Inkjet Printing Functionalization of SOFC LSCF Cathodes
Abstract
1. Introduction
2. Infiltration Strategies
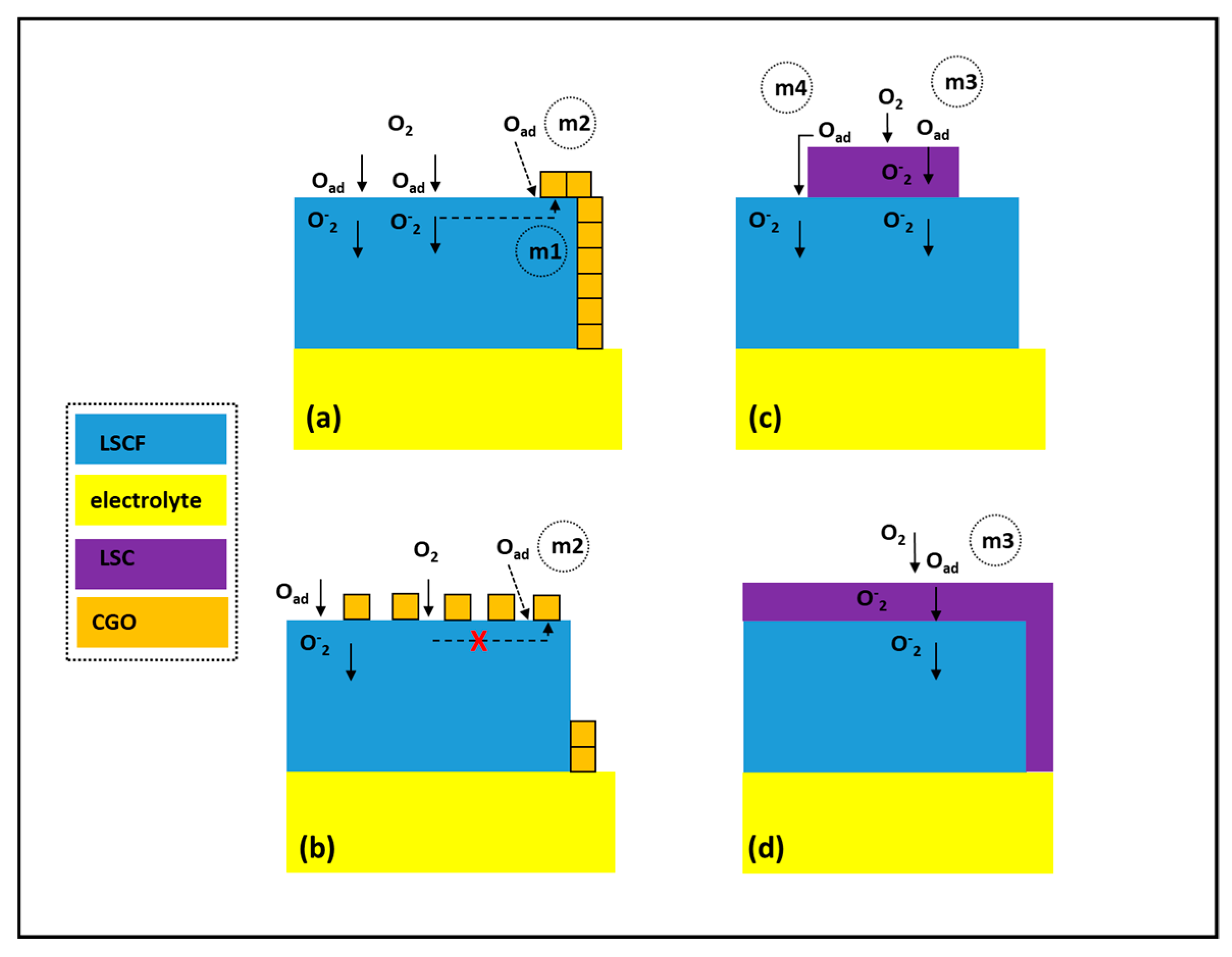
3. Infiltration of Ion Conductive Phase
4. Infiltration of MIEC Phase
5. Methodology
6. Experimental
7. Results and Discussions
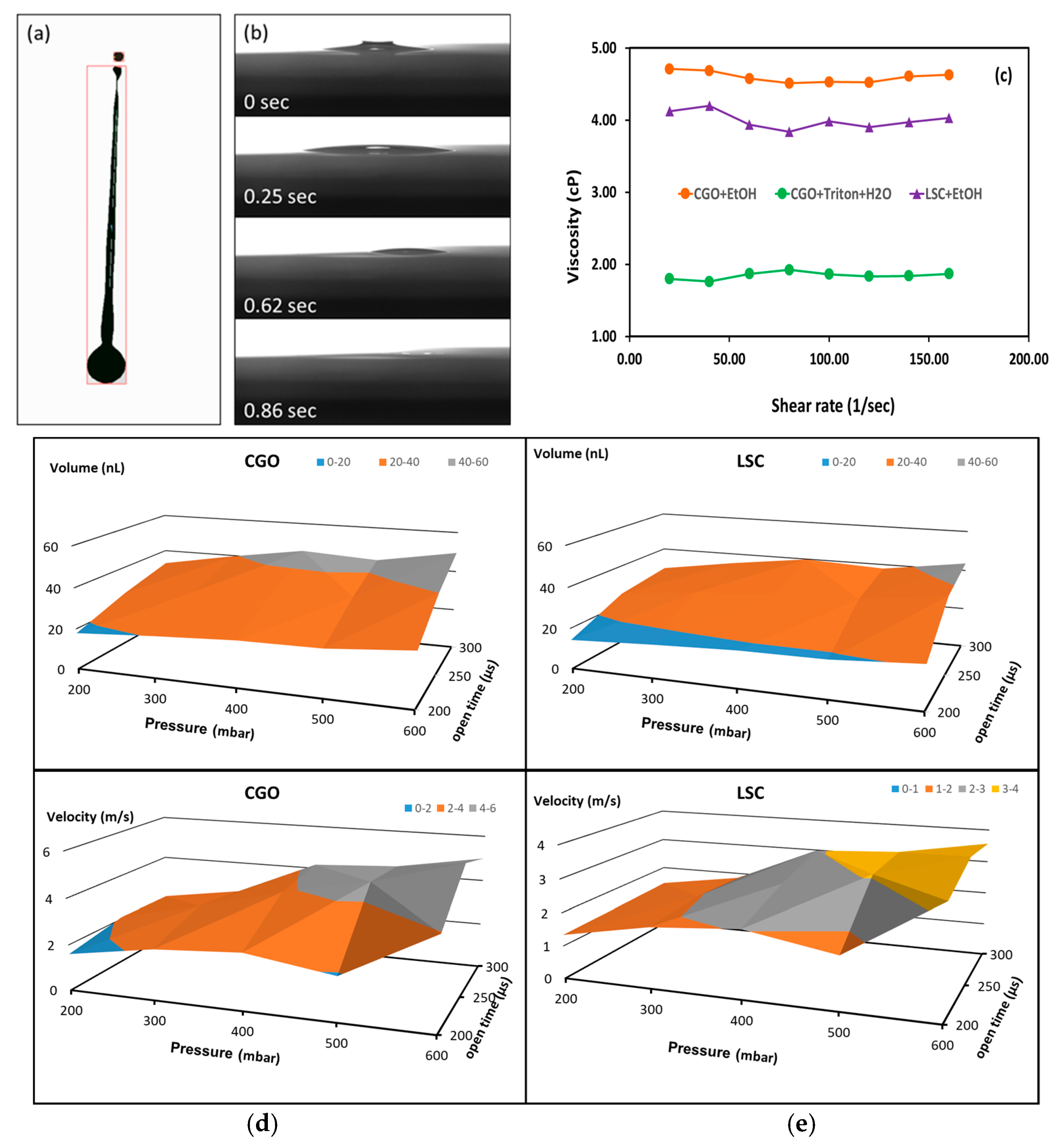
7.1. Inkjet Printing Infiltration
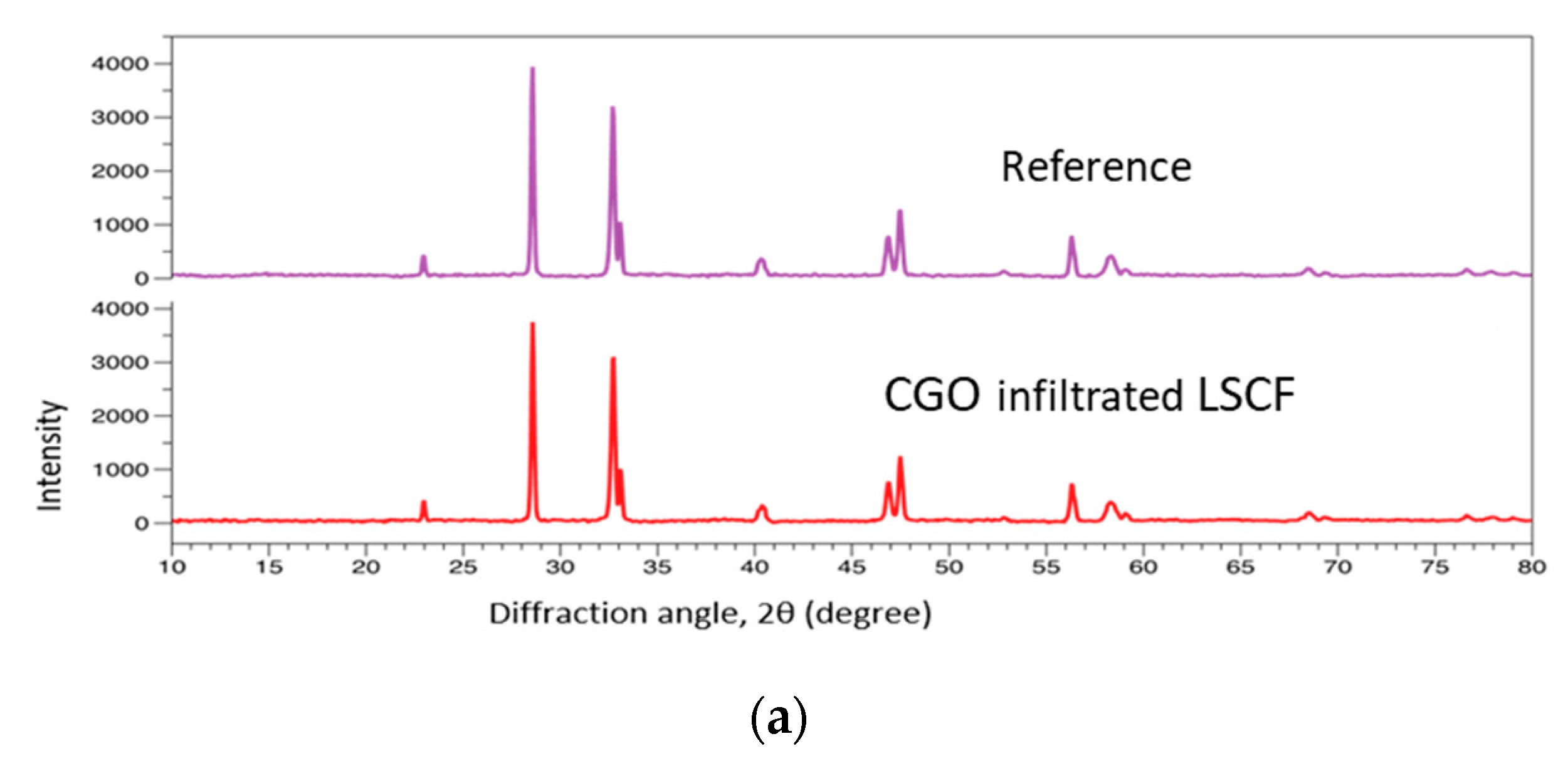
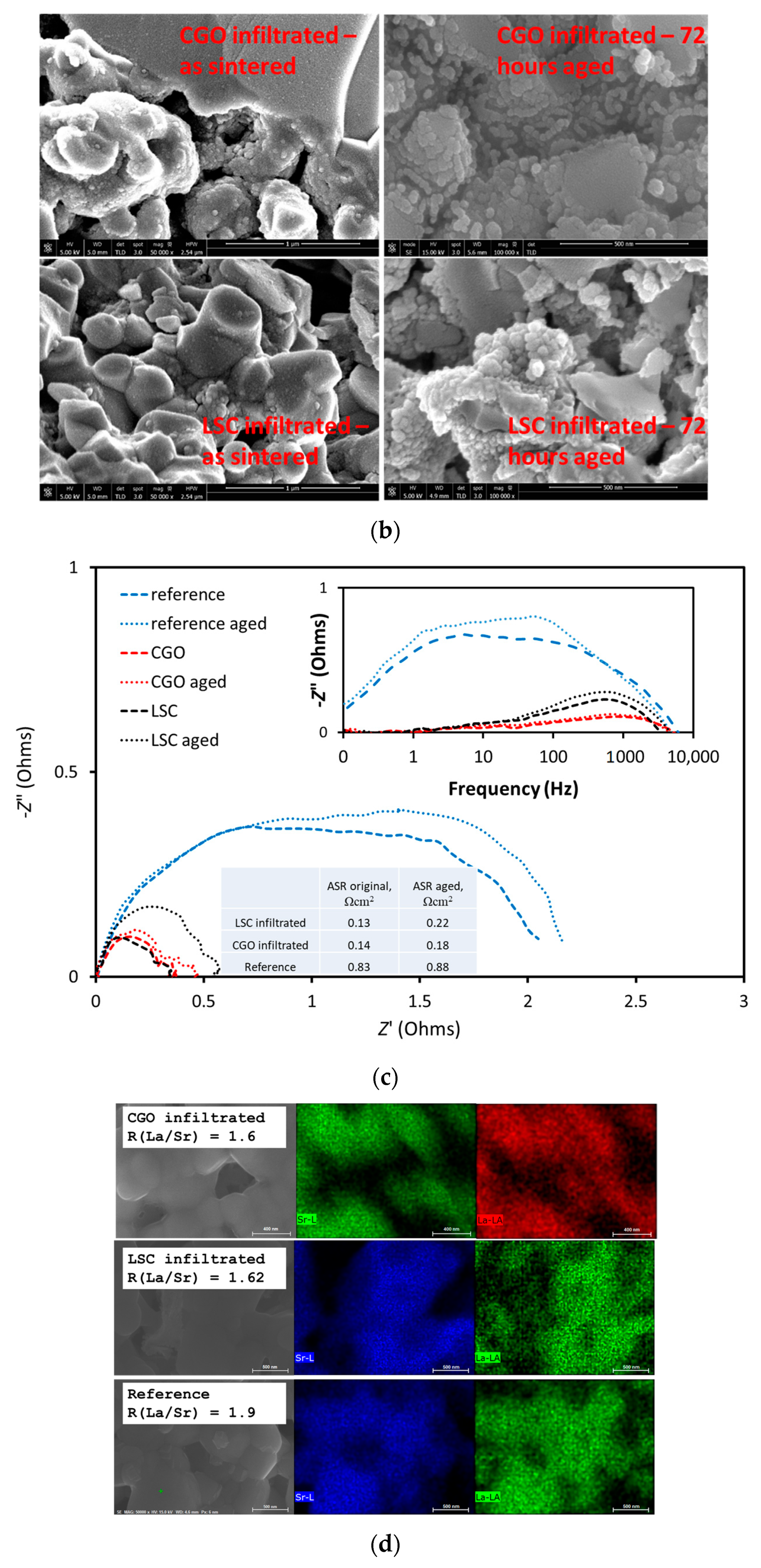
7.2. Characterization
8. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Behling, N. Fuel Cells Current Technology Challenges and Future Research Needs, 1st ed.; Elsevier: Amsterdam, The Netherlands, 2012; ISBN 9780444563262. [Google Scholar]
- Singhal, S.C. Solid oxide fuel cells for stationary, mobile, and military applications. Solid State Ion. 2002, 152, 405–410. [Google Scholar] [CrossRef]
- Liu, M.; Lynch, M.E.; Blinn, K.; Alamgir, F.M.; Choi, Y. Rational SOFC material design: New advances and tools. Mater. Today 2011, 14, 534–546. [Google Scholar] [CrossRef]
- Steele, B.C.H. Material science and engineering: The enabling technology for the commercialisation of fuel cell systems. J. Mater. Sci. 2001, 36, 1053–1068. [Google Scholar] [CrossRef]
- Adler, S.B. Factors Governing Oxygen Reduction in Solid Oxide Fuel Cell Cathodes. Chem. Rev. 2004, 104, 4791–4843. [Google Scholar] [CrossRef]
- Ding, D.; Li, X.; Lai, S.Y.; Gerdes, K.; Liu, M. Enhancing SOFC cathode performance by surface modification through infiltration. Energy Environ. Sci. 2014, 7, 552–575. [Google Scholar] [CrossRef]
- Jiang, S.P. Nanoscale and nano-structured electrodes of solid oxide fuel cells by infiltration: Advances and challenges. Int. J. Hydrog. Energy 2012, 37, 449–470. [Google Scholar] [CrossRef]
