Reducing the Crystallite Size of Spherulites in PEO-Based Polymer Nanocomposites Mediated by Carbon Nanodots and Ag Nanoparticles
Abstract
1. Introduction
2. Experimental Methodology
2.1. Composite Preparation
2.2. Characterization Techniques
3. Results and Discussion
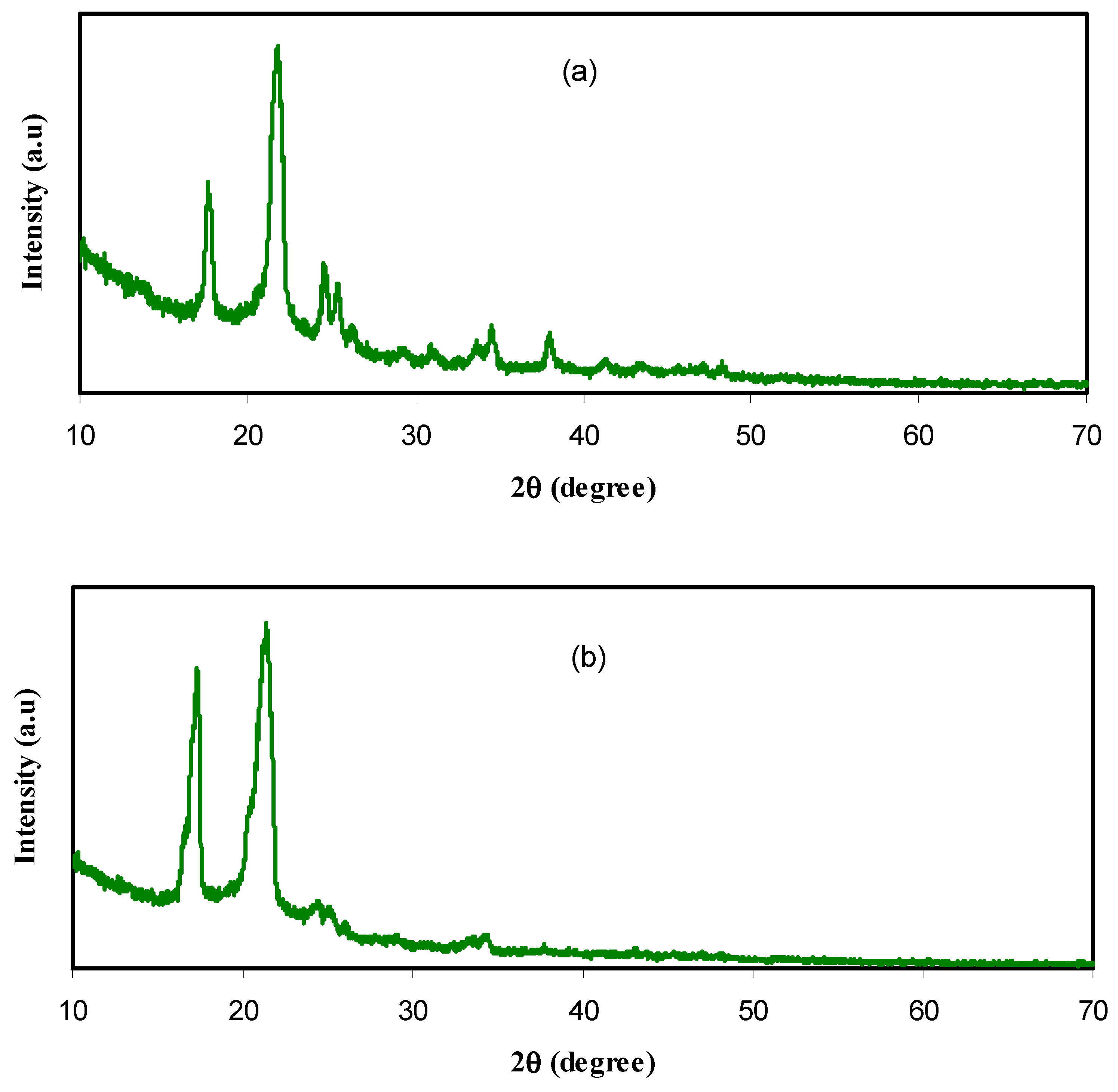
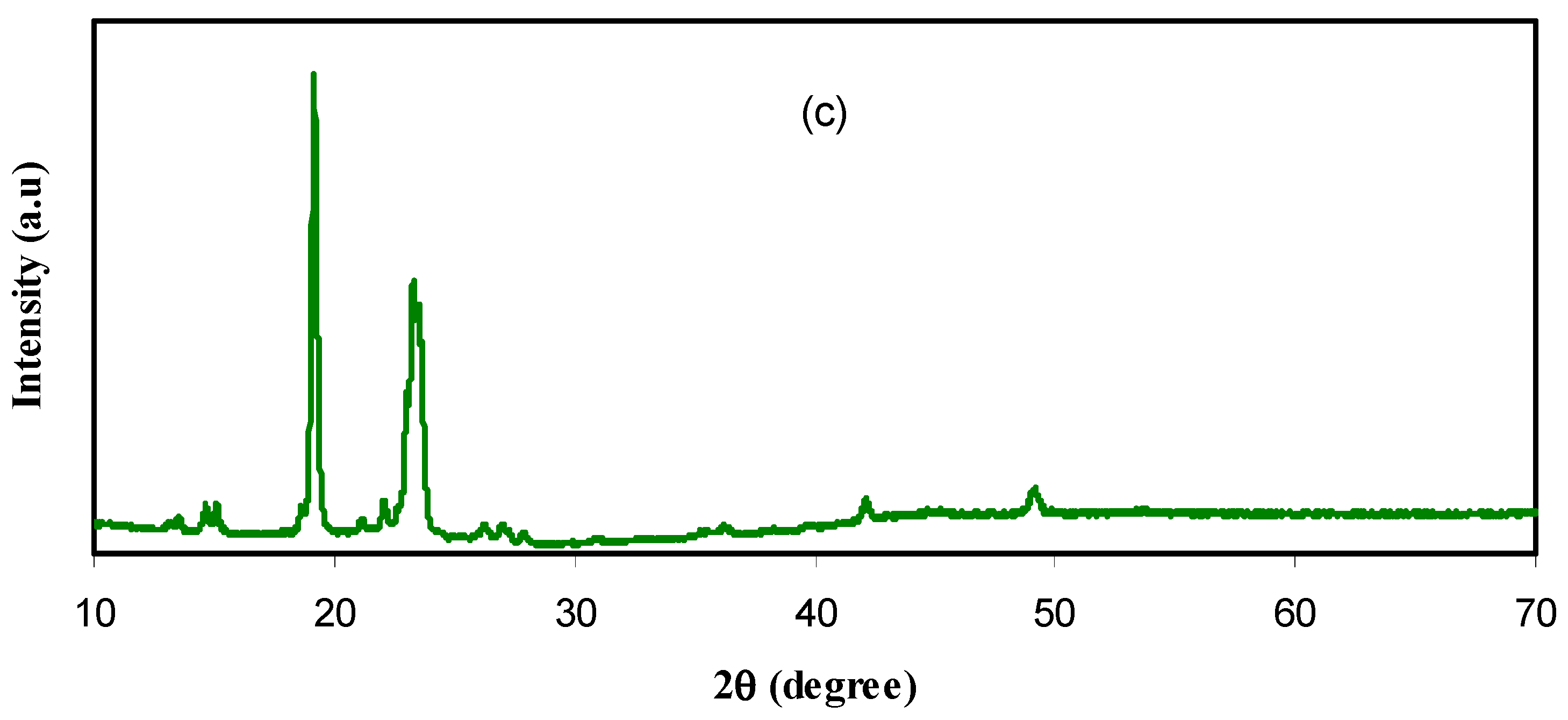
3.1. XRD Study
3.2. Morphology Study
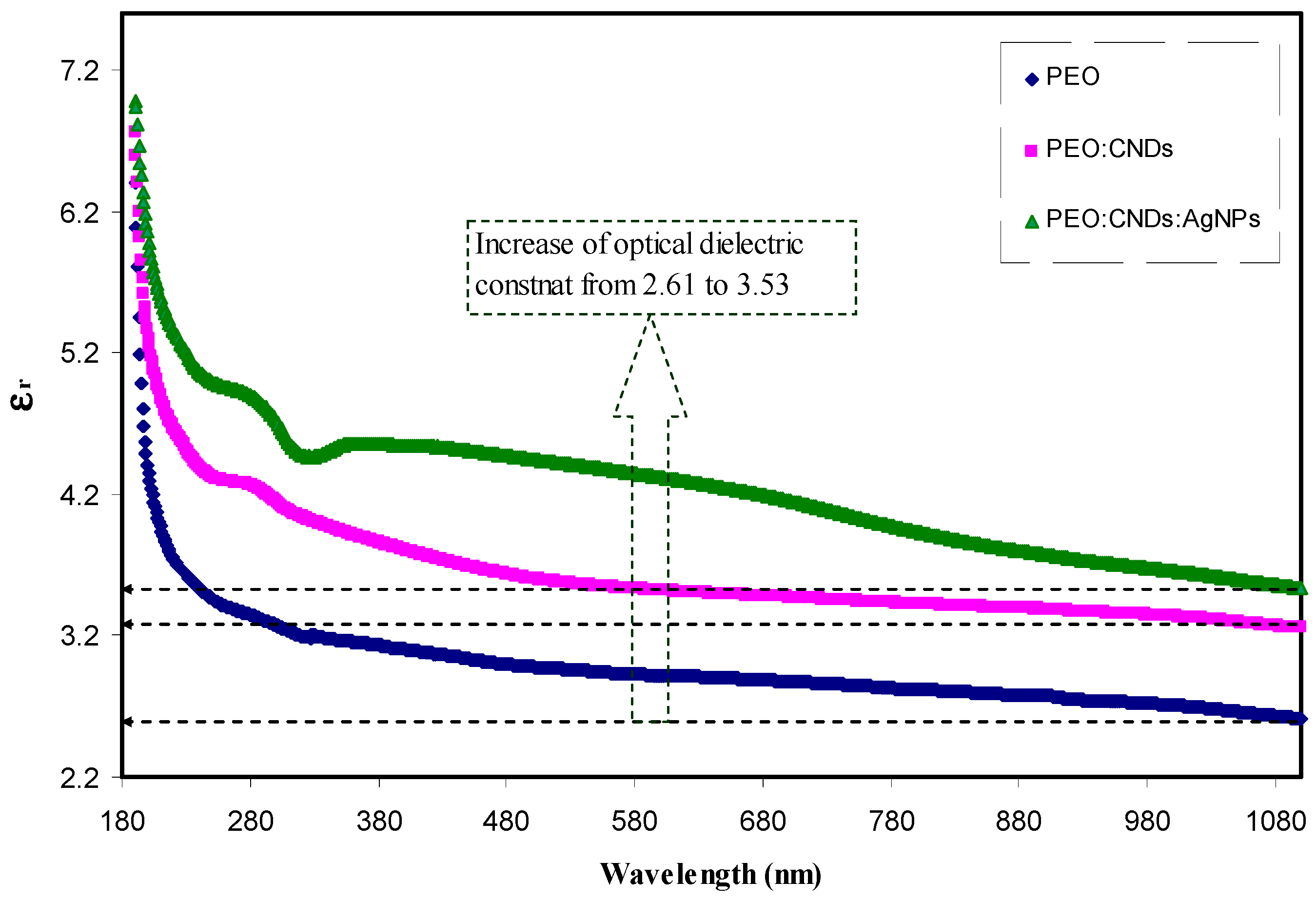
3.3. Optical Properties
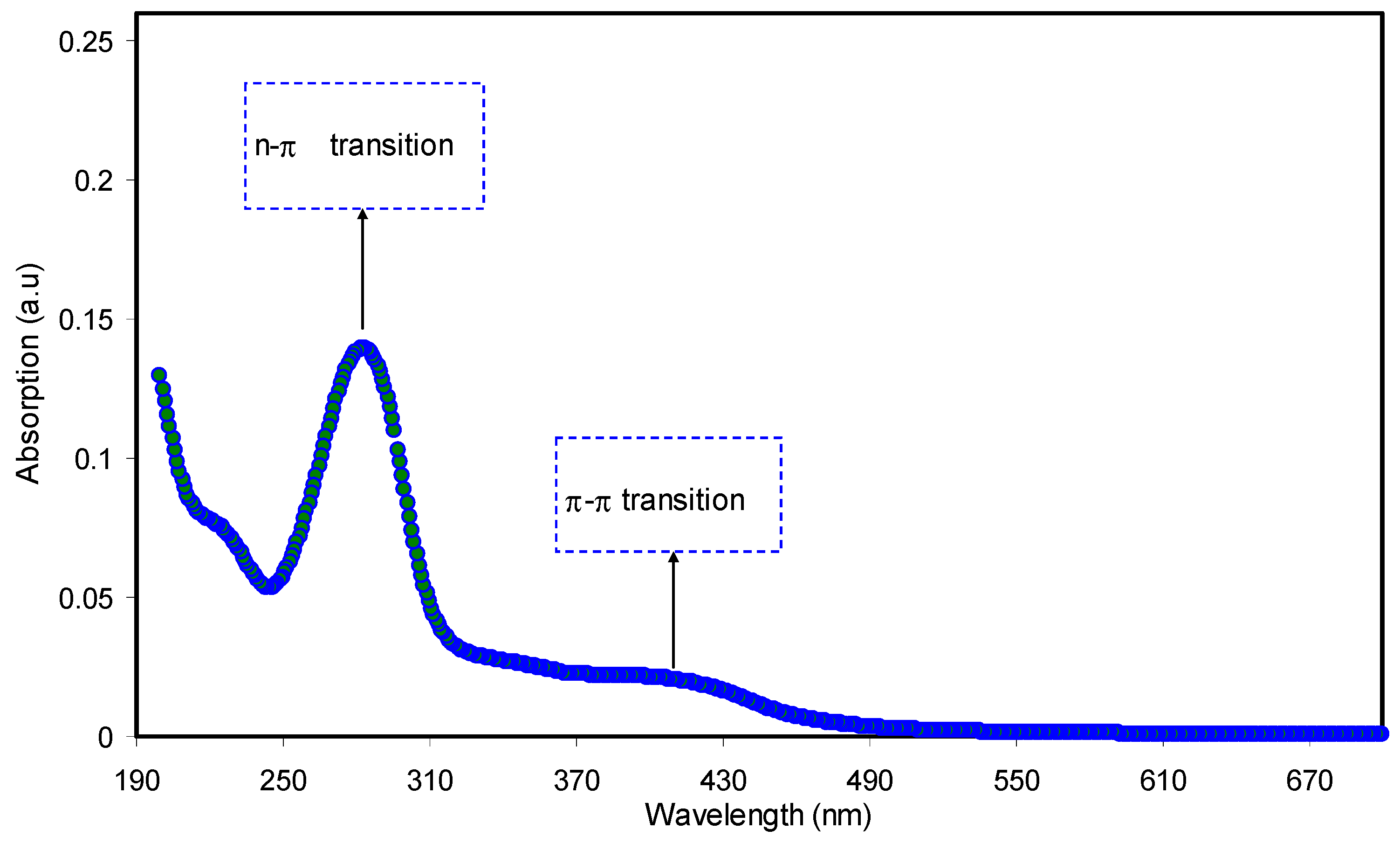
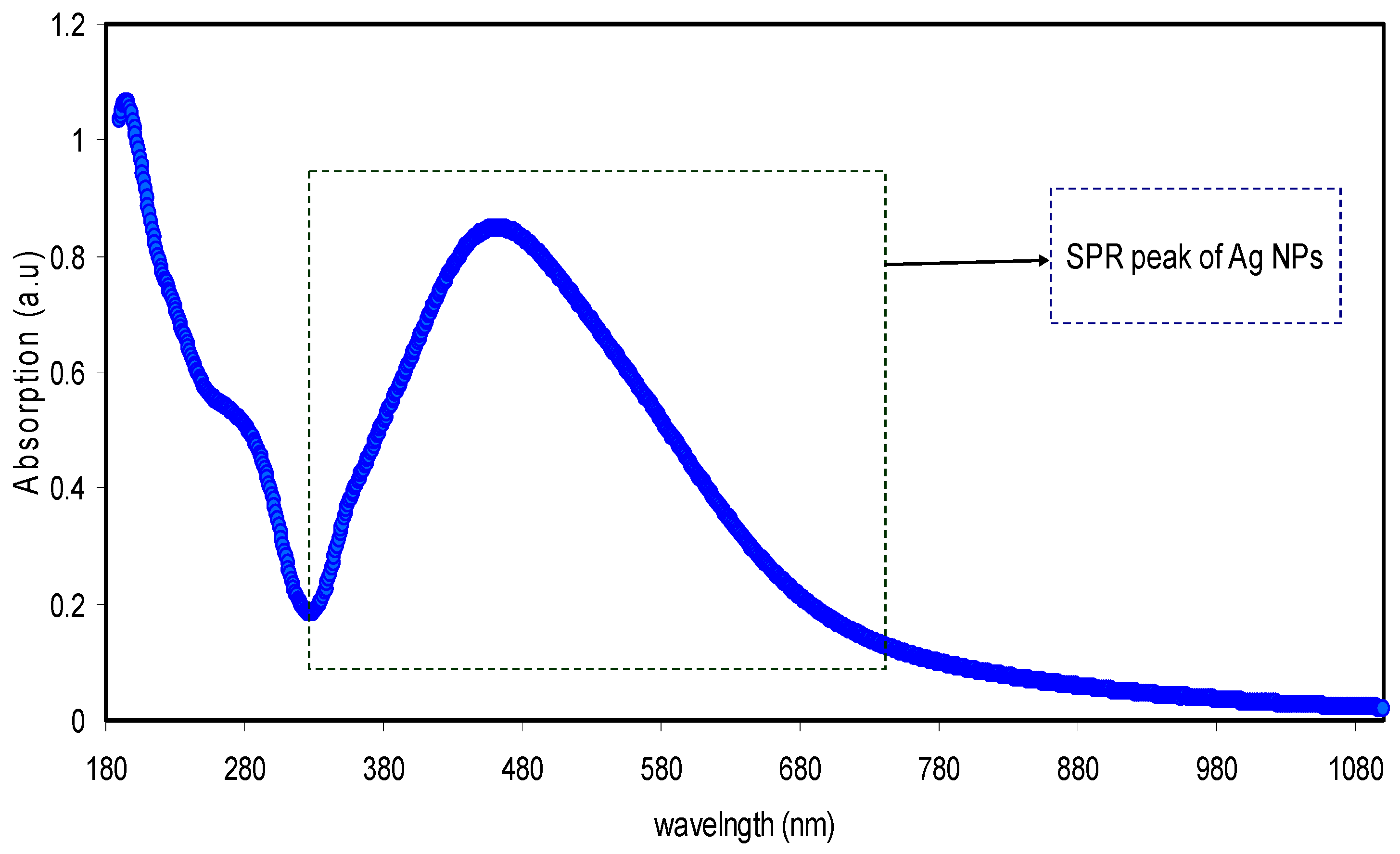
3.3.1. Absorption Study
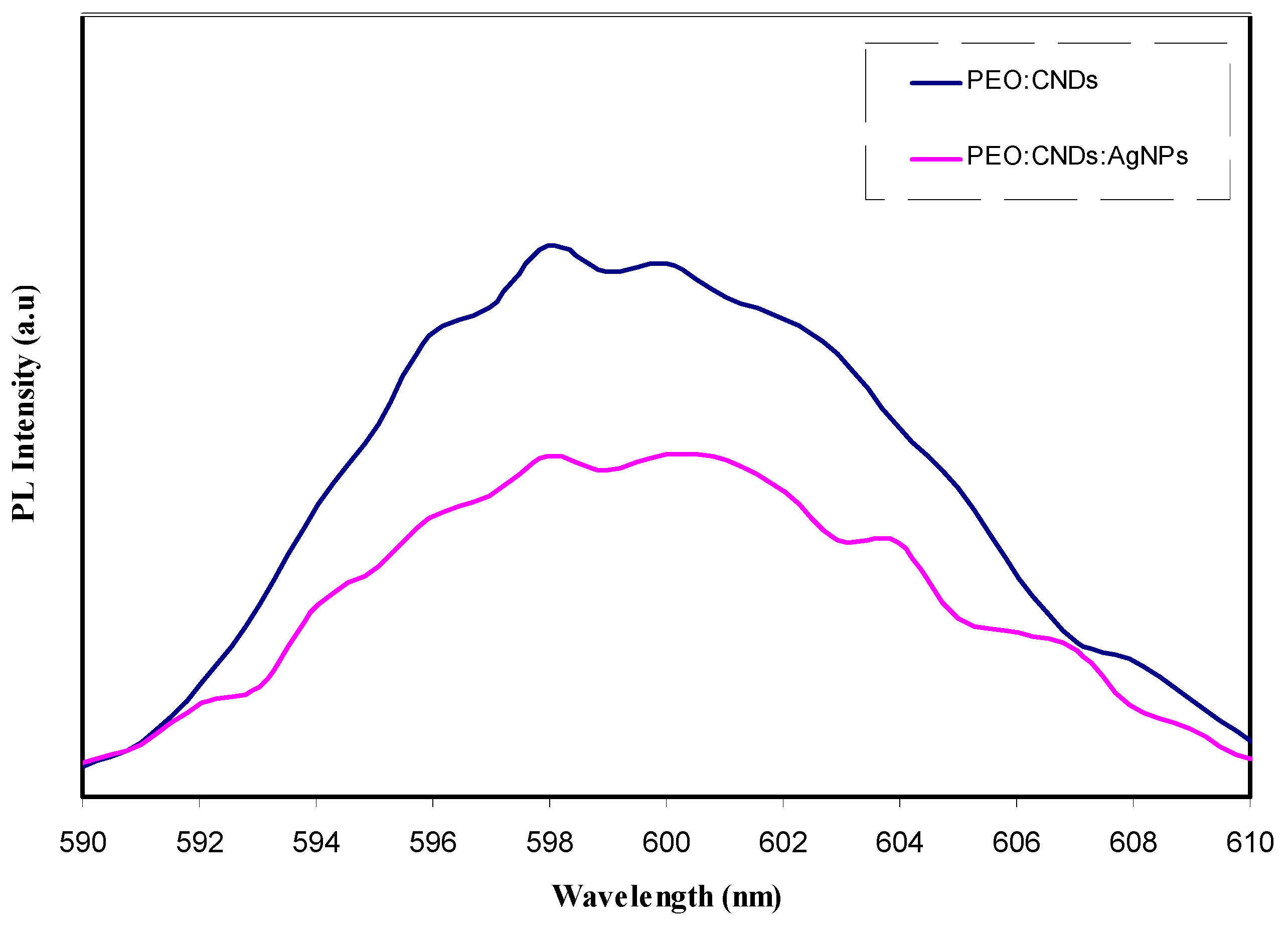
3.3.2. Photoluminescence (PL) Study
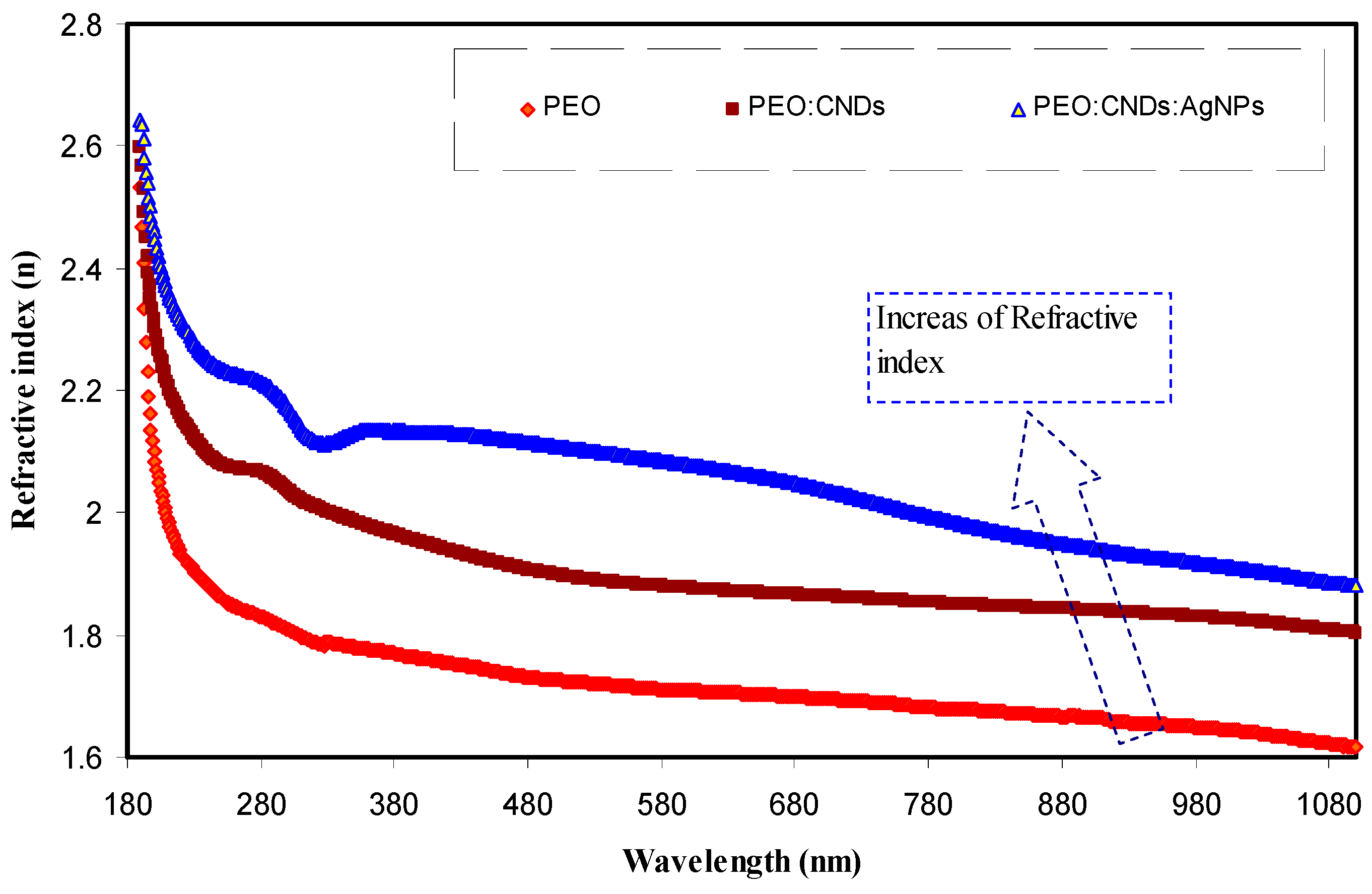
3.3.3. Refractive Index Study
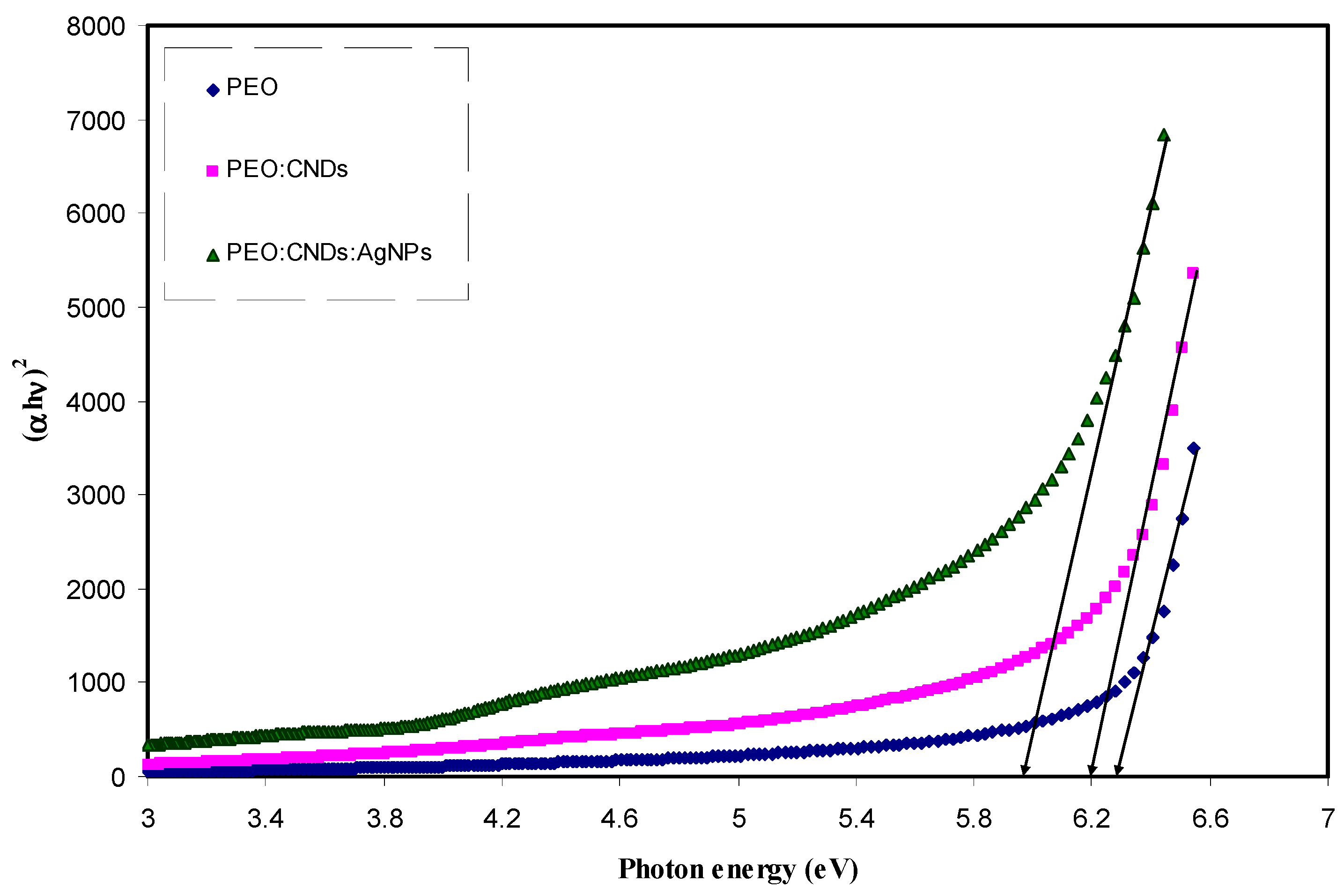
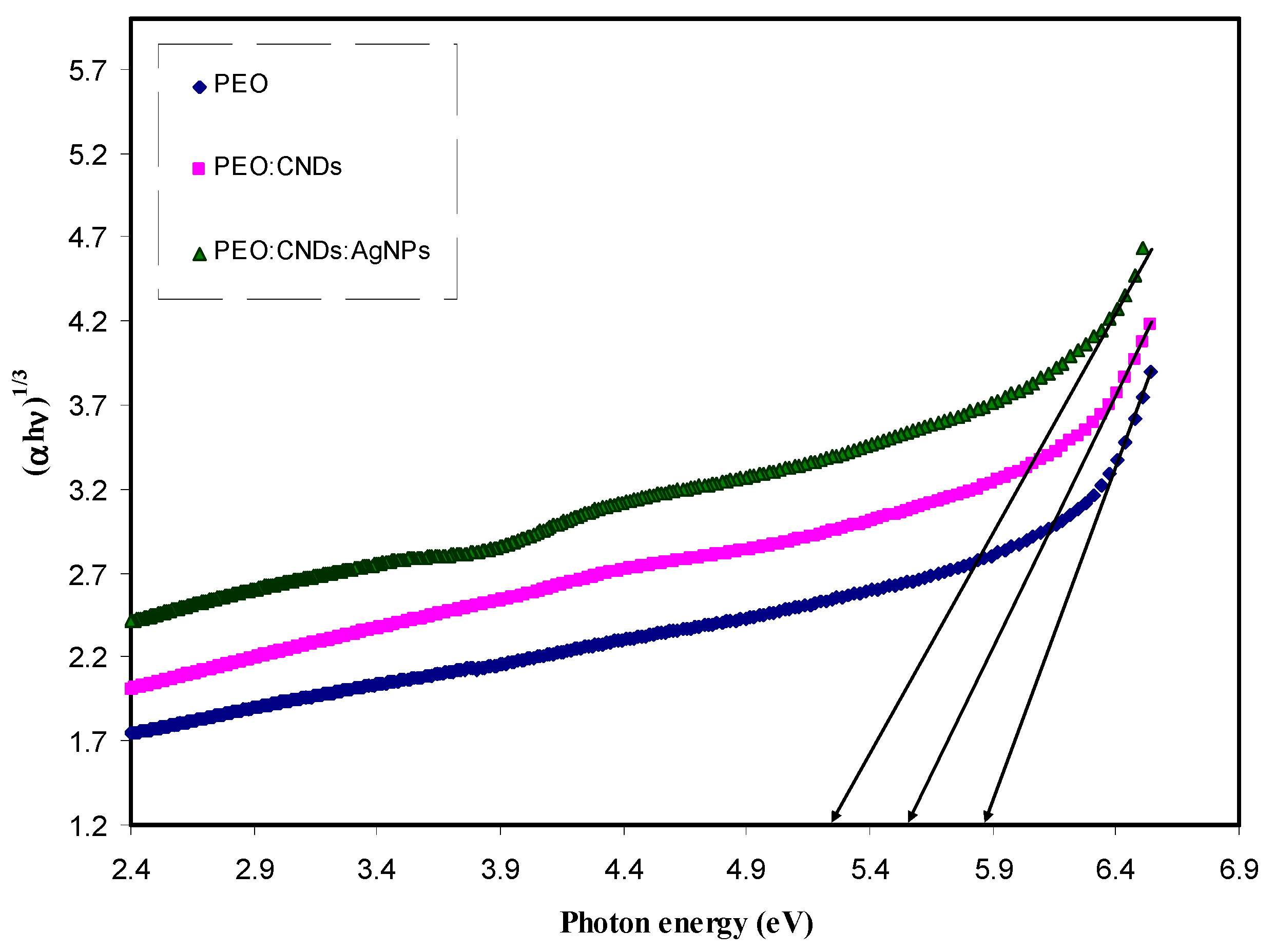
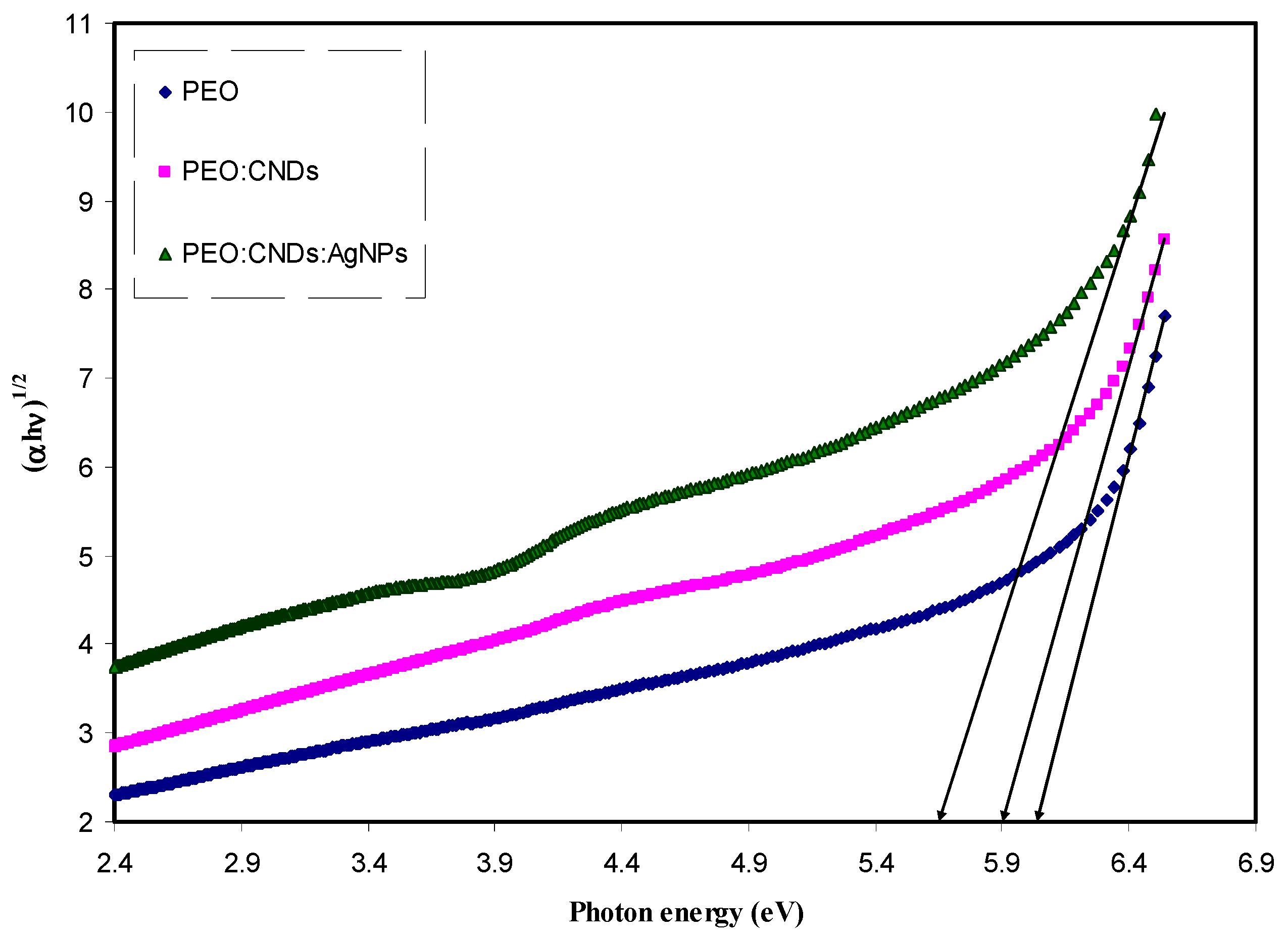
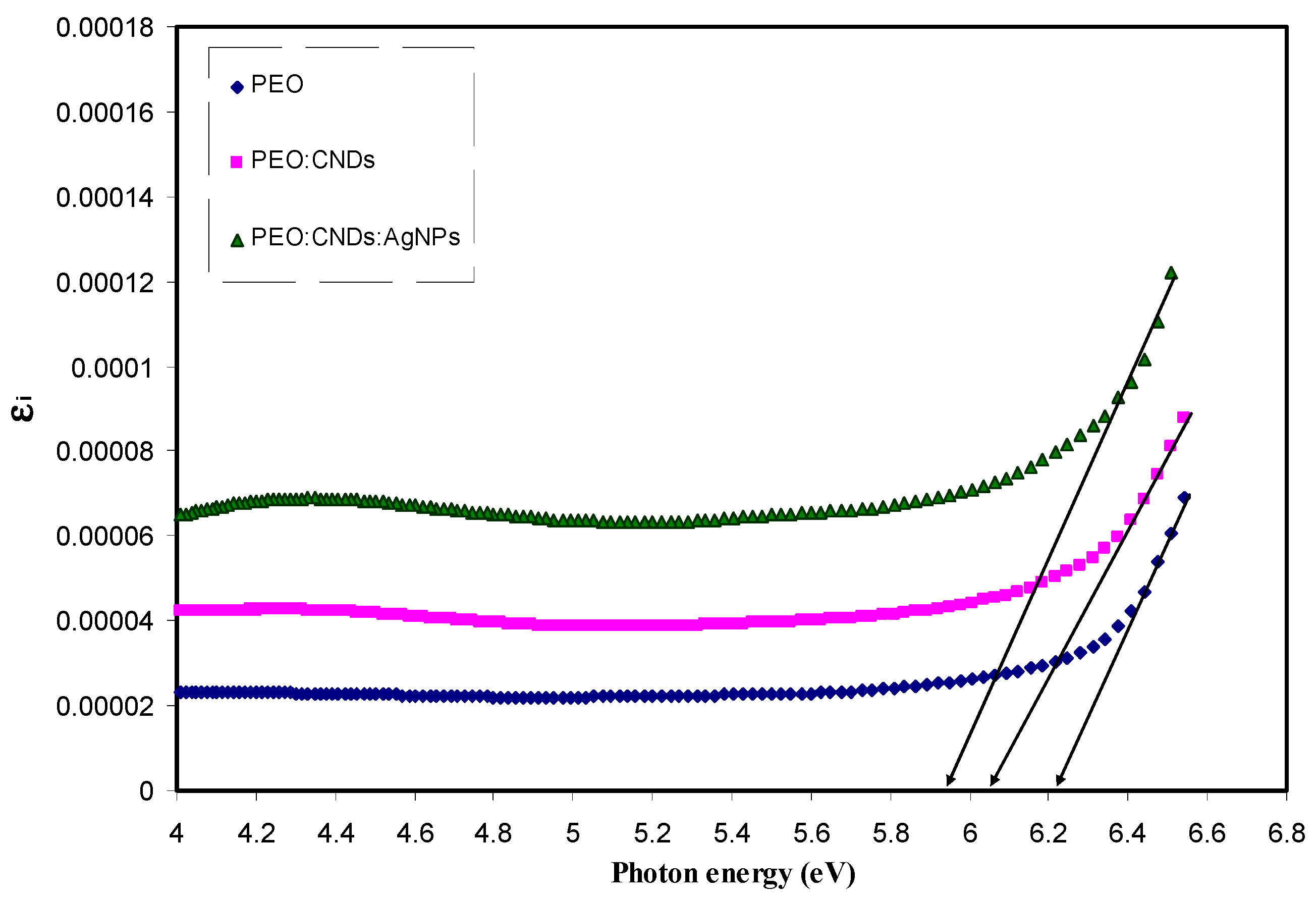
3.3.4. Band Gap Study
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Stepanov, A.L. Optical properties of polymer nanocomposites with functionalized nanoparticles. In Polymer Composites with Functionalized Nanoparticles; Elsevier: Amsterdam, The Netherlands, 2019; pp. 325–355. [Google Scholar]
- Aziz, S.B.; Hassan, A.Q.; Mohammed, S.J.; Karim, W.O.; Kadir, M.F.Z.; Tajuddin, H.A.; Chan, N.N.M.Y. Structural and optical characteristics of PVA:C-Dot composites: Tuning the absorption of ultra violet (UV) region. Nanomaterials 2019, 9, 216. [Google Scholar] [CrossRef] [PubMed]
- Bockstaller, M.R.; Thomas, E.L. Optical properties of polymer-based photonic nanocomposite materials. J. Phys. Chem. B 2003, 107, 10017–10024. [Google Scholar] [CrossRef]
- Aziz, S.B.; Abdulwahid, R.T.; Rsaul, H.A.; Ahmed, H.M. In situ synthesis of CuS nanoparticle with a distinguishable SPR peak in NIR region. J. Mater. Sci. Mater. Electron. 2016, 27, 4163–4171. [Google Scholar] [CrossRef]
- Srivastava, S.; Haridas, M.; Basu, J.K. Optical properties of polymer nanocomposites. Bull. Mater. Sci. 2008, 31, 213–217. [Google Scholar] [CrossRef]
- Aziz, S.B. Modifying poly(Vinyl Alcohol) (PVA) from Insulator to small-bandgap polymer: A Novel approach for organic solar cells and optoelectronic devices. J. Electron. Mater. 2016, 45, 736–745. [Google Scholar] [CrossRef]
- Aziz, S.B.; Abdullah, O.G.; Hussein, A.M.; Ahmed, H.M. From insulating PMMA polymer to conjugated double bond behavior: Green chemistry as a novel approach to fabricate small band gap polymers. Polymers 2017, 9, 626. [Google Scholar] [CrossRef] [PubMed]
- Jeon, I.Y.; Baek, J.B. Nanocomposites derived from polymers and inorganic nanoparticles. Materials 2010, 3, 3654–3674. [Google Scholar] [CrossRef]
