One-Step Synthesis of Self-Supported Ni3S2/NiS Composite Film on Ni Foam by Electrodeposition for High-Performance Supercapacitors
Abstract
:1. Introduction
2. Experimental Section
2.1. Materials and Reagents
2.2. Preparation of Ni3S2/NiS Composite on Ni Foam
2.3. Material Characterizations
2.4. Electrochemical Measurement
3. Results and Discussion
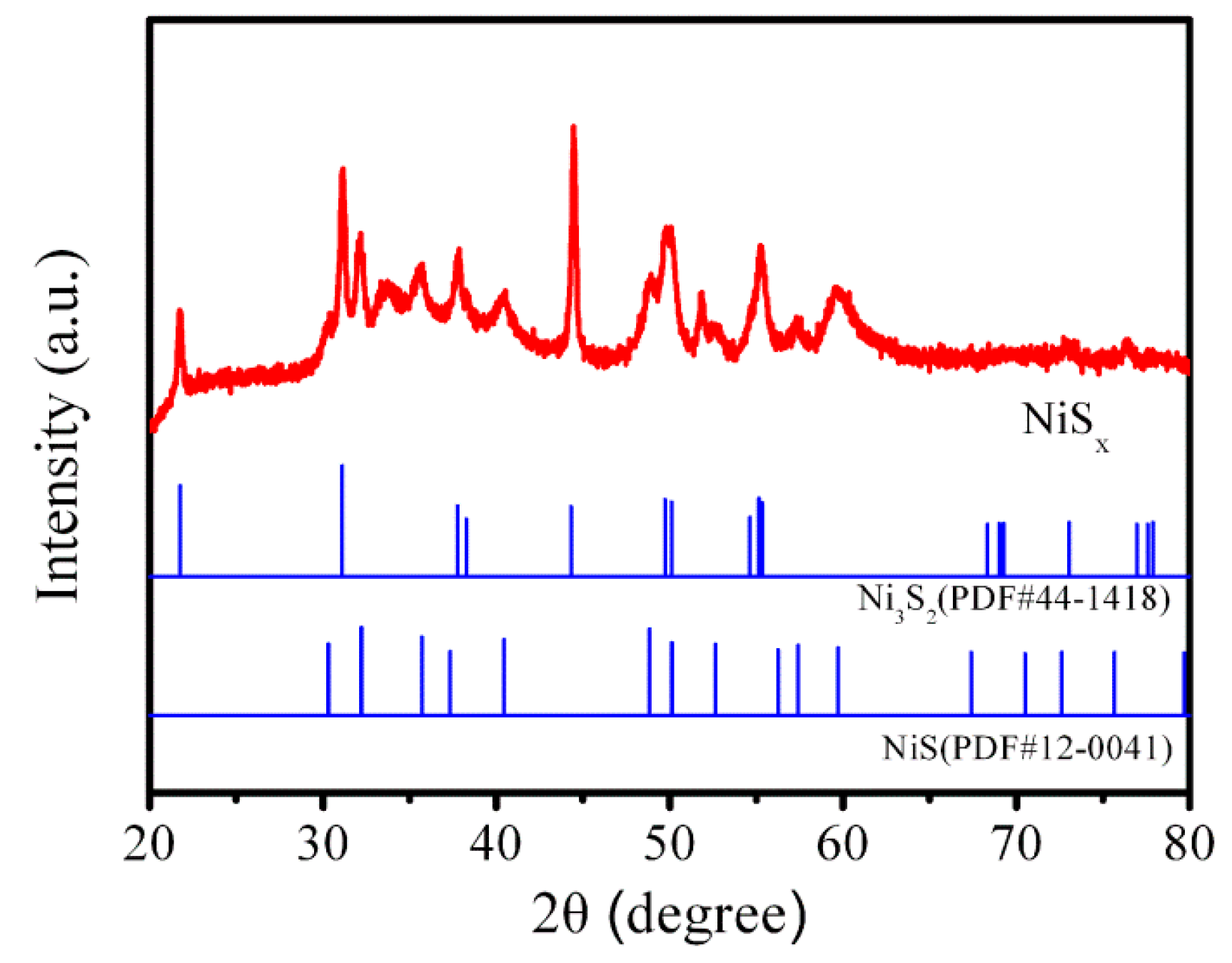
3.1. Characteristics
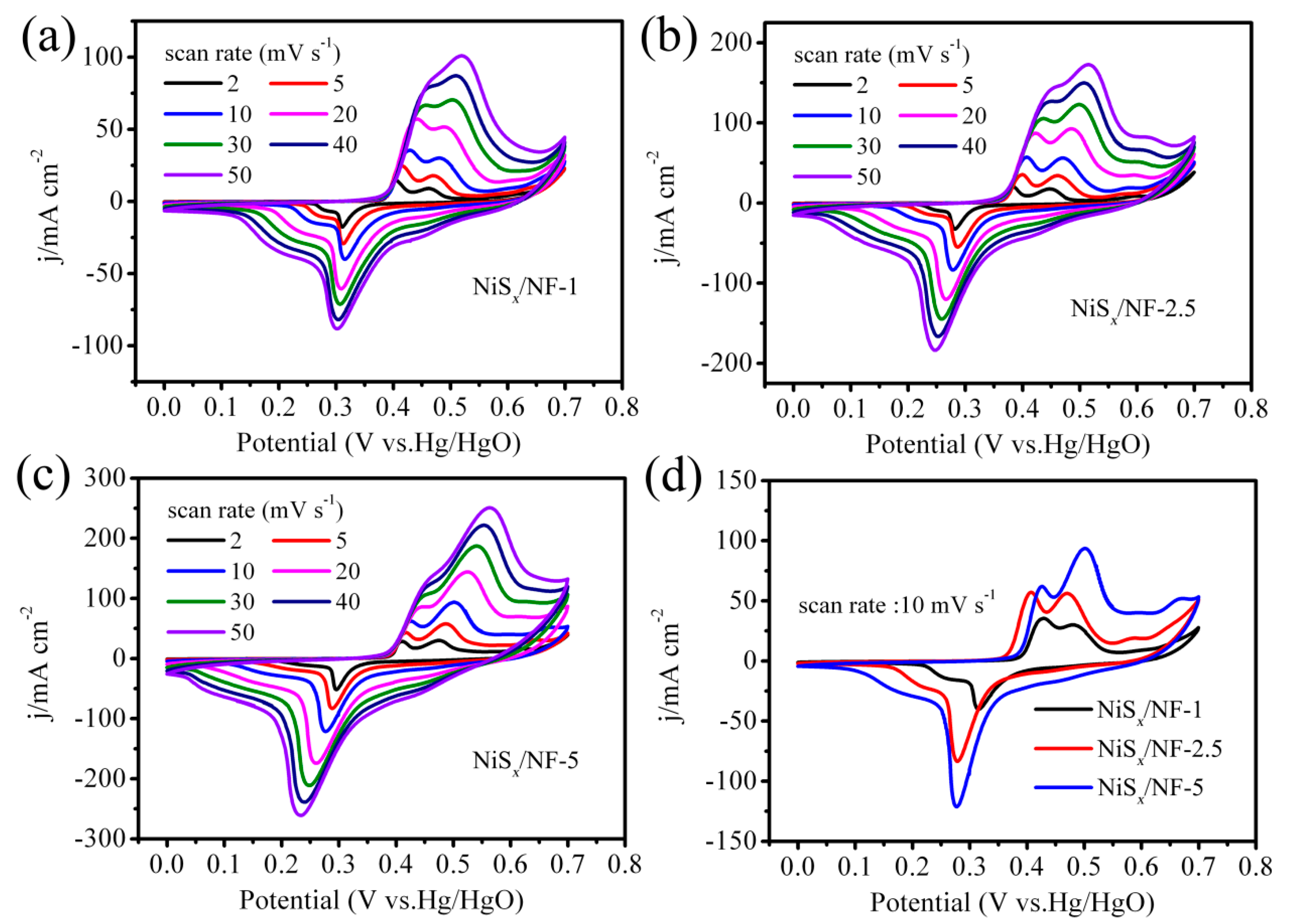
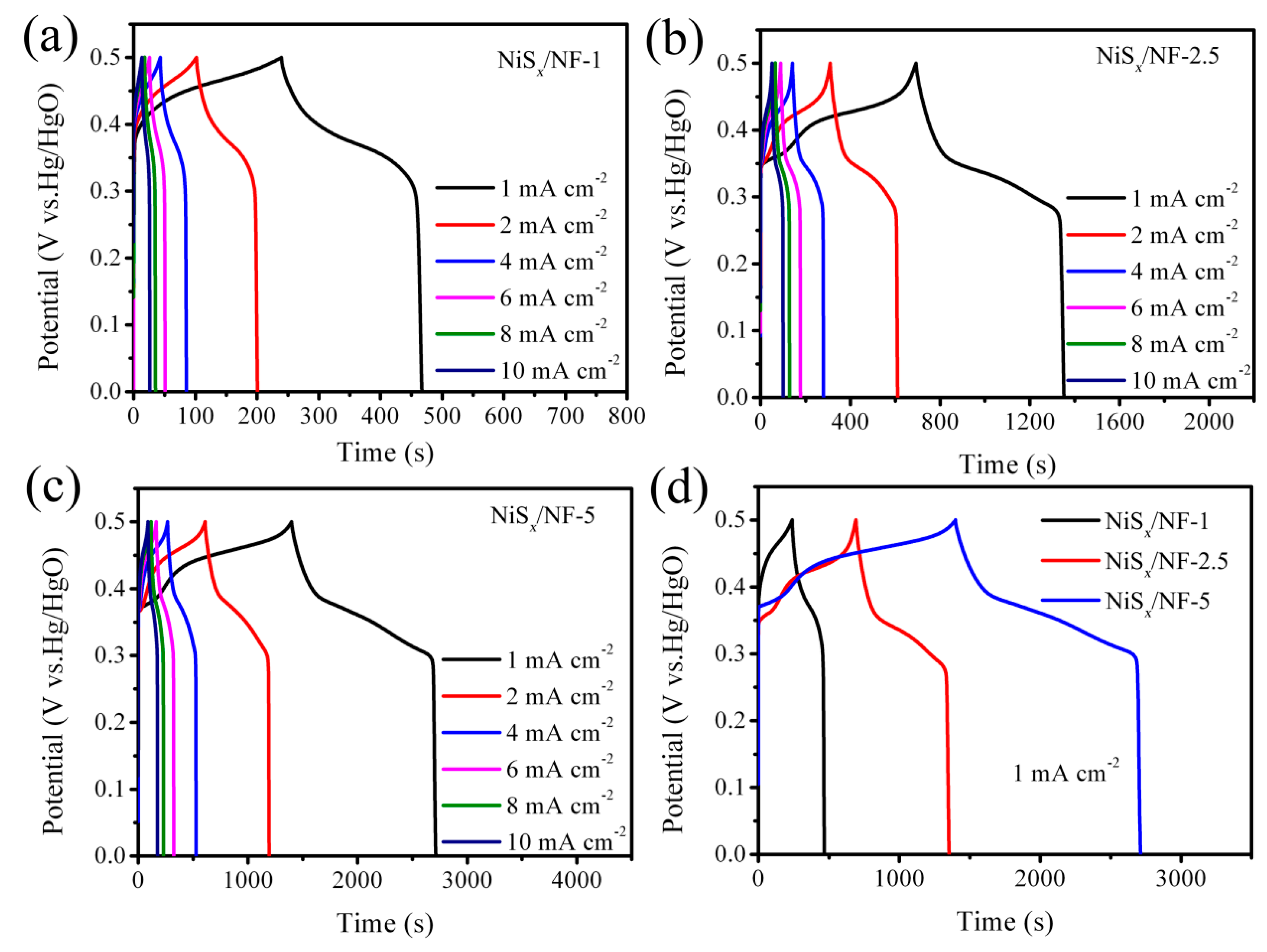
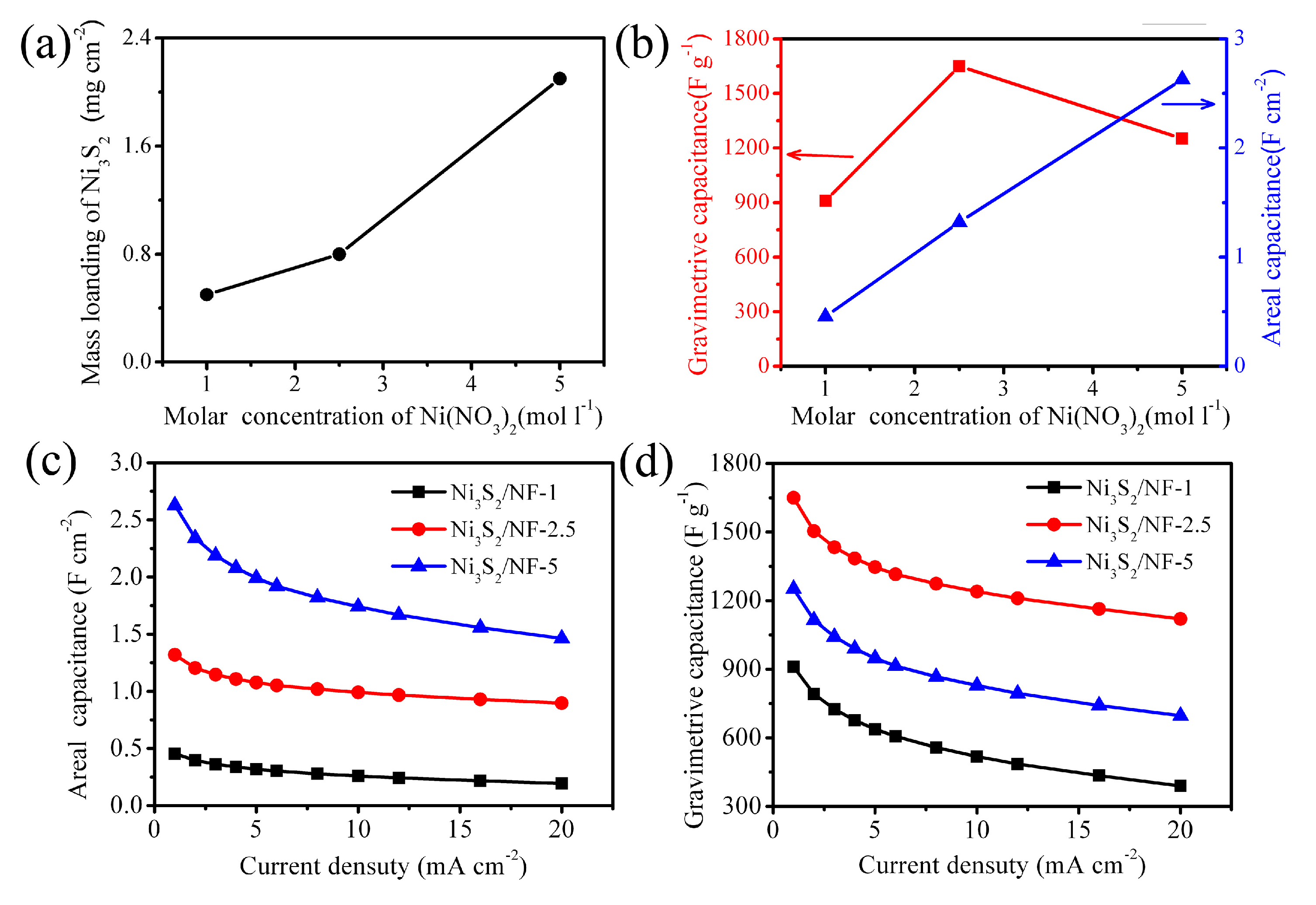
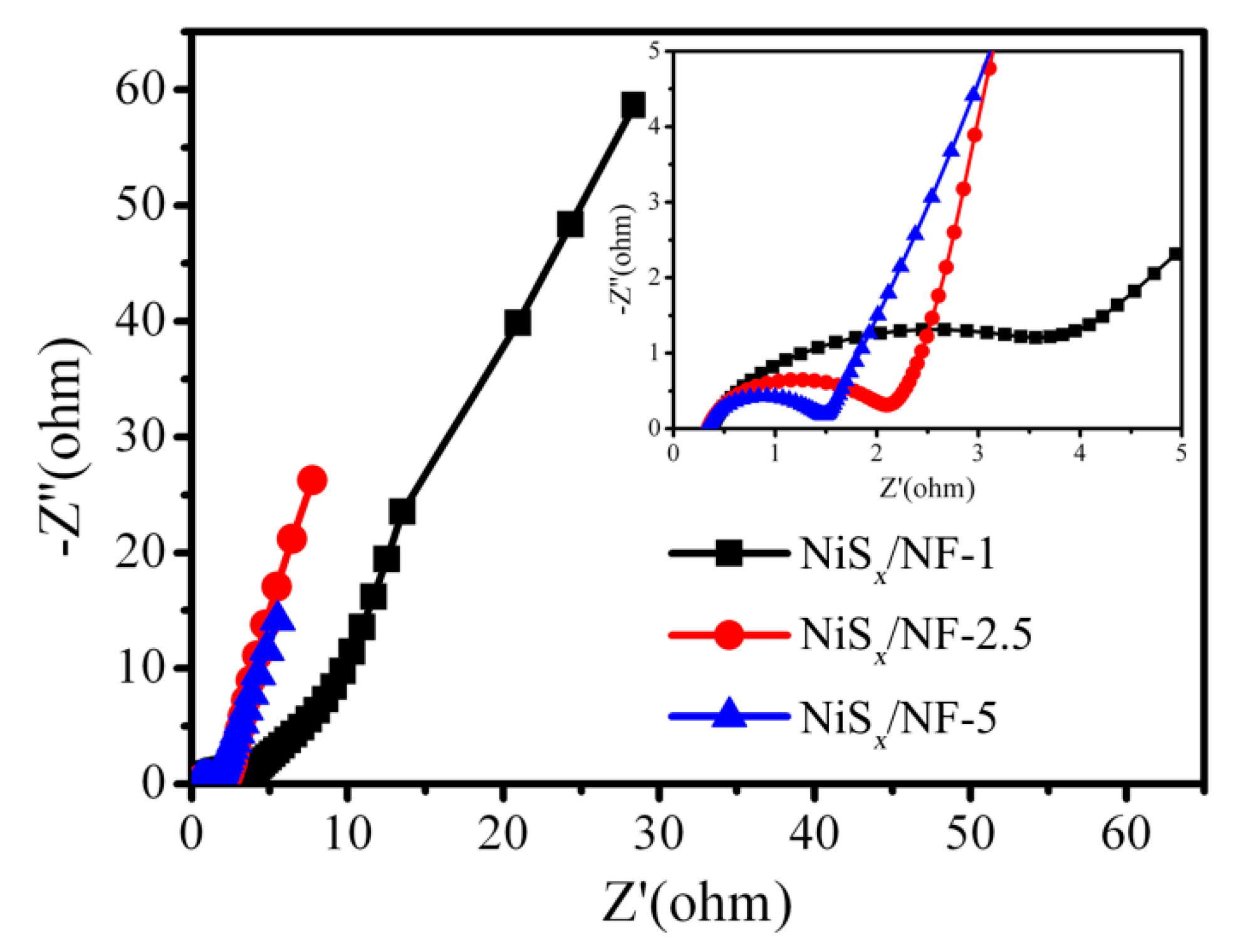
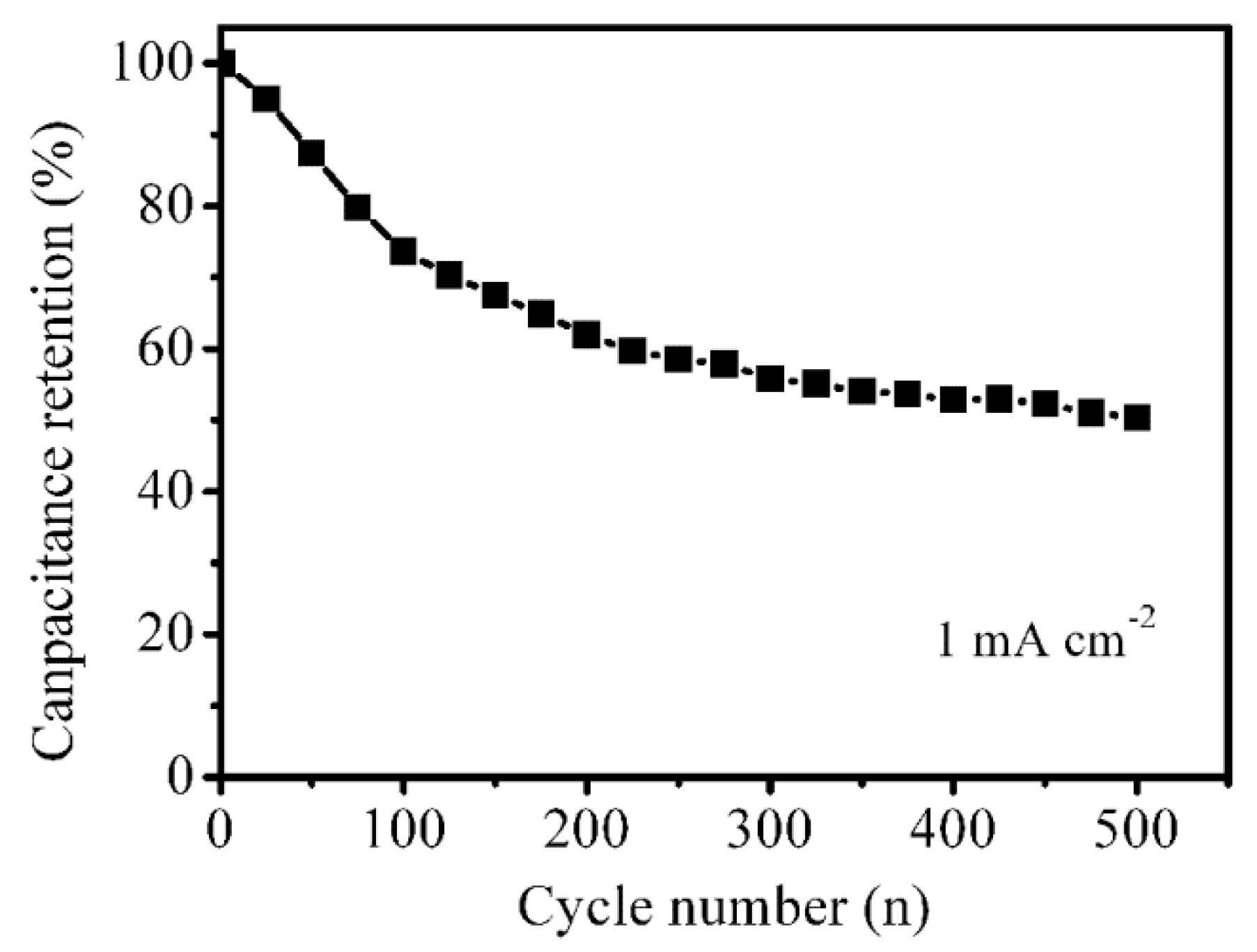
3.2. Electrochemical Performance
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Yang, Z.; Zhang, J.; Kintner-Meyer, M.C.; Lu, X.; Choi, D.; Lemmon, J.P.; Liu, J. Electrochemical energy storage for green grid. Chem. Rev. 2011, 111, 3577–3613. [Google Scholar] [CrossRef] [PubMed]
- Goodenough, J.B. Electrochemical energy storage in a sustainable modern society. Energy Environ. Sci. 2014, 7, 14–18. [Google Scholar] [CrossRef]
- Shao, Y.; El-Kady, M.F.; Sun, J.; Li, Y.; Zhang, Q.; Zhu, M.; Wang, H.; Dunn, B.; Kaner, R.B. Design and mechanisms of asymmetric supercapacitors. Chem. Rev. 2018, 118, 9233–9280. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Zhang, Q.; Zhu, J.; Duan, X.; Xu, Z.; Liu, Y.; Yang, H.; Lu, B. Nature of extra capacity in MoS2 electrodes: Molybdenum atoms accommodate with lithium. Energy Storage Mater. 2019, 16, 37–45. [Google Scholar] [CrossRef]