- Guo, S.; Wu, H.; Puleo, F.; Liotta, L.F. B-Site Metal (Pd, Pt, Ag, Cu, Zn, Ni) Promoted La1−xSrxCo1−yFeyO3–δPerovskite Oxides as Cathodes for IT-SOFCs. Catalysts 2015, 5, 366–391. [Google Scholar] [CrossRef]
- Kilner, J.A.; Burriel, M. Materials for Intermediate-Temperature Solid-Oxide Fuel Cells. Annu. Rev. Mater. Res. 2014, 44, 365–393. [Google Scholar] [CrossRef]
- Knöfel, C.; Wang, H.-J.; Thydén, K.T.S.; Mogensen, M. Modifications of interface chemistry of LSM–YSZ composite by ceria nanoparticles. Solid State Ion. 2011, 195, 36–42. [Google Scholar] [CrossRef]
- Howe, K.S.; Hanifi, A.R.; Kendall, K.; Zazulak, M.; Etsell, T.H.; Sarkar, P. Performance of microtubular SOFCs with infiltrated electrodes under thermal cycling. Int. J. Hydrog. Energy 2013, 38, 1058–1067. [Google Scholar] [CrossRef]
- Buyukaksoy, A.; Petrovsky, V.; Dogan, F. Stability and Performance of Solid Oxide Fuel Cells with Nanocomposite Electrodes. J. Electrochem. Soc. 2012, 159, B666–B669. [Google Scholar] [CrossRef]
- Kiebach, R.; Knöfel, C.; Bozza, F.; Klemensø, T.; Chatzichristodoulou, C. Infiltration of ionic-, electronic- and mixed-conducting nano particles into La0.75Sr0.25MnO3–Y0.16Zr0.84O2 cathodes—A comparative study of performance enhancement and stability at different temperatures. J. Power Sources 2013, 228, 170–177. [Google Scholar] [CrossRef]
- Lou, X.; Wang, S.; Liu, Z.; Yang, L.; Liu, M. Improving La0.6Sr0.4Co0.2Fe0.8O3−δ cathode performance by infiltration of a Sm0.5Sr0.5CoO3—δ coating. Solid State Ion. 2009, 180, 1285–1289. [Google Scholar] [CrossRef]
