Seed-Mediated Electroless Deposition of Gold Nanoparticles for Highly Uniform and Efficient SERS Enhancement
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Instrumentation
2.3. Procedures
2.3.1. Preparation of Gold Seed Solutions
2.3.2. Fabrication of Large-Area and Uniform Substrate Films by SMED
2.3.3. SERS Spectroscopy of CV on SERS-Active Au Nanoparticles Film
3. Results and Discussion
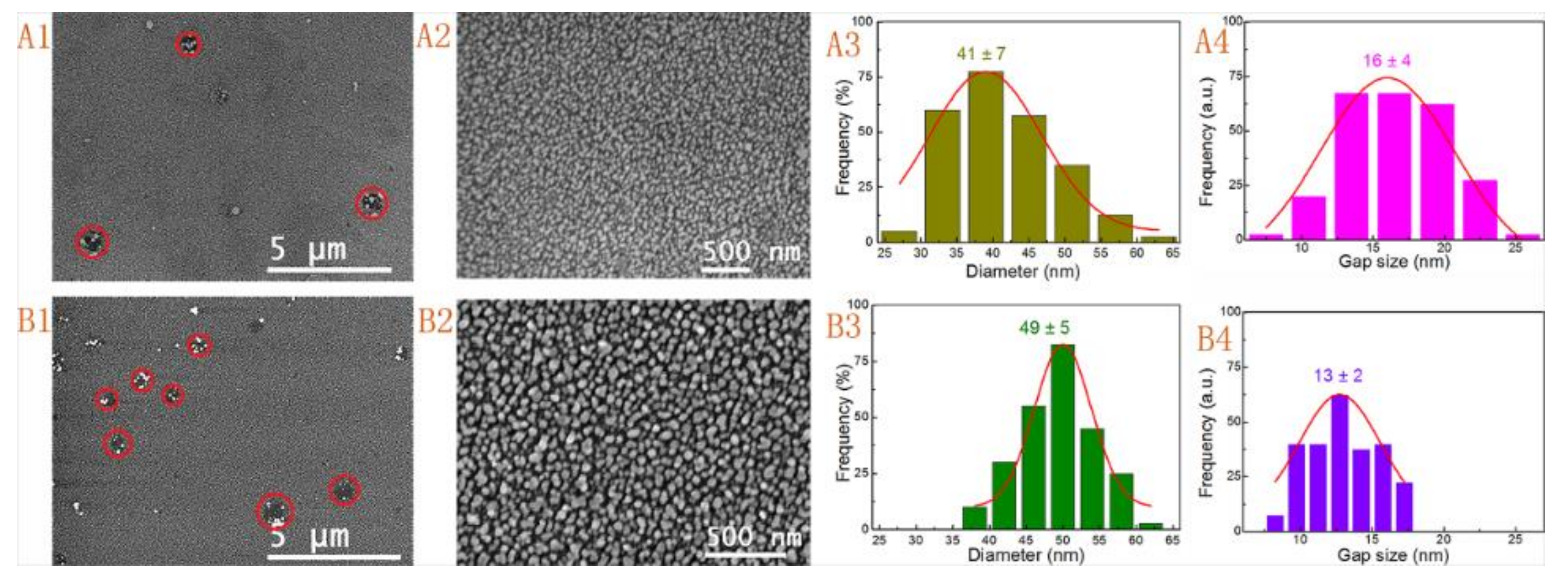
3.1. Effect of Gold Seed
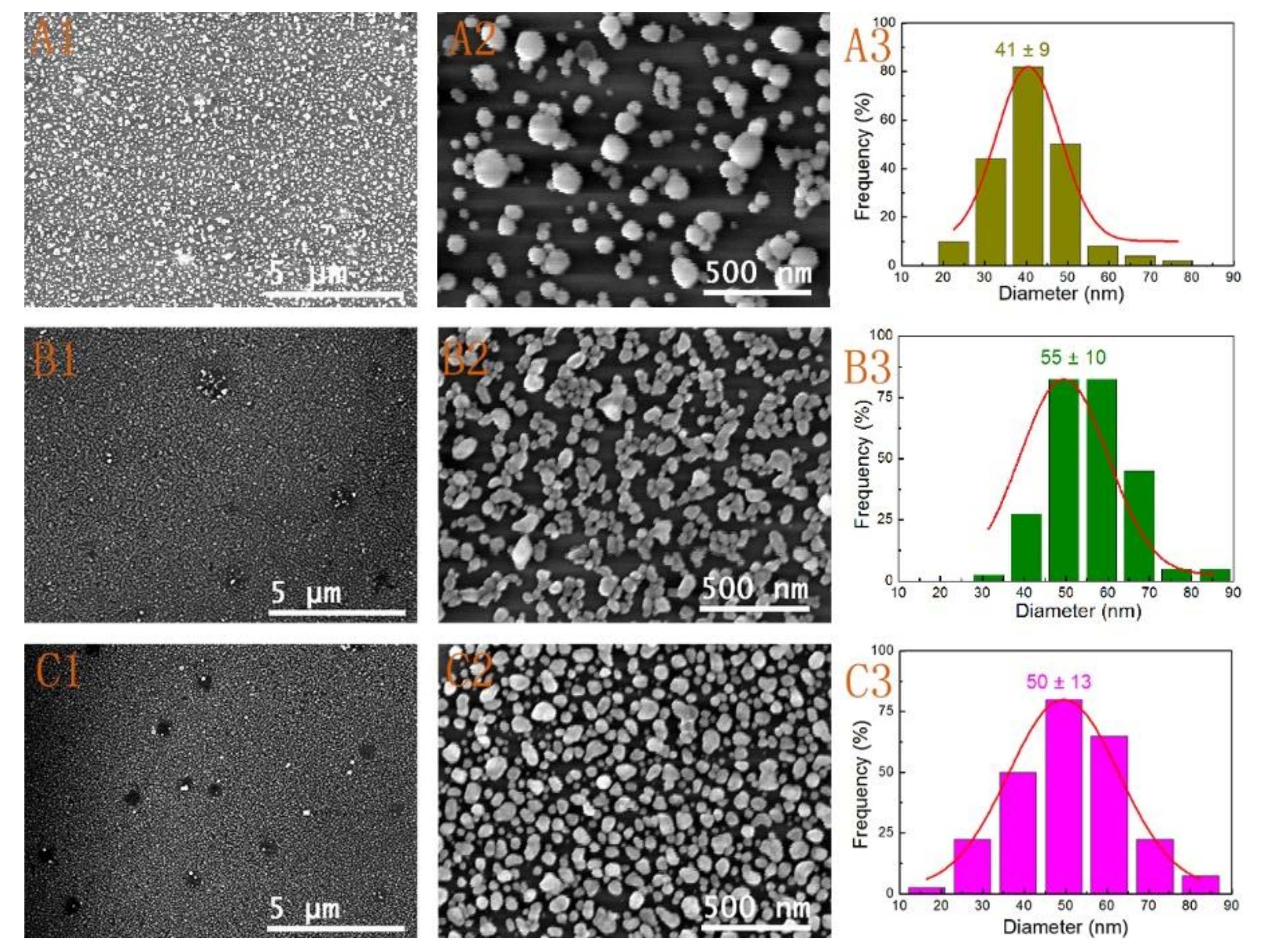
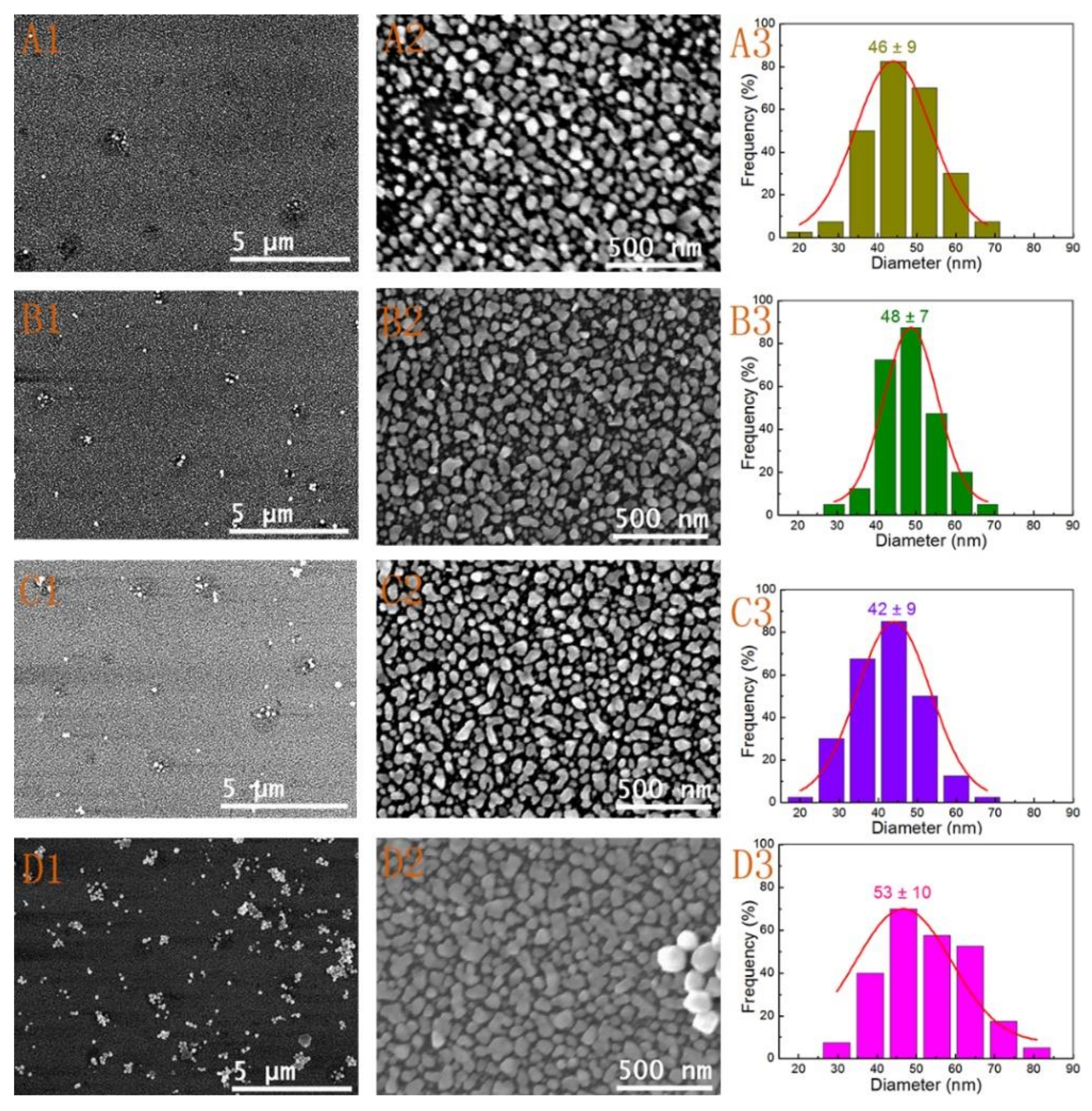
3.2. Effect of Reactant Concentration
3.3. Effect of Reaction Time
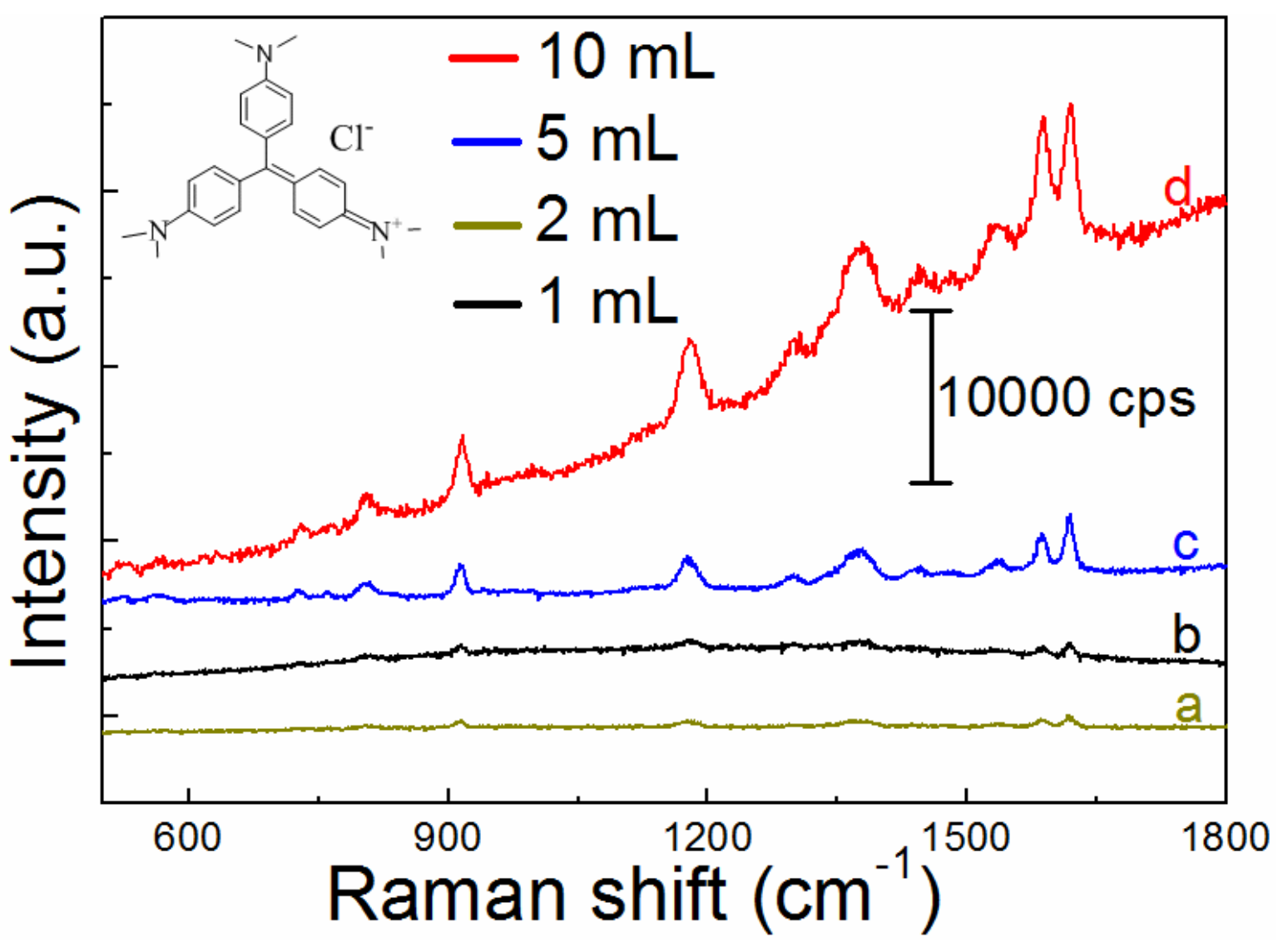
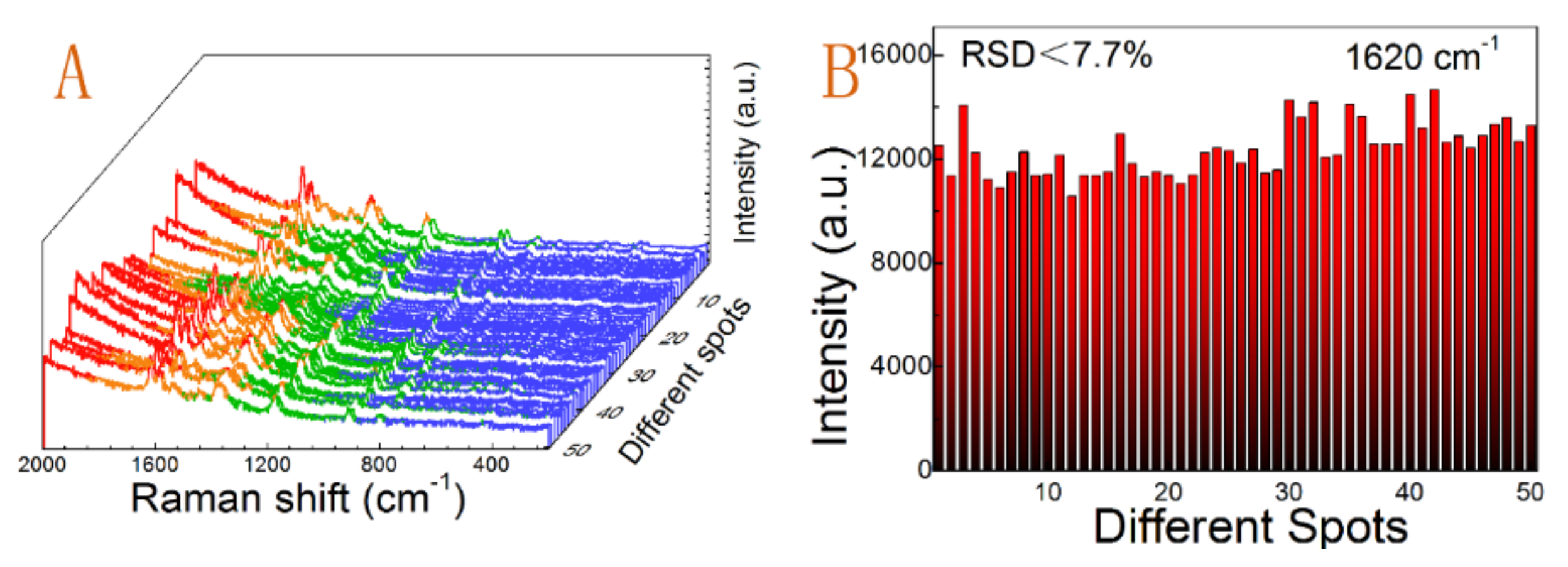
3.4. SERS Enhancement Performance
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lon, A.P.; Choi, H.C.; Ribbe, A.E.; Buriak, J.M. Controlled Electroless Deposition of Noble Metal Nanoparticle Films on Germanium Surfaces. Nano Lett. 2002, 2, 1067–1071. [Google Scholar] [CrossRef]
- Brenner, A. Nickel plating on steel by chemical reduction. J. Res. Natl. Bur. Stand. 1946, 37, 31–34. [Google Scholar] [CrossRef]
- Tsuneyoshi, T.; Ono, T. Metal-coated microcapsules with tunable magnetic properties synthesized via electroless plating. Mater. Sci. Eng. B 2017, 222, 49–54. [Google Scholar] [CrossRef]
- Raj, M.A.; John, S.A. Assembly of gold nanoparticles on graphene film via electroless deposition: Spontaneous reduction of Au3+ ions by graphene film. RSC Adv. 2015, 5, 4964–4971. [Google Scholar] [CrossRef]
- Milazzo, R.G.; Privitera, S.; Litrico, G.; Scalese, S.; Mirabella, S.; La Via, F.; Lombardo, S.; Rimini, E. Formation, Morphology, and Optical Properties of Electroless Deposited Gold Nanoparticles on 3C-SiC. J. Phys. Chem. C 2017, 121, 4304–4311. [Google Scholar] [CrossRef]
