Evaluating Nanoshells and a Potent Biladiene Photosensitizer for Dual Photothermal and Photodynamic Therapy of Triple Negative Breast Cancer Cells
Abstract
1. Introduction
2. Materials and Methods
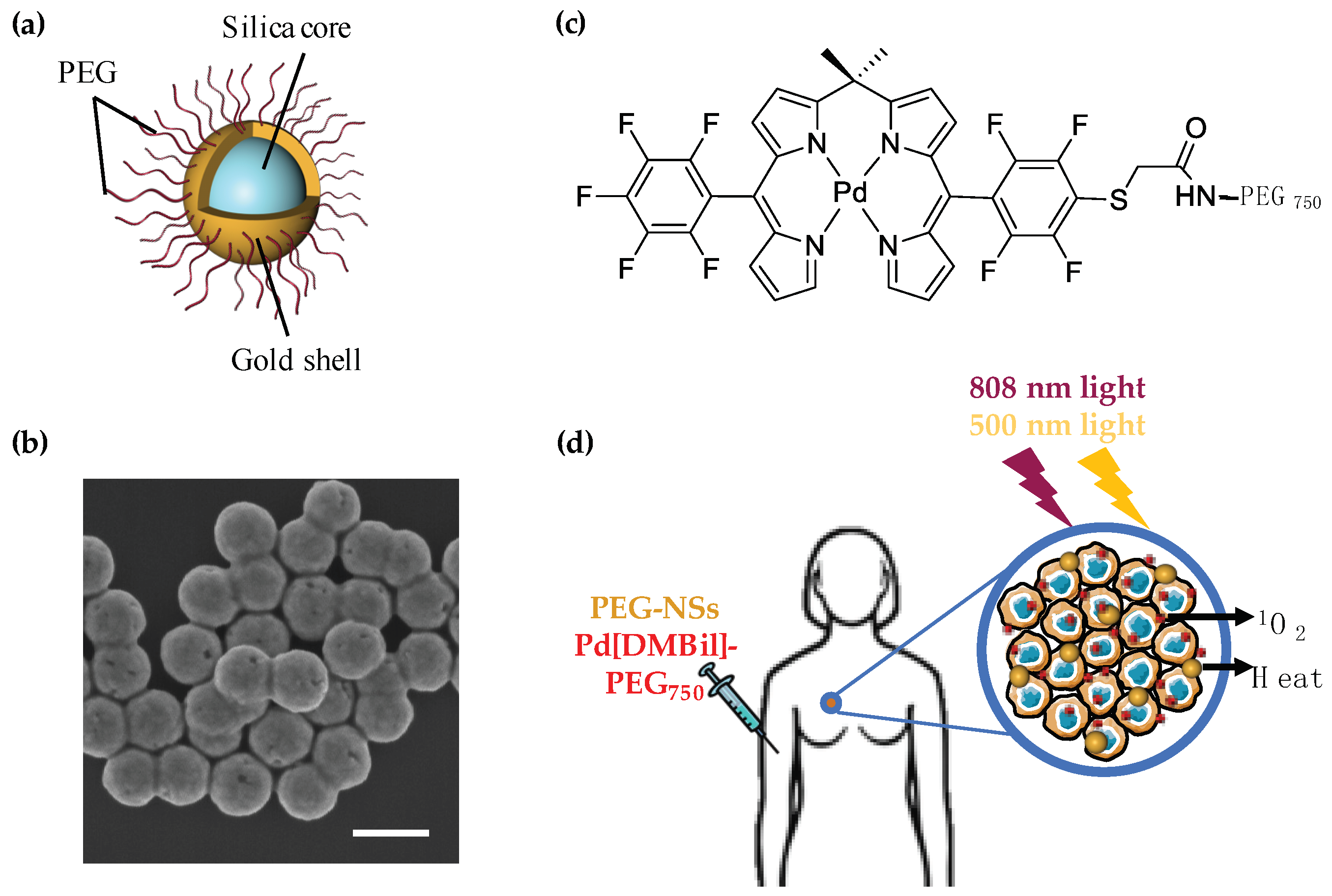
2.1. Nanoshell Synthesis and Functionalization
2.2. Pd[DMBil1]-PEG750 Synthesis
2.3. Characterization and Stability of PEG-NSs and Pd[DMBil1]-PEG750
2.4. Temperature and 1O2 Production during PTT and PDT
2.5. Cell Culture
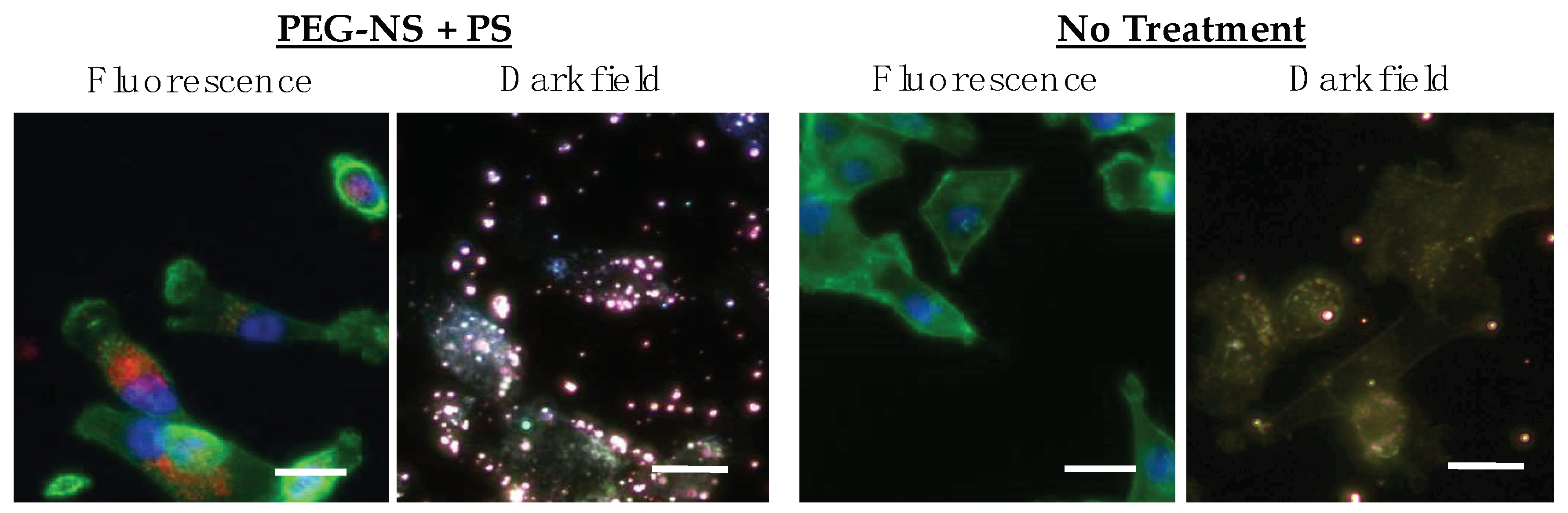
2.6. Cellular Binding and Uptake of Pd[DMBil1]-PEG750 and PEG-NSs
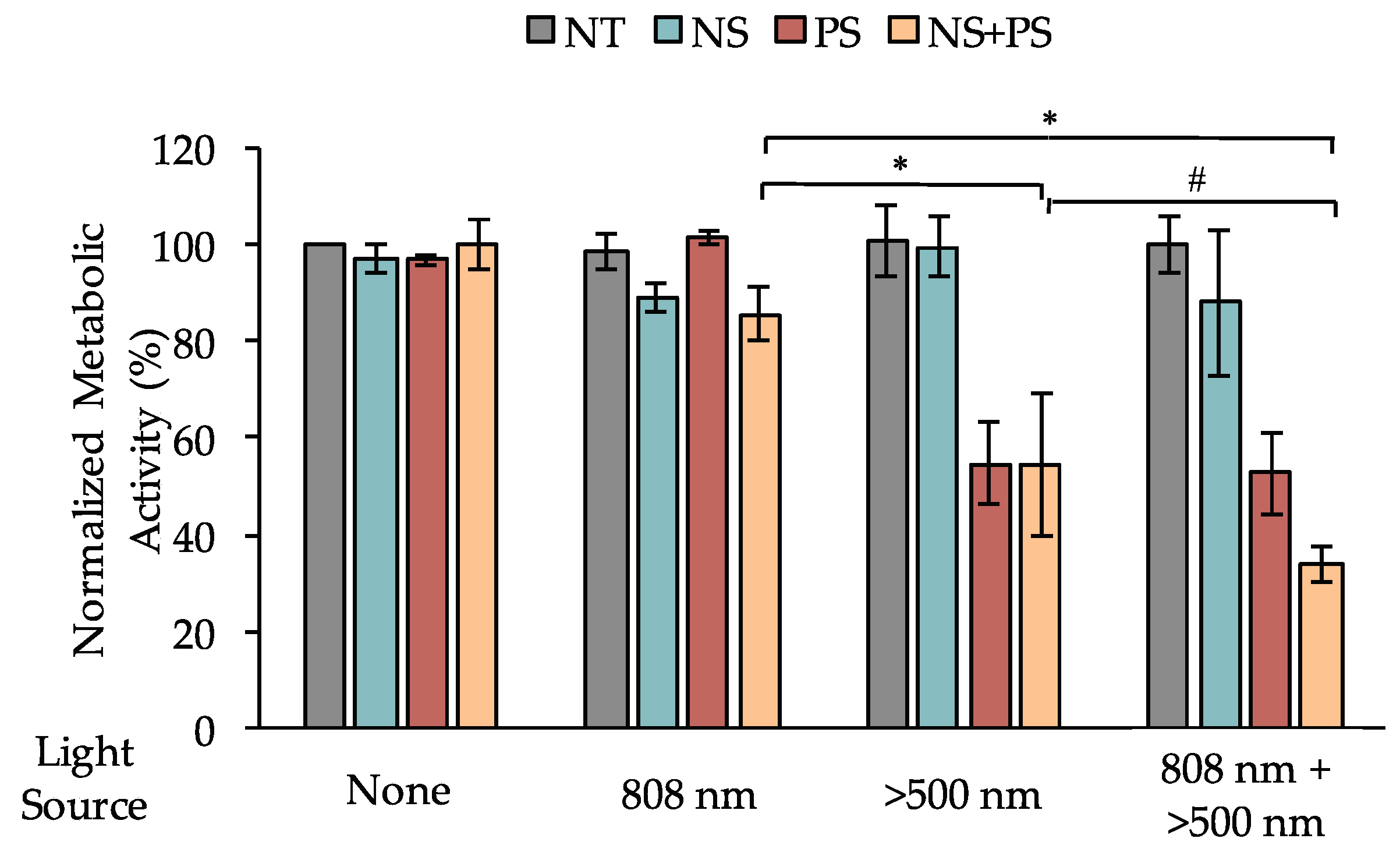
2.7. Assessment of Cells’ Metabolic Activity Following PTT and/or PDT
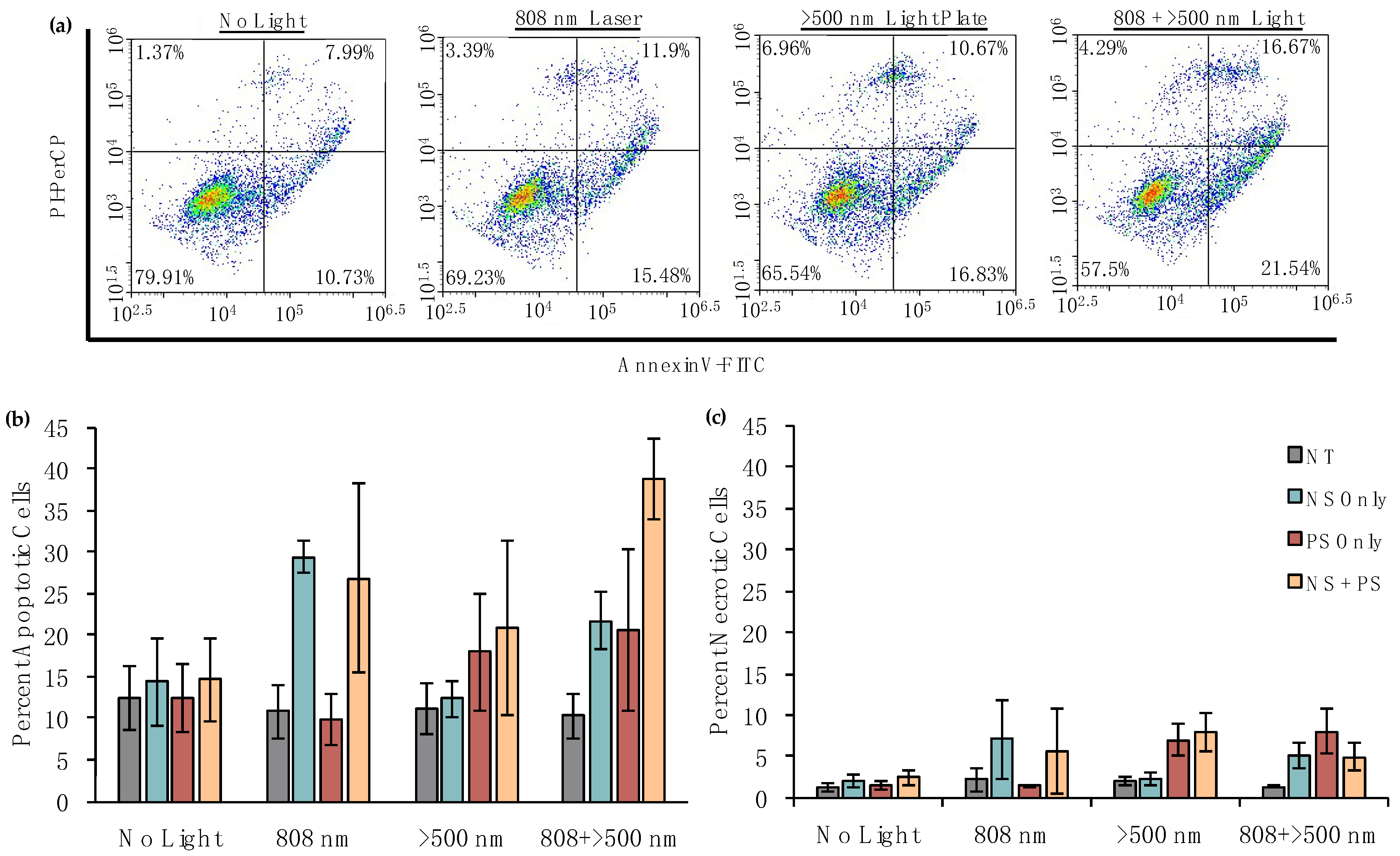
2.8. Analysis of the Mechanisms of Cell Death Induced by PTT and/or PDT
3. Results
3.1. PEG-NSs and Pd[DMBil1]-PEG750 Are Stable and Retain Their Individual Photophysical Properties When Combined
3.2. PEG-NSs and Pd[DMBil1]-PEG750 Produce Heat and 1O2 When Combined
3.3. PEG-NSs and Pd[DMBil1]-PEG750 Are Internalized within Cells
3.4. Dual PTT/PDT Is a Potent Strategy to Treat TNBC
