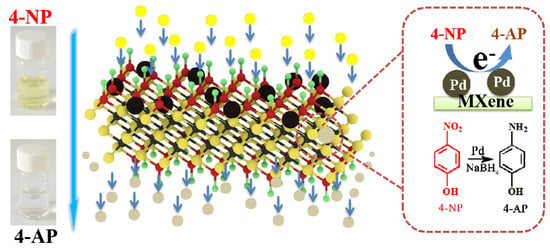
Highly Efficient Catalytic Performances of Nitro Compounds and Morin via Self-Assembled MXene-Pd Nanocomposites Synthesized through Self-Reduction Strategy
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
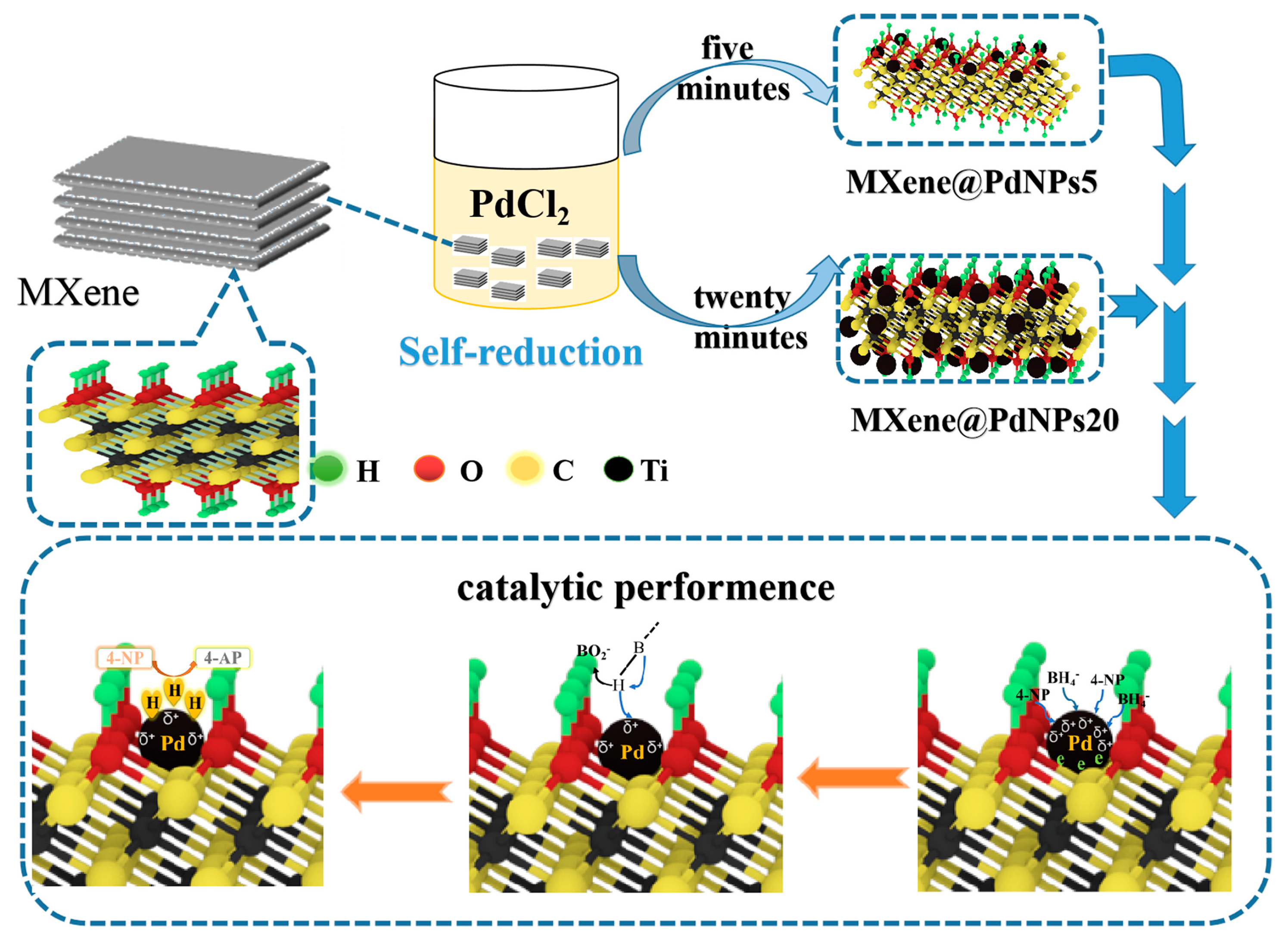
2.2. Preparation of MXene@PdNPs Composite
2.3. Catalytic Performance Test
2.4. Characterization
3. Results and Discussion
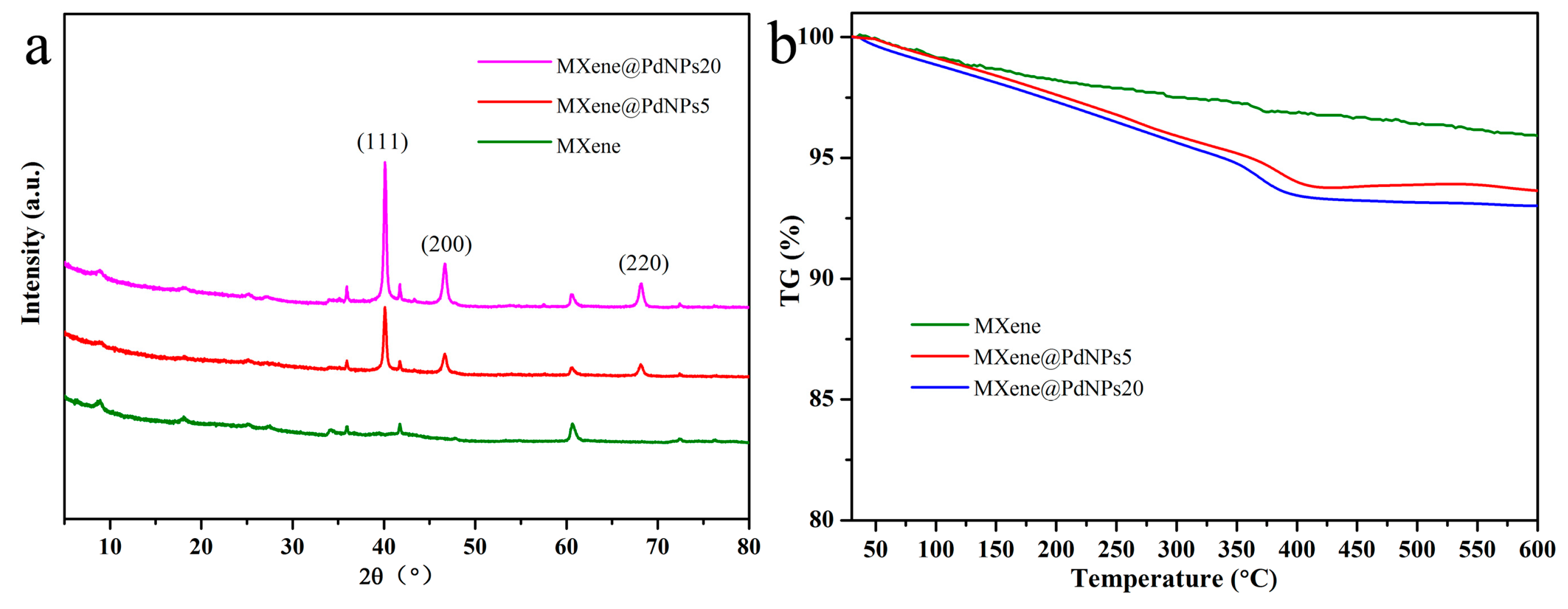
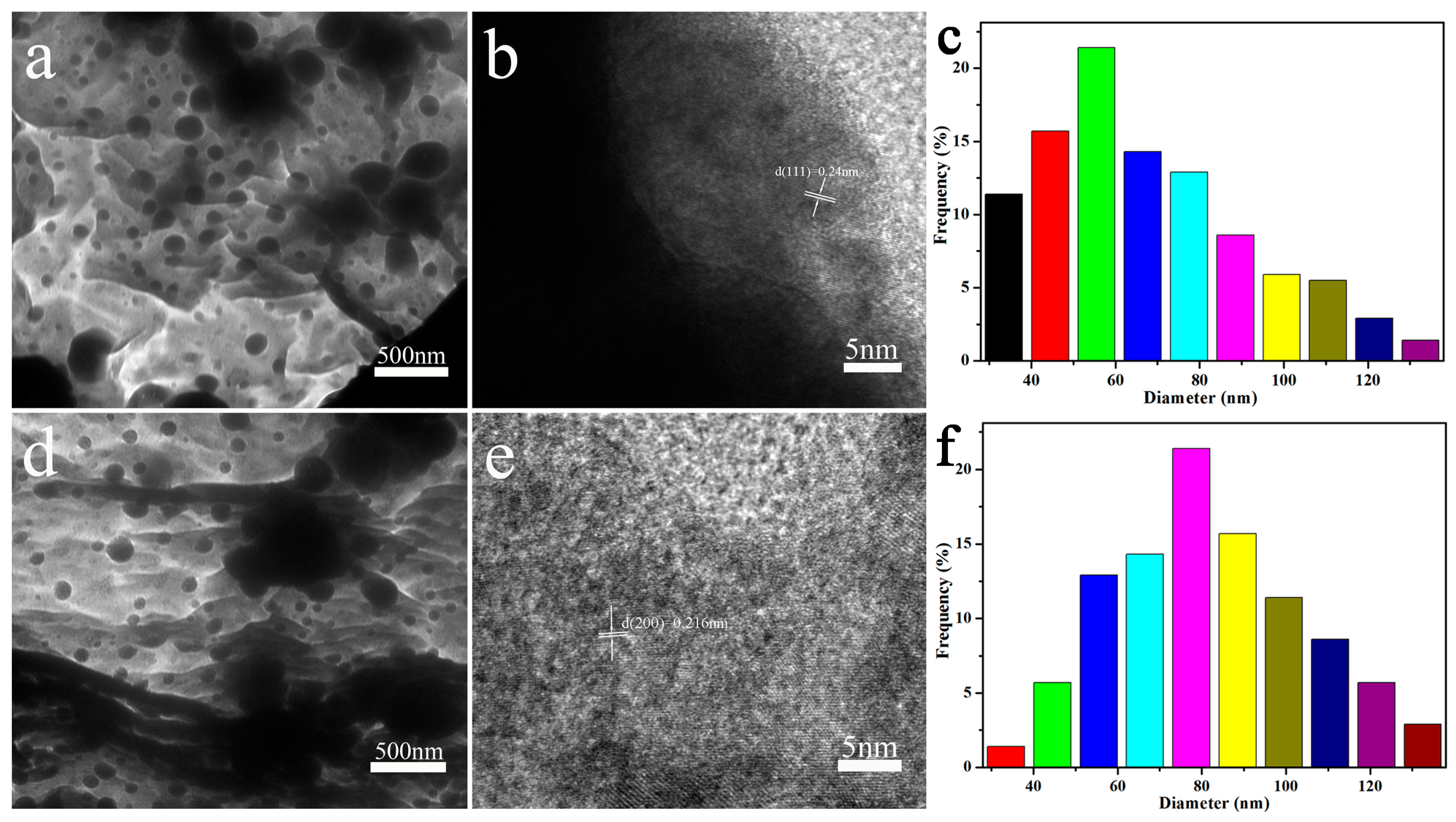
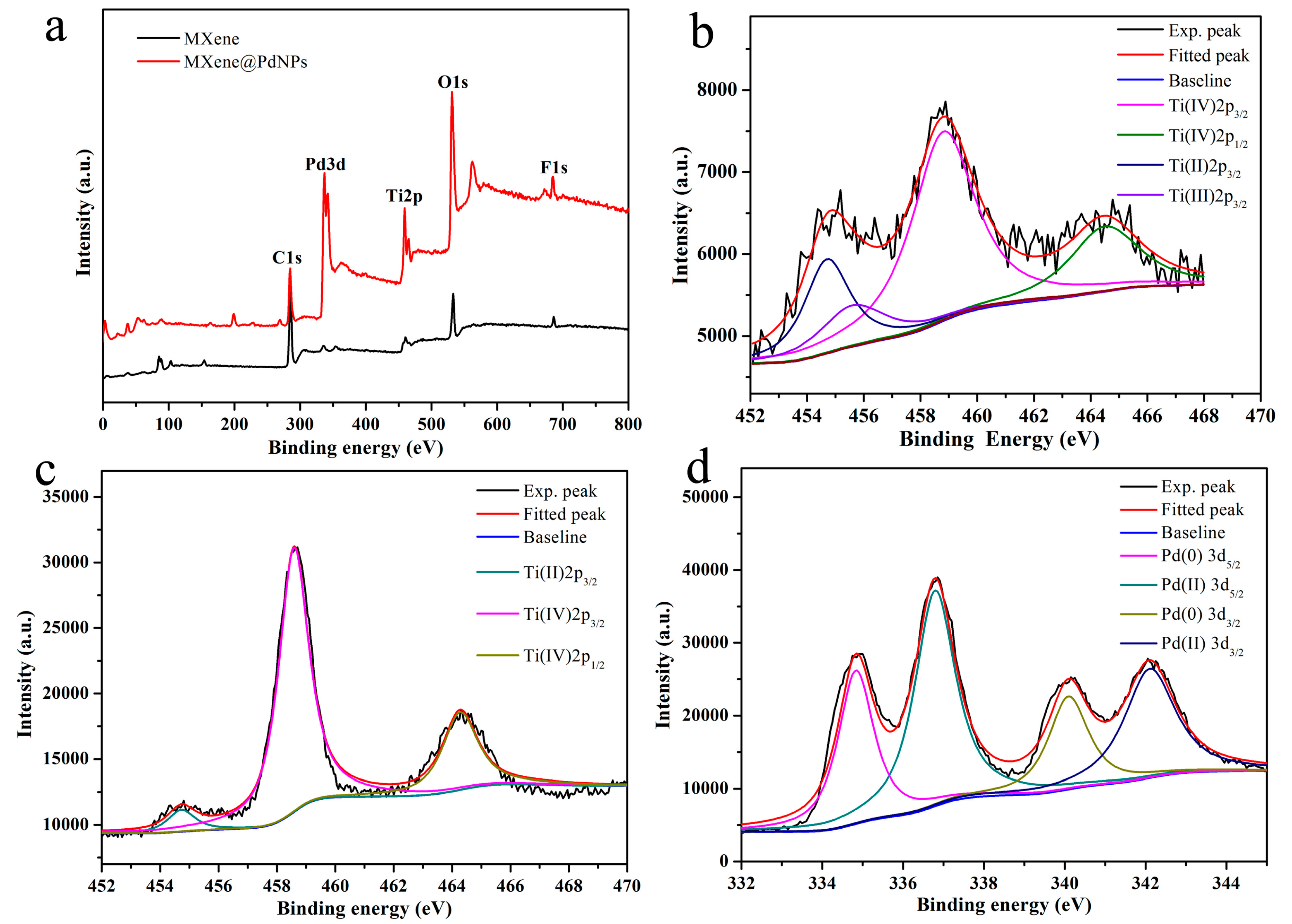
3.1. Structural Characterization of Nanocomposites
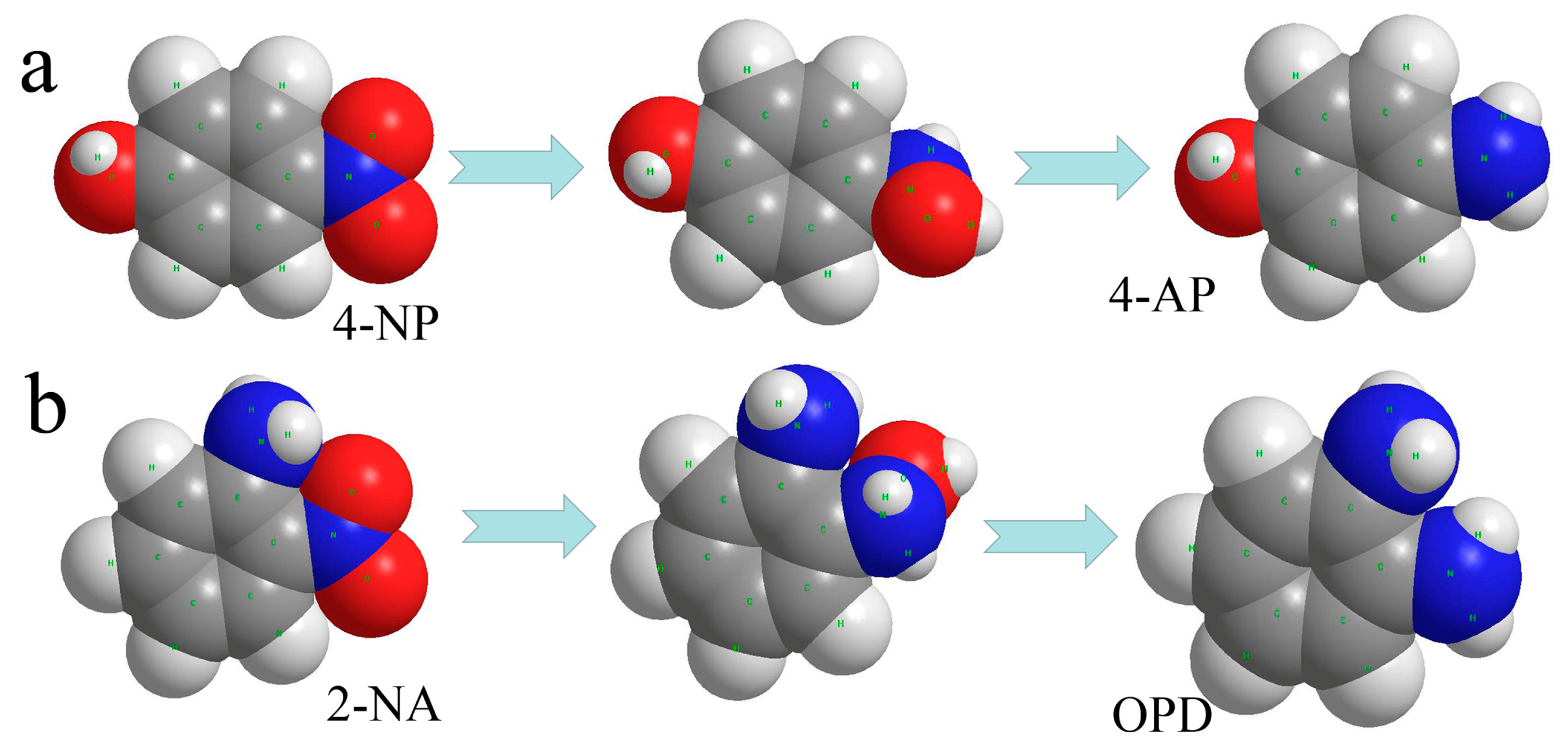
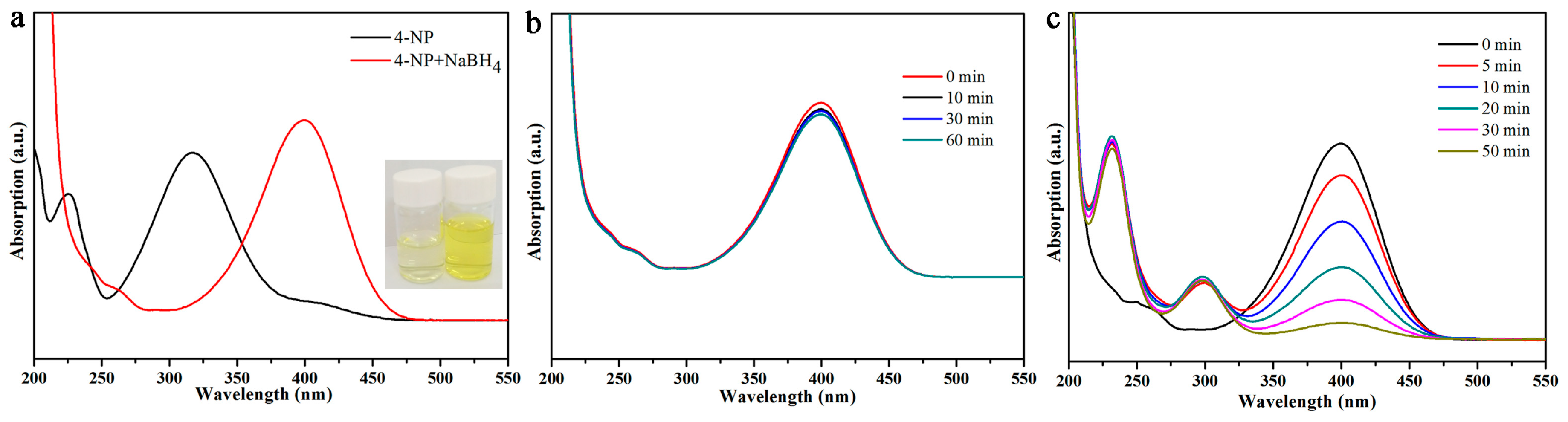
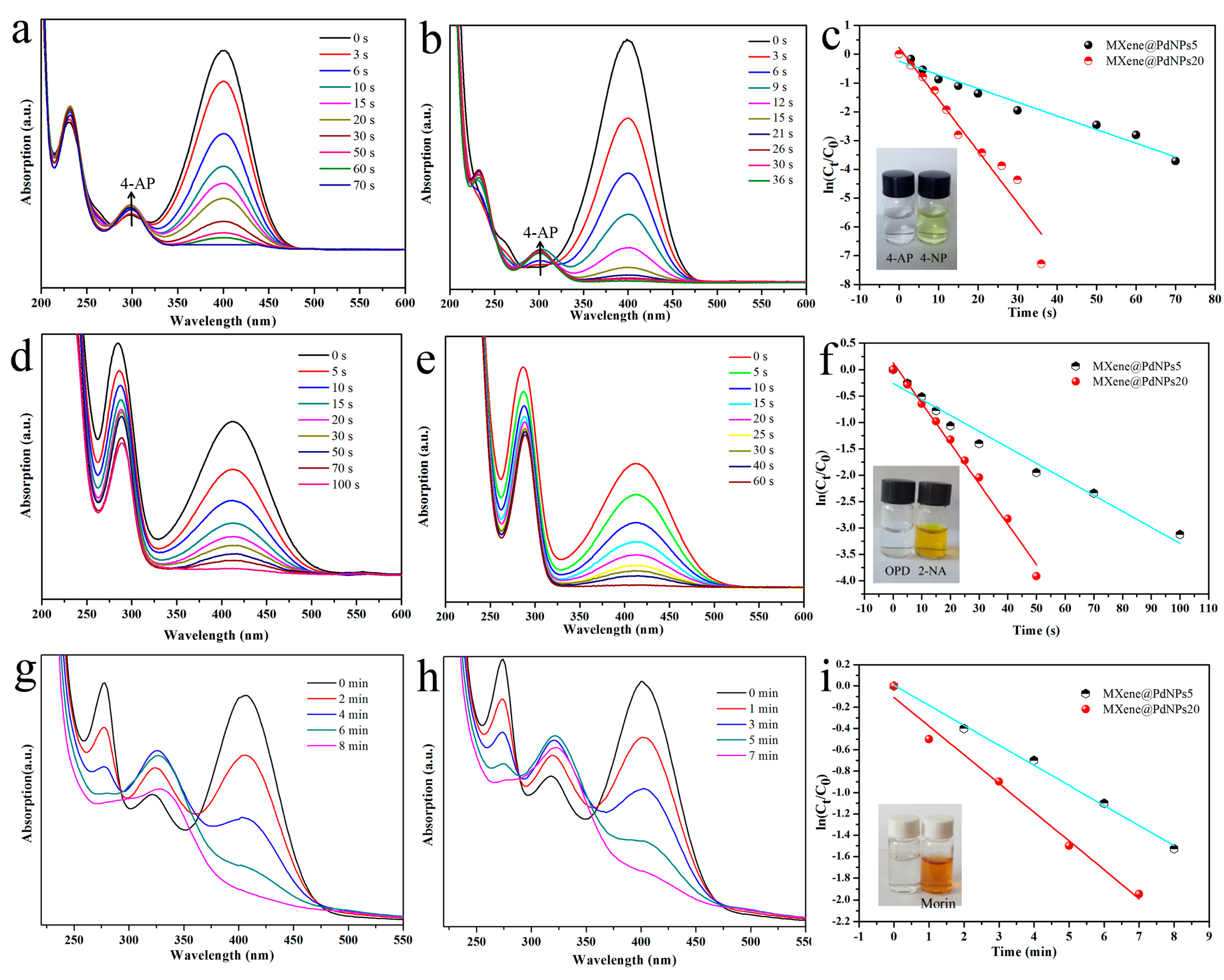
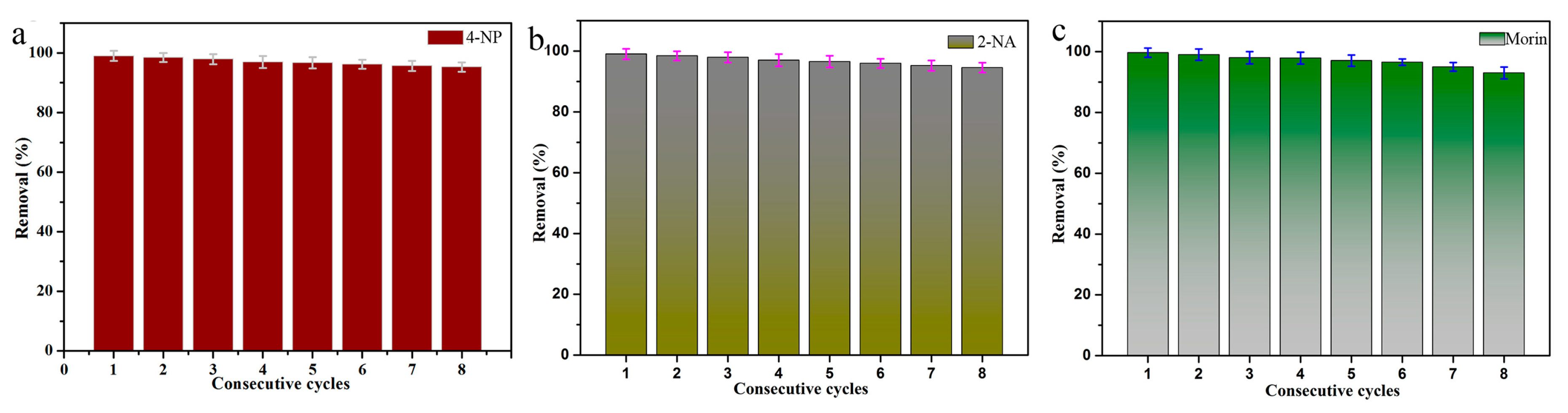
3.2. Dye Catalytic Performance of Nanocomposites
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Zhang, Y.J.; He, P.Y.; Chen, H.; Liu, L.C. Development of an eco-efficient CaMoO4/electroconductive geopolymer composite for recycling silicomanganese slag and degradation of dye wastewater. J. Clean. Prod. 2019, 208, 1476–1487. [Google Scholar]
- Kampouri, S.; Nguyen, T.N.; Spodaryk, M.; Palgrave, R.G.; Züttel, A.; Smit, B.; Stylianou, K.C. Concurrent Photocatalytic Hydrogen Generation and Dye Degradation Using MIL-125-NH2 under Visible Light Irradiation. Adv. Funct. Mater. 2018, 28, 1806368. [Google Scholar] [CrossRef]
- Anasori, B.; Lukatskaya, M.R.; Gogotsi, Y. 2D metal carbides and nitrides (MXenes) for energy storage. Nat. Rev. Mater. 2017, 2, 16098. [Google Scholar] [CrossRef]
- Li, Z.Y.; Wang, L.B.; Sun, D.D.; Zhang, Y.D.; Liu, B.Z.; Hu, Q.K.; Zhou, A.G. Synthesis and thermal stability of two-dimensional carbide MXene Ti3C2. Mater. Sci. Eng. B 2015, 191, 33–40. [Google Scholar] [CrossRef]
- Ghidiu, M.; Lukatskaya, M.R.; Zhao, M.Q.; Gogotsi, Y.; Barsoum, M.W. Conductive two-dimensional titanium carbide ‘clay’ with high volumetric capacitance. Nature 2014, 516, 78–81. [Google Scholar] [CrossRef] [PubMed]
- Alhabeb, M.; Maleski, K.; Mathis, T.S.; Sarycheva, A. Fabrication and thermal stability of two-dimensional carbide Ti3C2 nanosheets. Ceram. Int. 2016, 42, 8419–8424. [Google Scholar]
- Khazaei, M.; Ranjbar, A.; Ghorbani-Asl, M.; Arai, M. Nearly free electron states in MXenes. Phys. Rev. B 2016, 93, 205125–205135. [Google Scholar] [CrossRef]
- Wang, C.; Sun, S.; Zhang, L.; Yin, J.; Jiao, T.; Zhang, L.; Xu, Y.; Zhou, J.; Peng, Q. Facile preparation and catalytic performance characterization of AuNPs-loaded hierarchical electrospun composite fibers by solvent vapor annealing treatment. Colloid Surf. A 2019, 561, 283–291. [Google Scholar] [CrossRef]
