Recommendations for In Vitro and In Vivo Testing of Magnetic Nanoparticle Hyperthermia Combined with Radiation Therapy †
Abstract
:1. Introduction
2. Sample Preparation for In Vitro Evaluation
2.1. Colloidal Stability
2.2. Sterilization of the Aqueous MNP Solution
Sterilization of the Aqueous MNP Solution Using Filtration
2.3. MNP Samples Should Be Tested for Endotoxin and Used When Endotoxin-Free
2.4. A High Working Concentration of MNPs Is Recommended
2.5. The Formation of Protein Corona in the Presence of Fetal Bovine Serum
2.6. Sonication and Vortexing to Assure Homogeneity of the MNPs
2.7. Cells Should Be Confluent but Not Overgrown
2.8. Properties of the MNPs
3. In Vitro Evaluation of MNP-Cell Interactions
3.1. Toxicity
3.2. Cell Viability
3.3. Cell Proliferation
3.4. MNP Cellular Uptake Studies
3.5. Other Complementary Assays
4. Sample Preparation for In Vivo Evaluation
5. Magnetic Hyperthermia Combined with Radiation Therapy
5.1. Animal Physiology
5.2. Modelling and Simulations
5.3. Tumor Models
5.4. MNP Administration
5.5. Thermal Dosimetry
5.6. Treatment
5.7. Blood Flow
5.8. Thermotolerance
5.9. Heating Profile
5.10. Study Endpoints
5.11. Toxicity
5.12. Treatment Evaluation
5.13. Analysis of Results
6. Recommended Method for Pharmacokinetic and Biodistribution Studies of MNPs
6.1. Pharmacokinetics
6.2. Biodistribution Studies
6.3. Radiolabeling of MNPs, Selection of Radionuclides and Optimization of the Radiolabeling Procedure
6.4. Prediction of the Biodistribution Based on the Physicochemical Characteristics of MNPs
6.5. Particle Size and Charge Influence Biodistribution
6.6. The Nature of Coating Polymer Influences MNPs Pharmacokinetics
6.7. The Surface Charge of MNPs Modifies Their Biodistribution
6.8. Injected Quantity (Dose) of MNPs for Pharmacokinetic and Biodistribution Studies
6.9. Requirements for Animal Experimentation
6.10. Route of Administration
6.11. Metabolism and Excretion
- In urine: Urine is collected at the time of sacrifice and is centrifuged before High-performance liquid chromatography (HPLC) analysis;
- In serum: Blood is collected at the time of sacrifice and allowed to clot. After sample centrifugation (10 min, 2000× g), the supernatant is carefully removed, treated with twice the volume of cold ethanol and centrifuged once again. The supernatant is subjected to HPLC analysis;
- In other organs (and tumor, if relevant): The organ sample is cut into small pieces and homogenized, after addition of ice-cold Phosphate-buffered saline (PBS) solution. The resulting homogenate is centrifuged (15 min, 2000× g), filtered through a 0.2 μm filter and analyzed by HPLC.
6.12. Elemental Analysis by Inductively Coupled Plasma Mass Spectroscopy
7. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
Abbreviations
| AMF | Alternating Magnetic Field |
| ALP | Alkaline phosphatase |
| BrdU | 5-bromo-2′-deoxyuridine |
| CFSE | CarboxyFluoresceinSuccinimidyl Ester |
| CT | Computed Tomography |
| DLS | Dynamic Light Scattering |
| γ-GT | Gamma-glutamyl transferase |
| HPLC | High-performance liquid chromatography |
| HSP | Heat Shock Proteins |
| HT | Hyperthermia |
| ICP-MS | Inductively Coupled Plasma Mass Spectroscopy |
| ID | Injected Dose |
| MNP | Magnetic Nanoparticle |
| MH | Magnetic Hyperthermia |
| MPS | Mononuclear Phagocytic System |
| MRI | Magnetic Resonance Imaging |
| MTT | 3-(4,5-dimethylthiazolyl-2)-2,5-diphenyltetrazolium bromide |
| NP | Nanoparticle |
| PEG | Polyethylene glycol |
| PET | Positron Emission Tomography |
| PK | Pharmacokinetic |
| PI | Propidium Iodide |
| RES | Reticuloendothelial System |
| RT | Radiation Therapy |
| SGOT | Serum Glutamic-Oxaloacetic Transaminase |
| SPECT | Single-Photon Emission Computed Tomography |
| TEM | Transmission Electron Microscopy |
| X-ray | X-radiation |
| XTT | sodium 3′-[1-(phenylaminocarbonyl)-3,4-tetrazolium]-bis(4-methoxy-6-nitro) benzene sulfonic acid hydrate |
References
- Delaney, G.; Jacob, S.; Featherstone, C.; Barton, M. The role of radiotherapy in cancer treatment. Cancer 2005, 104, 1129–1137. [Google Scholar] [CrossRef] [PubMed]
- Schaue, D.; McBride, W.H. Opportunities and challenges of radiotherapy for treating cancer. Nat. Rev. Clin. Oncol. 2015, 12, 527–540. [Google Scholar] [CrossRef] [PubMed]
- Bortfeld, T. IMRT: A review and preview. Phys. Med. Biol. 2006, 51, R363–R379. [Google Scholar] [CrossRef] [PubMed]
- Staffurth, J. A review of the clinical evidence for intensity-modulated radiotherapy. Clin. Oncol. 2010, 22, 643–657. [Google Scholar] [CrossRef] [PubMed]
- Veldeman, L.; Madani, I.; Hulstaert, F.; De Meerleer, G.; Mareel, M.; De Neve, W. Evidence behind use of intensity-modulated radiotherapy: A systematic review of comparative clinical studies. Lancet Oncol. 2008, 9, 367–375. [Google Scholar] [CrossRef]
- Dawson, L.A.; Jaffray, D.A. Advances in image-guided radiation therapy. J. Clin. Oncol. 2007, 25, 938–946. [Google Scholar] [CrossRef] [PubMed]
- Jaffray, D.A. Image-guided radiotherapy: From current concept to future perspectives. Nat. Rev. Clin. Oncol. 2012, 9, 688–699. [Google Scholar] [CrossRef] [PubMed]
- Meyer, J.L.; Kavanagh, B.D.; Purdy, J.A.; Timmerman, R. IMRT, IGRT, SBRT: Advances in Treatment Planning and Delivery of Radiotherapy; S. Karger AG: Basel, Switzerland, 2007. [Google Scholar]
- Otto, K. Volumetric modulated arc therapy: IMRT in a single gantry arc. Med. Phys. 2008, 35, 310–317. [Google Scholar] [CrossRef] [PubMed]
- Teoh, M.; Clark, C.H.; Wood, K.; Whitaker, S.; Nisbet, A. Volumetric modulated arc therapy: A review of current literature and clinical use in practice. Br. J. Radiol. 2011, 84, 967–996. [Google Scholar] [CrossRef] [PubMed]
- Lo, S.S.; Fakiris, A.J.; Chang, E.L.; Mayr, N.A.; Wang, J.Z.; Papiez, L.; Teh, B.S.; McGarry, R.C.; Cardenes, H.R.; Timmerman, R.D. Stereotactic body radiation therapy: A novel treatment modality. Nat. Rev. Clin. Oncol. 2010, 7, 44–54. [Google Scholar] [CrossRef] [PubMed]
- Martin, A.; Gaya, A. Stereotactic body radiotherapy: A review. Clin. Oncol. 2010, 22, 157–172. [Google Scholar] [CrossRef] [PubMed]
- Sapkaroski, D.; Osborne, C.; Knight, K.A. A review of stereotactic body radiotherapy—Is volumetric modulated arc therapy the answer? J. Med. Radiat. Sci. 2015, 62, 142–151. [Google Scholar] [CrossRef] [PubMed]
- Allen, A.M.; Pawlicki, T.; Dong, L.; Fourkal, E.; Buyyounouski, M.; Cengel, K.; Plastaras, J.; Bucci, M.K.; Yock, T.I.; Bonilla, L.; et al. An evidence based review of proton beam therapy: The report of ASTRO’s emerging technology committee. Radiother. Oncol. 2012, 103, 8–11. [Google Scholar] [CrossRef] [PubMed]
