Doped N/Ag Carbon Dot Catalytic Amplification SERS Strategy for Acetamiprid Coupled Aptamer with 3,3′-Dimethylbiphenyl-4,4′-diamine Oxidizing Reaction
Abstract
:1. Introduction
2. Experimental Section
2.1. Instruments
2.2. Reagents
2.3. Procedure
3. Results and Discussions
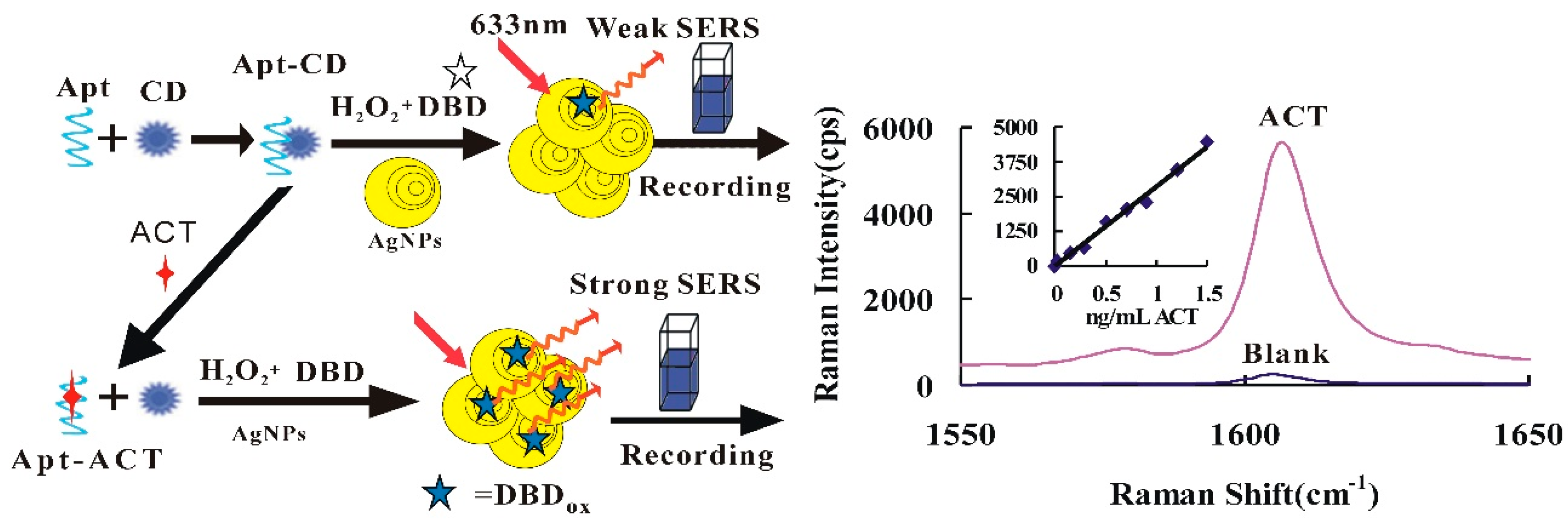
3.1. Analytical Principle
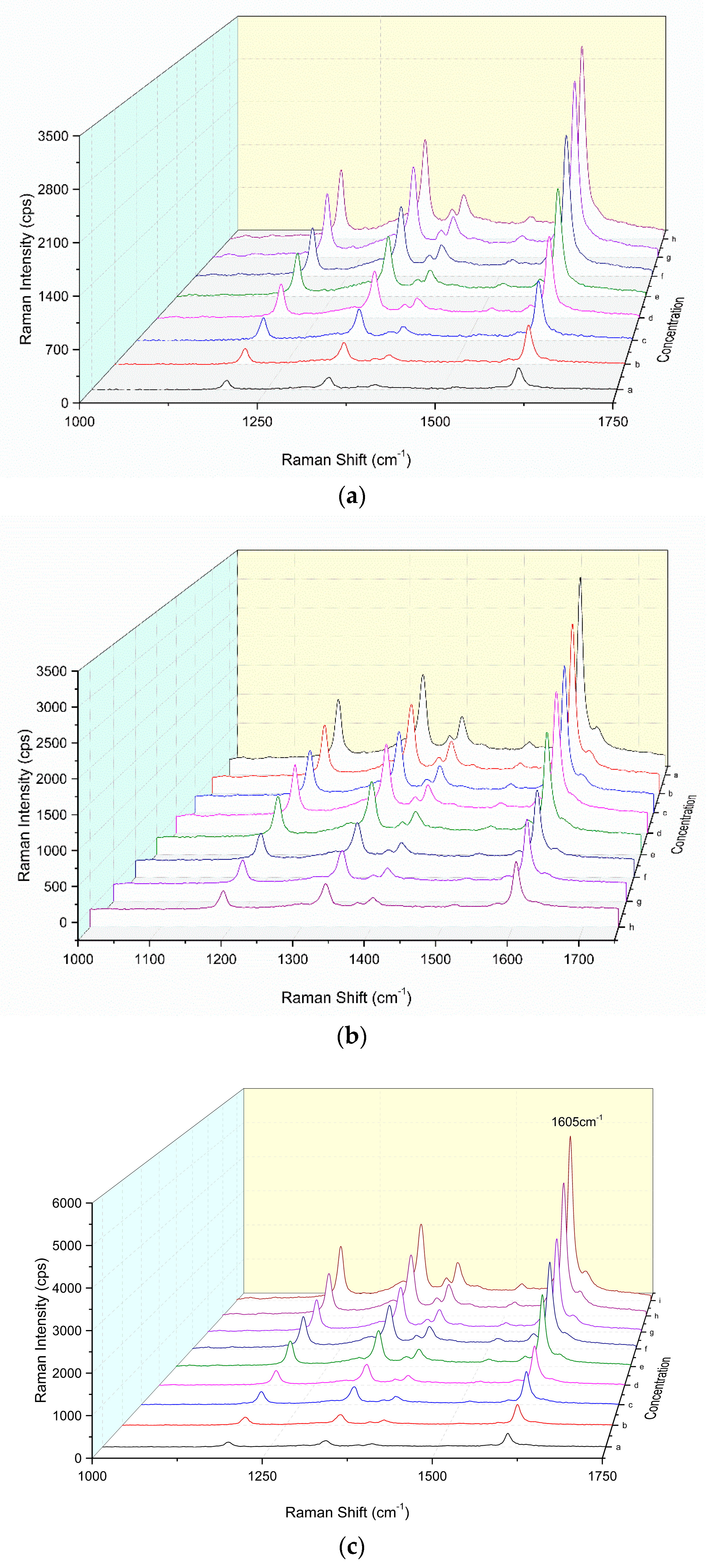
3.2. SERS Spectra
3.3. Nanocatalysis and Aptamer Inhibition
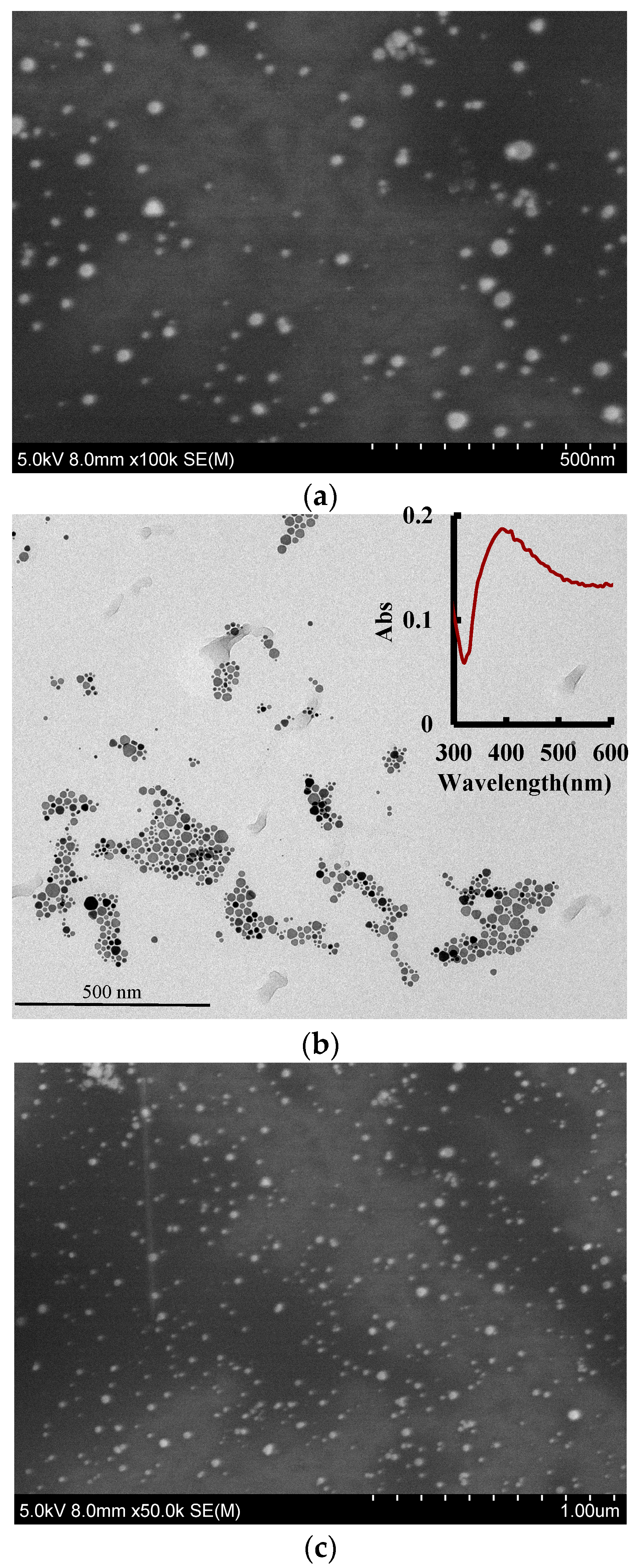
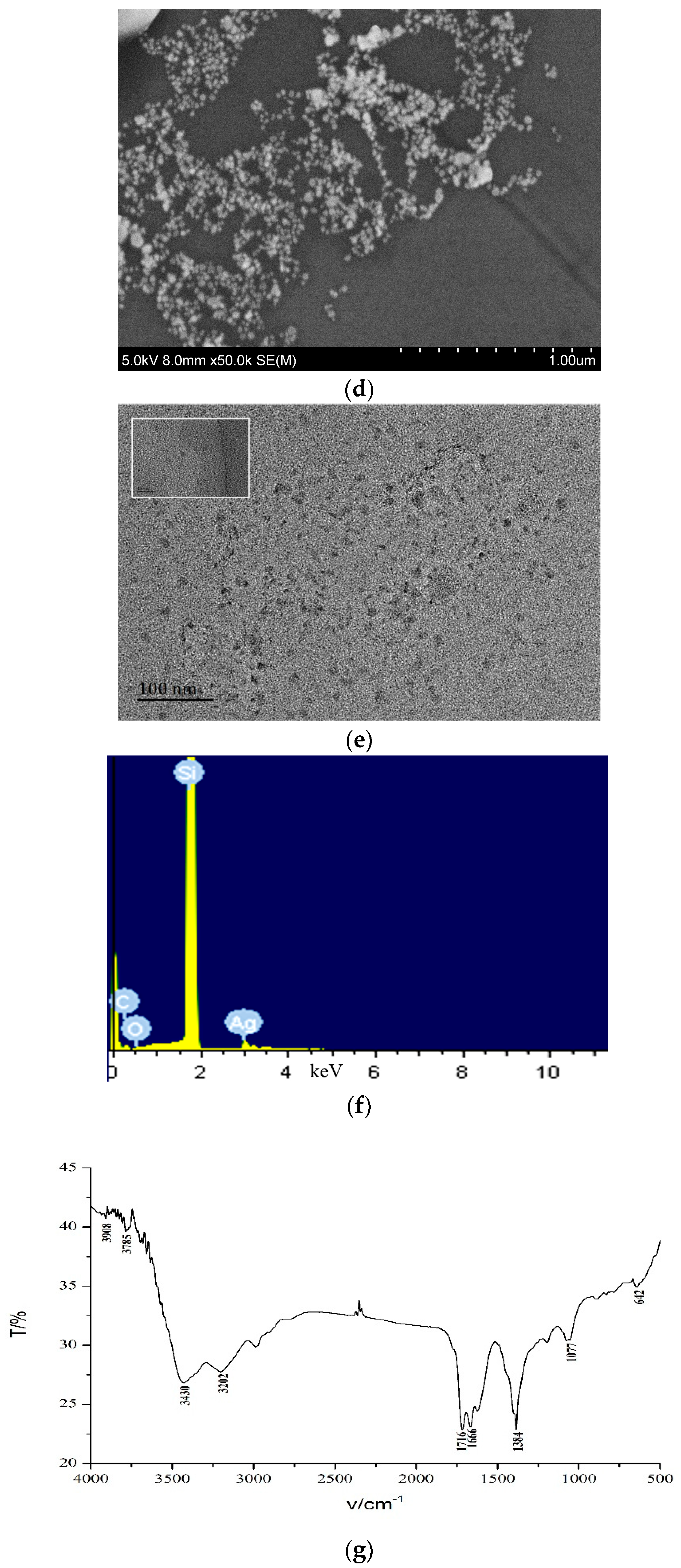
3.4. Electron Microscopy (EM) and Infrared Spectra
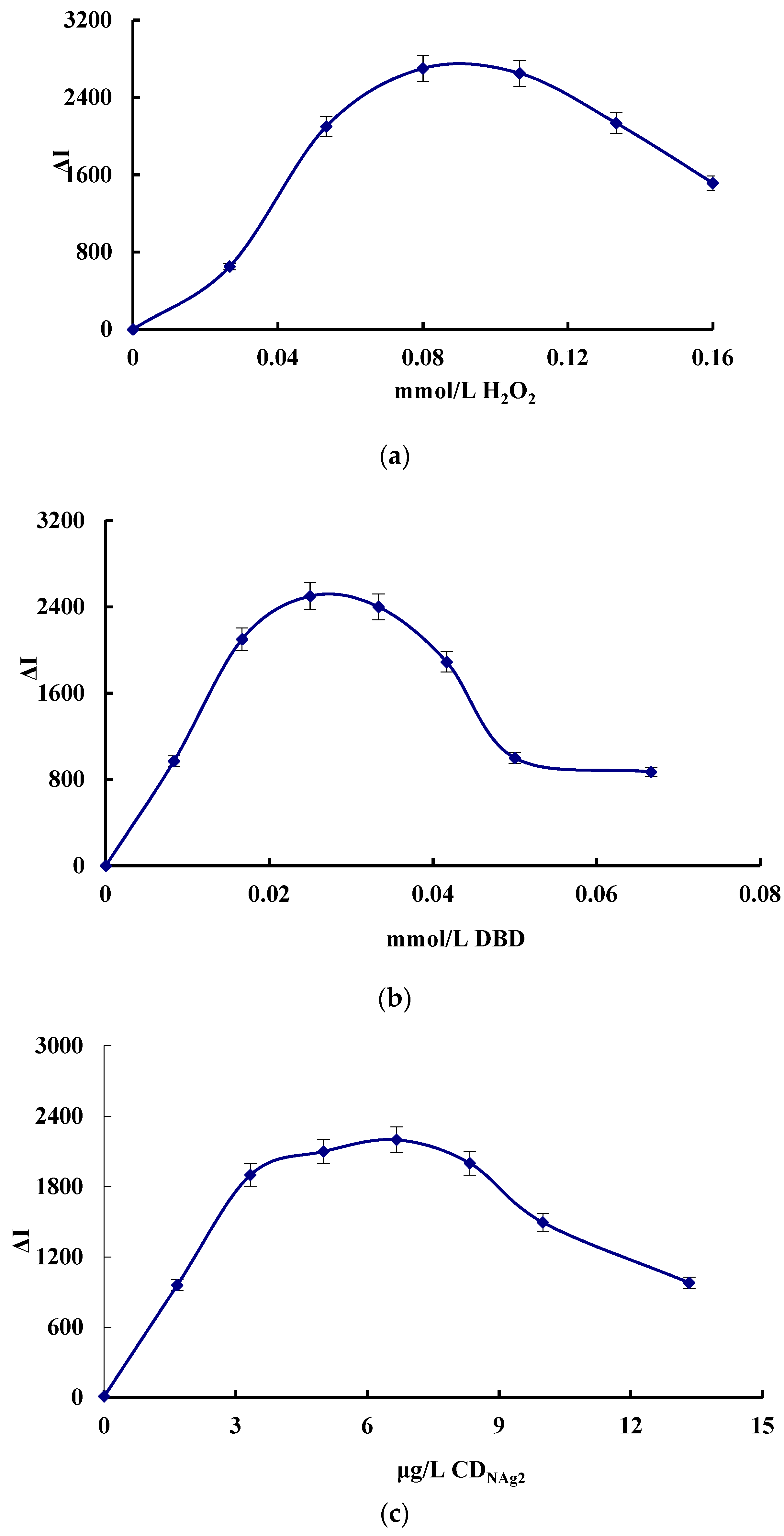
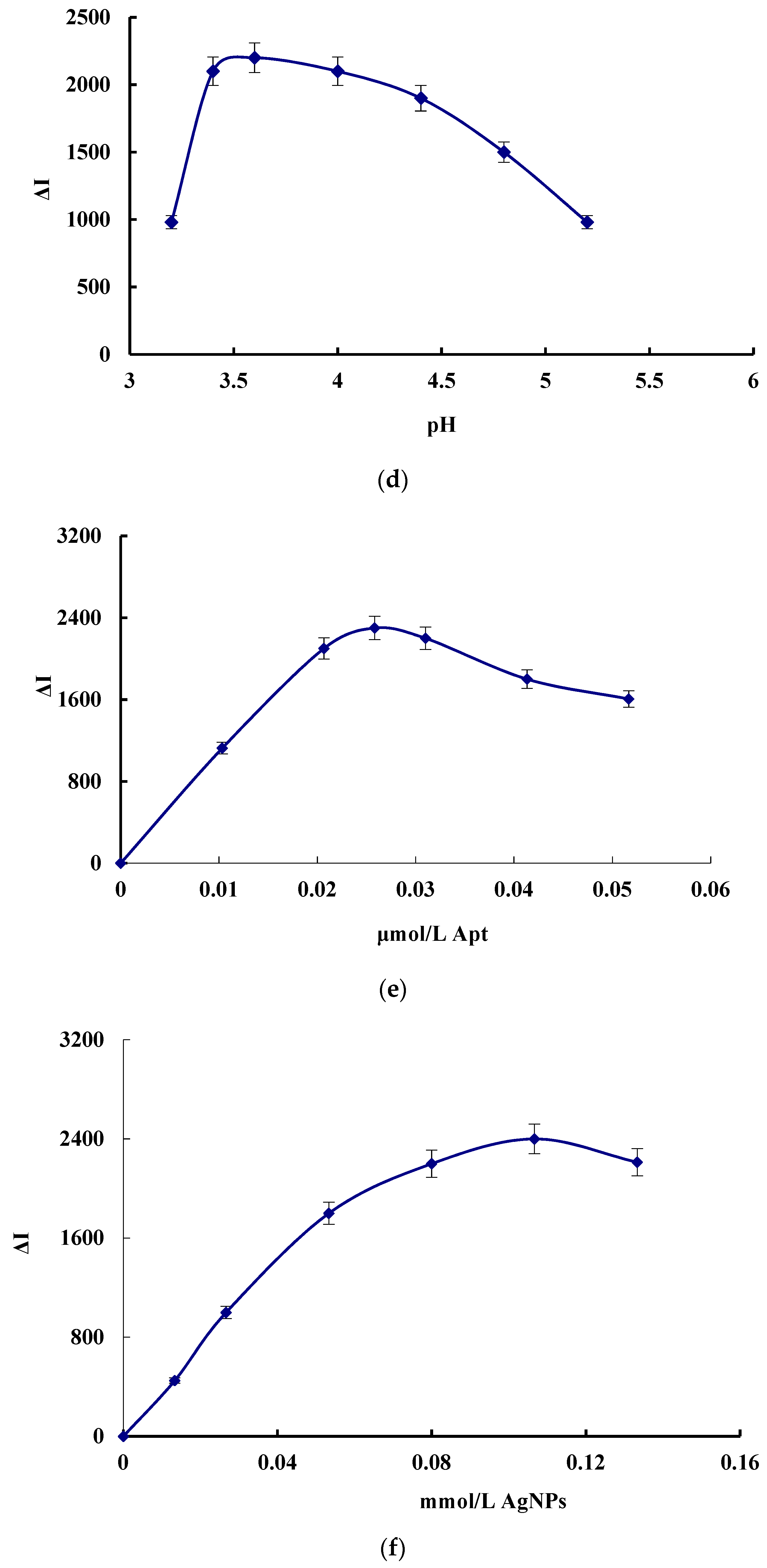
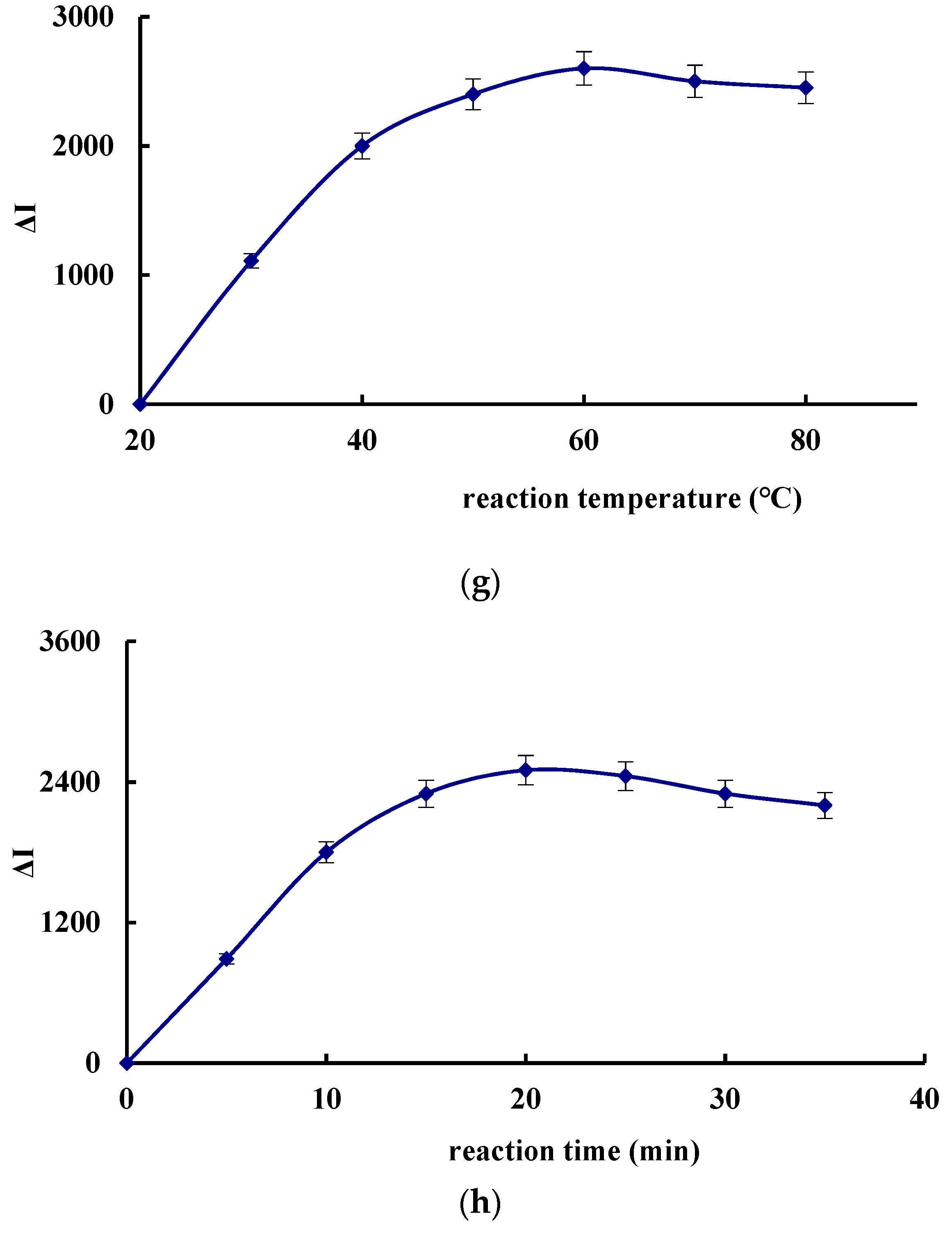
3.5. Optimization of the Analytical Conditions
3.6. Influence of Interfering Ions
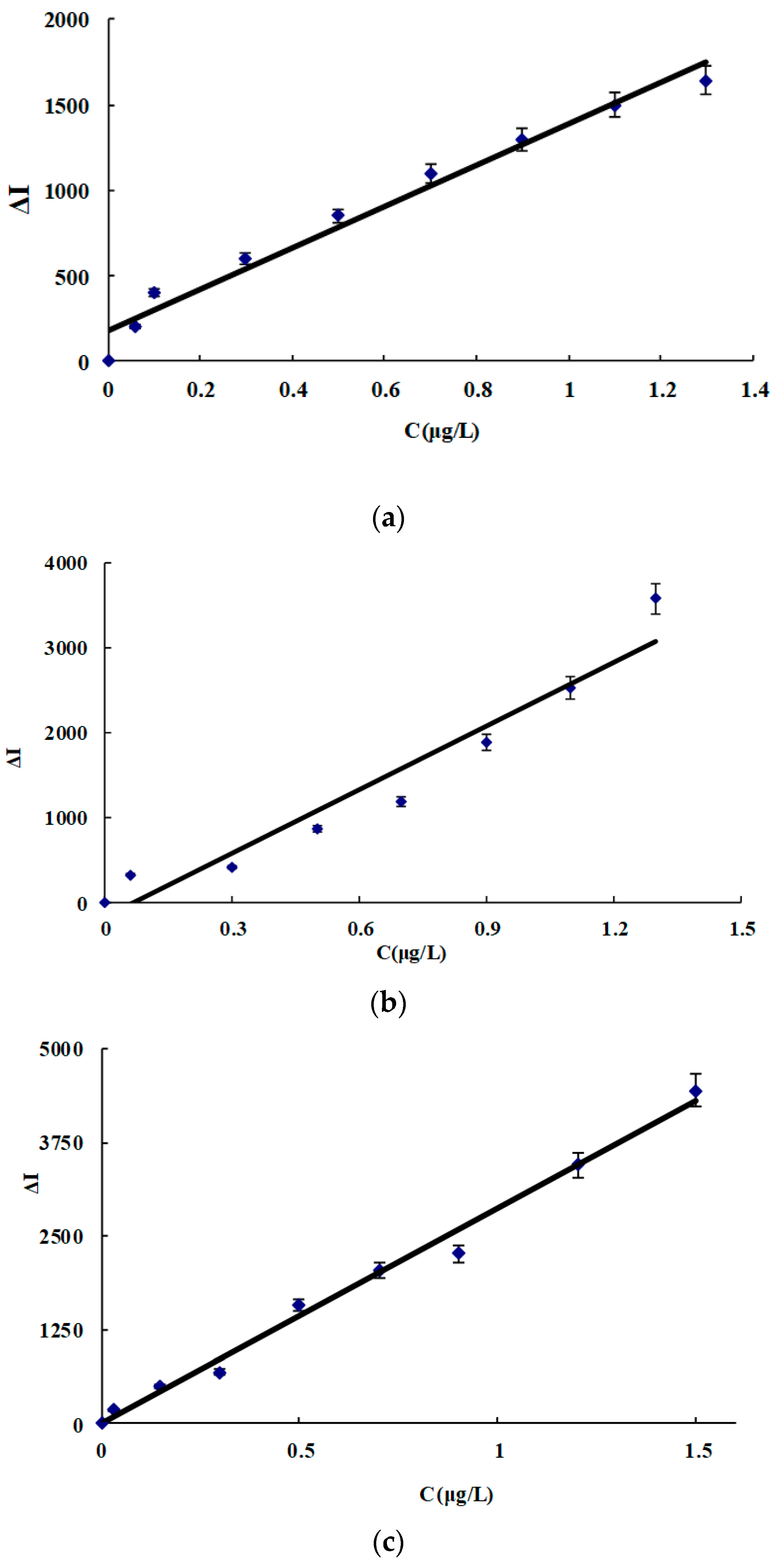
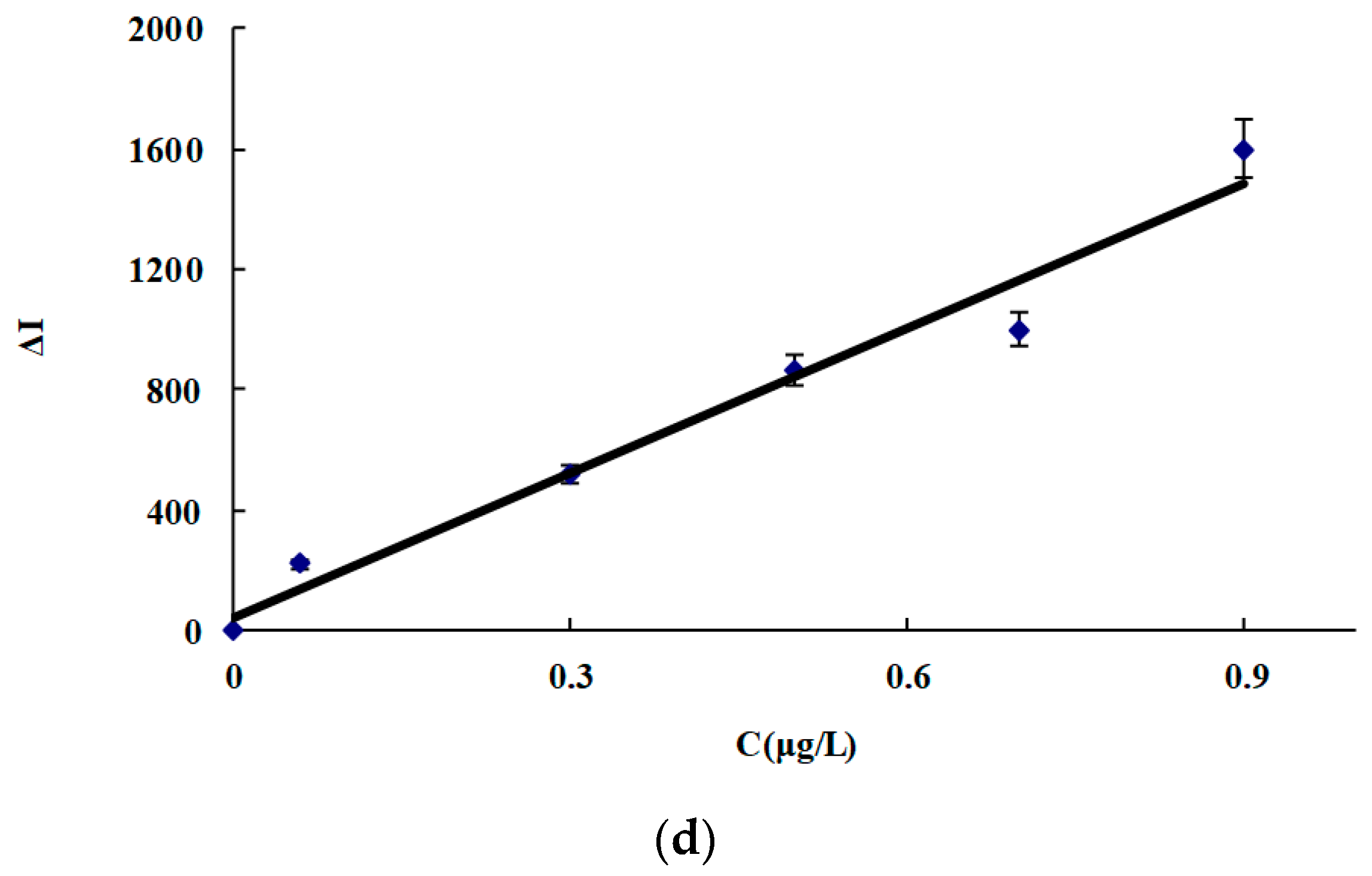
3.7. Working Curve
3.8. Sample Analysis
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Shahdordizadeh, M.; Taghdisi, S.M.; Ansari, N.; Langroodi, F.A.; Abnous, K.; Ramezani, M. Aptamer based biosensors for detection of Staphylococcus aureus. Sens. Actuators B Chem. 2017, 241, 619–635. [Google Scholar] [CrossRef]
- Huang, R.; Xi, Z.; He, N. Applications of aptamers for chemistry analysis, medicine and food security. Sci. China Chem. 2015, 58, 1122–1130. [Google Scholar] [CrossRef]
- Xu, X.; Ma, X.; Wang, H.; Wang, Z. Aptamer based SERS detection of Salmonella typhimurium, using DNA-assembled gold nanodimers. Microchim. Acta 2018, 185, 325. [Google Scholar] [CrossRef]
- Song, L.; Mao, K.; Zhou, X.; Hu, J. A novel biosensor based on Au@Ag core-shell nanoparticles for SERS detection of arsenic (III). Talanta 2016, 146, 285–290. [Google Scholar] [CrossRef]