- Khursheed, S.; Kumar, V.; Singh, V.K.; Sharma, J.; Swart, H.C. Optical properties of Sr3B2O6:Dy3+/PMMA polymer nanocomposites. Phys. B Condens. Matter 2018, 535, 184–188. [Google Scholar] [CrossRef]
- Aziz, S.B.; Abdullah, O.G.; Rasheed, M.A. A novel polymer composite with a small optical band gap: New approaches for photonics and optoelectronics. J. Appl. Polym. Sci. 2017, 134, 44847. [Google Scholar] [CrossRef]
- Aziz, S.B.; Mamand, S.M.; Saed, S.R.; Abdullah, R.M.; Hussein, S.A. New method for the development of plasmonic metal-semiconductor interface layer: Polymer composites with reduced energy band gap. J. Nanomater. 2017, 2017, 1–9. [Google Scholar] [CrossRef]
- Aziz, S. Morphological and optical characteristics of chitosan(1−x):Cuox (4 ≤ x ≤ 12) based polymer nano-composites: Optical dielectric loss as an alternative method for tauc’s model. Nanomaterials 2017, 7, 444. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Alexandridis, P. Composite polymer electrolytes: Nanoparticles affect structure and properties. Polymers 2016, 8, 387. [Google Scholar] [CrossRef]
- Wang, G.X.; Yang, L.; Wang, J.Z.; Liu, H.K.; Dou, S.X. Enhancement of ionic conductivity of PEO based polymer electrolyte by the addition of nanosize ceramic powders. J. Nanosci. Nanotechnol. 2005, 5, 1135–1140. [Google Scholar] [CrossRef] [PubMed]
- Mohan, V.M.; Bhargav, P.B.; Raja, V.; Sharma, A.K.; Rao, V.V.R.N. Optical and electrical properties of pure and doped PEO polymer electrolyte films. Soft Mater. 2007, 5, 33–46. [Google Scholar] [CrossRef]
- Vodnik, V.V.; Božanić, D.K.; Džunuzović, E.; Vuković, J.; Nedeljković, J.M. Thermal and optical properties of silver-poly(methylmethacrylate) nanocomposites prepared by in-situ radical polymerization. Eur. Polym. J. 2010, 46, 137–144. [Google Scholar] [CrossRef]
- Loo, Y.Y.; Chieng, B.W.; Nishibuchi, M.; Radu, S. Synthesis of silver nanoparticles by using tea leaf extract from Camellia Sinensis. Int. J. Nanomed. 2012, 7, 4263–4267. [Google Scholar]
- Kouvaris, P.; Delimitis, A.; Zaspalis, V.; Papadopoulos, D.; Tsipas, S.A.; Michailidis, N. Green synthesis and characterization of silver nanoparticles produced using Arbutus Unedo leaf extract. Mater. Lett. 2012, 76, 18–20. [Google Scholar] [CrossRef]
- Reverberi, A.P.; Vocciante, M.; Lunghi, E.; Pietrelli, L.; Fabiano, B. New trends in the synthesis of nanoparticles by green methods. Chem. Eng. Trans. 2017, 61, 667–672. [Google Scholar]
- Kamal, S.S.K.; Sahoo, P.K.; Vimala, J.; Premkumar, M.; Ram, S.; Durai, L. A novel green chemical route for synthesis of silver nanoparticles using Camellia Sinensis. Acta Chim. Slov. 2010, 57, 808–812. [Google Scholar]
