Single-Step Synthesis of Vertically Aligned Carbon Nanotube Forest on Aluminium Foils
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
3.1. Synthesis and Characteristics of Vertically Aligned Carbon Nanotube (VACNT) on Aluminium Foils
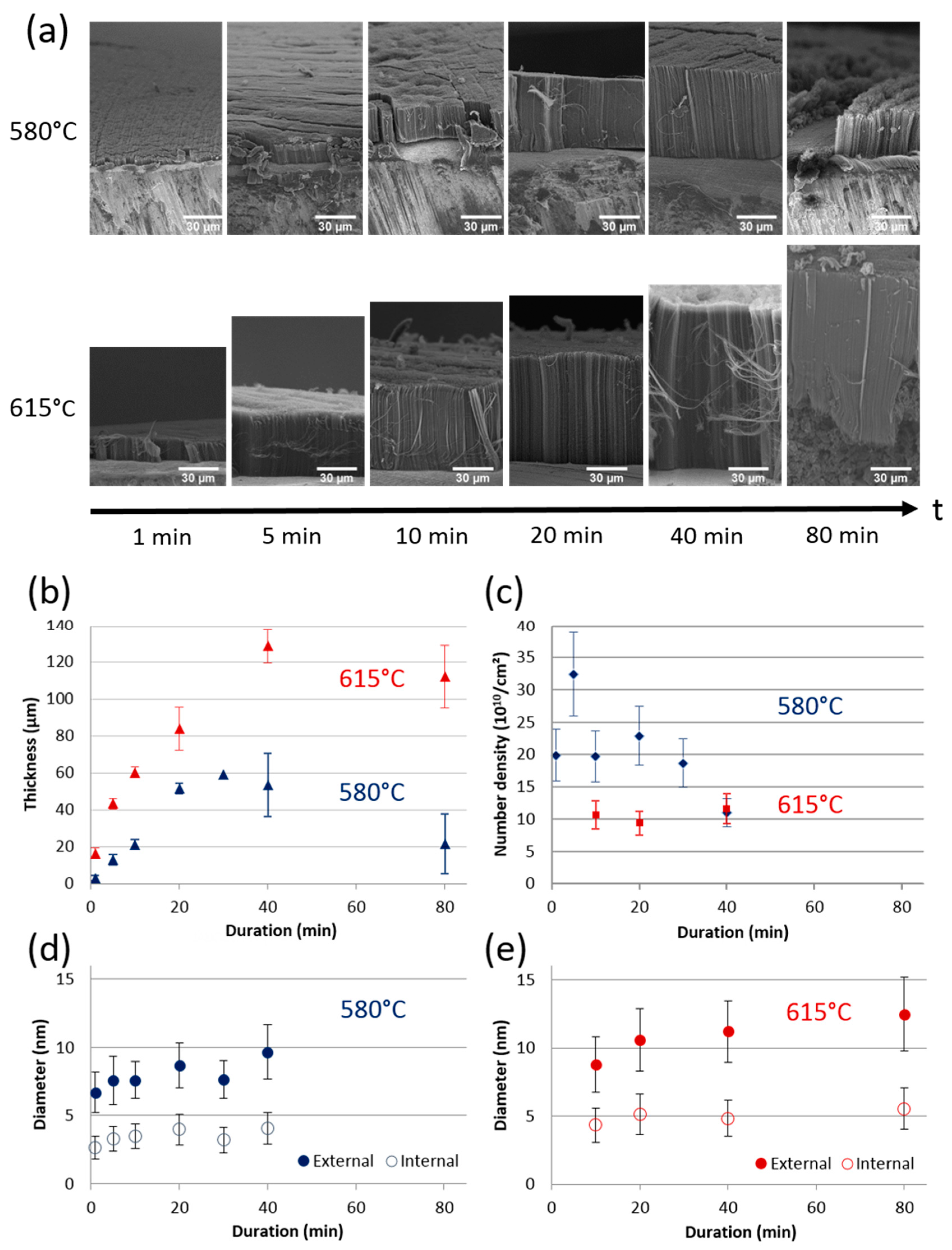
3.1.1. Low-Temperature Conditions to Form Suitable VACNT Carpets
3.1.2. Morphological and Structural Characterization
3.1.3. Electrochemical Characterization
3.1.4. Effect of Synthesis Duration on VACNT Thickness, CNT Diameter and Density
3.2. Understanding the Growth Rate Changes Occurring at Low Temperature
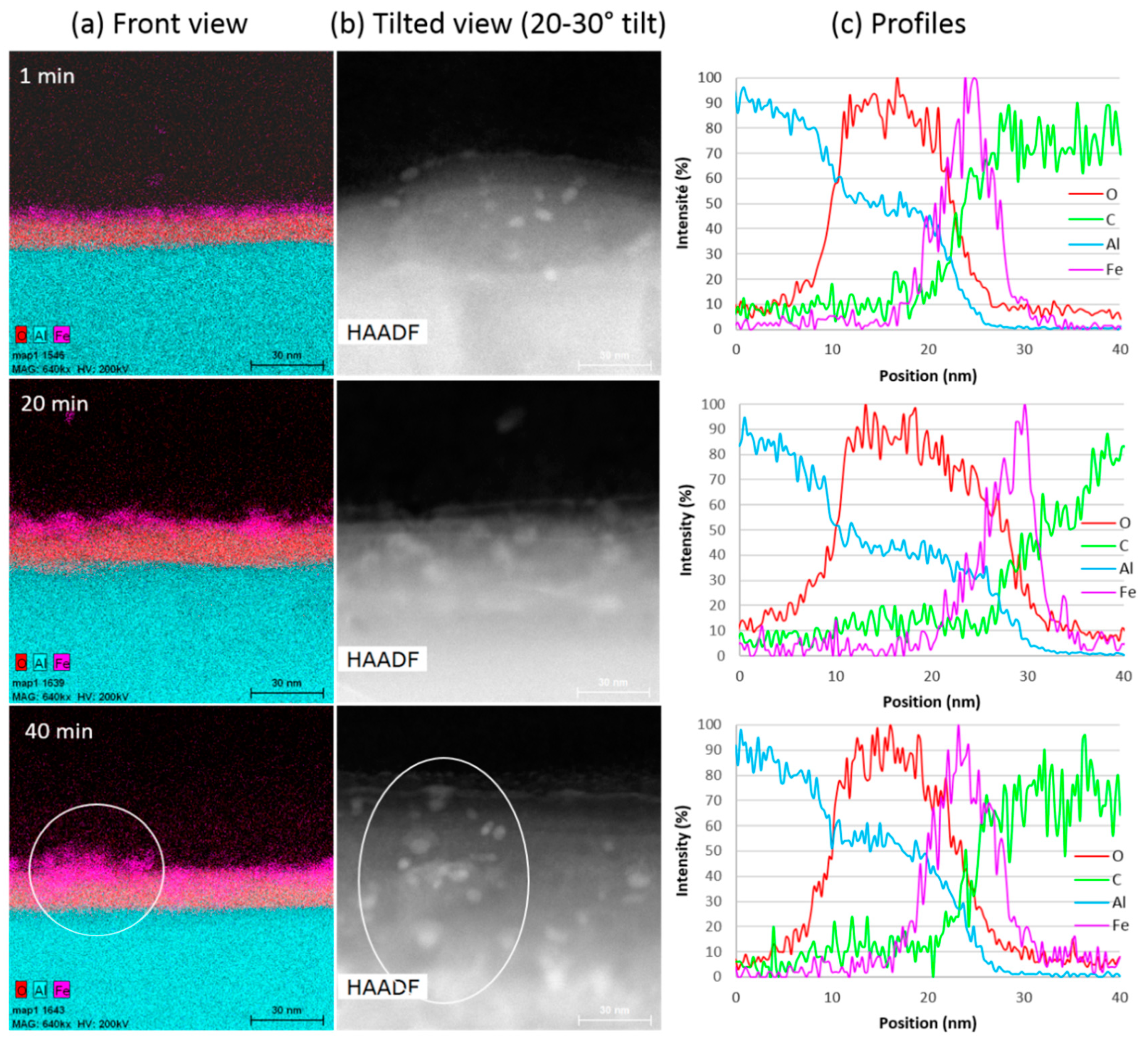
3.2.1. Analysis of the Aluminium (Al)/VACNT Interface by Scanning Transmission Electron Microscopy coupled with Energy Dispersive X-Ray Spectroscopy (STEM/EDX)
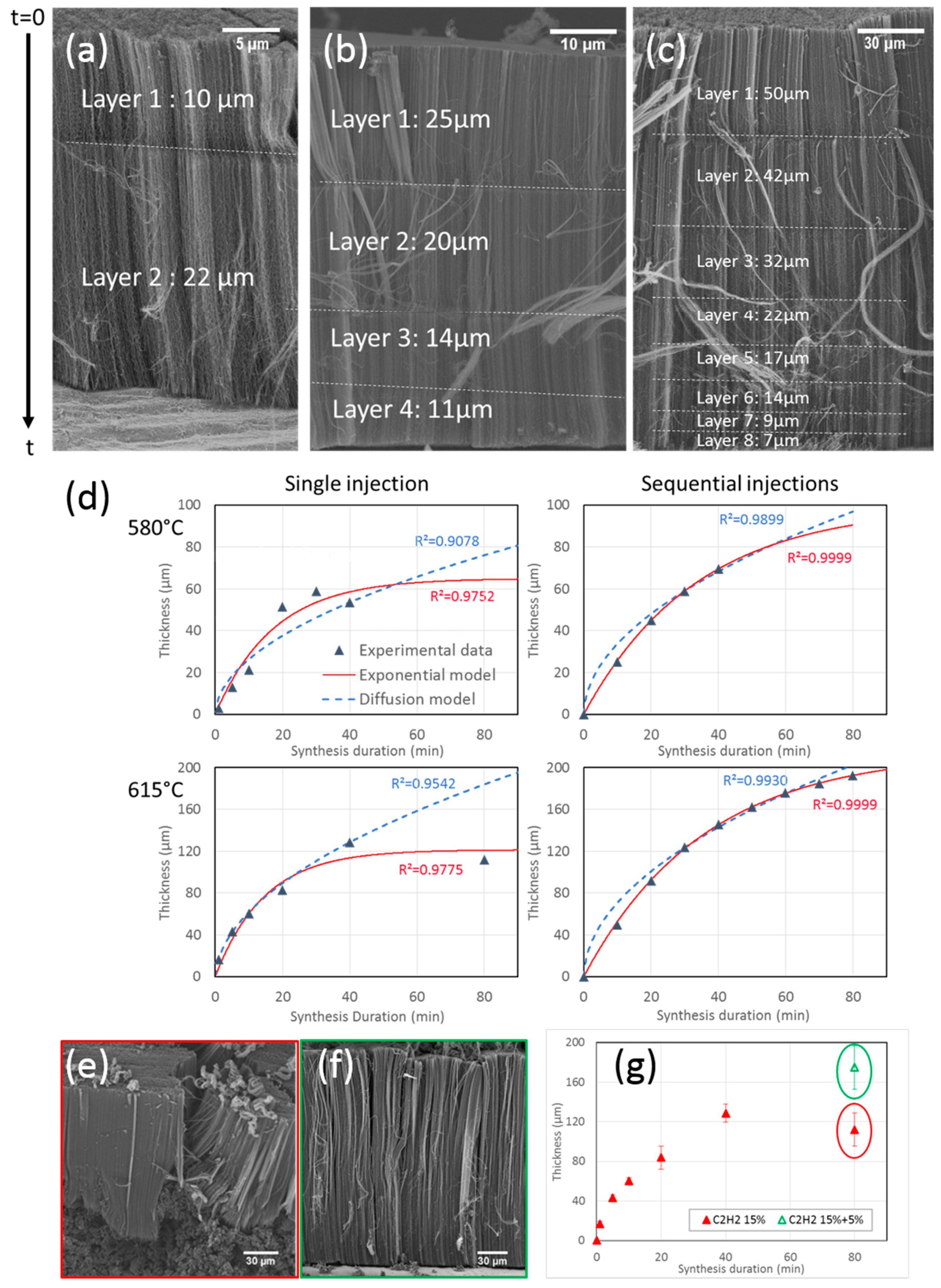
3.2.2. Sequential Synthesis
3.2.3. Growth Rate Model
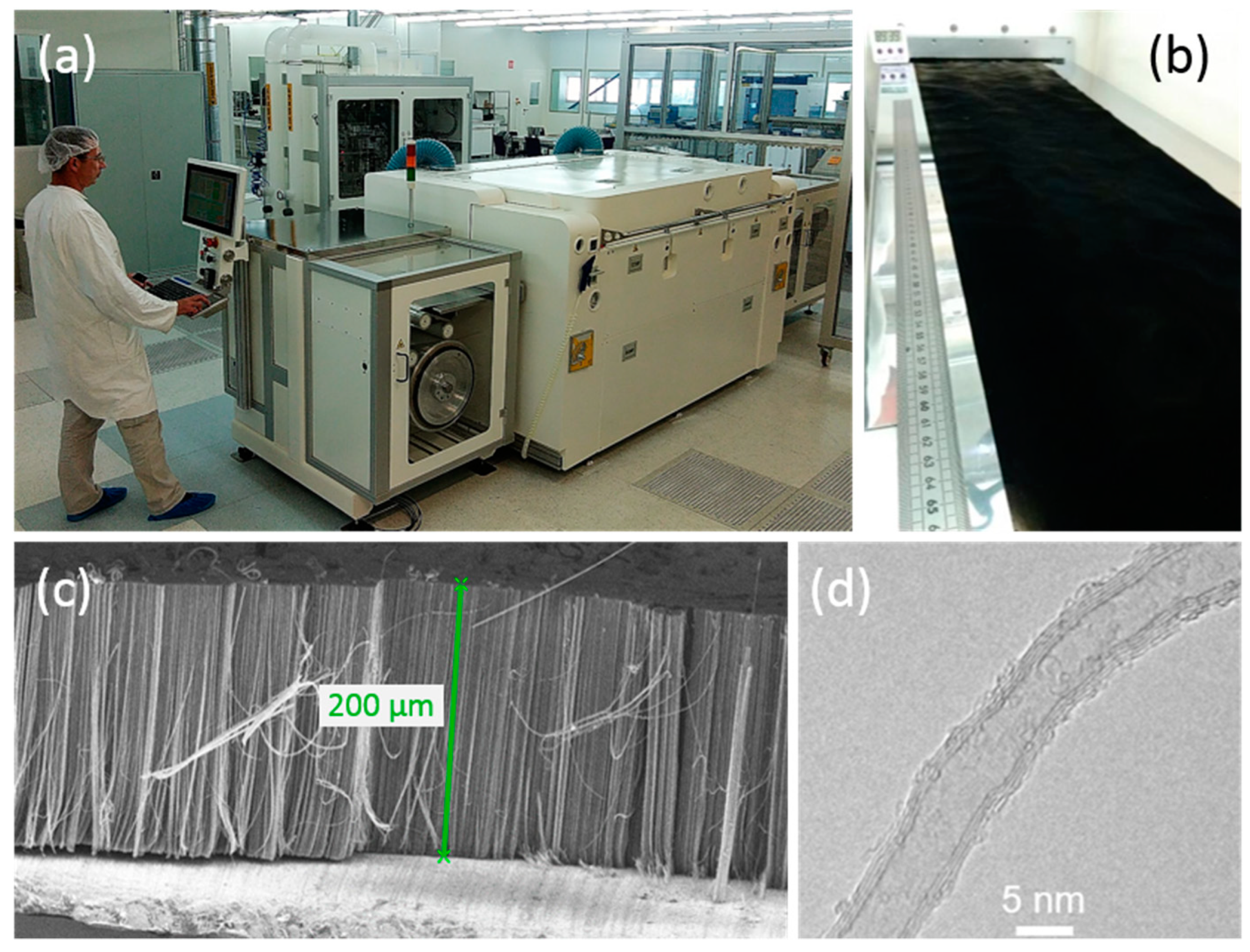
3.3. Toward Application: Industrial Process
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Burke, A. Ultracapacitors: Why, how, and where is the technology. J. Power Sources 2000, 91, 37–50. [Google Scholar] [CrossRef]
- Qu, D.; Shi, H. Studies of the activated carbons used in double-layer supercapacitors. J. Power Sources 1998, 74, 99–107. [Google Scholar] [CrossRef]
- Pandolfo, A.G.; Hollenkamp, A.F. Carbon properties and their role in supercapacitors. J. Power Sources 2006, 157, 11–27. [Google Scholar] [CrossRef]
- Simon, P.; Gogotsi, Y. Capacitive Energy Storage in Nanostructured Carbon-Electrolyte Systems. Acc. Chem. Res. 2013, 46, 1094–1103. [Google Scholar] [PubMed]
- Emmenegger, C.; Bonard, J.M.; Mauron, P.; Sudan, P.; Lepora, A.; Grobety, B.; Züttel, A.; Schlapbach, L. Synthesis of carbon nanotubes over Fe catalyst on aluminium and suggested growth mechanism. Carbon N. Y. 2003, 41, 539–547. [Google Scholar] [CrossRef]
