Influence of Sintering Strategy on the Characteristics of Sol-Gel Ba1−xCexTi1−x/4O3 Ceramics
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Preparation
2.2. Sample Characterization
3. Results and Discussion
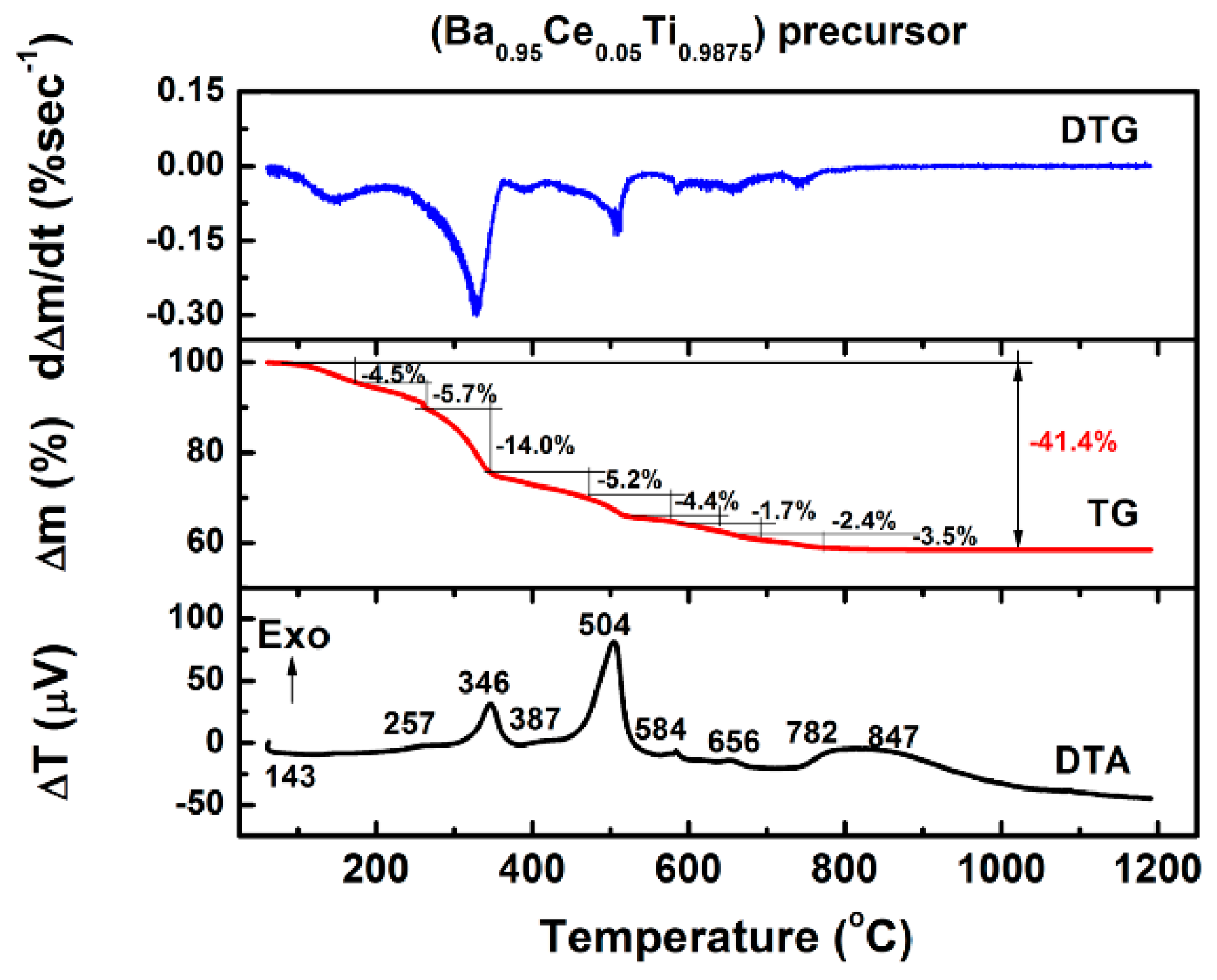
3.1. Thermal Behaviour of Precursors
3.2. Ce3+-Doped BaTiO3 Powders
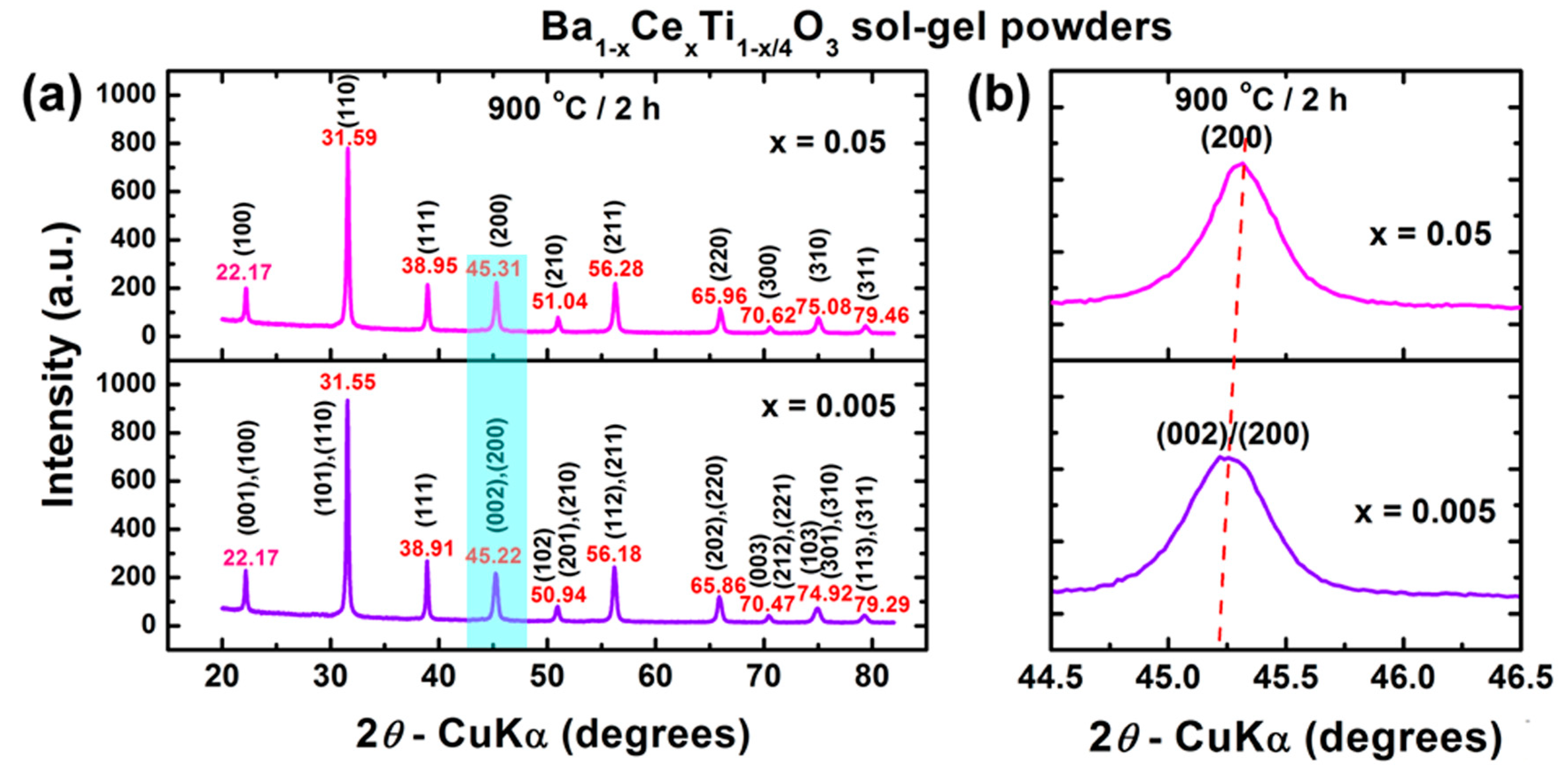
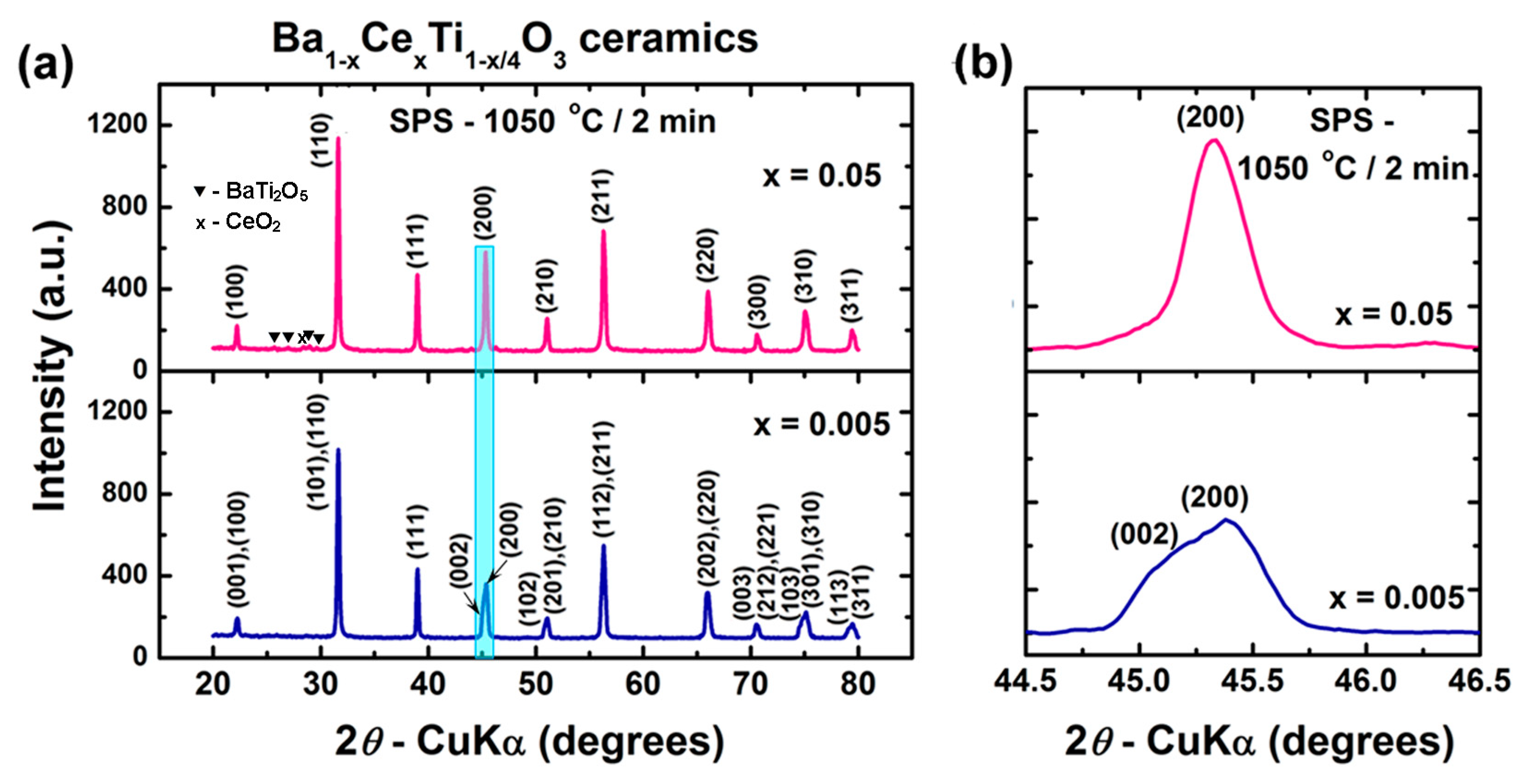
3.2.1. Phase Composition and Crystalline Structure
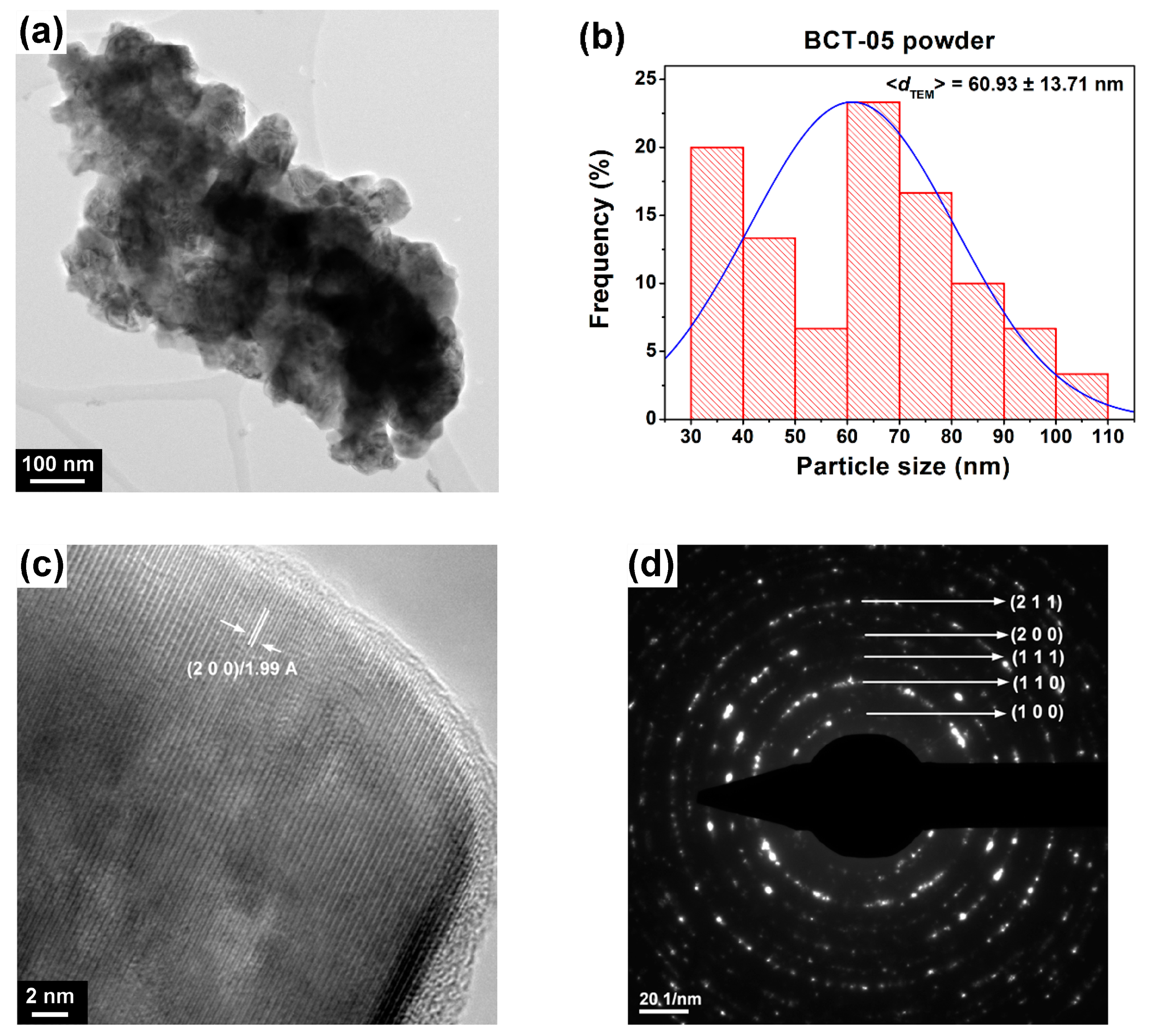
3.2.2. Morphology
3.3. Ce3+-Doped BaTiO3 Ceramics
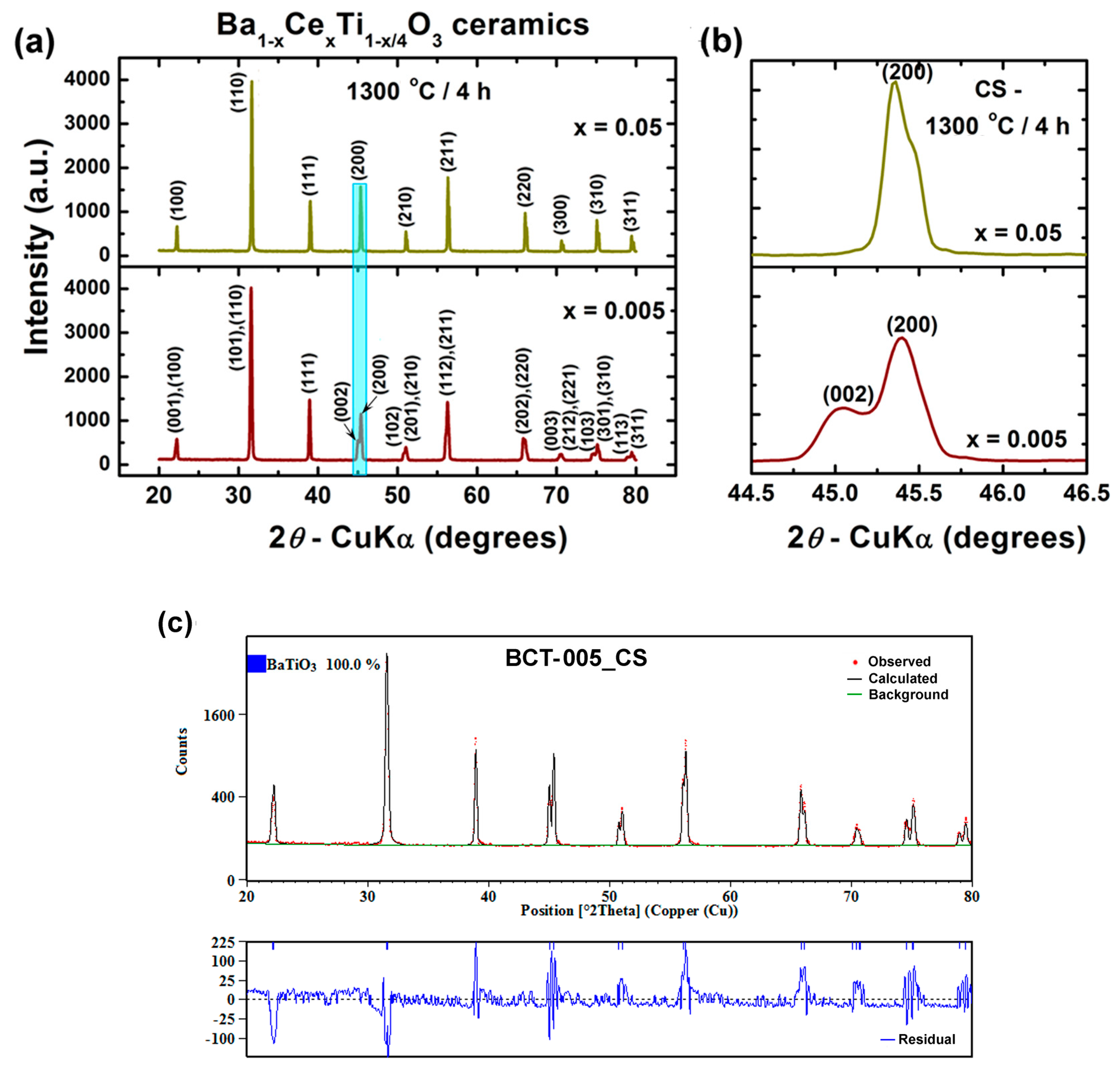
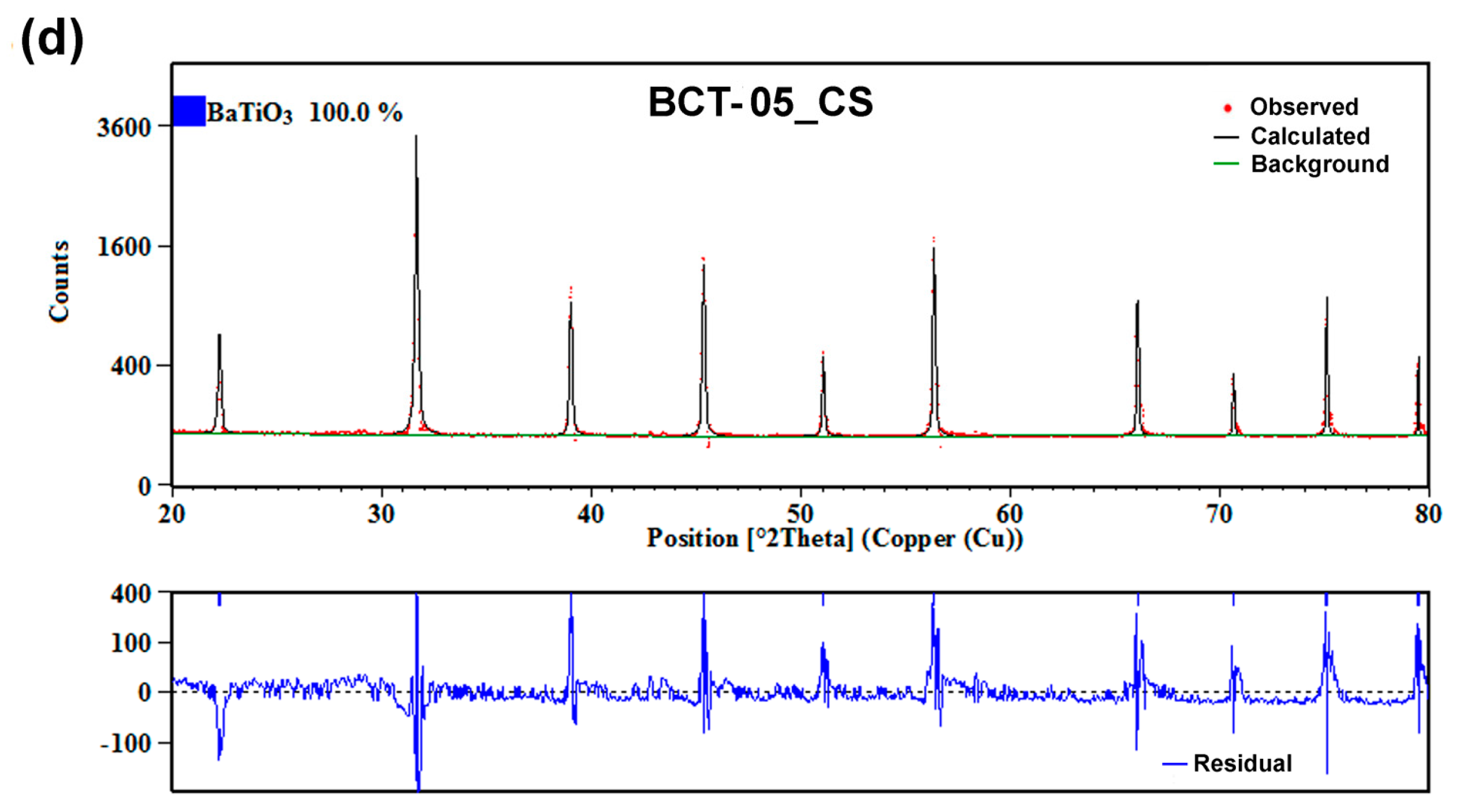
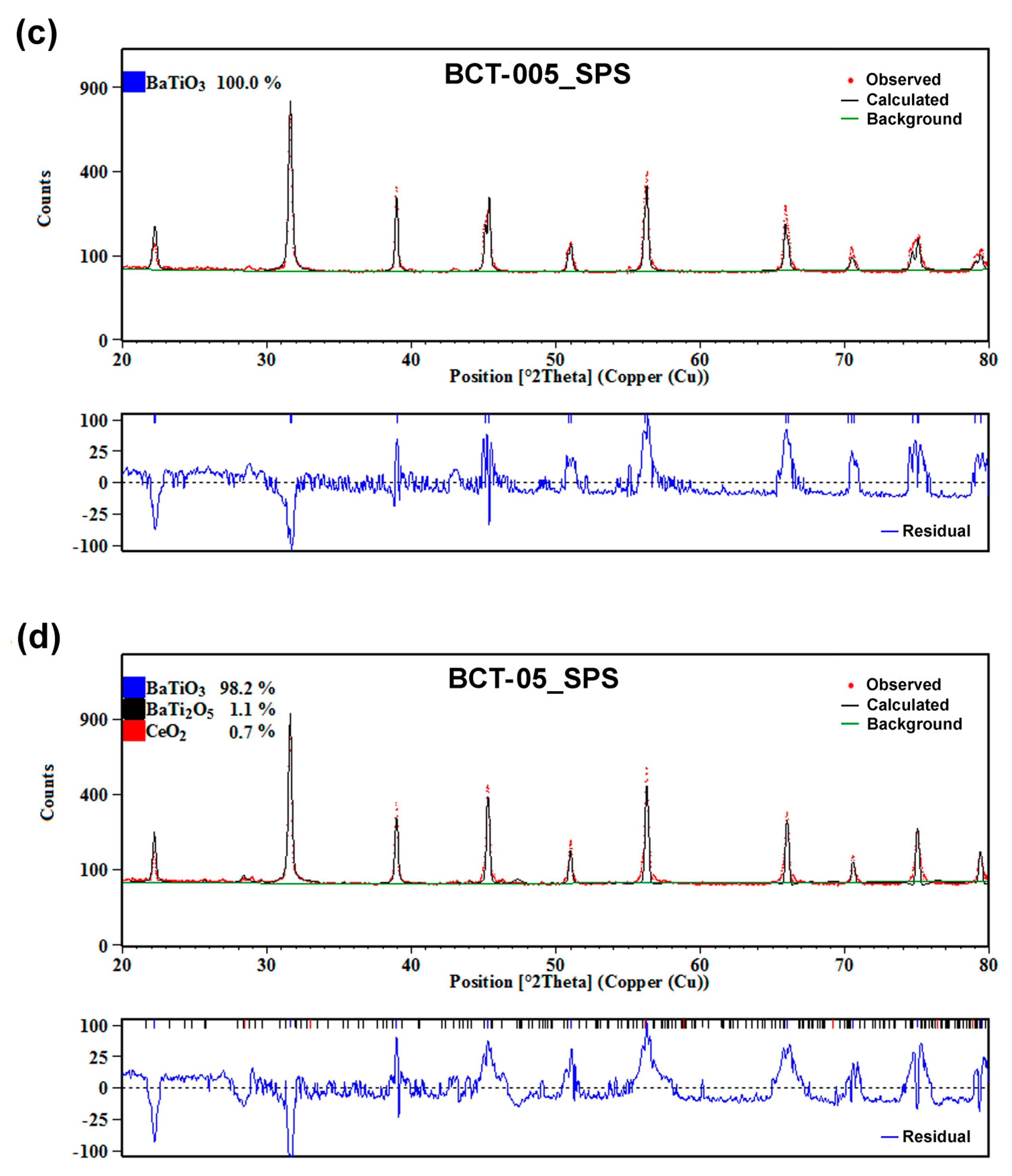
3.3.1. Phase Composition and Crystalline Structure
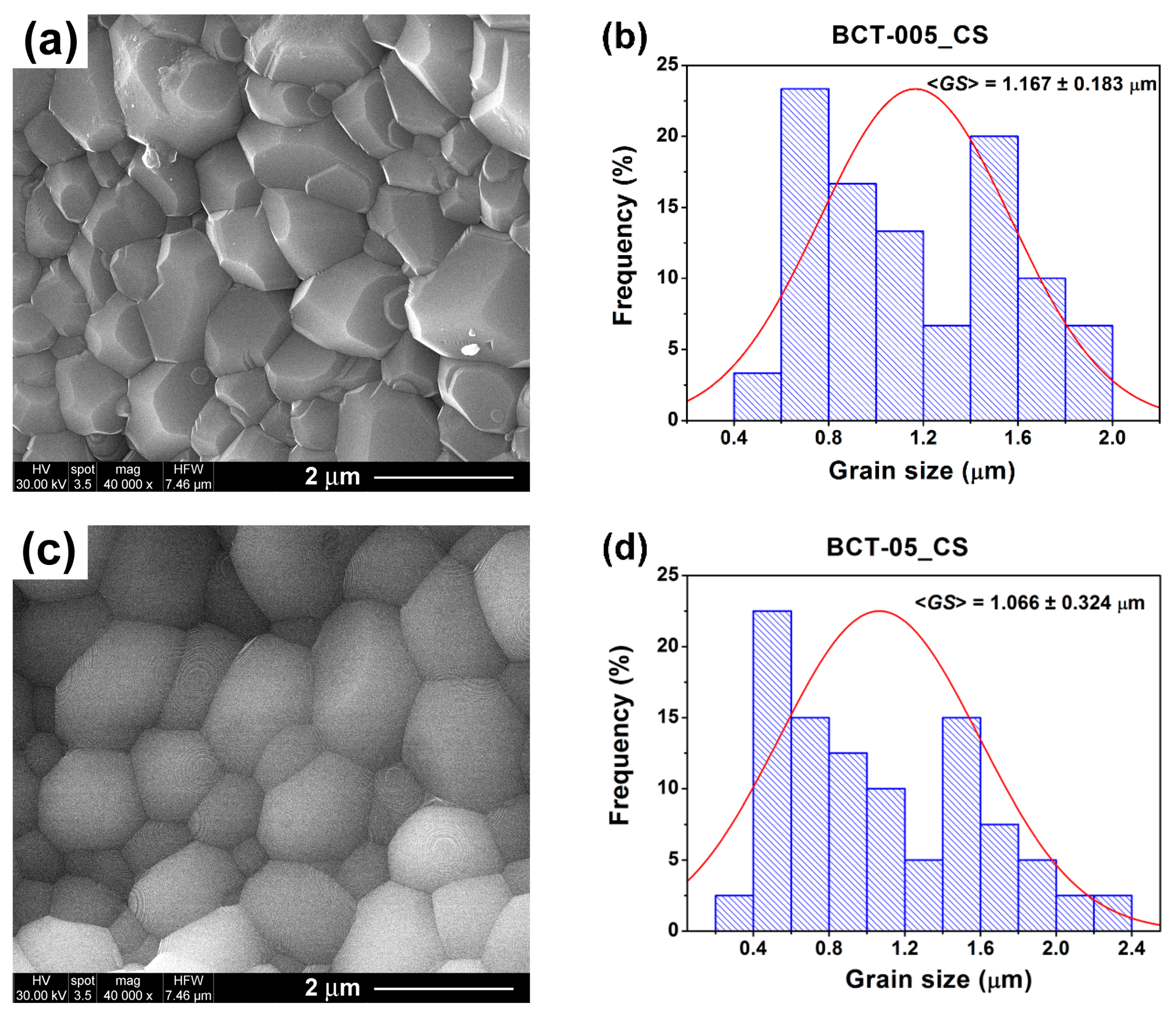
3.3.2. Microstructure
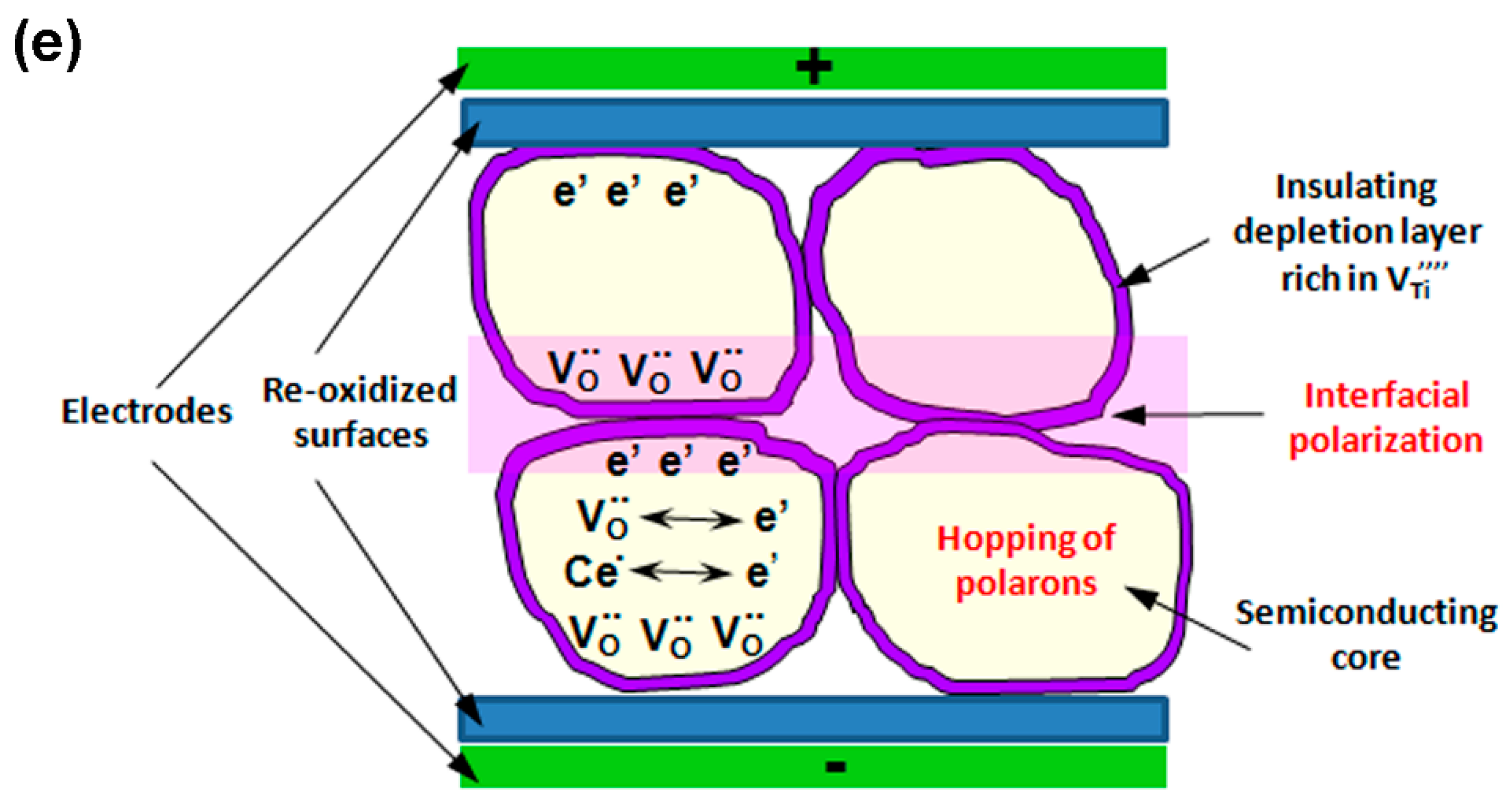
3.3.3. Dielectric and Ferroelectric Properties
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Acosta, M.; Novak, N.; Rojas, V.; Patel, S.; Vaish, R.; Koruza, J.; Rossetti, G.A.; Rödel, J. BaTiO3-based piezoelectrics: Fundamentals, current status, and perspectives. Appl. Phys. Rev. 2017, 4, 041305. [Google Scholar] [CrossRef]
- Lines, M.E.; Glass, A.M. Principles and Applications of Ferroelectrics and Related Materials; Clarendon, Oxford University Press: Oxford, UK, 1977. [Google Scholar]
- Haertling, G.H. Ferroelectric Ceramics: History and Technology. J. Am. Ceram. Soc. 1999, 82, 797–818. [Google Scholar] [CrossRef]
- Rae, A.; Chu, M.; Ganine, V. Dielectric Ceramic Materials. In Ceram Trans Barium Titanate: Past, Present and Future; Nair, K.M., Bhalla, A.S., Eds.; The American Ceramic Society: Westerville, OH, USA, 2007; Volume 100, pp. 1–12. [Google Scholar]
- Randall, C.A. Scientific and Engineering Issues of the State-of-the-Art and Future Multilayer Capacitors. J. Ceram. Soc. Jpn. 2001, 109, S2–S6. [Google Scholar] [CrossRef]
- Morrison FDSinclair, D.C.; West, A.R. Doping mechanisms and electrical properties of La-doped BaTiO3 ceramics. Int. J. Inorg. Mater. 2001, 3, 1205–1210. [Google Scholar] [CrossRef]
- Morrison, F.D.; Sinclair, D.C.; West, A.R. Electrical and structural characteristics of lanthanum-doped barium titanate ceramics. J. Appl. Phys. 1999, 86, 6355–6366. [Google Scholar] [CrossRef]
