Small-Angle Scattering as a Structural Probe for Nucleic Acid Nanoparticles (NANPs) in a Dynamic Solution Environment
Abstract
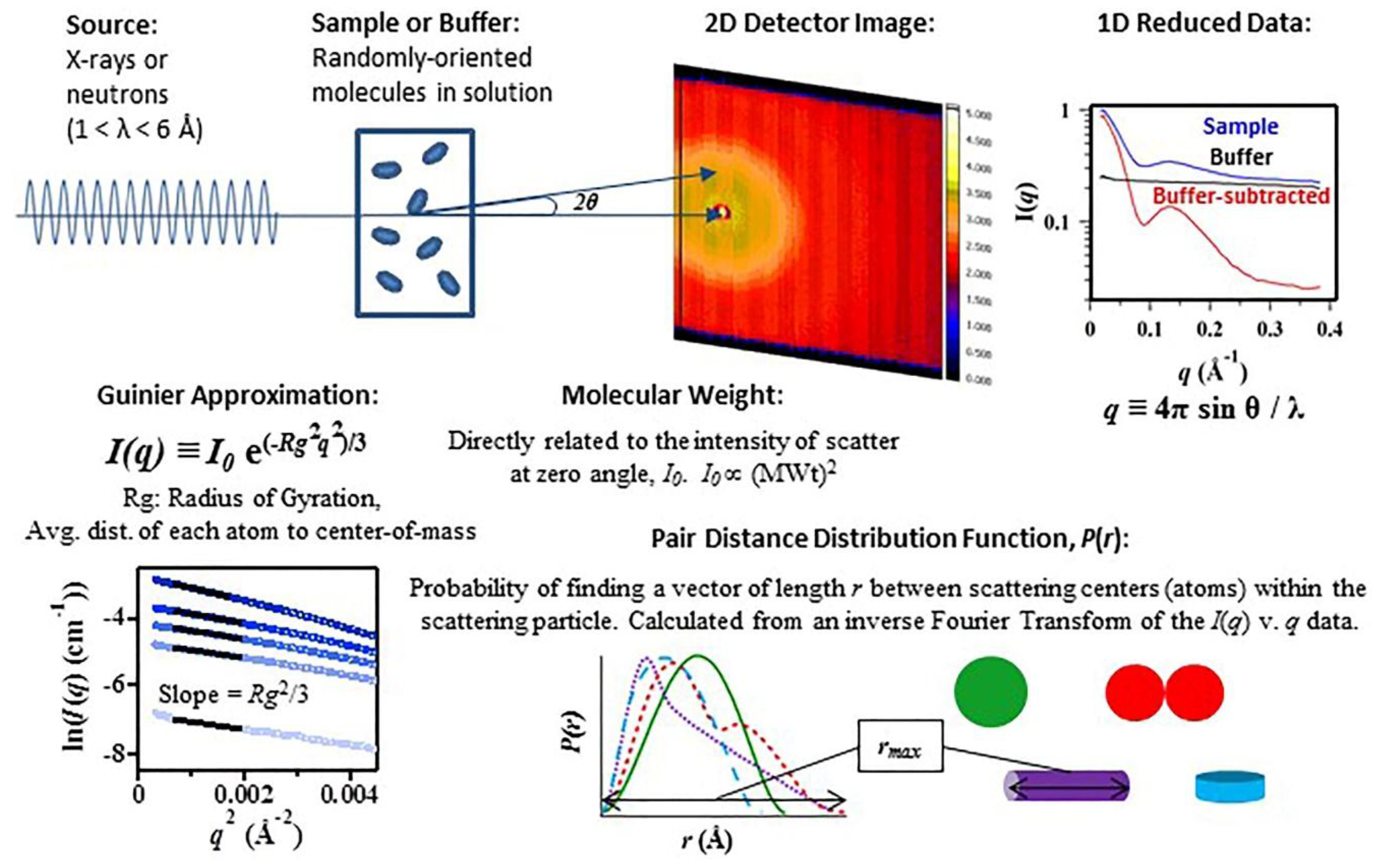
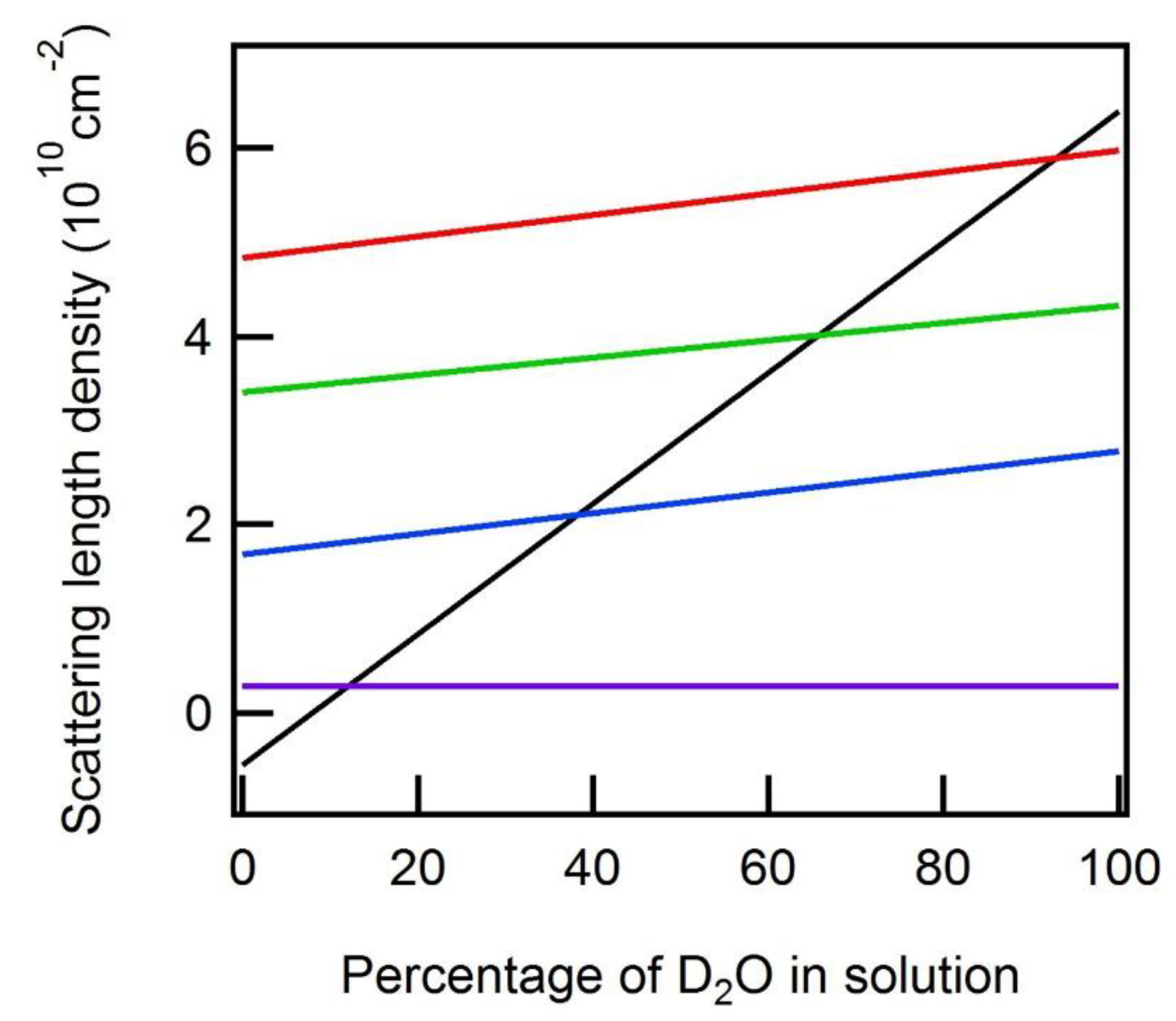
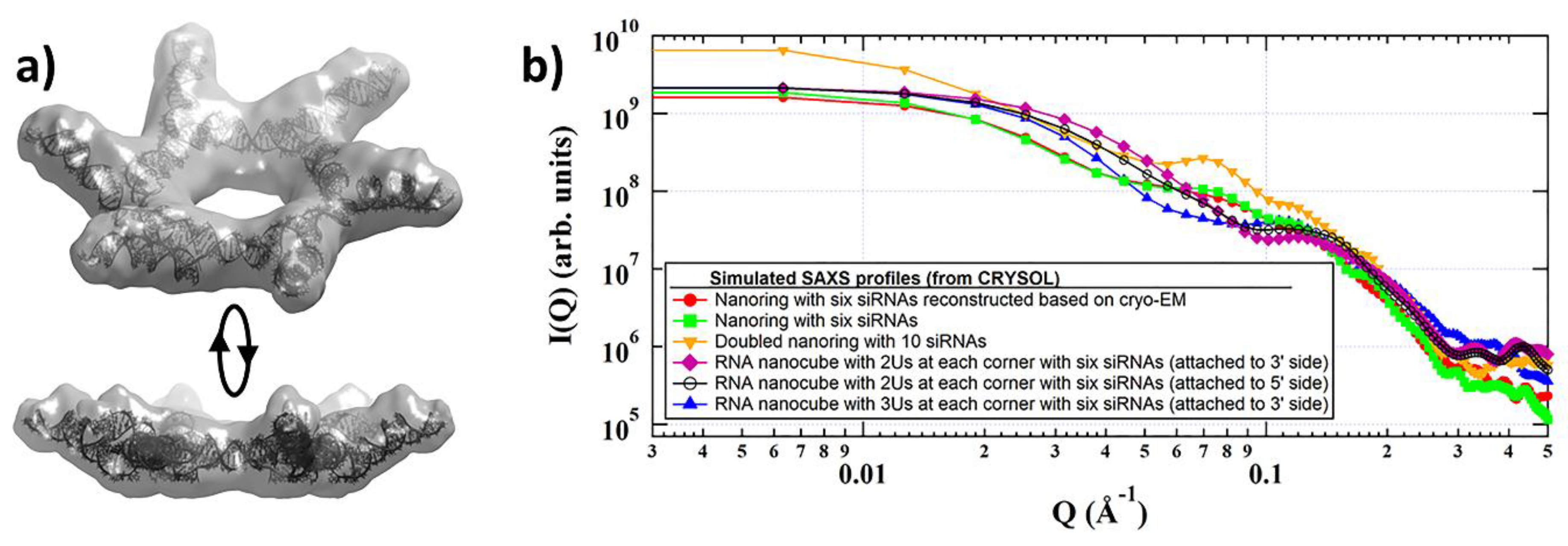
1. Introduction
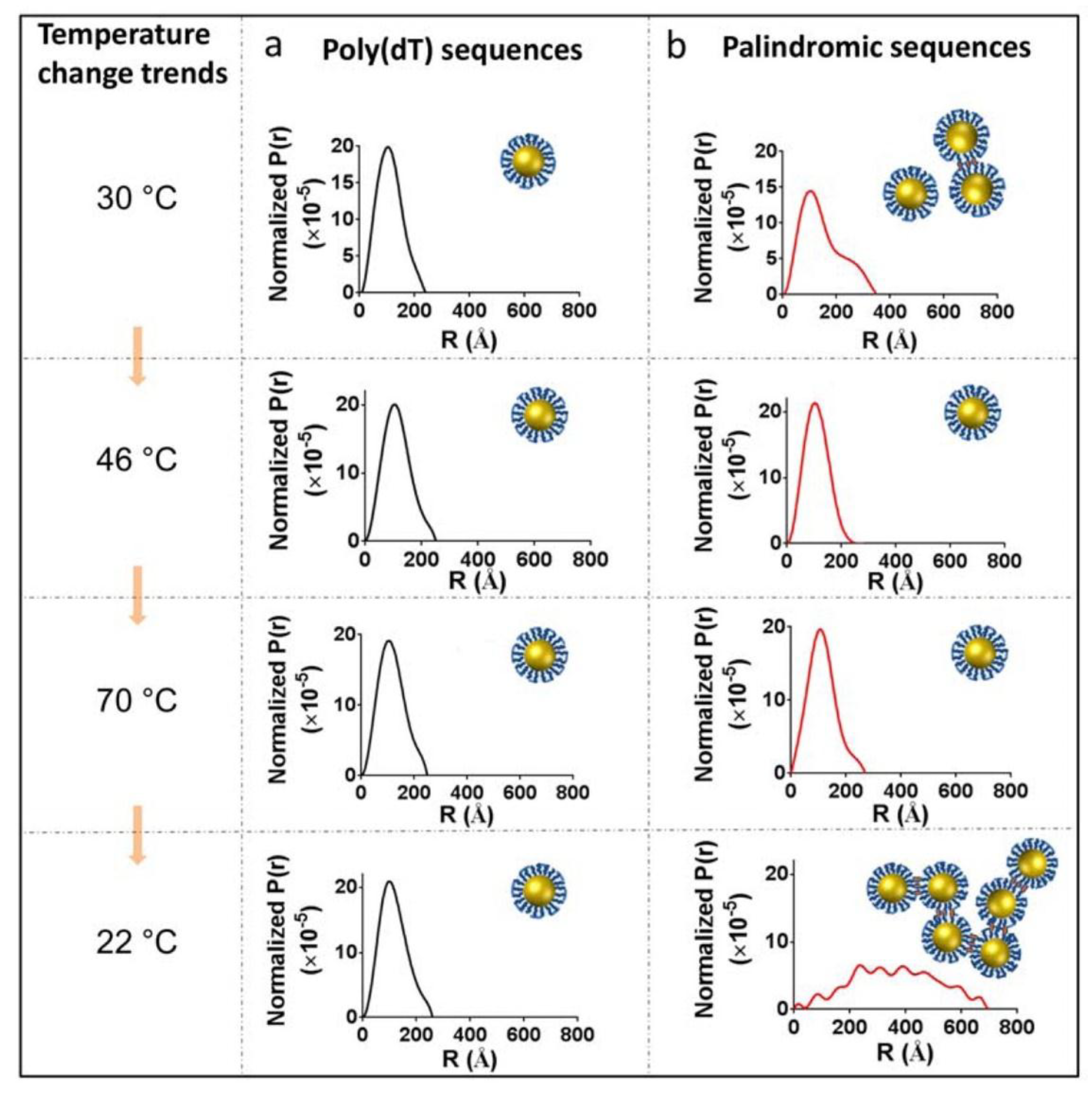
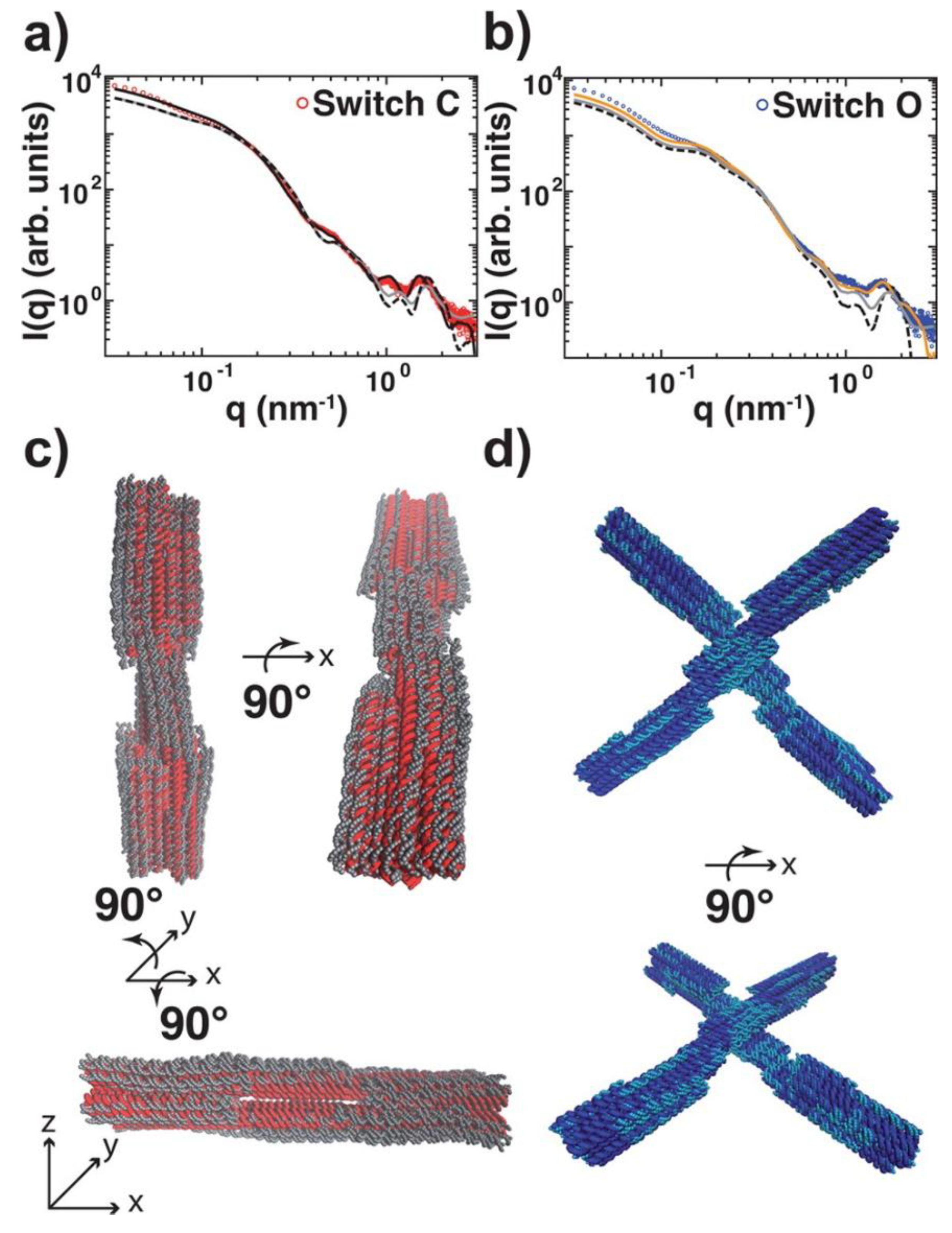
2. Discussion
3. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Guo, P.X. The Emerging Field of RNA Nanotechnology. Nat. Nanotechnol. 2010, 5, 833–842. [Google Scholar] [CrossRef] [PubMed]
- Shukla, G.C.; Haque, F.; Tor, Y.; Wilhelmsson, L.M.; Toulmé, J.J.; Isambert, H.; Guo, P.; Rossi, J.J.; Tenenbaum, S.A.; Shapiro, B.A. A Boost for the Emerging Field of RNA Nanotechnology. ACS Nano 2011, 5, 3405–3418. [Google Scholar] [CrossRef]
- Li, H.; Lee, T.; Dziubla, T.; Pi, F.; Guo, S.; Xu, J.; Li, C.; Haque, F.; Liang, X.J.; Guo, P. RNA as a Stable Polymer to Build Controllable and Defined Nanostructures for Material and Biomedical Applications. Nano Today 2015, 10, 631–655. [Google Scholar] [CrossRef] [PubMed]
- Kirill, A.A.; Kasprzak, W.; Bindewald, E.; Praneet, S.; Puppala Alex, R.; Diehl Kenneth, T.; Hall Tae Jin Kim Michael, T.; Zimmermann Robert, L.; Jernigan, L.J.; Shapiro, B.A. Computational and Experimental Characterization of RNA Cubic Nanoscaffolds. Methods 2014, 67, 256–265. [Google Scholar]
- Kirill, A.A.; Kasprzak, W.K.; Bindewald, E.; Kireeva, M.; Viard, M.; Kashlev, M.; Shapiro, B.A. In Silico Design and Enzymatic Synthesis of Functional RNA Nanoparticles. Account. Chem. Res. 2014, 47, 1731–1741. [Google Scholar]
- Afonin, K.A.; Viard, M.; Kagiampakis, I.; Case, C.L.; Dobrovolskaia, M.A.; Hofmann, J.; Vrzak, A.; Kireeva, M.; Kasprzak, W.K.; KewalRamani, V.N.; et al. Triggering of RNA Interference with RNA-RNA, RNA-DNA, and DNA-RNA Nanoparticles. ACS Nano 2015, 9, 251–259. [Google Scholar] [CrossRef]
- Afonin, K.A.; Viard, M.; Koyfman, A.Y.; Martins, A.N.; Kasprzak, W.K.; Panigaj, M.; Desai, R.; Santhanam, A.; Grabow, W.W.; Jaeger, L.; et al. Multifunctional RNA Nanoparticles. Nano Lett. 2014, 14, 5662–5671. [Google Scholar] [CrossRef] [PubMed]
- Dao, B.N.; Viard, M.; Martins, A.N.; Kasprzak, W.K.; Shapiro, B.A.; Afonin, K.A. Triggering Rnai with Multifunctional RNA Nanoparticles and Their Delivery. DNA RNA Nanotechnol. 2015, 1, 27–38. [Google Scholar] [CrossRef][Green Version]
- Ohno, H.; Kobayashi, T.; Kabata, R.; Endo, K.; Iwasa, T.; Yoshimura, S.H.; Takeyasu, K.; Inoue, T.; Saito, H. Synthetic RNA-Protein Complex Shaped Like an Equilateral Triangle. Nat. Nanotechnol. 2011, 6, 116–120. [Google Scholar] [CrossRef]
- Shibata, T.; Fujita, Y.; Ohno, H.; Suzuki, Y.; Hayashi, K.; Komatsu, K.R.; Kawasaki, S.; Hidaka, K.; Yonehara, S.; Sugiyama, H.; et al. Protein-Driven RNA Nanostructured Devices That Function in Vitro and Control Mammalian Cell Fate. Nat. Commun. 2017, 8, 540. [Google Scholar] [CrossRef]
- Lee, H.; Lytton-Jean, A.K.; Chen, Y.; Love, K.T.; Park, A.I.; Karagiannis, E.D.; Sehgal, A.; Querbes, W.; Zurenko, C.S.; Jayaraman, M.; et al. Molecularly Self-Assembled Nucleic Acid Nanoparticles for Targeted in Vivo Sirna Delivery. Nat. Nanotechnol. 2012, 7, 389–393. [Google Scholar] [CrossRef]
