Ag/H-ZIF-8 Nanocomposite as an Effective Antibacterial Agent Against Pathogenic Bacteria
Abstract
1. Introduction
2. Experimental
2.1. Materials
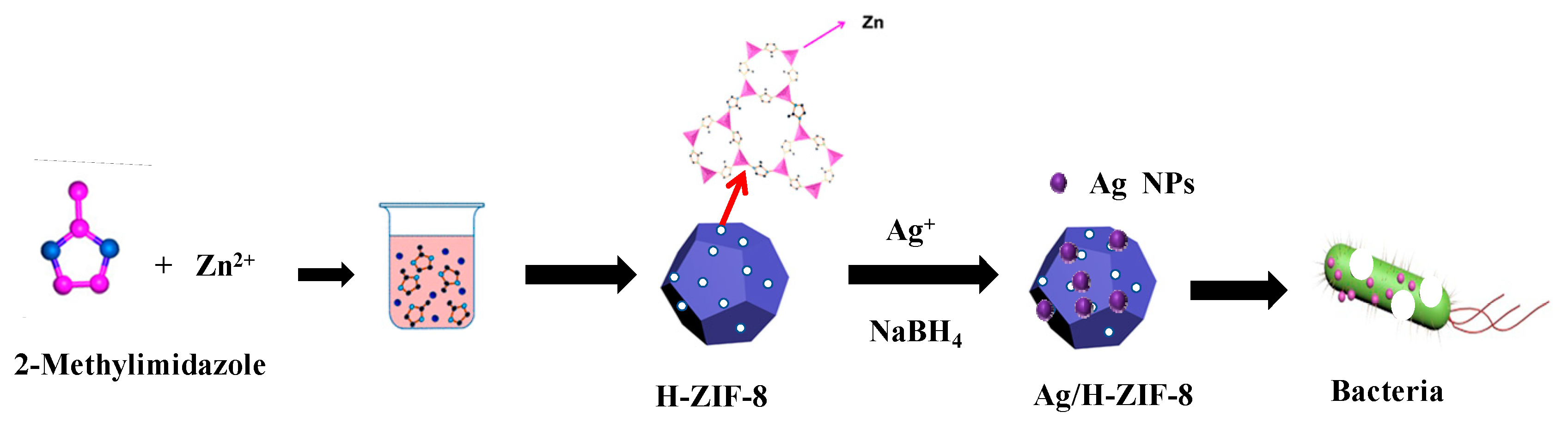
2.2. Synthesis of Hierarchical ZIF-8
2.3. Loading of Ag Nanoparticles into the H-ZIF-8
2.4. Antibacterial Assay
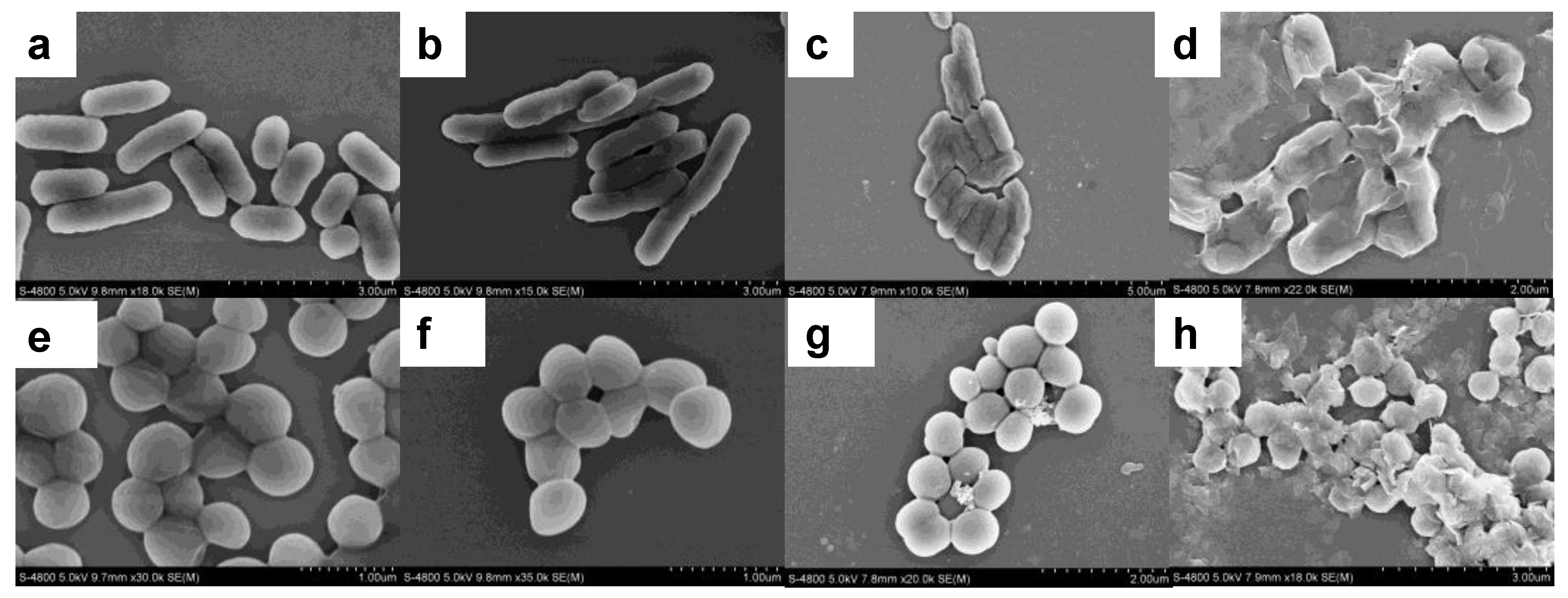
2.5. Preparation of Bacterial Samples for SEM
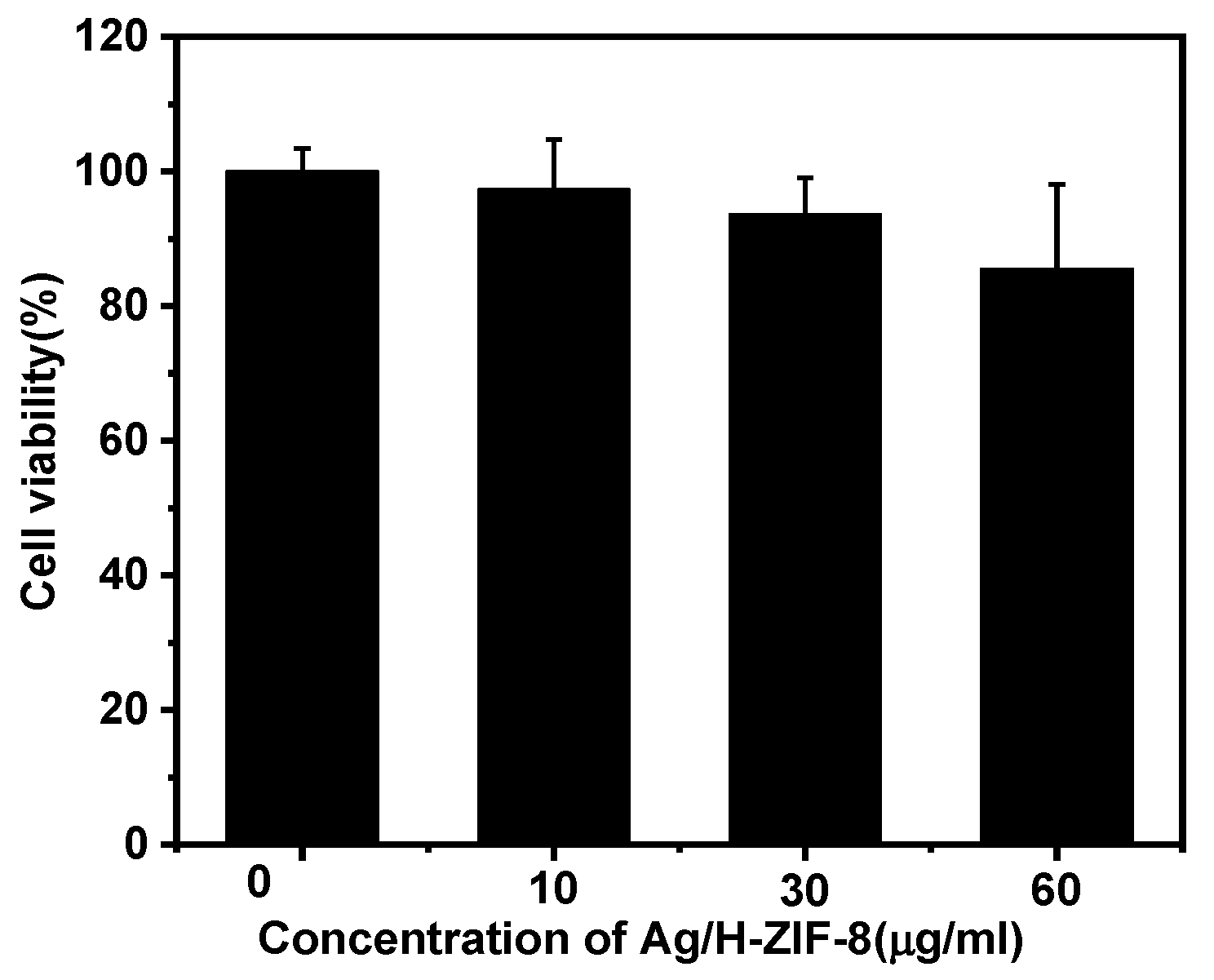
2.6. Cytotoxicity of Ag/H-ZIF-8
3. Results and Discussion
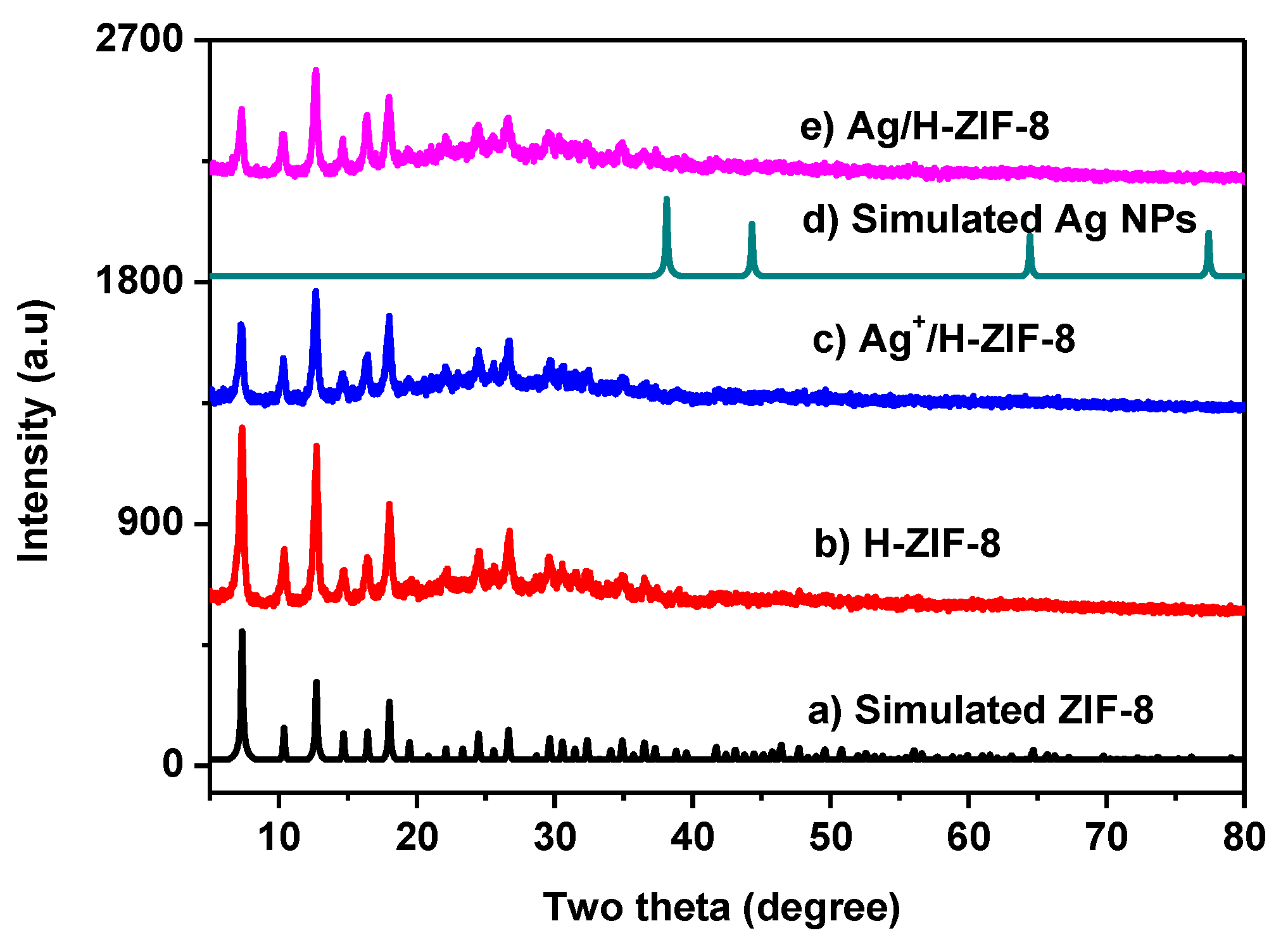
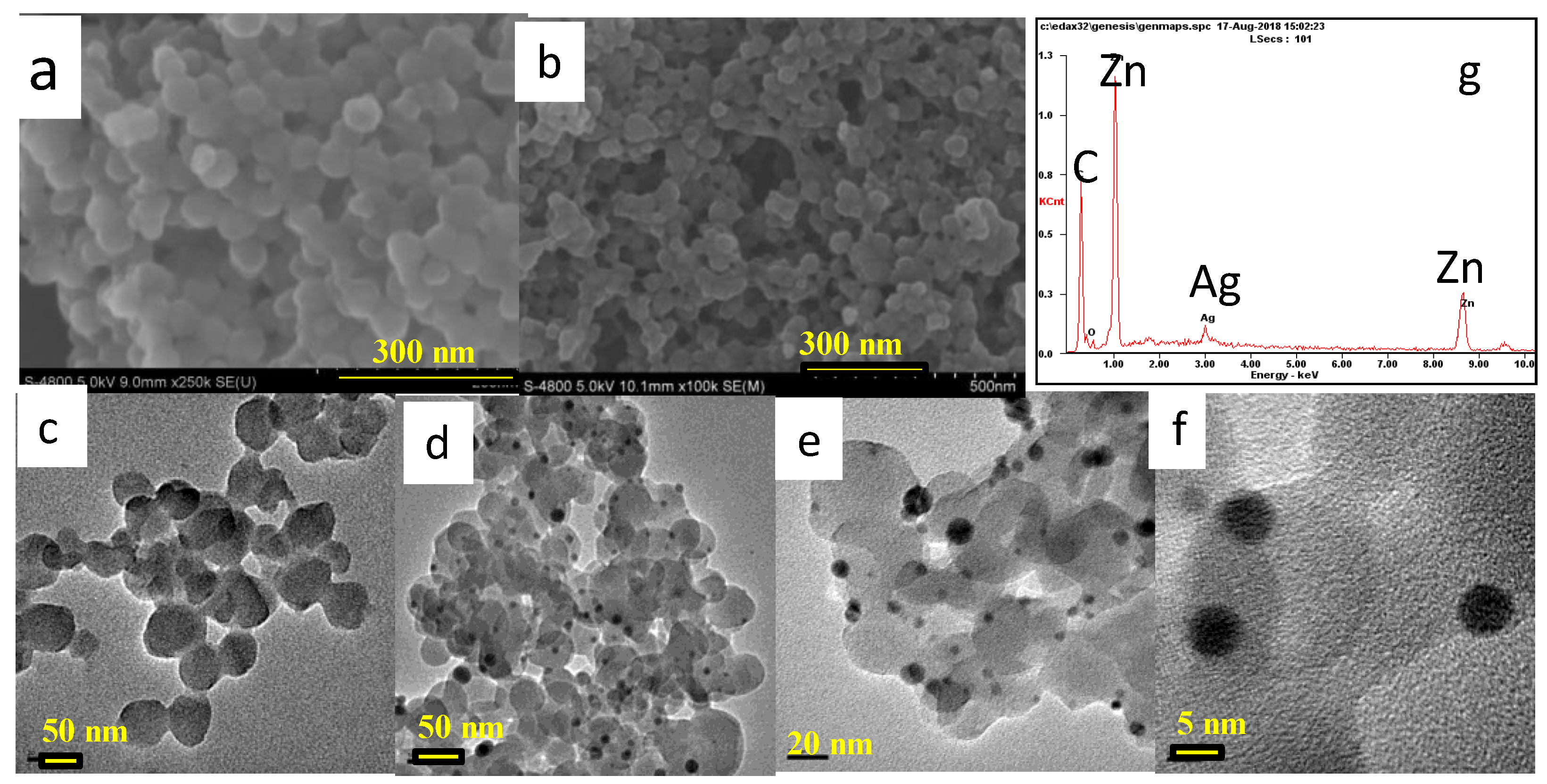
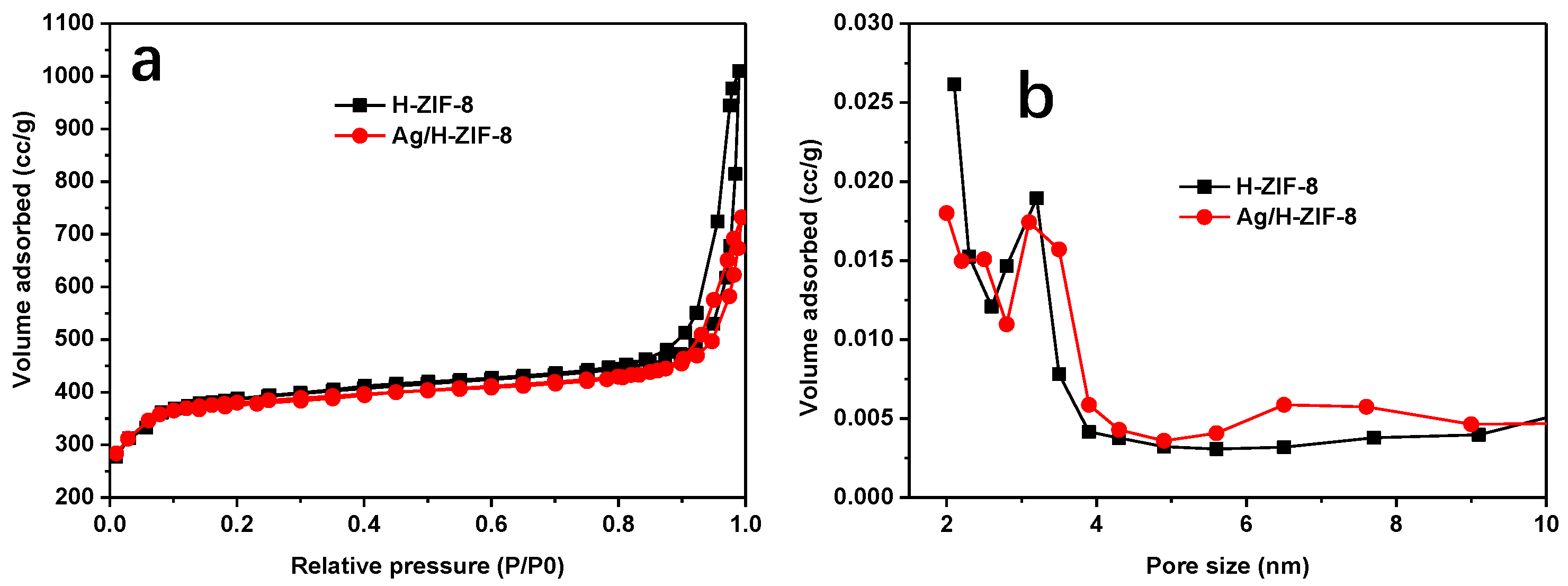
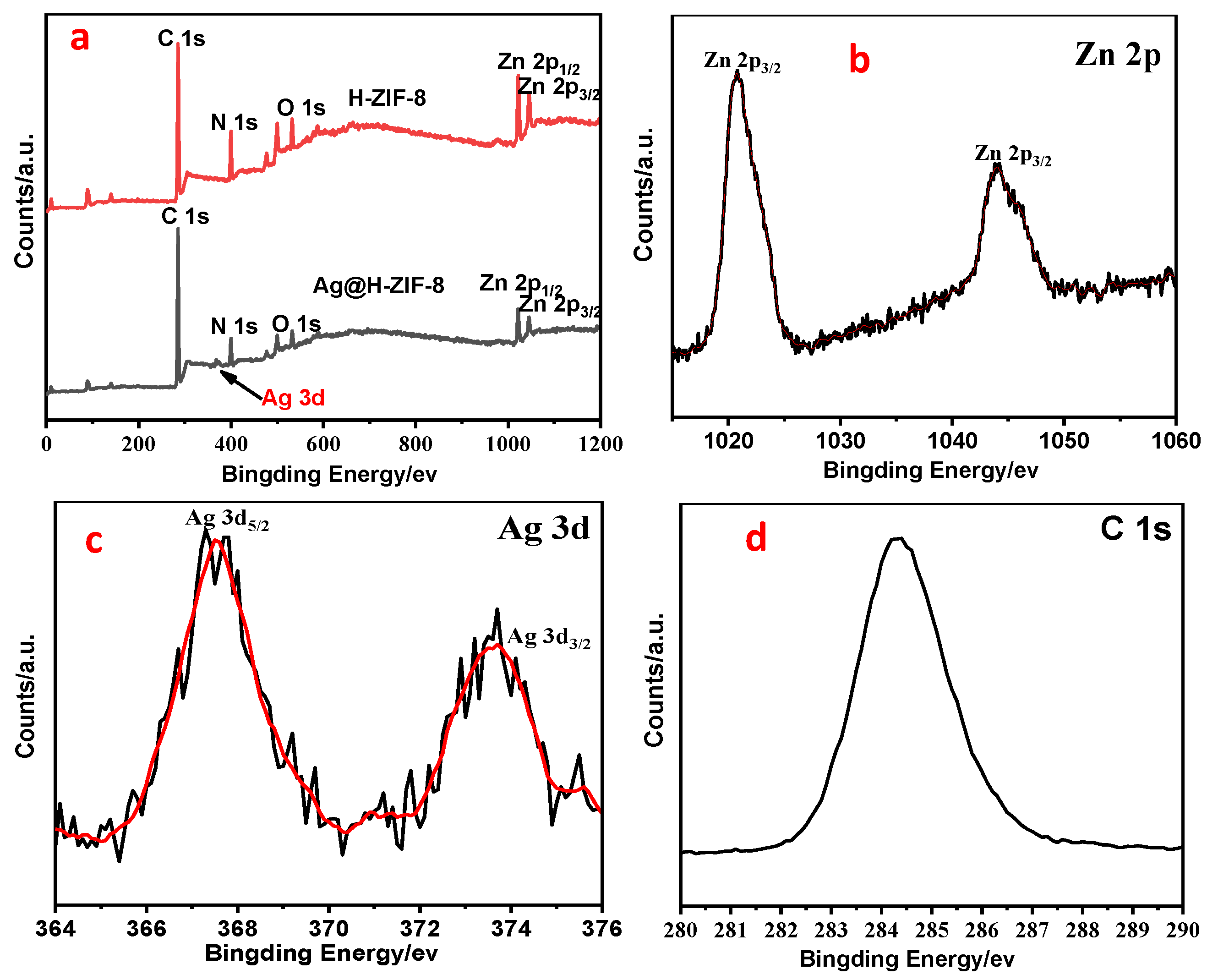
3.1. Characterization of the Synthesized Materials
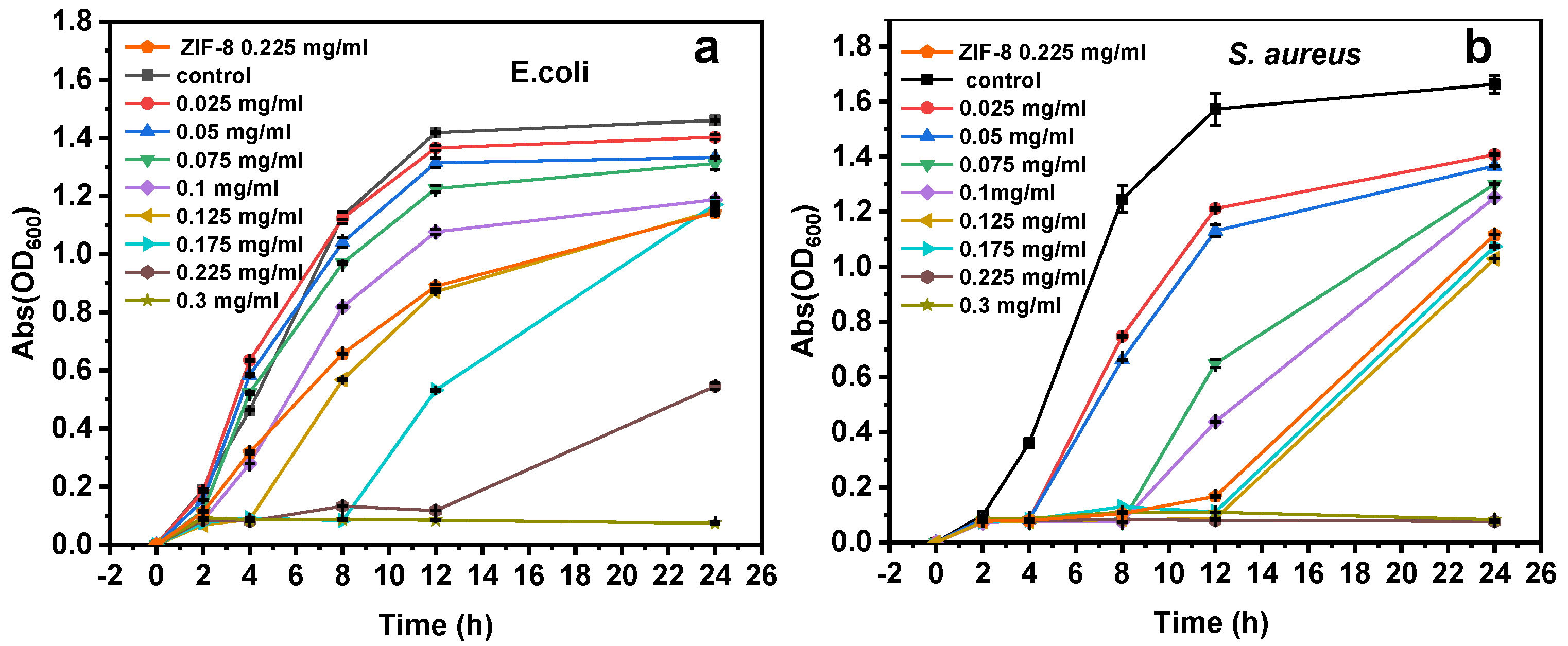
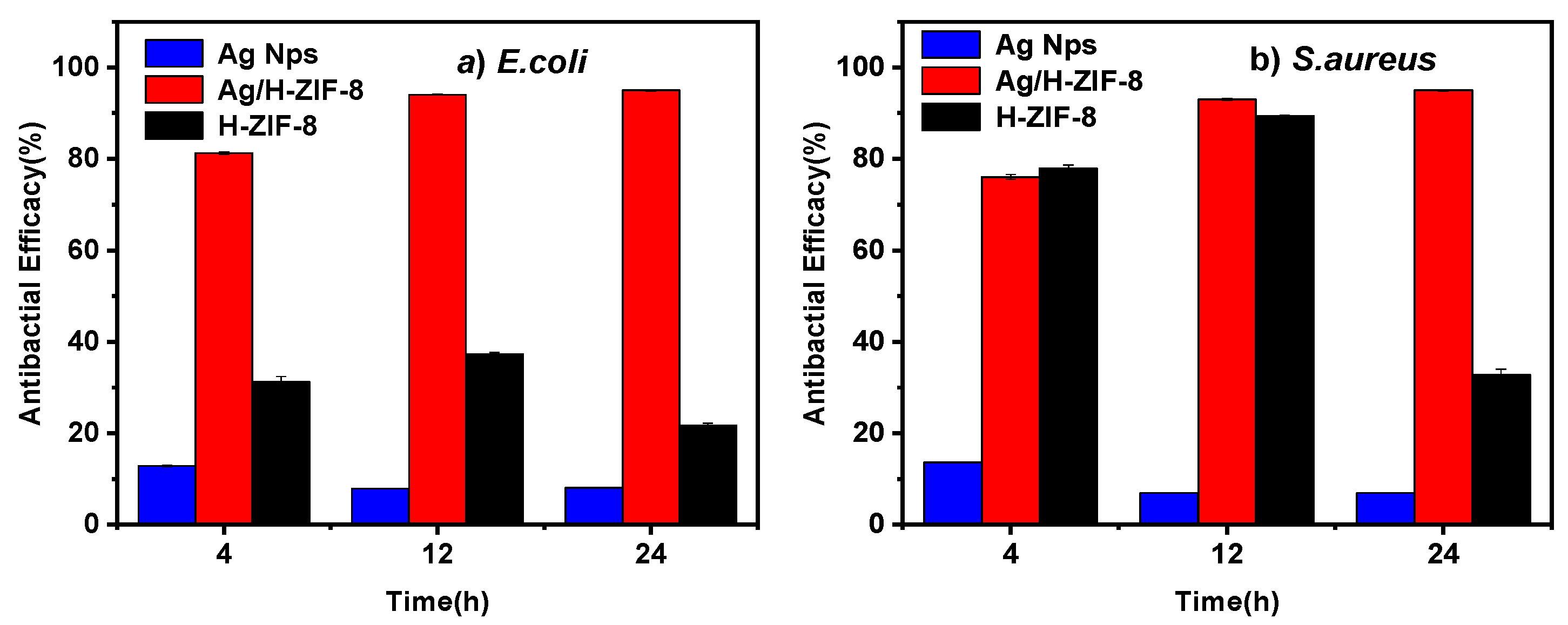
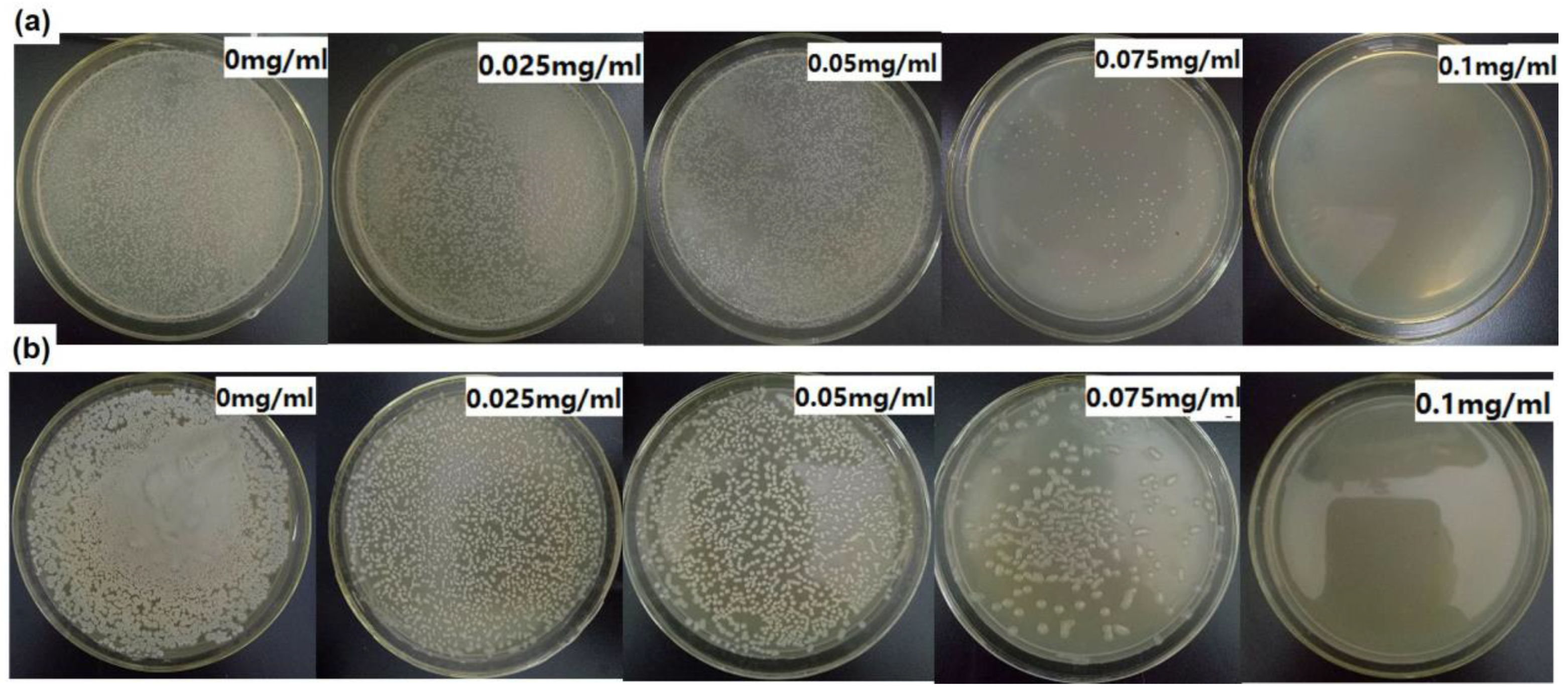
3.2. Antibacterial Activity
3.3. Cytotoxicity Assay
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Rizzello, L.; Pompa, P.P. Nanosilver-based antibacterial drugs and devices: Mechanisms, methodological drawbacks, and guidelines. Chem. Soc. Rev. 2014, 43, 1501–1518. [Google Scholar] [CrossRef] [PubMed]
- Hajipour, M.J.; Fromm, K.M.; Ashkarran, A.A.; de Aberasturi, D.J.; de Larramendi, I.R.; Rojo, T.; Serpooshan, V.; Parak, W.J.; Mahmoudi, M. Antibacterial properties of nanoparticles. Trends Biotechnol. 2012, 30, 499–511. [Google Scholar] [CrossRef] [PubMed]