- Mohan, R.; Grosshans, D. Proton therapy—Present and future. Adv. Drug Deliv. Rev. 2017, 109, 26–44. [Google Scholar] [CrossRef] [PubMed]
- Newhauser, W.D.; Zhang, R. The physics of proton therapy. Phys. Med. Biol. 2015, 60, R155. [Google Scholar] [CrossRef] [PubMed]
- Weichselbaum, R.R.; Liang, H.; Deng, L.; Fu, Y.-X. Radiotherapy and immunotherapy: A beneficial liaison? Nat. Rev. Clin. Oncol. 2017, 14, 365. [Google Scholar] [CrossRef] [PubMed]
- Overgaard, J. The current and potential role of hyperthermia in radiotherapy. Int. J. Radiat. Oncol. Biol. Phys. 1989, 16, 535–549. [Google Scholar] [CrossRef]
- Overgaard, J. The heat is (still) on—The past and future of hyperthermic radiation oncology. Radiother. Oncol. 2013, 109, 185–187. [Google Scholar] [CrossRef] [PubMed]
- Van der Zee, J. Heating the patient: A promising approach? Ann. Oncol. 2002, 13, 1173–1184. [Google Scholar] [CrossRef] [PubMed]
- Wust, P.; Hildebrandt, B.; Sreenivasa, G.; Rau, B.; Gellermann, J.; Riess, H.; Felix, R.; Schlag, P.M. Hyperthermia in combined treatment of cancer. Lancet Oncol. 2002, 3, 487–497. [Google Scholar] [CrossRef]
- Gerweck, L.E.; Nygaard, T.G.; Burlett, M. Response of cells to hyperthermia under acute and chronic hypoxic conditions. Cancer Res. 1979, 39, 966–972. [Google Scholar] [PubMed]
- Nielsen, O.S. Effect of fractionated hyperthermia on hypoxic cells in vitro. Int. J. Radiat. Biol. Relat. Stud. Phys. Chem. Med. 1981, 39, 73–82. [Google Scholar] [CrossRef] [PubMed]
- Overgaard, J.; Bichel, P. The influence of hypoxia and acidity on the hyperthermic response of malignant cells in vitro. Radiology 1977, 123, 511–514. [Google Scholar] [CrossRef] [PubMed]
- Overgaard, J.; Nielsen, O.S. The role of tissue environmental factors on the kinetics and morphology of tumor cells exposed to hyperthermia. Ann. N. Y. Acad. Sci. 1980, 335, 254–280. [Google Scholar] [CrossRef] [PubMed]
- Suit, H.D.; Gerweck, L.E. Potential for hyperthermia and radiation therapy. Cancer Res. 1979, 39, 2290–2298. [Google Scholar] [PubMed]
- Coss, R.A.; Dewey, W.C.; Bamburg, J.R. Effects of hyperthermia on dividing chinese hamster ovary cells and on microtubules in vitro. Cancer Res. 1982, 42, 1059–1071. [Google Scholar] [PubMed]
- Kim, S.H.; Kim, J.H.; Hahn, E.W. The enhanced killing of irradiated HeLa cells in synchronous culture by hyperthermia. Radiat. Res. 1976, 66, 337–345. [Google Scholar] [CrossRef] [PubMed]
- Westra, A.; Dewey, W.C. Variation in sensitivity to heat shock during the cell-cycle of Chinese hamster cells in vitro. Int. J. Radiat. Biol. Relat. Stud. Phys. Chem. Med. 1971, 19, 467–477. [Google Scholar] [CrossRef] [PubMed]
- Engin, K.; Leeper, D.B.; Thistlethwaite, A.J.; Tupchong, L.; Phil, D.; McFarlane, J.D. Tumor extracellular pH as a prognostic factor in thermoradiotherapy. Int. J. Radiat. Oncol. Biol. Phys. 1994, 29, 125–132. [Google Scholar] [CrossRef]
- Gerweck, L.E.; Richards, B.; Michaels, H.B. Influence of low pH on the development and decay of 42 °C thermotolerance in CHO cells. Int. J. Radiat. Oncol. Biol. Phys. 1982, 8, 1935–1941. [Google Scholar] [CrossRef]
- Koutcher, J.A.; Barnett, D.; Kornblith, A.B.; Cowburn, D.; Brady, T.J.; Gerweck, L.E. Relationship of changes in pH and energy status to hypoxic cell fraction and hyperthermia sensitivity. Int. J. Radiat. Oncol. Biol. Phys. 1990, 18, 1429–1435. [Google Scholar] [CrossRef]
- Song, C.W. Effect of local hyperthermia on blood flow and microenvironment: A review. Cancer Res. 1984, 44, 4721s–4730s. [Google Scholar] [PubMed]
- Wahl, M.L.; Bobyock, S.B.; Leeper, D.B.; Owen, C.S. Effects of 42 °C hyperthermia on intracellular pH in ovarian carcinoma cells during acute or chronic exposure to low extracellular pH. Int. J. Radiat. Oncol. Biol. Phys. 1997, 39, 205–212. [Google Scholar] [CrossRef]
- Chicheł, A.; Skowronek, J.; Kubaszewska, M.; Kanikowski, M. Hyperthermia—Description of a method and a review of clinical applications. Rep. Pract. Oncol. Radiother. 2007, 12, 267–275. [Google Scholar] [CrossRef]
- Hildebrandt, B.; Wust, P.; Ahlers, O.; Dieing, A.; Sreenivasa, G.; Kerner, T.; Felix, R.; Riess, H. The cellular and molecular basis of hyperthermia. Crit. Rev. Oncol. Hematol. 2002, 43, 33–56. [Google Scholar] [CrossRef]
- Vaupel, P.; Horsman, M.R. Tumour perfusion and associated physiology: Characterization and significance for hyperthermia. Int. J. Hyperth. 2010, 26, 209–210. [Google Scholar] [CrossRef] [PubMed]
- Vaupel, P.W.; Kelleher, D.K. Pathophysiological and vascular characteristics of tumours and their importance for hyperthermia: Heterogeneity is the key issue. Int. J. Hyperth. 2010, 26, 211–223. [Google Scholar] [CrossRef] [PubMed]
- Emami, B.; Scott, C.; Perez, C.A.; Asbell, S.; Swift, P.; Grigsby, P.; Montesano, A.; Rubin, P.; Curran, W.; Delrowe, J.; et al. Phase III study of interstitial thermoradiotherapy compared with interstitial radiotherapy alone in the treatment of recurrent or persistent human tumors: A prospectively controlled randomized study by the Radiation Therapy Oncology Group. Int. J. Radiat. Oncol. Biol. Phys. 1996, 34, 1097–1104. [Google Scholar] [CrossRef]
- Panikkanvalappil, S.R.; Hooshmand, N.; El-Sayed, M.A. Intracellular assembly of nuclear-targeted gold nanosphere enables selective plasmonic photothermal therapy of cancer by shifting their absorption wavelength toward near-infrared region. Bioconjug. Chem. 2017, 28, 2452–2460. [Google Scholar] [CrossRef] [PubMed]
- Dutz, S.; Hergt, R. Magnetic particle hyperthermia—A promising tumour therapy? Nanotechnology 2014, 25, 452001. [Google Scholar] [CrossRef] [PubMed]
- Gao, N.; Bozeman, E.N.; Qian, W.; Wang, L.; Chen, H.; Lipowska, M.; Staley, C.A.; Wang, Y.A.; Mao, H.; Yang, L. Tumor penetrating theranostic nanoparticles for enhancement of targeted and image-guided drug delivery into peritoneal tumors following intraperitoneal delivery. Theranostics 2017, 7, 1689–1704. [Google Scholar] [CrossRef] [PubMed]
- Mil’to, I.V.; Sukhodolo, I.V.; Ivanova, V.V.; Yusubov, M.S. Ultrastructure of rat kidneys after intravenous administration of modified magnetite nanoparticles. Bull. Exp. Biol. Med. 2017, 164, 218–222. [Google Scholar] [CrossRef] [PubMed]
- Coene, A.; Crevecoeur, G.; Leliaert, J.; Dupré, L. Toward 2D and 3D imaging of magnetic nanoparticles using EPR measurements. Med. Phys. 2015, 42, 5007–5014. [Google Scholar] [CrossRef] [PubMed]
