On the Mechanism of Magnesium Storage in Micro- and Nano-Particulate Tin Battery Electrodes
Abstract
1. Introduction
2. Materials and Methods
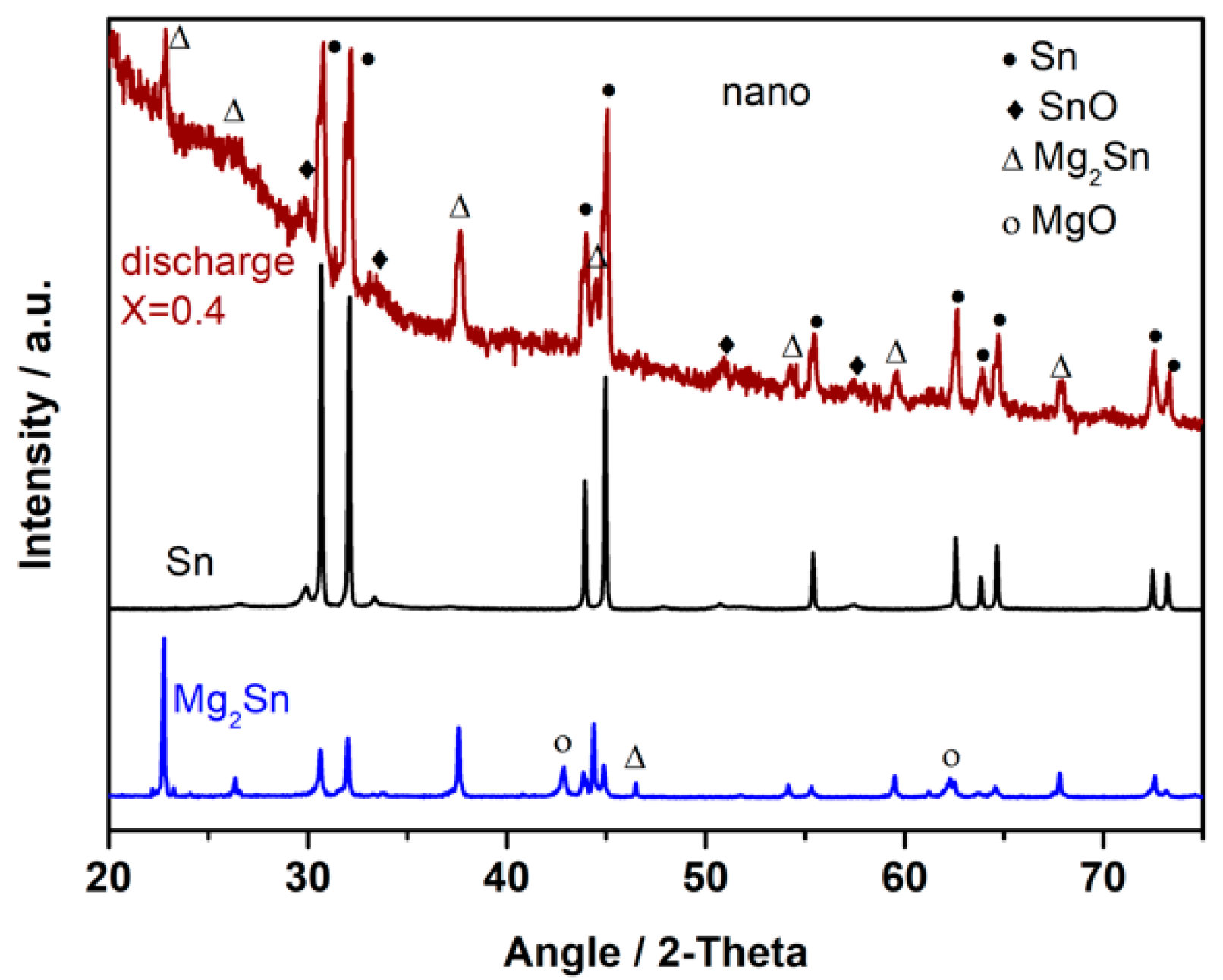
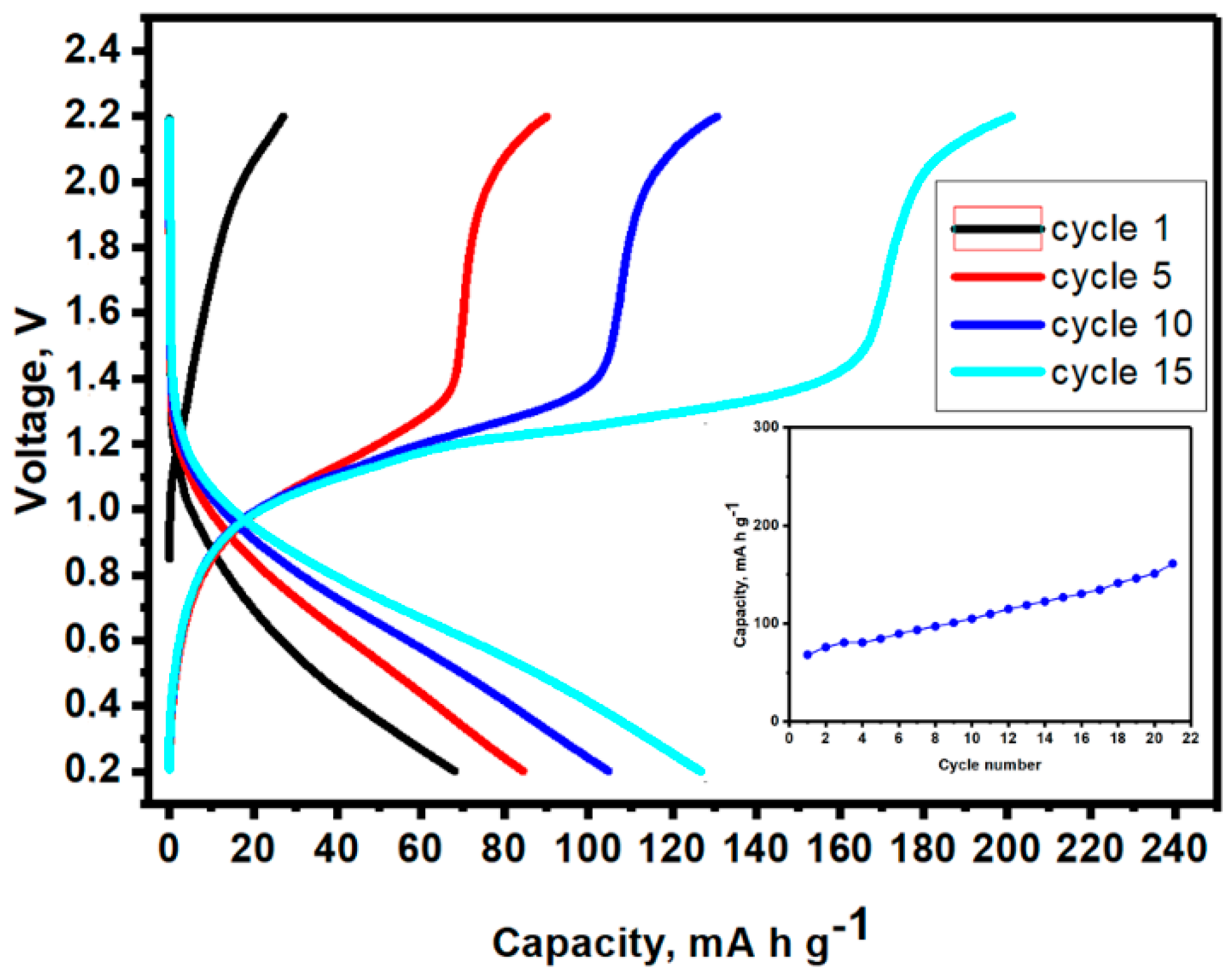
3. Results
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gerst, M.D.; Graedel, T.E. In-Use Stocks of Metals: Status and Implications. Environ. Sci. Technol. 2008, 42, 7038–7044. [Google Scholar] [CrossRef] [PubMed]
- Tarascon, J.M. Is lithium the new gold? Nat. Chem. 2010, 2, 510. [Google Scholar] [CrossRef] [PubMed]
- Wadia, C.; Albertus, P.; Srinivasan, V. Resource constraints on the battery energy storage potential for grid and transportation applications. J. Power Sources 2011, 196, 1593–1598. [Google Scholar] [CrossRef]
- Speirs, J.; Contestabile, M.; Houari, Y.; Gross, R. The future of lithium availability for electric vehicle batteries. Renew. Sustain. Energy Rev. 2014, 35, 183–193. [Google Scholar] [CrossRef]
- Lyon, R.E.; Walters, R.N. Energetics of lithium ion battery failure. J. Hazard. Mater. 2016, 318, 164–172. [Google Scholar] [CrossRef] [PubMed]