- Huang, H.; Lei, C.; Luo, G.; Li, G.; Liang, X.; Tang, S.; Du, Y. UV-assisted reduction of graphene oxide on Ni foam as high performance electrode for supercapacitors. Carbon 2016, 107, 917–924. [Google Scholar] [CrossRef]
- Feng, Y.; Chen, S.; Wang, J.; Lu, B. Carbon foam with microporous structure for high performance symmetric potassium dual-ion capacitor. J. Energy Chem. 2020, 43, 129–138. [Google Scholar] [CrossRef]
- Fan, L.; Ma, R.; Zhang, Q.; Jia, X.; Lu, B. Graphite Anode for Potassium Ion Battery with Unprecedented Performance. Angew. Chem. Int. Ed. 2019, 85, 10500–10505. [Google Scholar] [CrossRef]
- Wang, J.; Dong, S.; Ding, B.; Wang, Y.; Hao, X.; Dou, H.; Xia, Y.; Zhang, X. Pseudocapacitive materials for electrochemical capacitors: From rational synthesis to capacitance optimization. Nat. Sci. Rev. 2016, 4, 71–90. [Google Scholar] [CrossRef]
- Yu, Z.; Kang, Z.; Hu, Z.; Lu, J.; Zhou, Z.; Jiao, S. Hexagonal NiS nanobelts as advanced cathode materials for rechargeable Al-ion batteries. Chem. Commun. 2016, 52, 10427–10430. [Google Scholar] [CrossRef]