- Khavrus, V.O.; Weiser, M.; Fritsch, M.; Ummethala, R.; Salvaggio, M.G.; Schneider, M.; Kusnezoff, M.; Leonhardt, A. Application of carbon nanotubes directly grown on aluminum foils as electric double layer capacitor electrodes. Chem. Vap. Depos. 2012, 18, 53–60. [Google Scholar]
- Chen, T.; Dai, L. Carbon nanomaterials for high-performance supercapacitors. Mater. Today 2013, 16, 272–280. [Google Scholar]
- Zhang, H.; Cao, G.; Yang, Y.; Gu, Z. Comparison Between Electrochemical Properties of Aligned Carbon Nanotube Array and Entangled Carbon Nanotube Electrodes. J. Electrochem. Soc. 2008, 155, K19. [Google Scholar]
- Fedorovskaya, E.O.; Bulusheva, L.G.; Kurenya, A.G.; Asanov, I.P.; Rudina, N.A.; Funtov, K.O.; Lyubutin, I.S.; Okotrub, A.V. Supercapacitor performance of vertically aligned multiwall carbon nanotubes produced by aerosol-assisted CCVD method. Electrochim. Acta 2014, 139, 165–172. [Google Scholar]
- Lagoutte, S.; Aubert, P.-H.; Pinault, M.; Tran-Van, F.O.; Mayne-L’hermite, M.; Chevrot, C. Poly(3-methylthiophene)/Vertically Aligned Multi-walled Carbon Nanotubes: Electrochemical Synthesis, Characterizations and Electrochemical Storage Properties in Ionic Liquids. Electrochim. Acta 2014, 130, 754–765. [Google Scholar] [CrossRef]
- Lou, F.; Zhou, H.; Huang, F.; Vullum-Bruer, F.; Tran, T.D.; Chen, D. Facile synthesis of manganese oxide/aligned carbon nanotubes over aluminium foil as 3D binder free cathodes for lithium ion batteries. J. Mater. Chem. A 2013, 1, 3757. [Google Scholar] [CrossRef]
- Chen, H.; Zeng, S.; Chen, M.; Zhang, Y.; Li, Q. Fabrication and functionalization of carbon nanotube films for high-performance flexible supercapacitors. Carbon N. Y. 2015, 92, 271–296. [Google Scholar] [CrossRef]