- Sharma, V.K.; Yngard, R.A.; Lin, Y. Silver nanoparticles: Green synthesis and their antimicrobial activities. Adv. Colloid Interface Sci. 2009, 145, 83–96. [Google Scholar] [CrossRef]
- Mittal, A.K.; Chisti, Y.; Banerjee, U.C. Synthesis of metallic nanoparticles using plant extracts. Biotechnol. Adv. 2013, 31, 346–356. [Google Scholar] [CrossRef] [PubMed]
- Perva-Uzunalić, A.; Škerget, M.; Knez, Ž.; Weinreich, B.; Otto, F.; Grüner, S. Extraction of active ingredients from green tea (Camellia sinensis): Extraction efficiency of major catechins and caffeine. Food Chem. 2006, 96, 597–605. [Google Scholar] [CrossRef]
- Tariq, M.; Naveed, A.; Barkat, A.K. The morphology, characteristics, and medicinal properties of Camellia sinensis tea. J. Med. Plants Res. 2010, 4, 2028–2033. [Google Scholar] [CrossRef]
- Koduru, M.; Golla, N.; Dillip, G.R.; Praveen, B.; Bojja, S.; Ch, L.; Reddy, B.V.S.; Raju Borelli, D.P.; Raju, P. Green synthesis of silver nanoparticles using Ocimum leaf extract and their characterization. Dig. J. Nanomater. Biostruct. 2011, 6, 181–186. [Google Scholar]
- Dubey, S.P.; Lahtinen, M.; Sillanpää, M. Green synthesis and characterizations of silver and gold nanoparticles using leaf extract of Rosa rugosa. Colloids Surf. A Physicochem. Eng. Asp. 2010, 364, 34–41. [Google Scholar] [CrossRef]
- Aziz, S.B.; Abdullah, O.G.; Saber, D.R.; Rasheed, M.A.; Ahmed, H.M. Investigation of metallic silver nanoparticles through UV-Vis and optical micrograph techniques. Int. J. Electrochem. Sci. 2017, 12, 363–373. [Google Scholar] [CrossRef]
- Ramya, M.; Subapriya, M.S. Green synthesis of silver nanoparticles. Int. J. Pharm. Med. Biol. Sci. 2012, 1, 1. [Google Scholar]
- Yeshchenko, O.A.; Dmitruk, I.M.; Alexeenko, A.A.; Losytskyy, M.Y.; Kotko, A.V.; Pinchuk, A.O. Size-dependent surface-plasmon-enhanced photoluminescence from silver nanoparticles embedded in silica. Phys. Rev. B Condens. Matter Mater. Phys. 2009, 79, 235438. [Google Scholar] [CrossRef]
- Ambasankar, K.N.; Bhattacharjee, L.; Jat, S.K.; Bhattacharjee, R.R.; Mohanta, K. Study of electrical charge storage in polymer-carbon quantum dot composite. ChemistrySelect 2017, 2, 4241–4247. [Google Scholar] [CrossRef]
- Atabaev, T. Doped carbon dots for sensing and bioimaging applications: A minireview. Nanomaterials 2018, 8, 342. [Google Scholar] [CrossRef]
- Wang, R.; Lu, K.Q.; Tang, Z.R.; Xu, Y.J. Recent progress in carbon quantum dots: Synthesis, properties and applications in photocatalysis. J. Mater. Chem. A 2017, 5, 3717–3734. [Google Scholar] [CrossRef]