- Radhakrishnan, S.; Kim, H.Y.; Kim, B.S. Expeditious and eco-friendly fabrication of highly uniform microflower superstructures and their applications in highlydurable methanol oxidation and high-performance supercapacitors. J. Mater. Chem. A 2016, 4, 12253–12262. [Google Scholar] [CrossRef]
- Radhakrishnan, S.; Kim, H.Y. Facile fabrication of NiS and reduced graphene oxide hybrid film for nonenzymatic detection of glucose. RSC Adv. 2015, 5, 44346–44352. [Google Scholar] [CrossRef]
- Zhang, C.; Huang, Y.; Tang, S.; Deng, M.; Du, Y. High-energy all-solid-state symmetric supercapacitor based on Ni3S2 mesoporous nanosheet-decorated three-dimensional reduced graphene oxide. ACS Energy Lett. 2017, 2, 759–768. [Google Scholar] [CrossRef]
- Huang, H.; Zhang, H.; Fan, Y.; Deng, X.; Li, G.; Liang, X.; Zhou, W.; Guo, J.; Tang, S. Serrated-like NiCoO2 nanoarrays on Ni foam for high-performance supercapacitors. Appl. Surf. Sci. 2019, 481, 1220–1227. [Google Scholar] [CrossRef]
- Naoi, K.; Naoi, W.; Aoyagi, S.; Miyamoto, J.-I.; Kamino, T. New generation “nanohybrid supercapacitor”. Acc. Chem. Res. 2012, 46, 1075–1083. [Google Scholar] [CrossRef]
- Liu, T.; Jiang, C.; Cheng, B.; You, W.; Yu, J. Hierarchical NiS/N-doped carbon composite hollow spheres with excellent supercapacitor performance. J. Mater. Chem. A 2017, 5, 21257–21265. [Google Scholar] [CrossRef]