- Halman, J.R.; Satterwhite, E.; Roark, B.; Chandler, M.; Viard, M.; Ivanina, A.; Bindewald, E.; Kasprzak, W.K.; Panigaj, M.; Bui, M.N.; et al. Functionally-Interdependent Shape-Switching Nanoparticles with Controllable Properties. Nucleic Acids Res. 2017, 45, 2210–2220. [Google Scholar] [CrossRef]
- Hong, E.P.; JHalman, R.; Shah, A.B.; Khisamutdinov, E.F.; Dobrovolskaia, M.A.; Afonin, K.A. Structure and Composition Define Immunorecognition of Nucleic Acid Nanoparticles. Nano Lett. 2018, 18, 4309–4321. [Google Scholar] [CrossRef]
- Douglas, S.M.; Bachelet, I.; Church, G.M. A Logic-Gated Nanorobot for Targeted Transport of Molecular Payloads. Science 2012, 335, 831–834. [Google Scholar] [CrossRef]
- Bindewald, E.; Afonin, K.A.; Viard, M.; Zakrevsky, P.; Kim, T.; Shapiro, B.A. Multistrand Structure Prediction of Nucleic Acid Assemblies and Design of RNA Switches. Nano Lett. 2016, 16, 1726–1735. [Google Scholar] [CrossRef]
- Roark, B.K.; Tan, L.A.; Ivanina, A.; Chandler, M.; Castaneda, J.; Kim, H.S.; Jawahar, S.; Viard, M.; Talic, S.; Wustholz, K.L.; et al. Fluorescence Blinking as an Output Signal for Biosensing. ACS Sens. 2016, 1, 1295–1300. [Google Scholar] [CrossRef] [PubMed]
- Andersen, E.S.; Dong, M.; Nielsen, M.M.; Jahn, K.; Subramani, R.; Mamdouh, W.; Golas, M.M.; Sander, B.; Stark, H.; Oliveira, C.L.; et al. Self-Assembly of a Nanoscale DNA Box with a Controllable Lid. Nature 2009, 459, 73–76. [Google Scholar] [CrossRef]
- Binzel, D.W.; Shu, Y.; Li, H.; Sun, M.; Zhang, Q.; Shu, D.; Guo, B.; Guo, P. Specific Delivery of Mirna for High Efficient Inhibition of Prostate Cancer by RNA Nanotechnology. Mol. Ther. 2016, 24, 1267–1277. [Google Scholar] [CrossRef]
- Shu, D.; Li, H.; Shu, Y.; Xiong, G.; Carson, W.E.; Haque, F.; Xu, R.; Guo, P. Systemic Delivery of Anti-Mirna for Suppression of Triple Negative Breast Cancer Utilizing RNA Nanotechnology. ACS Nano 2015, 9, 9731–9740. [Google Scholar] [CrossRef]
- Feng, L.; Li, S.K.; Liu, H.; Liu, C.Y.; LaSance, K.; Haque, F.; Shu, D.; Guo, P. Ocular Delivery of Prna Nanoparticles: Distribution and Clearance after Subconjunctival Injection. Pharm. Res. 2014, 31, 1046–1058. [Google Scholar] [CrossRef]
- Shu, Y.; Shu, D.; Haque, F.; Guo, P. Fabrication of Prna Nanoparticles to Deliver Therapeutic Rnas and Bioactive Compounds into Tumor Cells. Nat. Protoc. 2013, 8, 1635–1659. [Google Scholar] [CrossRef]
- Afonin, K.A.; Viard, M.; Martins, A.N.; Lockett, S.J.; Maciag, A.E.; Freed, E.O.; Heldman, E.; Jaeger, L.; Blumenthal, R.; Shapiro, B.A. Activation of Different Split Functionalities on Re-Association of RNA-DNA Hybrids. Nat. Nanotechnol. 2013, 8, 296–304. [Google Scholar] [CrossRef] [PubMed]
- Kirill, A.A.; Grabow, W.W.; Walker, F.M.; Bindewald, E.; Dobrovolskaia, M.A.; Shapiro, B.A.; Jaeger, L. Design and Self-Assembly of Sirna-Functionalized RNA Nanoparticles for Use in Automated Nanomedicine. Nat. Protoc. 2011, 6, 2022–2034. [Google Scholar]
- Sajja, S.; Chandler, M.; Fedorov, D.; Kasprzak, W.K.; Lushnikov, A.; Viard, M.; Shah, A.; Dang, D.; Dahl, J.; Worku, B.; et al. Dynamic Behavior of RNA Nanoparticles Analyzed by Afm on a Mica/Air Interface. Langmuir 2018, 34, 15099–15108. [Google Scholar] [CrossRef]