- Klemensø, T.; Chatzichristodoulou, C.; Nielsen, J.; Bozza, F.; Thydén, K.; Kiebach, R.; Ramousse, S. Characterization of impregnated GDC nano structures and their functionality in LSM based cathodes. Solid State Ion. 2012, 224, 21–31. [Google Scholar] [CrossRef]
- Wang, C.; Tomov, R.I.; Mitchell-Williams, T.B.; Kumar, R.V.; Glowacki, B.A. Inkjet printing infiltration of Ni-Gd:CeO2 anodes for low temperature solid oxide fuel cells. J. Appl. Electrochem. 2017, 47, 1227–1238. [Google Scholar] [CrossRef]
- Doshi, R.; Richards, V.L.; Carter, J.D.; Wang, X.; Krumpelt, M. Development of Solid-Oxide Fuel Cells That Operate at 500°C. J. Electrochem. Soc. 1999, 146, 1273–1278. [Google Scholar] [CrossRef]
- Teraoka, T.; Zhang, H.M.; Okamoto, K.; Yamazoe, N. Mixed ionic-electronic conductivity of La1−xSrxCo1−yFeyO3−δ perovskite-type oxides. Mater. Res. Bull. 1988, 23, 51–58. [Google Scholar] [CrossRef]
- Tai, L.-W.; Nasrallah, M.M.; Anderson, H.U.; Sparlin, D.M.; Sehlin, S.R. Structure and electrical properties of La1-xSrxCo1-yFeyO3. Part 2. The system La1-xSrxCo0.2Fe0.803. Solid State Ion. 1995, 76, 273–283. [Google Scholar] [CrossRef]
- Ding, H.; Virkar, A.V.; Liu, M.; Liu, F. Suppression of Sr surface segregation in La1−xSrxCo1−yFeyO3−δ: A first principles study. Phys. Chem. Chem. Phys. 2013, 15, 489–496. [Google Scholar] [CrossRef]
- Hong, T.; Zhang, L.; Chen, F.; Xia, C. Oxygen surface exchange properties of La0.6Sr0.4Co0.8Fe0.2O3-δ coated with SmxCe1-xO2-δ. J. Power Sources 2012, 218, 254–260. [Google Scholar] [CrossRef]
- Hu, B.; Wang, Y.; Xia, C. Effects of Ceria Conductivity on the Oxygen Incorporation at the LSCF-SDC-Gas Three-Phase Boundary. J. Electrochem. Soc. 2015, 162, F33–F39. [Google Scholar] [CrossRef]