- Novák, P.; Imhof, R.; Haas, O. Magnesium insertion electrodes for rechargeable nonaqueous batteries—A competitive alternative to lithium? Electrochim. Acta 1999, 45, 351–367. [Google Scholar] [CrossRef]
- Aurbach, D.; Lu, Z.; Schechter, A.; Gofer, Y.; Gizbar, H.; Turgeman, R.; Cohen, Y.; Moshkovich, M.; Levi, E. Prototype systems for rechargeable magnesium batteries. Nature 2000, 407, 724–727. [Google Scholar] [CrossRef] [PubMed]
- Saha, P.; Datta, M.K.; Velikokhatnyi, O.I.; Manivannan, A.; Alman, D.; Kumta, P.N. Rechargeable magnesium battery: Current status and key challenges for the future. Prog. Mater. Sci. 2014, 66, 1–86. [Google Scholar] [CrossRef]
- Zhao-Karger, Z.; Zhao, X.Y.; Wang, D.; Thomas, D.; Behm, R.J.; Fichtner, M. Performance Improvement of Magnesium Sulfur Batteries with Modified Non-Nucleophilic Electrolytes. Adv. Energy Mater. 2015, 5, 1401155. [Google Scholar] [CrossRef]
- Song, J.; Sahadeo, E.; Noked, M.; Lee, S.B. Mapping the Challenges of Magnesium Battery. J. Phys. Chem. Lett. 2016, 7, 1736–1749. [Google Scholar] [CrossRef] [PubMed]