- Afonin, K.A.; Bindewald, E.; Yaghoubian, A.J.; Voss, N.; Jacovetty, E.; Shapiro, B.A.; Jaeger, L. In Vitro Assembly of Cubic RNA-Based Scaffolds Designed in Silico. Nat. Nanotechnol. 2010, 5, 676–682. [Google Scholar] [CrossRef] [PubMed]
- Davis, J.H.; Tonelli, M.; Scott, L.G.; Jaeger, L.; Williamson, J.R.; Butcher, S.E. RNA Helical Packing in Solution: Nmr Structure of a 30 Kda Gaaa Tetraloop-Receptor Complex. J. Mol. Biol. 2005, 351, 371–382. [Google Scholar] [CrossRef]
- Dibrov, S.M.; McLean, J.; Parsons, J.; Hermann, T. Self-Assembling RNA Square. Proc. Natl. Acad. Sci. USA 2011, 108, 6405–6408. [Google Scholar] [CrossRef]
- Manaia, E.B.; Abuçafy, M.P.; Chiari-Andréo, B.G.; Silva, B.L.; Junior JA, O.; Chiavacci, L.A. Physicochemical Characterization of Drug Nanocarriers. Int. J. Nanomed. 2017, 12, 4991–5011. [Google Scholar] [CrossRef] [PubMed]
- Xavier, P.L.; Chandrasekaran, A.R. DNA-Based Construction at the Nanoscale: Emerging Trends and Applications. Nanotechnology 2018, 29, 062001. [Google Scholar] [CrossRef]
- Ukraintsev, V.; Banke, B. Review of Reference Metrology for Nanotechnology: Significance, Challenges, and Solutions. J. Micro Nanolithogr. MEMS MOEMS 2012, 11, 011010. [Google Scholar] [CrossRef]
- Bald, I.; Keller, A. Molecular Processes Studied at a Single-Molecule Level Using DNA Origami Nanostructures and Atomic Force Microscopy. Molecular 2014, 19, 13803–13823. [Google Scholar] [CrossRef]
- Song, Y.; Chen, S. Janus Nanoparticles: Preparation, Characterization, and Applications. Chem. Asian J. 2014, 9, 418–430. [Google Scholar] [CrossRef]
- Svergun, D.I.; Koch, M.H. Small-Angle Scattering Studies of Biological Macromolecules in Solution. Rep. Prog. Phys. 2013, 66, 1735–1782. [Google Scholar] [CrossRef]
- Jacques, D.A.; Trewhella, J. Small-Angle Scattering for Structural Biology-Expanding the Frontier While Avoiding the Pitfalls. Protein Sci. 2010, 19, 642–657. [Google Scholar] [CrossRef]
- Vestergaard, B.; Sayers, Z. Investigating Increasingly Complex Macromolecular Systems with Small-Angle X-Ray Scattering. IUCrJ 2014, 1, 523–529. [Google Scholar] [CrossRef]
- Heller, W.T. Small-Angle Neutron Scattering and Contrast Variation: A Powerful Combination for Studying Biological Structures. Acta Crystallogr. Sect. D Biol. Crystallogr. 2010, 66, 1213–1217. [Google Scholar] [CrossRef]
- Heller, W.T.; Littrell, K.C. Small-Angle Neutron Scattering for Molecular Biology: Basics and Instrumentation. Micro Nano Technol. Bioanal. Methods Protoc. 2009, 544, 293–305. [Google Scholar]
- Hong, E.; Halman, J.R.; Shah, A.; Cedrone, E.; Truong, N.; Afonin, K.A.; Dobrovolskaia, M.A. Toll-Like Receptor-Mediated Recognition of Nucleic Acid Nanoparticles (Nanps) in Human Primary Blood Cells. Molecules 2019, 24, 1094. [Google Scholar] [CrossRef]
- Chandler, M.; Afonin, K.A. Smart-Responsive Nucleic Acid Nanoparticles (Nanps) with the Potential to Modulate Immune Behavior. Nanomaterials 2019, 9, 611. [Google Scholar] [CrossRef]
- Rychahou, P.; Haque, F.; Shu, Y.; Zaytseva, Y.; Weiss, H.L.; Lee, E.Y.; Mustain, W.; Valentino, J.; Guo, P.; Evers, B.M. Delivery of RNA Nanoparticles into Colorectal Cancer Metastases Following Systemic Administration. ACS Nano 2015, 9, 1108–1116. [Google Scholar] [CrossRef] [PubMed]
