High-Density Plasmonic Nanoparticle Arrays Deposited on Nanoporous Anodic Alumina Templates for Optical Sensor Applications
Abstract
1. Introduction
2. Experimental
2.1. Materials
2.2. Methods
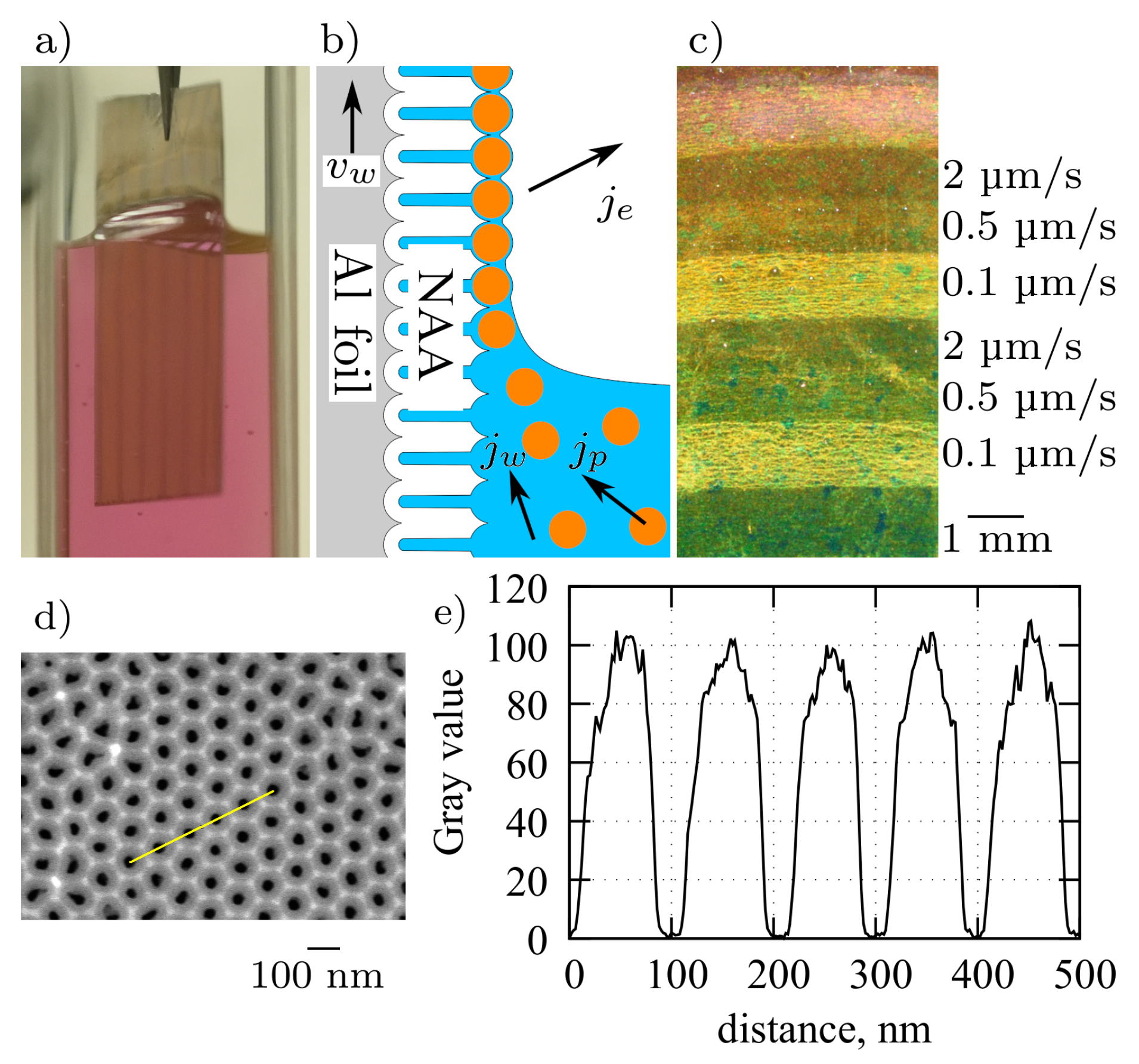
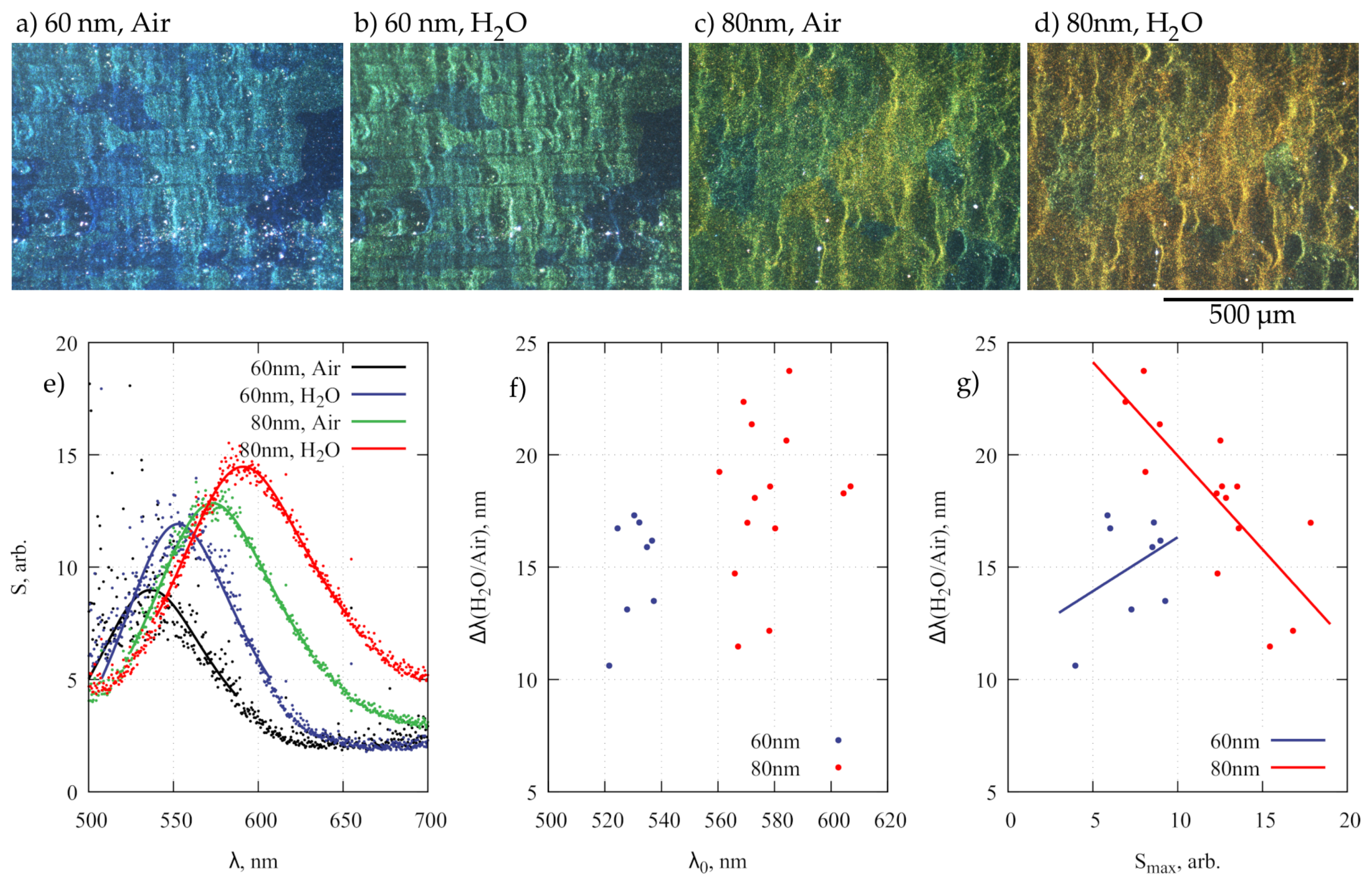
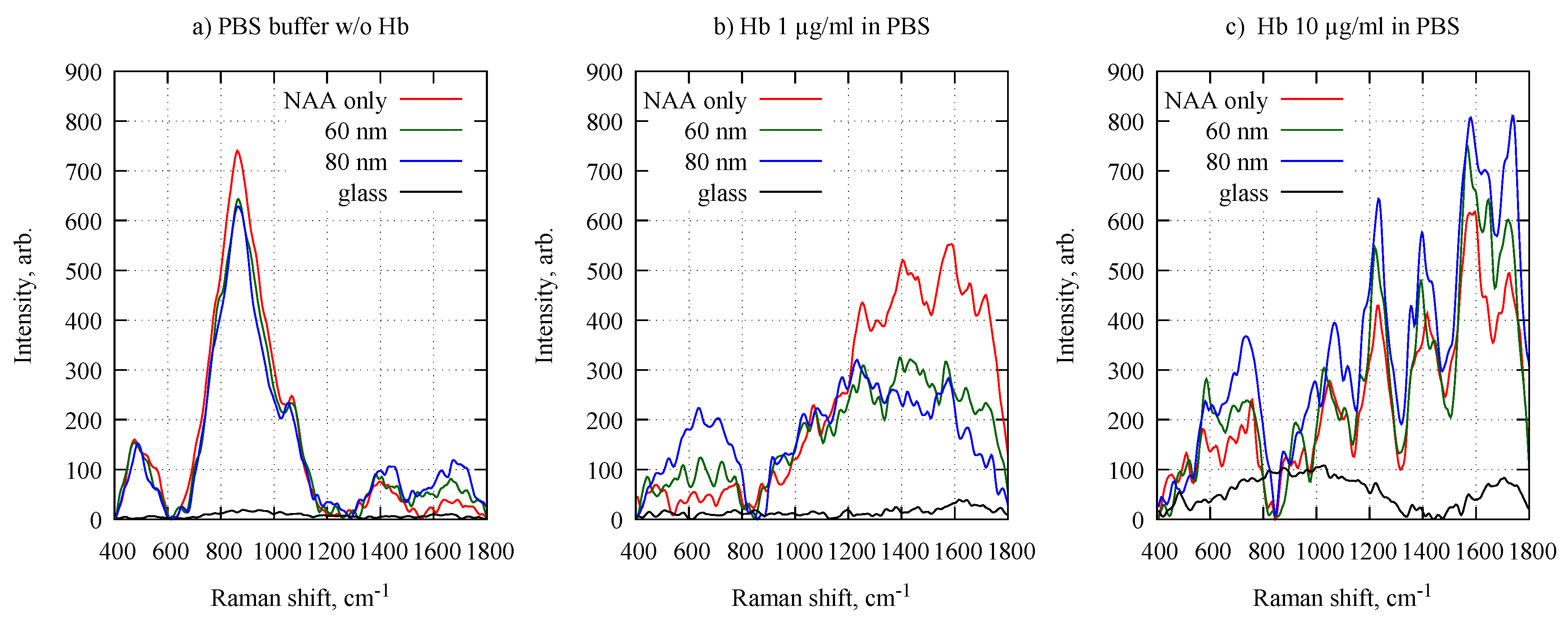
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Willets, K.A.; Van Duyne, R.P. Localized Surface Plasmon Resonance Spectroscopy and Sensing. Annu. Rev. Phys. Chem. 2007, 58, 267–297. [Google Scholar] [CrossRef]
- Halas, N.J.; Lal, S.; Chang, W.S.; Link, S.; Nordlander, P. Plasmons in Strongly Coupled Metallic Nanostructures. Chem. Rev. 2011, 111, 3913–3961. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.Y.; Forestiere, C.; Pasquale, A.J.; Trevino, J.; Walsh, G.; Galli, P.; Romagnoli, M.; Negro, L.D. Plasmon-enhanced structural coloration of metal films with isotropic Pinwheel nanoparticle arrays. Opt. Express 2011, 19, 23818–23830. [Google Scholar] [CrossRef] [PubMed]
- Zou, S.; Schatz, G.C. Narrow plasmonic/photonic extinction and scattering line shapes for one and two dimensional silver nanoparticle arrays. J. Chem. Phys. 2004, 121, 12606–12612. [Google Scholar] [CrossRef]
- Kravets, V.G.; Kabashin, A.V.; Barnes, W.L.; Grigorenko, A.N. Plasmonic Surface Lattice Resonances: A Review of Properties and Applications. Chem. Rev. 2018, 118, 5912–5951. [Google Scholar] [CrossRef]
- Chau, Y.F.C.; Chou Chao, C.T.; Lim, C.M.; Huang, H.J.; Chiang, H.P. Depolying Tunable Metal-Shell/Dielectric Core Nanorod Arrays as the Virtually Perfect Absorber in the Near-Infrared Regime. ACS Omega 2018, 3, 7508–7516. [Google Scholar] [CrossRef]
- Shiigi, H.; Nagaoka, T. Molecularly Bridged Gold Nanoparticle Array for Sensing Applications. Anal. Sci. 2014, 30, 89–96. [Google Scholar] [CrossRef]