- Kurtoglu, M.; Naguib, M.; Gogotsi, Y.; Barsoum, M. First principles study of two-dimensional early transition metal carbides. MRS Commun. 2012, 2, 133–137. [Google Scholar] [CrossRef]
- Zhang, Z.W.; Li, H.N.; Zou, G.D.; Fernández, C.M. Self-reduction synthesis of new MXene/Ag composites with unexpected electrocatalytic activity. ACS Sustain. Chem. Eng. 2016, 4, 6763–6771. [Google Scholar] [CrossRef]
- Luo, S.; Wang, R.; Yin, J.; Jiao, T.; Chen, K.; Zou, G.; Zhang, L.; Zhou, J.; Zhang, L.; Peng, Q. Preparation and dye degradation performances of self-assembled MXene-Co3O4 nanocomposites synthesized via solvothermal approach. ACS Omega 2019, 4, 3946–3953. [Google Scholar] [CrossRef]
- Xu, Y.; Ren, B.; Wang, R.; Zhang, L.; Jiao, T.; Liu, Z. Facile preparation of rod-like MnO nanomixtures via hydrothermal approach and highly efficient removal of methylene blue for wastewater Treatment. Nanomaterials 2019, 9, 10. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.R.; Teng, J.; Zou, G.D.; Peng, Q.M.; Du, Q.; Jiao, T.F.; Xiang, J. Efficient phosphate sequestration for water purification by unique sandwich-like MXene/magnetic iron oxide nanocomposites. Nanoscale 2016, 8, 7085–7093. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Wang, R.; Jiao, T.; Zou, G.; Zhan, F.; Yin, J.; Zhang, L.; Zhou, J.; Peng, Q. Facile preparation of hierarchical AgNPs-loaded MXene/Fe3O4/polymers nanocomposites by electrospinning with enhanced catalytic performance for wastewater treatment. ACS Omega 2019, 4, 1897–1906. [Google Scholar] [CrossRef]
- Wang, C.; Yin, J.; Han, S.; Jiao, T.; Bai, Z.; Zhou, J.; Peng, Q. Preparation of Palladium Nanoparticles Decorated Polyethyleneimine/Polycaprolactone Composite Fibers Constructed by Electrospinning with Highly Efficient and Recyclable Catalytic Performances. Catalysts 2019, 9, 559. [Google Scholar] [CrossRef]
- Zhang, S.; Gai, S.; He, F.; Ding, S.; Li, L.; Yang, P. In situ assembly of well-dispersed ni nanoparticles on silica nanotubes and excellent catalytic activity in 4-nitrophenol reduction. Nanoscale 2014, 6, 11181–11188. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Zhao, G.; Chen, M.; Gao, W.; Zhou, X.; Xie, X.; Du, G. Facile and Green Approach to Prepare Nanostructured Au@MnO2 and Its Applications for Catalysis and Fluorescence Sensing of Glutathione in Human Blood. ACS Sustain. Chem. Eng. 2018, 6, 3948–3956. [Google Scholar] [CrossRef]