- Grabow, W.W.; Zakrevsky, P.; Afonin, K.A.; Chworos, A.; Shapiro, B.A.; Jaeger, L. Self-Assembling RNA Nanorings Based on RNAi/Ii Inverse Kissing Complexes. Nano Lett. 2011, 11, 878–887. [Google Scholar] [CrossRef]
- Wu, P.W.; Yu, Y.; McGhee, C.E.; Tan, L.H.; Lu, Y. Applications of Synchrotron-Based Spectroscopic Techniques in Studying Nucleic Acids and Nucleic Acid-Functionalized Nanomaterials. Adv. Mater. 2014, 26, 7849–7872. [Google Scholar] [CrossRef] [PubMed]
- Dervichian, D.G.; Fournet, G.; Guinier, A. X-Ray Scattering Study of the Modifications Which Certain Proteins Undergo. Biochim. Biophys. Acta 1952, 8, 145–149. [Google Scholar] [CrossRef]
- Glatter, O.; Kratky, O. Small Angle X-Ray Scattering; Academic Press: London, UK; New York, NY, USA, 1982. [Google Scholar]
- Franke, D.; Petoukhov, M.V.; Konarev, P.V.; Panjkovich, A.; Tuukkanen, A.; Mertens, H.D.T.; Kikhney, A.G.; Hajizadeh, N.R.; Franklin, J.M.; Jeffries, C.M.; et al. Atsas 2.8: A Comprehensive Data Analysis Suite for Small-Angle Scattering from Macromolecular Solutions. J. Appl. Crystallogr. 2017, 50, 1212–1225. [Google Scholar] [CrossRef]
- Fang, X.; Wang, J.; O’Carroll, I.P.; Mitchell, M.; Zuo, X.; Wang, Y.; Yu, P.; Liu, Y.; Rausch, J.W.; Dyba, M.A.; et al. An Unusual Topological Structure of the Hiv-1 Rev Response Element. Cell 2013, 155, 594–605. [Google Scholar] [CrossRef]
- Hennig, J.; Wang, I.; Sonntag, M.; Gabel, F.; Sattler, M. Combining Nmr and Small Angle X-Ray and Neutron Scattering in the Structural Analysis of a Ternary Protein-RNA Complex. J. Biomol. NMR 2013, 56, 17–30. [Google Scholar] [CrossRef] [PubMed]
- Rambo, R.P.; Tainer, J.A. Bridging the Solution Divide: Comprehensive Structural Analyses of Dynamic RNA, DNA, and Protein Assemblies by Small-Angle X-Ray Scattering. Curr. Opin. Struct. Biol. 2010, 20, 128–137. [Google Scholar] [CrossRef]
- Rambo, R.P.; Tainer, J.A. Improving Small-Angle X-Ray Scattering Data for Structural Analyses of the RNA World. RNA 2010, 16, 638–646. [Google Scholar] [CrossRef] [PubMed]
- Etoukhov, M.V.; Konarev, P.V.; Kikhney, A.G.; Svergun, D.I. Atsas 2.1—Towards Automated and Web-Supported Small-Angle Scattering Data Analysis. J. Appl. Crystallogr. 2007, 40, S223–S228. [Google Scholar] [CrossRef]
- Svergun, D.I. Restoring low resolution structure of biological macromolecules from solution scattering using simulated annealing. Biophys. J. 1999, 76, 2879–2886. [Google Scholar] [CrossRef]
- Yang, S. Methods for Saxs-Based Structure Determination of Biomolecular Complexes. Adv. Mater. 2014, 26, 7902–7910. [Google Scholar] [CrossRef] [PubMed]
- Maguire, C.M.; Rosslein, M.; Wick, P.; Prina-Mello, A. Characterisation of Particles in Solution—A Perspective on Light Scattering and Comparative Technologies. Sci. Technol. Adv. Mater. 2018, 19, 732–745. [Google Scholar] [CrossRef]
- Trewhella, J. Small-Angle Scattering and 3d Structure Interpretation. Curr. Opin. Struct. Biol. 2016, 40, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Petoukhov, M.V.; Svergun, D.I. Ambiguity Assessment of Small-Angle Scattering Curves from Monodisperse Systems. Acta Crystallogr. Sect. D 2015, 71, 1051–1058. [Google Scholar] [CrossRef]
- Mahieu, E.; Gabel, F. Biological Small-Angle Neutron Scattering: Recent Results and Development. Acta Crystallogr. D Struct. Biol. 2018, 74, 715–726. [Google Scholar] [CrossRef] [PubMed]