- Yadav, R.; Balasubramanian, K. Metallization of electrospun PAN nanofibers via electroless gold plating. RSC Adv. 2015, 5, 24990–24996. [Google Scholar] [CrossRef]
- Cheng, M.L.; Yang, J. Seed-mediated growth method for electroless deposition of AgNPs on glass substrates for use in SERS measurements. J. Raman Spectrosc. 2010, 41, 167–174. [Google Scholar] [CrossRef]
- Ahmed, S.R.; Kim, J.; Tran, V.T.; Suzuki, T.; Neethirajan, S.; Lee, J.; Park, E.Y. In situ self-assembly of gold nanoparticles on hydrophilic and hydrophobic substrates for influenza virus-sensing platform. Sci. Rep. 2017, 7, 44495. [Google Scholar] [CrossRef]
- Sánchez-Iglesias, A.; Aldeanueva-Potel, P.; Ni, W.; Pérez-Juste, J.; Pastoriza-Santos, I.; Alvarez-Puebla, R.A.; Mbenkum, B.N.; Liz-Marzán, L.M. Chemical seeded growth of Ag nanoparticle arrays and their application as reproducible SERS substrates. Nano Today 2010, 5, 21–27. [Google Scholar] [CrossRef]
- Hou, Z.; Abbott, N.L.; Stroeve, P. Electroless Gold as a Substrate for Self-Assembled Monolayers. Langmuir 1998, 14, 3287–3297. [Google Scholar] [CrossRef]
- Cui, J.H.; Yang, Y.H.; Zheng, M.T.; Liu, Y.L.; Xiao, Y.; Lei, B.F.; Chen, W. Facile fabrication of graphene oxide loaded with silver nanoparticles as antifungal materials. Mater. Res. Express 2014, 1, 045007. [Google Scholar] [CrossRef]
- Wang, C.; Xu, Y.; Zhao, H.; Gang, C.; Lai, C.; Liao, X.; Wang, R. Detection and analysis of SERS effect of nano gold by self-assembly chemical plating composite method. Appl. Surf. Sci. 2015, 353, 750–756. [Google Scholar] [CrossRef]
- Zhang, Y.H.; Shi, Y.; Wu, M.M.; Zhang, K.; Man, B.Y.; Liu, M. Synthesis and Surface-Enhanced Raman Scattering of Ultrathin SnSe2 Nanoflakes by Chemical Vapor Deposition. Nanomaterials 2018, 8, 515. [Google Scholar] [CrossRef] [PubMed]
