Thylakoid-Deposited Micro-Pillar Electrodes for Enhanced Direct Extraction of Photosynthetic Electrons
Abstract
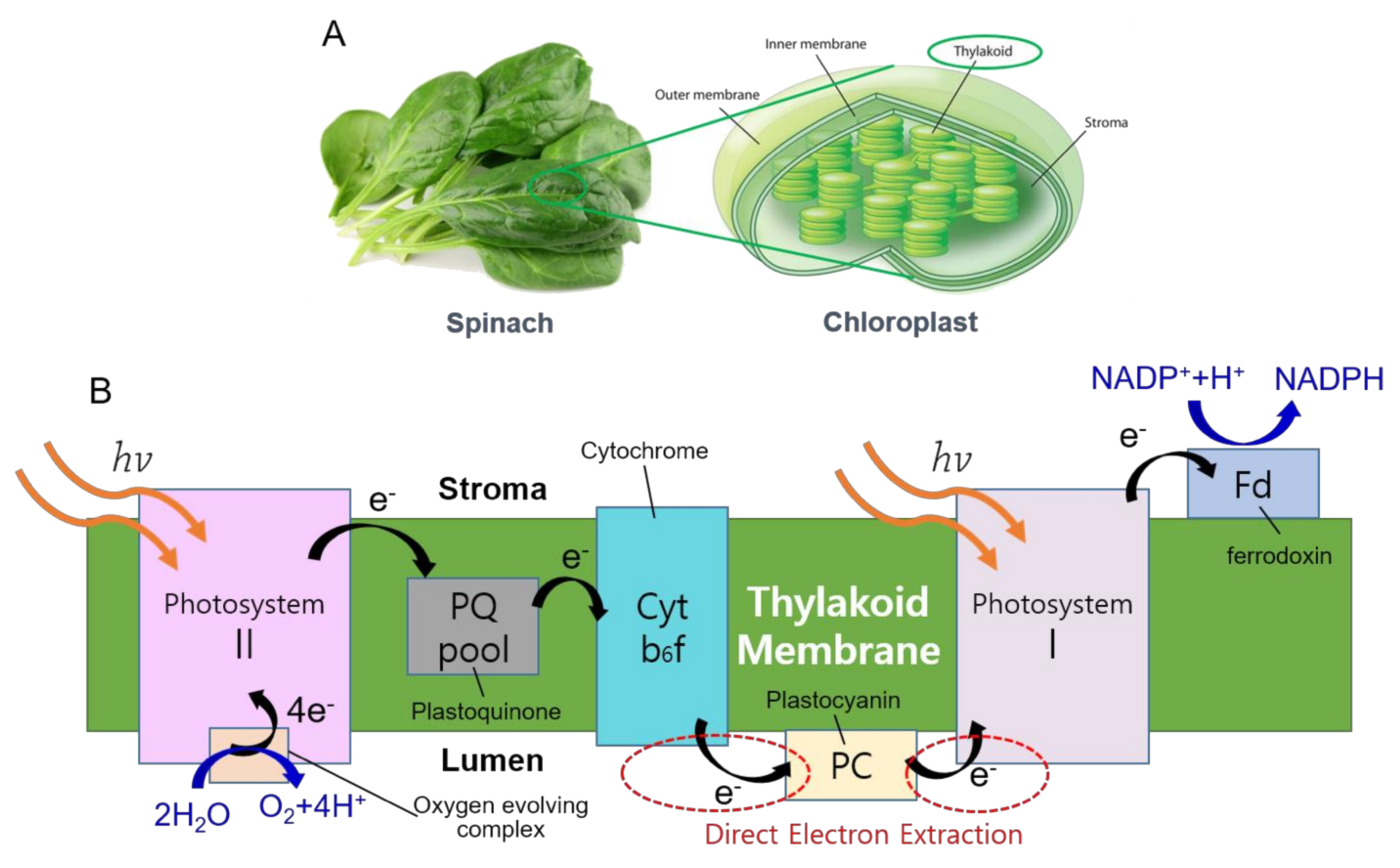
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Thylakoids Isolation
2.3. Measurement of Chlorophyll Concentration
2.4. SEM Imaging of Thylakoids
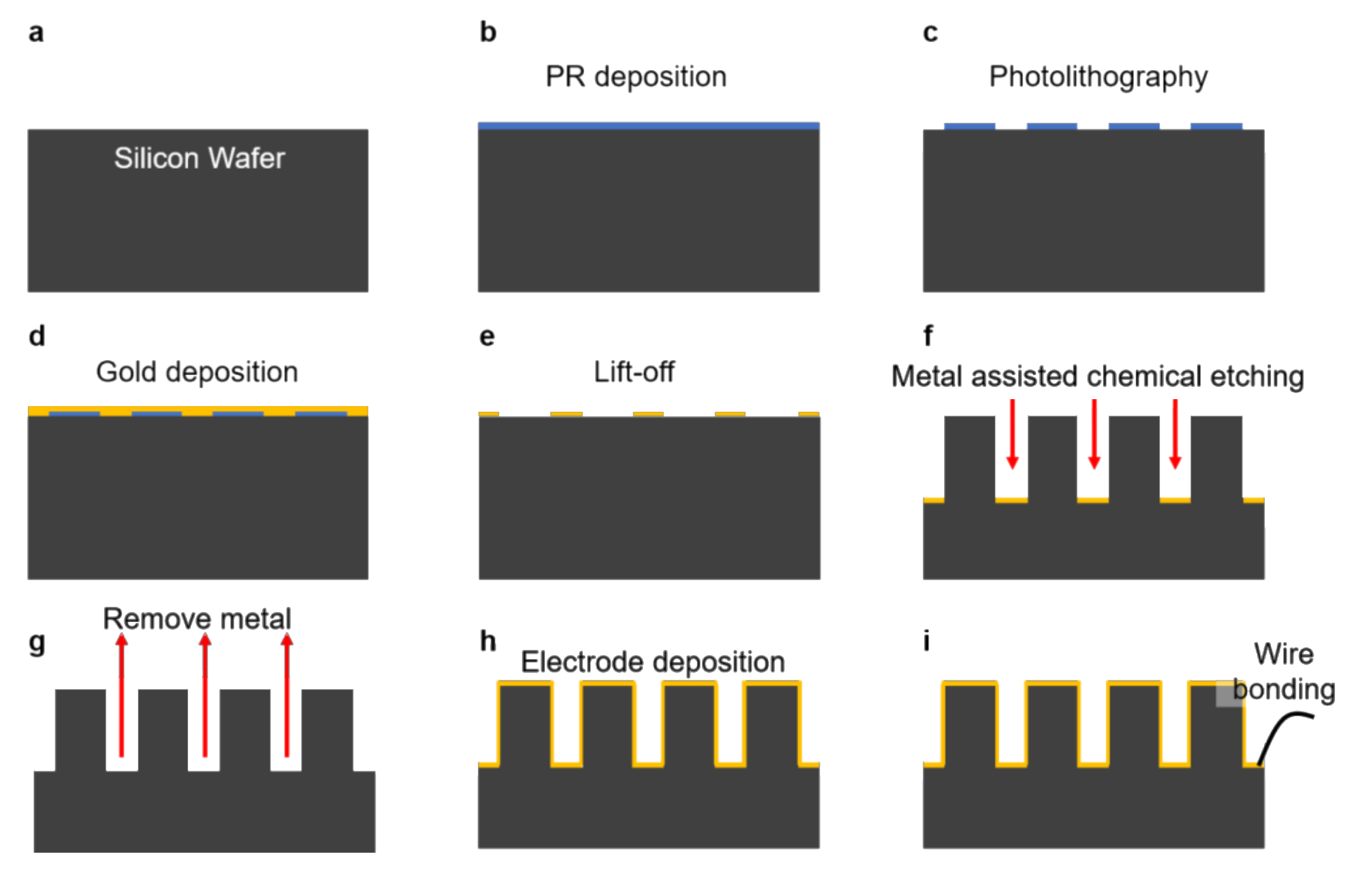
