Continuously Improved Photocatalytic Performance of Zn2SnO4/SnO2/Cu2O Composites by Structural Modulation and Band Alignment Modification
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Synthesis of Pristine Zn2SnO4/SnO2 Hierarchical Architectures (P-ZSO/SnO2)
2.3. Synthesis of Etching Zn2SnO4/SnO2 Hierarchical Architectures (ET-ZSO/SnO2)
2.4. Synthesis of Zn2SnO4/SnO2/Cu2O Hierarchical Architectures
2.5. Characterizations
2.6. Photocatalytic Experiments
2.7. Photocurrent Measurements
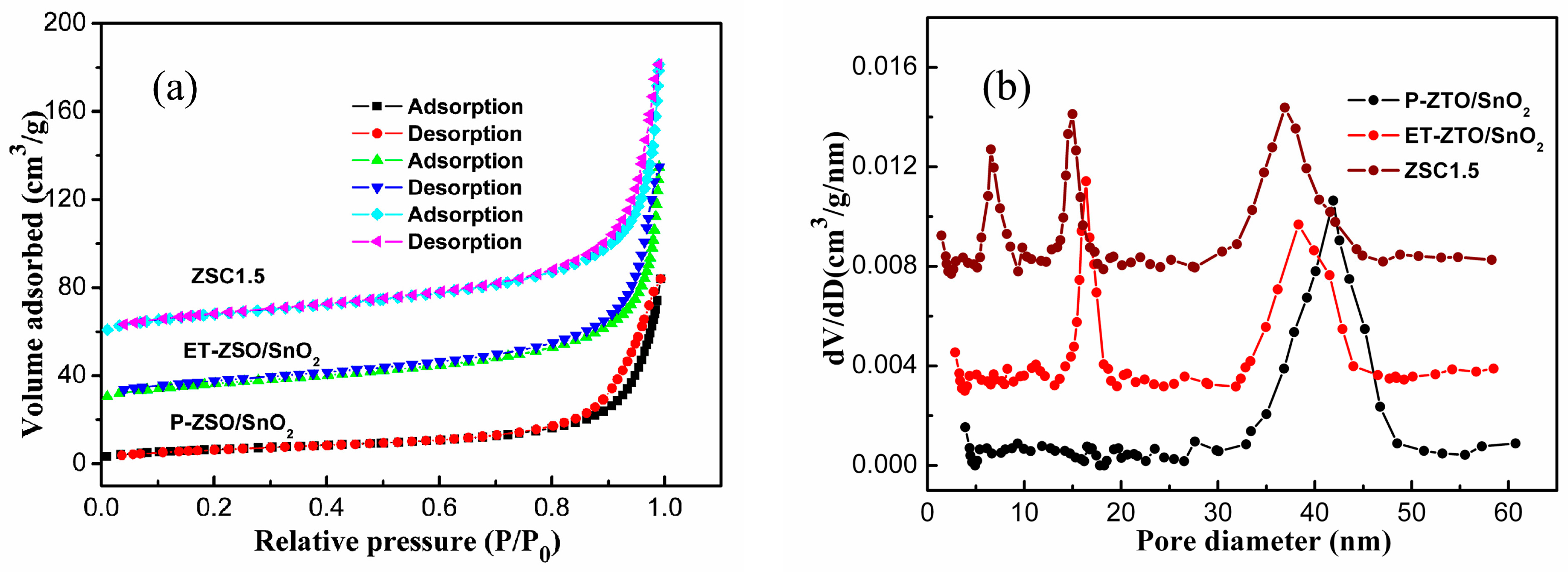
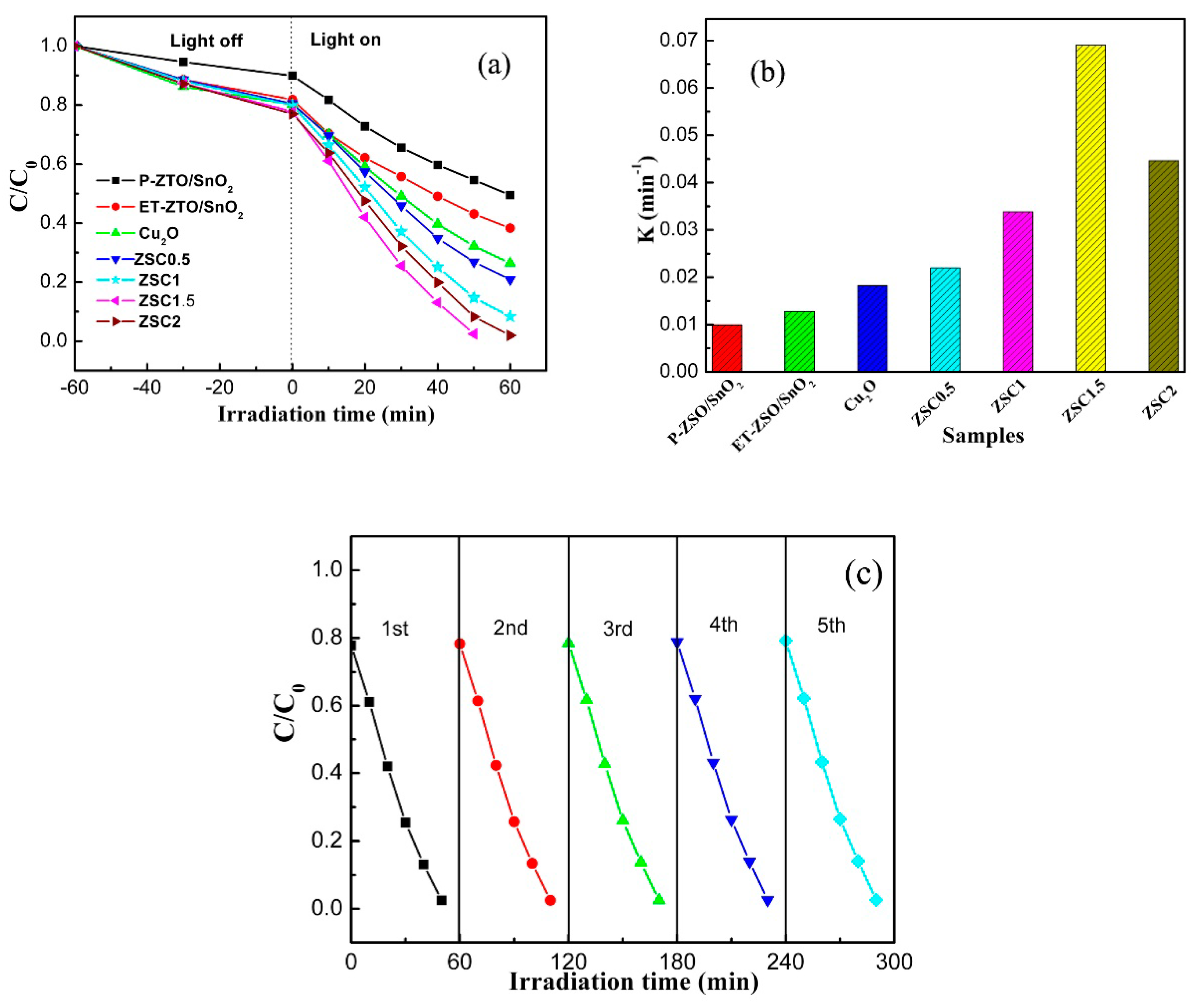
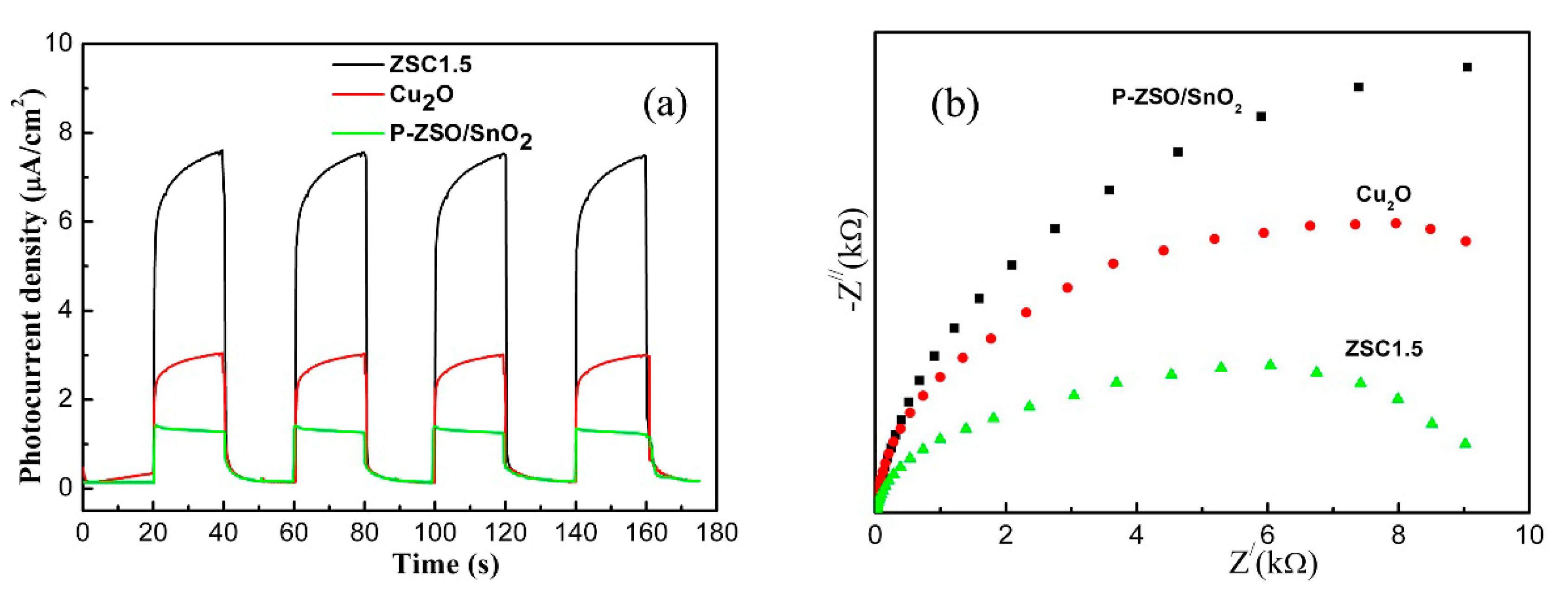
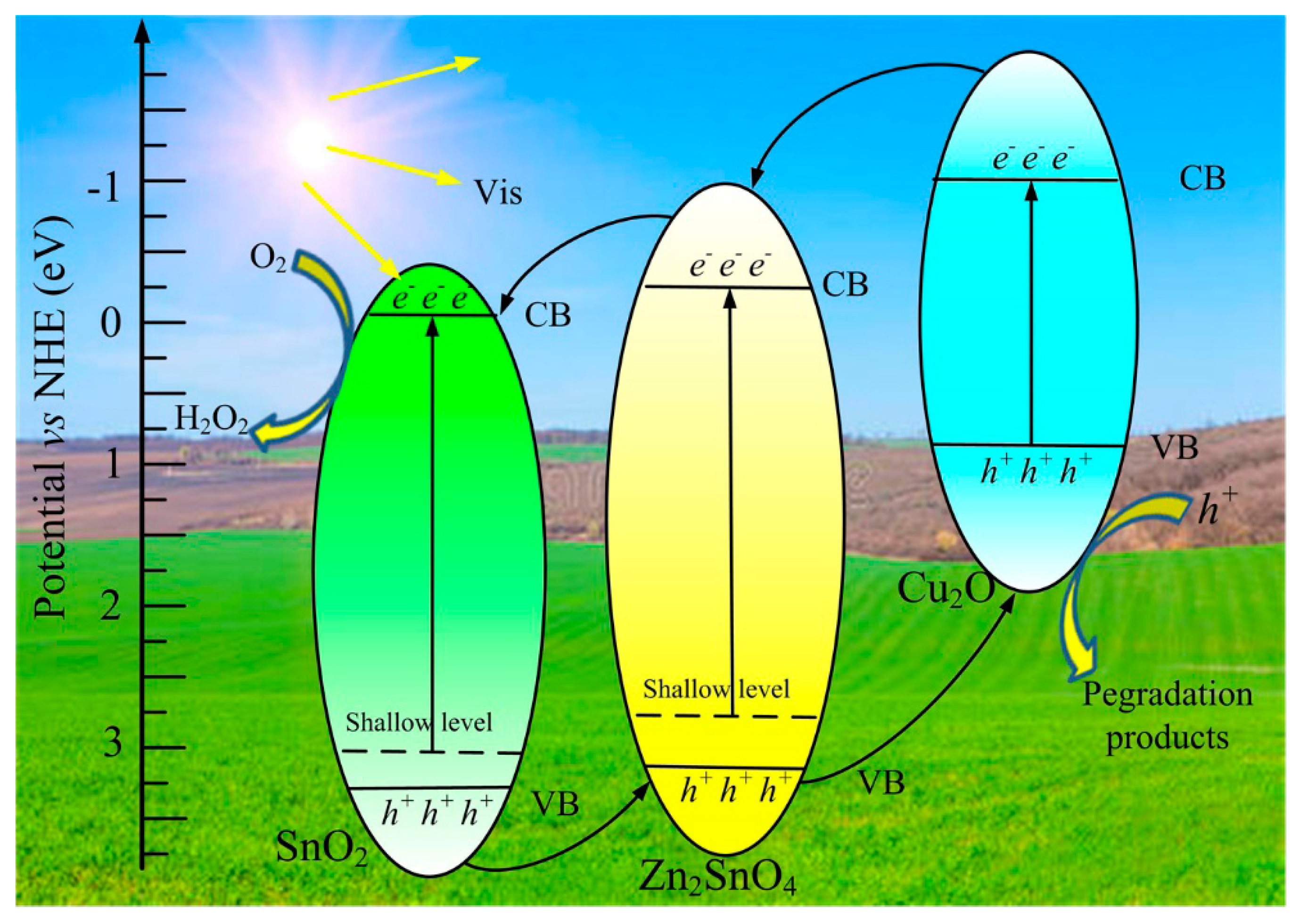
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Tan, B.; Toman, E.; Li, Y.G.; Wu, Y.Y. Zinc stannate (Zn2SnO4) dye-sensitized solar cells. J. Am. Chem. Soc. 2007, 129, 4162–4163. [Google Scholar] [CrossRef] [PubMed]
- Sumithra, S.; VictorJaya, N. Band gap tuning and room temperature ferromagnetism in Co doped Zinc stannate nanostructures. Physica B 2016, 493, 35–42. [Google Scholar] [CrossRef]
- Lou, X.; Jia, X.; Xu, J.; Liu, S.; Cao, Q. Hydrothermal synthesis, characterization and photocatalytic properties of Zn2SnO4 nanocrystal. Mater. Sci. Eng. A 2006, 432, 221–225. [Google Scholar] [CrossRef]
- Zhao, Q.Q.; Deng, X.L.; Ding, M.; Huang, J.Z.; Ju, D.X.; Xu, X.J. Synthesis of hollow cubic Zn2SnO4 sub-microstructures with enhanced photocatalytic performance. J. Alloys Compd. 2016, 671, 328–333. [Google Scholar] [CrossRef]
- Li, Z.; Zhou, Y.; Zhang, J.; Tu, W.; Liu, Q.; Yu, T.; Zou, Z. Hexagonal nanoplate-textured micro-octahedron Zn2SnO4: Combined effects toward enhanced efficiencies of dye-sensitized solar cell and photoreduction of CO2 into hydrocarbon fuels. Cryst. Growth Des. 2012, 12, 1476–1481. [Google Scholar] [CrossRef]
