The Adsorption of Methylene Blue by an Amphiphilic Block Co-Poly(Arylene Ether Nitrile) Microsphere-Based Adsorbent: Kinetic, Isotherm, Thermodynamic and Mechanistic Studies
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
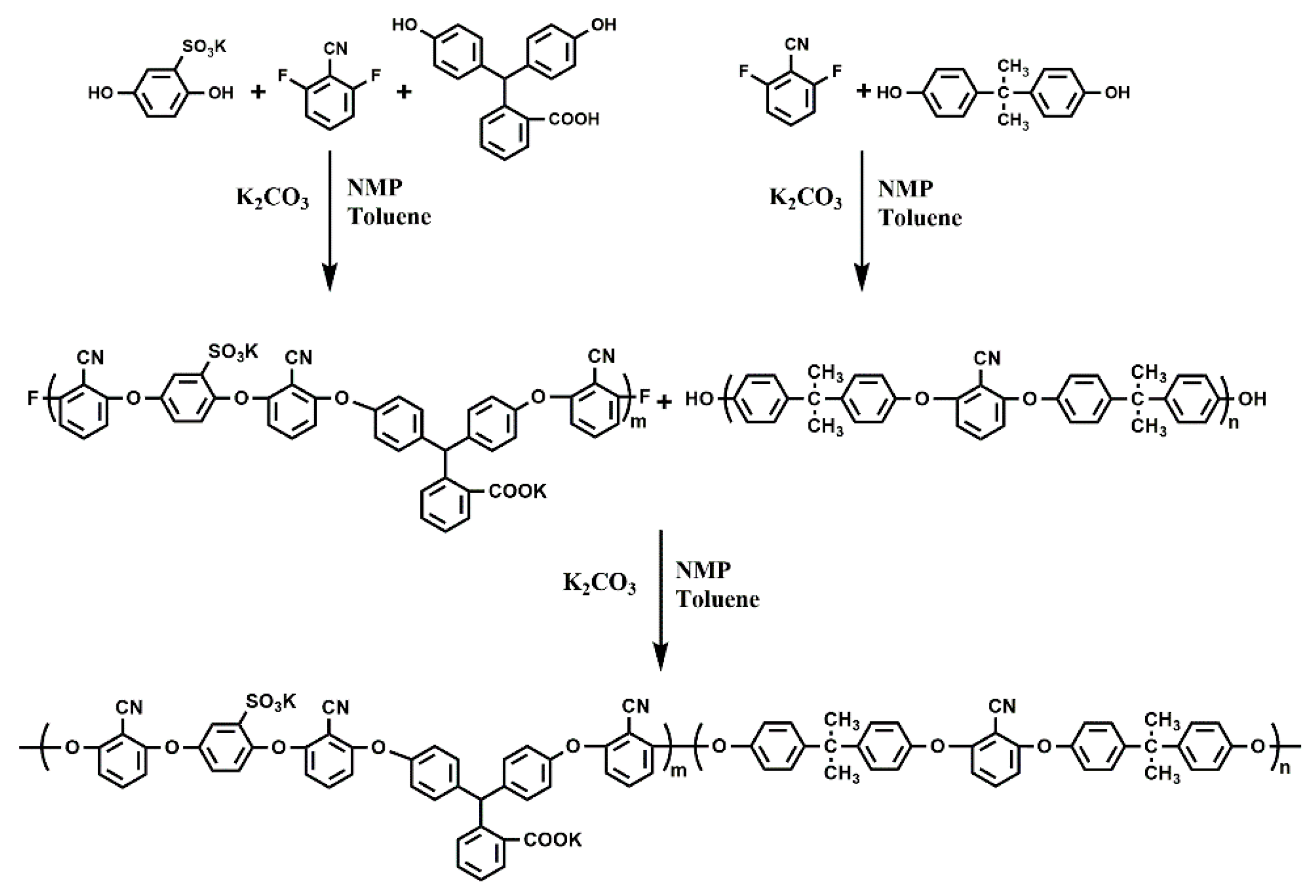
2.2. The Synthesis of B-b-S-P
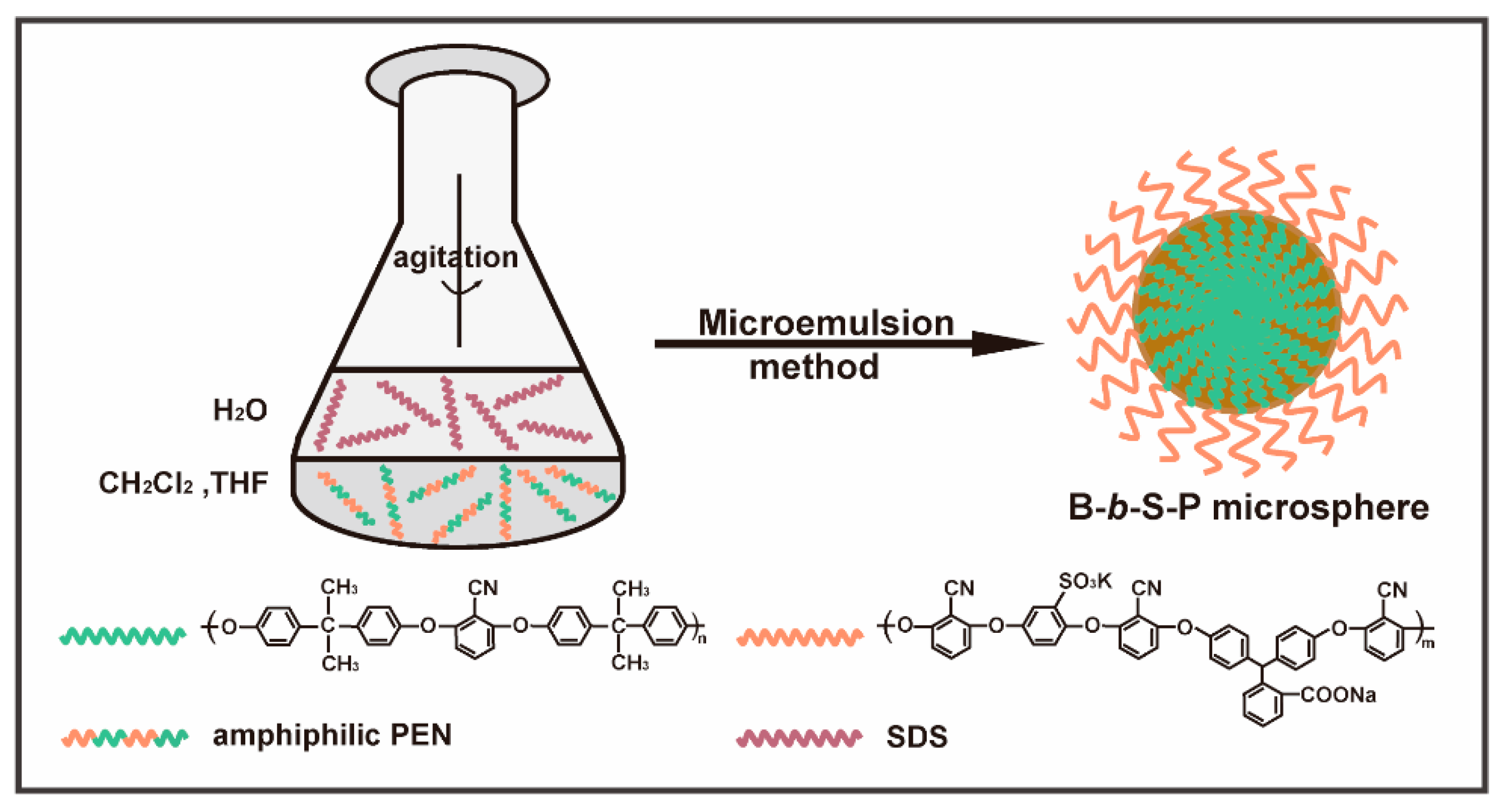
2.3. Preparation of the B-b-S-P Microspheres
2.4. Batch-of-Dye Adsorption
2.5. Characterization
3. Results and Discussion
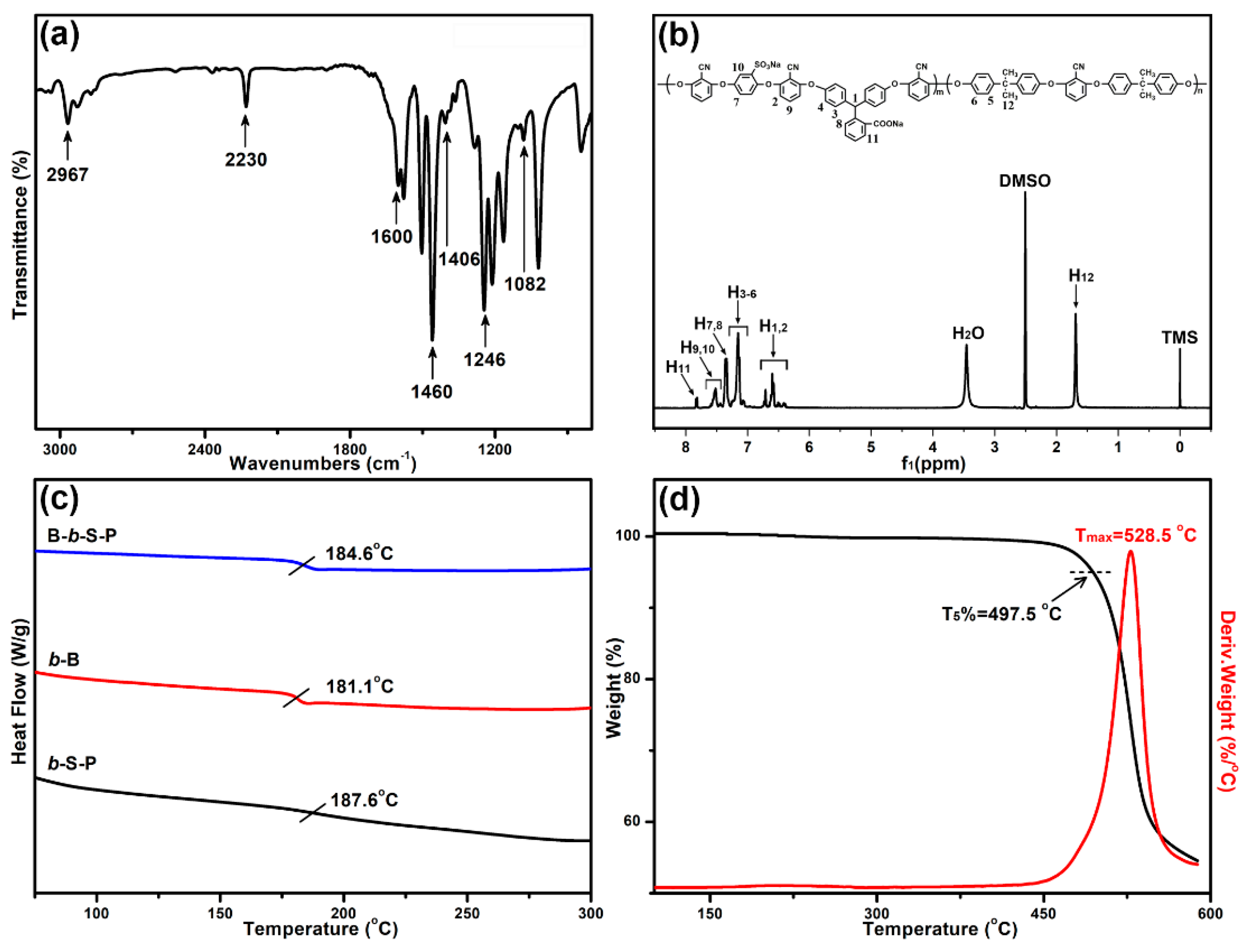
3.1. Characterization of B-b-S-P
3.2. Preparation of the B-b-S-P Microspheres
3.3. Adsorption Kinetics
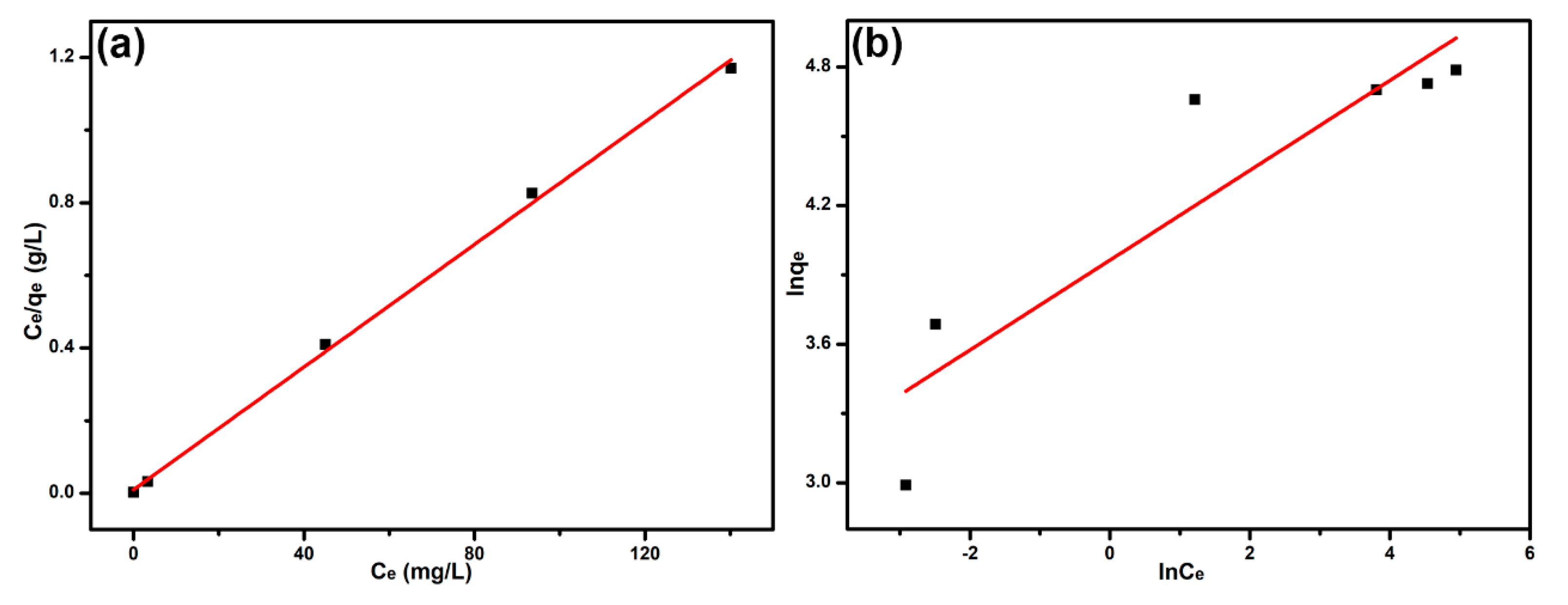
3.4. Adsorption Isotherm
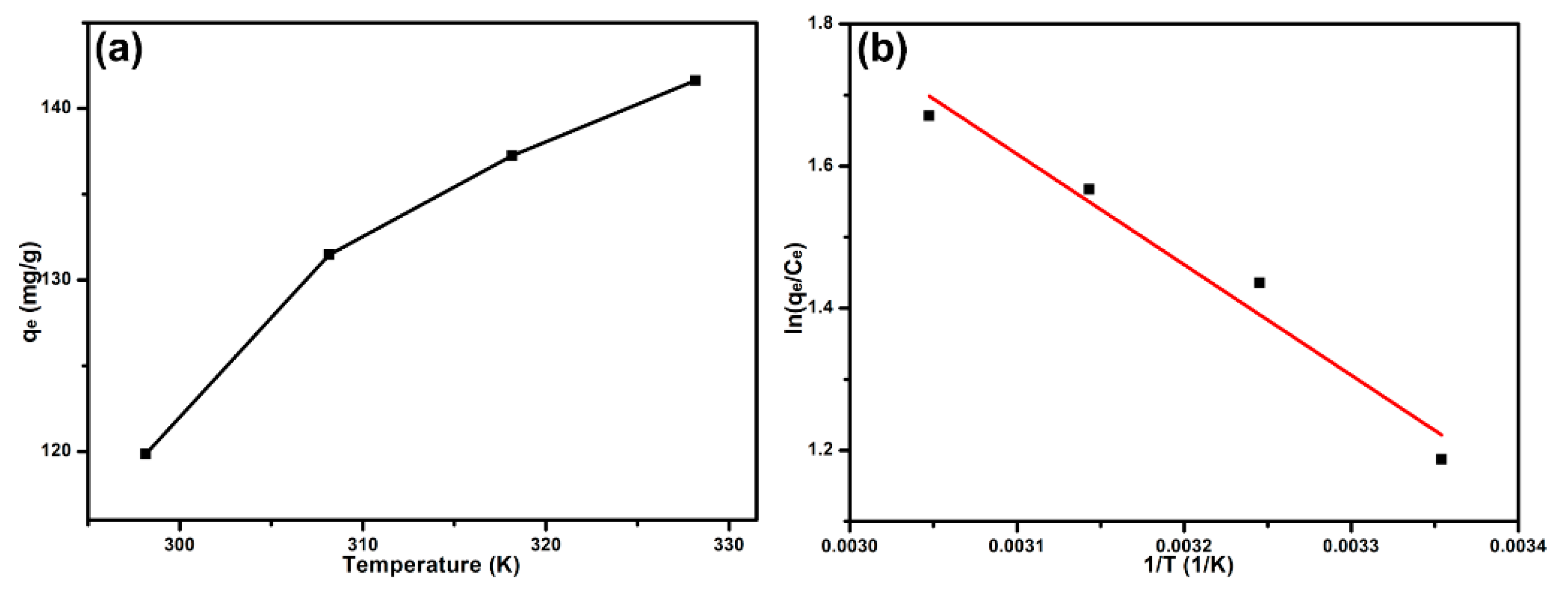
3.5. Adsorption Thermodynamics
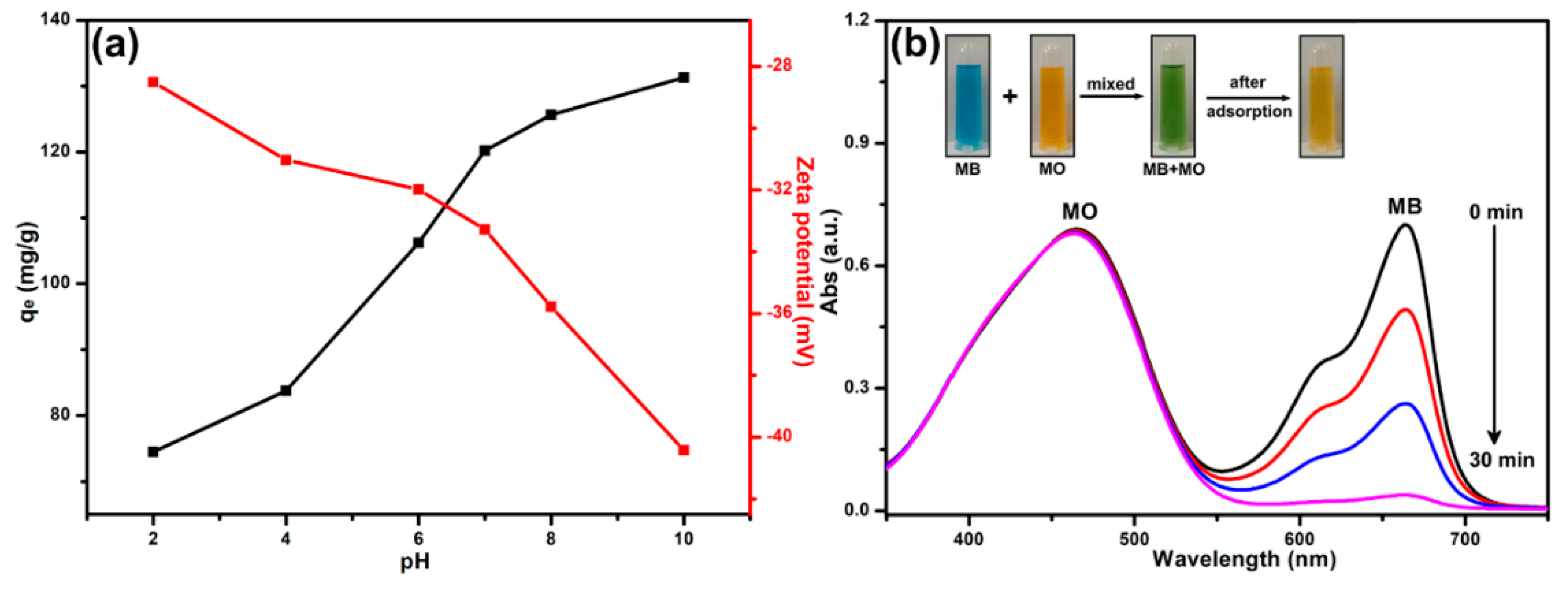
3.6. The Effect of Solution pH and Selective Adsorption for MB
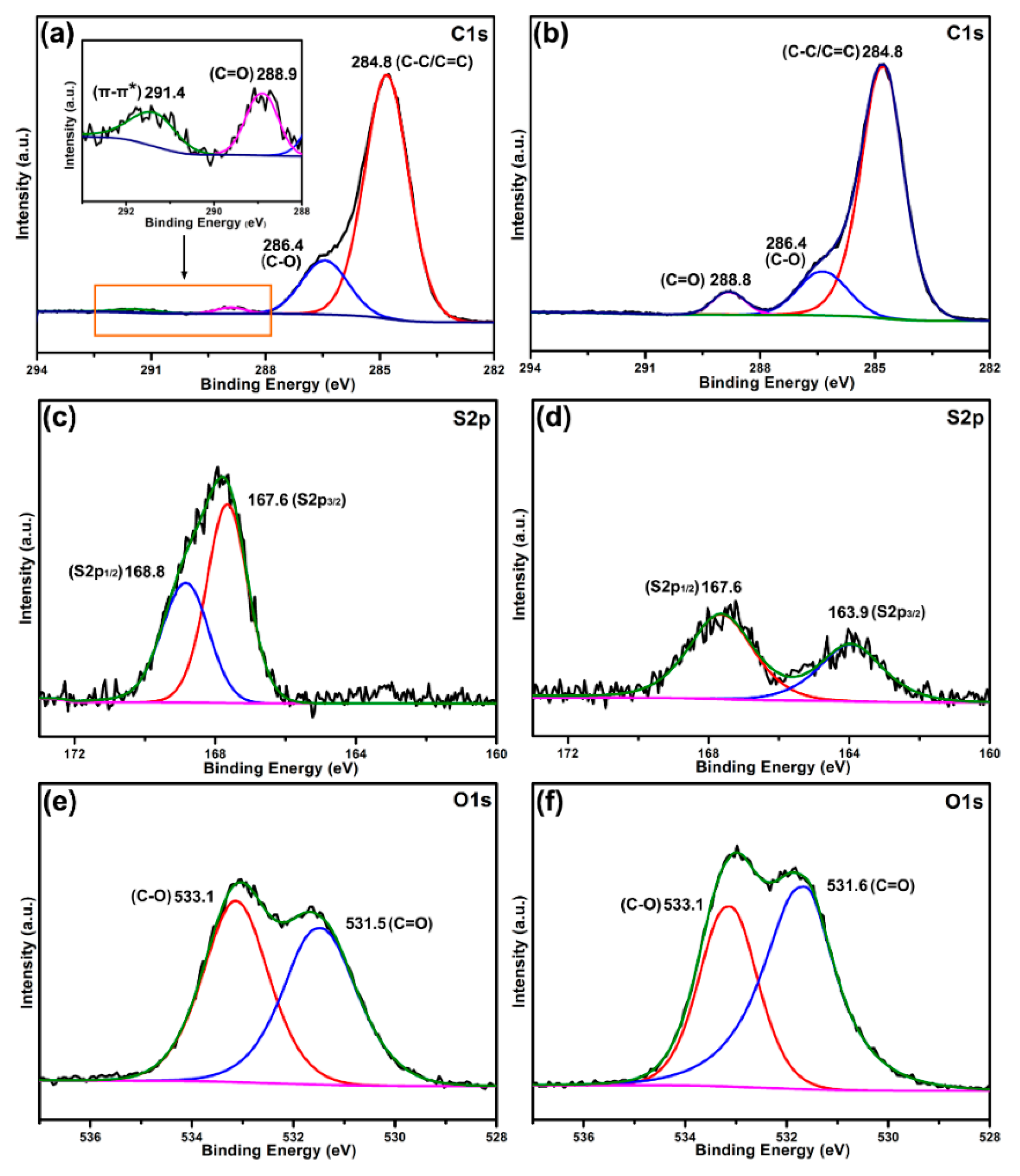
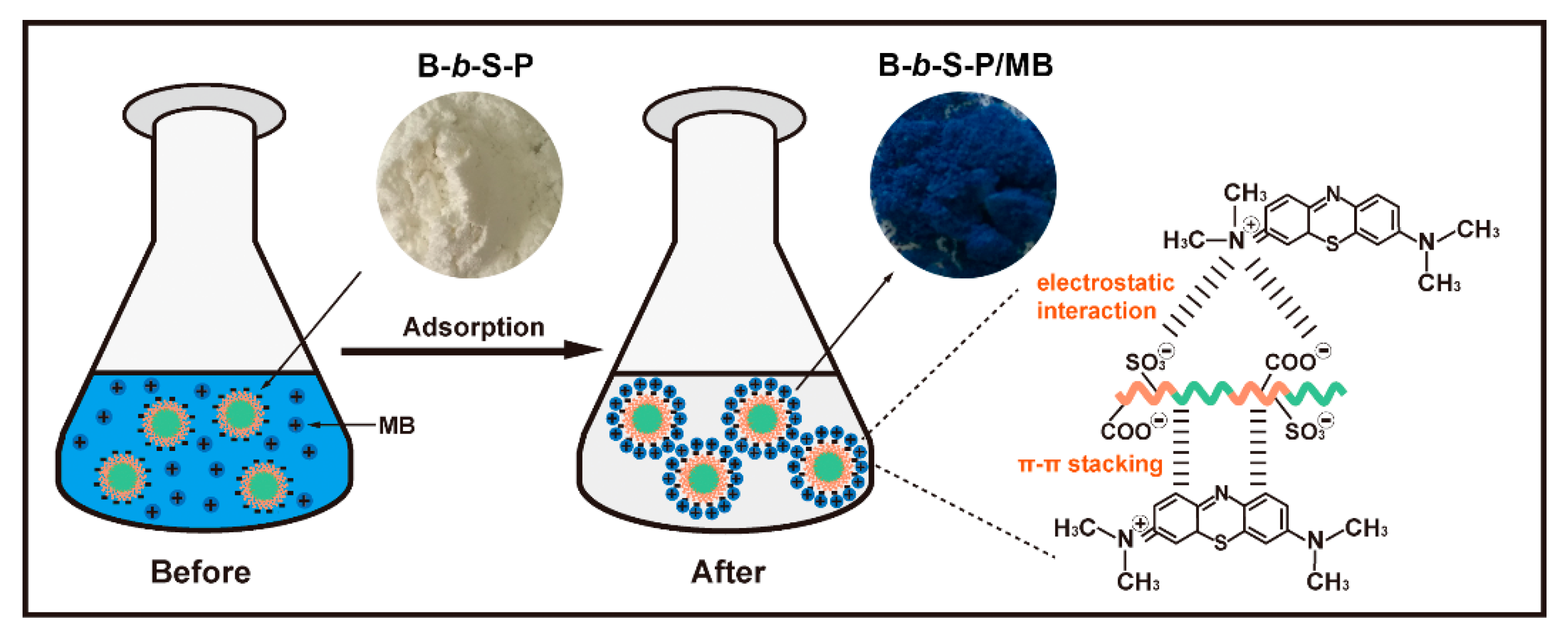
3.7. Adsorption Mechanism
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Holkar, C.R.; Jadhav, A.J.; Pinjari, D.V.; Mahamuni, N.M.; Pandit, A.B. A critical review on textile wastewater treatments: Possible approaches. J. Environ. Manag. 2016, 182, 351–366. [Google Scholar] [CrossRef] [PubMed]
- Dixit, S.; Yadav, A.; Dwivedi, P.D.; Das, M. Toxic hazards of leather industry and technologies to combat threat: A review. J. Clean. Prod. 2015, 87, 39–49. [Google Scholar] [CrossRef]
- Zhang, R.Z.; Quan, S.; Xia, M.; Wang, Q.; Zhang, W.; Yang, J.M. Effect of surface charge status of amorphous porous coordination polymer particles on the adsorption of organic dyes from an aqueous solution. J. Colloid Interface Sci. 2018, 525, 54–61. [Google Scholar] [CrossRef] [PubMed]