- Li, J.-J.; Hu, Y.-X.; Liu, M.-C.; Kong, L.-B.; Hu, Y.-M.; Han, W.; Luo, Y.-C.; Kang, L. Mechanical alloying synthesis of Ni3S2 nanoparticles as electrode material for pseudocapacitor with excellent performances. J. Alloys Compds. 2016, 656, 138–145. [Google Scholar] [CrossRef]
- Xu, S.; Wang, T.; Ma, Y.; Jiang, W.; Wang, S.; Hong, M.; Hu, N.; Su, Y.; Zhang, Y.; Yang, Z. Cobalt Doping to Boost the Electrochemical Properties of Ni@Ni3S2 Nanowire Films for High-Performance Supercapacitors. ChemSusChem 2017, 10, 4056–4065. [Google Scholar] [CrossRef]
- Yang, J.; Duan, X.; Guo, W.; Li, D.; Zhang, H.; Zheng, W. Electrochemical performances investigation of NiS/rGO composite as electrode material for supercapacitors. Nano Energy 2014, 5, 74–81. [Google Scholar] [CrossRef]
- Qu, C.; Zhang, L.; Meng, W.; Liang, Z.; Zhu, B.; Dang, D.; Dai, S.; Zhao, B.; Tabassum, H.; Gao, S.; et al. MOF-derived α-NiS nanorods on graphene as an electrode for high-energy-density supercapacitors. J. Mater. Chem. A 2018, 6, 4003–4012. [Google Scholar] [CrossRef]
- Krishnamoorthy, K.; Veerasubramani, G.K.; Radhakrishnan, S.; Kim, S.J. One pot hydrothermal growth of hierarchical nanostructured Ni3S2 on Ni foam for supercapacitor application. Chem. Engine J. 2014, 251, 116–122. [Google Scholar] [CrossRef]
- Yu, L.; Yang, B.; Liu, Q.; Liu, J.; Wang, X.; Song, D.; Wang, J.; Jing, X. Interconnected NiS nanosheets supported by nickel foam: Soaking fabrication and supercapacitors application. J. Electroanal. Chem. 2015, 739, 156–163. [Google Scholar] [CrossRef]