- Jia, X.; Wang, C.; Ranganathan, V.; Napier, B.; Yu, C.; Chao, Y.; Forsyth, M.; Omenetto, F.G.; MacFarlane, D.R.; Wallace, G.G. A Biodegradable Thin-Film Magnesium Primary Battery Using Silk Fibroin–Ionic Liquid Polymer Electrolyte. ACS Energy Lett. 2017, 2, 831–836. [Google Scholar] [CrossRef]
- Muldoon, J.; Bucur, C.B.; Gregory, T. Quest for Nonaqueous Multivalent Secondary Batteries: Magnesium and Beyond. Chem. Rev. 2014, 114, 11683–11720. [Google Scholar] [CrossRef] [PubMed]
- Canepa, P.; Gautam, G.S.; Hannah, D.C.; Malik, R.; Liu, M.; Gallagher, K.G.; Persson, K.A.; Ceder, G. Odyssey of Multivalent Cathode Materials: Open Questions and Future Challenges. Chem. Rev. 2017, 117, 4287–4341. [Google Scholar] [CrossRef] [PubMed]
- Okamoto, S.; Ichitsubo, T.; Kawaguchi, T.; Kumagai, Y.; Oba, F.; Yagi, S.; Shimokawa, K.; Goto, N.; Doi, T.; Matsubara, E. Intercalation and Push-Out Process with Spinel-to-Rocksalt Transition on Mg Insertion into Spinel Oxides in Magnesium Batteries. Adv. Sci. 2015, 2, 1500072. [Google Scholar] [CrossRef] [PubMed]
- Knight, J.C.; Therese, S.; Manthiram, A. On the Utility of Spinel Oxide Hosts for Magnesium-Ion Batteries. ACS Appl. Mater. Interfaces 2015, 7, 22953–22961. [Google Scholar] [CrossRef] [PubMed]