- Blachnio, M.; Budnyak, T.M.; Derylo-Marczewska, A.; Marczewski, A.W.; Tertykh, V.A. Chitosan-Silica Hybrid Composites for Removal of Sulfonated Azo Dyes from Aqueous Solutions. Langmuir 2018, 34, 2258–2273. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.; Zhang, Q.; Zhan, X.; Chen, F. A multifunctional gelatin-based aerogel with superior pollutants adsorption, oil/water separation and photocatalytic properties. Chem. Eng. J. 2019, 358, 1539–1551. [Google Scholar] [CrossRef]
- Verma, A.K.; Dash, R.R.; Bhunia, P. A review on chemical coagulation/flocculation technologies for removal of colour from textile wastewaters. J. Environ. Manag. 2012, 93, 154–168. [Google Scholar] [CrossRef] [PubMed]
- Solís, M.; Solís, A.; Pérez, H.I.; Manjarrez, N.; Flores, M. Microbial decolouration of azo dyes: A review. Process Biochem. 2012, 47, 1723–1748. [Google Scholar] [CrossRef]
- Geng, Z.; Yang, X.; Boo, C.; Zhu, S.; Lu, Y.; Fan, W.; Elimelech, M.; Yang, X. Self-cleaning anti-fouling hybrid ultrafiltration membranes via side chain grafting of poly (aryl ether sulfone) and titanium dioxide. J. Membr. Sci. 2017, 529, 1–10. [Google Scholar] [CrossRef]
- Zeng, G.; Ye, Z.; He, Y.; Yang, X.; Ma, J.; Shi, H.; Feng, Z. Application of dopamine-modified halloysite nanotubes/PVDF blend membranes for direct dyes removal from wastewater. Chem. Eng. J. 2017, 323, 572–583. [Google Scholar] [CrossRef]
- Lv, Y.; Xing, B.; Zheng, M.; Yi, G.; Huang, G.; Zhang, C.; Yuan, R.; Chen, Z.; Cao, Y. Hydrothermal Synthesis of Ultra-Light Coal-Based Graphene Oxide Aerogel for Efficient Removal of Dyes from Aqueous Solutions. Nanomaterials 2018, 8, 670. [Google Scholar] [CrossRef]
- Yagub, M.T.; Sen, T.K.; Afroze, S.; Ang, H.M. Dye and its removal from aqueous solution by adsorption: A review. Adv. Colloid Interface Sci. 2014, 209, 172–184. [Google Scholar] [CrossRef] [PubMed]