- Dipalo, M.; Messina, G.C.; Amin, H.; La Rocca, R.; Shalabaeva, V.; Simi, A.; Maccione, A.; Zilio, P.; Berdondini, L.; De Angelis, F. 3D plasmonic nanoantennas integrated with MEA biosensors. Nanoscale 2015, 7, 3703–3711. [Google Scholar] [CrossRef] [PubMed]
- Lee, T.; Wi, J.S.; Oh, A.; Na, H.K.; Lee, J.; Lee, K.; Lee, T.G.; Haam, S. Highly robust, uniform and ultra-sensitive surface-enhanced Raman scattering substrates for microRNA detection fabricated by using silver nanostructures grown in gold nanobowls. Nanoscale 2018, 10, 3680–3687. [Google Scholar] [CrossRef] [PubMed]
- Komiyama, H. Fabrication of a Vertically Aligned Au Nanorod Array via Block-Copolymer-Templated Electroplating. Chemistryselect 2018, 3, 4944–4950. [Google Scholar] [CrossRef]
- Mohacek-Grosev, V.; Gebavi, H.; Bonifacio, A.; Sergo, V.; Dakovic, M.; Bajuk-Bogdanovic, D. Binding of p-mercaptobenzoic acid and adenine to gold-coated electroless etched silicon nanowires studied by surface-enhanced Raman scattering. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2018, 200, 102–109. [Google Scholar] [CrossRef]
- Pliatsikas, N.; Vourlias, G.; Patsalas, P. Facile Synthesis of Tunable Nanostructured Plasmonic Templates by Electroless Deposition. Plasmonics 2018, 13, 467–474. [Google Scholar] [CrossRef]
- Wu, C.; Cai, C.; Yang, P.; Liu, W. An atom-induced situ-growth method for constructing a highly sensitive and reproducible large area SERS substrate. Colloids Surf. Phys. Eng. Asp. 2018, 545, 205–211. [Google Scholar] [CrossRef]
- Wu, Y.; Hang, T.; Komadina, J.; Ling, H.; Li, M. High-adhesive superhydrophobic 3D nanostructured silver films applied as sensitive, long-lived, reproducible and recyclable SERS substrates. Nanoscale 2014, 6, 9720–9726. [Google Scholar] [CrossRef]
- Yap, F.L.; Thoniyot, P.; Krishnan, S.; Krishnamoorthy, S. Nanoparticle Cluster Arrays for High-Performance SERS through Directed Self-Assembly on Flat Substrates and on Optical Fibers. ACS Nano 2012, 6, 2056–2070. [Google Scholar] [CrossRef] [PubMed]