- Gobbo, O.L.; Sjaastad, K.; Radomski, M.W.; Volkov, Y.; Prina-Mello, A. Magnetic nanoparticles in cancer theranostics. Theranostics 2015, 5, 1249–1263. [Google Scholar] [CrossRef] [PubMed]
- Hilger, I.; Kaiser, W.A. Iron oxide-based nanostructures for MRI and magnetic hyperthermia. Nanomedicine 2012, 7, 1443–1459. [Google Scholar] [CrossRef] [PubMed]
- Du, Z.; Su, R.; Liu, W.; Huang, Z. Magnetic nanoparticle thermometer: An investigation of minimum error transmission path and AC bias error. Sensors 2015, 15, 8624–8641. [Google Scholar] [CrossRef] [PubMed]
- Weaver, J.B.; Rauwerdink, A.M.; Hansen, E.W. Magnetic nanoparticle temperature estimation. Med. Phys. 2009, 36, 1822–1829. [Google Scholar] [CrossRef] [PubMed]
- Kossatz, S.; Grandke, J.; Couleaud, P.; Latorre, A.; Aires, A.; Crosbie-Staunton, K.; Ludwig, R.; Dähring, H.; Ettelt, V.; Lazaro-Carrillo, A.; et al. Efficient treatment of breast cancer xenografts with multifunctionalized iron oxide nanoparticles combining magnetic hyperthermia and anti-cancer drug delivery. Breast Cancer Res. 2015, 17, 66. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Jena, P.K.; Behera, S.; Lockey, R.F.; Mohapatra, S.; Mohapatra, S. Multifunctional magnetic nanoparticles for targeted delivery. Nanomed. Nanotechnol. Biol. Med. 2010, 6, 64–69. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.; Huang, S. Magnetic nanoparticles in cancer diagnosis, drug delivery and treatment (review). Mol. Clin. Oncol. 2017, 7, 738–746. [Google Scholar] [CrossRef] [PubMed]
- Hilger, I. In vivo applications of magnetic nanoparticle hyperthermia. Int. J. Hyperth. 2013, 29, 828–834. [Google Scholar] [CrossRef] [PubMed]
- Maier-Hauff, K.; Ulrich, F.; Nestler, D.; Niehoff, H.; Wust, P.; Thiesen, B.; Orawa, H.; Budach, V.; Jordan, A. Efficacy and safety of intratumoral thermotherapy using magnetic iron-oxide nanoparticles combined with external beam radiotherapy on patients with recurrent glioblastoma multiforme. J. Neurooncol. 2011, 103, 317–324. [Google Scholar] [CrossRef] [PubMed]
- Tombácz, E.; Farkas, K.; Földesi, I.; Szekeres, M.; Illés, E.; Tóth, I.Y.; Nesztor, D.; Szabó, T. Polyelectrolyte coating on superparamagnetic iron oxide nanoparticles as interface between magnetic core and biorelevant media. Interface Focus 2016, 6, 20160068. [Google Scholar] [CrossRef] [PubMed]
- Lim, J.; Yeap, S.P.; Che, H.X.; Low, S.C. Characterization of magnetic nanoparticle by dynamic light scattering. Nanoscale Res. Lett. 2013, 8, 381. [Google Scholar] [CrossRef] [PubMed]
- Santos-Martinez, M.J.; Inkielewicz-Stepniak, I.; Medina, C.; Rahme, K.; D’Arcy, D.M.; Fox, D.; Holmes, J.D.; Zhang, H.; Radomski, M.W. The use of quartz crystal microbalance with dissipation (QCM-D) for studying nanoparticle-induced platelet aggregation. Int. J. Nanomed. 2012, 7, 243–255. [Google Scholar] [CrossRef] [PubMed]
- Santos-Martinez, M.J.; Rahme, K.; Corbalan, J.J.; Faulkner, C.; Holmes, J.D.; Tajber, L.; Medina, C.; Radomski, M.W. Pegylation increases platelet biocompatibility of gold nanoparticles. J. Biomed. Nanotechnol. 2014, 10, 1004–1015. [Google Scholar] [CrossRef] [PubMed]
- Masson, V.; Maurin, F.; Fessi, H.; Devissaguet, J.P. Influence of sterilization processes on poly(ε-caprolactone) nanospheres. Biomaterials 1997, 18, 327–335. [Google Scholar] [CrossRef]
- Özcan, İ.; Bouchemal, K.; Segura-Sánchez, F.; Abaci, Ö.; Özer, Ö.; Güneri, T.; Ponchel, G. Effects of sterilization techniques on the PEGylated poly (γ-benzyl-l-glutamate) (PBLG) nanoparticles. ACTA Pharm. Sci. 2009, 51, 211–218. [Google Scholar]
- Godara, A.; Raabe, D.; Green, S. The influence of sterilization processes on the micromechanical properties of carbon fiber-reinforced peek composites for bone implant applications. Acta Biomater. 2007, 3, 209–220. [Google Scholar] [CrossRef] [PubMed]
- Zheng, J.; Clogston, J.D.; Patri, A.K.; Dobrovolskaia, M.A.; McNeil, S.E. Sterilization of silver nanoparticles using standard gamma irradiation procedure affects particle integrity and biocompatibility. J. Nanomed. Nanotechnol. 2011, S5. [Google Scholar] [CrossRef] [PubMed]
- Vetten, M.A.; Yah, C.S.; Singh, T.; Gulumian, M. Challenges facing sterilization and depyrogenation of nanoparticles: Effects on structural stability and biomedical applications. Nanomed. Nanotechnol. Biol. Med. 2014, 10, 1391–1399. [Google Scholar] [CrossRef] [PubMed]
- França, Á.; Pelaz, B.; Moros, M.; Sánchez-Espinel, C.; Hernández, A.; Fernández-López, C.; Grazú, V.; de la Fuente Jesús, M.; Pastoriza-Santos, I.; Liz-Marzán Luis, M.; et al. Sterilization matters: Consequences of different sterilization techniques on gold nanoparticles. Small 2009, 6, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Quinn, P.J. Endotoxins: Lipopolysaccharides of Gram-negative bacteria. In Endotoxins: Structure, Function and Recognition; Wang, X., Quinn, P.J., Eds.; Springer: Dordrecht, The Netherlands, 2010; Volume 53, pp. 3–25. [Google Scholar]
- Mbeh, D.A.; Mireles, L.K.; Stanicki, D.; Tabet, L.; Maghni, K.; Laurent, S.; Sacher, E.; Yahia, L.H. Human alveolar epithelial cell responses to core–shell superparamagnetic iron oxide nanoparticles (SPIONs). Langmuir 2015, 31, 3829–3839. [Google Scholar] [CrossRef] [PubMed]
- Azaroff, L.V. Elements of X-ray Crystallography; McGraw-Hill: New Yrok, NY, USA, 1968. [Google Scholar]
- Häfeli, U.O.; Riffle, J.S.; Harris-Shekhawat, L.; Carmichael-Baranauskas, A.; Mark, F.; Dailey, J.P.; Bardenstein, D. Cell uptake and in vitro toxicity of magnetic nanoparticles suitable for drug delivery. Mol. Pharm. 2009, 6, 1417–1428. [Google Scholar] [CrossRef] [PubMed]
- Hoskins, C.; Wang, L.; Cheng, W.P.; Cuschieri, A. Dilemmas in the reliable estimation of the in-vitro cell viability in magnetic nanoparticle engineering: Which tests and what protocols? Nanoscale Res. Lett. 2012, 7, 77. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Walton, J.R. The systemic appraisal of cellular injury. Agents Actions 1975, 5, 394–400. [Google Scholar] [CrossRef] [PubMed]
- Walton, J.R.; Buckley, L.K. Cell models in the study of mechanisms of toxicity. Agents Actions 1975, 5, 69–88. [Google Scholar] [CrossRef] [PubMed]
- Patil, S.U.; Adireddy, S.; Jaiswal, A.; Mandava, S.; Lee, R.B.; Chrisey, B.D. In vitro/in vivo toxicity evaluation and quantification of iron oxide nanoparticles. Int. J. Mol. Sci. 2015, 16, 24417–24450. [Google Scholar] [CrossRef] [PubMed]