- Alaba, P.A.; Oladoja, N.A.; Sani, Y.M.; Ayodele, O.B.; Mohammed, I.Y.; Olupinla, S.F.; Daud, W.M.W. Insight into wastewater decontamination using polymeric adsorbents. J. Environ. Chem. Eng. 2018, 6, 1651–1672. [Google Scholar] [CrossRef]
- Yao, T.; Guo, S.; Zeng, C.; Wang, C.; Zhang, L. Investigation on efficient adsorption of cationic dyes on porous magnetic polyacrylamide microspheres. J. Hazard. Mater. 2015, 292, 90–97. [Google Scholar] [CrossRef] [PubMed]
- Fu, J.; Chen, Z.; Wang, M.; Liu, S.; Zhang, J.; Zhang, J.; Han, R.; Xu, Q. Adsorption of methylene blue by a high-efficiency adsorbent (polydopamine microspheres): Kinetics, isotherm, thermodynamics and mechanism analysis. Chem. Eng. J. 2015, 259, 53–61. [Google Scholar] [CrossRef]
- Zhou, M.; Wang, T.; He, Z.; Xu, Y.; Yu, W.; Shi, B.; Huang, K. Synthesis of Yolk–Shell Magnetic Porous Organic Nanospheres for Efficient Removal of Methylene Blue from Water. ACS. Sustain. Chem. Eng. 2019, 7, 2924–2932. [Google Scholar] [CrossRef]
- Lin, Z.; Liu, S.; Mao, W.; Tian, H.; Wang, N.; Zhang, N.; Tian, F.; Han, L.; Feng, X.; Mai, Y. Tunable Self-Assembly of Diblock Copolymers into Colloidal Particles with Triply Periodic Minimal Surfaces. Angew. Chem. Int. Ed. Engl. 2017, 56, 7135–7140. [Google Scholar] [CrossRef] [PubMed]
- McClements, D.J. Nanoemulsions versus microemulsions: Terminology, differences, and similarities. Soft Matter 2012, 8, 1719–1729. [Google Scholar] [CrossRef]
- Wang, J.; Li, W.; Zhu, J. Encapsulation of inorganic nanoparticles into block copolymer micellar aggregates: Strategies and precise localization of nanoparticles. Polymer 2014, 55, 1079–1096. [Google Scholar] [CrossRef] [Green Version]