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Matsumura, Y.; Maeda, H. A new concept for macromolecular therapeutics in cancer chemotherapy: Mechanism of tumoritropic accumulation of proteins and the antitumor agent smancs. Cancer Res. 1986, 46, 6387–6392. [Google Scholar] [PubMed]
- Maeda, H. The enhanced permeability and retention (EPR) effect in tumor vasculature: The key role of tumor-selective macromolecular drug targeting. Adv. Enzyme Regul. 2001, 41, 189–207. [Google Scholar] [CrossRef]
- Riley, R.S.; Day, E.S. Gold nanoparticle-mediated photothermal therapy: Applications and opportunities for multimodal cancer treatment. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2017, 9, e1449. [Google Scholar] [CrossRef] [PubMed]
- Riley, R.S.; Dang, M.N.; Billingsley, M.M.; Abraham, B.; Gundlach, L.; Day, E.S. Evaluating the mechanisms of light-triggered siRNA release from nanoshells for temporal control over gene regulation. Nano Lett. 2018, 18, 3565–3570. [Google Scholar] [CrossRef] [PubMed]
- Day, E.S.; Morton, J.G.; West, J.L. Nanoparticles for thermal cancer therapy. J. Biomech. Eng. 2009, 131, 074001. [Google Scholar] [CrossRef] [PubMed]