- Chen, J.; Liang, F.; Chi, B.; Pu, J.; Jiang, S.P.; Jian, L. Palladium and ceria infiltrated La0.8Sr0.2Co0.5Fe0.5O3−δ cathodes of solid oxide fuel cells. J. Power Sources 2009, 194, 275–280. [Google Scholar] [CrossRef]
- Nie, L.F.; Liu, M.F.; Zhang, Y.J.; Liu, M.L. La0.6Sr0.4Co0.2Fe0.8O3-δ cathodes infiltrated with samarium-doped cerium oxide for solid oxide fuel cells. J. Power Sources 2010, 195, 4704–4708. [Google Scholar] [CrossRef]
- Liu, M.; Ding, D.; Blinn, K.; Li, X.; Nie, L.; Liu, M. Enhanced performance of LSCF cathode through surface modification. Int. J. Hydrog. Energy 2012, 37, 8613–8620. [Google Scholar] [CrossRef]
- Tomov, R.I.; Mitchell-Williams, T.; Gao, C.H.; Kumar, R.V.; Glowacki, B.A. Performance optimization of LSCF/Gd:CeO2 composite cathodes via single-step inkjet printing infiltration. J. Appl. Electrochem. 2017, 47, 641–651. [Google Scholar] [CrossRef]
- Tsipis, E.V.; Kharton, V.V. Electrode materials and reaction mechanisms in solid oxide fuel cells: A brief review II. Electrochemical behavior vs. materials science aspects. J. Solid State Electrochem. 2008, 12, 1367–1391. [Google Scholar] [CrossRef]
- Lee, S.H.; Miller, N.; Abernathy, H.; Gerdes, K.; Manivannan, A. Effect of Sr-Doped LaCoO3 and LaZrO3 Infiltration on the Performance of SDC-LSCF Cathode. J. Electrochem. Soc. 2011, 158, B735–B742. [Google Scholar] [CrossRef]
- Clematis, D.; Barbucci, A.; Presto, S.; Viviani, M.; Carpanese, M.P. Electrocatalytic activity of perovskite-based cathodes for solid oxide fuel cells. Int. J. Hydrog. Energy 2019, 6212–6222. [Google Scholar] [CrossRef]
- Ai, N.; Jiang, S.P.; Lu, Z.; Chen, K.F.; Su, W.H. Nanostructured ( Ba , Sr ) ( Co , Fe ) O3 − δImpregnated ( La , Sr ) MnO3 Cathode for Intermediate-Temperature Solid Oxide Fuel Cells. J. Electrochem. Soc. 2010, 157, B1033–B1039. [Google Scholar] [CrossRef]