- Wong, C.K.; Mason, A.F.; Stenzel, M.H.; Thordarson, P. Formation of non-spherical polymersomes driven by hydrophobic directional aromatic perylene interactions. Nat. Commun. 2017, 8, 1240. [Google Scholar] [CrossRef] [PubMed]
- Mai, Y.; Eisenberg, A. Self-assembly of block copolymers. Chem. Soc. Rev. 2012, 41, 5969–5985. [Google Scholar] [CrossRef] [PubMed]
- Pu, Z.; Tang, H.; Huang, X.; Yang, J.; Zhan, Y.; Zhao, R.; Liu, X. Effect of surface functionalization of SiO2 particles on the interfacial and mechanical properties of PEN composite films. Colloid Surf. A 2012, 415, 125–133. [Google Scholar] [CrossRef]
- You, Y.; Liu, S.; Tu, L.; Wang, Y.; Zhan, C.; Du, X.; Wei, R.; Liu, X. Controllable Fabrication of Poly (Arylene Ether Nitrile) Dielectrics for Thermal-Resistant Film Capacitors. Macromolecules 2019, 52, 5850–5859. [Google Scholar] [CrossRef]
- Zheng, P.; Liu, J.; Liu, X.; Jia, K. Cross-linked sulfonated poly (arylene ether nitrile) s with high selectivity for proton exchange membranes. Solid State Ionics 2017, 303, 126–131. [Google Scholar] [CrossRef]
- Feng, M.; Huang, Y.; Cheng, Y.; Liu, J.; Liu, X. Rational design of sulfonated poly (ether ether ketone) grafted graphene oxide-based composites for proton exchange membranes with enhanced performance. Polymer 2018, 144, 7–17. [Google Scholar] [CrossRef]
- Tang, H.; Pu, Z.; Huang, X.; Wei, J.; Liu, X.; Lin, Z. Novel blue-emitting carboxyl-functionalized poly (arylene ether nitrile)s with excellent thermal and mechanical properties. Polym. Chem. 2014, 5, 3673. [Google Scholar] [CrossRef]
- Zhou, X.; Jia, K.; He, X.; Wei, S.; Wang, P.; Liu, X. Microemulsion self-assembling of novel amphiphilic block co-polyarylene ether nitriles and photosensitizer ZnPc towards hybrid superparticles for photocatalytic degradation of Rhodamine B. Mater. Chem. Phys. 2018, 207, 212–220. [Google Scholar] [CrossRef]
