Enhanced Catalytic Reduction of 4-Nitrophenol Driven by Fe3O4-Au Magnetic Nanocomposite Interface Engineering: From Facile Preparation to Recyclable Application
Abstract
1. Introduction
2. Experimental Section
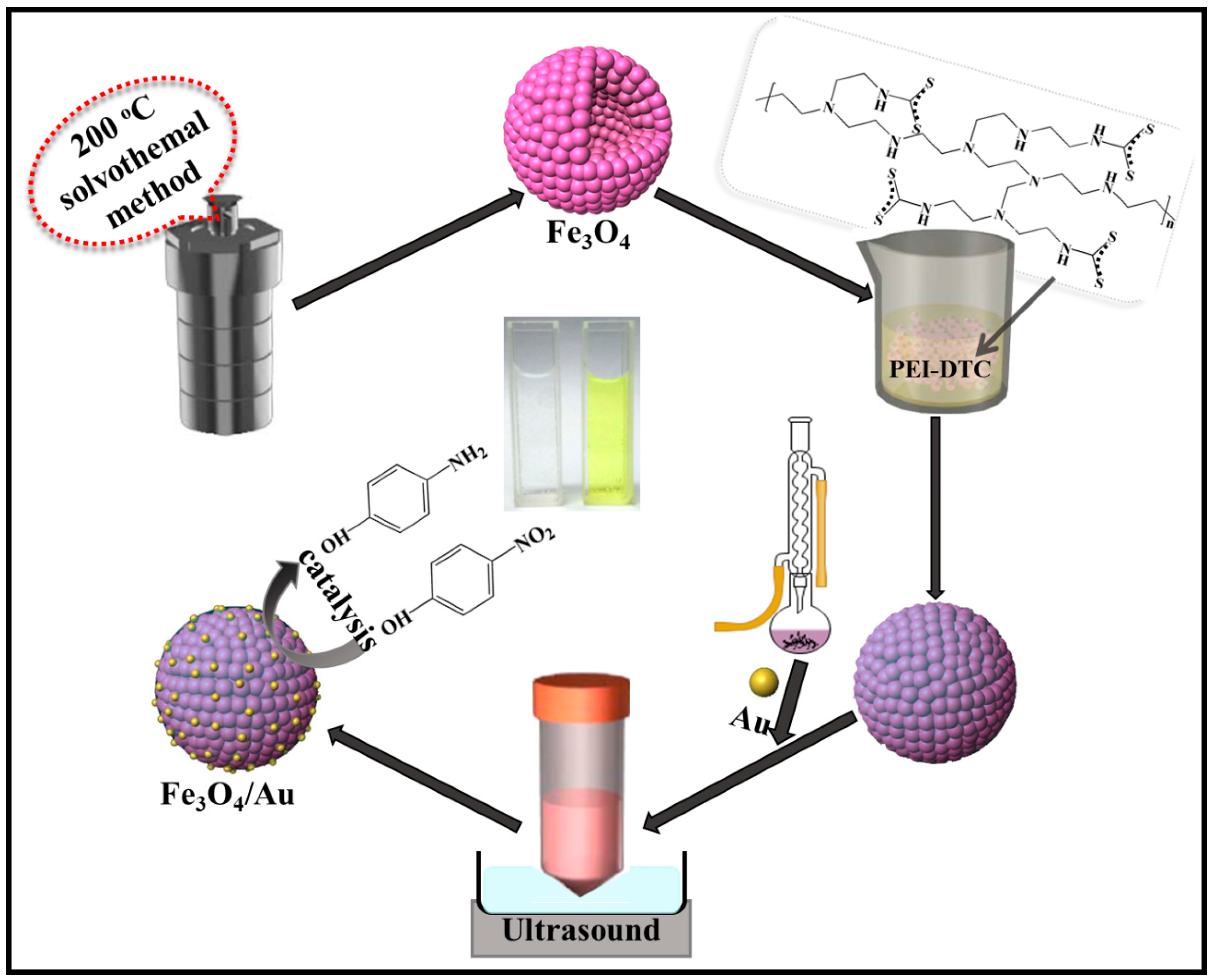
2.1. Materials’ Development
2.2. Synthesis of Fe3O4 Hollow Microspheres
2.3. Gold Seeds Synthesis
2.4. PEI-DTC Synthesis
2.5. Fe3O4@PEI-DTC-Au Seeds’ Synthesis
2.6. Application of Fe3O4-Au Magnetic Nanocomposites for Catalytic Reduction of 4-NP
2.7. Characterizations
3. Results and Discussion
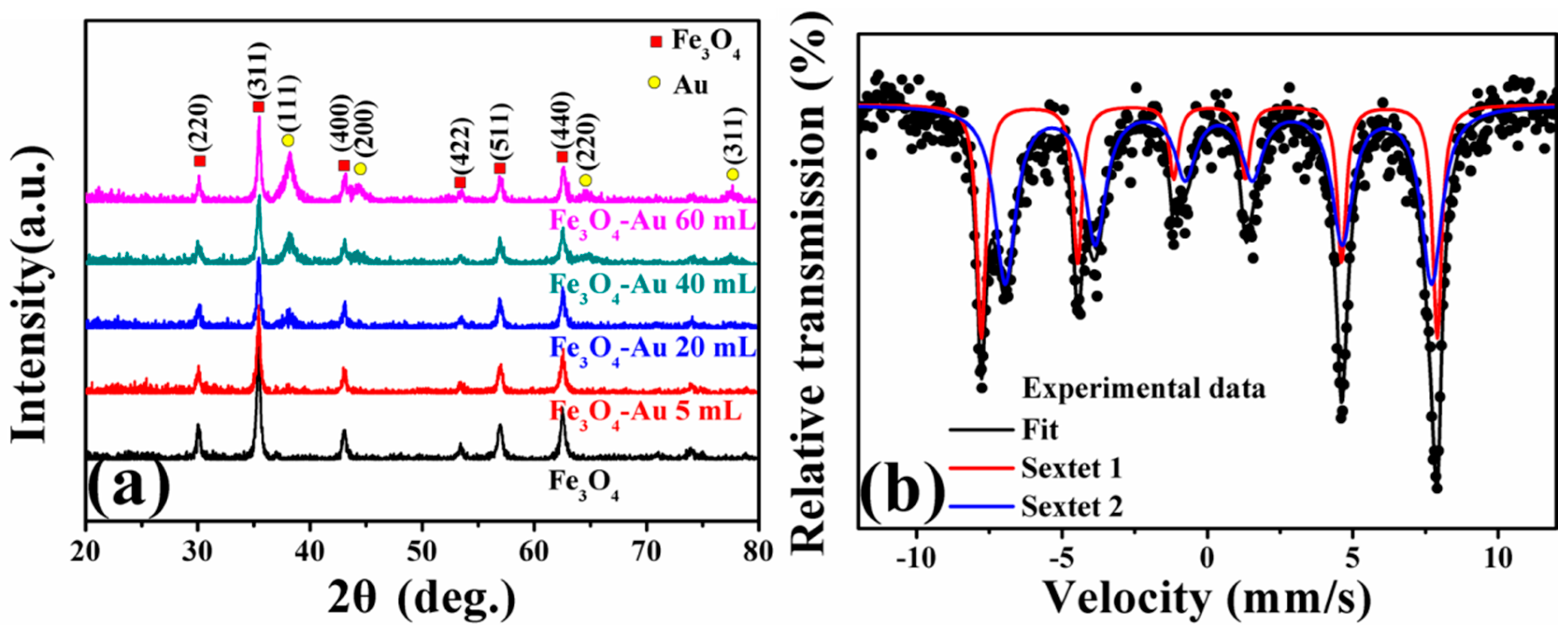
3.1. X-ray Diffraction of the Fe3O4-Au Magnetic Nanocomposites
3.2. Morphology of the Fe3O4-Au Magnetic Nanocomposites
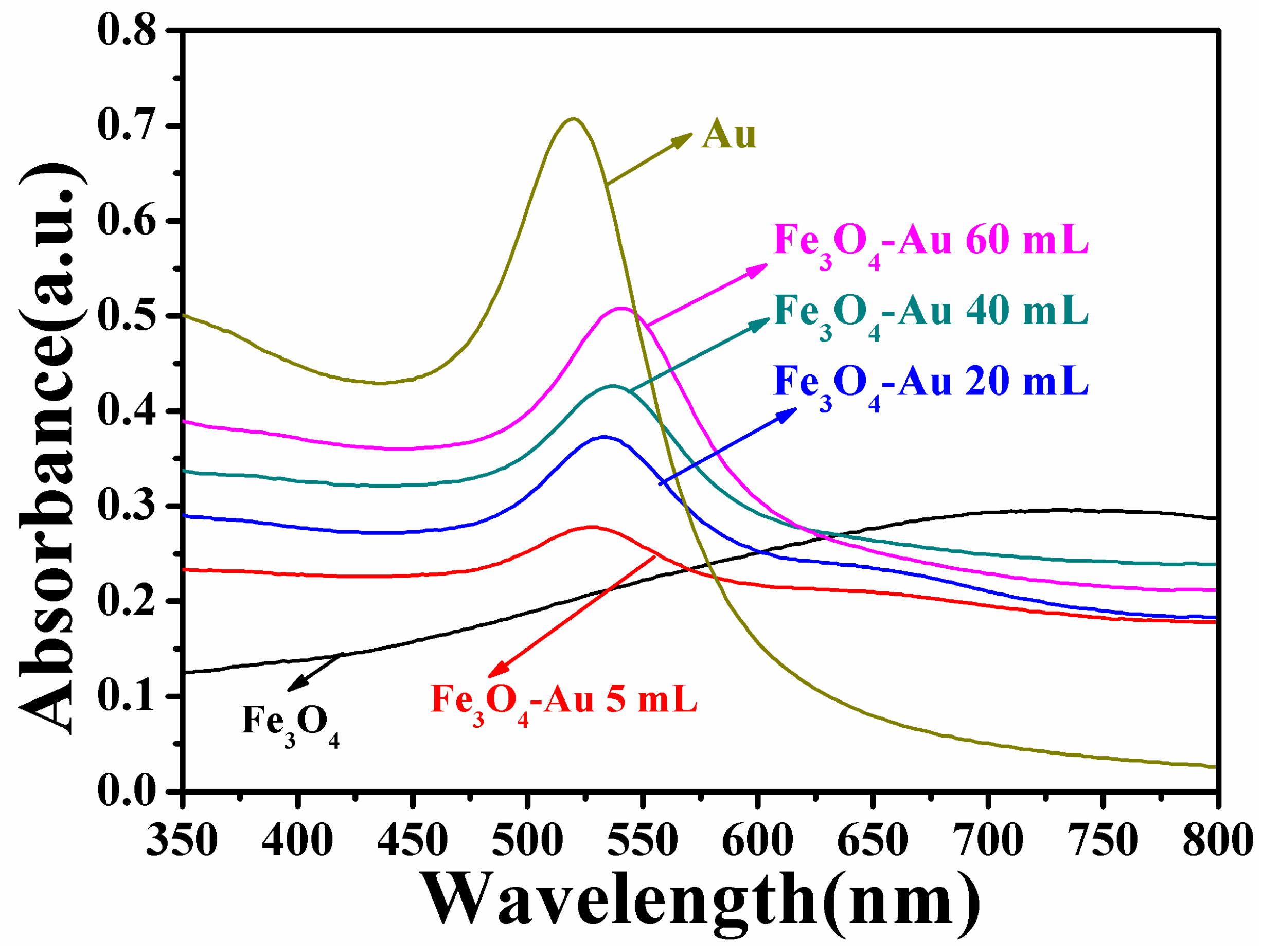
3.3. Optical Properties of the Fe3O4-Au Magnetic Nanocomposites
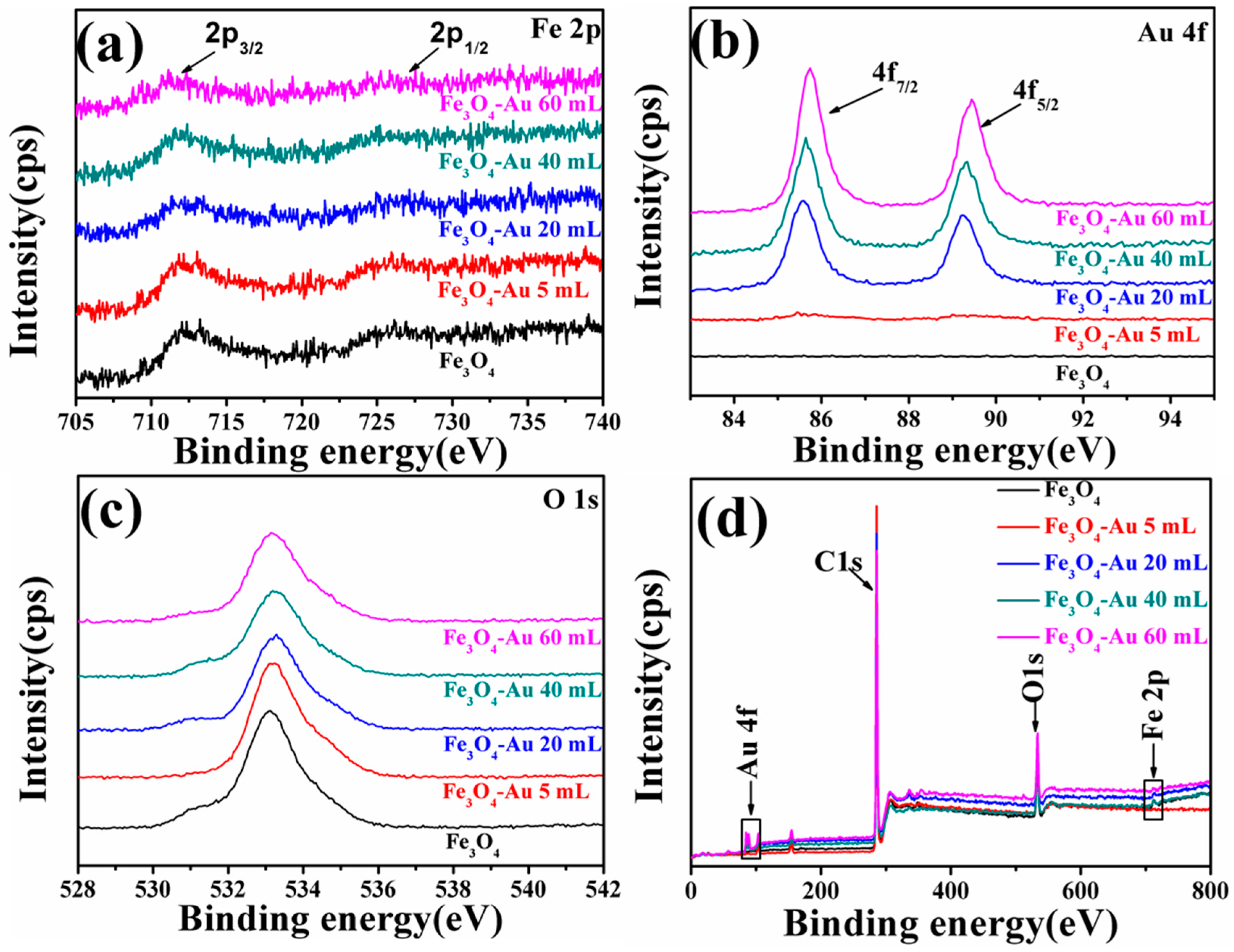
3.4. XPS of the Fe3O4-Au Magnetic Nanocomposites
3.5. Magnetic Properties of the Fe3O4-Au Magnetic Nanocomposites
3.6. Catalytic Activity of Fe3O4-Au Magnetic Nanocomposites to 4-NP
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Zhang, X.F.; Zhu, X.Y.; Feng, J.J.; Wang, A.J. Solvothermal synthesis of N-doped graphene supported PtCo nanodendrites with highly catalytic activity for 4-nitrophenol reduction. Appl. Surf. Sci. 2018, 428, 798–808. [Google Scholar] [CrossRef]
- Bordbar, M.; Negahdar, N.; Nasrollahzadeh, M. Melissa Officinalis L. leaf extract assisted green synthesis of CuO/ZnO nanocomposite for the reduction of 4-nitrophenol and Rhodamine B. Sep. Purif. Technol. 2018, 191, s295–s300. [Google Scholar] [CrossRef]
- Nag, S.; Pramanik, A.; Chattopadhyay, D.; Bhattacharyya, M. Green-fabrication of gold nanomaterials using Staphylococcus warneri from Sundarbans estuary: An effective recyclable nanocatalyst for degrading nitro aromatic pollutants. Environ. Sci. Pollut. Res. 2017, 24, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Meziane, D.; Benadda-Kordjani, A.; Nezzal, G.; Benammar, S.; Djadoun, A. Para-nitrophenol reduction on solvothermally prepared cobalt@silica core-shell catalysts. React. Kinet. Mech. Catal. 2017, 122, 1145–1158. [Google Scholar] [CrossRef]
- He, L.L.; Tong, Z.F.; Wang, Z.H.; Chen, M.; Huang, N.; Zhang, W. Effects of calcination temperature and heating rate on the photocatalytic properties of ZnO prepared by pyrolysis. J. Colloid Interface Sci. 2017, 509, 448–456. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Yang, W.; Sun, W.; Li, Q.; Shang, J.K. Creation of Cu2O@TiO2 composite photocatalysts with p-n heterojunctions formed on exposed Cu2O facets, their energy band alignment study, and their enhanced photocatalytic activity under visible light illumination. ACS Appl. Mater. Interfaces 2015, 7, 1465–1476. [Google Scholar] [CrossRef] [PubMed]
- Andreou, D.; Iordanidou, D.; Tamiolakis, I.; Armatas, G.S.; Lykakis, I.N. Reduction of nitroarenes into aryl amines and n-aryl hydroxylamines via activation of NaBH4 and ammonia-borane complexes by Ag/TiO2 catalyst. Nanomaterials 2016, 6, 54. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Wang, Y.; Feng, W.H.; Lei, H.T.; Li, J. Preparation of Cu(II) porphyrin-TiO2 composite in one-pot method and research on photocatalytic property. RSC Adv. 2017, 7, 52738–52746. [Google Scholar] [CrossRef]