- Wang, Y.; Yan, B.; Chen, L. SERS tags: Novel optical nanoprobes for bioanalysis. Chem. Rev. 2013, 113, 1391–1428. [Google Scholar] [CrossRef]
- Cialla, D.; März, A.; Bohme, R.; Theil, F.; Weber, K.; Schmitt, M.; Popp, J. Surface-enhanced Raman spectroscopy (SERS): Progress and trends. Anal. Bioanal. Chem. 2012, 403, 27–54. [Google Scholar] [CrossRef] [PubMed]
- Fisk, H.; Westley, C.; Turner, N.J.; Goodacre, R. Achieving optimal SERS through enhanced experimental design. J. Raman Spectrosc. 2016, 47, 59–66. [Google Scholar] [CrossRef]
- Lu, T.; Lee, Y.; Yen, Y.; Yu, C.; Chen, H. Astronomical liquid mirrors as highly ultrasensitive, broadband-operational surface-enhanced Raman scattering-active substrates. J. Colloid Interface Sci. 2016, 466, 80–90. [Google Scholar] [CrossRef] [PubMed]
- Ge, J.; Li, Y.; Wang, J.; Pu, Y.; Xue, W.; Liu, X. Green synthesis of graphene quantum dots and silver nanoparticles compounds with excellent surface enhanced Raman scattering performance. J. Alloys Compd. 2016, 663, 166–171. [Google Scholar] [CrossRef]
- Koleva, M.E.; Nedyalkov, N.N.; Atanasov, P.A.; Gerlach, J.W.; Hirsch, D.; Prager, A. Porous plasmonic nanocomposites for SERS substrates fabricated by two-step laser method. J. Alloys Compd. 2016, 665, 282–287. [Google Scholar] [CrossRef]
- Wen, G.Q.; Zhou, L.P.; Li, T.S.; Liang, A.H.; Jiang, Z.L. A sensitive surface-enhanced Raman scattering method for determination of melamine with aptamer-modified nanosilver probe. Chin. J. Chem. 2012, 30, 869–874. [Google Scholar] [CrossRef]
- Ye, L.; Wen, G.Q.; Dong, J.; Luo, Y.; Liu, Q.Y.; Liang, A.H.; Jiang, Z.L. A simple label-free rhodamine 6G SERS probe for quantitative analysis of trace As3+ in an aptamer-nanosol. RSC Adv. 2014, 4, 32960–32964. [Google Scholar] [CrossRef]