- Ghosh, D.; Das, C.K. Hydrothermal growth of hierarchical Ni3S2 and Co3S4 on a reduced graphene oxide hydrogel@ Ni foam: A high-energy-density aqueous asymmetric supercapacitor. ACS Appl. Mater. Interfaces 2015, 7, 1122–1131. [Google Scholar] [CrossRef] [PubMed]
- Deng, X.; Fan, Y.; Zhou, Q.; Huang, H.; Zhou, W.; Lan, Z.; Liang, X.; Li, G.; Guo, J.; Tang, S. Self-supported Ni3S2/NiCo2O4 core-shell flakes-arrays on Ni foam for enhanced charge storage properties. Electrochim. Acta 2019, 319, 783–790. [Google Scholar] [CrossRef]
- Zhang, G.; Lou, X.W. General solution growth of mesoporous NiCo2O4 nanosheets on various conductive substrates as high-performance electrodes for supercapacitors. Adv. Mater. 2013, 25, 976–979. [Google Scholar] [CrossRef]
- Chen, Y.; Qu, B.; Hu, L.; Xu, Z.; Li, Q.; Wang, T. High-performance supercapacitor and lithium-ion battery based on 3D hierarchical NH4F-induced nickel cobaltate nanosheet–nanowire cluster arrays as self-supported electrodes. Nanoscale 2013, 5, 9812–9820. [Google Scholar] [CrossRef]
- Chen, J.S.; Guan, C.; Gui, Y.; Blackwood, D.J. Rational Design of Self-Supported Ni3S2 Nanosheets Array for Advanced Asymmetric Supercapacitor with a Superior Energy Density. ACS Appl. Mater. Interfaces 2017, 9, 496–504. [Google Scholar] [CrossRef]