- Xu, N.S.; Li, X.; Zhao, X.; Zhao, H.L.; Huang, K. One-Step Infiltration of Mixed Conducting Electrocatalysts for Reducing Cathode Polarization of a Commercial Cathode-Supported SOFC. Electrochem. Solid-State Lett. 2012, 15, B1–B4. [Google Scholar] [CrossRef]
- Derby, B. Inkjet Printing of Functional and Structural Materials: Fluid Property Requirements, Feature Stability, and Resolution. Annu. Rev. Mater. Res. 2010, 40, 395–414. [Google Scholar] [CrossRef]
- de Gans, B.-J.; Duineveld, P.C.; Schubert, U.S. Inkjet Printing of Polymers: State of the Art and Future Developments. Adv. Mater. 2004, 16, 203–213. [Google Scholar] [CrossRef]
- Magdassi, S.; Kamyshny, A. Nanomaterials for 2D and 3D Printing; Wiley-VCH: Weinheim, Germany, 2017; pp. 6–20. ISBN 978-3-527-68582-0. [Google Scholar]
- Lee, K.T.; Yoon, H.S.; Ahn, J.S.; Wachsman, E.D. Bimodally integrated anode functional layer for lower temperature solid oxide fuel cells. J. Mater. Chem. 2012, 22, 17113–17120. [Google Scholar] [CrossRef]
- Kiebach, R.; Zielke, P.; Høgh, J.V.T.; Thyden, K.; Wang, H.-J.; Barford, R.; Hendriksen, P.V. Infiltration of SOFC stacks: Evaluation of the electrochemical performance enhancement and the underlying changes in the microstructure. Fuel Cells 2016, 16, 80–88. [Google Scholar] [CrossRef]
- Dowd, R.P., Jr.; Lee, S.; Fan, Y.; Gerdes, K. Engineering the solid oxide fuel cell electrocatalyst infiltration technique for industrial use. Int. J. Hydrog. Energy 2016, 41, 14971–14981. [Google Scholar] [CrossRef]
- Mitchel-Williams, T.B.; Tomov, R.I.; Saadabadi, S.A.; Krauz, M.; Aravind, P.V.; Glowacki, B.A.; Kumar, R.V. Infiltration of commercially available, anode supported SOFC’s via inkjet printing. Mater. Renew. Sustain. Energy 2017, 6, 12. [Google Scholar] [CrossRef]
