Enhanced Protective Coatings Based on Nanoparticle fullerene C60 for Oil & Gas Pipeline Corrosion Mitigation
Abstract
1. Introduction
2. Experiment
2.1. Material
2.2. Fabrication of Nanofiller-Reinforced Epoxy Composites and Test Sample Preparation
2.3. Characterization Method
2.3.1. Characterization
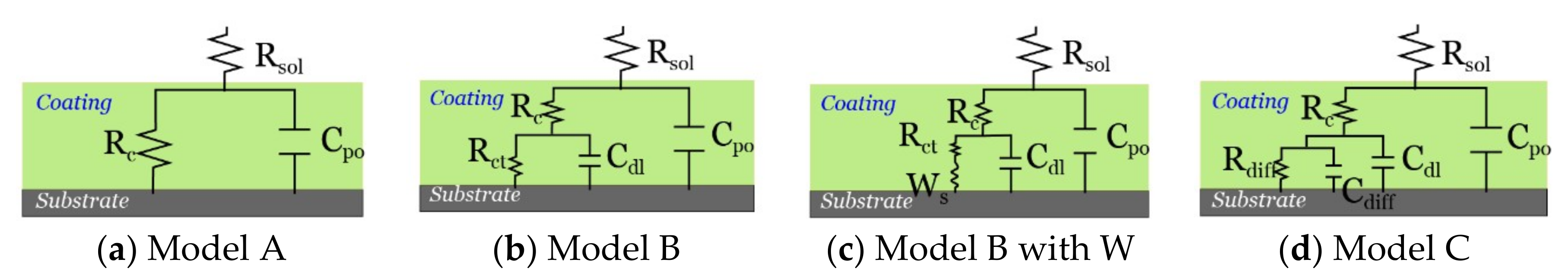
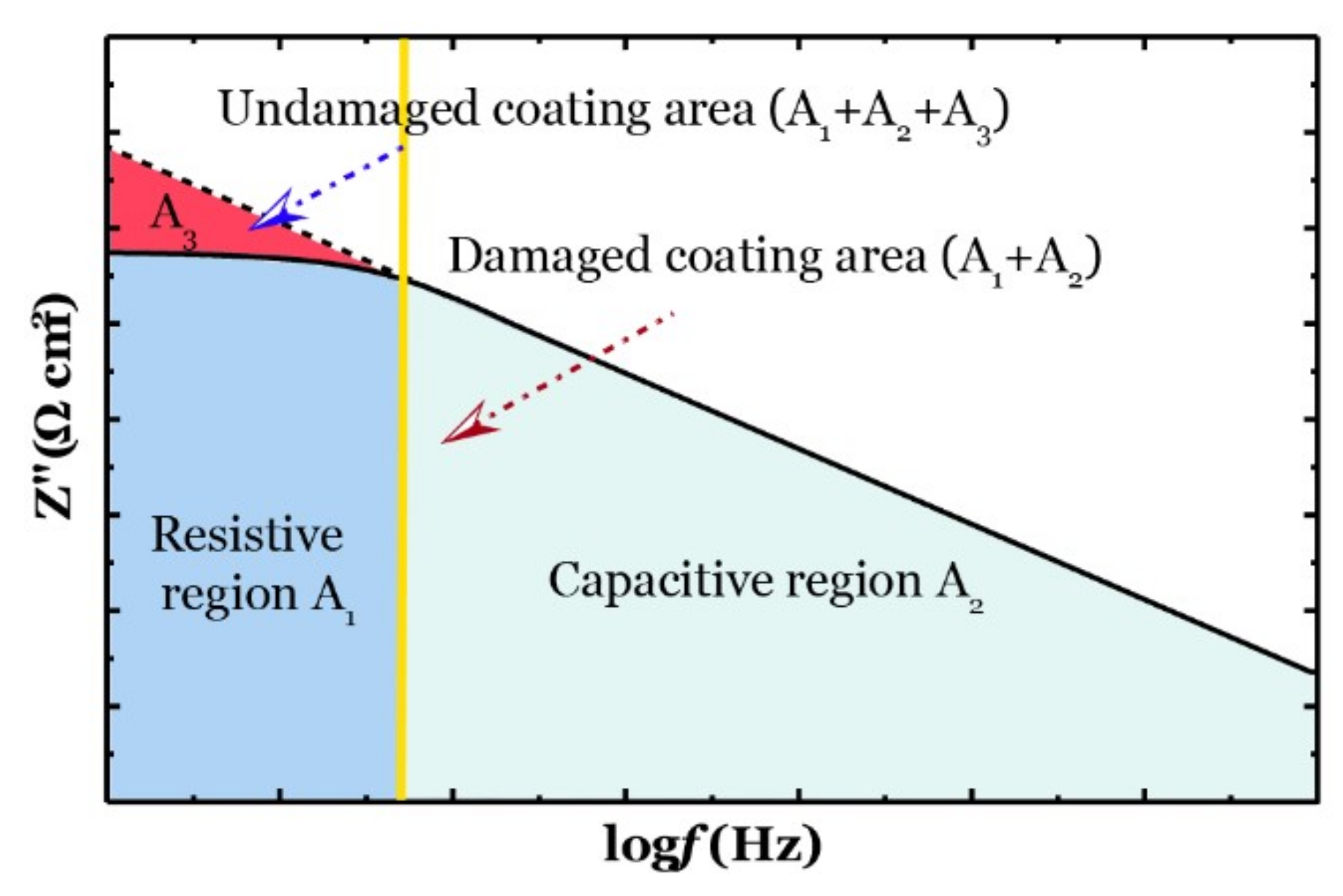
2.3.2. Corrosion Resistance of the Composite Coating Using EIS
2.3.3. Adhesion of the Composite Coating Using Tensile Button Testing
2.3.4. Tensile Strength, Ultimate Strain, and Young’s Modulus
3. Results and Discussion
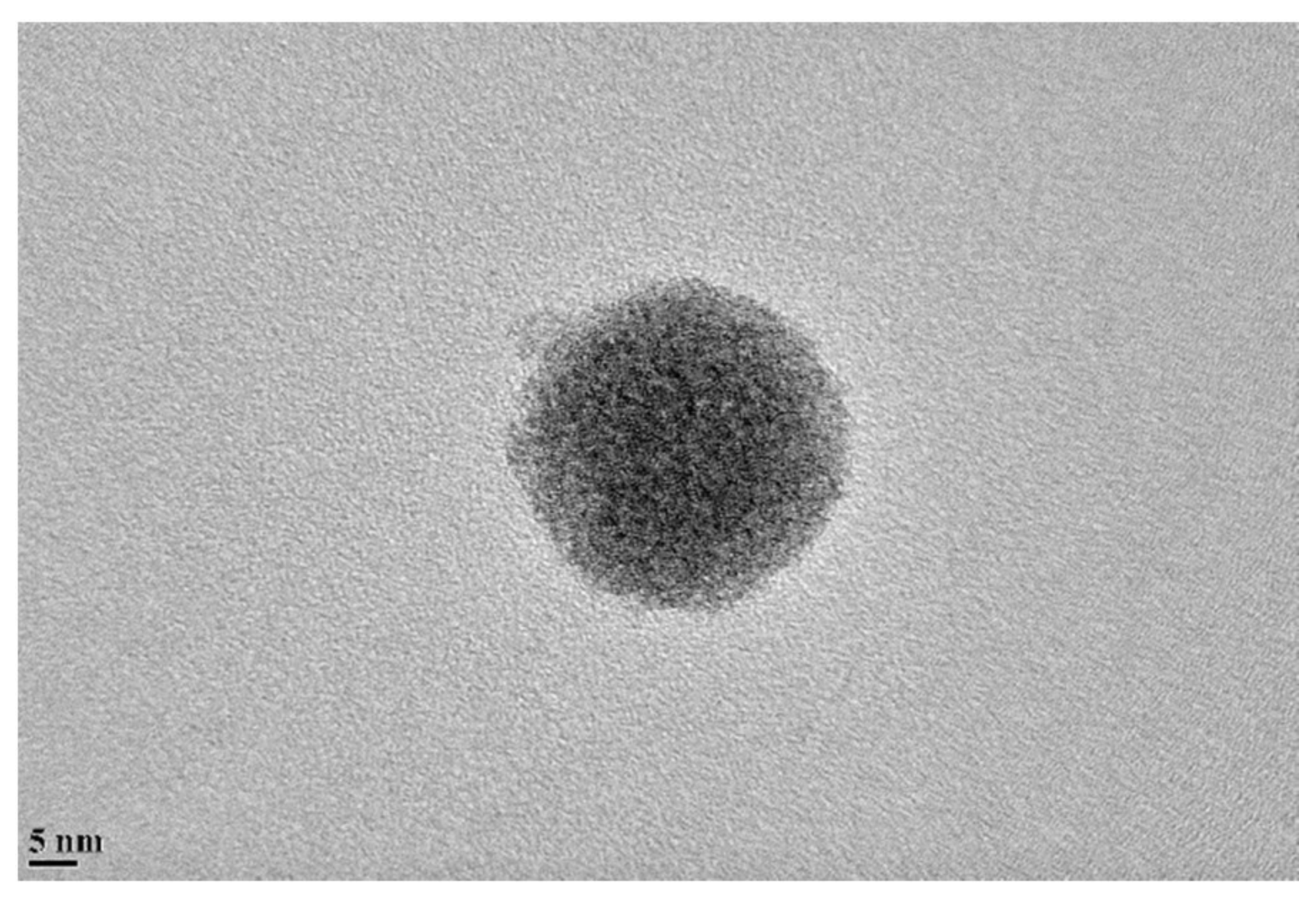
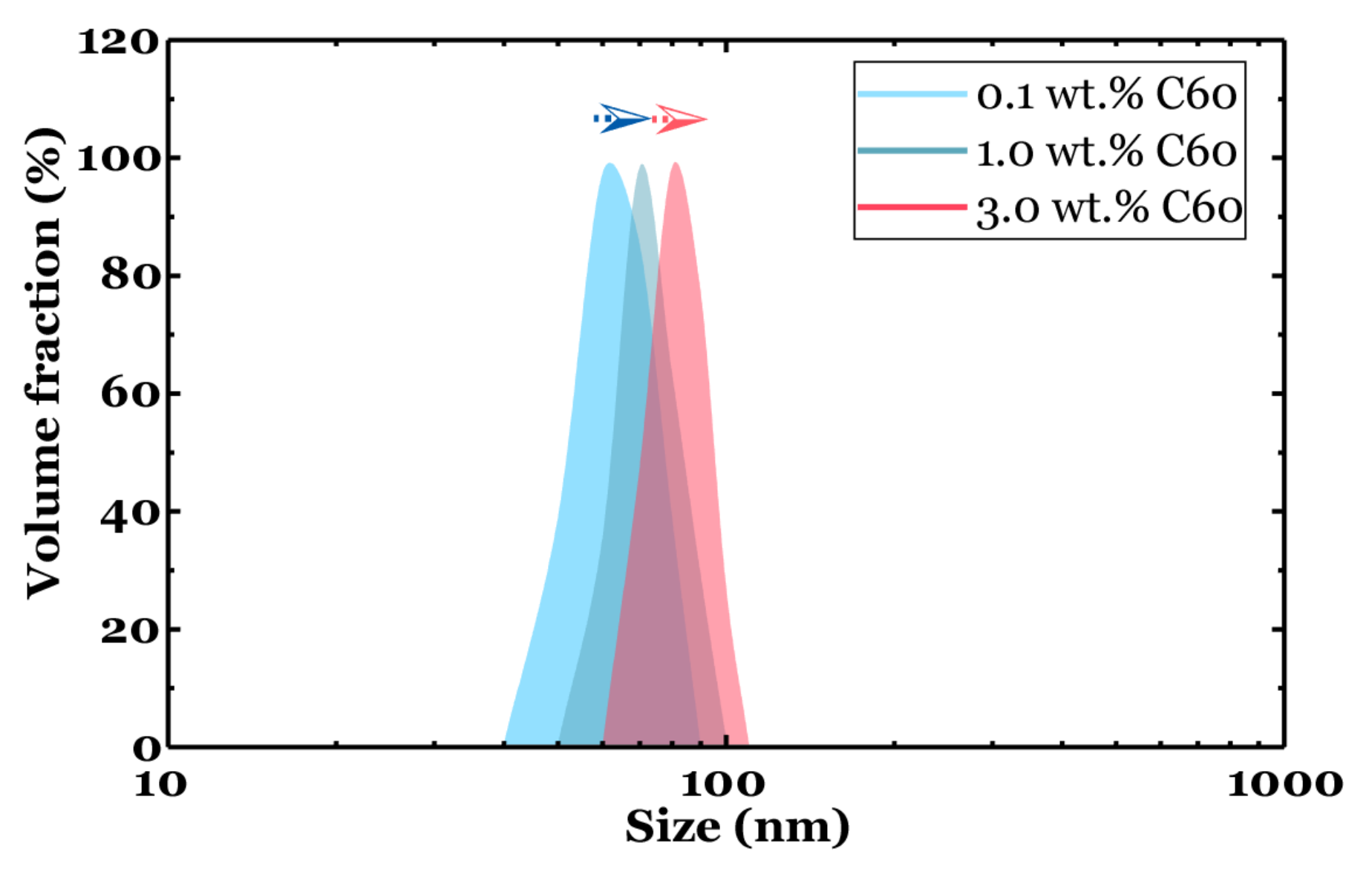
3.1. Particle Size Distribution and Dispersion
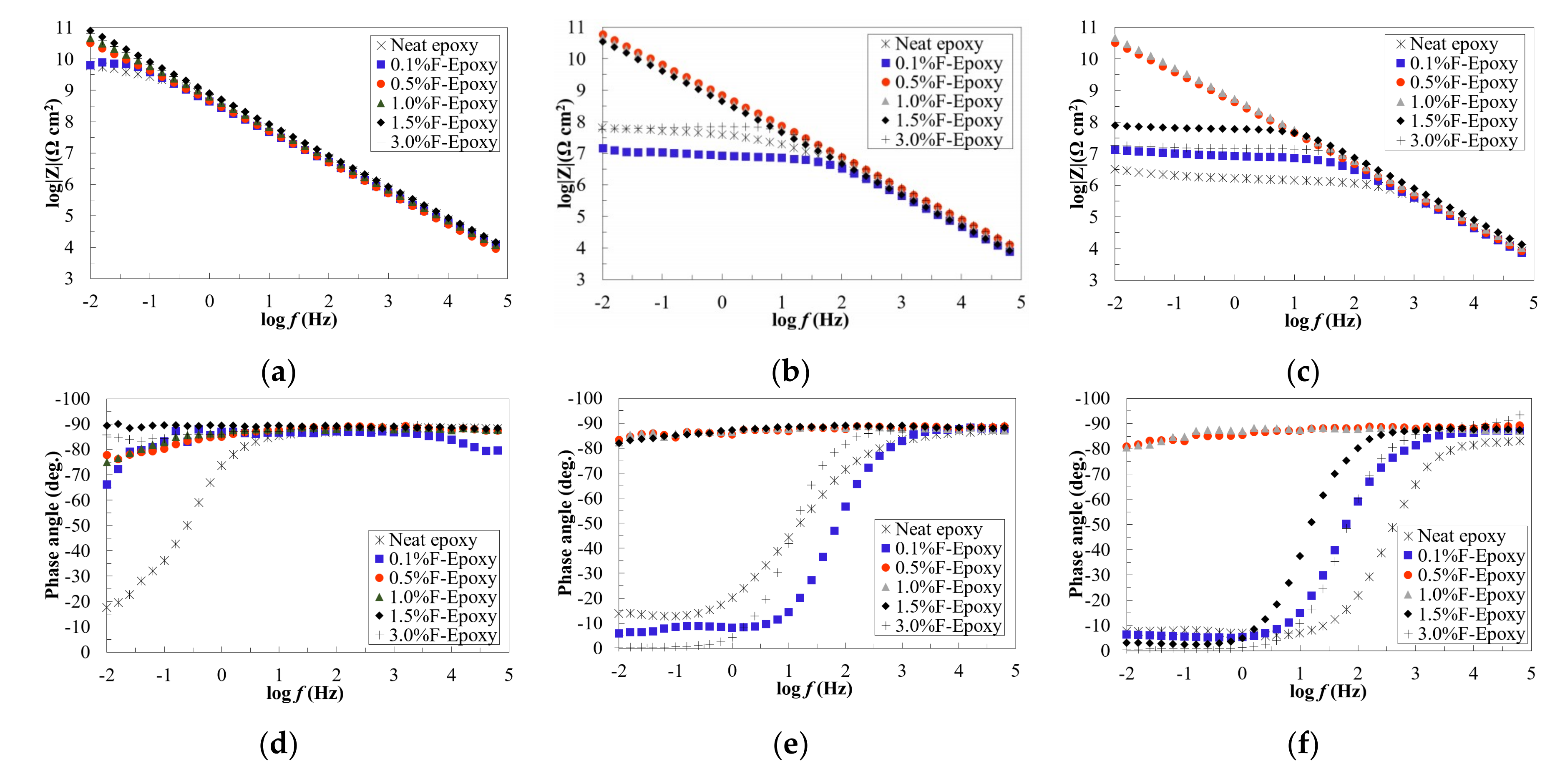
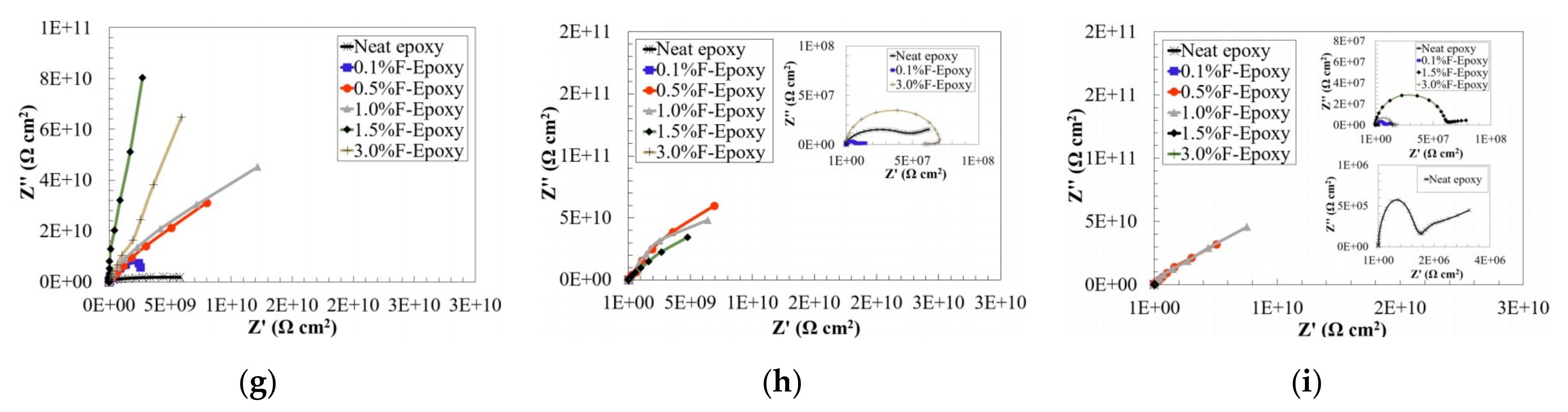
3.2. Barrier Performance of the New Composite Coatings
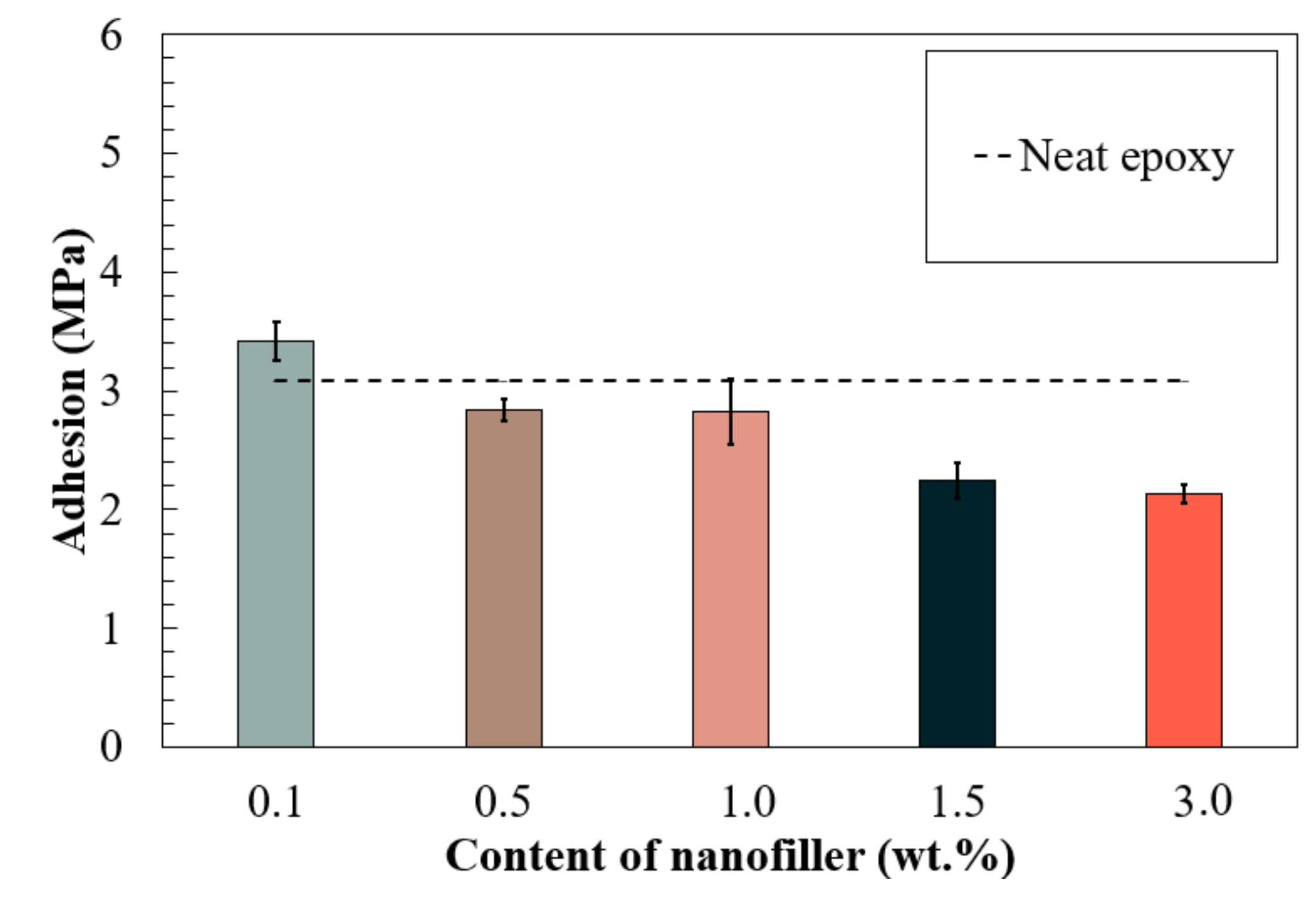
3.3. Adhesive Bond Strength of Nano-Reinforced Composites to the Substrate
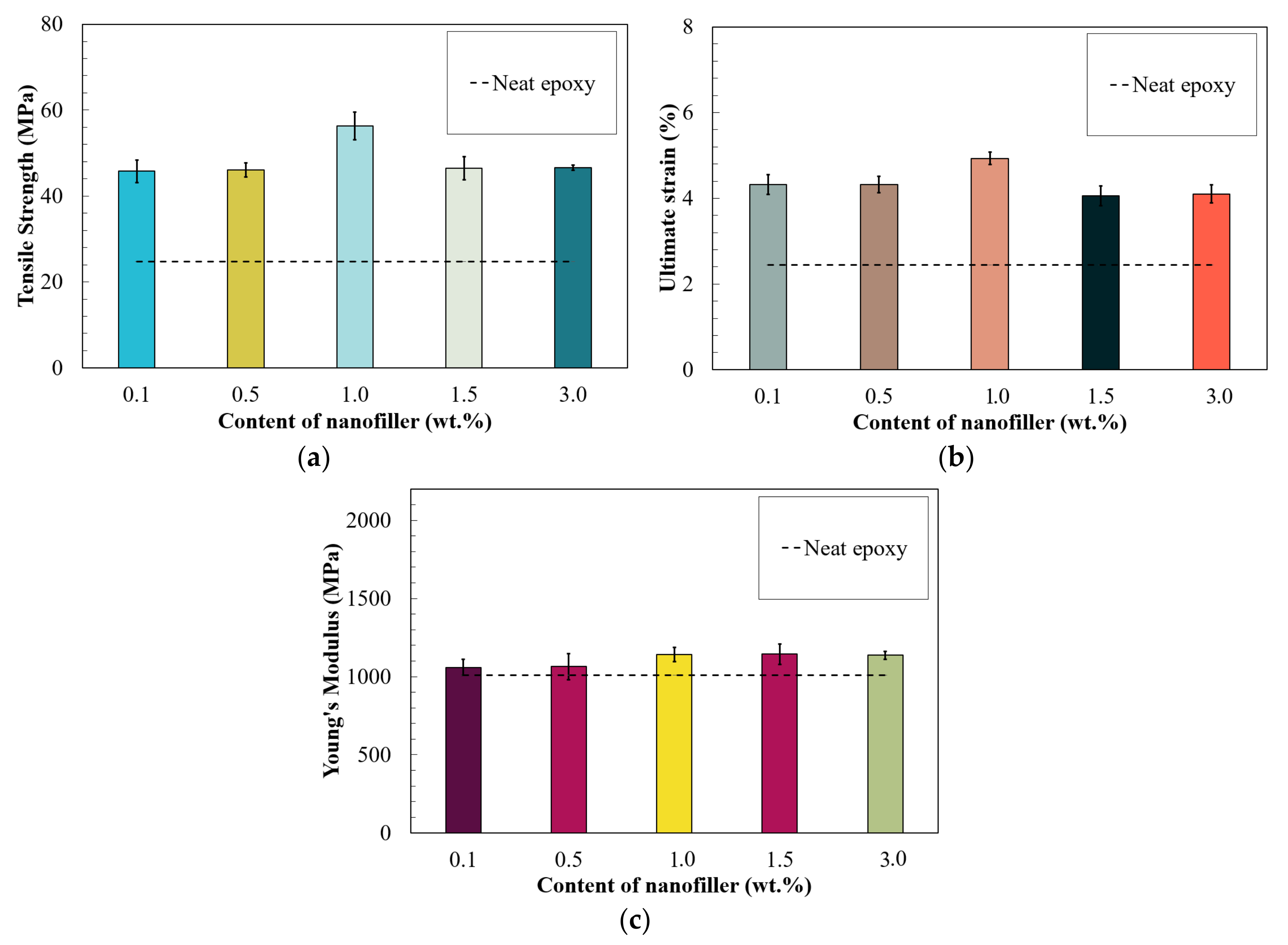
3.4. Tensile Behavior of the Nanocomposite Coating
4. Conclusions
- (a)
- The fullerene-C60/epoxy coatings exhibited improved electrochemical, mechanical properties with excellent durability, indicating the coatings enabled protection of the substrate against a harsh environment with corrosive media that oil/gas pipelines often experience.
- (b)
- Particle distribution results from DLS measurements revealed the developed dispersion method effectively overcome agglomeration, and no large particles were observed in all the tested samples.
- (c)
- The incorporation of fullerene-C60 as a coating additive led to dramatically improved corrosion resistance, as suggested by EIS results. Excellent barrier performance was observed in the samples with higher content fullerene-C60 particles (from 0.5 to 3.0 wt.%).
- (d)