- Cabello, M.; Alcántara, R.; Nacimiento, F.; Ortiz, G.; Lavela, P.; Tirado, J.L. Electrochemical and chemical insertion/deinsertion of magnesium in spinel-type MgMn2O4 and lambda-MnO2 for both aqueous and non-aqueous magnesium-ion batteries. CrystEngComm 2015, 17, 8728–8735. [Google Scholar] [CrossRef]
- Singh, N.; Arthur, T.S.; Ling, C.; Matsui, M.; Mizuno, F. A high energy-density tin anode for rechargeable magnesium-ion batteries. Chem. Commun. 2013, 49, 149–151. [Google Scholar] [CrossRef] [PubMed]
- Courtney, I.A.; Dunlap, R.A.; Dahn, J.R. In-situ 119Sn Mössbauer effect studies of the reaction of lithium with SnO and SnO:0.25 B2O3:0.25 P2O5 glass. Electrochim. Acta 1999, 45, 51–58. [Google Scholar] [CrossRef]
- Fernández-Madrigal, F.J.; Lavela, P.; Pérez-Vicente, C.; Tirado, J.L.; Jumas, J.C.; Olivier-Fourcade, J. X-ray Diffraction, 7Li MAS NMR Spectroscopy, and 119Sn Mössbauer Spectroscopy Study of SnSb-Based Electrode Materials. Chem. Mater. 2002, 14, 2962–2968. [Google Scholar] [CrossRef]
- Alcántara, R.; Ortiz, G.F.; Lavela, P.; Tirado, J.L. Electrochemical and 119Sn Mössbauer studies of the reaction of Co2SnO4 with lithium. Electrochem. Commun. 2006, 8, 731–736. [Google Scholar]