- Rice, K.M.; Ginjupalli, G.K.; Manne, N.D.P.K.; Jones, C.B.; Blough, E.R. A review of the antimicrobial potential of precious metal derived nanoparticle constructs. Nanotechnology 2019, 30, 372001. [Google Scholar] [CrossRef] [PubMed]
- Chernousova, S.; Epple, M. Silver as antibacterial agent: Ion, nanoparticle, and metal. Angew. Chem. Int. Ed. 2013, 52, 1636–1653. [Google Scholar] [CrossRef] [PubMed]
- Deng, Z.W.; Zhu, H.B.; Peng, B.; Chen, H.; Sun, Y.F.; Gang, X.D.; Jin, P.J.; Wang, J.L. Synthesis of PS/Ag nanocomposite spheres with catalytic and antibacterial activities. ACS Appl. Mater. Interfaces 2012, 4, 5625–5632. [Google Scholar] [CrossRef]
- Huang, F.; Gao, Y.; Zhang, Y.M.; Cheng, T.J.; Ou, H.L.; Yang, L.J.; Cheng, T.J.; Ou, H.L.; Yang, L.J.; Liu, J.J.; et al. Silver-decorated polymeric micelles combined with curcumin for enhanced antibacterial activity. ACS Appl. Mater. Interfaces 2017, 9, 16881–16890. [Google Scholar] [CrossRef]
- Qin, D.Z.; Yang, G.R.; Wang, Y.B.; Zhou, Y.B.; Zhang, L. Green synthesis of biocompatible trypsin-conjugated Ag nanocomposite with antibacterial activity. Appl. Surf. Sci. 2019, 469, 528–536. [Google Scholar] [CrossRef]