- Zhang, J.; Lin, J.; Wu, J.; Xu, R.; Lai, M.; Gong, C.; Chen, X.; Zhou, P. Excellent Electrochemical Performance Hierarchical Co3O4@Ni3S2 core/shell nanowire arrays for Asymmetric Supercapacitors. Electrochim. Acta 2016, 207, 87–96. [Google Scholar] [CrossRef]
- Li, R.; Wang, S.; Wang, J.; Huang, Z. Ni3S2@CoS core–shell nano-triangular pyramid arrays on Ni foam for high-performance supercapacitors. Phys. Chem. Chem. Phys. 2015, 17, 16434–16442. [Google Scholar] [CrossRef]
- Brousse, T.; Bélanger, D.; Long, J.W. To be or not to be pseudocapacitive? J. Electrochem. Soc. 2015, 162, A5185–A5189. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhao, C.; Min, S.; Qian, X. A facile one-step route to RGO/Ni3S2 for high-performance supercapacitors. Electrochim. Acta 2014, 144, 100–110. [Google Scholar] [CrossRef]
- Xing, Z.; Chu, Q.; Ren, X.; Ge, C.; Qusti, A.H.; Asiri, A.M.; Al-Youbi, A.O.; Sun, X. Ni3S2 coated ZnO array for high-performance supercapacitors. J. Power Sources 2014, 245, 463–467. [Google Scholar] [CrossRef]
- Li, W.; Wang, S.; Xin, L.; Wu, M.; Lou, X. Single-crystal β-NiS nanorod arrays with a hollow-structured Ni3S2 framework for supercapacitor applications. J. Mater. Chem. A 2016, 4, 7700–7709. [Google Scholar] [CrossRef]