- Morrison, F.D.; Sinclair, D.C.; Skakle, J.M.S.; West, A.R. Novel Doping Mechanism for Very-High-Permittivity Barium Titanate Ceramics. J. Am. Ceram. Soc. 1998, 81, 1957–1960. [Google Scholar] [CrossRef]
- Ianculescu, A.; Mocanu, Z.V.; Curecheriu, L.P.; Mitoseriu, L.; Padurariu, L.; Trusca, R. Dielectric and tunability properties of La-doped BaTiO3 ceramics. J. Alloy. Compd. 2011, 509, 10040–10049. [Google Scholar] [CrossRef]
- Ianculescu, A.C.; Vasilescu, C.A.; Crisan, M.; Raileanu, M.; Vasile, B.S.; Calugaru, M.; Crisan, D.; Dragan, N.; Curecheriu, L.; Mitoseriu, L. Formation mechanism and characteristics of lanthanum-doped BaTiO3 powders and ceramics prepared by the sol-gel process. Mater. Charact. 2015, 106, 195–207. [Google Scholar] [CrossRef]
- Murugaraj, P.; Kutty, T.R.N.; Subba, R.M. Diffuse phase transformations in neodymium-doped BaTiO3 ceramics. J. Mater. Sci. 1986, 21, 3521–3527. [Google Scholar] [CrossRef]
- Yao, Z.; Liu, H.; Liu, Y.; Wu, Z.; Shen, Z.; Liu, Y.; Cao, M. Structure and dielectric behavior of Nd-doped BaTiO3 perovskites. Mater. Chem. Phys. 2008, 109, 475–481. [Google Scholar] [CrossRef]
- Tagantsev, A.K.; Sherman, V.O.; Astafiev, K.F.; Venkatesh, J.; Setter, N. Ferroelectric Materials for Microwave Tunable Applications. J. Electroceramics 2003, 11, 5–66. [Google Scholar] [CrossRef]
- Ianculescu, A.; Berger, D.; Viviani, M.; Ciomaga, C.; Mitoseriu, L.; Vasile, E.; Drăgan, N.; Crişan, D. Investigation of Ba1-xSrxTiO3 ceramics prepared from powders synthesized by the modified Pechini route. J. Eur. Ceram. Soc. 2007, 27, 3655–3658. [Google Scholar] [CrossRef]
- Ianculescu, A.; Berger, D.; Mitoşeriu, L.; Curecheriu, L.P.; Drăgan, N.; Crişan, D.; Vasile, E. Properties of Ba1-xSrxTiO3 ceramics prepared by the modified Pechini method. Ferroelectrics 2008, 369, 22–34. [Google Scholar] [CrossRef]
- Kadira, L.; Elmesbahi, A.; Sayouri, S. Dielectric study of calcium doped barium titanate Ba1−xCaxTiO3 ceramics. Int. J. Phys. Sci. 2016, 11, 71–79. [Google Scholar]
- Zhang, L.; Thakur, O.P.; Feteira, A.; Keith, G.M.; Mould, A.G.; Sinclair, D.C.; West, A.R. Comment on the use of calcium as a dopant in X8R BaTiO3-based ceramics. Appl. Phys. Lett. 2007, 90, 142914. [Google Scholar] [CrossRef]
- Chan, N.H.; Harmer, M.P.; Smyth, D.M. Compensating defects in highly donor-doped BaTiO3. J. Am. Ceram. Soc. 1986, 69, 507–510. [Google Scholar] [CrossRef]