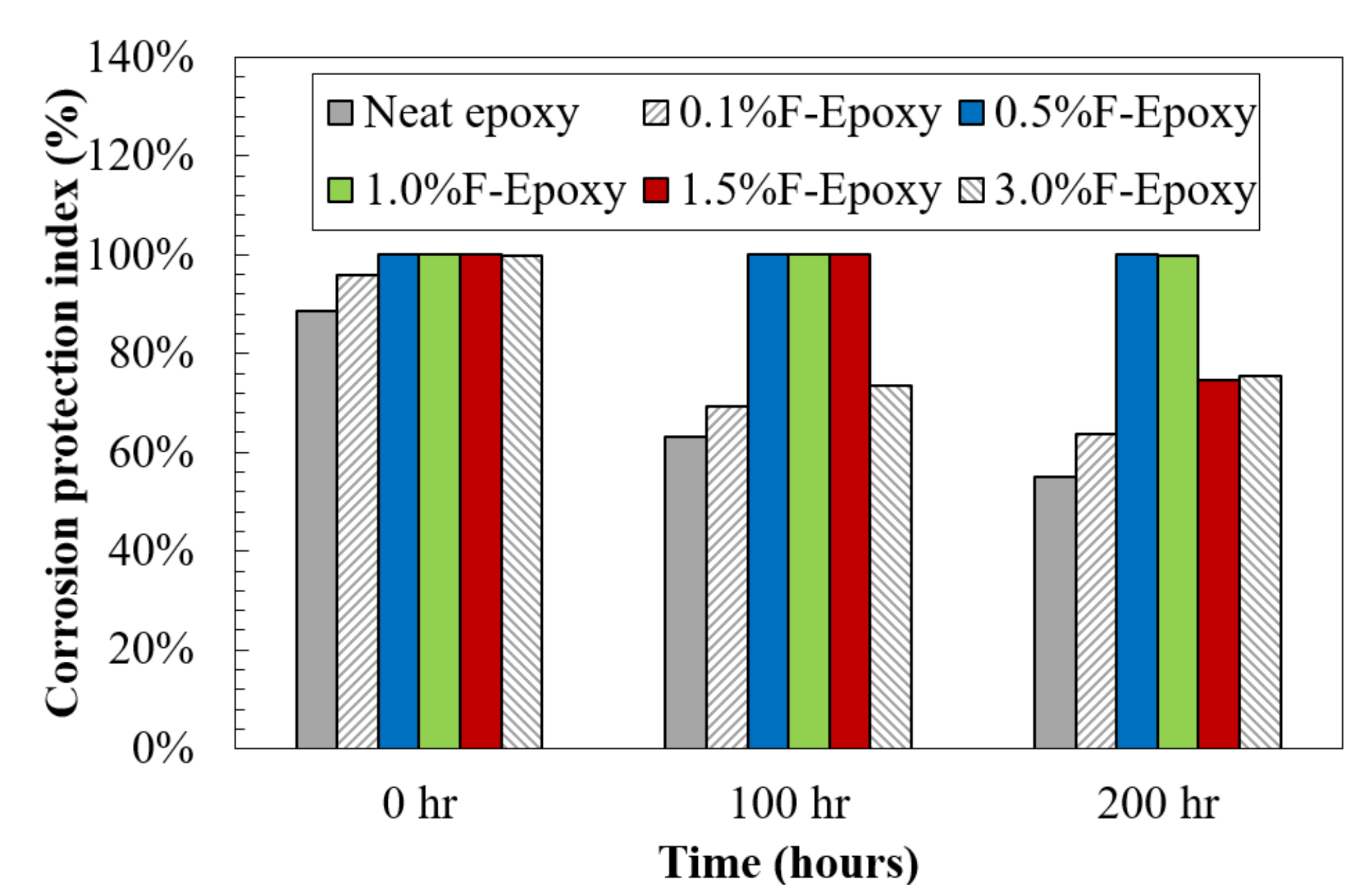
- EIS results after salt fog exposure confirmed that nanofiller coatings could provide a much longer life as compared with the neat epoxy. Particularly, as compared to an over 50% reduction in the control samples, the coatings with 0.5 and 1.0 wt.% of fullerene-C60 particles remained intact even after 200-h exposure to salt spray, as identified on their impedance values in Bode plots.
- (e)
- Enhancement in mechanical properties was observed in all the coatings with fullerene-C60 particles. The 1.0% F-Epoxy group exhibited the highest increase in tensile properties, including increased strength, strain, and Young’s modulus. In addition, improvement on adhesion was observed in the coating with low content of fullerene-C60 particles (0.1 wt.%).
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wang, X.; Qi, X.; Li, M.; Lin, Z.; Battocchi, D. Characterization of Graphene Reinforced Epoxy Coatings for Internal Surface of Oil and Gas Pipelines. In Proceedings of the ASCE Pipelines 2019 Conference, Nashville, TN, USA, 21–24 July 2019. [Google Scholar]
- Zi, Z.; Wang, X.; Pan, H.; Lin, Z. Corrosion-induced damage identification in metallic structures using machine learning approaches. In Proceedings of the 2019 Defense TechConnect Innovation Summit, National Harbor, MD, USA, 7–10 October 2019. [Google Scholar]
- Zi, Z.; Pan, H.; Lin, Z. Data-Driven Identification for Early-Age Corrosion-Induced Damage in Metallic Structures. In Proceedings of the Bridge Engineering Institute Conference 2019, Honolulu, HI, USA, 22–25 July 2019. [Google Scholar]
- Pan, H.; Azimi, M.; Yan, F.; Lin, Z. Time-frequency-based data-driven structural diagnosis and damage detection for cable-stayed bridges. J. Bridge Eng. 2018, 23, 04018033. [Google Scholar] [CrossRef]