- Liu, C.; Röder, R.; Zhang, L.; Ren, Z.; Chen, H.; Zhang, Z.; Ronning, C.; Gao, P.X. Highly efficient visible-light driven photocatalysts: A case of zinc stannate based nanocrystal assemblies. J. Mater. Chem. A 2014, 2, 4157–4167. [Google Scholar] [CrossRef]
- Wang, J.; Li, H.; Meng, S.; Zhang, L.; Fu, X.; Chen, S. One-pot hydrothermal synthesis of highly efficient SnOx-Zn2SnO4 composite photocatalyst for the degradation of methyl orange and gaseous benzene. Appl. Catal. B Environ. 2017, 200, 10–19. [Google Scholar] [CrossRef]
- Junploy, P.; Phuruangrat, A.; Plubphon, N.; Thongtem, S.; Thongtem, T. Photocatalytic degradation of methylene blue by Zn2SnO4-SnO2 system under UV visible radiation. Mat. Sci. Semicon. Proc. 2017, 66, 56–61. [Google Scholar] [CrossRef]
- Jia, T.K.; Zhao, J.W.; Fu, F.; Deng, Z.; Wang, W.M.; Fu, Z.Y.; Meng, F.C. Synthesis, characterization and photcatalytic activity Zn-doped SnO2/Zn2SnO4 coupled nanocomposites. Int. J. Photoenergy 2014, 2014, 7. [Google Scholar] [CrossRef]
- Jia, T.K.; Fu, F.; Long, F.; Min, Z.Y.; Zhao, J.W.; Chen, J.; Li, J.L. Synthesis, characterization and enhanced visible-light photocatalytic activity of Zn2SnO4/C nanocomposites with truncated octahedron morphology. Ceram. Int. 2016, 42, 3808–3815. [Google Scholar] [CrossRef]
- Jia, T.K.; Liu, M.; Yu, D.Y.; Long, F.; Mo, S.; Deng, Z.; Wang, W. A facile approach for the synthesis of Zn2SnO4/BiOBr nanocomposites with improved visible light photocatalytic performance. Nanomaterials 2018, 8, 313. [Google Scholar] [CrossRef] [PubMed]
- Jia, T.K.; Fu, F.; Li, J.; Wang, W.; Hu, X. Constructing a novel Zn2SnO4/C/AgBr nanocomposite with extended spectral Response and improved photocatalytic performance. J. Alloys Compd. 2019, 783, 687–696. [Google Scholar] [CrossRef]
- Sun, L.Q.; Han, X.; Jiang, Z.; Ye, Y.T.; Li, R.; Zhao, X.S.; Han, X.G. Fabrication of cubic Zn2SnO4/SnO2 complex hollow structures and their sunlight-driven photocatalytic activity. Nanoscale 2016, 8, 12858–12862. [Google Scholar] [CrossRef] [PubMed]