- Pal, S.K.; Talapatra, S.; Kar, S.; Ci, L.; Vajtai, R.; Schadler, L.S.; Ajayan, P.M.; Borca-Tasciuc, T.; Schadler, L.S.; Ajayan, P.M. Time and temperature dependence of multi-walled carbon nanotube growth on Inconel 600. Nanotechnology 2008, 19, 045610. [Google Scholar] [CrossRef] [PubMed]
- Delmas, M.; Pinault, M.; Patel, S.; Porterat, D.; Reynaud, C.; Mayne-L’Hermite, M. Growth of long and aligned multi-walled carbon nanotubes on carbon and metal substrates. Nanotechnology 2012, 23, 105604. [Google Scholar] [CrossRef]
- Arcila-Velez, M.R.; Zhu, J.; Childress, A.; Karakaya, M.; Podila, R.; Rao, A.M.; Roberts, M.E. Roll-to-roll synthesis of vertically aligned carbon nanotube electrodes for electrical double layer capacitors. Nano Energy 2014, 8, 9–16. [Google Scholar] [CrossRef]
- Jourdain, V.; Bichara, C. Current understanding of the growth of carbon nanotubes in catalytic chemical vapour deposition. Carbon N. Y. 2013, 58, 2–39. [Google Scholar] [CrossRef]
- Mayne, M.; Grobert, N.; Terrones, M.; Kamalakaran, R.; Rühle, M.; Kroto, H.W.; Walton, D.R.M. Pyrolytic production of aligned carbon nanotubes from homogeneously dispersed benzene-based aerosols. Chem. Phys. Lett. 2001, 338, 101–107. [Google Scholar] [CrossRef]
- Singh, C.; Shaffer, M.S.P.; Windle, A.H. Production of controlled architectures of aligned carbon nanotubes by an injection chemical vapour deposition method. Carbon N. Y. 2003, 41, 359–368. [Google Scholar] [CrossRef]
- Andrews, R.; Jacques, D.; Rao, A.M.; Derbyshire, F.; Qian, D.; Fan, X.; Dickey, E.C.; Chen, J. Continuous production of aligned carbon nanotubes: A step closer to commercial realization. Chem. Phys. Lett. 1999, 303, 467–474. [Google Scholar] [CrossRef]
- Meysami, S.S.; Koós, A.A.; Dillon, F.; Dutta, M.; Grobert, N. Aerosol-assisted chemical vapour deposition synthesis of multi-wall carbon nanotubes: III. Towards upscaling. Carbon N. Y. 2015, 88, 148–156. [Google Scholar] [CrossRef]