- Dong, N.; Hu, Y.; Yang, K.; Liu, J. Development of aptamer-modified SERS nanosensor and oligonucleotide chip to quantitatively detect melamine in milk with high sensitivity. Sens. Actuators B Chem. 2016, 228, 85–93. [Google Scholar] [CrossRef]
- Lin, X.M.; Cui, Y.; Xu, Y.H.; Ren, B.; Tian, Z.Q. Surface-enhanced Raman spectroscopy: Substrate-related issues. Anal. Bioanal. Chem. 2009, 394, 1729–1745. [Google Scholar] [CrossRef] [PubMed]
- Bassi, B.; Albini, B.; Agostino, A.D.; Dacarro, G.; Pallavicini, P.; Galinetto, P.; Taglietti, A. Robust, reproducible, recyclable SERS substrates: Monolayers of gold nanostars grafted on glass and coated with a thin silica layer. Nanotechnology 2019, 30, 025302. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.G.; Lei, Y. Fluorescent carbon dots and their sensing applications. TrAC Trends Anal. Chem. 2017, 89, 163–180. [Google Scholar] [CrossRef]
- Atabaev, T.S. Doped carbon dots for sensing and bioimaging applications: A minireview. Nanomaterials 2018, 8, 342. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Q.; Liang, Y.; Zhu, F.W.; Lu, S.Y.; Huang, S. Microwave-assisted one-pot synthesis of highly luminescent N-doped carbon dots for cellular imaging and multi-ion probing. Microchim. Acta 2017, 184, 2429–2438. [Google Scholar] [CrossRef]
- Wang, W.; Li, Y.M.; Cheng, L.; Cao, Z.Q.; Liu, W.G. Water-soluble and phosphorus-containing carbon dots with strong green fluorescence for cell labeling. J. Mater. Chem. B 2013, 2, 46–48. [Google Scholar] [CrossRef]
- Liu, M.L.; Chen, B.B.; Li, C.M.; Huang, C.Z. Carbon dots: Synthesis, formation mechanism, fluorescence origin and sensing applications. Green Chem. 2019, 21, 449–471. [Google Scholar] [CrossRef]