- Liu, Y.; Zhang, Y.Y.; Kou, Q.W.; Chen, Y.; Han, D.L.; Wang, D.D.; Lu, Z.Y.; Chen, L.; Yang, J.H.; Xing, S. Eco-friendly seeded Fe3O4-Ag nanocrystals: A new type of highly efficient and low cost catalyst for methylene blue reduction. RSC Adv. 2018, 8, 2209–2218. [Google Scholar] [CrossRef]
- Shah, M.T.; Balouch, A.; Sirajuddin; Pathan, A.A.; Abdullah; Mahar, A.M.; Sabir, S.; Khattak, R.; Umar, A.A. SiO2 caped Fe3O4 nanostructures as an active heterogeneous catalyst for 4-nitrophenol reduction. Microsyst. Technol. 2017, 23, 5745–5758. [Google Scholar] [CrossRef]
- Svedendahl, M.; Verre, R.; Kall, M. Refractometric Biosensing Based on Optical Phase Flips in Sparse and Short-Range-Ordered Nanoplasmonic Layers. Light Sci. Appl. 2014, 3, e220. [Google Scholar] [CrossRef]
- Zhu, Z.; Bai, B.; You, O.; Li, Q.; Fan, S. Fano resonance boosted cascaded optical field enhancement in a plasmonic nanoparticle-in-cavity nanoantenna array and its SERS application. Light Sci. Appl. 2015, 4, e296. [Google Scholar] [CrossRef]
- Wang, P.; Wang, Y.; Tong, L. Graphene-doped polymer nanofibers for low-threshold nonlinear optical waveguiding. Light Sci. Appl. 2013, 2, e102. [Google Scholar] [CrossRef]
- Li, X.; Xing, L.; Zheng, K.; Wei, P.; Du, L.; Shen, M.; Shi, X. Formation of gold nanostar-coated hollow mesoporous silica for tumor multimodality imaging and photothermal therapy. ACS Appl. Mater. Interfaces 2017, 9, 5817–5827. [Google Scholar] [CrossRef] [PubMed]
- Aslam, U.; Chavez, S.; Linic, S. Controlling energy flow in multimetallic nanostructures for plasmonic catalysis. Nat Nano 2017, 12, 1000–1005. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Wang, H. Facet-Dependent Catalytic Activities of Au Nanoparticles Enclosed by High-Index Facets. ACS Catal. 2014, 4, 4027–4033. [Google Scholar] [CrossRef]
- Karabchevsky, A.; Mosayyebi, A.; Kavokin, A.V. Tuning the chemiluminescence of a luminol flow using plasmonic nanoparticles. Light Sci. Appl. 2016, 5, 1–7. [Google Scholar] [CrossRef]
- Linnenbank, H.; Grynko, Y.; Forstner, J.; Linden, S. Second harmonic generation spectroscopy on hybrid plasmonic/dielectric nanoantennas. Light Sci. Appl. 2016, 5, e16013. [Google Scholar] [CrossRef]
- Blum, O.; Shaked, N.T. Prediction of photothermal phase signatures from arbitrary plasmonic nanoparticles and experimental verification. Light Sci. Appl. 2015, 4, e322. [Google Scholar] [CrossRef]
- Gangarapu, M.; Sarangapany, S.; Veerabhali, K.K.; Devipriya, S.P.; Arava, V.B.R. A high-performance catalytic and recyclability of phyto-synthesized silver nanoparticles embedded in natural polymer. J. Clust. Sci. 2017, 28, 3127–3138. [Google Scholar] [CrossRef]
- Jin, C.J.; Qu, Y.; Wang, M.G.; Han, J.; Hu, Y.M.; Guo, R. Aqueous solution-based Fe3O4 seed-mediated route to hydrophilic Fe3O4-Au janus nanoparticles. Langmuir 2016, 32, 4595–4601. [Google Scholar] [CrossRef] [PubMed]
- Navalón, S.; García, H. Nanoparticles for Catalysis. Nanomaterials 2016, 6, 123. [Google Scholar] [CrossRef] [PubMed]
- Maham, M.; Nasrollahzadeh, M.; Sajadi, S.M.; Nekoei, M. Biosynthesis of Ag/reduced graphene oxide/Fe3O4 using Lotus Garcinii leaf extract and its application as a recyclable nanocatalyst for the reduction of 4-nitrophenol and organic dyes. J. Colloid Interface Sci. 2017, 497, 33–42. [Google Scholar] [CrossRef] [PubMed]