- Jing, N.; Li, H.Y.; Yu, H.; Yan, Z.W.; Xu, Q.H.; Wang, Z. Synthesis of Ag nanoparticles via "Molecular Cage" method for antibacterial application. J. Nanosci. Nanotechnol. 2019, 19, 780–785. [Google Scholar] [CrossRef]
- Aydin, M.T.A.; Hosgun, H.L.; Dede, A.; Guven, K. Synthesis, characterization and antibacterial activity of silver-doped TiO2 nanotubes. Spectrochim. Acta Part A 2018, 205, 503–507. [Google Scholar] [CrossRef]
- Li, S.; Wang, Q.T.; Yu, H.Q.; Ben, T.; Xu, H.J.; Zhang, J.C.; Du, Q.Y. Preparation of effective Ag-loaded zeolite antibacterial materials by solid phase ionic exchange method. J. Porous Mater. 2018, 25, 1797–1804. [Google Scholar] [CrossRef]
- Furukawa, H.; Cordova, K.E.; O’Keeffe, M.; Yaghi, O.M. The chemistry and applications of metal-organic frameworks. Science 2013, 341, 1230444. [Google Scholar] [CrossRef] [PubMed]
- Guan, H.Y.; LeBlanc, R.J.; Xie, S.Y.; Yue, Y.F. Recent progress in the syntheses of mesoporous metal-organic framework materials. Coord. Chem. Rev. 2018, 369, 76–90. [Google Scholar] [CrossRef]
- Chen, L.Y.; Luque, R.; Li, Y.W. Controllable design of tunable nanostructures inside metal-organic frameworks. Chem. Soc. Rev. 2017, 46, 4614–4630. [Google Scholar] [CrossRef] [PubMed]