- Gaspar, A.S.; Wagner, F.E.; Amaral, V.S.; Costa Lima, S.A.; Khomchenko, V.A.; Santos, J.G.; Costa, B.F.O.; Durães, L. Development of a biocompatible magnetic nanofluid by incorporating SPIONs in Amazonian oils. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2017, 172, 135–146. [Google Scholar] [CrossRef] [PubMed]
- Matuszak, J.; Baumgartner, J.; Zaloga, J.; Juenet, M.; da Silva, A.E.; Franke, D.; Almer, G.; Texier, I.; Faivre, D.; Metselaar, J.M.; et al. Nanoparticles for intravascular applications: Physicochemical characterization and cytotoxicity testing. Nanomedicine 2016, 11, 597–616. [Google Scholar] [CrossRef] [PubMed]
- Poller, J.M.; Zaloga, J.; Schreiber, E.; Unterweger, H.; Janko, C.; Radon, P.; Eberbeck, D.; Trahms, L.; Alexiou, C.; Friedrich, R.P. Selection of potential iron oxide nanoparticles for breast cancer treatment based on in vitro cytotoxicity and cellular uptake. Int. J. Nanomed. 2017, 12, 3207–3220. [Google Scholar] [CrossRef] [PubMed]
- Roehm, N.W.; Rodgers, G.H.; Hatfield, S.M.; Glasebrook, A.L. An improved colorimetric assay for cell proliferation and viability utilizing the tetrazolium salt XTT. J. Immunol. Methods 1991, 142, 257–265. [Google Scholar] [CrossRef]
- Weislow, O.S.; Kiser, R.; Fine, D.L.; Bader, J.; Shoemaker, R.H.; Boyd, M.R. New soluble-formazan assay for HIV-1 cytopathic effects: Application to high-flux screening of synthetic and natural products for AIDS-antiviral activity. JNCI J. Natl. Cancer Inst. 1989, 81, 577–586. [Google Scholar] [CrossRef] [PubMed]
- Präbst, K.; Engelhardt, H.; Ringgeler, S.; Hübner, H. Basic colorimetric proliferation assays: MTT, WST, and Resazurin. In Cell Viability Assays. Methods in Molecular Biology; Gilbert, D., Friedrich, O., Eds.; Humana Press: New York, NY, USA, 2017; Volume 1601. [Google Scholar]
- Tantra, R.; Knight, A. Cellular uptake and intracellular fate of engineered nanoparticles: A review on the application of imaging techniques. Nanotoxicology 2011, 5, 381–392. [Google Scholar] [CrossRef] [PubMed]
- Ayache, J.; Beaunier, L.; Boumendil, J.; Ehret, G.; Laub, D. Sample Preparation Handbook for Transmission Electron Microscopy; Springer-Verlag: New York, NY, USA, 2010; Volume XXV, p. 338. [Google Scholar]
- Friedrich, R.P.; Janko, C.; Poettler, M.; Tripal, P.; Zaloga, J.; Cicha, I.; Dürr, S.; Nowak, J.; Odenbach, S.; Slabu, I.; et al. Flow cytometry for intracellular SPION quantification: Specificity and sensitivity in comparison with spectroscopic methods. Int. J. Nanomed. 2015, 10, 4185–4201. [Google Scholar] [CrossRef] [PubMed]
- Stranz, M.; Kastango, E.S. A review of ph and osmolarity. Int. J. Pharm. Compd. 2002, 6, 216–220. [Google Scholar] [PubMed]
- Lee, Y.-C.; Zocharski, P.D.; Samas, B. An intravenous formulation decision tree for discovery compound formulation development. Int. J. Pharm. 2003, 253, 111–119. [Google Scholar] [CrossRef]
- Kumar, C.S.S.R.; Mohammad, F. Magnetic nanomaterials for hyperthermia-based therapy and controlled drug delivery. Adv. Drug Deliv. Rev. 2011, 63, 789–808. [Google Scholar] [CrossRef] [PubMed]
- Kappiyoor, R.; Liangruksa, M.; Ganguly, R.; Puri, I.K. The effects of magnetic nanoparticle properties on magnetic fluid hyperthermia. J. Appl. Phys. 2010, 108, 094702. [Google Scholar] [CrossRef]
- Keblinski, P.; Cahill, D.G.; Bodapati, A.; Sullivan, C.R.; Taton, T.A. Limits of localized heating by electromagnetically excited nanoparticles. J. Appl. Phys. 2006, 100, 054305. [Google Scholar] [CrossRef]
- Yu, L.; Liu, J.; Wu, K.; Klein, T.; Jiang, Y.; Wang, J.-P. Evaluation of hyperthermia of magnetic nanoparticles by dehydrating DNA. Sci. Rep. 2014, 4, 7216. [Google Scholar] [CrossRef] [PubMed]
- Stylianopoulos, T.; Economides, E.-A.; Baish, J.W.; Fukumura, D.; Jain, R.K. Towards optimal design of cancer nanomedicines: Multi-stage nanoparticles for the treatment of solid tumors. Ann. Biomed. Eng. 2015, 43, 2291–2300. [Google Scholar] [CrossRef] [PubMed]
- Rao, L.; Xu, J.-H.; Cai, B.; Liu, H.; Li, M.; Jia, Y.; Xiao, L.; Guo, S.-S.; Liu, W.; Zhao, X.-Z. Synthetic nanoparticles camouflaged with biomimetic erythrocyte membranes for reduced reticuloendothelial system uptake. Nanotechnology 2016, 27, 085106. [Google Scholar] [CrossRef] [PubMed]
- Ozcelikkale, A.; Moon, H.-R.; Linnes, M.; Han, B. In vitro microfluidic models of tumor microenvironment to screen transport of drugs and nanoparticles. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2017, 9, e1460. [Google Scholar] [CrossRef] [PubMed]
- Veiseh, O.; Gunn, J.W.; Zhang, M. Design and fabrication of magnetic nanoparticles for targeted drug delivery and imaging. Adv. Drug Deliv. Rev. 2010, 62, 284–304. [Google Scholar] [CrossRef] [PubMed]
- Jain, R.K. Normalizing tumor microenvironment to treat cancer: Bench to bedside to biomarkers. J. Clin. Oncol. 2013, 31, 2205–2218. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Li, Y.; Orza, A.; Lu, Q.; Guo, P.; Wang, L.; Yang, L.; Mao, H. Magnetic nanoparticle facilitated drug delivery for cancer therapy with targeted and image-guided approaches. Adv. Funct. Mater. 2016, 26, 3818–3836. [Google Scholar] [CrossRef] [PubMed]
- Alexiou, C.; Jurgons, R.; Seliger, C.; Brunke, O.; Iro, H.; Odenbach, S. Delivery of superparamagnetic nanoparticles for local chemotherapy after intraarterial infusion and magnetic drug targeting. Anticancer Res. 2007, 27, 2019–2022. [Google Scholar] [PubMed]
- Kudr, J.; Haddad, Y.; Richtera, L.; Heger, Z.; Cernak, M.; Adam, V.; Zitka, O. Magnetic nanoparticles: From design and synthesis to real world applications. Nanomaterials 2017, 7, 243. [Google Scholar] [CrossRef] [PubMed]
- Tietze, R.; Lyer, S.; Dürr, S.; Alexiou, C. Nanoparticles for cancer therapy using magnetic forces. Nanomedicine 2012, 7, 447–457. [Google Scholar] [CrossRef] [PubMed]
- Byrne, J.D.; Betancourt, T.; Brannon-Peppas, L. Active targeting schemes for nanoparticle systems in cancer therapeutics. Adv. Drug Deliv. Rev. 2008, 60, 1615–1626. [Google Scholar] [CrossRef] [PubMed]
- Danhier, F.; Le Breton, A.; Préat, V. RGD-based strategies to target alpha(v) beta(3) integrin in cancer therapy and diagnosis. Mol. Pharm. 2012, 9, 2961–2973. [Google Scholar] [CrossRef] [PubMed]
- Reinmuth, N.; Liu, W.; Ahmad, S.A.; Fan, F.; Stoeltzing, O.; Parikh, A.A.; Bucana, C.D.; Gallick, G.E.; Nickols, M.A.; Westlin, W.F.; et al. Αvβ3 integrin antagonist s247 decreases colon cancer metastasis and angiogenesis and improves survival in mice. Cancer Res. 2003, 63, 2079–2087. [Google Scholar] [PubMed]