- Aziz, S.B.; Marif, R.B.; Brza, M.A.; Hassan, A.N.; Ahmad, H.A.; Faidhalla, Y.A.; Kadir, M.F. Structural, thermal, morphological and optical properties of PEO filled with biosynthesized Ag nanoparticles: New insights to band gap study. Results Phys. 2019, 13, 102220. [Google Scholar] [CrossRef]
- Vogel, M.; Herbers, C.; Koch, B. Effects of salt and nanoparticles on the segmental motion of poly(ethylene oxide) in its crystalline and amorphous phases: 2H and 7Li NMR studies. J. Phys. Chem. B 2008, 112, 11217–11226. [Google Scholar] [CrossRef] [PubMed]
- Koduru, H.K.; Iliev, M.T.; Kondamareddy, K.K.; Karashanova, D.; Vlakhov, T.; Zhao, X.-Z.; Scaramuzza, N. Investigations on poly (ethylene oxide) (PEO)—Blend based solid polymer electrolytes for sodium ion batteries. J. Phys. Conf. Ser. 2016, 764, 012006. [Google Scholar] [CrossRef]
- Aziz, S.B.; Abdullah, R.M. Crystalline and amorphous phase identification from the tanδ relaxation peaks and impedance plots in polymer blend electrolytes based on [CS:AgNt]x:PEO(x−1) (10 ≤ x ≤ 50). Electrochim. Acta 2018, 285, 30–46. [Google Scholar] [CrossRef]
- Huang, C.I.; Chen, J.R. Crystallization and chain conformation of semicrystalline and amorphous polymer blends studied by wide-angle and small-angle scattering. J. Polym. Sci. Part B Polym. Phys. 2001, 39, 2705–2715. [Google Scholar] [CrossRef]
- Johan, M.R.; Ting, L.M. Structural, thermal and electrical properties of nano manganese-composite polymer electrolytes. Int. J. Electrochem. Sci. 2011, 6, 4737–4748. [Google Scholar]
- Hamsan, M.H.; Aziz, S.B.; Shukur, M.F.; Kadir, M.F.Z. Protonic cell performance employing electrolytes based on plasticized methylcellulose-potato starch-NH4NO3. Ionics 2019, 25, 559. [Google Scholar] [CrossRef]
- Aziz, S.B.; Abidin, Z.H.Z.; Arof, A.K. Effect of silver nanoparticles on the DC conductivity in chitosansilver triflate polymer electrolyte. Phys. B Condens. Matter 2010, 405, 4429–4433. [Google Scholar] [CrossRef]
- Aziz, S.B.; Abidin, Z.H.Z.; Kadir, M.F.Z. Innovative method to avoid the reduction of silver ions to silver nanoparticles (Ag+→Ago) in silver ion conducting based polymer electrolytes. Phys. Scr. 2015, 90, 035808. [Google Scholar] [CrossRef]
- Hana, S.W.; Shi, W.; Jin, J. Morphology and Crystallization of Crystalline/Amorphous Polymer Blends. In Encyclopedia of Polymers and Composites; Palsule, S., Ed.; Springer: Berlin/Heidelberg, Germany, 2013; pp. 1–19. [Google Scholar]