- Eloy, J.O.; Petrilli, R.; Lopez, R.F.V.; Lee, R.J. Stimuli-responsive nanoparticles for siRNA delivery. Curr. Pharm. Des. 2015, 21, 4131–4144. [Google Scholar] [CrossRef] [PubMed]
- Lucky, S.S.; Soo, K.C.; Zhang, Y. Nanoparticles in photodynamic therapy. Chem. Rev. 2015, 115, 1990–2042. [Google Scholar] [CrossRef] [PubMed]
- Hussein, E.A.; Zagho, M.M.; Nasrallah, G.K.; Elzatahry, A.A. Recent advances in functional nanostructures as cancer photothermal therapy. Int. J. Nanomed. 2018, 13, 2897–2906. [Google Scholar] [CrossRef] [PubMed]
- Melamed, J.R.; Edelstein, R.S.; Day, E.S. Elucidating the fundamental mechanisms of cell death triggered by photothermal therapy. ACS Nano 2015, 9, 6–11. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Hernández, M.; Del Pino, P.; Mitchell, S.G.; Moros, M.; Stepien, G.; Pelaz, B.; Parak, W.J.; Gálvez, E.M.; Pardo, J.; De La Fuente, J.M. Dissecting the molecular mechanism of apoptosis during photothermal therapy using gold nanoprisms. ACS Nano 2015, 9, 52–61. [Google Scholar] [CrossRef] [PubMed]
- Mroz, P.; Yaroslavsky, A.; Kharkwal, G.B.; Hamblin, M.R. Cell death pathways in photodynamic therapy of cancer. Cancers 2011, 3, 2516–2539. [Google Scholar] [CrossRef] [PubMed]