- Desgrosellier, J.S.; Cheresh, D.A. Integrins in cancer: Biological implications and therapeutic opportunities. Nat. Rev. Cancer 2010, 10, 9–22. [Google Scholar] [CrossRef] [PubMed]
- Marschner, J.A.; Schäfer, H.; Holderied, A.; Anders, H.-J. Optimizing mouse surgery with online rectal temperature monitoring and preoperative heat supply. Effects on post-ischemic acute kidney injury. PLoS ONE 2016, 11, e0149489. [Google Scholar] [CrossRef] [PubMed]
- Matsukawa, T.; Sessler, D.I.; Sessler, A.M.; Schroeder, M.; Ozaki, M.; Kurz, A.; Cheng, C. Heat flow and distribution during induction of general anesthesia. Anesthesiology 1995, 82, 662–673. [Google Scholar] [CrossRef] [PubMed]
- Sessler, M.D.; Daniel, I. Temperature monitoring and perioperative thermoregulation. Anesthesiology 2008, 109, 318–338. [Google Scholar] [CrossRef] [PubMed]
- Di Ventura, B.; Lemerle, C.; Michalodimitrakis, K.; Serrano, L. From in vivo to in silico biology and back. Nature 2006, 443, 527. [Google Scholar] [CrossRef] [PubMed]
- Dunning, W.F. Prostate cancer in the rat. Natl. Cancer Inst. Monogr. 1963, 12, 351–369. [Google Scholar] [PubMed]
- Lein, M.; Koenig, F.; Misdraji, J.; McDougal, W.S.; Jung, K.; Loening, S.A.; Hasan, T.; Ortel, B. Laser-induced hyperthermia in rat prostate cancer: Role of site of tumor implantation. Urology 2000, 56, 167–172. [Google Scholar] [CrossRef]
- Echevarria-Uraga, J.J.; García-Alonso, I.; Plazaola, F.; Insausti, M.; Etxebarria, N.; Saiz-López, A.; Fernández-Ruanova, B. Study of the intra-arterial distribution of Fe3O4 nanoparticles in a model of colorectal neoplasm induced in rat liver by MRI and spectrometry. Int. J. Nanomed. 2012, 7, 2399–2410. [Google Scholar] [CrossRef] [PubMed]
- Fung, A.S.; Lee, C.; Yu, M.; Tannock, I.F. The effect of chemotherapeutic agents on tumor vasculature in subcutaneous and orthotopic human tumor xenografts. BMC Cancer 2015, 15, 112. [Google Scholar] [CrossRef] [PubMed]
- Pham, T.B.; Colvin, K.E.; Pham, T.N.; Kim, J.B.; Fuller, S.E.; Moon, A.E.; Barbey, R.; Yuen, S.; Rickman, H.B.; Bryce, S.N.; et al. Biodistribution and clearance of stable superparamagnetic maghemite iron oxide nanoparticles in mice following intraperitoneal administration. Int. J. Mol. Sci. 2018, 19, 205. [Google Scholar] [CrossRef] [PubMed]
- Zulauf, G.D.; Trembly, B.S.; Giustini, A.J.; Flint, B.R.; Strawbridge, R.R.; Hoopes, P.J. Targeting of Systemically-Delivered Magnetic Nanoparticle Hyperthermia Using a Noninvasive, Static, External Magnetic Field. In Proceedings of the SPIE 8584, Energy-Based Treatment of Tissue and Assessment VII, San Francisco, CA, USA, 26 February 2013; p. 85840C. [Google Scholar]
- Merle, P.; Camus, P.; Abergel, A.; Pageaux, G.P.; Masliah, C.; Bronowicki, J.P.; Zarski, J.P.; Pelletier, G.; Bouattour, M.; Farloux, L.; et al. Safety and efficacy of intra-arterial hepatic chemotherapy with doxorubicin-loaded nanoparticles in hepatocellular carcinoma. ESMO Open 2017, 2. [Google Scholar] [CrossRef] [PubMed]
- Deipolyi, R.A.; Zhang, S.Y.; Khademhosseini, A.; Naidu, S.; Borad, M.; Sahin, B.; Mathur, K.A.; Oklu, R. Portal vein embolization: Impact of chemotherapy and genetic mutations. J. Clin. Med. 2017, 6, 26. [Google Scholar] [CrossRef] [PubMed]
- MacFall, J.R.; Soher, B.J. MR imaging in hyperthermia. Radiographics 2007, 27, 1809–1818. [Google Scholar] [CrossRef] [PubMed]
- Shuhendler, A.J.; Staruch, R.; Oakden, W.; Gordijo, C.R.; Rauth, A.M.; Stanisz, G.J.; Chopra, R.; Wu, X.Y. Thermally-triggered ‘off–on–off’ response of gadolinium-hydrogel–lipid hybrid nanoparticles defines a customizable temperature window for non-invasive magnetic resonance imaging thermometry. J. Control. Release 2012, 157, 478–484. [Google Scholar] [CrossRef] [PubMed]
- Chung, S.H.; Mehta, R.; Tromberg, B.J.; Yodh, A.G. Non-invasive measurement of deep tissue temperature changes caused by apoptosis during breast cancer neoadjuvant chemotherapy: A case study. J. Innov. Opt. Health Sci. 2011, 4, 361–372. [Google Scholar] [CrossRef] [PubMed]
- Pearce, J.A. Comparative analysis of mathematical models of cell death and thermal damage processes. Int. J. Hyperth. 2013, 29, 262–280. [Google Scholar] [CrossRef] [PubMed]
- Van Rhoon, G.C. Is CEM43 still a relevant thermal dose parameter for hyperthermia treatment monitoring? Int. J. Hyperth. 2016, 32, 50–62. [Google Scholar] [CrossRef] [PubMed]
- Dewhirst, M.W.; Sim, D.A. The utility of thermal dose as a predictor of tumor and normal tissue responses to combined radiation and hyperthermia. Cancer Res. 1984, 44, 4772s–4780s. [Google Scholar] [PubMed]
- Kapp, D.S.; Cox, R.S. Thermal treatment parameters are most predictive of outcome in patients with single tumor nodules per treatment field in recurrent adenocarcinoma of the breast. Int. J. Radiat. Oncol. Biol. Phys. 1995, 33, 887–899. [Google Scholar] [CrossRef]
- Seegenschmiedt, M.H.; Martus, P.; Fietkau, R.; Iro, H.; Brady, L.W.; Sauer, R. Multivariate analysis of prognostic parameters using interstitial thermoradiotherapy (IHT-IRT): Tumor and treatment variables predict outcome. Int. J. Radiat. Oncol. Biol. Phys. 1994, 29, 1049–1063. [Google Scholar] [CrossRef]
- Sherar, M.; Liu, F.-F.; Pintilie, M.; Levin, W.; Hunt, J.; Hill, R.; Hand, J.; Vernon, C.; van Rhoon, G.; van der Zee, J.; et al. Relationship between thermal dose and outcome in thermoradiotherapy treatments for superficial recurrences of breast cancer: Data from a phase III trial. Int. J. Radiat. Oncol. Biol. Phys. 1997, 39, 371–380. [Google Scholar] [CrossRef]
- Horsman, M.R.; Overgaard, J. Hyperthermia: A potent enhancer of radiotherapy. Clin. Oncol. 2007, 19, 418–426. [Google Scholar] [CrossRef] [PubMed]
- Sapareto, S.A.; Peter Raaphorst, G.; Dewey, W.C. Cell killing and the sequencing of hyperthermia and radiation. Int. J. Radiat. Oncol. Biol. Phys. 1979, 5, 343–347. [Google Scholar] [CrossRef]
- Li, G.C.; Kal, H.B. Effect of hyperthermia on the radiation response of two mammalian cell lines. Eur. J. Cancer 1977, 13, 65–69. [Google Scholar] [CrossRef]
- Sapareto, S.A.; Hopwood, L.E.; Dewey, W.C.; Raju, M.R.; Gray, J.W. Effects of hyperthermia on survival and progression of Chinese hamster ovary cells. Cancer Res. 1978, 38, 393–400. [Google Scholar] [PubMed]
- Falk, T.; Prasad, T.; Steffen, L.; Antje, D.; Andriy, F.; Robert, H.; Mathias, L.; Bernd, R.; Manfred, S.; Mechthild, K.; et al. Precise image-guided irradiation of small animals: A flexible non-profit platform. Phys. Med. Biol. 2016, 61, 3084–3108. [Google Scholar] [CrossRef]