- Castro, C.; Pinault, M.; Coste-Leconte, S.; Porterat, D.; Bendiab, N.; Reynaud, C.; Mayne-L’Hermite, M. Dynamics of catalyst particle formation and multi-walled carbon nanotube growth in aerosol-assisted catalytic chemical vapor deposition. Carbon N. Y. 2010, 48, 3807–3816. [Google Scholar] [CrossRef]
- Boulanger, P.; Belkadi, L.; Descarpentries, J.; Porterat, D.; Hibert, E.; Brouzes, A.; Mille, M.; Patel, S.; Pinault, M.; Reynaud, C.; et al. Towards large scale aligned carbon nanotube composites: An industrial safe-by-design and sustainable approach. J. Phys. Conf. Ser. 2013, 429, 012050. [Google Scholar] [CrossRef]
- Yoshikawa, N.; Asari, T.; Kishi, N.; Hayashi, S.; Sugai, T.; Shinohara, H. An efficient fabrication of vertically aligned carbon nanotubes on flexible aluminum foils by catalyst-supported chemical vapor deposition. Nanotechnology 2008, 19, 245607. [Google Scholar] [CrossRef] [PubMed]
- Gao, Z.; Zhang, X.; Zhang, K.; Yuen, M.M.F. Growth of Vertically Aligned Carbon Nanotube Arrays on Al Substrates through Controlled Diffusion of Catalyst. J. Phys. Chem. C 2015, 119, 15636–15642. [Google Scholar] [CrossRef]
- Miura, S.; Yoshihara, Y.; Asaka, M.; Hasegawa, K.; Sugime, H.; Ota, A.; Oshima, H.; Noda, S. Millimeter-tall carbon nanotube arrays grown on aluminum substrates. Carbon N. Y. 2018, 130, 834–842. [Google Scholar] [CrossRef]
- Liatard, S.; Benhamouda, K.; Fournier, A.; Ramos, R.; Barchasz, C.; Dijon, J. Vertically-aligned carbon nanotubes on aluminum as a light-weight positive electrode for lithium-polysulfide batteries. Chem. Commun. 2015, 51, 7749–7752. [Google Scholar] [CrossRef]