- Adnan, M.; Li, K.; Wang, J.H.; Xu, L.; Yan, Y.J. Hierarchical ZIF-8 toward immobilizing burkholderia cepacia lipase for application in biodiesel preparation. Int. J. Mol. Sci. 2018, 19, 1424. [Google Scholar] [CrossRef] [PubMed]
- Fletcher, N.D.; Lieb, H.C.; Mullaugh, K.M. Stability of silver nanoparticle sulfidation products. Sci. Total Environ. 2019, 648, 854–860. [Google Scholar] [CrossRef]
- Guo, Y.F.; Fang, W.J.; Fu, J.R.; Wu, Y.; Zheng, J.; Gao, G.Q.; Chen, C.; Yan, R.W.; Huang, S.G.; Wang, C.C. Facile synthesis of Ag@ZIF-8 core-shell heterostructure nanowires for improved antibacterial activities. Appl. Surf. Sci. 2018, 435, 149–155. [Google Scholar] [CrossRef]
- Song, Z.Y.; Wu, Y.; Wang, H.J.; Han, H.Y. Synergistic antibacterial effects of curcumin modified silver nanoparticles through ROS-mediated pathways. Mater. Sci. Eng. C 2019, 99, 255–263. [Google Scholar] [CrossRef]
- Tippayawat, P.; Phromviyo, N.; Boueroy, P.; Chompoosor, A. Green synthesis of silver nanoparticles in aloe vera plant extract prepared by a hydrothermal method and their synergistic antibacterial activity. PeerJ 2016, 4, 1–15. [Google Scholar] [CrossRef]
- Wu, R.F.; Chong, Y.; Fang, G.; Jiang, X.M.; Pan, Y.; Chen, C.Y.; Yin, J.J.; Ge, C.C. Synthesis of Pt hollow nanodendrites with enhanced peroxidase-like activity against bacterial infections: Implication for wound healing. Adv. Funct. Mater. 2018, 28, 1801484. [Google Scholar]
