The DaNa2.0 Knowledge Base Nanomaterials—An Important Measure Accompanying Nanomaterials Development
Abstract
:1. Introduction—Nanosafety: An International Issue
2. Study Design and Methodology
3. Knowledge Base—Content and Statistics
3.1. Information Platform to Support Sustainable Material Development
3.2. Platform Informing on the Applications of Advanced Materials, Its Implications for Consumers, and Safety Issues in an Integrative Way
4. Summary
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Appendix A
| Sector | Application in | Nanomaterials Involved | Estimated Exposure Potential Human | Estimated Exposure Potential Environment |
|---|---|---|---|---|
| Medical products & food | Bone cement Cancer therapy Contrast agent Dental prosthesis Dietary supplement Drugs Food additive Implants Pregnancy test Rapid diagnostics Vaccination Wound dressing | Barium sulphate Cellulose Gold Iron and Iron oxides Silicon dioxide Silver Titanium dioxide Zirconium dioxide Zeolite | High (exposure intended) | Medium (mainly via waste water) |
| Electronic goods | Color television tube Display Electrode Electronics LED Nano Wires Photovoltaic cell Processor RAM (Random-Access Memory) Touch screens | Carbon Nanotubes (CNT) Copper and copper oxides Diamond Gold Indium tin oxide (ITO) Quantum Dots Strontium carbonate Titanium dioxide | Low | Low (medium at end-of-life) |
| Construction and building | Abrasive and polishing agents Anti-fogging agents Cement Cobblestones Glass Glue Heated coatings Lacquer and plastics additive Lightweight construction Rubber Tools | Aluminum oxides Carbon Black Carbon Nanotubes (CNT) Cerium dioxide Diamond Gold Indium tin oxide (ITO) Silicon dioxide Titanium dioxide Tungsten carbide Tungsten carbide-Cobalt Zinc oxide Zirconium dioxide | Medium to low | low |
| House hold | Anti-fogging agents Cat litter Cleaning agent PET bottles Soft Toy Sports equipment Tennis racket Textiles Wallpaper | Carbon Nanotubes (CNT) Fullerenes Graphene Nanoclays Silicon dioxide Silver Titanium dioxide Titanium nitride Zeolite Zinc oxide | High to medium | Medium |
| Personal care products | Cosmetics Skin care Soap Suncream Tooth paste | Carbon Black Fullerenes Silicon dioxide Silver Titanium dioxide Zinc oxide | High (exposure intended) | Medium (mainly via waste water) |
| Pigments | Black pigments Facade and wall color Printing inkToner | Carbon Black Iron and Iron oxides Silicon dioxide Silver Titanium dioxide | High (when exposure intended, e.g., Tattoos) Medium for others | Medium to low (mainly via waste) |
| Automotive sector | Catalytic exhaust converter Diesel additive Fuel cell Lubricant Tires | Barium sulphate Carbon Black Cerium dioxide Copper and copper oxides Fullerenes Gold Platinum Silicon dioxide Zirconium dioxide | High to medium | High to medium |
| Agriculture and environmental sector | Environmental remediation Fertilizer Filtration Water softener Water treatment Wood preservatives | Aluminum oxides Copper and copper oxides Diamond Gold Iron and Iron oxides Zeolite | Medium to low | High to medium |
| Miscellaneous | Chemical catalyst Coating Film Pyrotechnics | Gold NanoClays Silicon dioxide Silver Strontium carbonate Titanium dioxide Zeolite | Low (Event related) | Medium to low |
References
- Nanosafety Cluster—Cluster Projects—Horizon 2020 Projects. Available online: https://www.nanosafetycluster.eu/eu-nanosafety-cluster-projects/horizon-2020-projects (accessed on 28 February 2018).
- Horizon 2020 Work Programme 2014–2015. Vol. 5. Leadership in Enabling and Industrial Technologies II. Nanotechnologies, Advanced Materials, Biotechnology and Advanced Manufacturing and Processing. 2013. Available online: http://ec.europa.eu/research/participants/data/ref/h2020/wp/2014_2015/main/h2020-wp1415-leit-nmp_en.pdf (accessed on 5 March 2018).