- Ding, S.Y.; You, E.M.; Tian, Z.Q.; Moskovits, M. Electromagnetic theories of surface-enhanced Raman spectroscopy. Chem. Soc. Rev. 2017, 46, 4042–4076. [Google Scholar] [CrossRef] [PubMed]
- Lu, X.F.; Huang, Y.J.; Liu, B.Q.; Zhang, L.; Song, L.P.; Zhang, J.W.; Zhang, A.F.; Chen, T. Light-Controlled Shrinkage of Large-Area Gold Nanoparticle Monolayer Film for Tunable SERS Activity. Chem. Mater. 2018, 30, 1989–1997. [Google Scholar] [CrossRef]
- Ye, X.; Qi, L. Two-dimensionally patterned nanostructures based on monolayer colloidal crystals: Controllable fabrication, assembly, and applications. Nano Today 2011, 6, 608–631. [Google Scholar] [CrossRef]
- Yi, Z.; Li, X.; Xu, X.; Chen, X.; Ye, X.; Yi, Y.; Duan, T.; Tang, Y.; Liu, J.; Yi, Y. Nanostrip-Induced High Tunability Multipolar Fano Resonances in a Au Ring-Strip Nanosystem. Nanomaterials 2018, 8, 568. [Google Scholar] [CrossRef] [PubMed]
- Xu, D.; Teng, F.; Wang, Z.; Lu, N. Droplet-confined Electroless Deposition of Silver Nanoparticles on Ordered Superhydrophobic Structures for High Uniform SERS Measurements. ACS Appl. Mater. Interfaces 2017, 9, 21548–21553. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.X.; Wu, F.F.; Xu, D.R.; Li, N.; Lu, N. Space confined electroless deposition of silver nanoparticles for highly-uniform SERS detection. Sens. Actuators B Chem. 2018, 255, 1401–1406. [Google Scholar] [CrossRef]
- Steinigeweg, D.; Schlucker, S. Monodispersity and size control in the synthesis of 20–100 nm quasi-spherical silver nanoparticles by citrate and ascorbic acid reduction in glycerol-water mixtures. Chem. Commun. 2012, 48, 8682–8684. [Google Scholar] [CrossRef]