- Ford, E.; Emery, R.; Huff, D.; Narayanan, M.; Schwartz, J.; Cao, N.; Meyer, J.; Rengan, R.; Zeng, J.; Sandison, G.; et al. An image-guided precision proton radiation platform for preclinical in vivo research. Phys. Med. Biol. 2017, 62, 43–58. [Google Scholar] [CrossRef] [PubMed]
- Vanstalle, M.; Constanzo, J.; Karakaya, Y.; Finck, C.; Rousseau, M.; Brasse, D. Analytical dose modeling for preclinical proton irradiation of millimetric targets. Med. Phys. 2018, 45, 470–478. [Google Scholar] [CrossRef] [PubMed]
- Verhaegen, F.; Dubois, L.; Gianolini, S.; Hill, M.A.; Karger, C.P.; Lauber, K.; Prise, K.M.; Sarrut, D.; Thorwarth, D.; Vanhove, C.; et al. ESTRO ACROP: Technology for precision small animal radiotherapy research: Optimal use and challenges. Radiother. Oncol. 2018, 126, 471–478. [Google Scholar] [CrossRef] [PubMed]
- Iwata, K.; Shakil, A.; Hur, W.J.; Makepeace, C.M.; Griffin, R.J.; Song, C.W. Tumour pO2 can be increased markedly by mild hyperthermia. Br. J. Cancer Suppl. 1996, 27, S217–S221. [Google Scholar] [PubMed]
- Song, C.W.; Shakil, A.; Osborn, J.L.; Iwata, K. Tumour oxygenation is increased by hyperthermia at mild temperatures. Int. J. Hyperth. 1996, 12, 367–373. [Google Scholar] [CrossRef]
- Vaupel, P.; Kallinowski, F.; Okunieff, P. Blood flow, oxygen and nutrient supply, and metabolic microenvironment of human tumors: A review. Cancer Res. 1989, 49, 6449–6465. [Google Scholar] [PubMed]
- Hurwitz, M.; Stauffer, P. Hyperthermia, radiation and chemotherapy: The role of heat in multidisciplinary cancer care. Semin. Oncol. 2014, 41, 714–729. [Google Scholar] [CrossRef] [PubMed]
- Corry, P.M.; Armour, E.P. The heat shock response: Role in radiation biology and cancer therapy. Int. J. Hyperth. 2005, 21, 769–778. [Google Scholar] [CrossRef] [PubMed]
- Repasky, E.; Issels, R. Physiological consequences of hyperthermia: Heat, heat shock proteins and the immune response. Int. J. Hyperth. 2002, 18, 486–489. [Google Scholar] [CrossRef] [PubMed]
- Roti Roti, J.L. Cellular responses to hyperthermia (40–46 °C): Cell killing and molecular events. Int. J. Hyperth. 2008, 24, 3–15. [Google Scholar] [CrossRef] [PubMed]
- Calderwood, S.K.; Theriault, J.R.; Gong, J. How is the immune response affected by hyperthermia and heat shock proteins? Int. J. Hyperth. 2005, 21, 713–716. [Google Scholar] [CrossRef] [PubMed]
- Coss, R.A. Inhibiting induction of heat shock proteins as a strategy to enhance cancer therapy. Int. J. Hyperth. 2005, 21, 695–701. [Google Scholar] [CrossRef] [PubMed]
- Lauber, K.; Brix, N.; Ernst, A.; Hennel, R.; Krombach, J.; Anders, H.; Belka, C. Targeting the heat shock response in combination with radiotherapy: Sensitizing cancer cells to irradiation-induced cell death and heating up their immunogenicity. Cancer Lett. 2015, 368, 209–229. [Google Scholar] [CrossRef] [PubMed]
- Welton, P.L.; Goffinet, D.R.; Kapp, D.S. Interstitial hyperthermia in head and neck oncology. In Carcinomas of the Head and Neck: Evaluation and Management; Jacobs, C., Ed.; Springer: Boston, MA, USA, 1990; Volume 52, pp. 83–93. [Google Scholar]
- Lindegaard, J.C.; Grau, C.; Overgaard, J. Effect of step-down heating on the interaction between heat and radiation in a C3H mammary carcinoma in vivo. Int. J. Radiat. Biol. 1991, 60, 707–721. [Google Scholar] [CrossRef] [PubMed]
- Lindegaard, J.C.; Overgaard, J. Effect of step-down heating on hyperthermic radiosensitization in an experimental tumor and a normal tissue in vivo. Radiother. Oncol. 1988, 11, 143–151. [Google Scholar] [CrossRef]
- Nielsen, O.S.; Overgaard, J.; Kamura, T. Influence of thermotolerance on the interaction between hyperthermia and radiation in a solid tumour in vivo. Br. J. Radiol. 1983, 56, 267–273. [Google Scholar] [CrossRef] [PubMed]
- Datta, N.R.; Puric, E.; Klingbiel, D.; Gomez, S.; Bodis, S. Hyperthermia and radiation therapy in locoregional recurrent breast cancers: A systematic review and meta-analysis. Int. J. Radiat. Oncol. Biol. Phys. 2016, 94, 1073–1087. [Google Scholar] [CrossRef] [PubMed]
- Datta, N.R.; Rogers, S.; Ordóñez, S.G.; Puric, E.; Bodis, S. Hyperthermia and radiotherapy in the management of head and neck cancers: A systematic review and meta-analysis. Int. J. Hyperth. 2016, 32, 31–40. [Google Scholar] [CrossRef] [PubMed]
- Franckena, M. Review of radiotherapy and hyperthermia in primary cervical cancer. Int. J. Hyperth. 2012, 28, 543–548. [Google Scholar] [CrossRef] [PubMed]
- Kaur, P.; Hurwitz, M.D.; Krishnan, S.; Asea, A. Combined hyperthermia and radiotherapy for the treatment of cancer. Cancers 2011, 3, 3799–3823. [Google Scholar] [CrossRef] [PubMed]
- Fu, Q.; Huang, T.; Wang, X.; Lu, C.; Liu, F.; Yang, G.; Wang, Y.; Wang, B. Association of elevated reactive oxygen species and hyperthermia induced radiosensitivity in cancer stem-like cells. Oncotarget 2017, 8, 101560–101571. [Google Scholar] [CrossRef] [PubMed]
- Suriyanto; Ng, E.Y.K.; Kumar, S.D. Physical mechanism and modeling of heat generation and transfer in magnetic fluid hyperthermia through Néelian and Brownian relaxation: A review. BioMed. Eng. OnLine 2017, 16, 36. [Google Scholar] [CrossRef] [PubMed]
- Saeed, M.; Ren, W.; Wu, A. Therapeutic applications of iron oxide based nanoparticles in cancer: Basic concepts and recent advances. Biomater. Sci. 2018. [Google Scholar] [CrossRef] [PubMed]
- Lagadec, C.; Vlashi, E.; Della Donna, L.; Meng, Y.; Dekmezian, C.; Kim, K.; Pajonk, F. Survival and self-renewing capacity of breast cancer initiating cells during fractionated radiation treatment. Breast Cancer Res. 2010, 12, R13. [Google Scholar] [CrossRef] [PubMed]
- Woodward, W.A.; Bristow, R.G. Radiosensitivity of cancer-initiating cells and normal stem cells (or what the Heisenberg uncertainly principle has to do with biology). Semin. Radiat. Oncol. 2009, 19, 87–95. [Google Scholar] [CrossRef] [PubMed]
- Hurley, K.R.; Ring, H.L.; Etheridge, M.; Zhang, J.; Gao, Z.; Shao, Q.; Klein, N.D.; Szlag, V.M.; Chung, C.; Reineke, T.M.; et al. Predictable heating and positive MRI contrast from a mesoporous silica-coated iron oxide nanoparticle. Mol. Pharm. 2016, 13, 2172–2183. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, M.M.; Guha, C.; Hodge, J.W.; Jaffee, E. Immunobiology of radiotherapy: New paradigms. Radiat. Res. 2014, 182, 123–125. [Google Scholar] [CrossRef] [PubMed]
- Demaria, S.; Golden, E.B.; Formenti, S.C. Role of local radiation therapy in cancer immunotherapy. JAMA Oncol. 2015, 1, 1325–1332. [Google Scholar] [CrossRef] [PubMed]