- Chan, N.H.; Smyth, D.M. Defect chemistry of donor-doped BaTiO3. J. Am. Ceram. Soc. 1984, 67, 285–288. [Google Scholar] [CrossRef]
- Hennings, D.F.K.; Schreinemacher, B.; Schreinemacher, H. High-permittivity dielectric ceramics with high endurance. J. Eur. Ceram. Soc. 1994, 13, 81–88. [Google Scholar] [CrossRef]
- Hwang, J.H.; Han, Y.H. Dielectric properties of (Ba1−xCex)TiO3. Jpn. J. Appl. Phys. 2000, 39, 2701–2704. [Google Scholar] [CrossRef]
- Hwang, J.H.; Han, Y.H. Electrical properties of cerium-doped BaTiO3. J. Am. Ceram. Soc. 2001, 84, 1750–1754. [Google Scholar] [CrossRef]
- Guha, J.P.; Kolar, D. Subsolidus equilibria in the system BaO-CeO2-TiO2. J. Am. Ceram. Soc. 1973, 56, 5–6. [Google Scholar] [CrossRef]
- Jing, Z.; Yu, Z.; Ang, C. Crystalline structure and dielectric properties of Ba(Ti1−yCey)O3. J. Mater. Sci. 2003, 38, 1057–1061. [Google Scholar] [CrossRef]
- Ang, C.; Jing, Z.; Yu, Z. Ferroelectric relaxor Ba(Ti,Ce)O3. J. Phy. Condens. Matter. 2002, 14, 8901–8912. [Google Scholar] [CrossRef]
- Canu, G.; Confalonieri, G.; Deluca, M.; Curecheriu, L.; Buscaglia, M.T.; Asandulesa, M.; Horchidan, N.; Dapiaggi, M.; Mitoseriu, L.; Buscaglia, V. Structure-property correlations and origin of relaxor behaviour in BaCexTi1−xO3. Acta Mater. 2018, 152, 258–268. [Google Scholar] [CrossRef]
- Makovec, D.; Samardžija, Z.; Kolar, D. Incorporation of cerium into the BaTiO3 lattice. In Third Euro-Ceramics, Proceedings of the 3rd European Ceramic Society Conference, Madrid, Spain, 12–17 September 1993; Durán, P., Fernández, J.P., Eds.; Faenza Editrice Ibérica, S. L.: Faenza, Italy, 1993; Volume 1, pp. 961–966. [Google Scholar]
- Makovec, D.; Samardžija, Z.; Kolar, D. Solid solubility of cerium in BaTiO3. J. Solid State Chem. 1996, 123, 30–38. [Google Scholar] [CrossRef]
- Jonker, G.H.; Havinga, E.E. The influence of foreign ions on the crystal lattice of BaTiO3. Mater. Res. Bull. 1982, 17, 345–350. [Google Scholar] [CrossRef]
- Lewis, C.V.; Catlow, C.R.A. Defect studies of doped and undoped barium titanate using computer simulation techniques. J. Phys. Chem. Solids 1986, 47, 89–97. [Google Scholar] [CrossRef]
- Jing, Z.; Yu, Z.; Ang, C. Crystalline structure and dielectric behavior of (Ce,Ba)TiO3 ceramics. J. Mater. Res. 2002, 17, 2787–2793. [Google Scholar] [CrossRef]