- Ahmed, G.H.G.; Laino, R.B.; Calzón, J.A.G.; Garcia, M.E.D. Highly fluorescent carbon dots as nanoprobes for sensitive and selective determination of 4-nitrophenol in surface waters. Microchim. Acta 2015, 182, 51–59. [Google Scholar] [CrossRef]
- Li, X.; Jiang, X.; Liu, Q.Y.; Liang, A.H.; Jiang, Z.L. Using N-doped carbon dots prepared rapidly by microwave as nanoprobe and nanocatalyst for fluorescence determination of ultratrace isocarbophos with label-free aptamer. Nanomaterials 2019, 9, 223. [Google Scholar] [CrossRef] [PubMed]
- Luo, J.; Shen, X.; Li, B.; Li, X.; Zhou, X. Signal amplification by strand displacement in a carbon dot based fluorometric assay for ATP. Microchim. Acta 2018, 185, 392–399. [Google Scholar] [CrossRef]
- Shi, M.; Cen, Y.; Sohail, M.; Xu, G.; Wei, F.; Ma, Y.; Song, Y.; Hu, Q. Aptamer based fluorometric β-lactoglobulin assay based on the use of magnetic nanoparticles and carbon dots. Microchim. Acta 2018, 185, 40–48. [Google Scholar] [CrossRef] [PubMed]
- Shi, H.; Zhao, G.; Liu, M. Aptamer-based colorimetric sensing of acetamiprid in soil samples: Sensitivity, selectivity and mechanism. J. Hazard. Mater. 2013, 260, 754–761. [Google Scholar] [CrossRef] [PubMed]
- El Hassani, A.K.; Dacher, M.; Gary, V. Effects of sublethal doses of acetamiprid and thiamethoxam on the behavior of the honeybee (Apis mellifera). Arch. Environ. Contam. Toxicol. 2008, 54, 653–661. [Google Scholar] [CrossRef] [PubMed]
- Imamura, T.; Yanagawa, Y.; Nishikawa, K. Two cases of acute poisoning with acetamiprid in humans. Clin. Toxicol. 2010, 48, 851–853. [Google Scholar] [CrossRef]