- Sun, S.Q.; Li, S.; Su, Y.; He, D.; Zhang, Z.T. Surface-disorder-engineered Zn2SnO4/SnO2 hollow microboxes with enhanced solar-driven photocatalytic activity. Appl. Surf. Sci. 2019, 463, 474–480. [Google Scholar] [CrossRef]
- Li, Y.; Wu, X.; Ho, W.; Lv, K.; Li, Q.; Li, M.; Lee, S.C. Graphene-induced formation of visible- light-responsive SnO2-Zn2SnO4 Z scheme photocatalyst with surface vacancy for the enhanced photoreactivity towards NO and acetone oxidation. Chem. Eng. J. 2018, 336, 200–210. [Google Scholar] [CrossRef]
- Ma, T.Y.; Cao, J.L.; Jaronies, M.; Qiao, S.Z. Interacting carbon nitride and titanium carbide nanosheets for high-performance oxygen evolution. Angew. Chem. Int. Edit. 2016, 55, 1138–1142. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Li, X.; Sun, H.; Sun, P.; Liang, X.; Liu, F.; Hu, X.; Lu, G.Y. Enhanced gas sensing properties of monodisperse Zn2SnO4 octahedron functionalized by PdO nanoparticles. Sensor. Actuat. B 2018, 266, 302–310. [Google Scholar]
- Paracchino, A.; Laporte, V.; Sivula, K.; Gratzel, M.; Thimsen, E. Highly active oxide photocathode for photoelectrochemical water reduction. Nat. Mater. 2011, 10, 456–461. [Google Scholar] [CrossRef]
- Hu, C.C.; Nian, J.N.; Teng, H. Electrodeposited p-type Cu2O as photocatalyst for H2 evolution from water reduction in the presence of WO3. Sol. Energy Mater. Sol. Cells 2008, 92, 1071–1076. [Google Scholar] [CrossRef]
- Zhang, Z.; Wang, P. Highly stable copper oxide composite as an effective photocathode for water splitting via a facile electrochemical synthesis strategy. J. Mater. Chem. 2012, 22, 2456–2464. [Google Scholar] [CrossRef]
- Siol, S.; Hellmann, J.C.; Tilley, D.S.; Graetzel, M.; Morasch, J.; Deuermeirer, J.; Jaegermann, W.; Klein, A. Band alignment engineering at Cu2O/ZnO heterointerfaces. ACS Appl. Mater. Interfaces 2016, 8, 21824–21831. [Google Scholar] [CrossRef] [PubMed]