- Zhou, X.; Zheng, P.; Wang, L.; Liu, X. Preparation of Sulfonated Poly (arylene ether nitrile)-Based Adsorbent as a Highly Selective and Efficient Adsorbent for Cationic Dyes. Polymers 2018, 11, 32. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Chen, Y.; Zhao, J.; Tong, Z.; Jin, S. Preparation of SA-g-(PAA-co-PDMC) polyampholytic superabsorbent polymer and its application to the anionic dye adsorption removal from effluents. Sep. Purif. Technol. 2017, 188, 329–340. [Google Scholar] [CrossRef]
- Singh, S.; Mahalingam, H.; Singh, P.K. Polymer-supported titanium dioxide photocatalysts for environmental remediation: A review. Appl. Catal. A Gen. 2013, 462, 178–195. [Google Scholar] [CrossRef]
- Xu, H.; Cheng, L.; Wang, C.; Ma, X.; Li, Y.; Liu, Z. Polymer encapsulated upconversion nanoparticle/iron oxide nanocomposites for multimodal imaging and magnetic targeted drug delivery. Biomaterials 2011, 32, 9364–9373. [Google Scholar] [CrossRef]
- Xu, B.; Zheng, H.; Wang, Y.; An, Y.; Luo, K.; Zhao, C.; Xiang, W. Poly (2-acrylamido-2-methylpropane sulfonic acid) grafted magnetic chitosan microspheres: Preparation, characterization and dye adsorption. Int. J. Biol. Macromol. 2018, 112, 648–655. [Google Scholar] [CrossRef] [PubMed]
- Shi, X.; Zhang, S.; Chen, X.; Mijowska, E. Evaluation of Nanoporous Carbon Synthesized from Direct Carbonization of a Metal(-)Organic Complex as a Highly Effective Dye Adsorbent and Supercapacitor. Nanomaterials 2019, 9, 601. [Google Scholar] [CrossRef] [PubMed]
- Benhouria, A.; Islam, M.A.; Zaghouane-Boudiaf, H.; Boutahala, M.; Hameed, B.H. Calcium alginate–bentonite–activated carbon composite beads as highly effective adsorbent for methylene blue. Chem. Eng. J. 2015, 270, 621–630. [Google Scholar] [CrossRef]