- Benov, L. Photodynamic therapy: Current status and future directions. Med. Princ. Pract. 2015, 24, 14–28. [Google Scholar] [CrossRef] [PubMed]
- Triesscheijn, M.; Baas, P.; Schellens, J.H.M.; Stewart, F.A. photodynamic therapy in oncology. Oncologist 2006, 11, 1034–1044. [Google Scholar] [CrossRef] [PubMed]
- Agostinis, P.; Berg, K.; Cengel, K.; Foster, T.; Girotti, A.; Gollnick, S.; Hahn, S.; Hamblin, M.; Juzeniene, A.; Kessel, D.; Koberlik, M.; et al. Photodynamic therapy of cancer: an update. CA Cancer J. Clin. 2011, 61, 250–281. [Google Scholar] [CrossRef] [PubMed]
- Ozog, D.M.; Rkein, A.M.; Fabi, S.G.; Gold, M.H.; Goldman, M.P.; Lowe, N.J.; Martin, G.M.; Munavalli, G.S. Photodynamic therapy: A clinical consensus guide. Dermatol. Surg. 2016, 42, 804–827. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Xu, Y.; Shi, J.; Gao, X.; Geng, L. Photodynamic therapy in the treatment of basal cell carcinoma: A systematic review and meta-analysis. Photodermatol. Photoimmunol. Photomed. 2015, 31, 44–53. [Google Scholar] [CrossRef] [PubMed]
- Nowis, D.; Stokłosa, T.; Legat, M.; Issat, T.; Jakóbisiak, M.; Goła̧b, J. The influence of photodynamic therapy on the immune response. Photodiagn. Photodyn. Ther. 2005, 2, 283–298. [Google Scholar] [CrossRef]