- Yasmin, S.; Choudhury, S.; Hakim, M.A.; Bhuiyan, A.H.; Rahman, M.J. Effect of cerium doping on microstructure and dielectric properties of BaTiO3 ceramics. J. Mater. Sci. Technol. 2011, 27, 759–763. [Google Scholar]
- Cernea, M.; Monereau, O.; Llewellyn, P.; Tortet, L.; Galassi, C. Sol–gel synthesis and characterization of Ce doped-BaTiO3. J. Eur. Ceram. Soc. 2006, 26, 3241–3246. [Google Scholar] [CrossRef]
- Pechini, M.P. Method of Preparing Lead and Alkaline Earth Titanates and Niobates and Coating Method using the Same for a Capacitor. U.S. Patent No. 3,330,697, 11 July 1967. [Google Scholar]
- Ianculescu, A.; Berger, D.C.; Vasilescu, C.A.; Olariu, M.; Vasile, B.S.; Curecheriu, L.P.; Gajovic, A.; Trusca, R. Incorporation mechanism and functional properties of Ce-doped BaTiO3 ceramics derived from nanopowders prepared by the modified-Pechini method. In Nanoscale Ferroelectrics and Multiferroics: Key Processing and Characterization Issues, and Nanoscale Effects; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2016; Volume 1, pp. 13–43. [Google Scholar]
- Ianculescu, A.; Pintilie, I.; Vasilescu, C.A.; Botea, M.; Iuga, A.; Melinescu, A.; Drăgan, N.; Pintilie, L. Intrinsic pyroelectric properties of thick, coarse grained Ba1-xSrxTiO3 ceramics. Ceram. Int. 2016, 42, 10338–10348. [Google Scholar] [CrossRef]
- Vasilescu, C.A.; Trupina, L.; Vasile, B.S.; Trusca, R.; Cernea, M.; Ianculescu, A.C. Characteristics of 5 mol% Ce3+-doped barium titanate nanowires prepared by a combined route involving sol-gel chemistry and polycarbonate membrane-templated process. J. Nanopart. Res. 2015, 17, 434. [Google Scholar] [CrossRef]
- Ianculescu, A.C.; Vasilescu, C.A.; Trupină, L.; Vasile, B.S.; Truşcă, R.; Cernea, M.; Pintilie, L.; Nicoară, A. Characteristics of Ce3+-doped barium titanate nanoshell tubes prepared by template-mediated colloidal chemistry. J. Eur. Ceram. Soc. 2016, 36, 1633–1642. [Google Scholar] [CrossRef]
- Takeuchi, T.; Suyama, Y.; Sinclair, D.C.; Kageyama, H. Spark-plasma-sintering of fine BaTiO3 powder prepared by a sol-crystal method. J. Mater. Sci. 2001, 36, 2329–2334. [Google Scholar] [CrossRef]
- Li, B.; Wang, X.; Cai, M.; Hao, L.; Li, L. Densification of uniformly small-grained BaTiO3 using spark-plasma-sintering. Mater. Chem. Phys. 2003, 82, 173–180. [Google Scholar] [CrossRef]