- Aziz, S.B.; Abdullah, R.M.; Kadir, M.F.Z.; Ahmed, H.M. Non suitability of silver ion conducting polymer electrolytes based on chitosan mediated by barium titanate (BaTiO3) for electrochemical device applications. Electrochim. Acta 2019, 296, 494–507. [Google Scholar] [CrossRef]
- Aziz, S.B.; Abidin, Z.H.Z. Ion-transport study in nanocomposite solid polymer electrolytes based on chitosan: Electrical and dielectric analysis. J. Appl. Polym. Sci. 2015, 132, 41774. [Google Scholar] [CrossRef]
- Aziz, S.B. Role of dielectric constant on ion transport: reformulated arrhenius equation. Adv. Mater. Sci. Eng. 2016, 2016, 11. [Google Scholar] [CrossRef]
- Lin, J.-H.; Woo, E.M.; Huang, Y.-P. Effects of lithium salt and poly(4-vinyl phenol) on crystalline and amorphous phases in poly(ethylene oxide). J. Polym. Sci. Part B Polym. Phys. 2006, 44, 3357–3368. [Google Scholar] [CrossRef]
- Bandara, T.M.W.J.; Karunathilaka, D.G.N.; Ratnasekera, J.L.; de Silva, L.A.; Herath, A.C.; Mellander, B.E. Electrical and complex dielectric behaviour of composite polymer electrolyte based on PEO, alumina and tetrapropylammonium iodide. Ionics 2017, 23, 1711–1719. [Google Scholar] [CrossRef]
- Emam, A.N.; Loutfy, S.A.; Mostafa, A.A.; Awad, H.; Mohamed, M.B. Cyto-toxicity, biocompatibility and cellular response of carbon dots-plasmonic based nano-hybrids for bioimaging. RSC Adv. 2017, 7, 23502–23514. [Google Scholar] [CrossRef]
- Li, X.; Zhang, S.; Kulinich, S.A.; Liu, Y.; Zeng, H. Engineering surface states of carbon dots to achieve controllable luminescence for solid-luminescent composites and sensitive Be2+ detection. Sci. Rep. 2014, 4, 4976. [Google Scholar] [CrossRef]
- Wang, Y.H. Carbon quantum dots: synthesis, properties and applications. J. Mater. Chem. C 2014, 2, 6921–6939. [Google Scholar] [CrossRef]
- Kumar, R.; Asad Alia, S.; Mahura, A.K.; Virk, H.S.; Singh, F.; Khan, S.A.; Avasthic, D.K.; Rajendra, P. Study of optical band gap and carbonaceous clusters in swift heavy ion irradiated polymers with UV-Vis spectroscopy. Nucl. Instr. Methods Phys. Res. Sect. B Beam Interact. Mater. Atoms. 2008, 266, 1788–1792. [Google Scholar] [CrossRef]
- Kovalchuk, A.; Huang, K.; Xiang, C.; Martí, A.A.; Tour, J.M. Luminescent Polymer composite films containing coal-derived graphene quantum dots. ACS Appl. Mater. Interfaces 2015, 7, 26063–26068. [Google Scholar] [CrossRef]
- Woelfle, C.; Claus, R.O. Transparent and flexible quantum dot-polymer composites using an ionic liquid as compatible polymerization medium. Nanotechnology 2007, 18, 9. [Google Scholar] [CrossRef]
- Camelio, S.; Babonneau, D.; Denanot, M.-F.; Girardeau, T.; Espiños, J.P.; Yubero, F.; Gonzalez-Elip, A.R. Morphology and surface-plasmon resonance of silver nanoparticles sandwiched between Si 3 N 4 and BN layers. J. Appl. Phys. 2005, 98, 114316. [Google Scholar]