- Jia, L.; Zhou, T.; Xu, J.; Li, F.H.; Xu, Z.Q.; Zhang, B.B.; Guo, S.L.; Shen, X.K.; Zhang, W.S. AuPd bimetallic nanocrystals embedded in magnetic halloysite nanotubes: Facile synthesis and catalytic reduction of nitroaromatic compounds. Nanomaterials 2017, 7, 333. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Z.Y.; Zhai, S.R.; Ma, X.P.; Zhao, Z.Y.; Wang, X.; Bai, H.; An, Q.D. Monolithic Cu/C hybrid beads with well developed porosity for reduction of 4-nitrophenol to 4-aminophenol. New J. Chem. 2017, 41, 13230–13234. [Google Scholar] [CrossRef]
- Zhang, K.H.; Wang, C.W.; Rong, Z.; Xiao, R.; Zhou, Z.; Wang, S.Q. Silver coated magnetic microflowers as an efficient and recyclable Catalyst for catalytic reduction. New J. Chem. 2017, 41, 14199–141208. [Google Scholar] [CrossRef]
- Sun, J.; Chen, L. Superparamagnetic POT/Fe3O4 nanoparticle composites with supported Au nanoparticles as recyclable high-performance nanocatalysts. Mater. Today Chem. 2017, 5, 43–51. [Google Scholar] [CrossRef]
- Wang, D.M.; Duan, H.C.; Lü, J.H.; Lü, C.L. Fabrication of thermo-responsive polymer functionalized reduced graphene oxide@Fe3O4@Au magnetic nanocomposites for enhanced catalytic applications. J. Mater. Chem. A 2017, 5, 5088–5097. [Google Scholar] [CrossRef]
- Rath, P.C.; Saikia, D.; Mishra, M.; Kao, H.M. Exceptional catalytic performance of ultrafine Cu2O nanoparticles confined in cubic mesoporous carbon for 4-nitrophenol reduction. Appl. Surf. Sci. 2018, 427, 1217–1226. [Google Scholar] [CrossRef]
- Meng, G.H.; Zhang, X.Y.; Liu, C.; Wu, J.N.; Guo, X.H.; Liu, Z.Y. Ag quantum dot/montmorillonite compositeswith fluorescent properties: An efficient catalyst. Res. Chem. Intermed. 2017, 43, 7137–7145. [Google Scholar] [CrossRef]
- Govan, J.; Gun’ko, Y.K. Recent advances in the application of magnetic nanoparticles as a support for homogeneous catalysts. Nanomaterials 2014, 4, 222–241. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Zhang, Y.Y.; Kou, Q.W.; Chen, Y.; Sun, Y.T.; Han, D.L.; Wang, D.D.; Lu, Z.Y.; Chen, L.; Yang, J.H.; Xing, S.G.Z. Highly Efficient, Low-Cost, and Magnetically Recoverable FePt–Ag Nanocatalysts Towards Green Reduction of Organic Dyes. Nanomaterials 2018, 8, 329. [Google Scholar] [CrossRef] [PubMed]
- Wysocka, I.; Kowalska, E.; Trzciński, K.; Łapiński, M.; Nowaczyk, G.; Zielińska-Jurek, A. UV-Vis-Induced Degradation of Phenol over Magnetic Photocatalysts Modified with Pt, Pd, Cu and Au Nanoparticles. Nanomaterials 2018, 2, 28. [Google Scholar] [CrossRef] [PubMed]
- Pardo, I.R.; Pons, M.R.; Heredia, A.A.; Usagre, J.V.; Ribera, A.; Galian, R.E.; Prieto, J.P. Fe3O4@Au@mSiO2 as enhancing nanoplatform for rose bengal photodynamic activity. Nanoscale 2017, 9, 10388–10396. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.S.; Wang, Y.; Xia, H.; Wang, G.; Zhang, Z.Y.; Han, D.D.; Lv, C.; Feng, J.; Sun, H.B. Preparation of Fe3O4-Au-GO nanocomposite for simultaneous treatment of oil/water separation and dye decomposition. Nanoscale 2016, 8, 17451–17457. [Google Scholar] [CrossRef] [PubMed]