- Zhang, B.; Pan, X.; Venne, L. Development of a method for the determination of 9 currently used cotton pesticides by gas chromatography with electron capture detection. Talanta 2008, 75, 1055–1060. [Google Scholar] [CrossRef]
- Vichapong, J.; Burakham, R.; Srijaranai, S. Vortex-assisted surfactant-enhanced-emulsification liquid-liquid microextraction with solidification of floating organic droplet combined with HPLC for the determination of neonicotinoid pesticides. Talanta 2013, 117, 221–228. [Google Scholar] [CrossRef]
- Mohan, C.; Kumar, Y.; Madan, J. Multiresidue analysis of neonicotinoids by solid-phase extraction technique using high-performance liquid chromatography. Environ. Monit. Assess. 2010, 165, 573–576. [Google Scholar] [CrossRef] [PubMed]
- Xie, W.; Han, C.; Qian, Y. Determination of neonicotinoid pesticides residues in agricultural samples by solid-phase extraction combined with liquid chromatography-tandem mass spectrometry. J. Chromatogr. A 2011, 1218, 4426–4433. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Mobley, N.; Zhang, J. Analysis of Agricultural Residues on Tea Using d-SPE Sample Preparation with GC-NCI-MS and UHPLC-MS/MS. J. Agric. Food Chem. 2010, 58, 11553–11560. [Google Scholar] [CrossRef] [PubMed]
- Jin, D.; Qin, X.; Yu, L. A novel sensor for the detection of acetamiprid in vegetables based on its photocatalytic degradation compound. Food Chem. 2016, 194, 959–965. [Google Scholar] [CrossRef]
- Wang, J.; Wu, Y.; Zhou, P.; Yang, W.; Tao, H.; Qiu, S.; Feng, C. A novel fluorescent aptasensor for ultrasensitive and selective detection of acetamiprid pesticide based on the inner filter effect between gold nanoparticles and carbon dots. Analyst 2018, 143, 5151–5160. [Google Scholar] [CrossRef]