- Schaadt, D.M.; Feng, B.; Yu, E.T. Enhanced semiconductor optical absorption via surface plasmon excitation in metal nanoparticles. Appl. Phys. Lett. 2005, 86, 063106. [Google Scholar] [CrossRef]
- Wu, B.; Wang, Q.K. Investigation of highly sensitive surface plasmon resonance biosensors with Au nanoparticles embedded dielectric film using rigorous coupled wave analysis. Opt. Appl. 2009, XXXIX, 32–40. [Google Scholar]
- Das, R.; Nath, S.S.; Chakdar, D.; Gope, G.; Bhattacharjee, R. Synthesis of silver nanoparticles and their optical properties. J. Exp. Nanosci. 2010, 5, 357–362. [Google Scholar] [CrossRef]
- Amendola, V.; Bakr, O.M.; Stellacci, F. A study of the surface plasmon resonance of silver nanoparticles by the discrete dipole approximation method: Effect of shape, size, structure, and assembly. Plasmonics 2010, 5, 85–97. [Google Scholar] [CrossRef]
- Hu, S.; Tian, R.; Dong, Y.; Yang, J.; Liu, J.; Chang, Q. Modulation and effects of surface groups on photoluminescence and photocatalytic activity of carbon dots. Nanoscale 2013, 5, 11665–11671. [Google Scholar] [CrossRef]
- Yang, G.; Wan, X.; Liu, Y.; Li, R.; Su, Y.; Zeng, X.; Tang, J. Luminescent poly(vinyl alcohol)/carbon quantum dots composites with tunable water-induced shape memory behavior in different pH and temperature environments. ACS Appl. Mater. Interfaces 2016, 8, 34744–34754. [Google Scholar] [CrossRef]
- Wooten, F. Optical Properties of Solids; Academic Press: New York, NY, USA, 1972. [Google Scholar]
- Dresselhaus, M.S. Solid State Physics Part II Optical Properties of Solids, vol. 6, 2001. Available online: http://web.mit.edu/course/6/6.732/www/6.732-pt2.pdf (accessed on 1 April 2019).
- Aziz, S.B.; Ahmed, H.M.; Hussein, A.M.; Fathulla, A.B.; Wsw, R.M.; Hussein, R.T. Tuning the absorption of ultraviolet spectra and optical parameters of aluminum doped PVA based solid polymer composites. J. Mater. Sci. Mater. Electron. 2015, 26, 8022. [Google Scholar] [CrossRef]
- Baláž, M.; Mate, B.; Ľudmila, B.; Nina, D.; Erika, D.; Miriama, B.; Zdenka, B.; Yaroslav, S. Plant-mediated synthesis of silver nanoparticles and their stabilization by wet stirred media milling. Nanoscale Res. Lett. 2017, 12, 83. [Google Scholar] [CrossRef]
- Horiuchi, Y.; Shimada, M.; Kamegawa, T.; Mori, K.; Yamashita, H. Size-controlled synthesis of silver nanoparticles on Ti-containing mesoporous silica thin film and photoluminescence enhancement of rhodamine 6G dyes by surface plasmon resonance. J. Mater. Chem. 2009, 19, 6745–6749. [Google Scholar] [CrossRef]