- Castano, A.P.; Mroz, P.; Hamblin, M. Photodynamic therapy and anti-tumor immunity. Nat. Rev. Cancer 2006, 6, 535–545. [Google Scholar] [CrossRef] [PubMed]
- Dolmans, D.; Fukumura, D.; Jain, R. Photodynamic therapy for cancer. Nat. Rev. Cancer 2003, 3, 380–387. [Google Scholar] [CrossRef] [PubMed]
- Mari, C.; Pierroz, V.; Ferrari, S.; Gasser, G. Combination of Ru(II) complexes and light: New frontiers in cancer therapy. Chem. Sci. 2015, 6, 2660–2686. [Google Scholar] [CrossRef] [PubMed]
- Ai, F.; Ju, Q.; Zhang, X.; Chen, X.; Wang, F.; Zhu, G. A core-shell-shell nanoplatform upconverting near-infrared light at 808 nm for luminescence imaging and photodynamic therapy of cancer. Sci. Rep. 2015, 5, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Day, E.S.; Thompson, P.A.; Zhang, L.; Lewinski, N.A.; Ahmed, N.; Drezek, R.A.; Blaney, S.M.; West, J.L. Nanoshell-mediated photothermal therapy improves survival in a murine glioma model. J. Neurooncol. 2011, 104, 55–63. [Google Scholar] [CrossRef] [PubMed]