- German Federal Government. Action Plan Nanotechnology 2020. 2016. Available online: https://www.bmbf.de/pub/Action_Plan_Nanotechnology.pdf (accessed on 5 March 2018).
- National Institute for Nanotechnology (Nint) Homepage. Available online: http://www.nint-innt.ca (accessed on 27 February 2018).
- Nanoportal. Available online: http://nanoportal.gc.ca/default.asp?lang=En&n=9156F3D3-1 (accessed on 27 February 2018).
- CDC—Nanotechnology—Niosh Workplace Safety and Health Topic. Available online: https://www.cdc.gov/niosh/topics/nanotech/ (accessed on 27 February 2018).
- Safety of Manufactured Nanomaterials—OECD. Available online: http://www.oecd.org/science/nanosafety/ (accessed on 28 February 2018).
- Harremoes, P.; Gee, D.; MacGarvin, M.; Stirling, A.; Keys, J.; Wynne, B.; Guedes Vaz, S. Late Lessons from Early Warnings: The Precautionary Principle 1896–2000; European Environment Agency: Copenhagen, Denmark, 2001; pp. 1896–2000. Available online: https://www.eea.europa.eu/publications/environmental_issue_report_2001_22 (accessed on 3 March 2018).
- Zimmerman, J.B.; Anastas, P.T.; Miller, G.W. Green chemistry as a leadership opportunity for toxicology: We must take the wheel. Toxicol. Sci. 2014, 141, 4–5. [Google Scholar] [CrossRef] [PubMed]
- Maertens, A.; Anastas, N.; Spencer, P.J.; Stephens, M.; Goldberg, A.; Hartung, T. Green toxicology. ALTEX 2014, 31, 243–249. [Google Scholar] [CrossRef] [PubMed]
- Krug, H.F.; Wick, P. Nanotoxicology: An interdisciplinary challenge. Angew. Chem. Int. Ed. Engl. 2011, 50, 1260–1278. [Google Scholar] [CrossRef] [PubMed]
- Kunzmann, A.; Andersson, B.; Thurnherr, T.; Krug, H.; Scheynius, A.; Fadeel, B. Toxicology of engineered nanomaterials: Focus on biocompatibility, biodistribution and biodegradation. Biochim. Biophys. Acta 2011, 1810, 361–373. [Google Scholar] [CrossRef] [PubMed]
- Kühnel, D.; Marquardt, C.; Nau, K.; Krug, H.F.; Mathes, B.; Steinbach, C. Environmental impacts of nanomaterials: Providing comprehensive information on exposure, transport and ecotoxicity—The project dana2.0. Environ. Sci. Eur. 2014, 26, 21. [Google Scholar] [CrossRef]
- Hristozov, D.R.; Gottardo, S.; Critto, A.; Marcomini, A. Risk assessment of engineered nanomaterials: A review of available data and approaches from a regulatory perspective. Nanotoxicology 2012, 6, 880–898. [Google Scholar] [CrossRef] [PubMed]
- Krug, H.F. Nanosafety research—Are we on the right track? Angew. Chem. Int. Ed. Engl. 2014, 53, 12304–12319. [Google Scholar] [CrossRef] [PubMed]
- Crist, R.M.; Grossman, J.H.; Patri, A.K.; Stern, S.T.; Dobrovolskaia, M.A.; Adiseshaiah, P.P.; Clogston, J.D.; McNeil, S.E. Common pitfalls in nanotechnology: Lessons learned from nci’s nanotechnology characterization laboratory. Integr. Biol. 2013, 5, 66–73. [Google Scholar] [CrossRef] [PubMed]