- Licheri, R.; Fadda, S.; Orrù, R.; Cao, G.; Buscaglia, V. Self-propagating high-temperature synthesis of barium titanate and subsequent densification by spark plasma sintering (SPS). J. Eur. Ceram. Soc. 2007, 27, 2245–2253. [Google Scholar] [CrossRef]
- Luan, W.; Gao, L.; Kawaoka, H.; Sekino, T.; Niihara, K. Fabrication and characteristics of fine-grained BaTiO3 ceramics by spark plasma sintering. Ceram. Int. 2004, 30, 405–410. [Google Scholar] [CrossRef]
- Mitoşeriu, L.; Harnagea, C.; Nanni, P.; Testino, A.; Buscaglia, M.T.; Buscaglia, V.; Viviani, M.; Zhao, Z.; Nygren, M. Local switching properties of dense nanocrystalline BaTiO3 ceramics. Appl. Phys. Lett. 2004, 84, 2418–2420. [Google Scholar] [CrossRef]
- Buscaglia, M.T.; Buscaglia, V.; Viviani, M.; Petzelt, J.; Savinov, M.; Mitoşeriu, L.; Testino, A.; Nanni, P.; Harnagea, C.; Zhao, Z.; et al. Ferroelectric properties of dense nanocrystalline BaTiO3 ceramics. Nanotechnology 2004, 15, 1113–1117. [Google Scholar] [CrossRef]
- Hungría, T.; Algueró, M.; Hungría, A.B.; Castro, A. Dense fine-grained Ba1−xSrxTiO3 ceramic prepared by the combination of mechanosynthesized nanopowders and spark plasma sintering. Chem. Mater. 2005, 17, 6205–6212. [Google Scholar] [CrossRef]
- Aldica, G.; Cernea, M.; Ganea, P. Dielectric Ba(Ti1-xSnx)O3 (x = 0.13) ceramics, sintered by spark plasma and conventional methods. J. Mater. Sci. 2010, 45, 2606–2610. [Google Scholar] [CrossRef]
- Maiwa, H. Structure and properties of Ba(Zr0.2Ti0.8)O3 ceramics prepared by spark plasma sintering. J. Mater. Sci. 2008, 43, 6385–6390. [Google Scholar] [CrossRef]
- Maiwa, H. Dielectric and Electromechanical Properties of Ba(ZrxTi1-x)O3 (x = 0.1 and 0.2) Ceramics Prepared by Spark Plasma Sintering. Jpn. J. Appl. Phys. 2007, 46, 7013–7017. [Google Scholar] [CrossRef]
- Shannon, R.D. Revised effective ionic radii and systematic studies of interatomic distances in halides and chalcogenides. Acta Cryst. A 1976, 32, 751–767. [Google Scholar] [CrossRef]
- Makovec, D.; Kolar, K. Internal oxidation of Ce3+–BaTiO3 solid solutions. J. Am. Ceram. Soc. 1997, 80, 45–52. [Google Scholar] [CrossRef]
- Ramoška, T.; Banys, J.; Sobiestianskas, R.; Vijatović Petrović, M.; Bobić, J.; Stojanović, B. Dielectric investigations of La-doped barium titanate. Proc. Appl. Ceram. 2010, 4, 193–198. [Google Scholar] [CrossRef]
- Cross, L.E. Relaxor Ferroelectrics: An Overview. Ferroelectrics 1994, 151, 305–320. [Google Scholar] [CrossRef]