- Jongh, P.E.; Vanmaekelbergh, D.; Kelly, J.J. Photoelectrochemistry of electrodeposited Cu2O. J. Electrochem. Soc. 2000, 147, 486–489. [Google Scholar] [CrossRef]
- Wang, Y.; Li, S.; Shi, H.; Yu, K. Facile synthesis of p-type Cu2O/n-type ZnO nanoheterojunctions with novel photoluminescence properties enhanced field emission and photocatalytic activities. Nanoscale 2012, 4, 7817–7824. [Google Scholar] [CrossRef]
- Yuan, W.; Yuan, J.; Xie, J.; Li, C.M. Polymer-mediated self-assembly of TiO2@Cu2O core-shell nanowire array for highly efficient photoelectrochemical water oxidation. ACS Appl. Mater. Interfaces 2016, 8, 6082–6092. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.Y.; Sun, L.; Lin, Z.Q.; Cai, J.H.; Xie, K.P.; Lin, C.J. P-N heterojunction photoelectrodes composed of Cu2O-loaded TiO2 nanotube arrays with enhanced photoelectrochemical and photoelectrocatalytic activities. Energy Environ. Sci. 2013, 6, 1211–1220. [Google Scholar] [CrossRef]
- Cheng, W.Y.; Yu, T.H.; Chao, K.J.; Lu, S.Y. Cu2O-decorated CdS nanostructures for high efficiency visible light driven hydrogen production. Int. J. Hydrogen Energy 2013, 38, 9665–9672. [Google Scholar] [CrossRef]
- Tian, L.; Rui, Y.; Sun, K.; Cui, W.; An, W.J. Surface decoration of ZnWO4 nanorods with Cu2O nanoparticles to build heterostructure with enhanced photocatalysis. Nanomaterials 2018, 8, 33. [Google Scholar] [CrossRef]
- Zhang, J.; Ma, H.; Liu, Z. Highly efficient photocatalyst based on all oxides WO3/Cu2O heterojunction for photoelectrochemical water splitting. Appl. Catal. B 2017, 201, 84–91. [Google Scholar] [CrossRef]
- Wang, W.Z.; Huang, X.W.; Wu, S.; Zhou, Y.X.; Wang, L.J.; Shi, H.L.; Liang, Y.J.; Zou, B. Preparation of p-n junction Cu2O/BiVO4 heterogeneous nanostructures with enhanced visible-light photocatalytic activity. Appl. Catal. B 2013, 134–135, 293–301. [Google Scholar] [CrossRef]