- Wan, Y.; Guo, Z.; Jiang, X.; Fang, K.; Lu, X.; Zhang, Y.; Gu, N. Quasi-spherical silver nanoparticles: Aqueous synthesis and size control by the seed-mediated Lee–Meisel method. J. Colloid Interface Sci. 2013, 394, 263–268. [Google Scholar] [CrossRef]
- Wang, Q.; Wang, Z.; Li, Z.; Xiao, J.; Shan, H.; Fang, Z.; Qi, L. Controlled growth and shape-directed self-assembly of gold nanoarrows. Sci. Adv. 2017, 3, 568. [Google Scholar] [CrossRef]
- Evcimen, N.I.; Coskun, S.; Kozanoglu, D.; Ertas, G.; Unalan, H.E.; Esenturk, E.N. Growth of branched gold nanoparticles on solid surfaces and their use as surface-enhanced Raman scattering substrates. RSC Adv. 2015, 5, 101656–101663. [Google Scholar] [CrossRef]
- Kambayashi, M.; Zhang, J.; Oyama, M. Crystal Growth of Gold Nanoparticles on Indium Tin Oxides in the Absence and Presence of 3-Mercaptopropyl-trimethoxysilane. Cryst. Growth Des. 2005, 5, 81–84. [Google Scholar] [CrossRef]
- Wang, J.; Cao, X.; Li, L.; Li, T.; Wang, R. Electrochemical Seed-Mediated Growth of Surface-Enhanced Raman Scattering Active Au(111)-Like Nanoparticles on Indium Tin Oxide Electrodes. J. Phys. Chem. C 2013, 117, 15817–15828. [Google Scholar] [CrossRef]
- Philip, A.; Ankudze, B.; Pakkanen, T.T. Polyethylenimine-assisted seed-mediated synthesis of gold nanoparticles for surface-enhanced Raman scattering studies. Appl. Surf. Sci. 2018, 444, 243–252. [Google Scholar] [CrossRef]
- Lim, D.K.; Jeon, K.S.; Hwang, J.H.; Kim, H.; Kwon, S.; Suh, Y.D.; Nam, J.M. Highly uniform and reproducible surface-enhanced Raman scattering from DNA-tailorable nanoparticles with 1-nm interior gap. Nat. Nanotechnol. 2011, 6, 452–460. [Google Scholar] [CrossRef] [PubMed]
- Jana, N.R.; Gearheart, L.; Murphy, C.J. Wet chemical synthesis of high aspect ratio cylindrical gold nanorods. J. Phys. Chem. B 2001, 105, 4065–4067. [Google Scholar] [CrossRef]