- Wang, H.; Levin, C.S.; Halas, N.J. Nanosphere Arrays with Controlled Sub-10-nm Gaps as Surface-Enhanced Raman Spectroscopy Substrates. J. Am. Chem. Soc. 2005, 127, 14992–14993. [Google Scholar] [CrossRef]
- Lee, S.J.; Guan, Z.; Xu, H.; Moskovits, M. Surface-Enhanced Raman Spectroscopy and Nanogeometry: The Plasmonic Origin of SERS. J. Phys. Chem. C 2007, 111, 17985–17988. [Google Scholar] [CrossRef]
- Venditti, I. Gold Nanoparticles in Photonic Crystals Applications: A Review. Materials 2017, 10, 97. [Google Scholar] [CrossRef]
- Prikulis, J.; Malinovskis, U.; Poplausks, R.; Apsite, I.; Bergs, G.; Erts, D. Optical Scattering by Dense Disordered Metal Nanoparticle Arrays. Plasmonics 2014, 9, 427–434. [Google Scholar] [CrossRef]
- Svedendahl, M.; Verre, R.; Käll, M. Refractometric biosensing based on optical phase flips in sparse and short-range-ordered nanoplasmonic layers. Light Sci. Appl. 2014, 3, e220. [Google Scholar] [CrossRef]
- Bergs, G.; Malinovskis, U.; Poplausks, R.; Apsite, I.; Erts, D.; Prikulis, J. Polarized interference imaging of dense disordered plasmonic nanoparticle arrays for biosensor applications. Phys. Scr. 2015, 90, 094002. [Google Scholar] [CrossRef]
- Masuda, H.; Satoh, M. Fabrication of Gold Nanodot Array Using Anodic Porous Alumina as an Evaporation Mask. Jpn. J. Appl. Phys. 1996, 35, L126–L129. [Google Scholar] [CrossRef]
- Sander, M.; Tan, L.S. Nanoparticle Arrays on Surfaces Fabricated Using Anodic Alumina Films as Templates. Adv. Funct. Mater. 2003, 13, 393–397. [Google Scholar] [CrossRef]
- Malinovskis, U.; Poplausks, R.; Apsite, I.; Meija, R.; Prikulis, J.; Lombardi, F.; Erts, D. Ultrathin Anodic Aluminum Oxide Membranes for Production of Dense Sub-20 nm Nanoparticle Arrays. J. Phys. Chem. C 2014, 118, 8685–8690. [Google Scholar] [CrossRef]