- Cui, W.; An, W.; Liu, L.; Hu, J.; Liang, Y. Novel Cu2O quantum dots coupled flower-like BiOBr for enhanced photocatalytic degradation of organic contaminant. J. Hazard. Mater. 2014, 280, 417–427. [Google Scholar] [CrossRef]
- Jia, T.K.; Fu, F.; Yu, D.; Cao, J.; Sun, G. Facile synthesis and characterization of N-doped TiO2/C nanocomposites with enhanced visible-light photocatalytic performance. Appl. Surf. Sci. 2018, 430, 438–447. [Google Scholar] [CrossRef]
- Jia, T.K.; Fu, F.; Li, J.L.; Deng, D.; Long, F.; Yu, D.; Cui, Q.; Wang, W.M. Rational construction of direct Z-scheme SnS-g-C3N4 hybrid photocatalyst for significant enhancement of visible-light photocatalytic activity. Appl. Surf. Sci. 2019, 499, 143941. [Google Scholar] [CrossRef]
- Wang, W.; Zhu, H.G.; Li, B. Synergy of Ti-O-based heterojunction and hierarchical 1D nanobelt/3D microflower heteroarchitectures for enhanced photocatalytic tetracycline degradation and photoelectrochemical water splitting. Chem. Eng. J. 2019, 378, 122072. [Google Scholar] [CrossRef]
- Jia, T.K.; Wang, W.M.; Long, F.; Fu, Z.Y.; Wang, H.; Zhang, Q.J. Synthesis, characterization, and photocatalytic activity of Zn-doped SnO2 hierarchical architectures assembled by nanocones. J. Phys. Chem. C 2009, 113, 9071–9077. [Google Scholar] [CrossRef]
- Lu, C.; Wang, J.; Xu, F.; Wang, A.Q.; Meng, D.W. Zn-doped SnO2 hierarchical structures formed by a hydrothermal route with remarkably enhanced photocatalytic performance. Ceram. Int. 2018, 44, 15145–15152. [Google Scholar] [CrossRef]
- Zhou, X.; Li, X.W.; Sun, H.B.; Sun, P.; Liang, X.S.; Liu, F.M.; Hu, X.L.; Lu, G.Y. Nanosheet-assembled ZnFe2O4 hollow microspheres for high-sensitive acetone sensor. ACS Appl. Mater. Interfaces 2015, 7, 15414–15421. [Google Scholar] [CrossRef] [PubMed]