- Jiang, H.L.; Liu, B.; Akita, T.; Haruta, M.; Sakurai, H.; Xu, Q. Au@ZIF-8: CO oxidation over gold nanoparticles deposited to metal-organic framework. J. Am. Chem. Soc. 2009, 131, 11302. [Google Scholar] [CrossRef]
- Dhakshinamoorthy, A.; Asiri, A.M.; Garcia, H. Metal organic frameworks as versatile hosts of Au nanoparticles in heterogeneous catalysis. ACS Catal. 2017, 7, 2896–2919. [Google Scholar] [CrossRef]
- Gozdziewska, M.; Cichowicz, G.; Markowska, K.; Zawada, K.; Megiel, E. Nitroxide-coated silver nanoparticles: Synthesis, surface physicochemistry and antibacterial activity. RSC Adv. 2015, 5, 58403–58415. [Google Scholar] [CrossRef]
- Maiti, R.; Sinha, T.K.; Bhattacharya, S.; Datta, P.K.; Ray, S.K. Facile one-pot synthesis of highly stable graphene-Ag-0 hybrid nanostructures with enhanced optical properties. J. Phys. Chem. C 2017, 121, 21591–21599. [Google Scholar] [CrossRef]
- Lu, G.; Li, S.Z.; Guo, Z.; Farha, O.K.; Hauser, B.G.; Qi, X.Y.; Wang, Y.; Wang, X.; Han, S.Y.; Liu, X.G.; et al. Imparting functionality to a metal-organic framework material by controlled nanoparticle encapsulation. Nat. Chem. 2012, 4, 310–316. [Google Scholar] [CrossRef] [PubMed]