2.5. Fabrication of MP Electrode
2.6. Measurement of Photosynthetic Currents
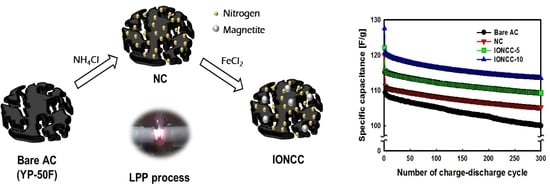
3. Results
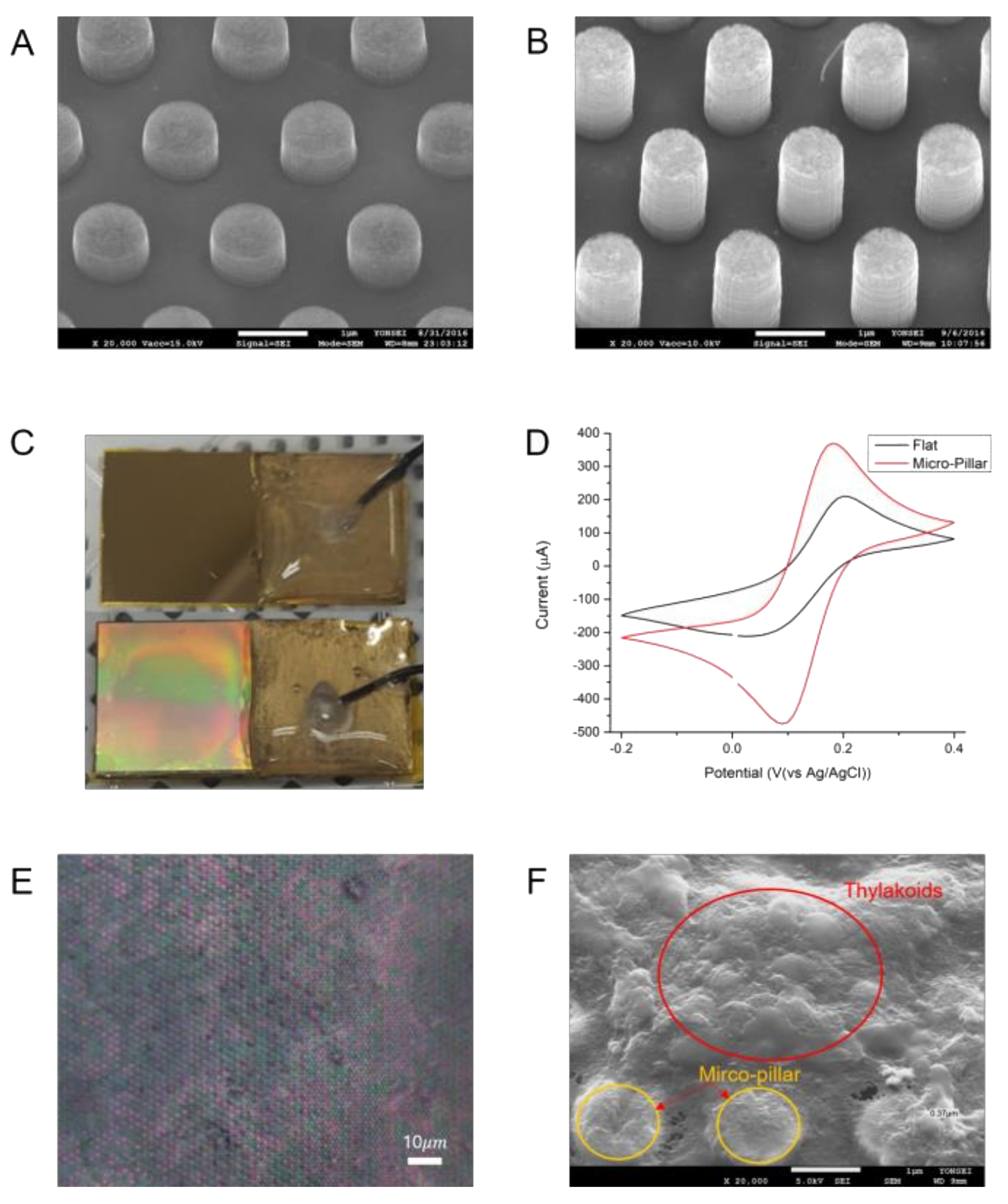
3.1. Design and Fabrication of MP Electrodes
3.2. Electrochemical Surface Area of MP Electrode
3.3. Electrochemical Impedance Spectroscopy
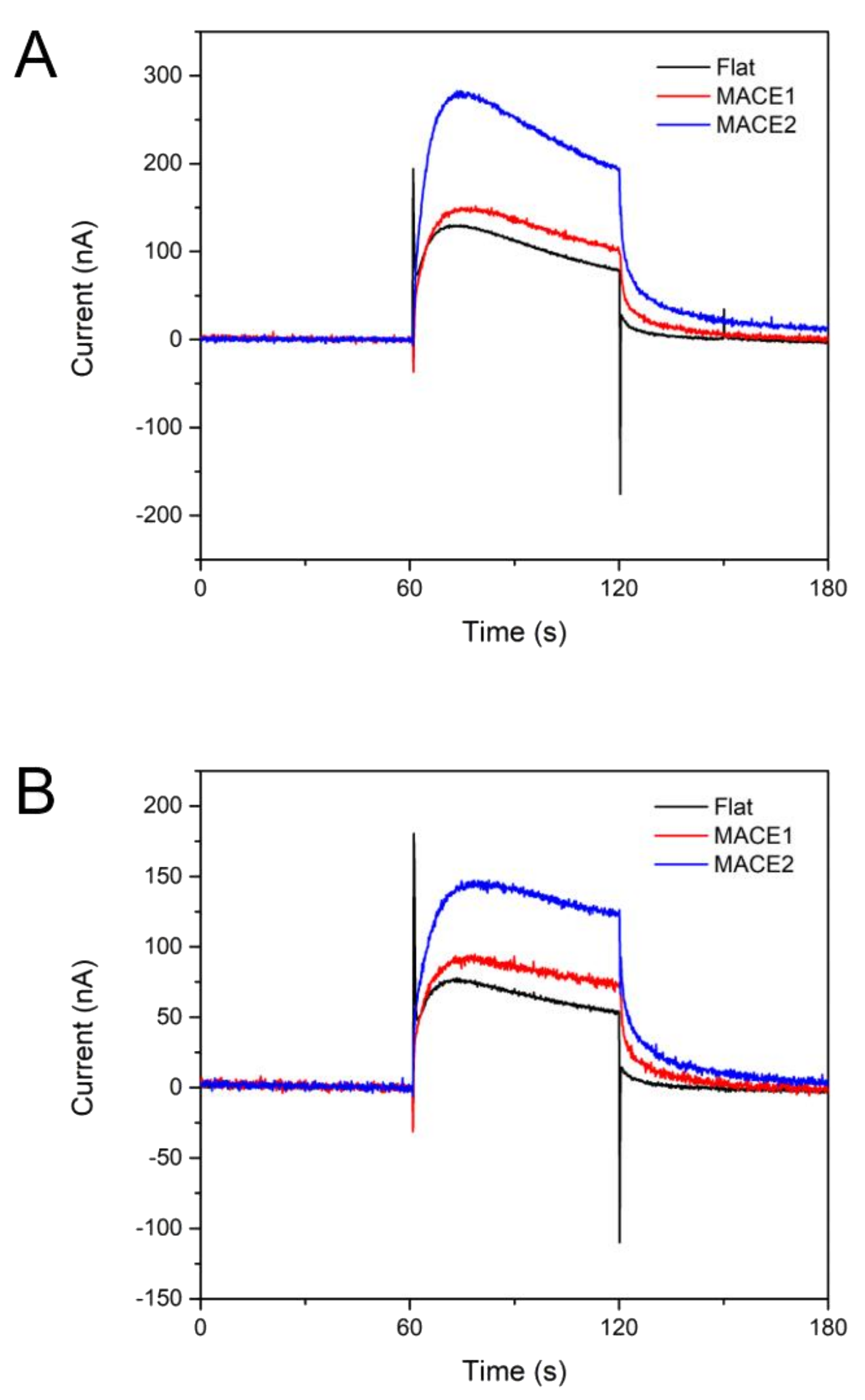