- Aldon, L.; Ionica, C.M.; Lippens, P.E.; Larcher, D.; Tarascon, J.M.; Olivier-Fourcade, J.; Jumas, J.C. In situ 119Sn Mössbauer spectroscopy used to study lithium insertion in c-Mg2Sn. Hyperfine Interact. 2006, 167, 729–732. [Google Scholar] [CrossRef]
- Alcántara, R.; Rodríguez, I.; Tirado, J.L. Structural and Electrochemical Properties of Micro-and Nano-Crystalline CoSn Electrode Materials. ChemPhysChem 2008, 9, 1171–1177. [Google Scholar] [CrossRef] [PubMed]
- Gogotsi, Y.; Penner, R.M. Energy Storage in Nanomaterials − Capacitive, Pseudocapacitive, or Battery-like? ACS Nano 2018, 12, 2081–2083. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Lee, J.; Xiang, G.; Glass, H.F.J.; Keyzer, E.N.; Dutton, S.E.; Grey, C.P. Insights into the Electrochemical Performances of Bi Anodes for Mg-Ion Batteries Using 25Mg Solid State NMR Spectroscopy. Chem. Commun. 2018, 4, 743–746. [Google Scholar] [CrossRef] [PubMed]
- Kapitanov, E.V.; Pakovl, E.K. Mössbauer Study of Phase Transitions (I) The Phase Transition of Mg2Sn. Phys. State Solids A 1979, 53, 473–480. [Google Scholar] [CrossRef]