- Lin, R.; Taberna, P.-L.; Fantini, S.; Presser, V.; Pérez, C.R.; Malbosc, F.; Rupesinghe, N.L.; Teo, K.B.K.; Gogotsi, Y.; Simon, P. Capacitive Energy Storage from −50 to 100 °C Using an Ionic Liquid Electrolyte. J. Phys. Chem. Lett. 2011, 2, 2396–2401. [Google Scholar] [CrossRef]
- Dörfler, S.; Felhösi, I.; Marek, T.; Thieme, S.; Althues, H.; Nyikos, L.; Kaskel, S. High power supercap electrodes based on vertical aligned carbon nanotubes on aluminum. J. Power Sources 2013, 227, 218–228. [Google Scholar] [CrossRef]
- Almkhelfe, H.; Carpena-Nuñez, J.; Back, T.C.; Amama, P.B. Gaseous product mixture from Fischer-Tropsch synthesis as an efficient carbon feedstock for low temperature CVD growth of carbon nanotube carpets. Nanoscale 2016, 8, 13476–13487. [Google Scholar] [CrossRef]
- Szabó, A.; Kecsenovity, E.; Pápa, Z.; Gyulavári, T.; Németh, K.; Horvath, E.; Hernadi, K. Influence of synthesis parameters on CCVD growth of vertically aligned carbon nanotubes over aluminum substrate. Sci. Rep. 2017, 7, 9557. [Google Scholar] [CrossRef]
- Pinault, M.; Pichot, V.; Khodja, H.; Launois, P.; Reynaud, C.; Mayne-L’Hermite, M. Evidence of sequential lift in growth of aligned multiwalled carbon nanotube multilayers. Nano Lett. 2005, 5, 2394–2398. [Google Scholar] [CrossRef] [PubMed]
- Castro, C.; Pinault, M.; Porterat, D.; Reynaud, C.; Mayne-L’Hermite, M. The role of hydrogen in the aerosol-assisted chemical vapor deposition process in producing thin and densely packed vertically aligned carbon nanotubes. Carbon N. Y. 2013, 61, 585–594. [Google Scholar] [CrossRef]
- Bokobza, L.; Bruneel, J.L.; Couzi, M. Raman spectroscopic investigation of carbon-based materials and their composites. Comparison between carbon nanotubes and carbon black. Chem. Phys. Lett. 2013, 590, 153–159. [Google Scholar] [CrossRef]
- DiLeo, R.A.; Landi, B.J.; Raffaelle, R.P. Purity assessment of multiwalled carbon nanotubes by Raman spectroscopy. J. Appl. Phys. 2007, 101, 064307. [Google Scholar] [CrossRef]