- Yeshchenko, O.A.; Bondarchuk, I.S.; Losytskyy, M.Y.; Alexeenko, A.A. Temperature dependence of photoluminescence from silver nanoparticles. Plasmonics 2014, 9, 93–101. [Google Scholar] [CrossRef]
- Gao, J.; Jun, F.; Cuikun, L.; Jun, L.; Yanchun, H.; Xiang, Y.; Caiyuan, P. Formation and photoluminescence of silver nanoparticles stabilized by a two-armed polymer with a crown ether core. Langmuir 2004, 20, 9775–9779. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, R.; Kumbhakar, P.; Mitra, A. Green synthesis of silver nanoparticles and its optical properties. Dig. J. Nanomater. Biostruct. 2010, 5, 491–496. [Google Scholar]
- Marder, M.P. Chapter 21 optical properties. In Condensed Matter Physics; John Wiley and Sons Ltd.: Hoboken, NJ, USA, 2000. [Google Scholar]
- Guo, Q.; Reza, G.; Thomas, W.; Andreas, A.; Evgeny, L.G.; Cemal, E.; Olaf, M.; Wei, C.; Boris, C.; Andreas, O. Comparison of in situ and ex situ methods for synthesis of two-photon polymerization polymer nanocomposites. Polymers 2014, 6, 2037–2050. [Google Scholar] [CrossRef]
- Jin, J.; Qi, R.; Su, Y.; Tong, M.; Zhu, J. Preparation of high-refractive-index PMMA/TiO2 nanocomposites by one-step in situ solvothermal method. Iran. Polym. J. 2013, 22, 767–774. [Google Scholar] [CrossRef]
- Mansur, H.S.; Mansur, A.A.P. CdSe quantum dots stabilized by carboxylic-functionalized PVA: Synthesis and UV-vis spectroscopy characterization. Mater. Chem. Phys. 2011, 125, 709–717. [Google Scholar] [CrossRef]
- Barajas-Ledesma, E.; García-Benjume, M.L.; Espitia-Cabrera, I.; Ortiz-Gutiérrez, M.; Espinoza-Beltrán, F.J.; Mostaghimi, J.; Contreras-García, M.E. Determination of the band gap of TiO2-Al2O3 films as a function of processing parameters. Mater. Sci. Eng. B Solid-State Mater. Adv. Technol. 2010, 174, 71–73. [Google Scholar] [CrossRef]
- Mohan, V.M.; Raja, V.; Bhargav, P.B.; Sharma, A.K.; Rao, V.V.R.N. Structural, electrical and optical properties of pure and NaLaF4 doped PEO polymer electrolyte films. J. Polym. Res. 2007, 14, 283–290. [Google Scholar] [CrossRef]
- Ibrahim, S.; Ahmad, R.; Johan, M.R. Conductivity and optical studies of plasticized solid polymer electrolytes doped with carbon nanotube. J. Lumin. 2012, 132, 147–152. [Google Scholar] [CrossRef]
- Yakuphanoglu, F.; Arslan, M. Determination of electrical conduction mechanism and optical band gap of a new charge transfer complex: TCNQ-PANT. Solid State Commun. 2004, 132, 229–234. [Google Scholar] [CrossRef]