- Sirkin, H.; Mingolo, E.; Nassif, N.; Arcondo, B. Increase of the glass-forming composition range of Mg-based binary alloys by addition of tin. J. Non-Cryst. Solids 1987, 93, 323–330. [Google Scholar] [CrossRef]
- Fries, S.G.; Lukas, H.L. Optimisation of the Mg-Sn System. J. Chim. Phys. 1993, 90, 181–187. [Google Scholar] [CrossRef]
- Chouvin, J.; Olivier-Fourcade, J.; Jumas, J.C.; Simon, B.; Biensan, P.; Fernández Madrigal, F.J.; Tirado, J.L.; Pérez-Vicente, C. SnO reduction in lithium cells: Study by X-ray absorption, 119Sn Mössbauer spectroscopy and X-ray diffraction. J. Electroanal. Chem. 2000, 494, 136–146. [Google Scholar] [CrossRef]
- Stockler, H.A.; Sano, H. Mössbauer recoilless fraction in tin compounds. Nucl. Instrum. Meth. 1966, 44, 103–108. [Google Scholar] [CrossRef]
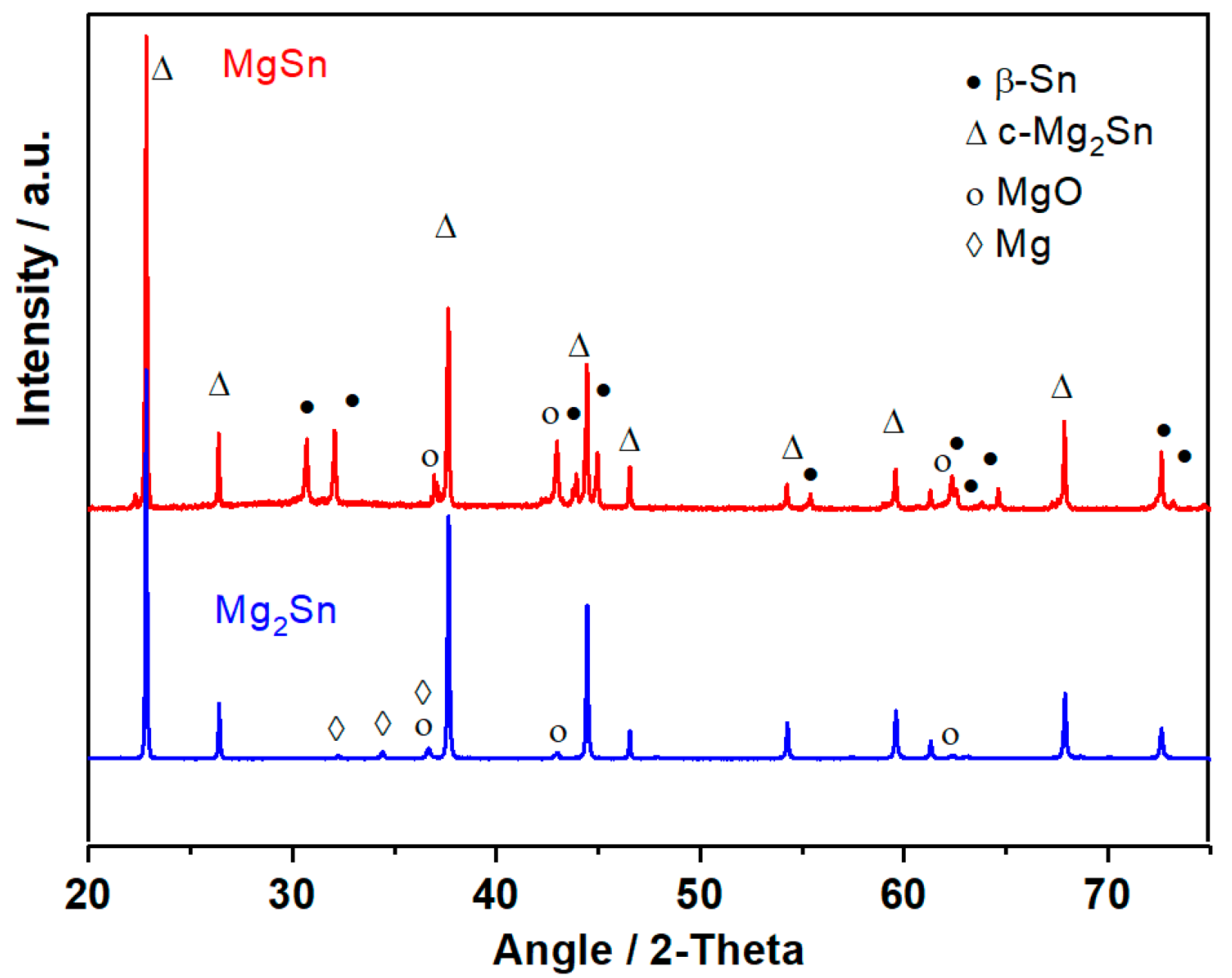
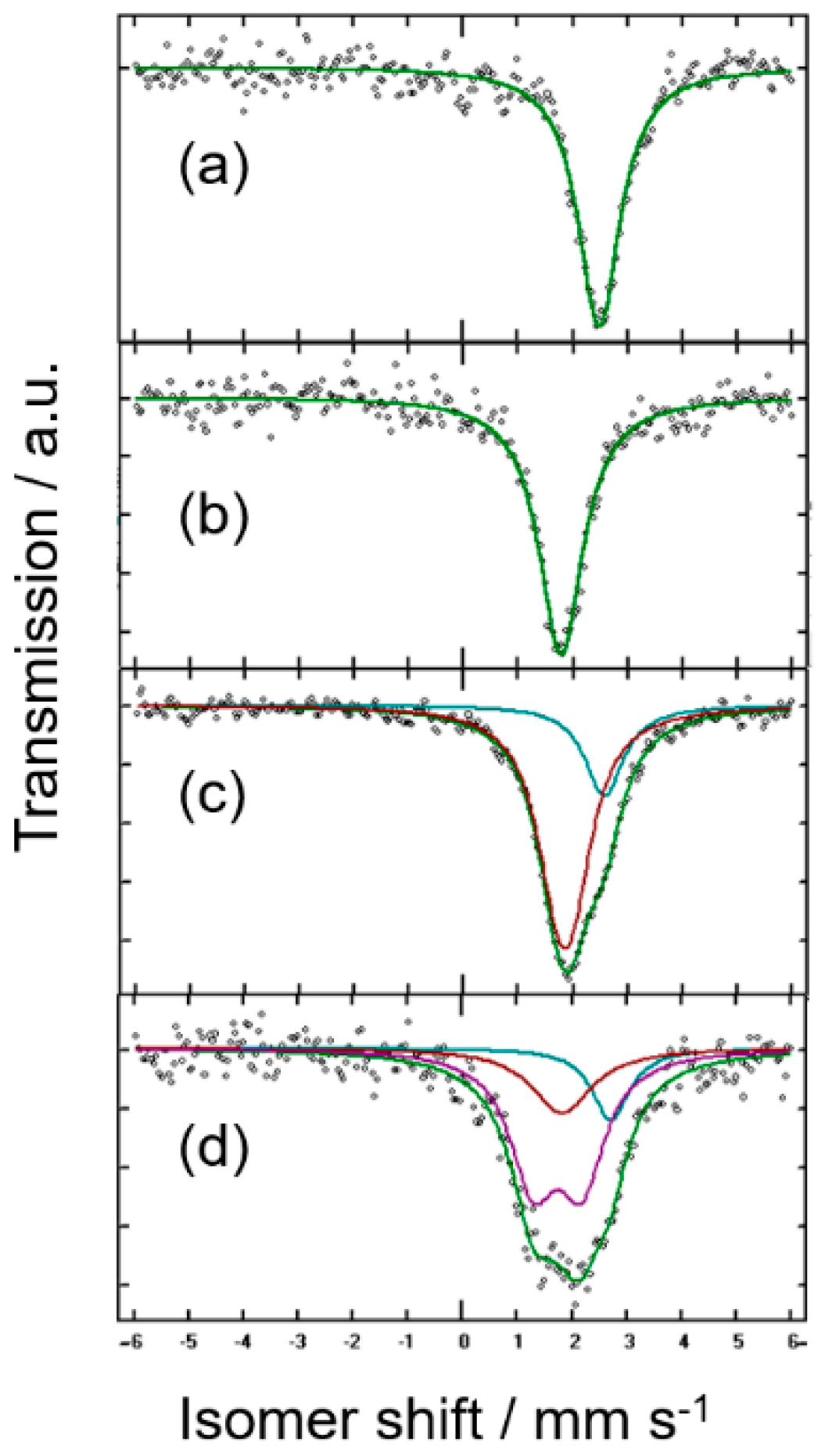
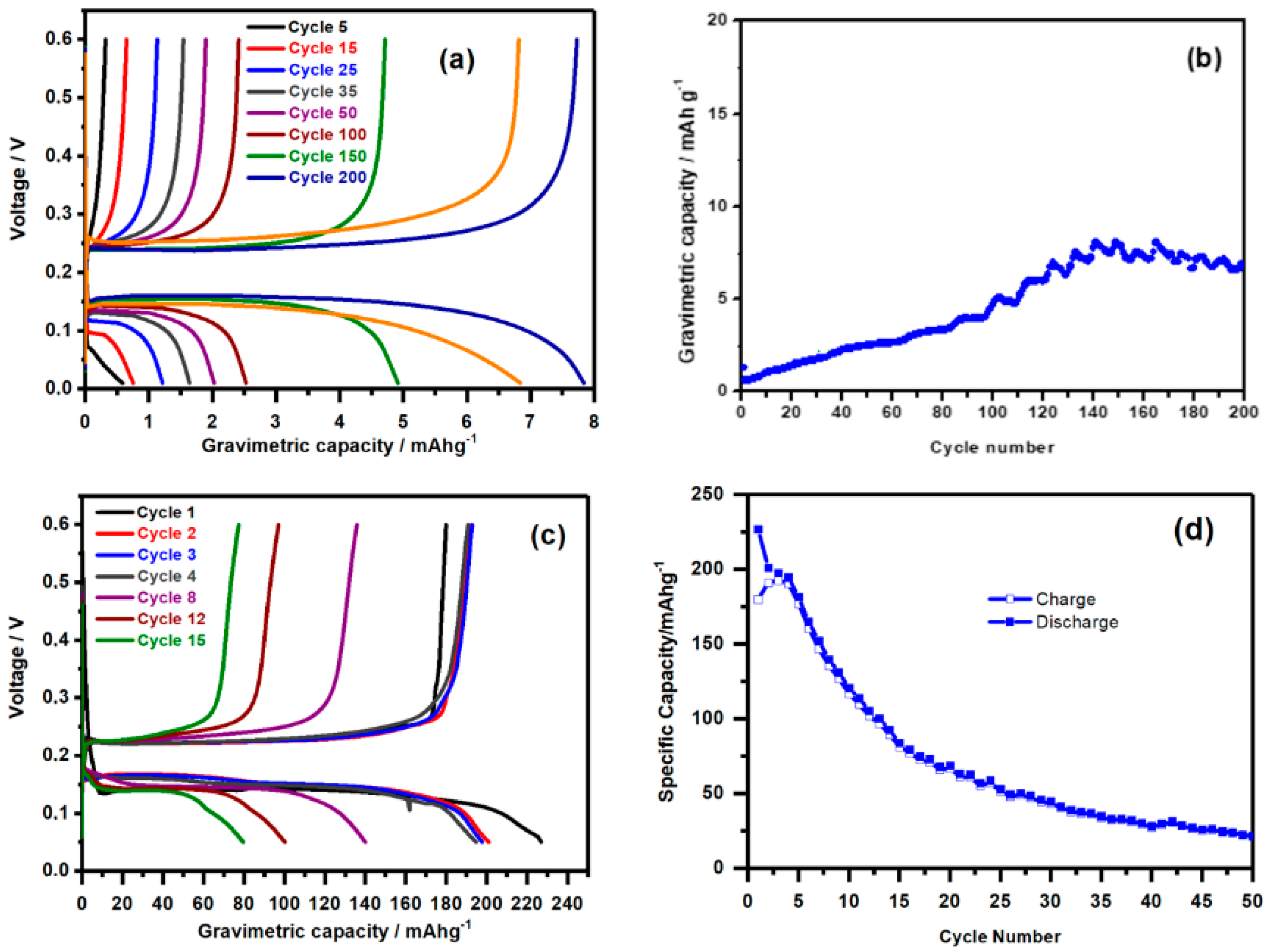
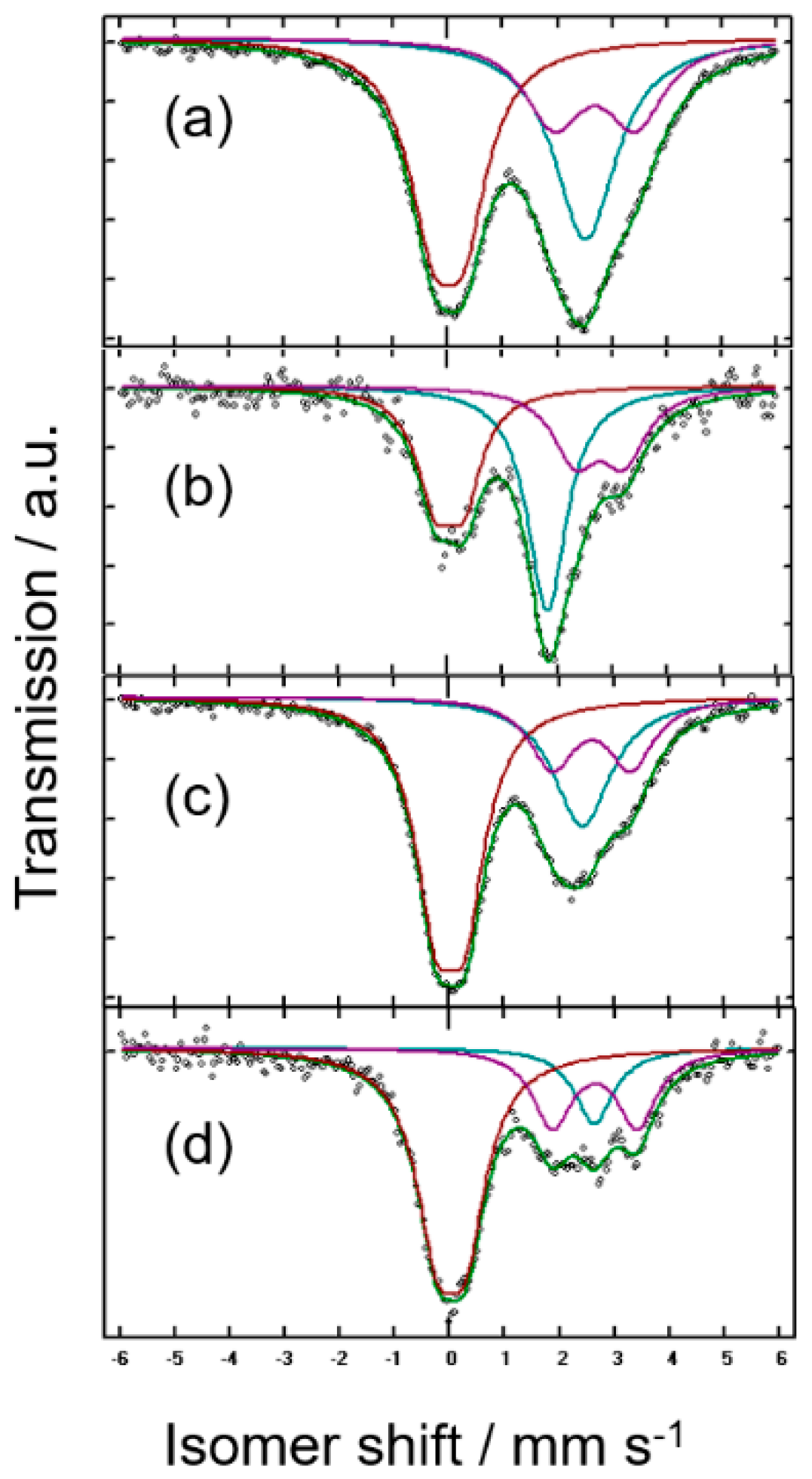

| Sample (Nominal) | IS/mms−1 | QS/mms−1 | LW/mms−1 | % | %corr * | Attribution |
|---|---|---|---|---|---|---|
| (a) micro-Sn | 2.5619 | - | 0.953 | 100 | 100 | β-Sn |