- Hirsch, C.; Roesslein, M.; Krug, H.F.; Wick, P. Nanomaterial cell interactions: Are current in vitro tests reliable? Nanomedicine 2011, 6, 837–847. [Google Scholar] [CrossRef] [PubMed]
- Kroll, A.; Pillukat, M.H.; Hahn, D.; Schnekenburger, J. Interference of engineered nanoparticles with in vitro toxicity assays. Arch. Toxicol. 2012, 86, 1123–1136. [Google Scholar] [CrossRef] [PubMed]
- Petersen, E.J.; Henry, T.B.; Zhao, J.; MacCuspie, R.I.; Kirschling, T.L.; Dobrovolskaia, M.A.; Hackley, V.; Xing, B.; White, J.C. Identification and avoidance of potential artifacts and misinterpretations in nanomaterial ecotoxicity measurements. Environ. Sci. Technol. 2014, 48, 4226–4246. [Google Scholar] [CrossRef] [PubMed]
- Rosslein, M.; Elliott, J.T.; Salit, M.; Petersen, E.J.; Hirsch, C.; Krug, H.F.; Wick, P. Use of cause-and-effect analysis to design a high-quality nanocytotoxicology assay. Chem. Res. Toxicol. 2015, 28, 21–30. [Google Scholar] [CrossRef] [PubMed]
- Schulze, C.; Schulze, C.; Kroll, A.; Schulze, C.; Kroll, A.; Lehr, C.M.; Schäfer, U.F.; Becker, K.; Schnekenburger, J.; Schulze Isfort, C.; et al. Not ready to use—Overcoming pitfalls when dispersing nanoparticles in physiological media. Nanotoxicology 2008, 2, 51–61. [Google Scholar] [CrossRef]
- Wittmaack, K. Excessive delivery of nanostructured matter to submersed cells caused by rapid gravitational settling. ACS Nano 2011, 5, 3766–3778. [Google Scholar] [CrossRef] [PubMed]
- Jemec, A.; Kahru, A.; Potthoff, A.; Drobne, D.; Heinlaan, M.; Bohme, S.; Geppert, M.; Novak, S.; Schirmer, K.; Rekulapally, R.; et al. An interlaboratory comparison of nanosilver characterisation and hazard identification: Harmonising techniques for high quality data. Environ. Int. 2016, 87, 20–32. [Google Scholar] [CrossRef] [PubMed]
- Donaldson, K.; Poland, C.A. Nanotoxicity: Challenging the myth of nano-specific toxicity. Curr. Opin. Biotechnol. 2013, 24, 724–734. [Google Scholar] [CrossRef] [PubMed]
- Oostingh, G.J.; Casals, E.; Italiani, P.; Colognato, R.; Stritzinger, R.; Ponti, J.; Pfaller, T.; Kohl, Y.; Ooms, D.; Favilli, F.; et al. Problems and challenges in the development and validation of human cell-based assays to determine nanoparticle-induced immunomodulatory effects. Part. Fibre Toxicol. 2011, 8, 8. [Google Scholar] [CrossRef] [PubMed]
- Pulskamp, K.; Diabate, S.; Krug, H.F. Carbon nanotubes show no sign of acute toxicity but induce intracellular reactive oxygen species in dependence on contaminants. Toxicol. Lett. 2007, 168, 58–74. [Google Scholar] [CrossRef] [PubMed]
- Smulders, S.; Kaiser, J.P.; Zuin, S.; Van Landuyt, K.L.; Golanski, L.; Vanoirbeek, J.; Wick, P.; Hoet, P.H. Contamination of nanoparticles by endotoxin: Evaluation of different test methods. Part. Fibre Toxicol. 2012, 9, 41. [Google Scholar] [CrossRef] [PubMed]
- Dana—Literature Criteria Checklist. Available online: https://nanopartikel.info/en/nanoinfo/methods/991-literature-criteria-checklist (accessed on 28 February 2018).