- Li, J.; Zhao, H.; Ma, C.; Han, Q.; Li, M.; Liu, H. Preparation of Fe3O4@polyoxometalates Nanocomposites and Their Efficient Adsorption of Cationic Dyes from Aqueous Solution. Nanomaterials 2019, 9, 649. [Google Scholar] [CrossRef] [PubMed]
- Fu, J.; Chen, Z.; Wu, X.; Wang, M.; Wang, X.; Zhang, J.; Zhang, J.; Xu, Q. Hollow poly(cyclotriphosphazene-co-phloroglucinol) microspheres: An effective and selective adsorbent for the removal of cationic dyes from aqueous solution. Chem. Eng. J. 2015, 281, 42–52. [Google Scholar] [CrossRef]
- Chen, Z.; Fu, J.; Wang, M.; Wang, X.; Zhang, J.; Xu, Q. Adsorption of cationic dye (methylene blue) from aqueous solution using poly(cyclotriphosphazene-co-4,4′-sulfonyldiphenol) nanospheres. Appl. Surf. Sci. 2014, 289, 495–501. [Google Scholar] [CrossRef]
- Basaleh, A.A.; Al-Malack, M.H.; Saleh, T.A. Methylene Blue removal using polyamide-vermiculite nanocomposites: Kinetics, equilibrium and thermodynamic study. J. Environ. Chem. Eng. 2019, 7, 103107. [Google Scholar] [CrossRef]
- Jamwal, H.S.; Kumari, S.; Chauhan, G.S.; Reddy, N.S.; Ahn, J.-H. Silica-polymer hybrid materials as methylene blue adsorbents. J. Environ. Chem. Eng. 2017, 5, 103–113. [Google Scholar] [CrossRef]
- He, K.; Zeng, G.; Chen, A.; Huang, Z.; Peng, M.; Huang, T.; Chen, G. Graphene hybridized polydopamine-kaolin composite as effective adsorbent for methylene blue removal. Compos. Part B Eng. 2019, 161, 141–149. [Google Scholar] [CrossRef]
- Azha, S.F.; Sellaoui, L.; Shamsudin, M.S.; Ismail, S.; Bonilla-Petriciolet, A.; Ben Lamine, A.; Erto, A. Synthesis and characterization of a novel amphoteric adsorbent coating for anionic and cationic dyes adsorption: Experimental investigation and statistical physics modelling. Chem. Eng. J. 2018, 351, 221–229. [Google Scholar] [CrossRef]