- Dupin, J.C.; Gonbeau, D.; Vinatier, P.; Levasseur, A. Systematic XPS studies of metal oxides, hydroxides and peroxides. Phys. Chem. Chem. Phys. 2000, 2, 1319–1324. [Google Scholar] [CrossRef]
- Li, F.T.; Wang, Q.; Ran, J.R.; Hao, Y.J.; Wang, X.J.; Zhao, D.S.; Qiao, S.Z. Ionic liquid self-combustion synthesis of BiOBr/Bi24O31Br10 heterojunctions with exceptional visible-light photocatalytic performances. Nanoscale 2015, 7, 1116–1126. [Google Scholar] [CrossRef]
- Lin, H.F.; Liao, S.C.; Hung, W.W.; Hu, C.T. Thermal plasma synthesis and optical properties of Zn2SnO4 nanopowders. Mater. Chem. Phys. 2009, 117, 9–13. [Google Scholar] [CrossRef]
- Pang, C.; Yan, B.; Liao, L.; Liu, B.; Zheng, Z.; Wu, T.; Yu, T. Synthesis characterization and opto-electrical properties of ternary Zn2SnO4 nanowires. Nanotechnology 2010, 21, 465706. [Google Scholar] [CrossRef]
- Zhu, A.; Zhao, Q.; Li, X.; Shi, Y. BiFeO3/TiO2 nanotube arrays composite electrode: Construction characterization and enhanced photoelectrochemical properties. ACS Appl. Mater. Interfaces 2014, 6, 671–679. [Google Scholar] [CrossRef] [PubMed]
- Huo, Y.; Yang, Y.; Dai, K.; Zhang, J.F. Construction of 2D/2D porous graphitic C3N4/SnS2 composite as a direct Z-scheme system for efficient visible photocatalytic activity. Appl. Surf. Sci. 2019, 481, 1260–1269. [Google Scholar] [CrossRef]
- Zhang, J.F.; Fu, J.W.; Wang, Z.L.; Cheng, B.; Dai, K.; Ho, W.K. Direct Z-scheme porous g-C3N4/BiOI heterojunction for enhanced visible-light photocatalytic activity. J. Alloys Compd. 2018, 766, 841–850. [Google Scholar] [CrossRef]
- Wang, Z.L.; Lv, J.L.; Zhang, J.F.; Dai, K.; Liang, C.H. Facile synthesis of Z-scheme BiVO4/porous graphite carbon nitride heterojunction for enhanced visible-light-driven photocatalyst. Appl. Surf. Sci. 2018, 430, 595–602. [Google Scholar] [CrossRef]