- Marquardt, C.; Kühnel, D.; Richter, V.; Krug, H.F.; Mathes, B.; Steinbach, C.; Nau, K. Latest research results on the effects of nanomaterials on humans and the environment: Dana—Knowledge base nanomaterials. J. Phys. Conf. Ser. 2013, 429, 012060. [Google Scholar] [CrossRef]
- Fadeel, B.; Fornara, A.; Toprak, M.S.; Bhattacharya, K. Keeping it real: The importance of material characterization in nanotoxicology. Biochem. Biophys. Res. Commun. 2015, 468, 498–503. [Google Scholar] [CrossRef] [PubMed]
- Mills, K.C.; Murry, D.; Guzan, K.A.; Ostraat, M.L. Nanomaterial registry: Database that captures the minimal information about nanomaterial physico-chemical characteristics. J. Nanopart. Res. 2014, 16, 2219. [Google Scholar] [CrossRef]
- Stefaniak, A.B.; Hackley, V.A.; Roebben, G.; Ehara, K.; Hankin, S.; Postek, M.T.; Lynch, I.; Fu, W.E.; Linsinger, T.P.; Thunemann, A.F. Nanoscale reference materials for environmental, health and safety measurements: Needs, gaps and opportunities. Nanotoxicology 2013, 7, 1325–1337. [Google Scholar] [CrossRef] [PubMed]
- Hendren, C.O.; Mesnard, X.; Droge, J.; Wiesner, M.R. Estimating production data for five engineered nanomaterials as a basis for exposure assessment. Environ. Sci. Technol. 2011, 45, 2562–2569. [Google Scholar] [CrossRef] [PubMed]
