE) | icorr (μA cm−2) | βc (mV dec−1) | βa (mV dec−1) | Crate (mpy) | |
|---|---|---|---|---|---|---|
| Blank | −245 | 2.185 | 60 | 55 | 1.00 | - |
| PVB | −147 | 1.839 | 179 | 75 | 0.85 | 15.8 |
| nano-SnS/PVB | −97 | 0.921 | 105 | 280 | 0.42 | 57.9 |
| nano-SnS-Zn/PVB | −107 | 0.757 | 99 | 317 | 0.35 | 65.4 |
| nano-MoS2/PVB | −98 | 0.710 | 102 | 297 | 0.33 | 67.5 |
| nano-MoS2-Zn/PVB | −62 | 0.591 | 84 | 190 | 0.27 | 72.9 |
| Coating | Rs (Ω cm2) | Rc (kΩ cm2) | Rct (kΩ cm2) | Cc (μF cm−2) | Cdl (μF cm−2) | W | |
|---|---|---|---|---|---|---|---|
| Blank | 7.94 | 0.04 | 0.32 | 13.08 | 128.30 | 0.002902 | - |
| PVB | 19.50 | 0.07 | 3.50 | 1.84 | 18.17 | 0.000620 | 90.86 |
| nano-SnS/PVB | 18.14 | 0.11 | 50.84 | 2.69 | 54.21 | - | 99.37 |
| nano-SnS-Zn/PVB | 22.54 | 0.30 | 74.36 | 2.49 | 69.49 | - | 99.57 |
| nano-MoS2/PVB | 20.07 | 0.26 | 97.84 | 3.07 | 60.39 | - | 99.67 |
| nano-MoS2-Zn/PVB | 25.09 | 0.30 | 126.20 | 1.92 | 43.34 | - | 99.75 |
| Classification | Samples | |
|---|---|---|
| Silicon carbide composite | POA–SiC/EP | 87.54 |
| Metal Organic Framework | ATT/ZIF-8 | 97.3 |
| Organic inhibitor | polyaspartic acid | 86.8 |
| System | Interaction Energy (kcal mol−1) |
|---|---|
| Cu+PVB | −852.33 |
| Cu+MoS2/PVB | −1838.253 |
| Cu+SnS/PVB | −1074.433 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qu, Z.; Wang, L.; Tang, H.; Ye, H.; Li, M. Effect of Nano-SnS and Nano-MoS2 on the Corrosion Protection Performance of the Polyvinylbutyral and Zinc-Rich Polyvinylbutyral Coatings. Nanomaterials 2019, 9, 956. https://doi.org/10.3390/nano9070956
Qu Z, Wang L, Tang H, Ye H, Li M. Effect of Nano-SnS and Nano-MoS2 on the Corrosion Protection Performance of the Polyvinylbutyral and Zinc-Rich Polyvinylbutyral Coatings. Nanomaterials. 2019; 9(7):956. https://doi.org/10.3390/nano9070956
Chicago/Turabian StyleQu, Zuopeng, Lei Wang, Hongyu Tang, Huaiyu Ye, and Meicheng Li. 2019. "Effect of Nano-SnS and Nano-MoS2 on the Corrosion Protection Performance of the Polyvinylbutyral and Zinc-Rich Polyvinylbutyral Coatings" Nanomaterials 9, no. 7: 956. https://doi.org/10.3390/nano9070956
APA StyleQu, Z., Wang, L., Tang, H., Ye, H., & Li, M. (2019). Effect of Nano-SnS and Nano-MoS2 on the Corrosion Protection Performance of the Polyvinylbutyral and Zinc-Rich Polyvinylbutyral Coatings. Nanomaterials, 9(7), 956. https://doi.org/10.3390/nano9070956