- Formenti, S.C.; Demaria, S. Combining radiotherapy and cancer immunotherapy: A paradigm shift. JNCI J. Natl. Cancer Inst. 2013, 105, 256–265. [Google Scholar] [CrossRef] [PubMed]
- Salama, A.K.S.; Postow, M.A.; Salama, J.K. Irradiation and immunotherapy: From concept to the clinic. Cancer 2016, 122, 1659–1671. [Google Scholar] [CrossRef] [PubMed]
- Dewan, M.Z.; Galloway, A.E.; Kawashima, N.; Dewyngaert, J.K.; Babb, J.S.; Formenti, S.C.; Demaria, S. Fractionated but not single-dose radiotherapy induces an immune-mediated abscopal effect when combined with anti–CTLA-4 antibody. Clin. Cancer Res. 2009, 15, 5379–5388. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.; Auh, S.L.; Wang, Y.; Burnette, B.; Wang, Y.; Meng, Y.; Beckett, M.; Sharma, R.; Chin, R.; Tu, T.; et al. Therapeutic effects of ablative radiation on local tumor require CD8+ T cells: Changing strategies for cancer treatment. Blood 2009, 114, 589–595. [Google Scholar] [CrossRef] [PubMed]
- Lugade, A.A.; Moran, J.P.; Gerber, S.A.; Rose, R.C.; Frelinger, J.G.; Lord, E.M. Local radiation therapy of B16 melanoma tumors increases the generation of tumor antigen-specific effector cells that traffic to the tumor. J. Immunol. 2005, 174, 7516–7523. [Google Scholar] [CrossRef] [PubMed]
- Schaue, D.; Ratikan, J.A.; Iwamoto, K.S.; McBride, W.H. Maximizing tumor immunity with fractionated radiation. Int. J. Radiat. Oncol. Biol. Phys. 2012, 83, 1306–1310. [Google Scholar] [CrossRef] [PubMed]
- Vranješ-Đurić, S.; Ignjatović, N. Radiolabeled functional nanoparticles in preventive and regenerative medicine. In Nanotechnologies in Preventive and Regenerative Medicine, 1st ed.; Uskokovic, V., Ed.; Elsevier: Cambridge, MA, USA, 2017; pp. 65–92. [Google Scholar]
- Ernsting, M.J.; Murakami, M.; Roy, A.; Li, S.-D. Factors controlling the pharmacokinetics, biodistribution and intratumoral penetration of nanoparticles. J. Control. Release 2013, 172, 782–794. [Google Scholar] [CrossRef] [PubMed]
- Kang, H.; Mintri, S.; Menon, A.V.; Lee, H.Y.; Choi, H.S.; Kim, J. Pharmacokinetics, pharmacodynamics and toxicology of theranostic nanoparticles. Nanoscale 2015, 7, 18848–18862. [Google Scholar] [CrossRef] [PubMed]
- Jankovic, D.; Vranjes-Djuric, S.; Djokic, D.; Markovic, M.; Ajdinovic, B.; Jaukovic, L.; Nikolic, N. 90Y-labeled tin fluoride colloid as a promising therapeutic agent: Preparation, characterization, and biological study in rats. J. Pharm. Sci. 2012, 101, 2194–2203. [Google Scholar] [CrossRef] [PubMed]
- Radović, M.; Calatayud, M.P.; Goya, G.F.; Ibarra, M.R.; Antić, B.; Spasojević, V.; Nikolić, N.; Janković, D.; Mirković, M.; Vranješ-Đurić, S. Preparation and in vivo evaluation of multifunctional 90Y-labeled magnetic nanoparticles designed for cancer therapy. J. Biomed. Mater. Res. Part A 2015, 103, 126–134. [Google Scholar] [CrossRef] [PubMed]
- Gobbo, O.L.; Wetterling, F.; Vaes, P.; Teughels, S.; Markos, F.; Edge, D.; Shortt, C.M.; Crosbie-Staunton, K.; Radomski, M.W.; Volkov, Y.; et al. Biodistribution and pharmacokinetic studies of spion using particle electron paramagnetic resonance, MRI and ICP-MS. Nanomedicine 2015, 10, 1751–1760. [Google Scholar] [CrossRef] [PubMed]
- Karageorgou, M.-A.; Vranješ-Djurić, S.; Radović, M.; Lyberopoulou, A.; Antić, B.; Rouchota, M.; Gazouli, M.; Loudos, G.; Xanthopoulos, S.; Sideratou, Z.; et al. Gallium-68 labeled iron oxide nanoparticles coated with 2,3-dicarboxypropane-1,1-diphosphonic acid as a potential pet/mr imaging agent: A proof-of-concept study. Contrast Media Mol. Imaging 2017, 2017, 13. [Google Scholar] [CrossRef] [PubMed]
- Pellico, J.; Ruiz-Cabello, J.; Saiz-Alía, M.; del Rosario, G.; Caja, S.; Montoya, M.; Fernández de Manuel, L.; Morales, M.P.; Gutiérrez, L.; Galiana, B.; et al. Fast synthesis and bioconjugation of 68Ga core-doped extremely small iron oxide nanoparticles for PET/MR imaging. Contrast Media Mol. Imaging 2016, 11, 203–210. [Google Scholar] [CrossRef] [PubMed]
- Xiao, K.; Li, Y.; Luo, J.; Lee, J.S.; Xiao, W.; Gonik, A.M.; Agarwal, R.G.; Lam, K.S. The effect of surface charge on in vivo biodistribution of PEG-oligocholic acid based micellar nanoparticles. Biomaterials 2011, 32, 3435–3446. [Google Scholar] [CrossRef] [PubMed]
- Diehl, K.-H.; Hull, R.; Morton, D.; Pfister, R.; Rabemampianina, Y.; Smith, D.; Vidal, J.-M.; Vorstenbosch, C.V.D. A good practice guide to the administration of substances and removal of blood, including routes and volumes. J. Appl. Toxicol. 2001, 21, 15–23. [Google Scholar] [CrossRef] [PubMed]
- Charan, J.; Kantharia, N. How to calculate sample size in animal studies? J. Pharmacol. Pharmacother. 2013, 4, 303–306. [Google Scholar] [CrossRef] [PubMed]
- Bouziotis, P.; Stellas, D.; Thomas, E.; Truillet, C.; Tsoukalas, C.; Lux, F.; Tsotakos, T.; Xanthopoulos, S.; Paravatou-Petsotas, M.; Gaitanis, A.; et al. 68Ga-radiolabeled AGuIX nanoparticles as dual-modality imaging agents for PET/MRI-guided radiation therapy. Nanomedicine 2017, 12, 1561–1574. [Google Scholar] [CrossRef] [PubMed]
- Antic, B.; Boskovic, M.; Nikodinovic-Runic, J.; Ming, Y.; Zhang, H.; Bozin, E.S.; Janković, D.; Spasojevic, V.; Vranjes-Djuric, S. Complementary approaches for the evaluation of biocompatibility of 90Y-labeled superparamagnetic citric acid (Fe,Er)3O4 coated nanoparticles. Mater. Sci. Eng. C 2017, 75, 157–164. [Google Scholar] [CrossRef] [PubMed]
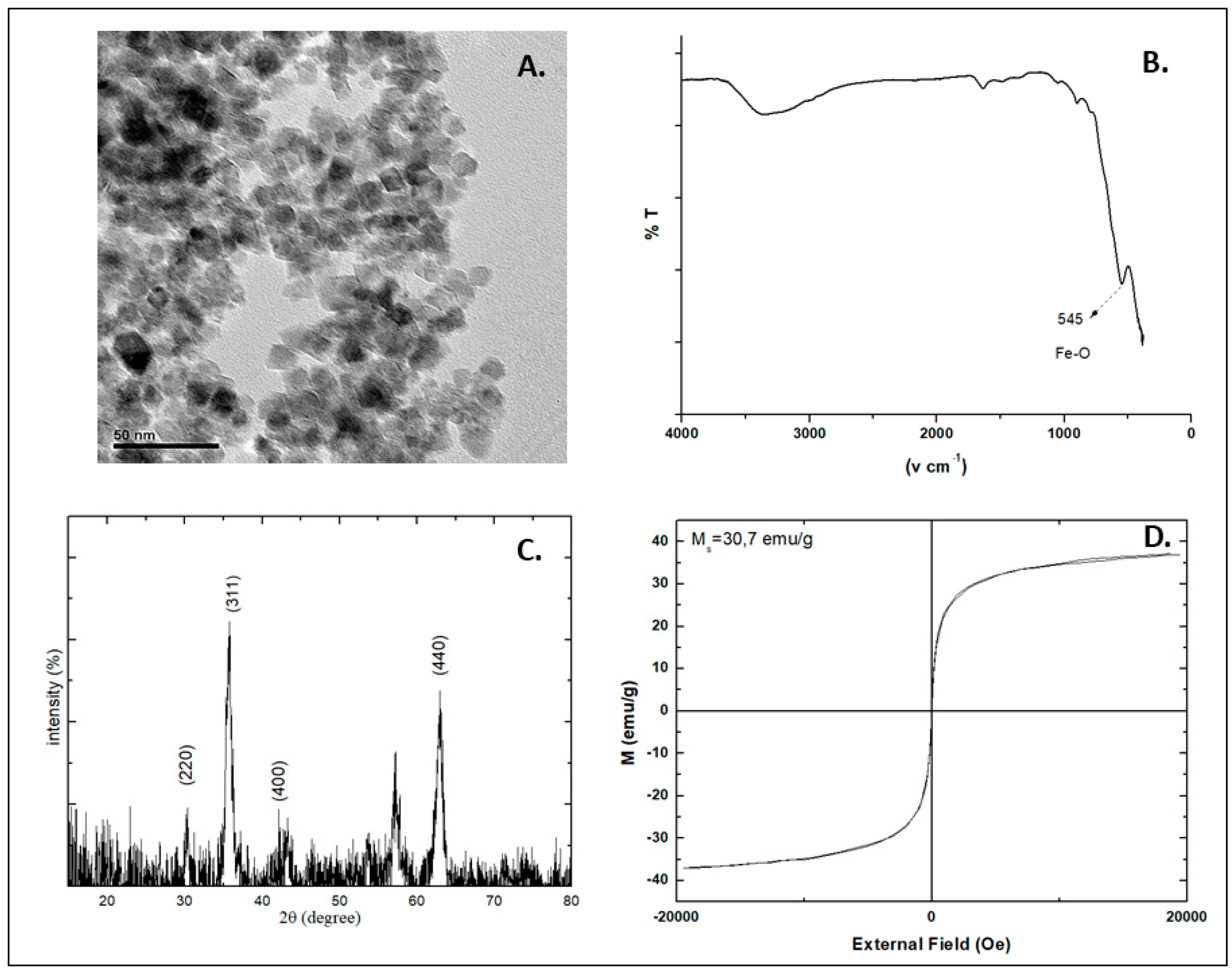
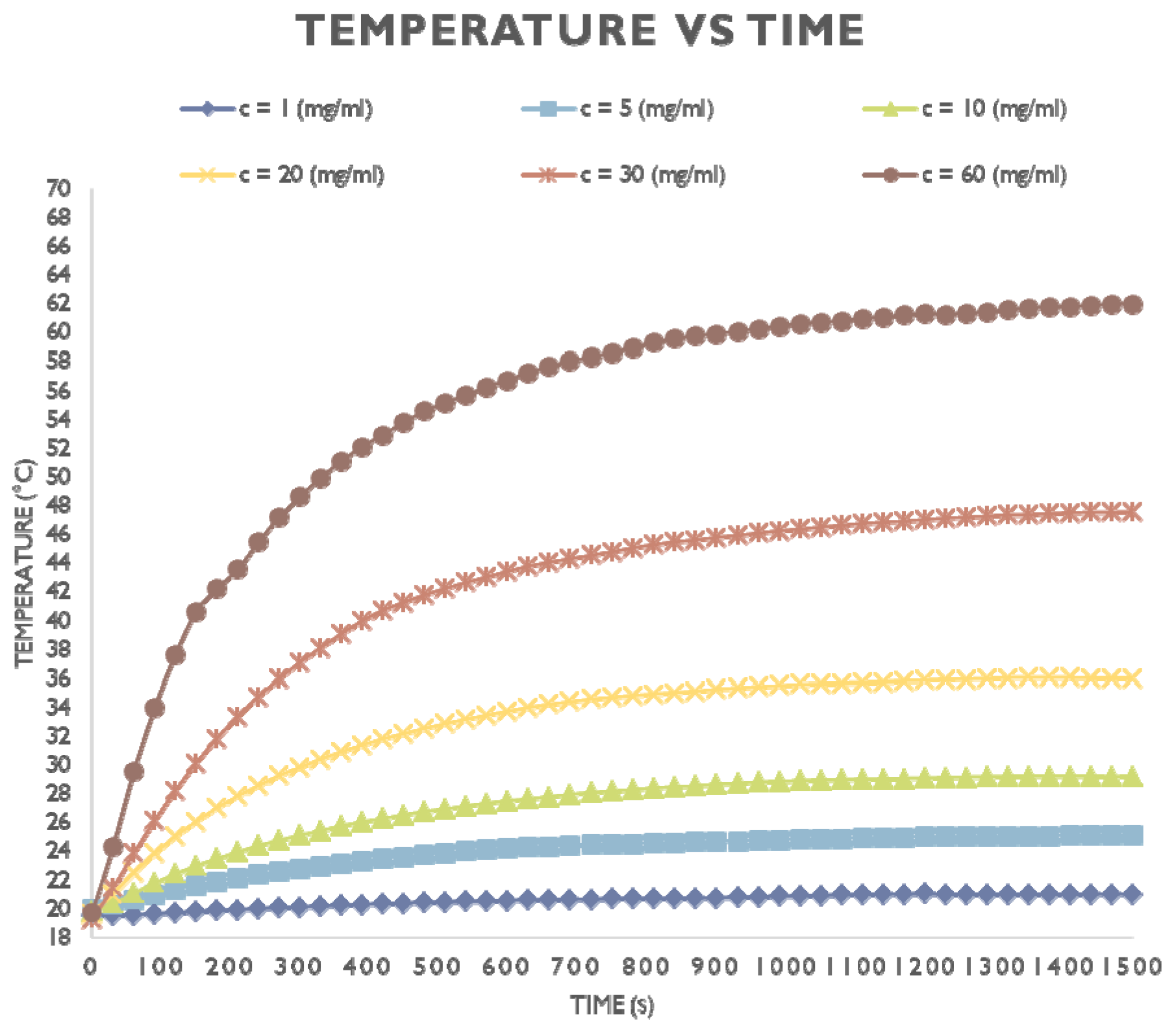
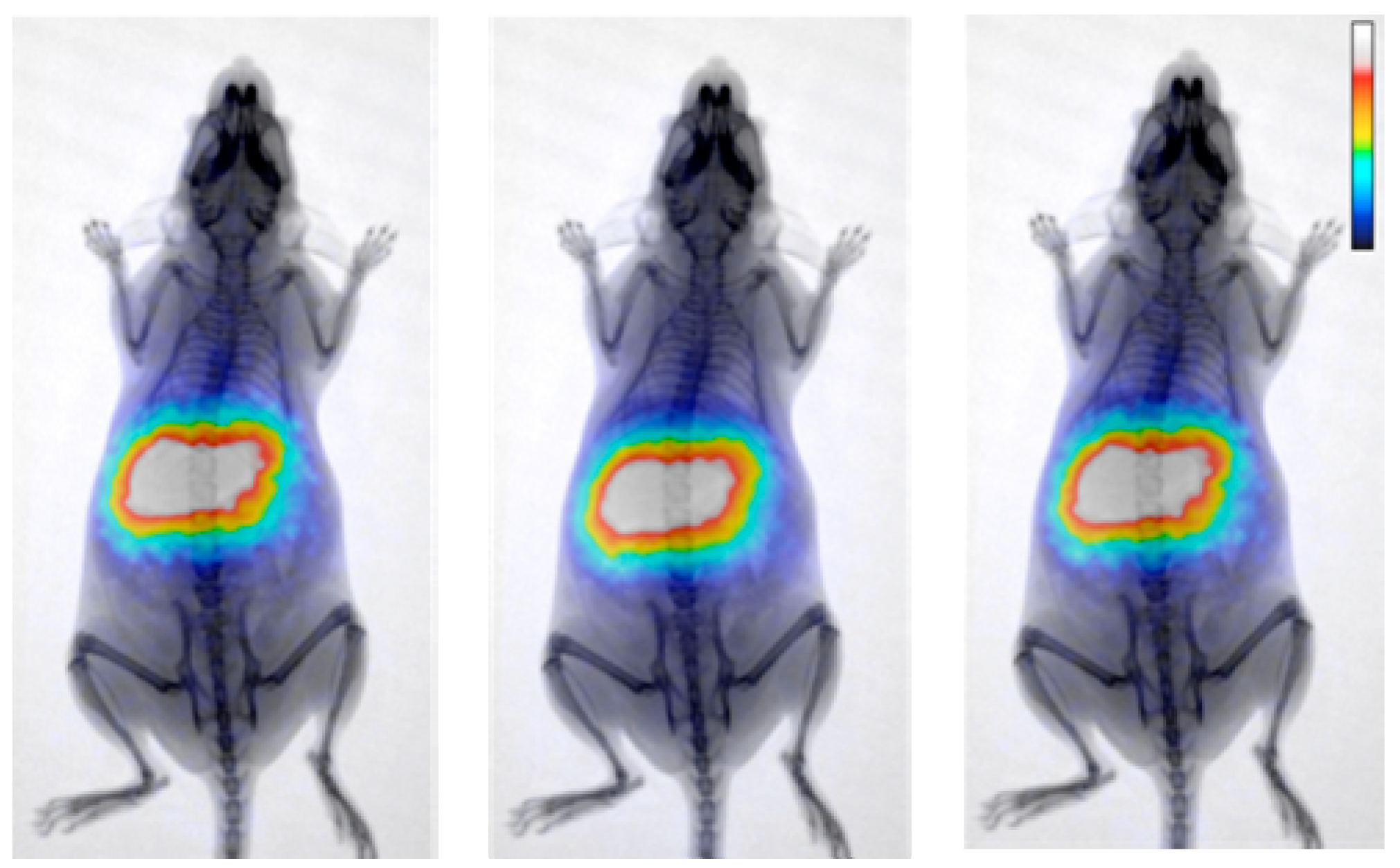
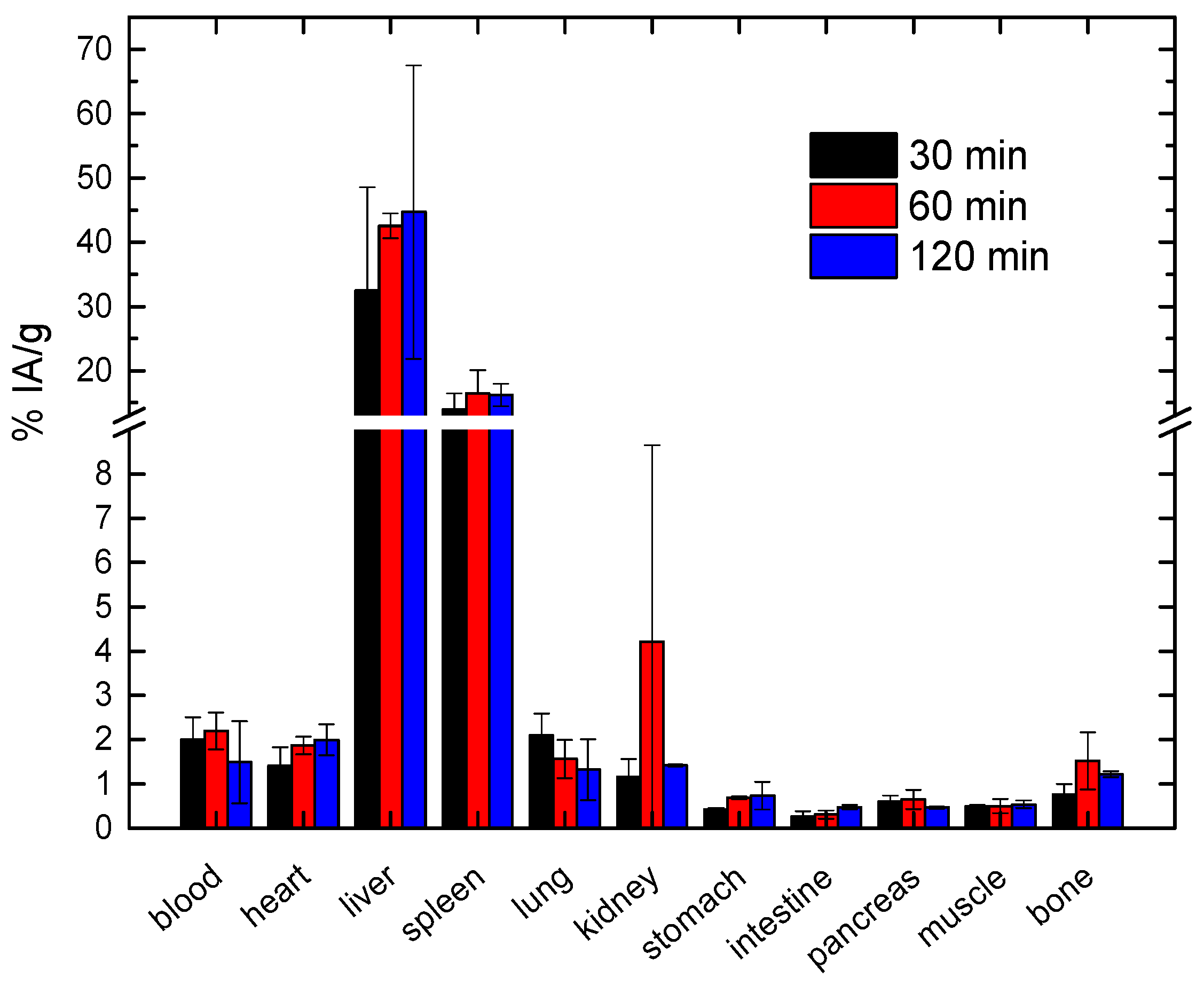
| Coating Agent | Types of IONPs | Diameter (nm) | Type of Cells | Dose | Incubation Time | Types of Assay | Brief Results | Ref. |
|---|---|---|---|---|---|---|---|---|
| Silica | Bare IONPs | 10 ± 3 | Human dermal fibroblasts (HDFs) and human fibrosarcoma (HT-1080) in DMEM media | 200–1000 μg/mL | 24 h | CCK-8 and LDH | APTMS-TEOS-Fe3O4 showed more cytotoxicity in terms of metabolic activity compared to other MNPs in HDFs. All MNPs induced LDH leakage in HDFs and HT-1080 cells. | [62] |
| TEOS-IONPs | 100–150 | |||||||
| APTMS-TEOS-IONPs | 100–150 | |||||||
| Bare IONPs | 10–50 | Peripheral blood lymphocytes in RPMI media | 1–100 μg/mL | 2 and 24 h | Annexin V-FIT Capoptosis detection | No significant difference between treated and untreated lymphocytes for 2 and 24 h. | [104] | |
| VTES-TEOS-IONPs | 10–50 | |||||||
| APTES/VTES-TEOS-IONPs | 10–50 | |||||||
| Bare IONPs | 150–200 L | L929 fibroblasts in DMEM media | 15–1000 mg/L | 24–72 h M | MTT | Silica coating reduced cell toxicity. Sulfhydryl modification improved cell-compatibility and haemocompatibility. | [105] | |