- Dana—Application & Nanomaterials Short Info—Knowledge Base Nanomaterials. Available online: https://www.nanopartikel.info/en/nanoinfo/knowledge-base (accessed on 28 February 2018).
- Piwik/Matomo—Free and Mobile Web Analytics Software. Available online: https://matomo.org/ (accessed on 28 February 2018).
- Google Analytics Solutions—Marketing Analytics and Measurement. Available online: https://www.google.com/intl/en_uk/analytics/ (accessed on 28 February 2018).
- Dana—Faq Nanoparticles—Knowledge Base Nanomaterials. Available online: http://www.nanopartikel.info/en/faq (accessed on 28 February 2018).
- Dana—Contact—Knoledge Base Nanomaterials. Available online: http://www.nanopartikel.info/en/contact (accessed on 28 February 2018).
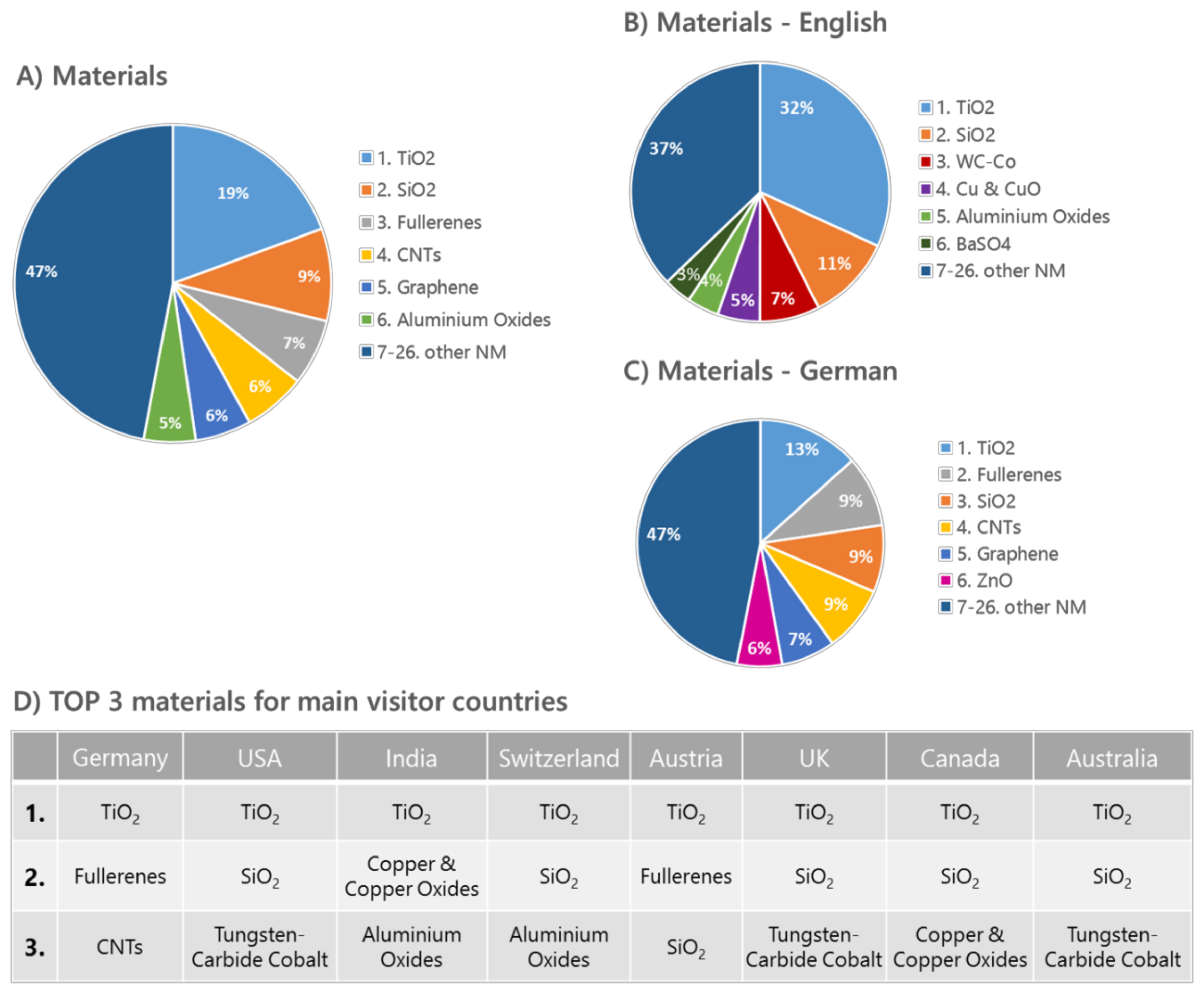
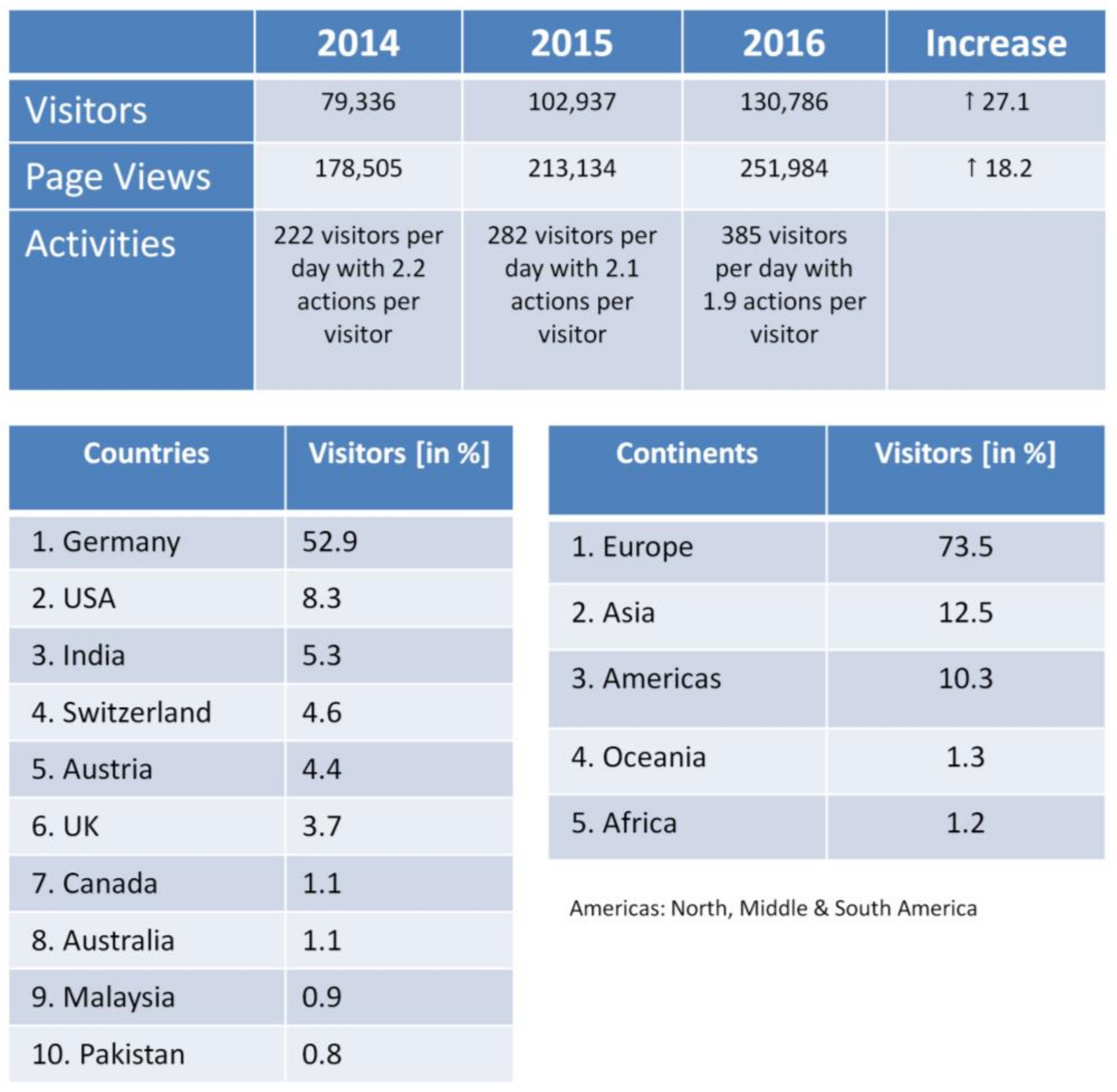
| Documents | Downloads | % of Total Downloads |
|---|---|---|
| Overall download activities | 6129 | 100% |
| DaNa SOP template | 139 | 2% |
| SOPs from Projects | 302 | 5% |
| Literature Criteria Checklist | 149 | 2% |
| NanoCare Datasheets | ||
| TiO2 | 3590 | 59% |