- Windiasti, G.; Feng, J.S.; Ma, L.N.; Hu, Y.X.; Hakeem, M.J.; Amoako, K.; Delaquis, P.; Lu, X.N. Investigating the synergistic antimicrobial effect of carvacrol and zinc oxide nanoparticles against Campylobacter jejuni. Food Control 2019, 96, 39–46. [Google Scholar] [CrossRef]
- Rajab, F.H.; Korshed, P.; Liu, Z.; Wang, T.; Li, L. How did the structural ZnO nanowire as antibacterial coatings control the switchable wettability. Appl. Surf. Sci. 2019, 469, 593–606. [Google Scholar] [CrossRef]
- Li, M.; Zhu, L.D.; Lin, D.H. Toxicity of ZnO nanoparticles to escherichia coli: Mechanism and the influence of medium components. Environ. Sci. Technol. 2011, 45, 1977–1983. [Google Scholar] [CrossRef]
- Zaidi, Z.; Vaghasiya, K.; Vijay, A.; Sharma, M.; Verma, R.K.; Vaidya, S. Hollow ZnO from assembly of nanoparticles: Photocatalytic and antibacterial activity. J. Mater. Sci. 2018, 53, 14964–14974. [Google Scholar] [CrossRef]
- Hong, D.; Cao, G.Z.; Qu, J.L.; Deng, Y.M.; Tang, J.N. Antibacterial activity of Cu2O and Ag co-modified rice grains-like ZnO nanocomposites. J. Mater. Sci. Technol. 2018, 34, 2359–2367. [Google Scholar] [CrossRef]
- Azam, A.; Ahmed, A.S.; Oves, M.; Khan, M.S.; Habib, S.S.; Memic, A. Antimicrobial activity of metal oxide nanoparticles against Gram-positive and Gram-negative bacteria: A comparative study. Int. J. Nanomed. 2012, 7, 6003–6009. [Google Scholar] [CrossRef]