- Guo, R.; Jiao, T.F.; Xing, R.R.; Chen, Y.; Guo, W.C.; Zhou, J.X.; Zhang, L.X.; Peng, Q.M. Hierarchical AuNPs-Loaded Fe3O4/Polymers Nanocomposites Constructed by Electrospinning with Enhanced and magnetically recyclable catalytic capacities. Nanomaterials 2017, 7, 317. [Google Scholar] [CrossRef] [PubMed]
- Tedsree, K.; Temnuch, N.; Sriplai, N.; Pinitsoontorn, S. Ag modified Fe3O4@TiO2 magnetic core-shell nanocomposites for photocatalytic degradation of methylene blue. Mater. Today 2017, 4, 6576–6584. [Google Scholar] [CrossRef]
- Yang, J.H.; Kou, Q.W.; Liu, Y.; Wang, D.D.; Lu, Z.Y.; Chen, L.; Zhang, Y.Y.; Wang, Y.X.; Zhang, Y.J.; Han, D.L.; et al. Effects of amount of benzyl ether and reaction time on the shape and magnetic properties of Fe3O4 nanocrystals. Powder Technol. 2017, 319, 53–59. [Google Scholar] [CrossRef]
- Duan, L.F.; Jia, S.S.; Wang, T.H.; Xue, B.; Wang, Y.Q.; Zhao, L.J. Synthesis and Characterization of hollow Fe3O4 submicrospheres by a Simple Solvothermal Synthesis. Met. Mater. Int. 2011, 17, 801–804. [Google Scholar] [CrossRef]
- Hu, Y.; Yang, J.; Wei, P.; Li, J.C.; Ding, L.; Zhang, G.X.; Shi, X.Y.; Shen, M.W. Facile synthesis of hyaluronic acid-modified Fe3O4/Au composite nanoparticles for targeted dual mode MR/CT imaging of tumors. J. Mater. Chem. B 2015, 3, 9098–9108. [Google Scholar] [CrossRef]
- Wang, C.W.; Li, P.; Wang, J.F.; Rong, Z.; Pang, Y.F.; Xu, J.W.; Dong, P.T.; Xiao, R.; Wang, S.Q. Polyethylenimine-interlayered core-shell-satellite 3D magnetic microspheres as versatile SERS substrate. Nanoscale 2015, 7, 18694–18707. [Google Scholar] [CrossRef] [PubMed]
- Yan, F.; Sun, R.Y. Facile Synthesis of Bifunctional Fe3O4/Au Nanocomposite and Their Application in Catalytic Reduction of 4-Nitrophenol. Mater. Res. Bull. 2014, 57, 293–299. [Google Scholar] [CrossRef]
- Liu, Y.; Kou, Q.W.; Wang, D.D.; Chen, L.; Sun, Y.T.; Lu, Z.Y.; Zhang, Y.Y.; Wang, Y.X.; Yang, J.H.; Xing, S. Rational synthesis and tailored optical and magnetic Characteristics of Fe3O4-Au composite nanoparticles. J. Mater. Sci. 2017, 52, 10163–10174. [Google Scholar] [CrossRef]
- Xing, G.Z.; Wang, Y.; Wong, J.I.; Shi, Y.M.; Huang, Z.X.; Li, S.; Yang, H.Y. Hybrid CuO/SnO2 nanocomposites: Towards cost-effective and high performance binder free lithium ion batteries anode materials. Appl. Phys. Lett. 2014, 105, 143905. [Google Scholar] [CrossRef]
- Xing, G.Z.; Wang, D.D.; Cheng, C.J.; He, M.; Li, S.; Wu, T. Emergent ferromagnetism in ZnO/Al2O3 core-shell nanowires: Towards oxide spinterfaces. Appl. Phys. Lett. 2013, 103, 022402. [Google Scholar] [CrossRef]
- Sun, Y.; Li, Q. Research of zinc oxide quantum dot light-emitting diodes based on preparation of chemical solutions. Chin. J. Liq. Cryst. Disp. 2016, 31, 635–642. [Google Scholar] [CrossRef]
- Chen, X.Y.; Tian, Z. Recent progress in terahertz dynamic modulation based on graphene. Chin. Opt. 2017, 10, 86–97. [Google Scholar] [CrossRef]
- Li, T.; Zhang, M.-L.; Wang, F.; Zhang, D.-M.; Wang, G.-P. Fabrication of optical waveguide amplifiers based on bonding-type NaYF4: Er nanoparticles-polymer. Chin. Opt. 2017, 10, 219–225. [Google Scholar] [CrossRef]
- Yang, J.H.; Pan, M.Q.; Shi, R.X.; Yang, L.L.; Wang, J.; Kong, X.W.; Yang, W.Q.; Wang, D.D.; Zhou, Z. Novel Fe3O4 hollow microspheres: Nontemplate hydrothermal synthesis, superparamagnetism and biocompatibility. Nanosci. Nanotech. Let. 2017, 9, 109–117. [Google Scholar] [CrossRef]