- Pourjavadi, A.; Abedin-Moghanaki, A. Ultrafast and efficient removal of cationic dyes using a magnetic nanocomposite based on functionalized cross-linked poly(methylacrylate). React. Funct. Polym. 2016, 105, 95–102. [Google Scholar] [CrossRef]
- Fu, J.; Xin, Q.; Wu, X.; Chen, Z.; Yan, Y.; Liu, S.; Wang, M.; Xu, Q. Selective adsorption and separation of organic dyes from aqueous solution on polydopamine microspheres. J. Colloid Interface Sci. 2016, 461, 292–304. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Wei, R.; Gu, J.; Liu, H.; Liu, C.; Luo, C.; Kong, J.; Shao, Q.; Wang, N.; Guo, Z.; et al. Ultralight, highly compressible and fire-retardant graphene aerogel with self-adjustable electromagnetic wave absorption. Carbon 2018, 139, 1126–1135. [Google Scholar] [CrossRef]

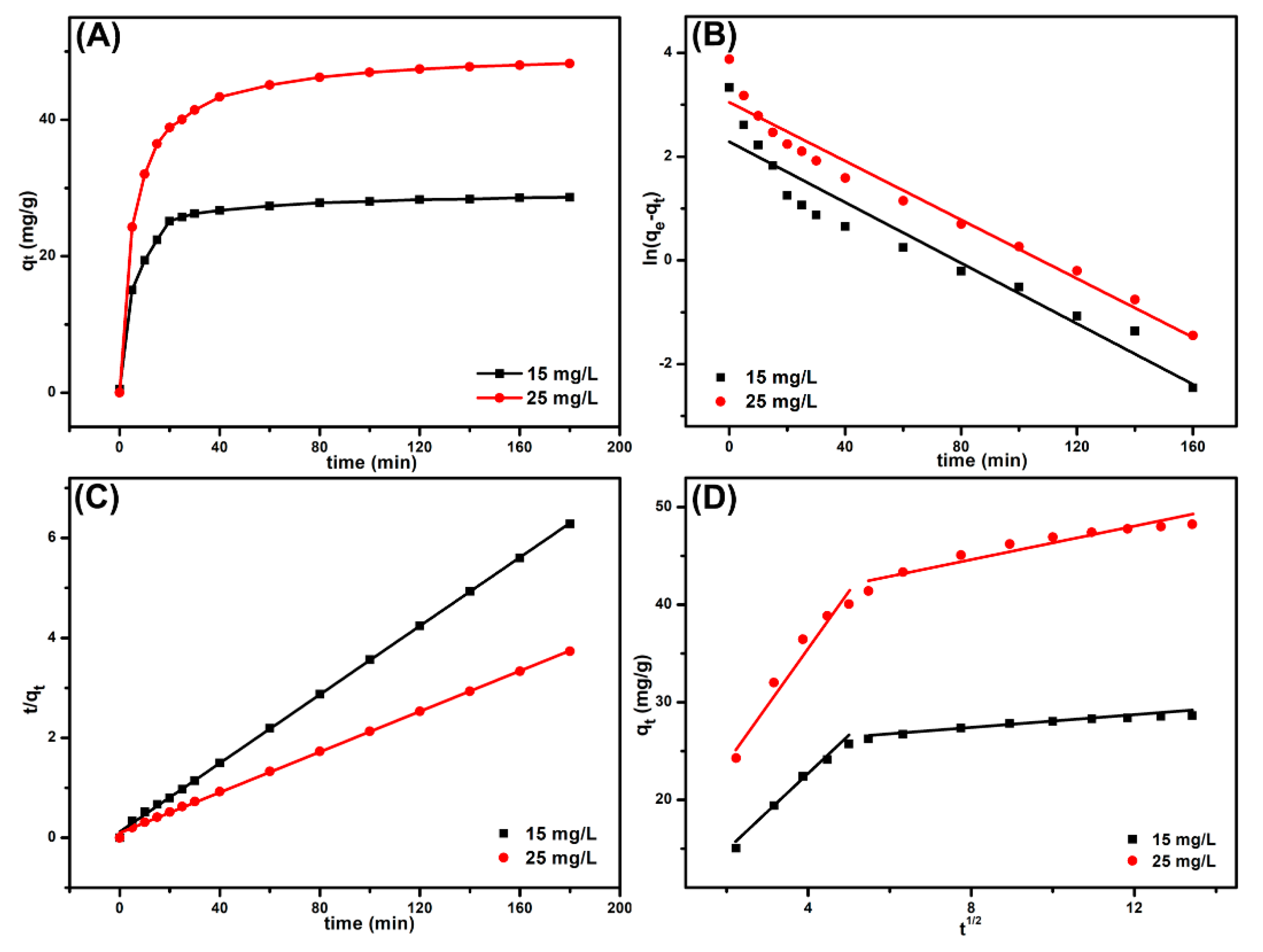
| Models | Parameters | 15 mg L−1 | 25 mg L−1 |
|---|---|---|---|
| Pseudo-first-order | k1 (min−1) | 0.0292 | 0.0283 |
| qe (cal.) (mg g−1) | 9.8384 | 21.066 | |
| qe (exp.) (mg g−1) | 28.643 | 48.243 | |
| R2 | 0.9163 | 0.9587 | |
| Pseudo-second-order | k2 (g mg−1 min−1) | 0.0120 | 0.0042 |
| qe (cal.) (mg g−1) | 29.155 | 49.505 | |
| qe (exp.) (mg g−1) | 28.643 | 48.243 | |
| R2 | 0.9996 | 0.9994 | |
| Intraparticle diffusion | ki1 (mg g−1 min−0.5) | 3.8658 | 5.7591 |
| C1 | 6.8537 | 12.744 | |
| R12 | 0.9843 | 0.9413 | |
| ki2 (mg g−1 min−0.5) | 0.2945 | 0.8029 | |
| C2 | 24.930 | 38.259 | |
| R22 | 0.9395 | 0.8994 |
| Isotherms | Parameters | Temperatures (K) 298.15 |
|---|---|---|
| Langmuir | qm (mg g−1) | 119.05 |
| KL (L mg−1) | 0.8660 | |
| R2 | 0.9981 | |
| Freundlich | KF (L mg−1) | 52.657 |
| n−1 | 0.1943 | |
| R2 | 0.7975 |
| Adsorbent | qm (mg g−1) | Reference |
|---|---|---|
| DPA microspheres | 90.7 | [14] |
| CS-MPONs nanocomposites | 104 | [15] |