- Hao, Q.; Huang, H.; Fan, X.; Hou, X.; Yin, Y.; Li, W.; Si, L.; Nan, H.; Wang, H.; Mei, Y.; Qiu, T.; Chu, P.K. Facile design of ultra-thin anodic aluminum oxide membranes for the fabrication of plasmonic nanoarrays. Nanotechnology 2017, 28. [Google Scholar] [CrossRef]
- Jung, M.; Kim, J.H.; Choi, Y.W. Preparation of Anodic Aluminum Oxide Masks with Size-Controlled Pores for 2D Plasmonic Nanodot Arrays. J. Nanomater. 2018, 2018, 6249890. [Google Scholar] [CrossRef]
- Pan, S.; Zeng, D.; Zhang, H.; Li, H. Preparation of ordered array of nanoscopic gold rods by template method and its optical properties. Appl. Phys. A 2000, 70, 637–640. [Google Scholar] [CrossRef]
- Wang, H.H.; Liu, C.Y.; Wu, S.B.; Liu, N.W.; Peng, C.Y.; Chan, T.H.; Hsu, C.F.; Wang, J.K.; Wang, Y.L. Highly Raman-Enhancing Substrates Based on Silver Nanoparticle Arrays with Tunable Sub-10 nm Gaps. Adv. Mater. 2006, 18, 491–495. [Google Scholar] [CrossRef]
- Baitimirova, M.; Pastare, A.; Katkevics, J.; Viksna, A.; Prikulis, J.; Erts, D. Gold nanowire synthesis by semi-immersed nanoporous anodic aluminium oxide templates in potassium dicyanoaurate-hexacyanoferrate electrolyte. Micro Nano Lett. 2014, 9, 761–765. [Google Scholar] [CrossRef]
- Polyakov, B.; Prikulis, J.; Grigorjeva, L.; Millers, D.; Daly, B.; Holmes, J.D.; Erts, D. Photoconductivity of Germanium Nanowire Arrays Incorporated in Anodic Aluminum Oxide. J. Phys. Conf. Ser. 2007, 61, 283–287. [Google Scholar] [CrossRef]
- Mori, T.; Mori, T.; Tanaka, Y.; Suzaki, Y.; Yamaguchi, K. Fabrication of single-crystalline plasmonic nanostructures on transparent and flexible amorphous substrates. Sci. Rep. 2017, 7, 42859. [Google Scholar] [CrossRef]
- Kumeria, T.; Losic, D. Controlling interferometric properties of nanoporous anodic aluminium oxide. Nanoscale Res. Lett. 2012, 7, 88. [Google Scholar] [CrossRef]