- Ramakrishnan, V.; Capel, M.; Kjeldgaard, M.; Engelman, D.M.; Moore, P.B. Positions of Protein-S14, Protein-S18 and Protein-S20 in the 30-S Ribosomal-Subunit of Escherichia-Coli. J. Mol. Biol. 1984, 174, 265–284. [Google Scholar] [CrossRef]
- Ramakrishnan, V.R.; Yabuki, S.; Sillers, I.Y.; Schindler, D.G.; Engelman, D.M.; Moore, P.B. Positions of Proteins S6, S11 and S15 in the 30-S Ribosomal-Subunit of Escherichia-Coli. J. Mol. Biol. 1981, 153, 739–760. [Google Scholar] [CrossRef]
- Ramakrishnan, V.; Engelman, D.M.; Moore, P.B. 3-Dimensional Localization of 12 Proteins of the 30s Ribosome of Escherichia-Coli. Fed. Proc. 1980, 39, 2122. [Google Scholar]
- Capel, M.S.; Engelman, D.M.; Freeborn, B.R.; Kjeldgaard, M.; Langer, J.A.; Ramakrishnan, V.; Schindler, D.G.; Schneider, D.K.; Schoenborn, B.P.; Sillers, I.Y.; et al. A Complete Mapping of the Proteins in the Small Ribosomal-Subunit of Escherichia-Coli. Science 1987, 238, 1403–1406. [Google Scholar] [CrossRef]
- Svergun, D.I.; Koch, M.H.J.; Pedersen, J.S.; Serdyuk, I.N. Structural Model of the 50-S Subunit of Escherichia-Coli Ribosomes from Solution Scattering.2. Neutron-Scattering Study. J. Mol. Biol. 1994, 240, 78–86. [Google Scholar] [CrossRef]
- Svergun, D.I.; Pedersen, J.S. Propagating Errors in Small-Angle Scattering Data Treatment. J. Appl. Crystallogr. 1994, 27, 241–248. [Google Scholar] [CrossRef]
- Svergun, D.I.; Pedersen, J.S.; Serdyuk, I.N.; Koch, M.H.J. Solution Scattering from 50s Ribosomal-Subunit Resolves Inconsistency between Electron-Microscopic Models. Proc. Natl. Acad. Sci. USA 1994, 91, 11826–11830. [Google Scholar] [CrossRef]
- Yang, W.; Lu, J.; Gilbert, E.P.; Knott, R.; He, L.; Cheng, W. Probing Soft Corona Structures of DNA-Capped Nanoparticles by Small Angle Neutron Scattering. J. Phys. Chem. C 2015, 119, 18773–18778. [Google Scholar] [CrossRef]
- Zuo, X.B.; Wang, J.B.; Yu, P.; Eyler, D.; Xu, H.; Starich, M.R.; Tiede, D.M.; Simon, A.E.; Kasprzak, W.; Schwieters, C.D.; et al. Solution Structure of the Cap-Independent Translational Enhancer and Ribosome-Binding Element in the 3′Utr of Turnip Crinkle Virus. Proc. Natl. Acad. Sci. USA 2010, 107, 1385–1390. [Google Scholar] [CrossRef]
- Wang, J.B.; Zuo, X.B.; Yu, P.; Xu, H.; Starich, M.R.; Tiede, D.M.; Shapiro, B.A.; Schwieters, C.D.; Wang, Y.X. A Method for Helical RNA Global Structure Determination in Solution Using Small-Angle X-Ray Scattering and Nmr Measurements. J. Mol. Biol. 2009, 393, 717–734. [Google Scholar] [CrossRef]
- Sosnick, T.; Pan, T.; Fang, X.W.; Shelton, V.; Thiyagarajan, P.; Littrel, K. Metal Ions and the Thermodynamics and Kinetics of Tertiary RNA Folding. Indian J. Chem. Sect. A Inorg. Bio Inorg. Phys. Theor. Anal. Chem. 2002, 41, 54–64. [Google Scholar]
- Fang, X.W.; Littrell, K.; Yang, X.; Henderson, S.J.; Siefert, S.; Thiyagarajan, P.; Pan, T.; Sosnick, T.R. Mg2+-Dependent Compaction and Folding of Yeast Trna(Phe) and the Catalytic Domain of the B-Subtilis Rnase P RNA Determined by Small-Angle X-Ray Scattering. Biochemistry 2000, 39, 11107–11113. [Google Scholar] [CrossRef]
- Serganov, A.; Nudler, E. A Decade of Riboswitches. Cell 2013, 152, 17–24. [Google Scholar] [CrossRef]