- Tang, X.; Li, X.; Ma, D.L.; Lu, L.; Qu, B. A label-free triplex-to-G-qadruplex molecular switch for sensitive fluorescent detection of acetamiprid. Talanta 2018, 189, 599–605. [Google Scholar] [CrossRef]
- Wang, C.; Chen, D.; Wang, Q.; Wang, Q. Aptamer-based resonance light scattering for sensitive detection of acetamiprid. Anal Sci. 2016, 32, 757–762. [Google Scholar] [CrossRef]
- Kang, Y.; Wu, T.; Han, X.; Gu, H.; Zhang, X. A needle-like reusable surface-enhanced Raman scattering substrate, and its application to the determination of acetamiprid by combining SERS and thin-layer chromatography. Microchim. Acta 2018, 185, 504. [Google Scholar] [CrossRef]
- Li, C.N.; Qin, Y.N.; Li, D.; Zhang, C.; Liang, A.H.; Wen, G.Q.; Lu, Z.J.; Jiang, Z.L. A highly sensitive enzyme catalytic SERS quantitative analysis method for ethanol with Victoria blue B molecular probe in the stable nanosilver sol substrate. Sens. Actuators B Chem. 2018, 255, 3464–3471. [Google Scholar] [CrossRef]
| Nanocatalytic System | Dynamic Range | Regress Equation | Coefficient |
|---|---|---|---|
| CDN | 1.7–13 µg/L CD | ΔI = 610C − 37 | 0.9058 |
| CDNAg1 | 11–450 µg/L CD | ΔI = 6102C + 180 | 0.9886 |
| CDNAg2 | 8–120 µg/L CD | ΔI = 8180C + 16 | 0.979 |
| CDNAg3 | 9–260 µg/L CD | ΔI = 7870C + 238 | 0.9198 |
| Apt-CDN0 | 3–26 nmol/L Apt | ΔI = 49C + 125 | 0.9307 |
| Apt-CDNAg1 | 5–30 nmol/L Apt | ΔI = 58C − 95 | 0.9387 |
| Apt-CDNAg2 | 3–36 nmol/L Apt | ΔI = 60C + 270 | 0.9598 |
| Apt-CDNAg3 | 10–50 nmol/L Apt | ΔI = 42C − 130 | 0.9508 |
| CES | Tolerance (CACT/CcES) | Error (%) | CES | Tolerance (CACT/CcES) | Error (%) |
|---|---|---|---|---|---|
| Zn2+ | 1000 | 3.0 | Fe3+ | 250 | 6.5 |