- Fay, B.L.; Melamed, J.R.; Day, E.S. Nanoshell-mediated photothermal therapy can enhance chemotherapy in inflammatory breast cancer cells. Int. J. Nanomed. 2015, 10, 6931–6941. [Google Scholar] [CrossRef]
- Day, E.S.; Zhang, L.; Thompson, P.A.; Zawaski, J.A.; Kaffes, C.C.; Gaber, M.W.; Blaney, S.M.; West, J.L. Vascular-targeted photothermal therapy of an orthotopic murine glioma model. Nanomedicine 2012, 7, 1133–1148. [Google Scholar] [CrossRef] [PubMed]
- Stern, J.M.; Solomonov, V.V.K.; Sazykina, E.; Schwartz, J.A.; Gad, S.C.; Goodrich, G.P. Initial evaluation of the safety of nanoshell-directed photothermal therapy in the treatment of prostate disease. Int. J. Toxicol. 2015, 35, 38–46. [Google Scholar] [CrossRef] [PubMed]
- Nanospectra Biosciences Inc. Efficacy Study of AuroLase Therapy in Subjects with Primary and/or Metastatic Lung Tumors. Available online: https://clinicaltrials.gov/ct2/show/NCT01679470?term=auroshell&rank=3 (accessed on 23 May 2016).
- Nanospectra Biosciences Inc. Pilot Study of AuroLase(tm) Therapy in Refractory and/or Recurrent Tumors of the Head and Neck. Available online: https://clinicaltrials.gov/ct2/show/NCT00848042?term=auroshell&rank=2 (accessed on 23 May 2016).