| PCPP microspheres | 50.7 | [35] |
| PZS nanospheres | 20.0 | [36] |
| Polyamide-vermiculite nanocomposites | 76.42 | [37] |
| Poly(methacrylate)/silica hybrid materials | 91.324 | [38] |
| PDA-rGO-kaolin composite | 39.663 | [39] |
| B-b-S-P microspheres | 119.84 | This work |
| T (K) | Thermodynamic Parameters | |||
|---|---|---|---|---|
| lnK | ΔGO (kJ mol−1) | ΔSO (J mol−1 K−1) | ΔHO (kJ mol−1) | |
| 298 | 1.1873 | −2.9431 | 53.545 | 1.2936 |
| 308 | 1.4360 | −3.6790 | — | — |
| 318 | 1.5672 | −4.1454 | — | — |
| 328 | 1.6709 | −4.5585 | — | — |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhou, X.; Xu, M.; Wang, L.; Liu, X. The Adsorption of Methylene Blue by an Amphiphilic Block Co-Poly(Arylene Ether Nitrile) Microsphere-Based Adsorbent: Kinetic, Isotherm, Thermodynamic and Mechanistic Studies. Nanomaterials 2019, 9, 1356. https://doi.org/10.3390/nano9101356
Zhou X, Xu M, Wang L, Liu X. The Adsorption of Methylene Blue by an Amphiphilic Block Co-Poly(Arylene Ether Nitrile) Microsphere-Based Adsorbent: Kinetic, Isotherm, Thermodynamic and Mechanistic Studies. Nanomaterials. 2019; 9(10):1356. https://doi.org/10.3390/nano9101356
Chicago/Turabian StyleZhou, Xuefei, Mingzhen Xu, Lingling Wang, and Xiaobo Liu. 2019. "The Adsorption of Methylene Blue by an Amphiphilic Block Co-Poly(Arylene Ether Nitrile) Microsphere-Based Adsorbent: Kinetic, Isotherm, Thermodynamic and Mechanistic Studies" Nanomaterials 9, no. 10: 1356. https://doi.org/10.3390/nano9101356
APA StyleZhou, X., Xu, M., Wang, L., & Liu, X. (2019). The Adsorption of Methylene Blue by an Amphiphilic Block Co-Poly(Arylene Ether Nitrile) Microsphere-Based Adsorbent: Kinetic, Isotherm, Thermodynamic and Mechanistic Studies. Nanomaterials, 9(10), 1356. https://doi.org/10.3390/nano9101356