| (b) thermal-Mg2Sn | 1.8479 | - | 0.993 | 100 | 100 | c-Mg2Sn |
| (c) thermal-MgSn | 2.643 | - | 0.877 | 24 | 49 | β-Sn |
| 1.913 | 0.304 | 0.964 | 76 | 51 | Mg2-δSn | |
| (d) micro-Mg0.4Sn | 2.798 | - | 1.831 | 15 | 38 | β-Sn |
| discharged | 1.82 | - | 1.45 | 21 | 9 | c-Mg2Sn |
| 1.795 | 0.9082 | 1.0512 | 64 | 53 | Mg2−δSn |
| Sample (Nominal) | IS/mms−1 | QS/mms−1 | LW/mms−1 | % | %corr * | Attribution |
|---|---|---|---|---|---|---|
| (a) nano-Sn | 2.5798 | - | 1.419 | 33 | 76 | β-Sn |
| 2.753 | 1.525 | 1.122 | 25 | 2 | SnO | |
| 0.0267 | 1.581 | 1.165 | 42 | 13 | SnO2 | |
| (b) nano-Mg1.3Sn | 1.871 | - | 0.884 | 40 | 50 | c-Mg2Sn |
| discharged | 2.856 | 0.906 | 1.01 | 27 | 29 | SnO |
| 0.052 | 0.533 | 0.836 | 33 | 21 | SnO2 | |
| (c) thermal-MgSn | 2.672 | - | 1.043 | 14 | 60 | d-MgδSn |
| discharged + charged | 1.903 | - | 1.044 | 9 | 7 | c-Mg2Sn |
| 2.812 | 1.223 | 1.041 | 23 | 14 | SnO | |
| 0.051 | 0.551 | 0.942 | 54 | 19 | SnO2 | |
| (d) micro-Mg0.4Sn | 2.708 | - | 0.874 | 15 | 60 | d-MgδSn |
| discharged + fully | 2.712 | 1.573 | 0.877 | 26 | 17 | SnO |