- Wang, C.; Hopkins, S.C.; Tomov, R.I.; Kumar, R.V.; Glowacki, B.A. Optimisation of CGO suspensions for inkjet-printed SOFC electrolytes. J. Eur. Ceram. Soc. 2012, 32, 2317–2324. [Google Scholar] [CrossRef]
- Tomov, R.I.; Krauz, M.; Tluczek, A.; Kluczowski, R.; Krishnan, V.V.; Balasubramanian, K.; Kumar, R.V.; Glowacki, B.A. Vacuum-sintered stainless steel porous supports for inkjet printing of functional SOFC coatings. Mater. Renew. Sustain. Energy 2015, 4, 14. [Google Scholar] [CrossRef]
- Kharton, V.V.; Yaremchenko, A.A.; Naumovich, E.N. Research on the electrochemistry of oxygen ion conductors in the former Soviet Union. II. Perovskite-related oxides. J. Solid State Electrochem. 1999, 3, 303–326. [Google Scholar] [CrossRef]
- Crumlin, E.J.; Mutoro, E.; Liu, Z.; Grass, M.E.; Biegalski, M.D.; Lee, Y.L.; Morgan, D.; Christen, H.M.; Bluhm, H.; Shao-Horn, Y. Surface strontium enrichment on highly active perovskites for oxygen electrocatalysis in solid oxide fuel cells. Energy Environ. Sci. 2012, 5, 6081–6088. [Google Scholar] [CrossRef]
- Harrison, W.A. Origin of Sr segregation at La1−xSrxMnO3 surfaces. Phys. Rev. B Condens. Matter Mater. Phys. 2011, 83, 155437. [Google Scholar] [CrossRef]
- Rupp, G.M.; Opitz, A.K.; Nenning, A.; Limbeck, A.; Fleig, J. Real-time impedance monitoring of oxygen reduction during surface modification of thin film cathodes. Nat. Mater. 2017, 16, 640–646. [Google Scholar] [CrossRef] [PubMed]
- Mutoro, E.; Crumlin, E.J.; Biegalski, M.D.; Christen, H.M.; Shao-Horn, Y. Enhanced oxygen reduction activity on surface-decorated perovskite thin films for solid oxide fuel cells. Energy Environ. Sci. 2011, 4, 3689–3696. [Google Scholar] [CrossRef]