- Stoddard, C.D.; Montange, R.K.; Hennelly, S.P.; Rambo, R.P.; Sanbonmatsu, K.Y.; Batey, R.T. Free State Conformational Sampling of the Sam-I Riboswitch Aptamer Domain. Structure 2010, 18, 787–797. [Google Scholar] [CrossRef]
- Fernandez-Castanon, J.; Bomboi, F.; Rovigatti, L.; Zanatta, M.; Paciaroni, A.; Comez, L.; Porcar, L.; Jafta, C.J.; Fadda, G.C.; Bellini, T.; et al. Small-Angle Neutron Scattering and Molecular Dynamics Structural Study of Gelling DNA Nanostars. J. Chem. Phys. 2016, 145, 084910. [Google Scholar] [CrossRef] [PubMed]
- Bruetzel, L.K.; Gerling, T.; Sedlak, S.M.; Walker, P.U.; Zheng, W.; Dietz, H.; Lipfert, J. Conformational Changes and Flexibility of DNA Devices Observed by Small-Angle X-Ray Scattering. Nano Lett. 2016, 16, 4871–4879. [Google Scholar] [CrossRef]
- Svergun, D.; Barberato, C.; Koch, M.H.J. Crysol—A Program to Evaluate X-Ray Solution Scattering of Biological Macromolecules from Atomic Coordinates. J. Appl. Crystallogr. 1995, 28, 768–773. [Google Scholar] [CrossRef]
- Bruetzel, L.K.; Walker, P.U.; Gerling, T.; Dietz, H.; Lipfert, J. Time-Resolved Small-Angle X-Ray Scattering Reveals Millisecond Transitions of a DNA Origami Switch. Nano Lett. 2018, 18, 2672–2676. [Google Scholar] [CrossRef]
- Afonin, K.A.; Kireeva, M.; Grabow, W.W.; Kashlev, M.; Jaeger, L.; Shapiro, B.A. Co-Transcriptional Assembly of Chemically Modified RNA Nanoparticles Functionalized with Sirnas. Nano Lett. 2012, 12, 5192–5195. [Google Scholar] [CrossRef]
- Afonin, K.A.; Viard, M.; Tedbury, P.; Bindewald, E.; Parlea, L.; Howington, M.; Valdman, M.; Johns-Boehme, A.; Brainerd, C.; Freed, E.O.; et al. The Use of Minimal RNA Toeholds to Trigger the Activation of Multiple Functionalities. Nano Lett. 2016, 16, 1746–1753. [Google Scholar] [CrossRef]
- Yingling, Y.G.; Shapiro, B.A. Computational Design of an RNA Hexagonal Nanoring and an RNA Nanotube. Nano Lett. 2007, 7, 2328–2334. [Google Scholar] [CrossRef] [PubMed]
- Ke, W.; Hong, E.; Saito, R.F.; Rangel, M.C.; Wang, J.; Viard, M.; Richardson, M.; Khisamutdinov, E.F.; Panigaj, M.; Dokholyan, N.V.; et al. RNA-DNA Fibers and Polygons with Controlled Immunorecognition Activate Rnai, Fret and Transcriptional Regulation of Nf-Kappab in Human Cells. Nucleic Acids Res. 2018, 47, 1350–1361. [Google Scholar] [CrossRef] [PubMed]
- Rackley, L.; Stewart, J.M.; Salotti, J.; Krokhotin, A.; Shah, A.; Halman, J.; Juneja, R.; Smollett, J.; Roark, B.; Viard, M.; et al. RNA Fibers as Optimized Nanoscaffolds for Sirna Coordination and Reduced Immunological Recognition. Adv. Funct. Mater. 2018, 28, 1805959. [Google Scholar] [CrossRef]
- Yourston, L.E.; Lushnikov, A.Y.; Shevchenko, O.A.; Afonin, K.A.; Krasnoslobodtsev, A.V. First Step Towards Larger DNA-Based Assemblies of Fluorescent Silver Nanoclusters: Template Design and Detailed Characterization of Optical Properties. Nanomaterials 2019, 9, 613. [Google Scholar] [CrossRef]
- Cruz-Acuna, M.; Halman, J.R.; Afonin, K.A.; Dobson, J.; Rinaldi, C. Magnetic Nanoparticles Loaded with Functional RNA Nanoparticles. Nanoscale 2018, 10, 17761–17770. [Google Scholar] [CrossRef]
- Gupta, K.; Mattingly, S.J.; Knipp, R.J.; Afonin, K.A.; Viard, M.; Bergman, J.T.; Stepler, M.; Nantz, M.H.; Puri, A.; Shapiro, B.A. Oxime Ether Lipids Containing Hydroxylated Head Groups Are More Superior Sirna Delivery Agents Than Their Nonhydroxylated Counterparts. Nanomedicine 2015, 10, 2805–2818. [Google Scholar] [CrossRef]