- Zou, G.; Zhang, Z.; Guo, J.; Liu, B.; Zhang, Q.; Fernandez, C.; Peng, Q. Synthesis of MXene/Ag composites for extraordinary long cycle lifetime lithium storage at high rates. ACS Appl. Mater. Interfaces 2016, 8, 22280–22286. [Google Scholar] [CrossRef]
- Yazdankhah, M.; Veisi, H.; Hemmati, S. In situ immobilized palladium nanoparticles (Pd NPs) on fritillaria imperialis flower extract-modified graphene and their catalytic activity for reduction of 4-nitrophenol. J. Taiwan Inst. Chem. Eng. 2018, 91, 38–46. [Google Scholar] [CrossRef]
- Guo, Y.; Xu, Y.T.; Gao, G.H.; Wang, T.; Zhao, B.; Fu, X.Z.; Sun, R.; Wong, C.P. Electro-oxidation of formaldehyde and methanol over hollow porous palladium nanoparticles with enhanced catalytic activity. Catal. Commun. 2015, 58, 40–45. [Google Scholar] [CrossRef]
- Li, C.L.; Su, Y.; Shi, H.J.; Yang, X.G.; Wang, Y.J. Enhanced ethanol electrooxidation of hollow Pd nanospheres prepared by galvanic exchange reactions. Mater. Lett. 2012, 69, 92–95. [Google Scholar] [CrossRef]
- Zhang, R.Y.; Xing, R.R.; Jiao, T.F.; Ma, K.; Chen, C.J.; Ma, G.H.; Yan, X.H. Carrier-free, chemophotodynamic dual nanodrugs via self-assembly for synergistic antitumor therapy. ACS Appl. Mater. Interfances 2016, 8, 13262–13269. [Google Scholar] [CrossRef]
- Filip, J.; Zavahir, S.; Lorencova, L.; Bertok, T.; Yousaf, A.B.; Mahmoud, K.A.; Kasak, P. Tailoring Electrocatalytic Properties of Pt Nanoparticles Grown on Ti3C2TX MXene Surface. J. Electrochem. Soc. 2019, 166, H54–H62. [Google Scholar] [CrossRef]
- Huo, S.W.; Duan, P.F.; Jiao, T.F.; Peng, Q.M.; Liu, M.H. Self-assembled luminescent quantum dots to generate full-color and white circularly polarized light. Angew. Chem. Int. Ed. 2017, 56, 12174–12178. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.N.; Ma, K.; Jiao, T.F.; Xing, R.R.; Ma, X.L.; Hu, J.; Huang, H.; Zhang, L.X.; Yan, X.H. Fabrication of hierarchical layer-by-layer assembled diamond-based core-shell nanocomposites as highly efficient dye absorbents for wastewater treatment. Sci. Rep. 2017, 7, 44076. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.M.; Ma, K.; Jiao, T.F.; Xing, R.R.; Shen, G.Z.; Yan, X.H. Water-insoluble photosensitizer nanocolloids stabilized by supramolecular interfacial assembly towards photodynamic therapy. Sci. Rep. 2017, 7, 42978. [Google Scholar] [CrossRef]