- Shah, S.T.; Yehye, W.A.; Saad, O.; Simarani, K.; Chowdhury, Z.Z.; Alhadi, A.A.; Al-Ani, L.A. Surface functionalization of iron oxide nanoparticles with gallic acid as potential antioxidant and antimicrobial agents. Nanomaterials 2017, 7, 306. [Google Scholar] [CrossRef] [PubMed]
- Jin, C.J.; Han, J.; Chu, F.Y.; Wang, X.X.; Guo, R. Fe3O4@PANI hybrid shell as multifunctional support for au nanocatalysts with remarkably improved catalytic performance. Langmuir 2017, 33, 4520–4527. [Google Scholar] [CrossRef] [PubMed]
- Miao, P.; Tang, Y.G.; Wang, L. DNA modified Fe3O4@Au magnetic nanoparticles as selective probes for simultaneous detection of heavy metal ions. ACS Appl. Mater. Interfaces 2017, 9, 3940–3947. [Google Scholar] [CrossRef] [PubMed]
- Baskakov, A.O.; Solov’eva, A.Y.; Ioni, Y.V.; Starchikov, S.S.; Lyubutin, I.S.; Khodos, I.I.; Avilov, A.S.; Gubin, S.P. Magnetic and interface properties of the core-shell Fe3O4/Au nanocomposites. Appl. Surf. Sci. 2017, 422, 638–644. [Google Scholar] [CrossRef]
- Li, X.A.; He, Y.Y.; Sui, H.; He, L. One-step fabrication of dual responsive lignin coated Fe3O4 nanoparticles for efficient removal of cationic and anionic dyes. Nanomaterials 2018, 8, 162. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.T.; Tang, B.T.; Wu, S.L.; Gao, Z.M.; Ju, B.Z.; Teng, X.X.; Zhang, S.F. Controllable 5-sulfosalicylic acid assisted solvothermal synthesis of monodispersed superparamagnetic Fe3O4 nanoclusters with tunable size. J. Magn. Magn. Mater. 2017, 423, 111–117. [Google Scholar] [CrossRef]
- Ghosh, R.; Pradhan, L.; Devi, Y.P.; Meena, S.S.; Tewari, R.; Kumar, A.; Sharma, S.; Gajbhiye, N.S.; Vatsa, R.K.; Pandey, B.N.; et al. Induction heating studies of Fe3O4 magnetic nanoparticles capped with oleic acid and polyethylene glycol for hyperthermia. J. Mater. Chem. 2011, 21, 13388–13398. [Google Scholar] [CrossRef]
- Ayyappan, S.; Thoguluva, R.R.; John, P. Superior thermal stability of polymer capped Fe3O4 magnetic nanoclusters. J. Am. Ceram. Soc. 2018, 101, 483–491. [Google Scholar] [CrossRef]
- Kazeminezhad, I.; Mosivand, S. Phase Transition of Electrooxidized Fe3O4 to γ and α-Fe2O3 Nanoparticles Using Sintering Treatment. Acta Phys. Pol. A 2014, 125, 1210–1214. [Google Scholar] [CrossRef]
- Zhang, M.; Xia, P.; Wang, L.; Zheng, J.; Wang, Y.; Xu, J.; Wang, L. Synthesis and fabrication of CNTs/Fe3O4@Pdop@Au nanocables by a facile approach. RSC Adv. 2014, 4, 44423–44426. [Google Scholar] [CrossRef]
- Matioli, E.; Brinkley, S.; Kelchner, K.M.; Hu, Y.L.; Nakamura, S.; DenBaars, S.; Speck, J.; Weisbuch, C. High-brightness polarized light-emitting diodes. Light Sci. Appl. 2012, 1, 479–482. [Google Scholar] [CrossRef]
- Li, L.; Guo, W.; Yan, Y.; Lee, S.; Wang, T. Label-free super-resolution imaging of adenoviruses by submerged microsphere optical nanoscopy. Light Sci. Appl. 2013, 2, e104. [Google Scholar] [CrossRef]
- Wang, D.D.; Wang, W.L.; Huang, M.Y.; Lek, A.; Lam, J.; Mai, Z.H. Failure mechanism analysis and process improvement on time-dependent dielectric breakdown of Cu/ultra-low-k dielectric based on complementary Raman and FTIR spectroscopy study. AIP Adv. 2014, 4, 107–110. [Google Scholar] [CrossRef]