- Juneja, R.; Lyles, Z.; Vadarevu, H.; Afonin, K.A.; Vivero-Escoto, J.L. Multimodal Polysilsesquioxane Nanoparticles for Combinatorial Therapy and Gene Delivery in Triple-Negative Breast Cancer. ACS Appl. Mater. Interfaces 2019, 11, 12308–12320. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.; Afonin, K.A.; Viard, M.; Koyfman, A.Y.; Sparks, S.; Heldman, E.; Grinberg, S.; Linder, C.; Blumenthal, R.P.; Shapiro, B.A. In Silico, in Vitro, and in Vivo Studies Indicate the Potential Use of Bolaamphiphiles for Therapeutic Sirnas Delivery. Mol. Ther. Nucleic Acids 2013, 2, e80. [Google Scholar] [CrossRef] [PubMed]
- Gupta, K.; Afonin, K.A.; Viard, M.; Herrero, V.; Kasprzak, W.; Kagiampakis, I.; Kim, T.; Koyfman, A.Y.; Puri, A.; Stepler, M.; et al. Bolaamphiphiles as Carriers for Sirna Delivery: From Chemical Syntheses to Practical Applications. J. Control. Release 2015, 213, 142–151. [Google Scholar] [CrossRef] [PubMed]
- Dakwar, G.R.; Hammad, I.A.; Popov, M.; Linder, C.; Grinberg, S.; Heldman, E.; Stepensky, D. Delivery of Proteins to the Brain by Bolaamphiphilic Nano-Sized Vesicles. J. Control. Release 2012, 160, 315–321. [Google Scholar] [CrossRef] [PubMed]
- Popov, M.; Grinberg, S.; Linder, C.; Waner, T.; Levi-Hevroni, B.; Deckelbaum, R.J.; Heldman, E. Site-Directed Decapsulation of Bolaamphiphilic Vesicles with Enzymatic Cleavable Surface Groups. J. Control. Release 2012, 160, 306–314. [Google Scholar] [CrossRef]
- Rudorf, S.; Radler, J.O. Self-Assembly of Stable Monomolecular Nucleic Acid Lipid Particles with a Size of 30 Nm. J. Am. Chem. Soc. 2012, 134, 11652–11658. [Google Scholar] [CrossRef]
| Technique | Parameters Analyzed/Advantages | Limitations |
|---|---|---|
| Solid State/Static Techniques | ||
| Crystallography [28] | High resolution molecular structure Broad Mass range Model building is well-developed | Static crystalline state structure; may not reflect dynamic or flexible structures Sample must form a crystal |
| Scanning Electron Microscopy (SEM) [28,29] | Particle size, size distributions, shape Sample preparation is relatively simple Structure in native state Allows analysis of hydrated materials without fixation, drying, freezing, or coating | Limited to larger molecules (up to ~200 nm) Highly dependent on electron microscopy (EM) techniques and access to costly equipment Cannot be used on certain biological materials due to degradation caused by the electron beam Low resolution |
| Transmission Electron Microscopy (TEM) [28] | Particle size, size distributions, shape Produces high resolution images that can provide information about structure and elemental composition High resolution TEM has Å resolution | Harsh chemical treatment of the sample Statistics are highly dependent on technique 2D images Samples need to be dehydrated, collected on metal mesh, and stained Small viewing section of sample |
| Atomic Force Microscopy (AFM) [30,31] | Provides a three-dimensional surface profile Minimal sample preparation Shown to give true atomic resolution in ultra-high vacuum (UHV) and, more recently, in liquid environments | Can only image a maximum height on the order of 10–20 micrometers and a maximum scanning area of about 150 × 150 micrometers Images can also be affected by hysteresis of the piezoelectric material Possibility of image artifacts Must immobilize the sample onto a substrate |