- Feng, Y.; Jiao, T.; Yin, J.; Zhang, L.; Zhang, L.X.; Zhou, J.; Peng, Q. Facile preparation of carbon nanotube-Cu2O nanocomposites as new catalyst materials for reduction of p-nitrophenol. Nanoscale Res. Lett. 2019, 14, 78. [Google Scholar] [CrossRef]
- Guo, R.; Jiao, T.F.; Li, R.L.; Chen, Y. Sandwiched Fe3O4/carboxylate graphene oxide nanostructures constructed by layer-by-layer assembly for highly efficient and magnetically recyclable dye removal. ACS Sustain. Chem. Eng. 2018, 6, 1279–1288. [Google Scholar] [CrossRef]
- Ji, Y.; Ghosh, K.; Shu, X.Z.; Li, B.Q.; Sokolov, J.C.; Prestwich, G.D.; Clark, R.A.F.; Rafailovich, M.H. Electrospun three-dimensional hyaluronic acid nanofibrous scaffolds. Biomaterials 2006, 27, 3782–3792. [Google Scholar] [CrossRef]
- Li, K.; Zou, G.; Jiao, T.; Xing, R.; Zhang, L.; Zhou, J.; Zhang, Q.; Peng, Q. Self-assembled MXene-based nanocomposites via layer-by-layer strategy for elevated adsorption capacities. Colloid Surf. A 2018, 553, 105–113. [Google Scholar] [CrossRef]
- Guo, H.Y.; Jiao, T.F.; Zhang, Q.R.; Guo, W.F.; Peng, Q.M.; Yan, X.H. Preparation of graphene oxidebased hydrogels as efficient dye adsorbents for wastewater treatment. Nanoscale Res. Lett. 2015, 10, 272. [Google Scholar] [CrossRef] [PubMed]
- Luo, X.N.; Ma, K.; Jiao, T.F.; Xing, R.R.; Zhang, L.X.; Zhou, J.X.; Li, B.B. Graphene oxide-polymer composite Langmuir films constructed by interfacial thiol-ene photopolymerization. Nanoscale Res. Lett. 2017, 12, 99. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.S.; Liu, H.; Lou, X.W.; Qiao, S.Z. Carbon-supported ultra-thin anatase TiO2 nanosheets for fast reversible lithium storage. J. Mater. Chem. 2011, 21, 5687–5692. [Google Scholar] [CrossRef]
- Hou, C.L.; Jiao, T.F.; Xing, R.R.; Chen, Y.; Zhou, J.X.; Zhang, L.X. Preparation of TiO2 nanoparticles modified electrospun nanocomposite membranes toward efficient dye degradation for wastewater treatment. J. Taiwan Inst. Chem. Eng. 2017, 78, 118–126. [Google Scholar] [CrossRef]
- Hu, H.W.; Xin, J.H.; Hu, H.; Wang, X.W.; Miao, D.G.; Liu, Y. Synthesis and stabilization of metal nanocatalysts for reduction reactions—A review. J. Mater. Chem. A 2015, 3, 11157–11182. [Google Scholar] [CrossRef]
- Pradhan, N.; Pal, A.; Pal, T. Catalytic reduction of aromatic nitro compounds by coinage metal nanoparticles. Langmuir 2001, 17, 1800–1802. [Google Scholar] [CrossRef]
- Ma, N.; Liu, X.h.; Yang, Z.Q.; Tai, G.J.; Yin, Y.R.; Liu, S.B.; Li, H.L.; Guo, P.Z.; Zhao, X.S. Carrageenan assisted synthesis of palladium nanoflowers and their electrocatalytic activity toward ethanol. ACS Sustain. Chem. Eng. 2017, 6, 1133–1140. [Google Scholar] [CrossRef]