- Sampath, S.; Bhattacharya, B.; Aryan, P.; Sohn, H. A Real-Time, Non-Contact Method for In-Line Inspection of Oil and Gas Pipelines Using Optical Sensor Array. Sensors 2019, 19, 3615. [Google Scholar] [CrossRef] [PubMed]
- Abbas, M.H.; Norman, R.; Charles, A. Neural network modelling of high pressure CO2 corrosion in pipeline steels. Process Saf. Environ. Prot. 2018, 119, 36–45. [Google Scholar] [CrossRef]
- Pan, H.; Gui, G.; Lin, Z.; Yan, C. Deep BBN Learning for Health Assessment toward Decision-Making on Structures under Uncertainties. KSCE J. Civ. Eng. 2018, 22, 928–940. [Google Scholar] [CrossRef]
- Lin, Z.; Pan, H.; Wang, X.; Li, M. Data-driven structural diagnosis and conditional assessment: From shallow to deep learning. In Proceedings of the International Society for Optics and Photonics, Denver, CO, USA, 27 March 2018; Volume 10598, p. 1059814. [Google Scholar]
- Gui, G.; Pan, H.; Lin, Z.; Li, Y.; Yuan, Z. Data-driven support vector machine with optimization techniques for structural health monitoring and damage detection. KSCE J. Civ. Eng. 2017, 21, 523–534. [Google Scholar] [CrossRef]
- Pan, H.; Ge, R.; Xingyu, W.; Jinhui, W.; Na, G.; Zhibin, L. Embedded Wireless Passive Sensor Networks for Health Monitoring of Welded Joints in Onshore Metallic Pipelines. In Proceedings of the ASCE 2017 Pipelines, Phoenix, AZ, USA, 6–9 August 2017. [Google Scholar]
- Wang, X.; Qi, X.; Pearson, M.; LI, M.; Lin, Z.; Battocchi, D. Design and Characterization of Functional Nanoengineered Epoxy-Resin Coatings for Pipeline Corrosion Control. In Proceedings of the Coating Trends and Technologies 2019, Rosemont, IL, USA, 10–11 September 2019. [Google Scholar]
- Varga, M.; Izak, T.; Vretenar, V.; Kozak, H.; Holovsky, J.; Artemenko, A.; Hulman, M.; Skakalova, V.; Lee, D.S.; Kromka, A. Diamond/carbon nanotube composites: Raman, FTIR and XPS spectroscopic studies. Carbon 2017, 111, 54–61. [Google Scholar] [CrossRef]