- Potocny, A.; Riley, R.; O’Sullivan, R.; Day, E.; Rosenthal, K. Photochemotherapeutic properties of a linear tetrapyrrole palladium(II) complex displaying an exceptionally high phototoxicity index. Inorg. Chem. 2018. [Google Scholar] [CrossRef] [PubMed]
- Oh, J.; Yoon, H.; Park, J.H. Nanoparticle platforms for combined photothermal and photodynamic therapy. Biomed. Eng. Lett. 2013, 3, 67–73. [Google Scholar] [CrossRef]
- Wang, L.; Meng, D.; Hao, Y.; Zhao, Y.; Li, D.; Zhang, B.; Zhang, Y.; Zhang, Z. Gold nanostars mediated combined photothermal and photodynamic therapy and X-ray imaging for cancer theranostic applications. J. Biomater. Appl. 2015, 30, 547–557. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Shi, Q.; Zhu, C.; Luo, Y.; Lu, Q.; Li, H.; Ye, R.; Du, D.; Lin, Y. Mitochondrial-targeted multifunctional mesoporous Au@Pt nanoparticles for dual-mode photodynamic and photothermal therapy of cancers. Nanoscale 2017, 9, 15813–15824. [Google Scholar] [CrossRef] [PubMed]
- Yin, W.; Bao, T.; Zhang, X.; Gao, Q.; Yu, J.; Dong, X.; Yan, L.; Gu, Z.; Zhao, Y. Biodegradable MoOxnanoparticles with efficient near-infrared photothermal and photodynamic synergetic cancer therapy at the second biological window. Nanoscale 2018, 10, 1517–1531. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.; Shi, S.; Huang, Y.; Tang, S.; Chen, X. Simultaneous photodynamic and photothermal therapy using photosensitizer-functionalized Pd nanosheets by single continuous wave laser. ACS Appl. Mater. Interfaces 2014, 6, 8878–8885. [Google Scholar] [CrossRef] [PubMed]
- Tan, X.; Pang, X.; Lei, M.; Ma, M.; Guo, F.; Wang, J.; Yu, M.; Tan, F.; Li, N. An efficient dual-loaded multifunctional nanocarrier for combined photothermal and photodynamic therapy based on copper sulfide and chlorin e6. Int. J. Pharm. 2016, 503, 220–228. [Google Scholar] [CrossRef] [PubMed]
- Griffiths, C.L.; Olin, J.L. Triple negative breast cancer: a brief review of its characteristics and treatment options. J. Pharm. Pract. 2012, 25, 319–323. [Google Scholar] [CrossRef] [PubMed]
- Oldenburg, S.; Averitt, R.; Westcott, S.; Halas, N. Nanoengineering of optical resonances. Chem. Phys. Lett. 1998, 288, 243–247. [Google Scholar] [CrossRef]
- Duff, D.G.; Baiker, A.; Edwards, P.P. A new hydrosol of gold clusters. 1. Formation and Particle Size Variation. ACS Langmuir 1993, 9, 2301–2309. [Google Scholar] [CrossRef]
- Pistner, A.J.; Yap, G.P.A.; Rosenthal, J. A tetrapyrrole macrocycle displaying a multielectron redox chemistry and tunable absorbance profile. J. Phys. Chem. C 2012, 116, 16918–16924. [Google Scholar] [CrossRef] [PubMed]
- Pistner, A.J.; Pupillo, R.C.; Yap, G.P.A.; Lutterman, D.A.; Ma, Y.Z.; Rosenthal, J. Electrochemical, spectroscopic, and1O2 sensitization characteristics of 10,10-dimethylbiladiene complexes of zinc and copper. J. Phys. Chem. A 2014, 118, 10639–10648. [Google Scholar] [CrossRef] [PubMed]