- Kumeria, T.; Santos, A.; Losic, D. Ultrasensitive Nanoporous Interferometric Sensor for Label-Free Detection of Gold(III) Ions. ACS Appl. Mater. Interfaces 2013, 5, 11783–11790. [Google Scholar] [CrossRef]
- Yan, W.; Overbury, S.H.; Dai, S. Gold Catalysts Supported on Nanostructured Materials: Support Effects. In Nanotechnology in Catalysis; Springer: New York, NY, USA, 2007; pp. 55–71. [Google Scholar] [CrossRef]
- Malinovskis, U.; Berzins, A.; Gahbauer, F.; Ferber, R.; Kitenbergs, G.; Muiznieks, I.; Erts, D.; Prikulis, J. Colloidal nanoparticle sorting and ordering on anodic alumina patterned surfaces using templated capillary force assembly. Surf. Coat. Technol. 2017, 326, 264–269. [Google Scholar] [CrossRef]
- Venditti, I.; Hassanein, T.; Fratoddi, I.; Fontana, L.; Battocchio, C.; Rinaldi, F.; Carafa, M.; Marianecci, C.; Diociaiuti, M.; Agostinelli, E.; et al. Bioconjugation of gold-polymer core–shell nanoparticles with bovine serum amine oxidase for biomedical applications. Colloids Surf. B Biointerfaces 2015, 134, 314–321. [Google Scholar] [CrossRef]
- Matricardi, C.; Hanske, C.; Garcia-Pomar, J.L.; Langer, J.; Mihi, A.; Liz-Marzan, L.M. Gold Nanoparticle Plasmonic Superlattices as Surface-Enhanced Raman Spectroscopy Substrates. ACS Nano 2018, 12, 8531–8539. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Yang, A.; Hryn, A.J.; Schatz, G.C.; Odom, T.W. Superlattice Plasmons in Hierarchical Au Nanoparticle Arrays. ACS Photonics 2015, 2, 1789–1794. [Google Scholar] [CrossRef]
- Yan, B.; Boriskina, S.V.; Reinhard, B.M. Design and Implementation of Noble Metal Nanoparticle Cluster Arrays for Plasmon Enhanced Biosensing. J. Phys. Chem. C 2011, 115, 24437–24453. [Google Scholar] [CrossRef]