- Wang, X.; Tang, F.; Qi, X.; Lin, Z. Mechanical, electrochemical, and durability behavior of graphene nano-platelet loaded epoxy-resin composite coatings. Compos. Part B Eng. 2019, 107103. [Google Scholar] [CrossRef]
- Shadlou, S.; Alishahi, E.; Ayatollahi, M. Fracture behavior of epoxy nanocomposites reinforced with different carbon nano-reinforcements. Compos. Struct. 2013, 95, 577–581. [Google Scholar] [CrossRef]
- Kuilla, T.; Bhadra, S.; Yao, D.; Kim, N.H.; Bose, S.; Lee, J.H. Recent advances in graphene based polymer composites. Prog. Polym. Sci. 2010, 35, 1350–1375. [Google Scholar] [CrossRef]
- Roy, S.; Mitra, K.; Desai, C.; Petrova, R.; Mitra, S. Detonation nanodiamonds and carbon nanotubes as reinforcements in epoxy composites—A comparative study. J. Nanotechnol. Eng. Med. 2013, 4, 011008. [Google Scholar] [CrossRef]
- Liu, D.; Zhao, W.; Liu, S.; Cen, Q.; Xue, Q. Comparative tribological and corrosion resistance properties of epoxy composite coatings reinforced with functionalized fullerene C60 and graphene. Surf. Coat. Technol. 2016, 286, 354–364. [Google Scholar] [CrossRef]
- Bhattacharya, M. Polymer nanocomposites—A comparison between carbon nanotubes, graphene, and clay as nanofillers. Materials 2016, 9, 262. [Google Scholar] [CrossRef] [PubMed]
- Abbasi, H.; Antunes, M.; Velasco, J.I. Recent advances in carbon-based polymer nanocomposites for electromagnetic interference shielding. Prog. Mater. Sci. 2019, 103, 319–373. [Google Scholar] [CrossRef]
- Wang, X.; Qi, X.; Pearson, M.; Lin, Z.; Battocchi, D. NanoModified Protective Coatings for Pipeline Corrosion Control and Mitigation. In Proceedings of the 2019 TechConnect World Innovation Conference, Boston, MA, USA, 17–19 June 2019. [Google Scholar]