| ZrO2 | 403 | 7% |
| Carbon Black | 256 | 4% |
| CeO2 | 248 | 4% |
| ZnO | 226 | 4% |
| BaSO4 | 194 | 3% |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Krug, H.F.; Bohmer, N.; Kühnel, D.; Marquardt, C.; Nau, K.; Steinbach, C. The DaNa2.0 Knowledge Base Nanomaterials—An Important Measure Accompanying Nanomaterials Development. Nanomaterials 2018, 8, 204. https://doi.org/10.3390/nano8040204
Krug HF, Bohmer N, Kühnel D, Marquardt C, Nau K, Steinbach C. The DaNa2.0 Knowledge Base Nanomaterials—An Important Measure Accompanying Nanomaterials Development. Nanomaterials. 2018; 8(4):204. https://doi.org/10.3390/nano8040204
Chicago/Turabian StyleKrug, Harald F., Nils Bohmer, Dana Kühnel, Clarissa Marquardt, Katja Nau, and Christoph Steinbach. 2018. "The DaNa2.0 Knowledge Base Nanomaterials—An Important Measure Accompanying Nanomaterials Development" Nanomaterials 8, no. 4: 204. https://doi.org/10.3390/nano8040204
APA StyleKrug, H. F., Bohmer, N., Kühnel, D., Marquardt, C., Nau, K., & Steinbach, C. (2018). The DaNa2.0 Knowledge Base Nanomaterials—An Important Measure Accompanying Nanomaterials Development. Nanomaterials, 8(4), 204. https://doi.org/10.3390/nano8040204