| Ca2+ | 1000 | −7.0 | Bi3+ | 250 | 2.9 |
| Ni2+ | 1000 | 5.0 | Cu2+ | 250 | 4.5 |
| Mn2+ | 1000 | 3.0 | Pb2+ | 250 | 5.0 |
| Co2+ | 1000 | 4.7 | Al3+ | 250 | 6.0 |
| NH4Cl | 1000 | 4.8 | Cr6+ | 100 | 6.0 |
| CO32− | 1000 | 6.5 | Fe2+ | 100 | 8.0 |
| Ba2+ | 500 | −5.4 | Hg2+ | 250 | −5.0 |
| Mg2+ | 500 | −5.1 | imidacloprid | 200 | 6.0 |
| K+ | 500 | −4.2 | atrazine | 200 | 3.0 |
| HCO3− | 500 | 5.2 | carbendazim | 100 | 7.0 |
| NO2− | 400 | −4.0 | chlorpyrifos | 200 | 4.3 |
| System | Linear Range (μg/L) | Regress Equation | Coefficient | DL (μg/L) |
|---|---|---|---|---|
| CDN | 0.06−1.3 | ΔI = 1240C + 180 | 0.9754 | 0.026 |
| CDNAg1 | 0.06−1.3 | ΔI = 2500C − 170 | 0.9367 | 0.029 |
| CDNAg2 | 0.01−1.5 | ΔI = 2800C + 20 | 0.9967 | 0.006 |
| CDNAg3 | 0.06−0.9 | ΔI = 1650C + 50 | 0.9832 | 0.027 |
| Method | Principle | Linear Range | Detection Limt | Comment | Ref. |
|---|---|---|---|---|---|
| Fluorescence | The aptasensor for ACT based on the inner filter effect between nanogold and CD. | 5–100 μg/L | 1.08 μg/L | High precision but low sensitivity. | [34] |
| Fluorescence | A label-free triplex-to-G-quadruplex molecular switch for ACT. | 10–500 nM | 2.38 nM | Fast but low sensitivity. | [35] |
| Resonance Light Scattering | Nanogold modified Apt-based resonance light scattering for ACT. | 0–100 nM | 1.2 nM | High sensitivity but complicated operation. | [36] |
| Colorimetry | Aptamer-based colorimetric sensing of ACT in soil samples based on aggregation of Au nanoparticles. | 0.075–7.5 μM | 5 nM | Fast and selective but low sensitivity. | [25] |
| SERS | Gold on carbon fiber needle-like SERS substrate for ACT by combining with thin-layer chromatography. | 0.1–10 μg/mL | 0.05 μg/mL | Selective but low sensitivity. | [37] |