- Ferrari, A.C.; Robertson, J. Resonant Raman spectroscopy of disordered, amorphous, and diamondlike carbon. Phys. Rev. B Condens. Matter Mater. Phys. 2001, 64, 1–13. [Google Scholar] [CrossRef]
- Frackowiak, E.; Metenier, K.; Bertagna, V.; Beguin, F. Supercapacitor electrodes from multiwalled carbon nanotubes. Appl. Phys. Lett. 2000, 77, 2421–2423. [Google Scholar] [CrossRef]
- Reit, R.; Nguyen, J.; Ready, W.J. Growth time performance dependence of vertically aligned carbon nanotube supercapacitors grown on aluminum substrates. Electrochim. Acta 2013, 91, 96–100. [Google Scholar] [CrossRef]
- Xia, J.; Chen, F.; Li, J.; Tao, N. Measurement of the quantum capacitance of graphene. Nat. Nanotechnol. 2009, 4, 505–509. [Google Scholar] [CrossRef]
- Bouillonnec, J. Elaboration et Etude des Propriétés Mécaniques et Thermiques de Matériaux Constitués de Nanotubes de Carbone Verticalement Alignés. Ph.D. Thesis, Université Toulouse III–Paul Sabatier, Toulouse, France, 2015. [Google Scholar]
- Xiang, R.; Luo, G.; Qian, W.; Zhang, Q.; Wang, Y.; Wei, F.; Li, Q.; Cao, A. Encapsulation, compensation, and substitution of catalyst particles during continuous growth of carbon nanotubes. Adv. Mater. 2007, 19, 2360–2363. [Google Scholar] [CrossRef]
- Jeong, S.; Lee, J.; Kim, H.C.; Hwang, J.Y.; Ku, B.C.; Zakharov, D.N.; Maruyama, B.; Stach, E.A.; Kim, S.M. Direct observation of morphological evolution of a catalyst during carbon nanotube forest growth: New insights into growth and growth termination. Nanoscale 2016, 8, 2055–2062. [Google Scholar] [CrossRef]
- Puretzky, A.A.; Geohegan, D.B.; Jesse, S.; Ivanov, I.N.; Eres, G. In situ measurements and modeling of carbon nanotube array growth kinetics during chemical vapor deposition. Appl. Phys. A 2005, 81, 223–240. [Google Scholar] [CrossRef]