- Li, G.; Cong, Y.; Zhang, C.; Tao, H.; Sun, Y.; Wang, Y. Hierarchical nanosheet-based Ni3S2 microspheres grown on Ni foam for high-performance all-solid-state asymmetric supercapacitors. Nanotechnology 2017, 28, 425401. [Google Scholar] [CrossRef] [PubMed]
- Yan, X.; Tong, X.; Ma, L.; Tian, Y.; Cai, Y.; Gong, C.; Zhang, M.; Liang, L. Synthesis of porous NiS nanoflake arrays by ion exchange reaction from NiO and their high performance supercapacitor properties. Mater. Lett. 2014, 124, 133–136. [Google Scholar] [CrossRef]
- Lv, J.; Liang, T.; Yang, M.; Suzuki, K.; Miura, H. The plume-like Ni3S2 supercapacitor electrodes formed on nickel foam by catalysis of thermal reduced graphene oxide. J. Electroanal. Chem. 2017, 786, 8–13. [Google Scholar]
- Wu, P.; Wang, D.; Ning, J.; Zhang, J.; Feng, X.; Dong, J.; Hao, Y. Novel 3D porous graphene/Ni3S2 nanostructures for high-performance supercapacitor electrodes. J. Alloys Compds. 2018, 731, 1063–1068. [Google Scholar] [CrossRef]
- Qi, J.; Chang, Y.; Sui, Y.; He, Y.; Meng, Q.; Wei, F.; Zhao, Y.; Jin, Y. Facile Construction of 3D Reduced Graphene Oxide Wrapped Ni3S2 Nanoparticles on Ni Foam for High-Performance Asymmetric Supercapacitor Electrodes. Part. Part. Syst. Char. 2017, 34, 1700196. [Google Scholar] [CrossRef]