| charged | 0.051 | 0.542 | 0.963 | 61 | 23 | SnO2 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nacimiento, F.; Cabello, M.; Pérez-Vicente, C.; Alcántara, R.; Lavela, P.; Ortiz, G.F.; Tirado, J.L. On the Mechanism of Magnesium Storage in Micro- and Nano-Particulate Tin Battery Electrodes. Nanomaterials 2018, 8, 501. https://doi.org/10.3390/nano8070501
Nacimiento F, Cabello M, Pérez-Vicente C, Alcántara R, Lavela P, Ortiz GF, Tirado JL. On the Mechanism of Magnesium Storage in Micro- and Nano-Particulate Tin Battery Electrodes. Nanomaterials. 2018; 8(7):501. https://doi.org/10.3390/nano8070501
Chicago/Turabian StyleNacimiento, Francisco, Marta Cabello, Carlos Pérez-Vicente, Ricardo Alcántara, Pedro Lavela, Gregorio F. Ortiz, and José L. Tirado. 2018. "On the Mechanism of Magnesium Storage in Micro- and Nano-Particulate Tin Battery Electrodes" Nanomaterials 8, no. 7: 501. https://doi.org/10.3390/nano8070501
APA StyleNacimiento, F., Cabello, M., Pérez-Vicente, C., Alcántara, R., Lavela, P., Ortiz, G. F., & Tirado, J. L. (2018). On the Mechanism of Magnesium Storage in Micro- and Nano-Particulate Tin Battery Electrodes. Nanomaterials, 8(7), 501. https://doi.org/10.3390/nano8070501