- Xing, G.; Wang, D.; Yi, J.; Yang, L.; Gao, M.; He, M.; Yang, J.; Ding, J.; Sum, T.C.; Wu, T. Correlated d0 ferromagnetism and photoluminescence in undoped ZnO nanowires. Appl. Phys. Lett. 2010, 96, 112511. [Google Scholar] [CrossRef]
- Cheng, X.; Zhang, J.; Ding, T.; Wei, Z.; Li, H.; Wang, Z. The effect of an electric field on the thermomechanical damage of nodular defects in dielectric multilayer coatings irradiated by nanosecond laser pulses. Light Sci. Appl. 2013, 2, e80. [Google Scholar] [CrossRef]
- Wang, D.D.; Xing, G.Z.; Yan, F.; Yan, Y.S.; Li, S. Ferromagnetic (Mn, N)-codoped ZnO nanopillars array: Experimental and computational insights. Appl. Phys. Lett. 2014, 104, 022412. [Google Scholar] [CrossRef]
- Pincella, F.; Isozaki, K.; Miki, K. A visible light-driven plasmonic photocatalyst. Light Sci. Appl. 2014, 3, 110–118. [Google Scholar] [CrossRef]
- Xing, G.Z.; Fang, X.S.; Zhang, Z.; Wang, D.D.; Huang, X.; Guo, J.; Liao, L.; Zheng, Z.; Xu, H.R.; Yu, T. Ultrathin single-crystal ZnO nanobelts: Ag-catalyzed growth and field emission property. Nanotechnology 2010, 21, 255701. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Qu, S. Absorption spectra and near-electric field enhancement effects of Au- and Ag-Fe3O4 dimers. Appl. Surf. Sci. 2014, 292, 1002–1008. [Google Scholar] [CrossRef]
- Zhang, X.; Song, L.; Cai, L.; Tian, X.; Zhang, Q.; Qi, X.; Zhou, W.; Zhang, N.; Yang, F.; Fan, Q.; et al. Optical visualization and polarized light absorption of the single-wall carbon nanotube to verify intrinsic thermal applications. Light Sci. Appl. 2015, 4, e318. [Google Scholar] [CrossRef]
- Xing, G.Z.; Yi, J.B.; Wang, D.D.; Liao, L.; Yu, T.; Shen, Z.X.; Huan, C.H.A.; Sum, T.C.; Ding, J.; Wu, T. Strong correlation between ferromagnetism and oxygen deficiency in Cr-doped In2O3−δ nanostructures. Phys. Rev. B 2009, 79, 174406. [Google Scholar] [CrossRef]
- Xing, G.Z.; Yi, J.B.; Tao, J.G.; Liu, T.; Wong, L.M.; Zhang, Z.; Li, G.P.; Wang, S.J.; Ding, J.; Sum, T.C.; et al. Comparative study of room-temperature ferromagnetism in cu-doped zno nanowires enhanced by structural inhomogeneity. Adv. Mater. 2008, 20, 3521–3527. [Google Scholar] [CrossRef]
- Tran, V.T.; Zhou, H.J.; Lee, S.; Hong, S.C.; Kim, J.; Jeong, S.; Lee, J. Magnetic-assembly mechanism of superparamagnetoplasmonic nanoparticles on a charged surface. ACS Appl. Mater. Interfaces 2015, 7, 8650–8658. [Google Scholar] [CrossRef] [PubMed]
- Ye, M.; Wei, Z.; Hu, F.; Wang, J.X.; Ge, G.L.; Hu, Z.Y.; Shao, M.W.; Lee, S.; Liu, J. Fast assembling microarrays of superparamagnetic Fe3O4@Au nanoparticle clusters as reproducible substrates for surface-enhanced Raman scattering. Nanoscale 2015, 7, 13427–13437. [Google Scholar] [CrossRef] [PubMed]
- Du, J.J.; Xu, J.W.; Sun, Z.L.; Jing, C.Y. Au nanoparticles grafted on Fe3O4 as effective SERS substrates for label-free detection of the 16 EPA priority polycyclic aromatic hydrocarbons. Anal. Chim. Acta 2016, 915, 81–89. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Pang, S.; He, L.; Nugen, S.R. Highly sensitive and selective detection of nitrite ions using Fe3O4@SiO2/Au magnetic nanoparticles by surface-enhanced raman spectroscopy. Biosens. Bioelectron. 2016, 85, 726–733. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Yu, Z.F.; Zhao, L.Y.; Xue, T. Synthesis and application of homogeneous Fe3O4 core/Au shell nanoparticles with strong SERS effect. RSC Adv. 2016, 6, 10352–10357. [Google Scholar] [CrossRef]