- Sholklapper, T.Z.; Kurokawa, H.; Jacobson, C.P.; Visco, S.J.; De Jonghe, L.C. Nanostructured solid oxide fuel cell electrodes. Nanoletters 2007, 7, 2136–2141. [Google Scholar] [CrossRef]
- Leonide, A.; Sonn, V.; Weber, A.; Ivers-Tiffée, E. Evaluation and Modeling of the Cell Resistance in Anode-Supported Solid Oxide Fuel Cells. J. Electrochem. Soc. 2007, 155, B36–B41. [Google Scholar] [CrossRef]
- Barfod, R.; Mogensen, M.; Klemensoø, T.; Hagen, A.; Liu, Y.-L.; Vang Hendriksen, P. Detailed Characterization of Anode-Supported SOFCs by Impedance Spectroscopy. J. Electrochem. Soc. 2007, 154, B371–B378. [Google Scholar] [CrossRef]
- Nielsen, J.; Mogensen, M. SOFC LSM:YSZ cathode degradation induced by moisture: An impedance spectroscopy study. Solid State Ion. 2011, 189, 74–81. [Google Scholar] [CrossRef]
- Shi, W.; Jia, C.; Zhang, Y.; Lü, Z.; Han, M. Differentiation and decomposition of solid oxide fuel cell electrochemical impedance spectra. Acta Phys. Chim. Sin. 2019, 35, 509–516. [Google Scholar] [CrossRef]
- Esquirol, A.; Kilner, J.; Brandon, N. Oxygen transport in La0.6Sr0.4Co0.2Fe0.8O3-δ/Ce0.8Ge0.2O2-x composite cathode for IT-SOFCs. Solid State Ion 2004, 175, 63–67. [Google Scholar] [CrossRef]



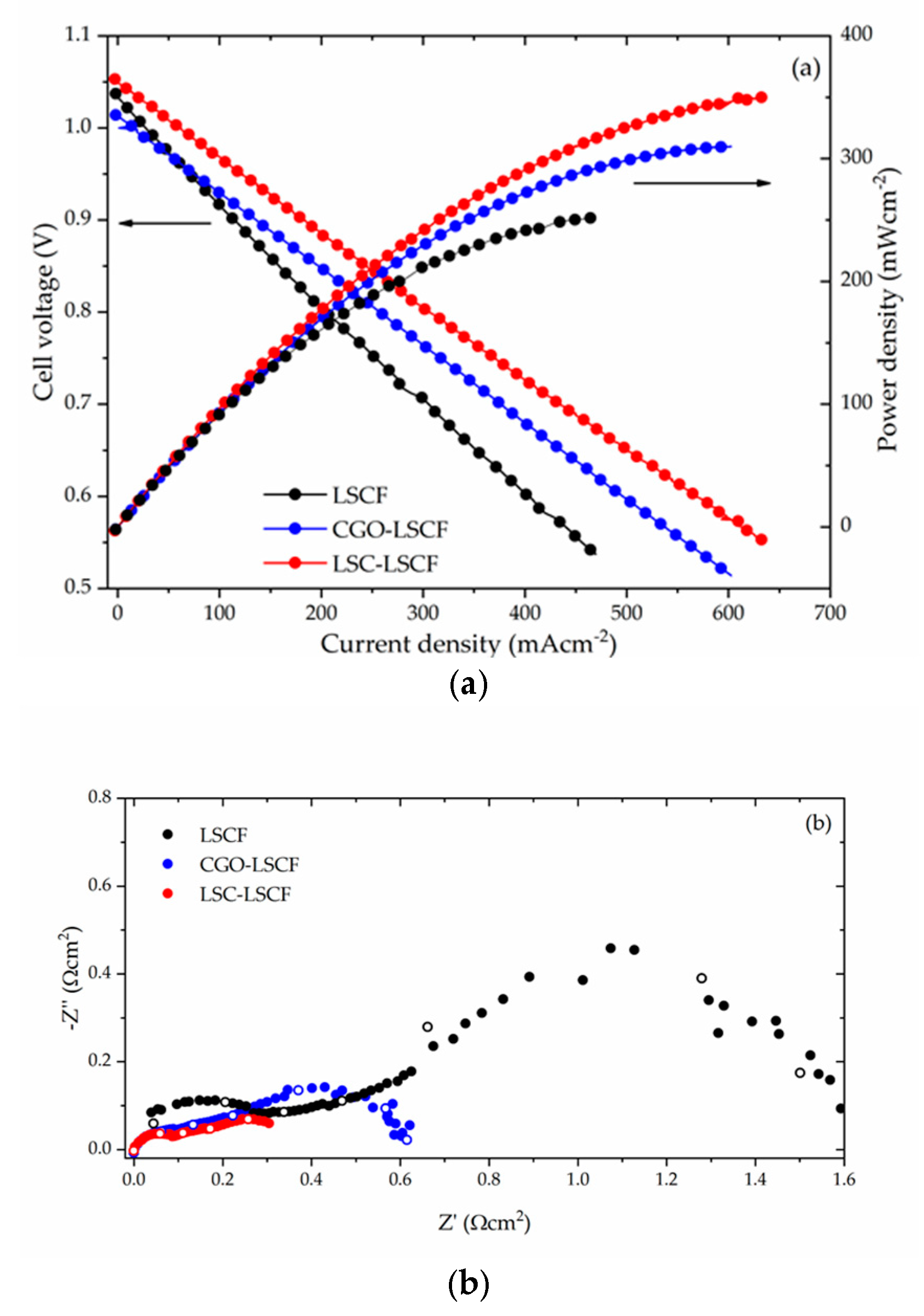
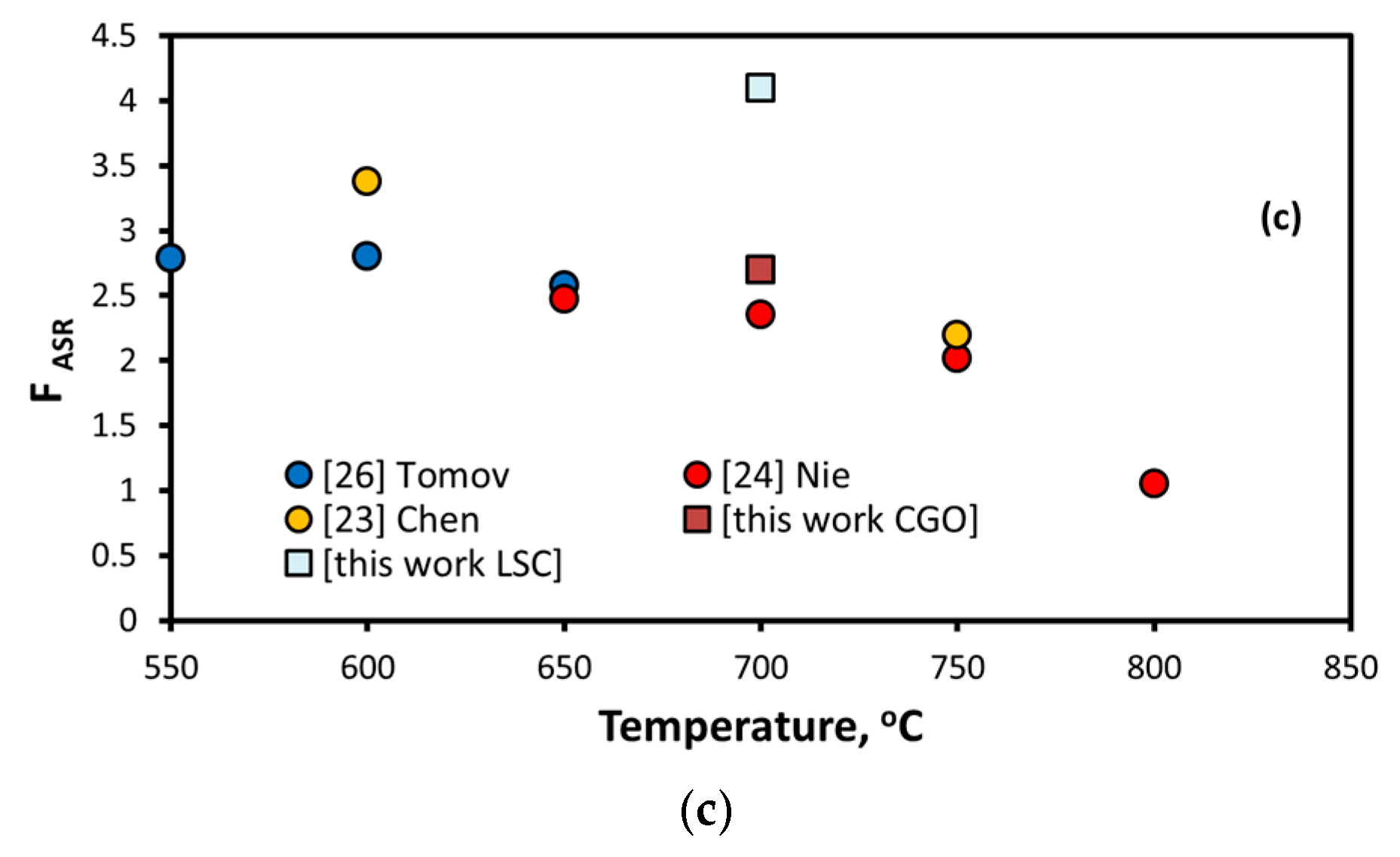
| Source | Infiltrated Ink | Scaffold | T, °C | FASR |
|---|---|---|---|---|
| Chen et al. [23] | Gd0.2Ce0.8O2−x | LSCF | 600 | 3.66 |
| 750 | 3.37 | |||
| Nie et al. [24] | Sm0.2Ce0.8O1.95 | LSCF | 750 | 2.02 |
| 800 | 1.05 | |||
| Liu et al. [25] | La0.4875Ca0.0125Ce0.5O2−δ | LSCF | 750 | 1.7 |