- Leng, W.; Kelley, A.M. Surface-Enhanced Hyper-Raman Spectra and Enhancement Factors for Three SERS Chromophores. SEHRS Spectra on Ag Films at Pulse Energies below 2 pJ. J. Am. Chem. Soc. 2006, 128, 3492–3493. [Google Scholar] [CrossRef] [PubMed]
- Dadosh, T.; Sperling, J.; Bryant, G.W.; Breslow, R.; Shegai, T.; Dyshel, M.; Haran, G.; Bar-Joseph, I. Plasmonic Control of the Shape of the Raman Spectrum of a Single Molecule in a Silver Nanoparticle Dimer. ACS Nano 2009, 3, 1988–1994. [Google Scholar] [CrossRef]
- Lee, K.; Irudayaraj, J. Correct Spectral Conversion between Surface-Enhanced Raman and Plasmon Resonance Scattering from Nanoparticle Dimers for Single-Molecule Detection. Small 2013, 9, 1106–1115. [Google Scholar] [CrossRef]
- Haiss, W.; Thanh, N.T.; Aveyard, J.; Fernig, D.G. Determination of size and concentration of gold nanoparticles from UV-vis spectra. Anal. Chem. 2007, 79, 4215–4221. [Google Scholar] [CrossRef]
- Jiang, X.C.; Pileni, M.P. Gold nanorods: Influence of various parameters as seeds, solvent, surfactant on shape control. Colloids Surf. Phys. Eng. Asp. 2007, 295, 228–232. [Google Scholar] [CrossRef]
- Ma, H.; Sun, J.; Yuan, Z.; Chao, B.; Xia, S.; Tong, Z. Label-free immunosensor based on one-step electrodeposition of chitosan-gold nanoparticles biocompatible film on Au microelectrode for determination of aflatoxin B 1 in maize. Biosens. Bioelectron. 2016, 80, 222–229. [Google Scholar] [CrossRef] [PubMed]
- Yang, G.; Nanda, J.; Wang, B.; Gang, C.; Hallinan, D.T. Self-Assembly of Large Gold Nanoparticles for Surface-Enhanced Raman Spectroscopy. ACS Appl. Mater. Interfaces 2017, 9, 13457–13470. [Google Scholar] [CrossRef] [PubMed]
- Tang, J.; Fu, X.; Ou, Q.; Gao, K.; Man, S.-Q.; Guo, J.; Liu, Y. Hydroxide assisted synthesis of monodisperse and biocompatible gold nanoparticles with dextran. Mater. Sci. Eng. C 2018, 93, 759–767. [Google Scholar] [CrossRef] [PubMed]