- Cross, L.E. Ferroic Materials and Composites: Past, Present, and Future. In Advanced Ceramics, 3rd ed.; Elsevier Applied Science: London, UK; New York, NY, USA, 1990; pp. 71–101. [Google Scholar]
- Samara, G.A. The relaxational properties of compositionally disordered ABO3 perovskites. J. Phys. Condens. Matter 2003, 15, R367–R411. [Google Scholar] [CrossRef]
- Ahn, C.W.; Wang, K.; Li, J.F.; Lee, J.-S.; Kim, I.W. A Brief Review on Relaxor Ferroelectrics and Selected Issues in Lead-Free Relaxors. J. Korean Phys. Soc. 2016, 68, 1481–1494. [Google Scholar] [CrossRef]
- Li, B.; Wang, X.; Li, L.; Zhou, H.; Liu, X.; Han, X.; Zhang, Y.; Qi, X.; Deng, X. Dielectric properties of fine-grained BaTiO3 prepared by spark-plasma-sintering. Mater. Chem. Phys. 2004, 83, 23–28. [Google Scholar] [CrossRef]
- Takeuchi, T.; Capiglia, C.; Balakrishnan, N.; Takeda, Y.; Kageyama, H. Preparation of fine grained BaTiO3 ceramics by spark plasma sintering. J. Mater. Res. 2002, 17, 575–581. [Google Scholar] [CrossRef]
- Deng, X.; Wang, X.; Wen, H.; Kang, A.; Gui, Z.; Li, L. Phase transitions in nanocrystalline barium titanate ceramics prepared by spark plasma sintering. J. Am. Ceram. Soc. 2006, 89, 1059–1064. [Google Scholar] [CrossRef]
- Zhao, Z.; Buscaglia, V.; Viviani, M.; Buscaglia, M.T.; Mitoseriu, L.; Testino, A.; Nygren, M.; Johnsson, M.; Nanni, P. Grain-size effects on the ferroelectric behavior of dense nanocrystalline BaTiO3 ceramics. Phys. Rev. B 2004, 70, 024107. [Google Scholar] [CrossRef]
- Buscaglia, M.T.; Viviani, M.; Buscaglia, V.; Mitoşeriu, L.; Testino, A.; Nanni, P.; Zhao, Z.; Nygren, M.; Harnagea, C.; Piazza, D.; et al. High dielectric constant and frozen macroscopic polarization in dense nanocrystalline BaTiO3 ceramics. Phys. Rev. B 2006, 73, 064114. [Google Scholar] [CrossRef]
- Curecheriu, L.; Balmus, S.B.; Nica, V.; Buscaglia, M.T.; Buscaglia, V.; Ianculescu, A.; Mitoseriu, L. Grain-size dependent properties of dense nanocrystalline barium titanate ceramics. J. Am. Ceram. Soc. 2012, 95, 3912–3921. [Google Scholar] [CrossRef]
- Yang, X.; Li, D.; Ren, Z.H.; Zeng, R.G.; Gong, S.Y.; Zhou, D.; Tian, H.; Li, J.X.; Xu, G.; Shen, Z.J.; et al. Colossal dielectric performance of pure barium titanate ceramics consolidated by spark plasma sintering. RSC Adv. 2016, 6, 75422. [Google Scholar] [CrossRef]