- Kubo, A.; Gorup, L.; Toffano, L.; Amaral, L.; Rodrigues-Filho, E.; Mohan, H.; Aroca, R.; Camargo, E. Nanostructured Assemblies of Gold and Silver Nanoparticles for Plasmon Enhanced Spectroscopy Using Living Biotemplates. Colloids Interfaces 2017, 1, 4. [Google Scholar] [CrossRef]
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 2012, 9, 671–675. [Google Scholar] [CrossRef] [PubMed]
- Cao, A.; Pandya, A.K.; Serhatkulu, G.K.; Weber, R.E.; Dai, H.; Thakur, J.S.; Naik, V.M.; Naik, R.; Auner, G.W.; Rabah, R.; et al. A robust method for automated background subtraction of tissue fluorescence. J. Raman Spectrosc. 2007, 38, 1199–1205. [Google Scholar] [CrossRef]
- Eilers, P.H.C. A Perfect Smoother. Anal. Chem. 2003, 75, 3631–3636. [Google Scholar] [CrossRef] [PubMed]
- Dimitrov, A.S.; Nagayama, K. Continuous Convective Assembling of Fine Particles into Two-Dimensional Arrays on Solid Surfaces. Langmuir 1996, 12, 1303–1311. [Google Scholar] [CrossRef]
- Prikulis, J.; Tamulevičius, T.; Poplausks, R.; Bergs, G.; Apsite, I.; Malinovskis, U.; Actins, A.; Erts, D. Optical properties of thin metal films with nanohole arrays on porous alumina–aluminum structures. RSC Adv. 2015, 5, 68143–68150. [Google Scholar] [CrossRef]
- Virganavičius, D.; Juodėnas, M.; Tamulevičius, T.; Schift, H.; Tamulevičius, S. Investigation of transient dynamics of capillary assisted particle assembly yield. Appl. Surf. Sci. 2017, 406, 136–143. [Google Scholar] [CrossRef]
- Malinovskis, U.; Berzins, A.; Smits, J.; Gahbauer, F.H.; Ferber, R.; Erts, D.; Prikulis, J. Fluorescent nanodiamond array deposition on porous anodized aluminum oxide using asperity assisted capillary force assembly. Proc. Estonian Acad. Sci. 2017, 66, 416. [Google Scholar] [CrossRef]
- Schäffer, E.; Wong, P.Z. Dynamics of Contact Line Pinning in Capillary Rise and Fall. Phys. Rev. Lett. 1998, 80, 3069–3072. [Google Scholar] [CrossRef]