- Potocny, A.M.; Pistner, A.J.; Yap, G.P.A.; Rosenthal, J. Electrochemical, spectroscopic, and 1O2 sensitization characteristics of synthetically accessible linear tetrapyrrole complexes of palladium and platinum. Inorg. Chem. 2017, 56, 12703–12711. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, A.Y.; McGann, J.P.; Geier, G.R. Dipyrromethane + dipyrromethanedicarbinol routes to an electron deficient meso-substituted phlorin with enhanced stability. J. Org. Chem. 2007, 72, 4084–4092. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Ye, H.L.; Zhang, G.; Yao, W.M.; Chen, X.Z.; Zhang, F.C.; Liang, G. Autophagy inhibition contributes to the synergistic interaction between EGCG and doxorubicin to kill the hepatoma Hep3B cells. PLoS ONE 2014, 9. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Lin, Z.; Zhang, B.; Guo, L.; Liu, S.; Li, H.; Zhang, J.; Ye, Q. Β-elemene sensitizes hepatocellular carcinoma cells to oxaliplatin by Preventing oxaliplatin-induced degradation of copper transporter 1. Sci. Rep. 2016, 6, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Riley, R.S.; Day, E.S. Frizzled7 antibody-functionalized nanoshells enable multivalent binding for wnt signaling inhibition in triple negative breast cancer cells. Small 2017, 13, 1700544. [Google Scholar] [CrossRef] [PubMed]
- Lal, S.; Clare, S.E.; Halas, N.J. Nanoshell-enabled photothermal cancer therapy: Impending clinical impact. Acc. Chem. Res. 2008, 41, 1842–1851. [Google Scholar] [CrossRef] [PubMed]
- Mocan, T.; Matea, C.T.; Cojocaru, I.; Ilie, I.; Tabaran, F.A.; Zaharie, F.; Iancu, C.; Bartos, D.; Mocan, L. Photothermal treatment of human pancreatic cancer using PEGylated multi-walled carbon nanotubes induces apoptosis by triggering mitochondrial membrane depolarization mechanism. J. Cancer 2014, 5, 679–688. [Google Scholar] [CrossRef] [PubMed]
- Tong, L.; Cheng, J.-X. Gold nanorod-mediated photothermolysis induces apoptosis of macrophages via damage of mitochondria. Nanomedicine 2009, 4, 265–276. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Xu, L.; Wang, C.; Yang, R.; Zhuang, Q.; Han, X.; Dong, Z.; Zhu, W.; Peng, R.; Liu, Z. Near-infrared-triggered photodynamic therapy with multitasking upconversion nanoparticles in combination with checkpoint blockade for immunotherapy of colorectal cancer. ACS Nano 2017, 11, 4463–4474. [Google Scholar] [CrossRef] [PubMed]


© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Riley, R.S.; O’Sullivan, R.K.; Potocny, A.M.; Rosenthal, J.; Day, E.S. Evaluating Nanoshells and a Potent Biladiene Photosensitizer for Dual Photothermal and Photodynamic Therapy of Triple Negative Breast Cancer Cells. Nanomaterials 2018, 8, 658. https://doi.org/10.3390/nano8090658
Riley RS, O’Sullivan RK, Potocny AM, Rosenthal J, Day ES. Evaluating Nanoshells and a Potent Biladiene Photosensitizer for Dual Photothermal and Photodynamic Therapy of Triple Negative Breast Cancer Cells. Nanomaterials. 2018; 8(9):658. https://doi.org/10.3390/nano8090658
Chicago/Turabian StyleRiley, Rachel S., Rachel K. O’Sullivan, Andrea M. Potocny, Joel Rosenthal, and Emily S. Day. 2018. "Evaluating Nanoshells and a Potent Biladiene Photosensitizer for Dual Photothermal and Photodynamic Therapy of Triple Negative Breast Cancer Cells" Nanomaterials 8, no. 9: 658. https://doi.org/10.3390/nano8090658
APA StyleRiley, R. S., O’Sullivan, R. K., Potocny, A. M., Rosenthal, J., & Day, E. S. (2018). Evaluating Nanoshells and a Potent Biladiene Photosensitizer for Dual Photothermal and Photodynamic Therapy of Triple Negative Breast Cancer Cells. Nanomaterials, 8(9), 658. https://doi.org/10.3390/nano8090658