- Lin, H.; Liu, F.; Wang, X.; Ai, Y.; Yao, Z.; Chu, L.; Han, S.; Zhuang, X. Graphene-Coupled Flower-Like Ni3S2 for a Free-Standing 3D Aerogel with an Ultra-High Electrochemical Capacity. Electrochim. Acta 2016, 191, 705–715. [Google Scholar] [CrossRef]


© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, H.; Deng, X.; Yan, L.; Wei, G.; Zhou, W.; Liang, X.; Guo, J. One-Step Synthesis of Self-Supported Ni3S2/NiS Composite Film on Ni Foam by Electrodeposition for High-Performance Supercapacitors. Nanomaterials 2019, 9, 1718. https://doi.org/10.3390/nano9121718
Huang H, Deng X, Yan L, Wei G, Zhou W, Liang X, Guo J. One-Step Synthesis of Self-Supported Ni3S2/NiS Composite Film on Ni Foam by Electrodeposition for High-Performance Supercapacitors. Nanomaterials. 2019; 9(12):1718. https://doi.org/10.3390/nano9121718
Chicago/Turabian StyleHuang, Haifu, Xiaoli Deng, Liqing Yan, Geng Wei, Wenzheng Zhou, Xianqing Liang, and Jin Guo. 2019. "One-Step Synthesis of Self-Supported Ni3S2/NiS Composite Film on Ni Foam by Electrodeposition for High-Performance Supercapacitors" Nanomaterials 9, no. 12: 1718. https://doi.org/10.3390/nano9121718
APA StyleHuang, H., Deng, X., Yan, L., Wei, G., Zhou, W., Liang, X., & Guo, J. (2019). One-Step Synthesis of Self-Supported Ni3S2/NiS Composite Film on Ni Foam by Electrodeposition for High-Performance Supercapacitors. Nanomaterials, 9(12), 1718. https://doi.org/10.3390/nano9121718