- Jiang, W.; Zong, X.; An, L.; Hua, S.; Miao, X.; Luan, S.; Wen, Y.; Tao, F.F.; Sun, Z. Consciously constructing heterojunction or direct Z-Scheme photocatalysts by regulating electron flow direction. ACS Catal. 2018, 8, 2209–2217. [Google Scholar] [CrossRef]
- Zhu, B.; Xia, P.; Ho, W.; Yu, J. Isoelectric point and adsorption activity of porous g-C3N4. Appl. Surf. Sci. 2015, 344, 188–195. [Google Scholar] [CrossRef]
- Li, S.T.; Shi, S.R.; Huang, G.C.; Xiong, Y.; Liu, S.W. Synergetic tuning charge dynamics and potentials of Ag3PO4 photocatalysts with boosting activity and stability by facile in-situ fluorination. Appl. Surf. Sci. 2018, 455, 1137–1149. [Google Scholar] [CrossRef]
- Lee, J.; Kang, Y.; Hwang, C.S.; Han, S.; Lee, S.C.; Cho, J.H. Effect of oxygen vacancy on the structural and electronic characteristics of crystalline Zn2SnO4. J. Mater. Chem. C 2014, 2, 8381–8387. [Google Scholar] [CrossRef]
- Dong, F.; Li, Q.; Sun, Y.; Ho, W.K. Noble metal-like behavior of plasmonic Bi particles as a cocatalyst deposited on (BiO)2CO3 microspheres for efficient visible light photocatalysis. ACS Catal. 2014, 4, 4341–4350. [Google Scholar] [CrossRef]
- Di, J.; Xia, J.; Ji, M.; Wang, B.; Li, X.; Zhang, Q.; Chen, Z.; Li, H.M. Nitrogen-doped carbon quantum dots/BiOBr ultrathin nanosheets: In situ strong coupling and improved molecular oxygen activation ability under visible light irradiation. ACS Sustain. Chem. Eng. 2016, 136, 136–146. [Google Scholar] [CrossRef]
- Shan, W.; Hu, Y.; Bai, Z.; Zheng, M.; Wei, C. In situ preparation of g-C3N4/bismuth-based oxide nanocomposites with enhanced photocatalytic activity. Appl. Catal. B Environ. 2016, 188, 12. [Google Scholar]
- Kim, J.; Lee, C.W.; Choi, W. Platinized WO3 as an environmental photocatalyst that generates OH radicals under visible light. Environ. Sci. Technol. 2010, 44, 6849–6854. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Li, S.; Yu, H.; Yu, J. In situ anion-exchange synthesis and photocatalytic activity of Ag8W4O16/AgCl—nanoparticle core—shell nanorods. J. Mol. Catal. A Chem. 2011, 334, 52–59. [Google Scholar] [CrossRef]