- Li, K.K.; Jiao, T.F.; Xing, R.R.; Zou, G.D.; Zhou, J.X.; Zhang, L.X.; Peng, Q.M. Fabrication of tunable hierarchical MXene@AuNPs nanocomposites constructed by self-reduction reactions with enhanced catalytic performances. Sci. China Mater. 2018, 61, 728–736. [Google Scholar] [CrossRef]
- Xue, J.; Han, G.T.; Ye, W.N.; Sang, Y.T. Structural regulation of PdCu2 nanoparticles and their electrocatalytic performance for ethanol oxidation. ACS Appl. Mater. Interfances 2016, 8, 34497–34505. [Google Scholar] [CrossRef]
- Le, X.; Dong, Z.; Li, X.; Zhang, W.; Le, M.; Ma, J. Fibrous nano-silica supported palladium nanoparticles: An efficient catalyst for the reduction of 4-nitrophenol and hydrodechlorination of 4-chlorophenol under mild conditions. Catal. Commun. 2015, 59, 21–25. [Google Scholar] [CrossRef]
- Sang, Y.T.; Cui, Y.Q.; Li, Z.; Ye, W.N.; Li, H.L.; Zhao, X.S.; Guo, P.Z. Electrochemical reaction of nitrobenzene and its derivatives on glassy carbon electrode modified with MnFe2O4 colloid nanocrystal assemblies. Sens. Actuators B 2016, 234, 46–52. [Google Scholar] [CrossRef]
- Zhan, F.; Wang, R.; Yin, J.; Han, Z.; Zhang, L.; Jiao, T.; Zhou, J.; Zhang, L.; Peng, Q. Facile solvothermal preparation of Fe3O4-Ag nanocomposite with excellent catalytic performance. RSC Adv. 2019, 9, 878–883. [Google Scholar] [CrossRef]
- Lebaschi, S.; Hekmati, M.; Veisi, H. Green synthesis of palladium nanoparticles mediated by black tea leaves (Camellia sinensis) extract: Catalytic activity in the reduction of 4-nitrophenol and Suzuki-Miyaura coupling reaction under ligand-free conditions. J. Colloid Interf. Sci. 2017, 485, 223–231. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Lu, C.; Zhang, W.; Yuan, G.; Xiong, R.; Zhang, X. A novel reagentless approach for synthesizing cellulose nanocrystal-supported palladium nanoparticles with enhanced catalytic performance. J. Mater. Chem. A 2013, 1, 8645–8652. [Google Scholar] [CrossRef]
- Chen, X.; Cai, Z.; Chen, X.; Oyama, M. AuPd bimetallic nanoparticles decorated on graphene nanosheets: Their green synthesis, growth mechanism and high catalytic ability in 4-nitrophenol reduction. J. Mater. Chem. A 2014, 2, 5668–5674. [Google Scholar] [CrossRef]