- Valdez-Nava, Z.; Guillemet-Fritsch, S.; Tenailleau, C.; Lebey, T.; Durand, B.; Chane-Ching, J.Y. Colossal dielectric permittivity of BaTiO3-based nanocrystalline ceramics sintered by spark plasma sintering. J. Electroceram. 2009, 22, 238–244. [Google Scholar] [CrossRef]
- Guillemet-Fritsch, S.; Valdez-Nava, Z.; Tenailleau, C.; Lebey, T.; Durand, B.; Chane-Ching, J.Y. Colossal Permittivity in Ultrafine Grain Size BaTiO3–x and Ba0.95La0.05TiO3–x Materials. Adv. Mater. 2008, 20, 551–555. [Google Scholar] [CrossRef]


| Formula | Ba0.995Ce0.005Ti0.99875O3 | Ba0.95Ce0.05Ti0.9875O3 | |
|---|---|---|---|
| Sample Symbol | BCT-005 | BCT-05 | |
| Phase composition | BCTss-100% | BCTss-100% | |
| BCTss structure | Tetragonal, P4mm | Cubic, Pm3m | |
| Unit cell parameters | a (Å) | 4.003105 ± 0.000113 | 4.001052 ± 0.001342 |
| b (Å) | 4.003105 ± 0.000113 | 4.001052 ± 0.001342 | |
| c (Å) | 4.016862 ± 0.000156 | 4.001052 ± 0.001342 | |
| Tetragonality, c/a | 1.0034 | 1.0000 | |
| Unit cell volume, V (Å3) | 64.36961 | 64.05051 | |
| Theoretical density, ρt (g/cm3) | 6.014 | 6.032 | |
| R profile, Rp | 5.04481 | 4.89943 | |
| Weighted R profile, Rwp | 6.56691 | 6.38165 | |
| Goodness of fit, χ2 | 0.01703 | 0.01539 | |
| Crystallite size, <D> (nm) | 33.66 ± 10.03 | 25.98 ± 5.35 | |
| Internal strains, <S> (%) | 0.27 ± 0.05 | 0.35 ± 0.07 | |
| Particle size, <dTEM> (nm) | 109.25 ± 30.68 | 60.93 ± 13.71 | |
| Formula | Ba0.995Ce0.005Ti0.99875O3 | Ba0.95Ce0.05Ti0.9875O3 | |||
|---|---|---|---|---|---|
| Sample Symbol | BCT-005_CS | BCT-005_SPS | BCT-05_CS | BCT-05_SPS | |
| Sintering procedure/conditions | CS 1300 °C/4 h | SPS 1050 °C/2 min | CS 1300 °C/4 h | SPS 1050 °C/2 min | |
| Phase composition | BCTss-100% | BCTss-100% | BCTss-100% | BCTss-98.2% BT2-1.1% C-0.7% | |
| BCTss structure | Tetragonal, P4mm | Tetragonal, P4mm | Cubic, Pm3m | Cubic, Pm3m | |
| Unit cell parameters | a (Å) | 3.994169 ± 0.000108 | 4.000073 ± 0.000273 | 3.999053 ± 0.000054 | 3.999968 ± 0.000167 |
| b (Å) | 3.994169 ± 0.000108 | 4.000073 ± 0.000273 | 3.999053 ± 0.000054 | 3.999968 ± 0.000167 | |
| c (Å) | 4.024993 ± 0.000127 | 4.021890 ± 0.000344 | 3.999053 ± 0.000054 | 3.999968 ± 0.000167 | |
| Tetragonality, c/a | 1.0077 | 1.0054 | 1.0000 | 1.0000 | |
| Unit cell volume, V (Å3) | 64.21228 | 64.35261 | 63.95455 | 63.99847 | |