- Zhu, L.; Hess, D.W.; Wong, C. Monitoring carbon nanotube growth by formation of nanotube stacks and investigation of the diffusion-controlled kinetics. J. Phys. Chem. B 2006, 110, 5445–5449. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Gregoire, J.M.; Van Dover, R.B.; Hart, A.J. Supporting Information for Ethanol-Promoted High-Yield Growth of Few-Walled Carbon Nanotubes. J. Phys. Chem. C 2010, 114, 6389–6395. [Google Scholar] [CrossRef]
- Futaba, D.N.; Hata, K.; Yamada, T.; Mizuno, K.; Yumura, M.; Iijima, S. Kinetics of water-assisted single-walled carbon nanotube synthesis revealed by a time-evolution analysis. Phys. Rev. Lett. 2005, 95, 056104. [Google Scholar] [CrossRef]
- Einarsson, E.; Murakami, Y.; Kadowaki, M.; Maruyama, S. Growth dynamics of vertically aligned single-walled carbon nanotubes from in situ measurements. Carbon N. Y. 2008, 46, 923–930. [Google Scholar] [CrossRef]
- Zhang, Y.; Gregoire, J.M.; Van Dover, R.B.; Hart, A.J. Ethanol-Promoted High-Yield Growth of Few-Walled Carbon Nanotubes. J. Phys. Chem. C 2010, 114, 6389–6395. [Google Scholar] [CrossRef]
- Landois, P.; Pinault, M.; Rouzière, S.; Porterat, D.; Mocuta, C.; Elkaim, E.; Mayne-L’Hermite, M.; Launois, P. In situ time resolved wide angle X-Ray diffraction study of nanotube carpet growth: Nature of catalyst particles and progressive nanotube alignment. Carbon N. Y. 2015, 87, 246–256. [Google Scholar] [CrossRef]


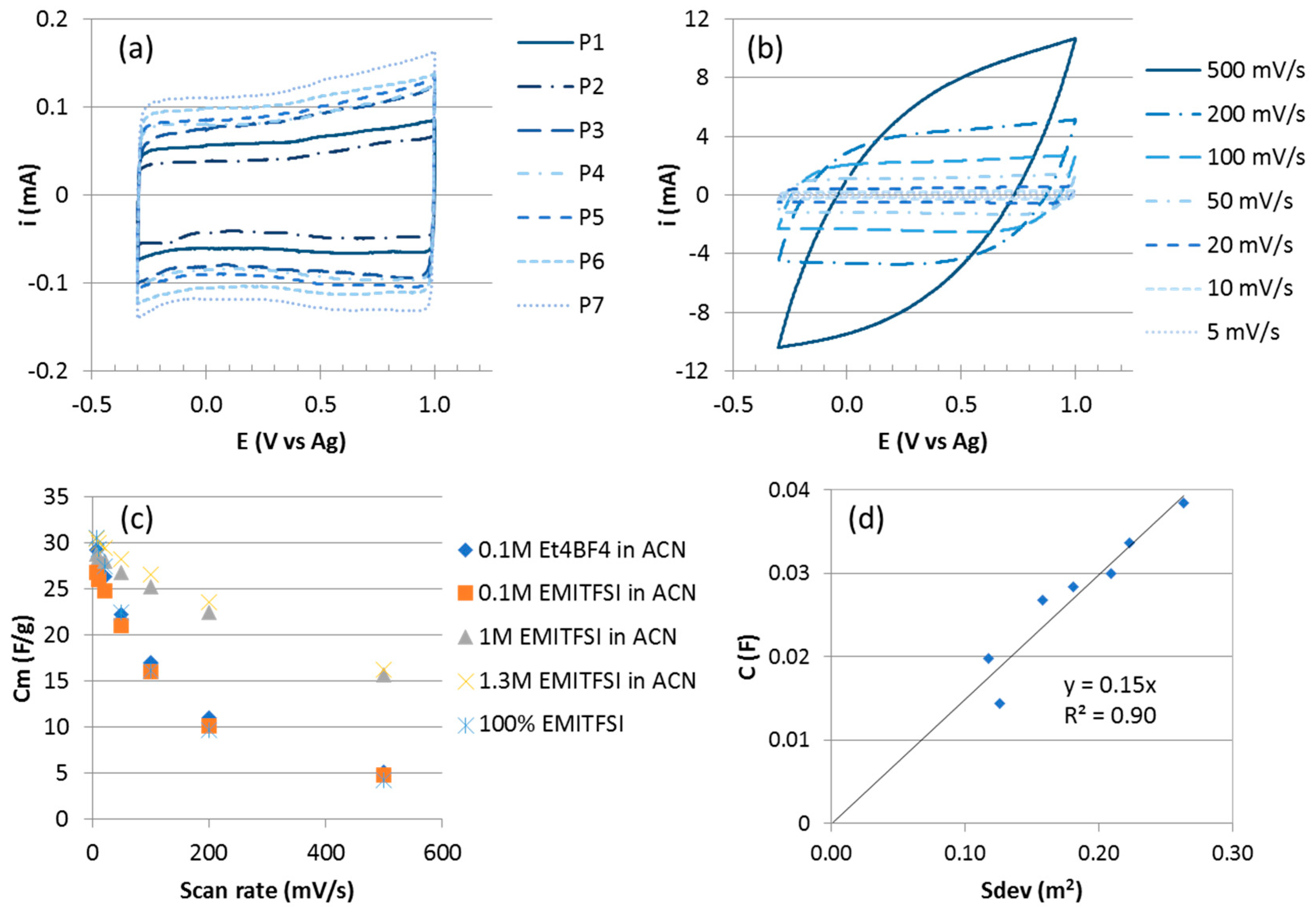
| Sample | Height (µm) | Mass (mg) | Diameter (nm) | Density (1010 CNT/cm2) | Sdev (m2) | Cm (F/g) | C (mF) |
|---|---|---|---|---|---|---|---|
| P1 | 99 | 0.42 | 8.1 | 5.9 | 0.117 | 47 | 19.7 |
| P2 | 96 | 0.45 | 8.1 | 6.6 | 0.126 | 32 | 14.4 |
| P3 | 49 | 0.57 | 8.3 | 15.7 | 0.158 | 47 | 26.8 |