- Eustis, S.; El-Sayed, M.A. Molecular mechanism of the photochemical generation of gold nanoparticles in ethylene glycol: Support for the disproportionation mechanism. J. Phys. Chem. B 2006, 110, 14014–14019. [Google Scholar] [CrossRef] [PubMed]
- Oyama, T.; Yamaguchi, S.; Rahman, M.R.; Okajima, T.; Ohsaka, T.; Oyama, N. EQCM Study of the (AuCl4)-Cl-III (-)- (AuCl2)-Cl-I (-)-Au(0) Redox System in 1-Ethyl-3-methylimidazolium Tetrafluoroborate Room-Temperature Ionic Liquid. Langmuir 2010, 26, 9069–9075. [Google Scholar] [CrossRef] [PubMed]
- Tang, J.; Zhao, Q.; Zhang, N.; Man, S.-Q. Facile fabrication of large-area and uniform silica nanospheres monolayer for efficient surface-enhanced Raman scattering. Appl. Surf. Sci. 2014, 308, 247–252. [Google Scholar] [CrossRef]
- Erdemir, S.; Kocyigit, O. Reversible “OFF-ON” fluorescent and colorimetric sensor based benzothiazole-bisphenol A for fluoride in MeCN. Sens. Actuators B: Chem. 2015, 221, 900–905. [Google Scholar] [CrossRef]
- Cecchini, M.P.; Turek, V.A.; Paget, J.; Kornyshev, A.A.; Edel, J.B. Self-assembled nanoparticle arrays for multiphase trace analyte detection. Nat. Mater. 2013, 12, 165–171. [Google Scholar] [CrossRef]
- Botta, R.; Upender, G.; Sathyavathi, R.; Narayana Rao, D.; Bansal, C. Silver nanoclusters films for single molecule detection using Surface Enhanced Raman Scattering (SERS). Mater. Chem. Phys. 2013, 137, 699–703. [Google Scholar] [CrossRef]
- Su, L.; Jia, W.; Manuzzi, D.P.; Zhang, L.; Li, X.; Gu, Z.; Lei, Y. Highly sensitive surface-enhanced Raman scattering using vertically aligned silver nanopetals. RSC Adv. 2012, 2, 1439–1443. [Google Scholar] [CrossRef]
- Lin, X.M.; Cui, Y.; Xu, Y.H.; Ren, B.; Tian, Z.Q. Surface-enhanced Raman spectroscopy: Substrate-related issues. Anal. Bioanal. Chem. 2009, 394, 1729–1745. [Google Scholar] [CrossRef] [PubMed]