- Cui, L.-J.; Wang, Y.-B.; Xiu, W.-J.; Wang, W.-Y.; Xu, L.-H.; Xu, X.-B.; Meng, Y.; Li, L.-Y.; Gao, J.; Chen, L.-T.; et al. Effect of functionalization of multi-walled carbon nanotube on the curing behavior and mechanical property of multi-walled carbon nanotube/epoxy composites. Mater. Des. 2013, 49, 279–284. [Google Scholar] [CrossRef]
- Zabet, M.; Moradian, S.; Ranjbar, Z.; Zanganeh, N. Effect of carbon nanotubes on electrical and mechanical properties of multiwalled carbon nanotubes/epoxy coatings. J. Coat. Technol. Res. 2016, 13, 191–200. [Google Scholar] [CrossRef]
- Jiang, J.; Xu, C.; Su, Y.; Guo, Q.; Liu, F.; Deng, C.; Yao, X.; Zhou, L. Influence of carbon nanotube coatings on carbon fiber by ultrasonically assisted electrophoretic deposition on its composite interfacial property. Polymers 2016, 8, 302. [Google Scholar] [CrossRef]
- Shimamura, Y.; Oshima, K.; Tohgo, K.; Fujii, T.; Shirasu, K.; Yamamoto, G.; Hashida, T.; Goto, K.; Ogasawara, T.; Naito, K.; et al. Tensile mechanical properties of carbon nanotube/epoxy composite fabricated by pultrusion of carbon nanotube spun yarn preform. Compos. Part A Appl. Sci. Manuf. 2014, 62, 32–38. [Google Scholar] [CrossRef]
- Wang, X.; Qi, X.; Pearson, M.; Li, M.; Lin, Z.; Battocchi, D. Characterization of Nano-Particle Reinforced Epoxy Coatings for Structural Corrosion Mitigation. In Proceedings of the Bridge Engineering Institute Conference 2019, Honolulu, HI, USA, 22–25 July 2019. [Google Scholar]
- Yu, Y.-H.; Lin, Y.-Y.; Lin, C.-H.; Chan, C.-C.; Huang, Y.-C. High-performance polystyrene/graphene-based nanocomposites with excellent anti-corrosion properties. Polym. Chem. 2014, 5, 535–550. [Google Scholar] [CrossRef]
- Chang, C.-H.; Huang, T.-C.; Peng, C.-W.; Yeh, T.-C.; Lu, H.-I.; Hung, W.-I.; Weng, C.-J.; Yang, T.-I.; Yeh, J.-M. Novel anticorrosion coatings prepared from polyaniline/graphene composites. Carbon 2012, 50, 5044–5051. [Google Scholar] [CrossRef]