- Jiang, G.M.; Huang, Y.X.; Zhang, S.; Zhu, H.Y.; Wu, Z.B.; Sun, S.H. Controlled synthesis of Au-Fe heterodimer nanoparticles and their conversion into Au Fe3O4 heterostructured nanoparticles. Nanoscale 2016, 8, 17947–17952. [Google Scholar] [CrossRef] [PubMed]
- Freitas, M.C.C.; Couto, M.M.; Barroso, M.F.; Pereira, C.; Delos-Santos-Álvarez, N.; Miranda-Ordieres, A.J.; Lobo-Castañón, M.J.; Delerue-Matos, C. Highly monodisperse Fe3O4@Au superparamagnetic nanoparticles as reproducible platform for genosensing genetically modified organisms. ACS Sens. 2016, 1, 1044–1053. [Google Scholar] [CrossRef]
- Abdullaalmamun, M.; Kusumoto, Y.; Zannat, T.; Horieand, Y.; Manaka, H. Au-ultrathin functionalized core–shell (Fe3O4@Au) monodispersed nanocubes for a combination of magnetic/plasmonic photothermal cancer cell killing. RSC Adv. 2013, 3, 7816–7827. [Google Scholar] [CrossRef]
- Zhao, H.Y.; Liu, S.; He, J.; Pan, C.C.; Li, H.; Zhou, Z.Y.; Ding, Y.; Huo, D.; Hu, Y. Synthesis and application of strawberry-like Fe3O4-Au nanoparticles as CT-MR dual-modality contrast agents in accurate detection of the progressive liver disease. Biomaterials 2015, 51, 194–207. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.Y.; Luo, J.; Fan, Q.; Suzuki, M.; Suzuki, I.S.; Engelhard, M.H.; Lin, Y.H.; Kim, N.; Wang, J.Q.; Zhong, C.J. Monodispersed Core-Shell Fe3O4@Au Nanoparticles. J. Phys. Chem. B 2005, 109, 21593–21601. [Google Scholar] [CrossRef] [PubMed]
- Hong, L.R.; Zhao, J.; Lei, Y.M.; Yuan, R.; Zhuo, Y. Efficient electrochemiluminescence from Ru(bpy)32+ enhanced by three-layer porous Fe3O4@SnO2@Au nanoparticles for label-free and sensitive bioanalysis. Electrochim. Acta 2017, 241, 291–298. [Google Scholar] [CrossRef]
- Wang, Y.; Li, H.; Zhang, J.J.; Yan, X.Y.; Chen, Z.X. Fe3O4 and Au nanoparticles dispersed on graphene support as a highly active catalyst toward the reduction of 4-nitrophenol. Phys. Chem. Chem. Phys. 2016, 18, 615–623. [Google Scholar] [CrossRef] [PubMed]
- Xia, Q.D.; Fu, S.S.; Ren, G.J.; Chai, F.; Jiang, J.J.; Qu, F.Y. Fabrication of Fe3O4@Au hollow spheres with recyclable and efficient catalytic properties. New J. Chem. 2016, 40, 818–824. [Google Scholar] [CrossRef]
- Yu, L.; Zhang, Y.T.; Chen, R.; Zhang, D.H.; Wei, X.H.; Chen, F.; Wang, J.X.; Xu, M.T. A highly sensitive resonance light scattering probe for Alzheimer’s amyloid-β peptide based on Fe3O4@Au composites. Talanta 2015, 131, 475–479. [Google Scholar] [CrossRef] [PubMed]
- Hou, Y.H.; Yuan, H.L.; Chen, H.; Shen, J.H.; Li, L.C. Controlled fabrication and microwave absorbing mechanism of hollow Fe3O4@C microspheres. Sci. China Chem. 2017, 60, 740–747. [Google Scholar] [CrossRef]
- Kwizera, E.A.; Chaffin, E.; Wang, Y.M.; Huang, X.H. Synthesis and properties of magnetic-optical core-shell nanoparticles. RSC Adv. 2017, 7, 17137–17153. [Google Scholar] [CrossRef] [PubMed]