- Hwang, S.K.; Jeong, S.H.; Hwang, H.Y.; Lee, O.J.; Lee, K.H. Fabrication of highly ordered pore array in anodic aluminum oxide. Korean J. Chem. Eng. 2002, 19, 467–473. [Google Scholar] [CrossRef]
- Nielsch, K.; Choi, J.; Schwirn, K.; Wehrspohn, R.B.; Gösele, U. Self-ordering Regimes of Porous Alumina: The 10% Porosity Rule. Nano Lett. 2002, 2, 677–680. [Google Scholar] [CrossRef]
- Jain, P.K.; Lee, K.S.; El-Sayed, I.H.; El-Sayed, M.A. Calculated Absorption and Scattering Properties of Gold Nanoparticles of Different Size, Shape, and Composition: Applications in Biological Imaging and Biomedicine. J. Phys. Chem. B 2006, 110, 7238–7248. [Google Scholar] [CrossRef]
- Lee, K.L.; Chang, C.C.; You, M.L.; Pan, M.Y.; Wei, P.K. Enhancing Surface Sensing Sensitivity of Metallic Nanostructures using Blue-Shifted Surface Plasmon Mode and Fano Resonance. Sci. Rep. 2018, 8, 9762. [Google Scholar] [CrossRef] [PubMed]
- Singh Sekhon, J.; Verma, S.S. Refractive Index Sensitivity Analysis of Ag, Au, and Cu Nanoparticles. Plasmonics 2011, 6, 311–317. [Google Scholar] [CrossRef]
- Rusciano, G.; De Luca, A.; Pesce, G.; Sasso, A. Raman Tweezers as a Diagnostic Tool of Hemoglobin-Related Blood Disorders. Sensors 2008, 8, 7818–7832. [Google Scholar] [CrossRef]
- Ramser, K.; Logg, K.; Goksör, M.; Enger, J.; Käll, M.; Hanstorp, D. Resonance Raman spectroscopy of optically trapped functional erythrocytes. J. Biomed. Opt. 2004, 9, 593. [Google Scholar] [CrossRef] [PubMed]
- Brunner, H.; Sussner, H. Resonance Raman scattering on haemoglobin. Biochim. Biophys. Acta Protein Struct. 1973, 310, 20–31. [Google Scholar] [CrossRef]
- Lazzara, T.D.; Mey, I.; Steinem, C.; Janshoff, A. Benefits and Limitations of Porous Substrates as Biosensors for Protein Adsorption. Anal. Chem. 2011, 83, 5624–5630. [Google Scholar] [CrossRef] [PubMed]