- Pikhurov, D.V.; Zuev, V.V. The effect of fullerene C60 on the dielectric behaviour of epoxy resin at low nanofiller loading. Chem. Phys. Lett. 2014, 601, 13–15. [Google Scholar] [CrossRef]
- Zuev, V.V. The mechanisms and mechanics of the toughening of epoxy polymers modified with fullerene C60. Polym. Eng. Sci. 2012, 52, 2518–2522. [Google Scholar] [CrossRef]
- Bai, J.; He, Q.; Shi, Z.; Tian, M.; Xu, H.; Ma, X.; Yin, J. Self-assembled elastomer nanocomposites utilizing C60 and poly (styrene-b-butadiene-b-styrene) via thermally reversible Diels-Alder reaction with self-healing and remolding abilities. Polymer 2017, 116, 268–277. [Google Scholar] [CrossRef]
- Ogasawara, T.; Ishida, Y.; Kasai, T. Mechanical properties of carbon fiber/fullerene-dispersed epoxy composites. Compos. Sci. Technol. 2009, 69, 2002–2007. [Google Scholar] [CrossRef]
- Li, P.; He, X.; Huang, T.-C.; White, K.L.; Zhang, X.; Liang, H.; Nishimura, R.; Sue, H.-J. Highly effective anti-corrosion epoxy spray coatings containing self-assembled clay in smectic order. J. Mater. Chem. A 2015, 3, 2669–2676. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhao, M.; Zhang, J.; Shao, Q.; Li, J.; Li, H.; Lin, B.; Yu, M.; Chen, S.; Guo, Z. Excellent corrosion protection performance of epoxy composite coatings filled with silane functionalized silicon nitride. J. Polym. Res. 2018, 25, 130. [Google Scholar] [CrossRef]
- Chhetri, S.; Samanta, P.; Murmu, N.C.; Kuila, T. Anticorrosion Properties of Epoxy Composite Coating Reinforced by Molybdate-Intercalated Functionalized Layered Double Hydroxide. J. Compos. Sci. 2019, 3, 11. [Google Scholar] [CrossRef]
- Yang, F.; Liu, T.; Li, J.; Zhao, H. Long Term Corrosion Protection of Epoxy Coating Containing Tetraaniline Nanofiber. Int. J. Electrochem. Sci. 2018, 13, 6843–6857. [Google Scholar] [CrossRef]