| Tomov et al. [26] | Gd0.1Ce0.8O2−x | LSCF/CGO | 650 | 2.56 |
| Piezoelectric | Electromagnetic | |
|---|---|---|
| Drop volume | 1–100 pL | 10–50 nL+ |
| Orifice size | 10–60 µm | 60–150 µm |
| Pressure regime | Vacuum (−50 mbar) | Positive (100–600 mbar) |
| Jetting distance | 1 mm | 4 mm |
| Maximum jetting rate | 10–50 kHz | 0.5–3 kHz |
| Multi-nozzles print head | ~512 | ~16 |
| Main advantage/Disadvantage | High resolution | Low cost robust technology |
| High jetting rate | Ink compatibility | |
| High throughput | Disassembling/assembling/cleaning option | |
| High cost | Low resolution | |
| Materials applications | Patterning | Coatings and infiltration |
| Thin layers | Suspensions |
| Ink | Viscosity, cP | Opening Time, µs | Drop Volume, nL | Drop Velocity, m·s−1 |
|---|---|---|---|---|
| 0.75 M CGO | 4.6 | 250 | 38 | 2.3 |
| 0.75M LCO | 4.2 | 250 | 32 | 2.4 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Venezia, E.; Viviani, M.; Presto, S.; Kumar, V.; Tomov, R.I. Inkjet Printing Functionalization of SOFC LSCF Cathodes. Nanomaterials 2019, 9, 654. https://doi.org/10.3390/nano9040654
Venezia E, Viviani M, Presto S, Kumar V, Tomov RI. Inkjet Printing Functionalization of SOFC LSCF Cathodes. Nanomaterials. 2019; 9(4):654. https://doi.org/10.3390/nano9040654
Chicago/Turabian StyleVenezia, Eleonora, Massimo Viviani, Sabrina Presto, Vasant Kumar, and Rumen I. Tomov. 2019. "Inkjet Printing Functionalization of SOFC LSCF Cathodes" Nanomaterials 9, no. 4: 654. https://doi.org/10.3390/nano9040654
APA StyleVenezia, E., Viviani, M., Presto, S., Kumar, V., & Tomov, R. I. (2019). Inkjet Printing Functionalization of SOFC LSCF Cathodes. Nanomaterials, 9(4), 654. https://doi.org/10.3390/nano9040654