| Theoretical density, t (g/cm3) | 6.027 | 6.014 | 6.041 | 6.037 | |
| R profile, Rp | 6.48709 | 6.75345 | 7.53511 | 7.3148 | |
| Weighted R profile, Rwp | 9.1197 | 10.6243 | 10.8429 | 10.79943 | |
| Goodness of fit, χ2 | 0.47706 | 0.45168 | 0.62327 | 0.48938 | |
| Crystallite size, <D> (nm) | 48.72 ± 4.31 | 45.85 ± 6.80 | 171.43 ± 7.49 | 45.05 ± 6.37 | |
| Internal strains, <S> (%) | 0.19 ± 0.07 | 0.21 ± 0.12 | 0.18 ± 0.08 | 0.22 ± 0.11 | |
| Relative density, ρr (%) | 91.7 | 95.8 | 97.1 | 98.6 | |
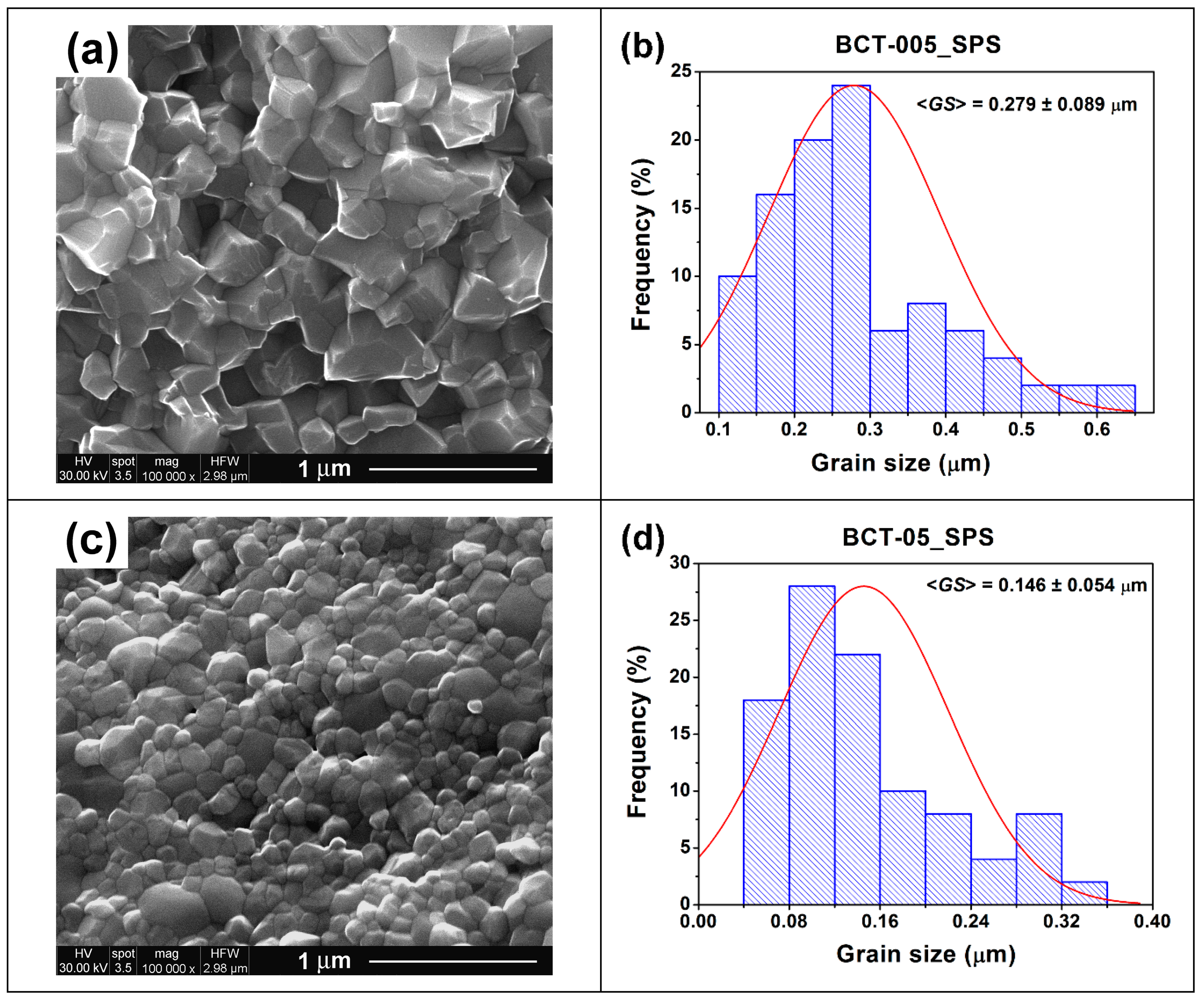
| Grain size, <GS > (µm) | 1.167 ± 0.183 | 0.279 ± 0.089 | 1.066 ± 0.324 | 0.146 ± 0.054 | |
| Formula | Ba0.995Ce0.005Ti0.99875O3 | Ba0.95Ce0.05Ti0.9875O3 | ||
|---|---|---|---|---|
| Sample Symbol | BCT-005_CS | BCT-005_SPS | BCT-05_CS | BCT-05_SPS |
| Sintering procedure/conditions | CS 1300 °C/4 h | SPS 1050 °C/2 min | CS 1300 °C/4 h | SPS 1050 °C/2 min |
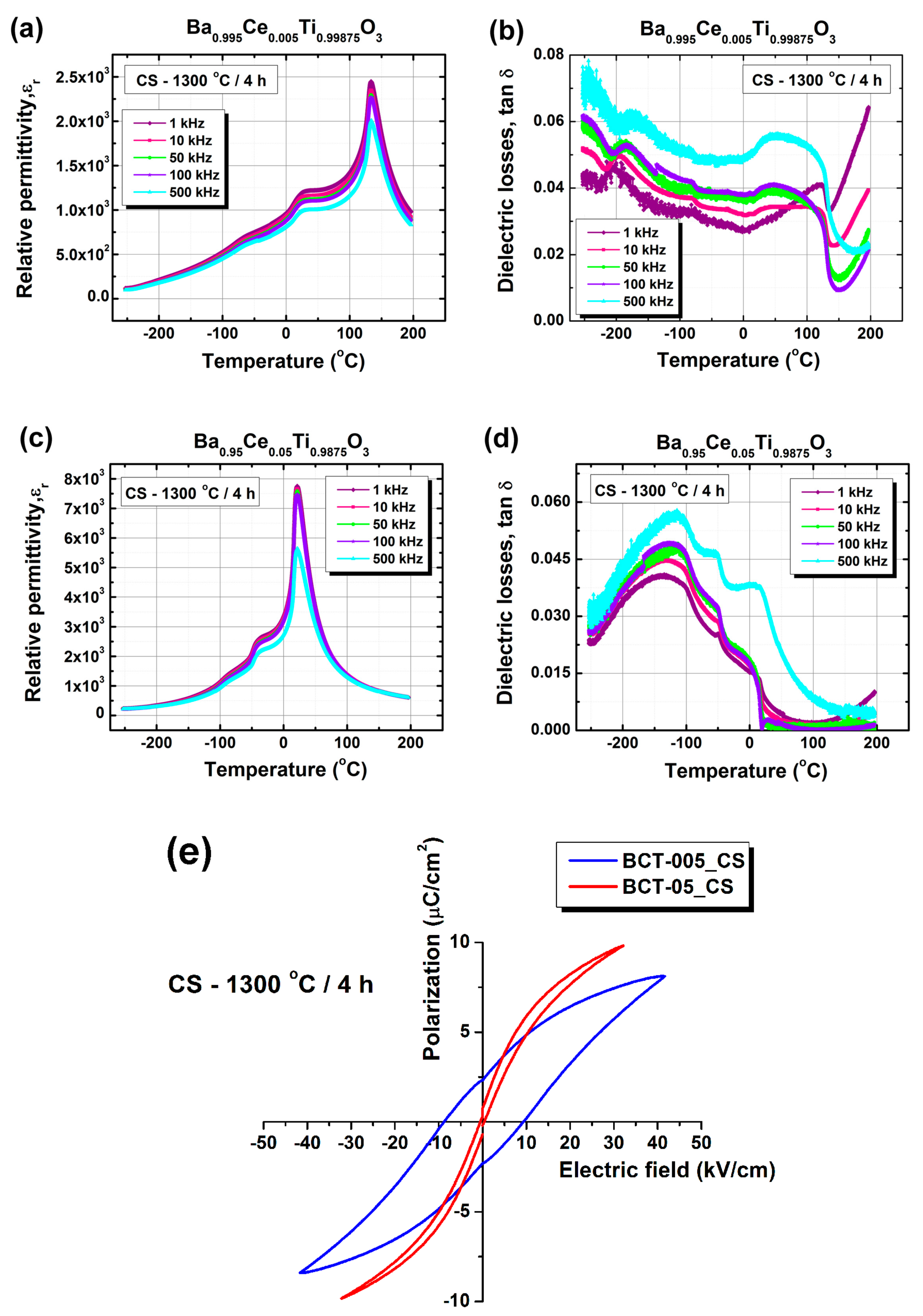
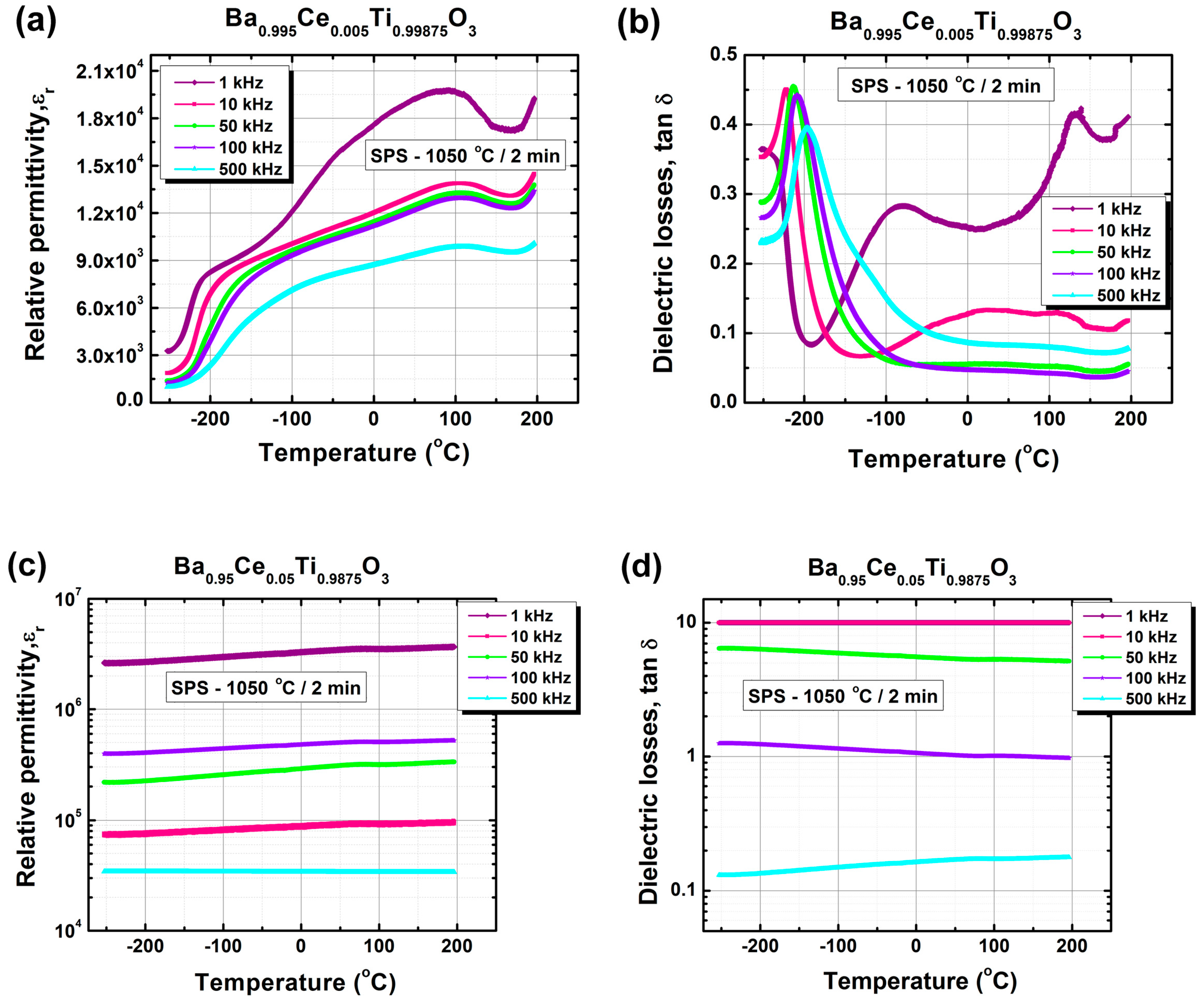
| ε′max | 2448 | 19782 | 7758 | 3.67 × 106 |
| TC | 133 | 91 | 21 | − |
| T1 | 25 | −49 | −44 | − |
| T2 | −69 | −214 | −93 | − |
| ε′RT | 1163 | 18367 | 7695 | 3.35 × 106 |
| tan δRT | 0.0287 | 0.2498 | 0.0098 | ~10 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stanciu, C.A.; Pintilie, I.; Surdu, A.; Truşcă, R.; Vasile, B.S.; Eftimie, M.; Ianculescu, A.C. Influence of Sintering Strategy on the Characteristics of Sol-Gel Ba1−xCexTi1−x/4O3 Ceramics. Nanomaterials 2019, 9, 1675. https://doi.org/10.3390/nano9121675
Stanciu CA, Pintilie I, Surdu A, Truşcă R, Vasile BS, Eftimie M, Ianculescu AC. Influence of Sintering Strategy on the Characteristics of Sol-Gel Ba1−xCexTi1−x/4O3 Ceramics. Nanomaterials. 2019; 9(12):1675. https://doi.org/10.3390/nano9121675
Chicago/Turabian StyleStanciu, Cătălina A., Ioana Pintilie, Adrian Surdu, Roxana Truşcă, Bogdan S. Vasile, Mihai Eftimie, and Adelina C. Ianculescu. 2019. "Influence of Sintering Strategy on the Characteristics of Sol-Gel Ba1−xCexTi1−x/4O3 Ceramics" Nanomaterials 9, no. 12: 1675. https://doi.org/10.3390/nano9121675
APA StyleStanciu, C. A., Pintilie, I., Surdu, A., Truşcă, R., Vasile, B. S., Eftimie, M., & Ianculescu, A. C. (2019). Influence of Sintering Strategy on the Characteristics of Sol-Gel Ba1−xCexTi1−x/4O3 Ceramics. Nanomaterials, 9(12), 1675. https://doi.org/10.3390/nano9121675