- Wang, X.; Qi, X.; Lin, Z.; Wang, J.; Gong, N. Electrochemical Characterization of the Soils Surrounding Buried or Embedded Steel Elements. In Proceedings of the ASCE 2016 Pipelines, Kansas City, MO, USA, 17–20 August 2016. [Google Scholar]
- Ammar, S.; Ramesh, K.; Ma, I.; Farah, Z.; Vengadaesvaran, B.; Ramesh, S.; Arof, A.K. Studies on SiO2-hybrid polymeric nanocomposite coatings with superior corrosion protection and hydrophobicity. Surf. Coat. Technol. 2017, 324, 536–545. [Google Scholar] [CrossRef]
- Ramezanzadeh, B.; Niroumandrad, S.; Ahmadi, A.; Mahdavian, M.; Moghadam, M.M. Enhancement of barrier and corrosion protection performance of an epoxy coating through wet transfer of amino functionalized graphene oxide. Corros. Sci. 2016, 103, 283–304. [Google Scholar] [CrossRef]
- Wang, X.; Qi, X.; Lin, Z.; Battocchi, D. Graphene Reinforced Composites as Protective Coatings for Oil and Gas Pipelines. Nanomaterials 2018, 8, 1005. [Google Scholar] [CrossRef]
- Ammar, S.; Ramesh, K.; Vengadaesvaran, B.; Ramesh, S.; Arof, A.K. A novel coating material that uses nano-sized SiO2 particles to intensify hydrophobicity and corrosion protection properties. Electrochim. Acta 2016, 220, 417–426. [Google Scholar] [CrossRef]
- Ramezanzadeh, B.; Haeri, Z.; Ramezanzadeh, M. A facile route of making silica nanoparticles-covered graphene oxide nanohybrids (SiO2-GO); fabrication of SiO2-GO/epoxy composite coating with superior barrier and corrosion protection performance. Chem. Eng. J. 2016, 303, 511–528. [Google Scholar] [CrossRef]
- Zhai, L.; Ling, G.; Wang, Y. Effect of nano-Al2O3 on adhesion strength of epoxy adhesive and steel. Int. J. Adhes. Adhes. 2008, 28, 23–28. [Google Scholar] [CrossRef]
- May, M.; Wang, H.; Akid, R. Effects of the addition of inorganic nanoparticles on the adhesive strength of a hybrid sol–gel epoxy system. Int. J. Adhes. Adhes. 2010, 30, 505–512. [Google Scholar] [CrossRef]