- Dong, Z.P.; Le, X.D.; Liu, Y.S.; Dong, C.X.; Ma, J.T. Metal organic framework derived magnetic porous carbon composite supported gold and palladium nanoparticles as highly efficient and recyclable catalysts for reduction of 4-nitrophenol and hydrodechlorination of 4-chlorophenol. J. Mater. Chem. A 2014, 2, 18775–18785. [Google Scholar] [CrossRef]
- Jiao, T.F.; Zhao, H.; Zhou, J.X.; Zhang, Q.R.; Luo, X.N.; Hu, J.; Peng, Q.M.; Yan, X.H. Self-Assembly Reduced Graphene Oxide Nanosheet Hydrogel Fabrication by Anchorage of Chitosan/Silver and Its Potential Efficient Application toward Dyes Degradation for Wastewater Treatments. ACS Sustain. Chem. Eng. 2015, 3, 3130–3139. [Google Scholar] [CrossRef]
- He, Y.; Wang, R.; Jiao, T.F.; Yan, X.Y.; Wang, M.L.; Zhang, L.X.; Bai, Z.H.; Zhang, Q.; Peng, Q.M. Facile Preparation of Self-Assembled Layered Double Hydroxide-Based Composite Dye Films as New Chemical Gas Sensors. ACS Sustain. Chem. Eng. 2019, 7, 10888–10899. [Google Scholar] [CrossRef]
- Jiao, T.; Guo, H.; Zhang, Q.; Peng, Q.; Tang, Y.; Yan, X.; Li, B. Reduced Graphene Oxide-Based Silver Nanoparticle-Containing Composite Hydrogel as Highly Efficient Dye Catalysts for Wastewater Treatment. Sci. Rep. 2015, 5, 11873. [Google Scholar] [CrossRef]
- Chen, K.Y.; Jiao, T.F.; Li, J.K.; Han, D.X.; Wang, R.; Tian, G.J.; Peng, Q.M. Chiral Nanostructured Composite Films via Solvent-Tuned Self-Assembly and Their Enantioselective Performances. Langmuir 2019, 35, 3337–3345. [Google Scholar] [CrossRef]
- Chen, K.Y.; Yan, X.Y.; Li, J.K.; Jiao, T.F.; Cai, C.; Zou, G.D.; Wang, R.; Wang, M.L.; Zhang, L.X.; Peng, Q.M. Preparation of Self-Assembled Composite Films Constructed by Chemically-Modified MXene and Dyes with Surface-enhanced Raman Scattering Characterization. Nanomaterials 2019, 9, 284. [Google Scholar] [CrossRef]
- Sun, S.X.; Wang, C.R.; Han, S.Q.; Jiao, T.F.; Wang, R.; Yin, J.J.; Li, Q.; Wang, Y.Q.; Geng, L.J.; Yu, X.D.; et al. Interfacial nanostructures and acidichromism behaviors in self-assembled terpyridine derivatives Langmuir-Blodgett films. Colloid Surf. A 2019, 564, 1–9. [Google Scholar] [CrossRef]
- Huang, X.X.; Jiao, T.F.; Liu, Q.Q.; Zhang, L.X.; Zhou, J.X.; Li, B.B.; Peng, Q.M. Hierarchical electrospun nanofibers treated by solvent vapor annealing as air filtration mat for high-efficiency PM2.5 capture. Sci. China Mater. 2019, 62, 423–436. [Google Scholar] [CrossRef]
- Wang, C.R.; Yin, J.J.; Wang, R.; Jiao, T.F.; Huang, H.M.; Zhou, J.X.; Zhang, L.X.; Peng, Q.M. Facile preparation of self-assembled polydopamine-modified electrospun fibers for highly effective removal of organic dyes. Nanomaterials 2019, 9, 116. [Google Scholar] [CrossRef]