| Solution State/Native Techniques | ||
| Static Light Scattering (SLS)/Dynamic Light Scattering (DLS)/ Zeta Potential [28] | Hydrodynamic particle size, size distributions, surface charge Sample volumes are small (μL) Particle size across a broad range (~0.1 nm to ~10 µm) Allows measurements under physiological conditions | Can only measure solid particles, polymers, and proteins dispersed in a solvent or emulsions Light absorption by the dispersant or sample can interfere with detection Concentration dependent Samples need to be homogenous Little shape information; size of particles can be under or over-estimated Dust/traces of agglomerates can interfere with results Cannot distinguish between similarly sized populations without coupling to a separation |
| Nuclear Magnetic Resonance (NMR) [32] | High resolution structure 3D structure in solution Dynamics can be studied | High sample purity and concentration required Computational simulation is challenging Sample MWs typically limited to below 40–50 kDa Water soluble samples |
| Small-Angle X-ray Scattering (SAXS) [28,33] | Structure in native state Particle size and shape, size distribution, particle interactions and interatomic distances: some parameters determined with sub Å precision Small sample size (10–25 μL solution; 0.01–10 mg/mL) Highflux synchrotron sources allow for time-resolved, kinetic studies | Low-resolution shape information interpreted from interatomic distance distributions Highest level of structural information requires pure, monodisperse samples |
| Small-Angle Neutron Scattering (SANS) [28,33] | Amenable to contrast variation Sensitive to fluctuations in the nuclei density of the sample | Experiments require access to user facilities with appropriate neutron source and instrumentation Flux of neutron source is intrinsically low |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oliver, R.C.; Rolband, L.A.; Hutchinson-Lundy, A.M.; Afonin, K.A.; Krueger, J.K. Small-Angle Scattering as a Structural Probe for Nucleic Acid Nanoparticles (NANPs) in a Dynamic Solution Environment. Nanomaterials 2019, 9, 681. https://doi.org/10.3390/nano9050681
Oliver RC, Rolband LA, Hutchinson-Lundy AM, Afonin KA, Krueger JK. Small-Angle Scattering as a Structural Probe for Nucleic Acid Nanoparticles (NANPs) in a Dynamic Solution Environment. Nanomaterials. 2019; 9(5):681. https://doi.org/10.3390/nano9050681
Chicago/Turabian StyleOliver, Ryan C., Lewis A. Rolband, Alanna M. Hutchinson-Lundy, Kirill A. Afonin, and Joanna K. Krueger. 2019. "Small-Angle Scattering as a Structural Probe for Nucleic Acid Nanoparticles (NANPs) in a Dynamic Solution Environment" Nanomaterials 9, no. 5: 681. https://doi.org/10.3390/nano9050681
APA StyleOliver, R. C., Rolband, L. A., Hutchinson-Lundy, A. M., Afonin, K. A., & Krueger, J. K. (2019). Small-Angle Scattering as a Structural Probe for Nucleic Acid Nanoparticles (NANPs) in a Dynamic Solution Environment. Nanomaterials, 9(5), 681. https://doi.org/10.3390/nano9050681