- Qiao, Y.; Yang, C.; Coady, D.J.; Ong, Z.Y.; Hedrick, J.L.; Yang, Y.Y. Highly dynamic biodegradable micelles capable of lysing Gram-positive and Gram-negative bacterial membrane. Biomaterials 2012, 33, 1146–1153. [Google Scholar] [CrossRef] [PubMed]
- Takahashia, T.; Imai, M.; Suzuki, I.; Sawai, J. Growth inhibitory effect on bacteria of chitosan membranes regulated with deacetylation degree. Biochem. Eng. J. 2008, 40, 485–491. [Google Scholar] [CrossRef]
- Lan, S.; Lu, Y.N.; Li, C.; Zhao, S.; Liu, N.R.; Sheng, X.L. Sesbania gum-supported hydrophilic electrospun fibers containing nanosilver with superior antibacterial activity. Nanomaterials 2019, 9, 592. [Google Scholar] [CrossRef] [PubMed]
- ErankaIllangakoon, U.E.; Mahalingam, S.; Wang, K.; Cheong, Y.K.; Canales, E.; Ren, G.G.; Cloutman-Green, E.; Edirisinghe, M.; Ciric, L. Gyrospun antimicrobial nanoparticle loaded fibrous polymeric filters. Mater. Sci. Eng. C 2017, 74, 315–324. [Google Scholar] [CrossRef] [PubMed]
- Cheong, Y.K.; Calvo-Castro, J.; Ciric, L.; Edirisinghe, M.; Cloutman-Green, E.; Illangakoon, U.E.; Kang, Q.; Mahalingam, S.; Matharu, R.K.; Wilson, R.M.; et al. Characterisation of the chemical composition and structural features of novel antimicrobial nanoparticles. Nanomaterials 2017, 7, 152. [Google Scholar] [CrossRef]