| TEOS-IONPs | ||||||||
| DMSA-TEOS-IONPs | ||||||||
| TEOS-IONPs | 15–20 | MCF-7 and HeLa cells in DMEM media | 0–200 μg/mL | 24 h M | MTT | MCF-7 and HeLa cells showed good biocompatibility at various concentrations. | [106] | |
| PEG | PEG-IONPs | ~30 | Hela cells and C6 cells in DMEM media | 0.01–1 mg/mL | 12 h | MTT | Cell viability was not affected at the concentration of 1 mg/mL. | [107] |
| PEG-IONPs | 10–15 | NIH/3T3 in DMEM | 1.5 to 192 μM | 24 and 48 h | MTT | PEG-IONPs showed good compatibility, 86% (24 h) and 67% (48 h) at 192 μM. | [108] | |
| Bare IONPs | 10–13 | Macrophages (mice) in RPMI media | 100 μg/mL | 1 h | MTT | No significant changes in viability after 1 h by all IONPs. Bare IONPs produced highest ROS compared to PEG and COOH-PEG-IONPs. | [109] | |
| PEG- IONPs | 100 | |||||||
| COOH-PEG-IONPs | 100 | |||||||
| PEG-550-IONPs | 8–11 | Bovine vascular smooth muscle cells (VSMCs) in DMEM media | 100–1000 ppm | 5–24 h | LIVE/DEAD viability/Cytotoxicity Kit | Dose dependent cytotoxic response was found. PEG-2K showed higher cell viability compared to PEG-10K at 100 ppm. | [110] | |
| PEG-2K-IONPs | ||||||||
| PEG-5K-IONPs | ||||||||
| PEG-10K-IONPs | ||||||||
| PEPABC: IONPs | 36 ± 5 | Mouse brain endothelial cell line (bEnd.3) in DMEM media | 0–10 mg/mL | 30 h | Resazurin dye assay | No cell death reported after 30 h exposure at 10 mg/mL. | [110] | |
| Dextran | Dextran-IONPs | 200–250 | Head and neck squamous cell carcinoma: tonsilla (UT-SCC-60A) and the metastasis (UT-SCC-60B) in DMEM media | 0.2–1.8 mM | 0–120 h | MTT, Annexin-V apoptosis detection assay | MTT: Decreased cell toxicity of dextran-IONPs compared to Resovist® Annexin-V-apoptosis: no changes in cell viability when cells were treated at the concentration of 1.8 mM. | [112] |
| Dextran-IONPs | 100 | Mouse melanoma cells (B16) and Chinese hamster lung; fibroblast cells (V79) in DMEM media | 0–400 μg/mL | 24 h | MTT | Slight changes in the cell viability were noticed as compared to control. | [113] | |
| Dextran-IONPs 9 | 9.12 ± 1.46 L | L929 fibroblast cells | 50–1000 μg/mL | 24 h | MTT | Significant reduction in cell viability at 1 mg/mL. Cells were 90% viable at 0.75 mg/mL. | [114] | |
| DEAE-dextran-IONPs | 27–50 | Murine mesenchymal stem/stromal cell (MSC) in DMEM media | 50 μg/mL | 3 h | CCK-8 | No significant changes I the cell viability were noticed. | [115] | |
| Bare Fe2O3 | 7 | Human bone marrow mesenchymal stromal cells (hBMSCs) hBMSCs-1: age 12 years; hBMSCs-2: age 54 years in α-modified eagle media (α MEM) | 15.4 g of iron/mL | 72 h | WST-1 | The study compared physicochemical properties of bare Fe2O3 and nanoparticles coated with different coating agents. hBMSCs-1: significant reduction in cell viability by PLL-Fe2O3and mannose-Fe2O3 NPs; hBMSCs-2: reduction in cell viability by all IONPs, mostly by uncoated-Fe2O3 and PLL-Fe O NPs. | [116] | |
| Endorem® (Fe3O4 coated with dextran) | 5.5 | |||||||
| PLL | PLL-Fe2O3 | 5.5 | ||||||
| PLL-dextran | PLL-Endorem | 5.6 | ||||||
| PDMAAm | PDMAAm-Fe2O3 | 7.5 | ||||||
| Mannose | Mannose-Fe2O3 | 7 | ||||||
| Mono-meric | IONPs-R1 | 6.5–7.5 | Murine primary brain cells (primary microglia, primary hippocampal neurons, and neuron–glia co-cultures) in DMEM media | 0.5, 1.5 or 3.0 mM | 6–24 h | PI staining | Extended incubation and dose dependent cell death was observed by all IONPs except Ferumoxytol. Ferumoxytol surprisingly increased the number of viable cells. IONPs-R1, R2 and Ferucarbotran were quickly ingested by microglial cells compared to Ferumoxytol. | [117] |
| citrate layer | IONPs-R2 | 7.5–8.7 | ||||||
| Carboxy-dextran | Ferucarbotran (Resovist®) | 60 | ||||||
| Carboxymeth-dextran | Ferumoxytol (Feraheme®) | 30 | ||||||
| Chitosan | Bare IONPs 5 | 50-100 | Human L-O2 hepatocytes in RPMI media | 1.25–20 μg/mL | 24 h | MTT | Bare IONPs showed more cytotoxicity compared to FAPLCS-IONPs in L-O2 hepatocytes. | [118] |
| FAPLCS-IONPs 1 | 136.60 ± 3.90 | |||||||
| Bare IONPs 1 | 18 | Primary human osteoblast cells (SV40) in DMEM media | 20–300 μg/mL | 48 h | CCK-8 | Decreased viability found when cells were treated with bare IONPs at 100 and 300 μg/mL. | [119] | |
| CS-IONPs | 35 | |||||||
| CS-IONPs | 2–8 | Cervical carcinoma cell lines (HeLa and SiHa) | 0–1000 μg/mL | 24 h | XTT | Bare and CS-IONPs showed reduction in cell viability by 5% and 2% respectively. SiHa cells showed 8% reduction in cell viability at 1000 μg/mL. | [120] | |
| Carbon | Fe@C/C | 5–140 | Human (HTB140), murine (B16-F10) melanoma cells and human dermal fibroblasts (HDF) in DMEM | 0.0001–100 μg/mL | 24 h | MTT | Decreased cell viability in melanoma cells. Murine melanoma cells were more sensitive to bare IONPs than human cells. Fe@C-COOH and Fe@C-CH2CH2-COOH showed weaker response to cells, and 80–100% cells remained viable. | [121] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Spirou, S.V.; Costa Lima, S.A.; Bouziotis, P.; Vranješ-Djurić, S.; Efthimiadou, E.Κ.; Laurenzana, A.; Barbosa, A.I.; Garcia-Alonso, I.; Jones, C.; Jankovic, D.; et al. Recommendations for In Vitro and In Vivo Testing of Magnetic Nanoparticle Hyperthermia Combined with Radiation Therapy. Nanomaterials 2018, 8, 306. https://doi.org/10.3390/nano8050306
Spirou SV, Costa Lima SA, Bouziotis P, Vranješ-Djurić S, Efthimiadou EΚ, Laurenzana A, Barbosa AI, Garcia-Alonso I, Jones C, Jankovic D, et al. Recommendations for In Vitro and In Vivo Testing of Magnetic Nanoparticle Hyperthermia Combined with Radiation Therapy. Nanomaterials. 2018; 8(5):306. https://doi.org/10.3390/nano8050306
Chicago/Turabian StyleSpirou, Spiridon V., Sofia A. Costa Lima, Penelope Bouziotis, Sanja Vranješ-Djurić, Eleni Κ. Efthimiadou, Anna Laurenzana, Ana Isabel Barbosa, Ignacio Garcia-Alonso, Carlton Jones, Drina Jankovic, and et al. 2018. "Recommendations for In Vitro and In Vivo Testing of Magnetic Nanoparticle Hyperthermia Combined with Radiation Therapy" Nanomaterials 8, no. 5: 306. https://doi.org/10.3390/nano8050306
APA StyleSpirou, S. V., Costa Lima, S. A., Bouziotis, P., Vranješ-Djurić, S., Efthimiadou, E. Κ., Laurenzana, A., Barbosa, A. I., Garcia-Alonso, I., Jones, C., Jankovic, D., & Gobbo, O. L. (2018). Recommendations for In Vitro and In Vivo Testing of Magnetic Nanoparticle Hyperthermia Combined with Radiation Therapy. Nanomaterials, 8(5), 306. https://doi.org/10.3390/nano8050306