| P4 | 65 | 0.81 | 10.6 | 10.6 | 0.181 | 35 | 28.4 |
| P5 | 53.5 | 0.79 | 8.7 | 18.2 | 0.209 | 38 | 30.0 |
| P6 | 56 | 0.84 | 8.7 | 18.5 | 0.223 | 40 | 33.6 |
| P7 | 125 | 1.28 | 11.1 | 7.7 | 0.263 | 30 | 38.4 |
| Coefficient | hmax (µm) | γ₀ (µm/min) | τ (min) |
|---|---|---|---|
| Single injection 580 °C | 65 | 3.8 | 17 |
| Single injection 615 °C | 121 | 8.3 | 15 |
| Sequential injection 580 °C | 100 | 3.0 | 33 |
| Sequential injection 615 °C | 215 | 6.0 | 36 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nassoy, F.; Pinault, M.; Descarpentries, J.; Vignal, T.; Banet, P.; Coulon, P.-E.; Goislard de Monsabert, T.; Hauf, H.; Aubert, P.-H.; Reynaud, C.; et al. Single-Step Synthesis of Vertically Aligned Carbon Nanotube Forest on Aluminium Foils. Nanomaterials 2019, 9, 1590. https://doi.org/10.3390/nano9111590
Nassoy F, Pinault M, Descarpentries J, Vignal T, Banet P, Coulon P-E, Goislard de Monsabert T, Hauf H, Aubert P-H, Reynaud C, et al. Single-Step Synthesis of Vertically Aligned Carbon Nanotube Forest on Aluminium Foils. Nanomaterials. 2019; 9(11):1590. https://doi.org/10.3390/nano9111590
Chicago/Turabian StyleNassoy, Fabien, Mathieu Pinault, Jérémie Descarpentries, Thomas Vignal, Philippe Banet, Pierre-Eugène Coulon, Thomas Goislard de Monsabert, Harald Hauf, Pierre-Henri Aubert, Cécile Reynaud, and et al. 2019. "Single-Step Synthesis of Vertically Aligned Carbon Nanotube Forest on Aluminium Foils" Nanomaterials 9, no. 11: 1590. https://doi.org/10.3390/nano9111590
APA StyleNassoy, F., Pinault, M., Descarpentries, J., Vignal, T., Banet, P., Coulon, P.-E., Goislard de Monsabert, T., Hauf, H., Aubert, P.-H., Reynaud, C., & Mayne-L’Hermite, M. (2019). Single-Step Synthesis of Vertically Aligned Carbon Nanotube Forest on Aluminium Foils. Nanomaterials, 9(11), 1590. https://doi.org/10.3390/nano9111590