- Höök, F.; Rodahl, M.; Kasemo, B.; Brzezinski, P. Structural changes in hemoglobin during adsorption to solid surfaces: Effects of pH, ionic strength, and ligand binding. Proc. Natl. Acad. Sci. USA 1998, 95, 12271–12276. [Google Scholar] [CrossRef]
- Santos, A.; Kumeria, T. Nanoporous Anodic Alumina for Optical Biosensing. In Nanoporous Alumina; Springer: Berlin, Germany, 2015; Volume 219, pp. 219–247. [Google Scholar] [CrossRef]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Malinovskis, U.; Poplausks, R.; Erts, D.; Ramser, K.; Tamulevičius, S.; Tamulevičienė, A.; Gu, Y.; Prikulis, J. High-Density Plasmonic Nanoparticle Arrays Deposited on Nanoporous Anodic Alumina Templates for Optical Sensor Applications. Nanomaterials 2019, 9, 531. https://doi.org/10.3390/nano9040531
Malinovskis U, Poplausks R, Erts D, Ramser K, Tamulevičius S, Tamulevičienė A, Gu Y, Prikulis J. High-Density Plasmonic Nanoparticle Arrays Deposited on Nanoporous Anodic Alumina Templates for Optical Sensor Applications. Nanomaterials. 2019; 9(4):531. https://doi.org/10.3390/nano9040531
Chicago/Turabian StyleMalinovskis, Uldis, Raimonds Poplausks, Donats Erts, Kerstin Ramser, Sigitas Tamulevičius, Asta Tamulevičienė, Yesong Gu, and Juris Prikulis. 2019. "High-Density Plasmonic Nanoparticle Arrays Deposited on Nanoporous Anodic Alumina Templates for Optical Sensor Applications" Nanomaterials 9, no. 4: 531. https://doi.org/10.3390/nano9040531
APA StyleMalinovskis, U., Poplausks, R., Erts, D., Ramser, K., Tamulevičius, S., Tamulevičienė, A., Gu, Y., & Prikulis, J. (2019). High-Density Plasmonic Nanoparticle Arrays Deposited on Nanoporous Anodic Alumina Templates for Optical Sensor Applications. Nanomaterials, 9(4), 531. https://doi.org/10.3390/nano9040531