- Guo, R.; Wang, R.; Yin, J.J.; Jiao, T.F.; Huang, H.M.; Zhao, X.M.; Zhang, L.X.; Li, Q.; Zhou, J.X.; Peng, Q.M. Fabrication and highly efficient dye removal characterization of beta-cyclodextrin-based composite polymer fibers by electrospinning. Nanomaterials 2019, 9, 127. [Google Scholar] [CrossRef]
- Ma, K.; Chen, W.; Jiao, T.; Jin, X.; Sang, Y.; Yang, D.; Zhou, J.; Liu, M.; Duan, P. Boosting Circularly Polarized Luminescence of Small Organic Molecules via Multi-dimensional Morphology Control. Chem. Sci. 2019. [Google Scholar] [CrossRef]
- Xing, R.; Liu, K.; Jiao, T.; Zhang, N.; Ma, K.; Zhang, R.; Zou, Q.; Ma, G.; Yan, X. An injectable self-assembling collagen-gold hybrid hydrogel for combinatorial antitumor photothermal/photodynamic therapy. Adv. Mater. 2016, 28, 3669–3676. [Google Scholar] [CrossRef]
- Xing, R.; Wang, W.; Jiao, T.; Ma, K.; Zhang, Q.; Hong, W.; Qiu, H.; Zhou, J.; Zhang, L.; Peng, Q. Bioinspired polydopamine sheathed nanofibers containing carboxylate graphene oxide nanosheet for high-efficient dyes scavenger. ACS Sustain. Chem. Eng. 2017, 5, 4948–4956. [Google Scholar] [CrossRef]
- Liu, Y.; Hou, C.; Jiao, T.; Song, J.; Zhang, X.; Xing, R.; Zhou, J.; Zhang, L.; Peng, Q. Self-assembled AgNP-containing nanocomposites constructed by electrospinning as efficient dye photocatalyst materials for wastewater treatment. Nanomaterials 2018, 8, 35. [Google Scholar] [CrossRef]
- Yin, Y.R.; Ma, N.; Xue, J.; Wang, G.Q.; Liu, S.B.; Li, H.L.; Guo, P.Z. Insights into the Role of Poly(vinylpyrrolidone) in the Synthesis of Palladium Nanoparticles and Their Electrocatalytic Properties. Langmuir 2019, 35, 787–795. [Google Scholar] [CrossRef]
- Liu, K.; Yuan, C.Q.; Zou, Q.L.; Xie, Z.C.; Yan, X.H. Self-Assembled Zinc/Cystine-Based Chloroplast Mimics Capable of Photoenzymatic Reactions for Sustainable Fuel Synthesis. Angew. Chem. Int. Ed. 2017, 56, 7876–7880. [Google Scholar] [CrossRef]
- Liu, K.; Xing, R.R.; Li, Y.X.; Zou, Q.L.; Möhwald, H.; Yan, X.H. Mimicking primitive photobacteria: Sustainable hydrogen evolution based on peptide-porphyrin co-assemblies with self-mineralized reaction center. Angew. Chem. Int. Ed. 2016, 55, 12503–12507. [Google Scholar] [CrossRef]
- Liu, K.; Xing, R.R.; Chen, C.J.; Shen, G.Z.; Yan, L.Y.; Zou, Q.L.; Ma, G.H.; Möhwald, H.; Yan, X.H. Peptide-induced hierarchical long-range order and photocatalytic activity of porphyrin assemblies. Angew. Chem. Int. Ed. 2015, 54, 500–505. [Google Scholar] [CrossRef]