| Label | Content of C60 (wt.%) | Exposure to Accelerated Environmental Stresses | |||||
|---|---|---|---|---|---|---|---|
| Onset | 100-h | 200-h | |||||
| Electrical Circuit Models (EEC) | |Zmod|0.01HZ | EEC | |Zmod|0.01HZ | EEC | |Zmod|0.01HZ | ||
| Neat epoxy | / | Model B with W | 6.10 × 109 | Model B with W | 6.46 × 107 | Model B with W | 3.29 × 106 |
| 0.1%F-Epoxy | 0.1 | Model B with W | 6.39 × 109 | Model B with W | 1.45 × 107 | Model B with W | 1.31 × 107 |
| 0.5%F-Epoxy | 0.5 | Model A | 3.21 × 1010 | Model A | 6.03 × 1010 | Model A | 3.22 × 1010 |
| 1.0%F-Epoxy | 1.0 | Model A | 4.69 × 1010 | Model A | 4.87 × 1010 | Model A | 4.64 × 1010 |
| 1.5%F-Epoxy | 1.5 | Model A | 8.04 × 1010 | Model A | 3.47 × 1010 | Model B | 7.86 × 107 |
| 3.0%F-Epoxy | 3.0 | Model A | 6.51 × 1010 | Model B | 5.85 × 107 | Model B | 1.83 × 107 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, X.; Tang, F.; Qi, X.; Lin, Z.; Battocchi, D.; Chen, X. Enhanced Protective Coatings Based on Nanoparticle fullerene C60 for Oil & Gas Pipeline Corrosion Mitigation. Nanomaterials 2019, 9, 1476. https://doi.org/10.3390/nano9101476
Wang X, Tang F, Qi X, Lin Z, Battocchi D, Chen X. Enhanced Protective Coatings Based on Nanoparticle fullerene C60 for Oil & Gas Pipeline Corrosion Mitigation. Nanomaterials. 2019; 9(10):1476. https://doi.org/10.3390/nano9101476
Chicago/Turabian StyleWang, Xingyu, Fujian Tang, Xiaoning Qi, Zhibin Lin, Dante Battocchi, and Xi Chen. 2019. "Enhanced Protective Coatings Based on Nanoparticle fullerene C60 for Oil & Gas Pipeline Corrosion Mitigation" Nanomaterials 9, no. 10: 1476. https://doi.org/10.3390/nano9101476
APA StyleWang, X., Tang, F., Qi, X., Lin, Z., Battocchi, D., & Chen, X. (2019). Enhanced Protective Coatings Based on Nanoparticle fullerene C60 for Oil & Gas Pipeline Corrosion Mitigation. Nanomaterials, 9(10), 1476. https://doi.org/10.3390/nano9101476