- Mitra, A.; Mohapatra, J.; Meena, S.S.; Tomy, C.V.; Aslam, M. Verwey Transition in Ultrasmallsized Octahedral Fe3O4 Nanoparticles. J. Phys. Chem. C 2012, 118, 19356–19362. [Google Scholar] [CrossRef]
- Zhu, Y.M.; Zhou, X.W.; Chen, D.S.; Li, F.; Xue, T.; Farag, A.S. Ternary Fe3O4@PANI@Au nanocomposites as a magnetic catalyst for degradation of organic dyes. Sci. China Technol. Sci. 2017, 60, 749–757. [Google Scholar] [CrossRef]
- Zhang, X.; Ding, S.N. Sandwich-structured electrogenerated chemiluminescence immunosensor based on dual-stabilizers-capped CdTe quantum dots as signal probes and Fe3O4-Au nanocomposites as magnetic separable carriers. Sensor. Actuators B Chem. 2017, 240, 1123–1133. [Google Scholar] [CrossRef]
- Hu, R.; Zheng, M.X.; Wu, J.C.; Li, C.; Shen, D.Q.; Yang, D.; Li, L.; Ge, M.F.; Chang, Z.M.; Dong, W.F. Core-shell magnetic gold nanoparticles for magnetic field-enhanced radio-photothermal therapy in cervical cancer. Nanomaterials 2017, 7, 111. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.Y.; Su, R.N.; Wang, D.; Wang, J.X.; Pu, Y.; Chen, J.F. Sulfurized graphene as efficient metal-free catalysts for reduction of 4-nitrophenol to 4-aminophenol. Ind. Eng. Chem. Res. 2017, 56, 13610–13617. [Google Scholar] [CrossRef]
- Liu, X.; Li, Y.; Xing, Z.; Zhao, X.H.; Liu, N.N.; Chen, F.Y. Monolithic carbon foam-supported Au nanoparticles with excellent catalytic performance in a fixed-bed system. New J. Chem. 2017, 41, 15027–15032. [Google Scholar] [CrossRef]


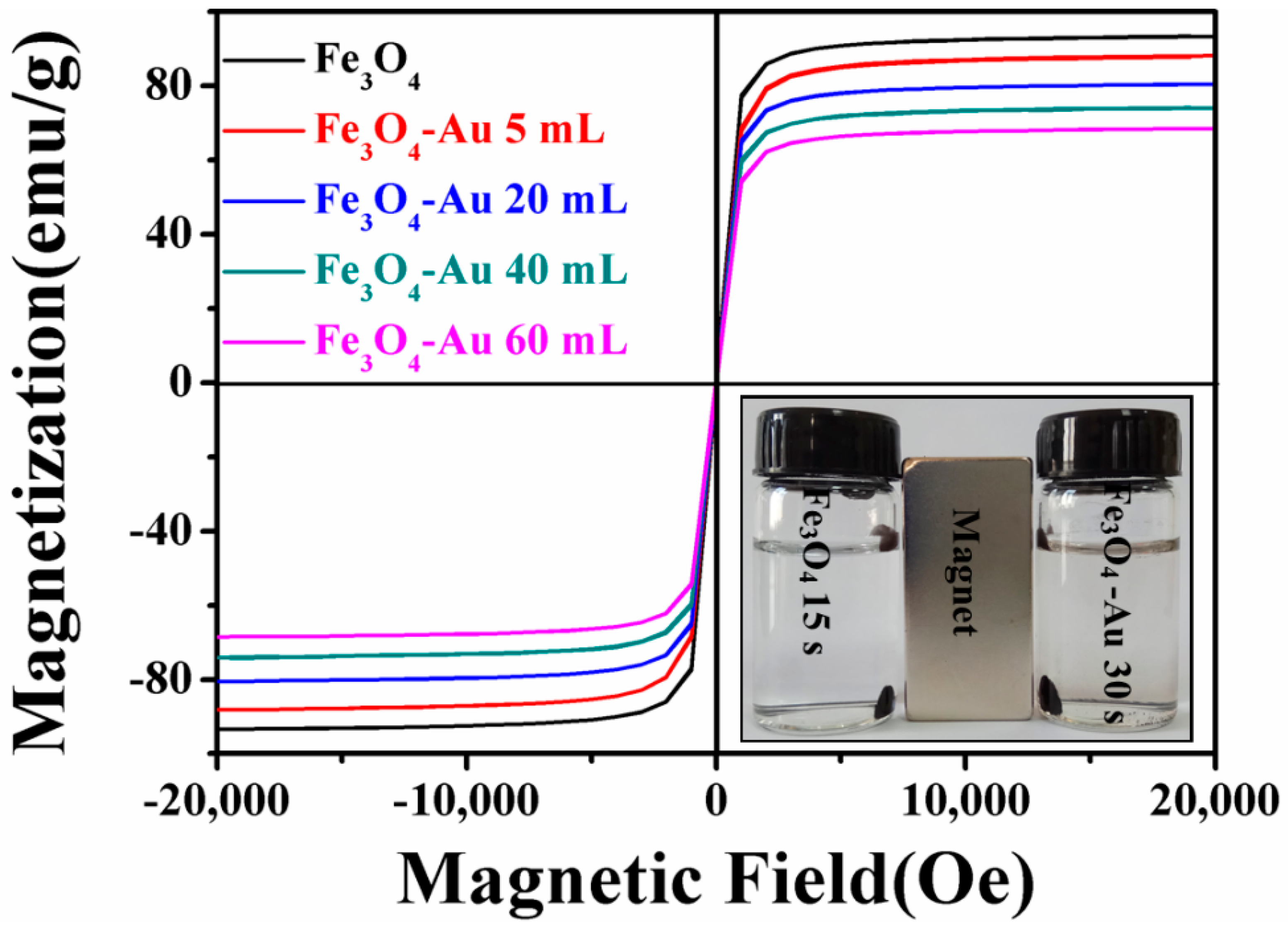
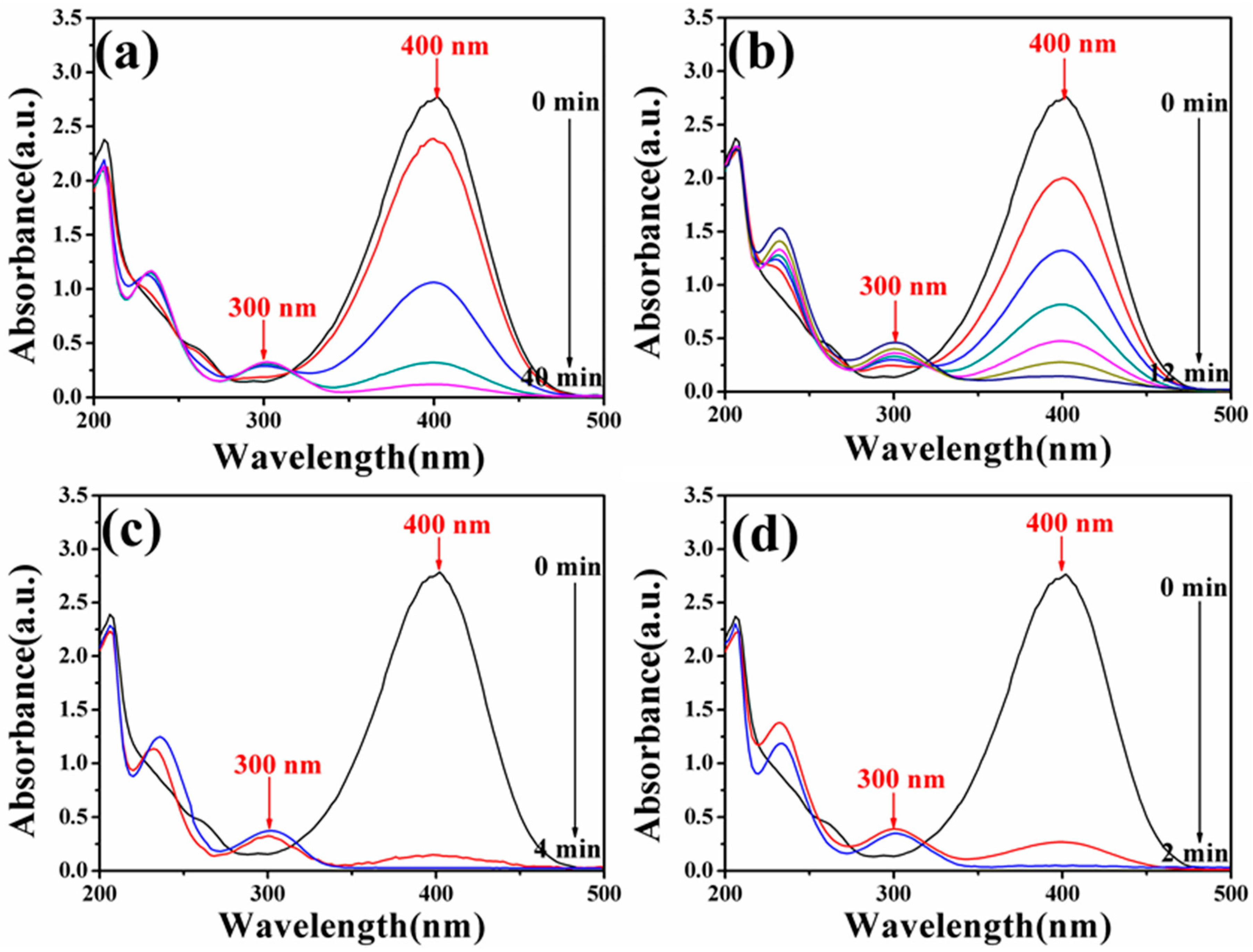
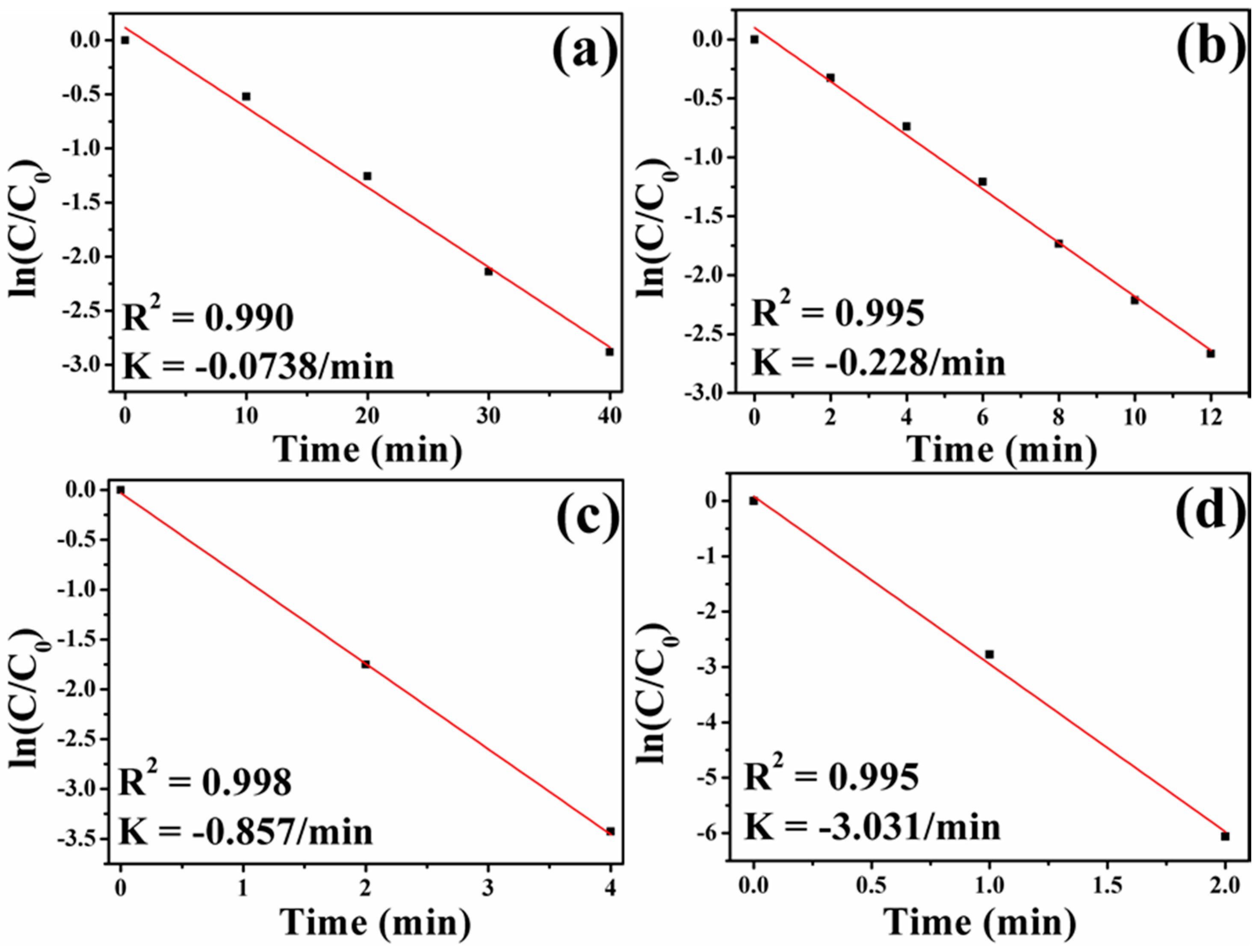
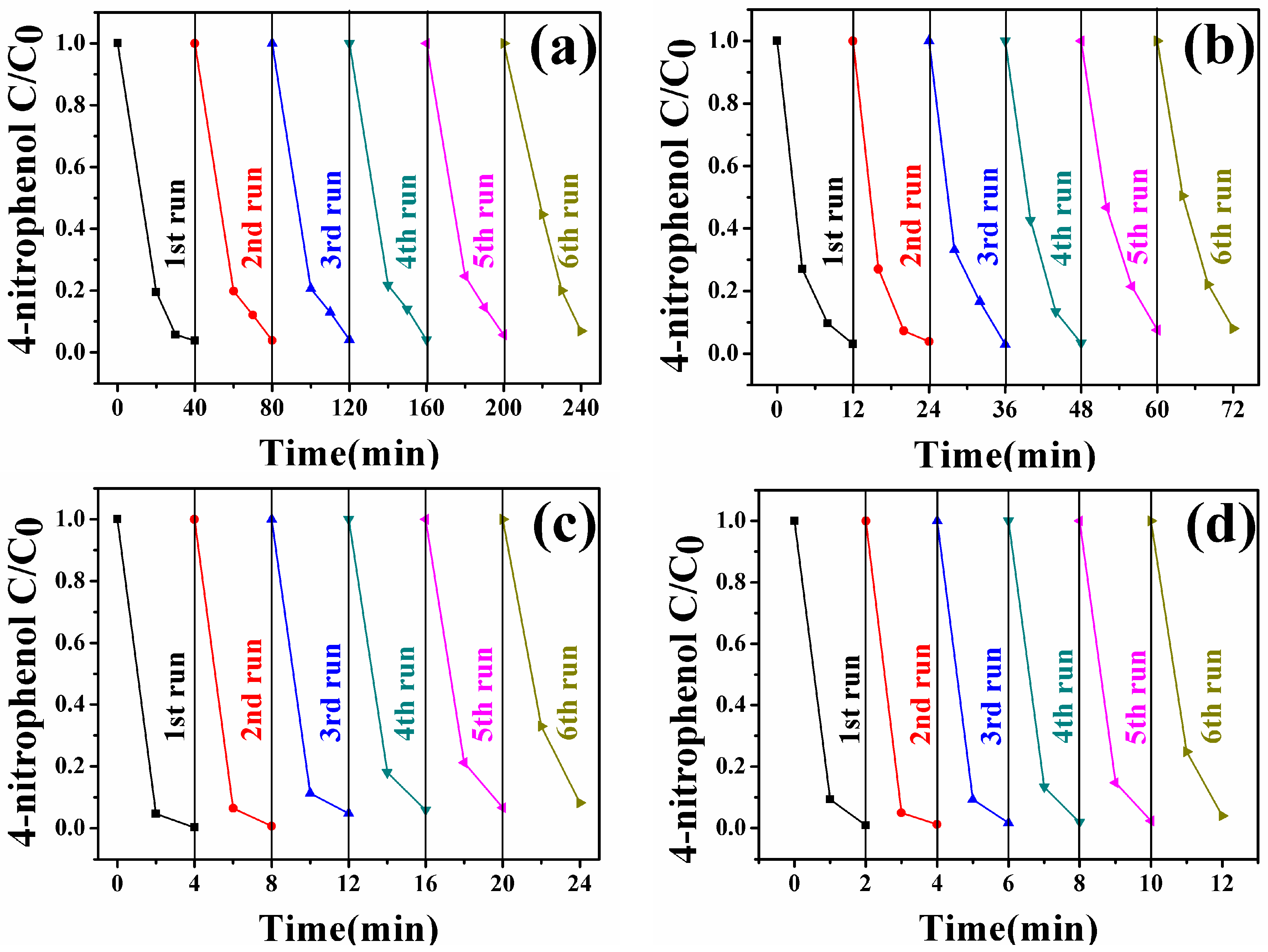
| Composition | IS (mm/s) | QS (mm/s) | HIN (T) | HWHM (mm/s) | AREA (%) |
|---|---|---|---|---|---|
| A | 0.287 | 0.015 | 48.5 | 0.186 | 34.3 |
| B | 0.603 | 0.012 | 45.5 | 0.433 | 65.7 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, Y.; Zhang, Y.; Kou, Q.; Liu, Y.; Han, D.; Wang, D.; Sun, Y.; Zhang, Y.; Wang, Y.; Lu, Z.; et al. Enhanced Catalytic Reduction of 4-Nitrophenol Driven by Fe3O4-Au Magnetic Nanocomposite Interface Engineering: From Facile Preparation to Recyclable Application. Nanomaterials 2018, 8, 353. https://doi.org/10.3390/nano8050353
Chen Y, Zhang Y, Kou Q, Liu Y, Han D, Wang D, Sun Y, Zhang Y, Wang Y, Lu Z, et al. Enhanced Catalytic Reduction of 4-Nitrophenol Driven by Fe3O4-Au Magnetic Nanocomposite Interface Engineering: From Facile Preparation to Recyclable Application. Nanomaterials. 2018; 8(5):353. https://doi.org/10.3390/nano8050353
Chicago/Turabian StyleChen, Yue, Yuanyuan Zhang, Qiangwei Kou, Yang Liu, Donglai Han, Dandan Wang, Yantao Sun, Yongjun Zhang, Yaxin Wang, Ziyang Lu, and et al. 2018. "Enhanced Catalytic Reduction of 4-Nitrophenol Driven by Fe3O4-Au Magnetic Nanocomposite Interface Engineering: From Facile Preparation to Recyclable Application" Nanomaterials 8, no. 5: 353. https://doi.org/10.3390/nano8050353
APA StyleChen, Y., Zhang, Y., Kou, Q., Liu, Y., Han, D., Wang, D., Sun, Y., Zhang, Y., Wang, Y., Lu, Z., Chen, L., Yang, J., & Xing, S. G. (2018). Enhanced Catalytic Reduction of 4-Nitrophenol Driven by Fe3O4-Au Magnetic Nanocomposite Interface Engineering: From Facile Preparation to Recyclable Application. Nanomaterials, 8(5), 353. https://doi.org/10.3390/nano8050353