- Rocquefelte, X.; Jobic, S.; Whangbo, M.H. Concept of optical channel as a guide for tuning the optical properties of insulating materials. Solid State Sci. 2007, 9, 600–603. [Google Scholar] [CrossRef]
- Rodríguez, A.; Vergara, M.E.S.; Montalvo, V.G.; Ortiz, A.; Alvarez, J.R. Thin films of molecular materials synthesized from C32H20N10M (M = Co, Pb, Fe): Film formation, electrical and optical properties. Appl. Surf. Sci. 2010, 256, 3374–3379. [Google Scholar] [CrossRef]
- Hossain, F.M.; Sheppard, L.; Nowotny, J.; Murch, G.E. Optical properties of anatase and rutile titanium dioxide: Ab initio calculations for pure and anion-doped material. J. Phys. Chem. Solids 2008, 69, 1820–1828. [Google Scholar] [CrossRef]
- Aziz, S.B.; Rasheed, M.A.; Hussein, A.M.; Ahmed, H.M. Fabrication of polymer blend composites based on [PVA-PVP](1−x):(Ag2S)x (0.01 ≤ x ≤ 0.03) with small optical band gaps: Structural and optical properties. Mater. Sci. Semicond. Process. 2017, 71, 197–203. [Google Scholar] [CrossRef]


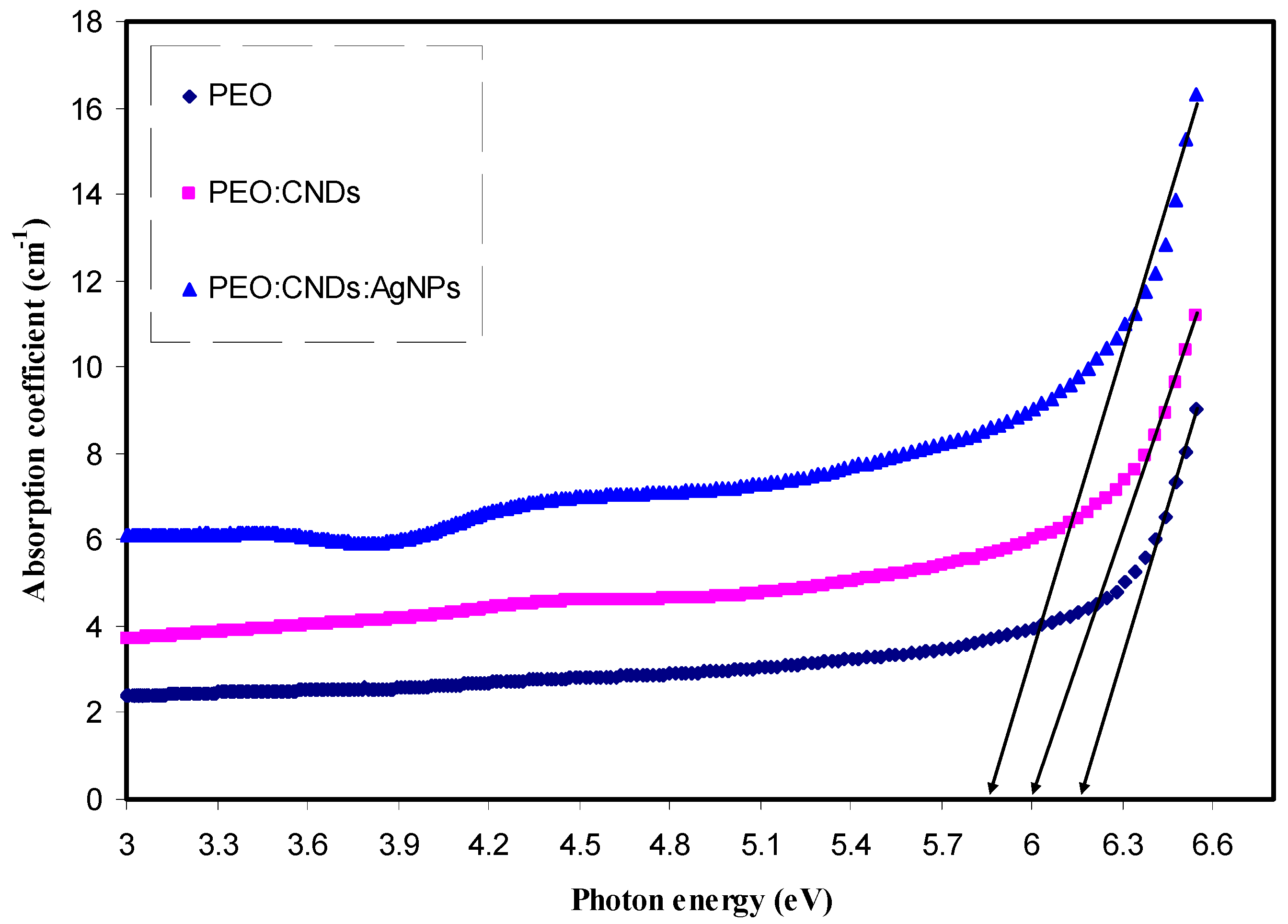
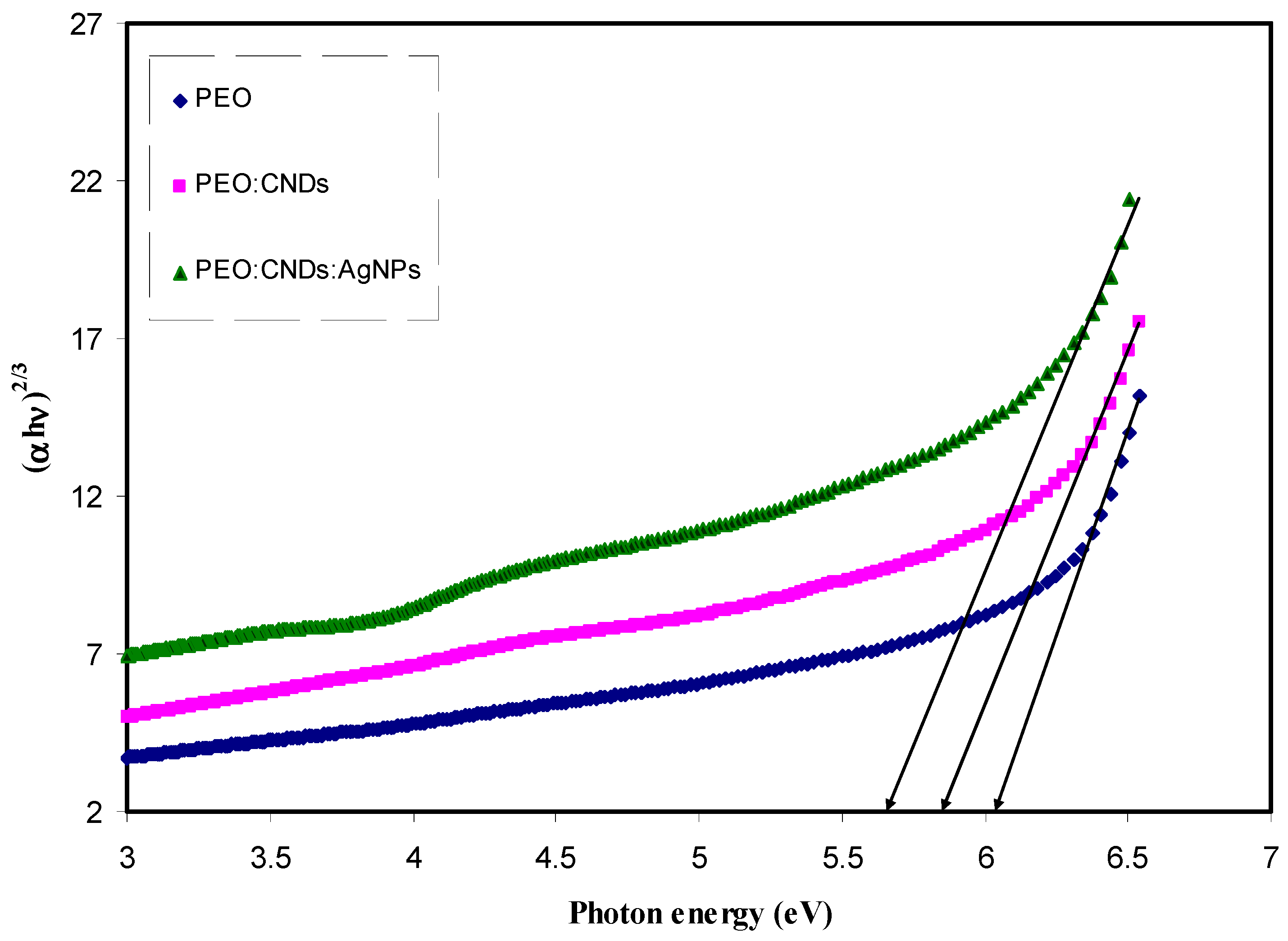
| Sample Code | Absorption Edge (eV) |
|---|---|
| PEPN 0 | 6.27 |
| PEPN 1 | 6 |
| PEPN2 | 5.84 |
| Sample Code | Eg for n = 1/2 | Eg for n = 2 | Eg for n = 3 | Eg for n = 3/2 | Eg from εi Plot |
|---|---|---|---|---|---|
| PEPN 0 | 6.26 | 6.07 | 5.83 | 6.07 | 6.22 |
| PEPN 1 | 6.2 | 5.9 | 5.58 | 5.84 | 6.05 |
| PEPN2 | 6 | 5.65 | 5.35 | 5.7 | 5.95 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abdullah, R.M.; Aziz, S.B.; Mamand, S.M.; Hassan, A.Q.; Hussein, S.A.; Kadir, M.F.Z. Reducing the Crystallite Size of Spherulites in PEO-Based Polymer Nanocomposites Mediated by Carbon Nanodots and Ag Nanoparticles. Nanomaterials 2019, 9, 874. https://doi.org/10.3390/nano9060874
Abdullah RM, Aziz SB, Mamand SM, Hassan AQ, Hussein SA, Kadir MFZ. Reducing the Crystallite Size of Spherulites in PEO-Based Polymer Nanocomposites Mediated by Carbon Nanodots and Ag Nanoparticles. Nanomaterials. 2019; 9(6):874. https://doi.org/10.3390/nano9060874
Chicago/Turabian StyleAbdullah, Ranjdar M., Shujahadeen B. Aziz, Soran M. Mamand, Aso Q. Hassan, Sarkawt A. Hussein, and M. F. Z. Kadir. 2019. "Reducing the Crystallite Size of Spherulites in PEO-Based Polymer Nanocomposites Mediated by Carbon Nanodots and Ag Nanoparticles" Nanomaterials 9, no. 6: 874. https://doi.org/10.3390/nano9060874
APA StyleAbdullah, R. M., Aziz, S. B., Mamand, S. M., Hassan, A. Q., Hussein, S. A., & Kadir, M. F. Z. (2019). Reducing the Crystallite Size of Spherulites in PEO-Based Polymer Nanocomposites Mediated by Carbon Nanodots and Ag Nanoparticles. Nanomaterials, 9(6), 874. https://doi.org/10.3390/nano9060874