- Yu, H.; Liu, R.; Wang, X.; Wang, P.; Yu, J. Enhanced visible-light photocatalytic activity of Bi2WO6 nanoparticles by Ag2O cocatalyst. Appl. Catal. B Environ. 2012, 111–112, 326–333. [Google Scholar] [CrossRef]
- Li, Y.; Wang, J.; Yang, Y.; Zhang, Y.; He, D.; An, Q.; Cao, G.Z. Seed-induced growing various TiO2 on nanostructures on g-C3N4 nanosheets with much enhanced photocatalytic activity under visible light. J. Hazard. Mater. 2015, 292, 79–89. [Google Scholar] [CrossRef] [PubMed]






© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jia, T.; An, J.; Yu, D.; Li, J.; Fu, F.; Wang, K.; Wang, W. Continuously Improved Photocatalytic Performance of Zn2SnO4/SnO2/Cu2O Composites by Structural Modulation and Band Alignment Modification. Nanomaterials 2019, 9, 1390. https://doi.org/10.3390/nano9101390
Jia T, An J, Yu D, Li J, Fu F, Wang K, Wang W. Continuously Improved Photocatalytic Performance of Zn2SnO4/SnO2/Cu2O Composites by Structural Modulation and Band Alignment Modification. Nanomaterials. 2019; 9(10):1390. https://doi.org/10.3390/nano9101390
Chicago/Turabian StyleJia, Tiekun, Junchao An, Dongsheng Yu, Jili Li, Fang Fu, Kun Wang, and Weimin Wang. 2019. "Continuously Improved Photocatalytic Performance of Zn2SnO4/SnO2/Cu2O Composites by Structural Modulation and Band Alignment Modification" Nanomaterials 9, no. 10: 1390. https://doi.org/10.3390/nano9101390
APA StyleJia, T., An, J., Yu, D., Li, J., Fu, F., Wang, K., & Wang, W. (2019). Continuously Improved Photocatalytic Performance of Zn2SnO4/SnO2/Cu2O Composites by Structural Modulation and Band Alignment Modification. Nanomaterials, 9(10), 1390. https://doi.org/10.3390/nano9101390