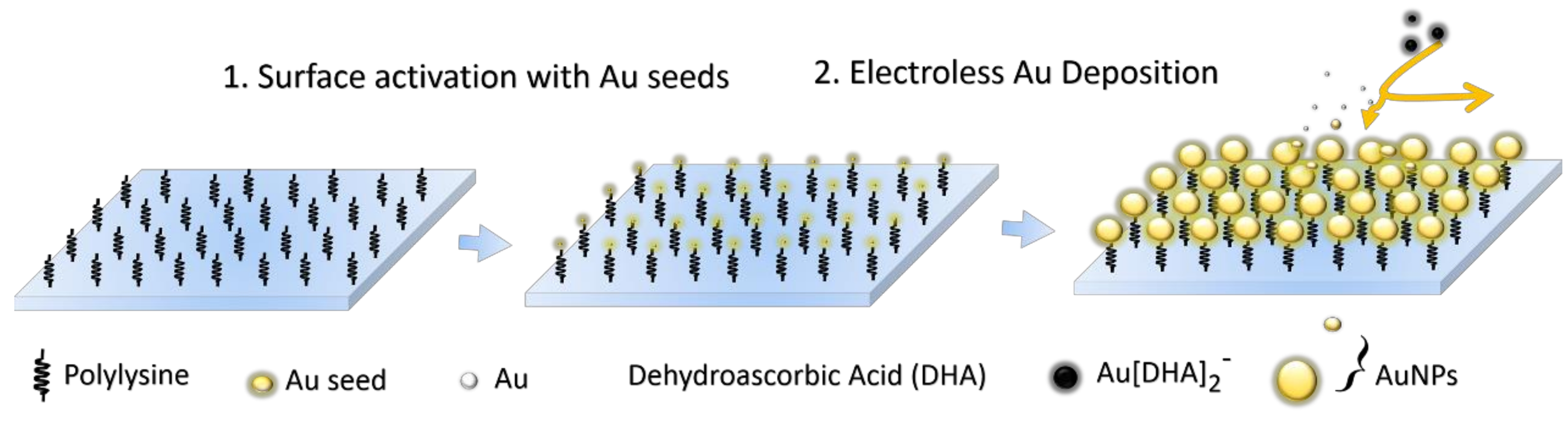
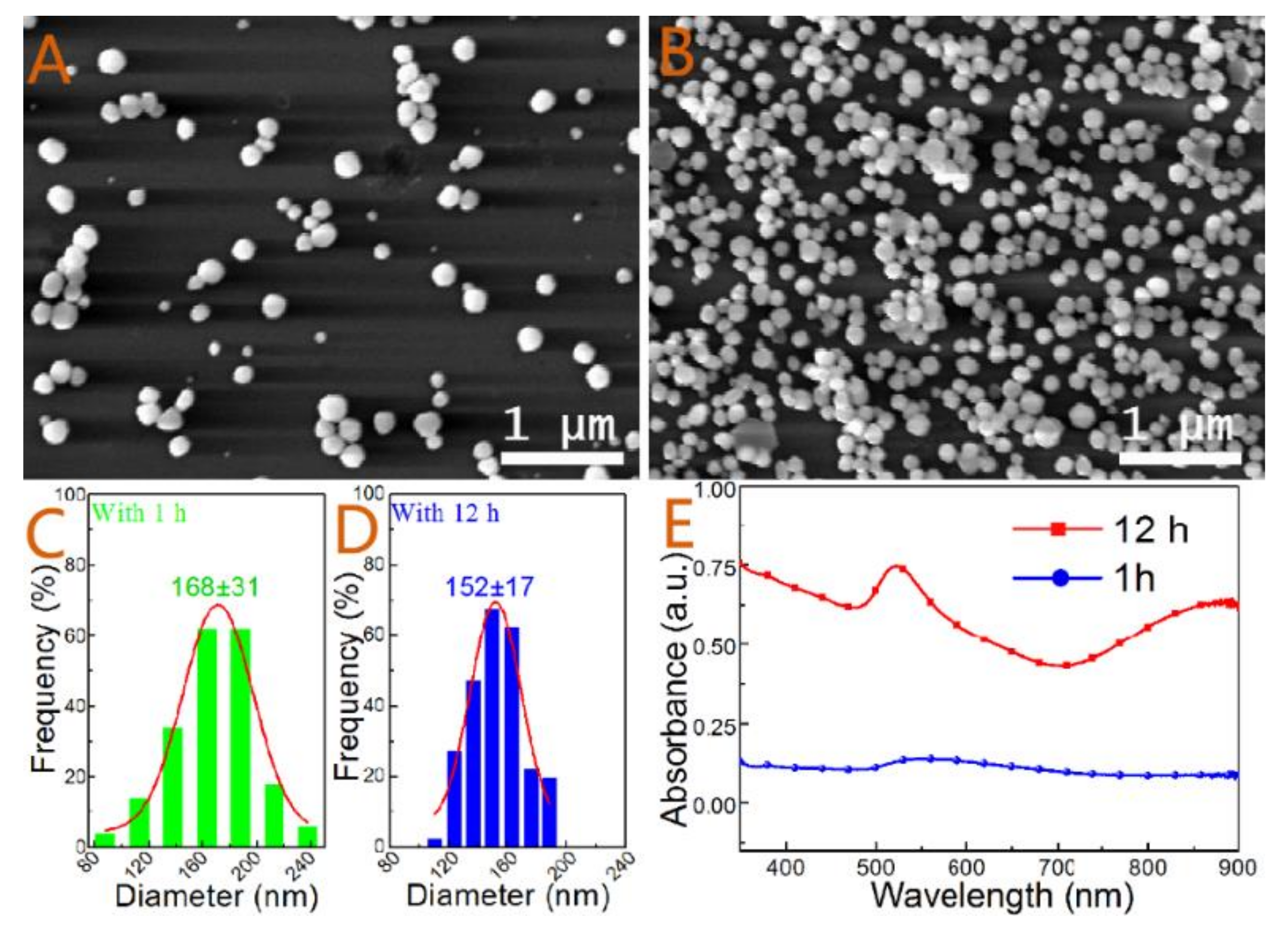
| Sample | Seeds | Reactant Concentration | Soaking Time | Figure ID |
|---|---|---|---|---|
| 1 | - | 10 mL | 1 h | 2(A,C) |
| 2 | - | 10 mL | 12 h | 2(B,D) |
| 3 | Seed I | 10 mL | 30 min | 3(A1–A4) |
| 4 | Seed II | 10 mL | 12 h | 3(B1–B4) |
| 5 | Seed I | 1 mL | 12 h | 4(A1–A3) |
| 6 | Seed I | 2 mL | 12 h | 4(B1–B3) |
| 7 | Seed I | 5 mL | 12 h | 4(C1–C3) |
| 8 | Seed II | 5 mL | 15 min | 5(A1–A3) |
| 9 | Seed II | 10 mL | 30 min | 5(B1–B3) |
| 10 | Seed II | 10 mL | 5 h | 5(C1–C3) |
| 11 | Seed II | 10 mL | 20 h | 5(D1–D3) |
| 12 | Seed I | 10 mL | 2 h | S3(A1–C3) |
| Peak Position (cm−1) | 1620 | 1589 | 1377 | 1183 | 917 | 807 |
|---|---|---|---|---|---|---|
| RSD values | 0.077 | 0.069 | 0.059 | 0.047 | 0.036 | 0.031 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tang, J.; Ou, Q.; Zhou, H.; Qi, L.; Man, S. Seed-Mediated Electroless Deposition of Gold Nanoparticles for Highly Uniform and Efficient SERS Enhancement. Nanomaterials 2019, 9, 185. https://doi.org/10.3390/nano9020185
Tang J, Ou Q, Zhou H, Qi L, Man S. Seed-Mediated Electroless Deposition of Gold Nanoparticles for Highly Uniform and Efficient SERS Enhancement. Nanomaterials. 2019; 9(2):185. https://doi.org/10.3390/nano9020185
Chicago/Turabian StyleTang, Junqi, Quanhong Ou, Haichun Zhou, Limin Qi, and Shiqing Man. 2019. "Seed-Mediated Electroless Deposition of Gold Nanoparticles for Highly Uniform and Efficient SERS Enhancement" Nanomaterials 9, no. 2: 185. https://doi.org/10.3390/nano9020185
APA StyleTang, J., Ou, Q., Zhou, H., Qi, L., & Man, S. (2019). Seed-Mediated Electroless Deposition of Gold Nanoparticles for Highly Uniform and Efficient SERS Enhancement. Nanomaterials, 9(2), 185. https://doi.org/10.3390/nano9020185