- Mahalingam, S.; Homer-Vanniasinkam, S.; Edirisinghe, M. Novel pressurised gyration device formaking core-sheath polymer fibres. Mater. Des. 2019, 178, 107846. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, Y.; Zhang, X.; Song, J.; Jin, L.; Wang, X.; Quan, C. Ag/H-ZIF-8 Nanocomposite as an Effective Antibacterial Agent Against Pathogenic Bacteria. Nanomaterials 2019, 9, 1579. https://doi.org/10.3390/nano9111579
Zhang Y, Zhang X, Song J, Jin L, Wang X, Quan C. Ag/H-ZIF-8 Nanocomposite as an Effective Antibacterial Agent Against Pathogenic Bacteria. Nanomaterials. 2019; 9(11):1579. https://doi.org/10.3390/nano9111579
Chicago/Turabian StyleZhang, Yanmei, Xin Zhang, Jie Song, Liming Jin, Xiaotong Wang, and Chunshan Quan. 2019. "Ag/H-ZIF-8 Nanocomposite as an Effective Antibacterial Agent Against Pathogenic Bacteria" Nanomaterials 9, no. 11: 1579. https://doi.org/10.3390/nano9111579
APA StyleZhang, Y., Zhang, X., Song, J., Jin, L., Wang, X., & Quan, C. (2019). Ag/H-ZIF-8 Nanocomposite as an Effective Antibacterial Agent Against Pathogenic Bacteria. Nanomaterials, 9(11), 1579. https://doi.org/10.3390/nano9111579