| DBDox SERS probe | Apt used to modulate CD catalysis to generate DBDox to detect ACT. | 0.01–1.5 μg/mL | 0.006 μg/L | High sensitivity and good selectivity. | This method |
| Sample | Content (μg/L) | Added (μg/L) | Found (μg/L) | Recovery (%) | Relative Standard Deviation (%) | Ref. Results (μg/L) |
|---|---|---|---|---|---|---|
| Pakchoi | — | 0.1 | 0. 0960 | 96.0 | 5.0 | — |
| Cucumber | 0.16 | 0.1 | 0.0957 | 95.7 | 3.5 | 0.15 |
| Tomato | 0.10 | 0.1 | 0.0995 | 99.5 | 5.6 | 0.12 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Feng, X.; Li, C.; Liang, A.; Luo, Y.; Jiang, Z. Doped N/Ag Carbon Dot Catalytic Amplification SERS Strategy for Acetamiprid Coupled Aptamer with 3,3′-Dimethylbiphenyl-4,4′-diamine Oxidizing Reaction. Nanomaterials 2019, 9, 480. https://doi.org/10.3390/nano9030480
Feng X, Li C, Liang A, Luo Y, Jiang Z. Doped N/Ag Carbon Dot Catalytic Amplification SERS Strategy for Acetamiprid Coupled Aptamer with 3,3′-Dimethylbiphenyl-4,4′-diamine Oxidizing Reaction. Nanomaterials. 2019; 9(3):480. https://doi.org/10.3390/nano9030480
Chicago/Turabian StyleFeng, Xiaozhen, Chongning Li, Aihui Liang, Yanghe Luo, and Zhiliang Jiang. 2019. "Doped N/Ag Carbon Dot Catalytic Amplification SERS Strategy for Acetamiprid Coupled Aptamer with 3,3′-Dimethylbiphenyl-4,4′-diamine Oxidizing Reaction" Nanomaterials 9, no. 3: 480. https://doi.org/10.3390/nano9030480
APA StyleFeng, X., Li, C., Liang, A., Luo, Y., & Jiang, Z. (2019). Doped N/Ag Carbon Dot Catalytic Amplification SERS Strategy for Acetamiprid Coupled Aptamer with 3,3′-Dimethylbiphenyl-4,4′-diamine Oxidizing Reaction. Nanomaterials, 9(3), 480. https://doi.org/10.3390/nano9030480