- Chen, H.; Zhang, S.; Zhao, Z.; Liu, M.; Zhang, Q. Application of Dopamine Functional Materials in Water Pollution Control. Prog. Chem. 2019, 31, 571–579. [Google Scholar]
- Yan, X.Y.; Wang, M.L.; Sun, X.; Wang, Y.H.; Shi, G.H.; Ma, W.L.; Hou, P. Sandwich-like Ag@Cu@CW SERS substrate with tunable nanogaps and component based on the Plasmonic nanonodule structures for sensitive detection crystal violet and 4-aminothiophenol. Appl. Surf. Sci. 2019, 479, 879–886. [Google Scholar] [CrossRef]

| No. | Materials | Kinetic Constant k/s−1 | Characteristics | Refs |
|---|---|---|---|---|
| 1 | Pd@B.tea NPs | 0.00059 | Natural bioactive black tea extract reducing agent | [43] |
| 2 | CN-supported PdNPs nanohybrids | 0.00570 | Cellulose nanocrystals as support matrix and reducing agent | [44] |
| 3 | AuPdNPs/graphene nanosheets (GNs) | 0.01445 | Bimetallic nanoparticles, monodisperse method, graphene nanosheet carrier | [45] |
| 4 | Pd/magnetic porous carbon (MPC) | 0.01200 | Porous carbon composite catalyst carrier, magnetic separation | [46] |
| 5 | MXene@PdNPs | 0.1800 | self-reduction, simple method | Present work |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yin, J.; Zhang, L.; Jiao, T.; Zou, G.; Bai, Z.; Chen, Y.; Zhang, Q.; Xia, M.; Peng, Q. Highly Efficient Catalytic Performances of Nitro Compounds and Morin via Self-Assembled MXene-Pd Nanocomposites Synthesized through Self-Reduction Strategy. Nanomaterials 2019, 9, 1009. https://doi.org/10.3390/nano9071009
Yin J, Zhang L, Jiao T, Zou G, Bai Z, Chen Y, Zhang Q, Xia M, Peng Q. Highly Efficient Catalytic Performances of Nitro Compounds and Morin via Self-Assembled MXene-Pd Nanocomposites Synthesized through Self-Reduction Strategy. Nanomaterials. 2019; 9(7):1009. https://doi.org/10.3390/nano9071009
Chicago/Turabian StyleYin, Juanjuan, Lun Zhang, Tifeng Jiao, Guodong Zou, Zhenhua Bai, Yan Chen, Qingrui Zhang, Meirong Xia, and Qiuming Peng. 2019. "Highly Efficient Catalytic Performances of Nitro Compounds and Morin via Self-Assembled MXene-Pd Nanocomposites Synthesized through Self-Reduction Strategy" Nanomaterials 9, no. 7: 1009. https://doi.org/10.3390/nano9071009
APA StyleYin, J., Zhang, L., Jiao, T., Zou, G., Bai, Z., Chen, Y., Zhang, Q., Xia, M., & Peng, Q. (2019). Highly Efficient Catalytic Performances of Nitro Compounds and Morin via Self-Assembled MXene-Pd Nanocomposites Synthesized through Self-Reduction Strategy. Nanomaterials, 9(7), 1009. https://doi.org/10.3390/nano9071009