3.4. Measurement of Photosynthetic Currents
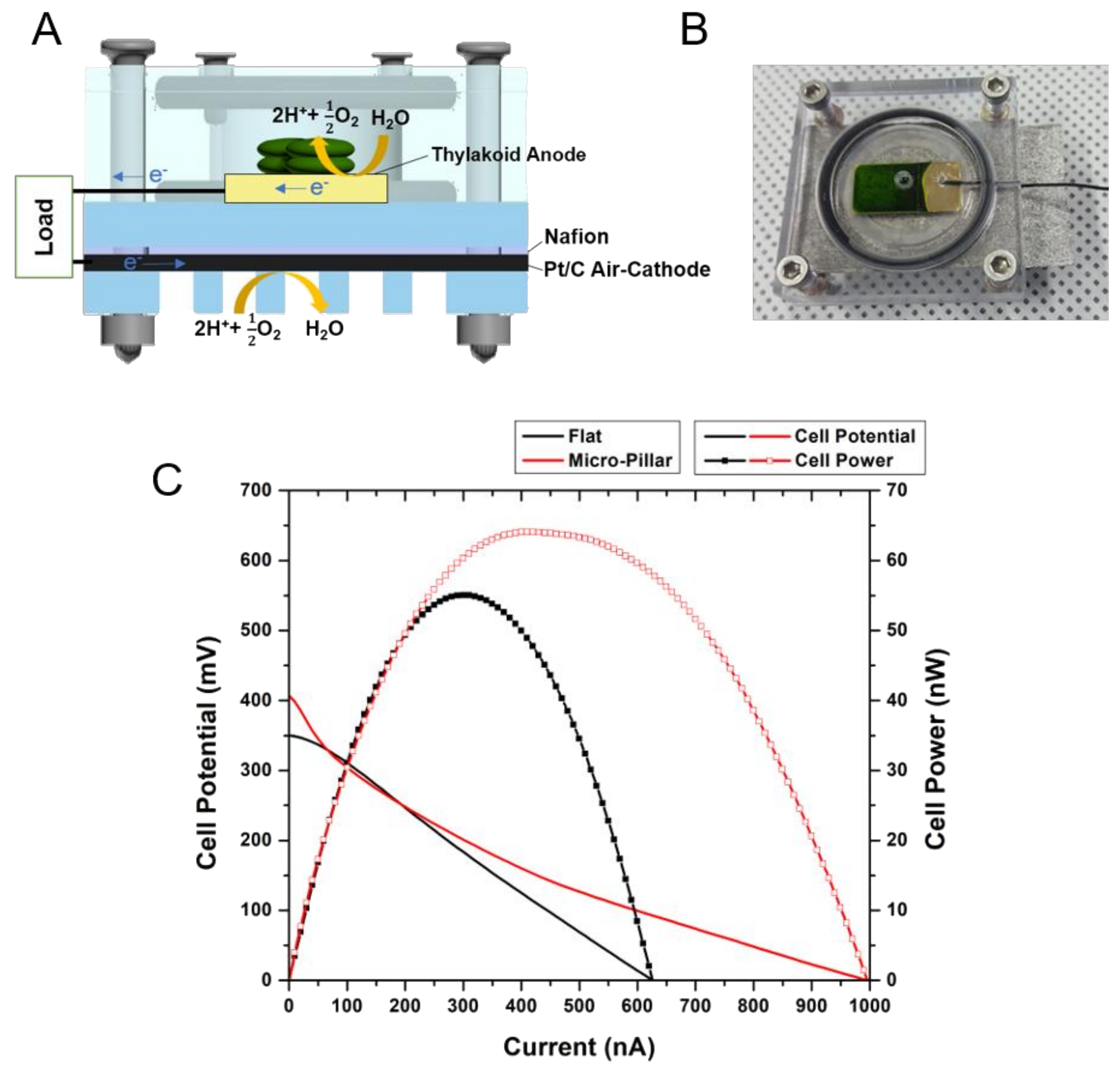
3.5. Photosynthetic Fuel Cell Operation
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Mohan, S.V.; Velvizhi, G.; Modestra, J.A.; Srikanth, S. Microbial fuel cell: Critical factors regulating bio-catalyzed electrochemical process and recent advancements. Renew. Sustain. Energy Rev. 2014, 40, 779–797. [Google Scholar] [CrossRef]
- Sekar, N.; Ramasamy, R.P. Recent advances in photosynthetic energy conversion. J. Photochem. Photobiol. C 2015, 22, 19–33. [Google Scholar] [CrossRef]
- Terasaki, N.; Iwai, M.; Yamamoto, N.; Hiraga, T.; Yamada, S.; Inoue, Y. Photocurrent generation properties of Histag-photosystem II immobilized on nanostructured gold electrode. Thin Solid Films 2008, 516, 2553–2557. [Google Scholar] [CrossRef][Green Version]
- Yehezkeli, O.; Tel-Vered, R.; Wasserman, J.; Trifonov, A.; Michaeli, D.; Nechushtai, R.; Willner, I. Integrated photosystem II-based photo-bioelectrochemical cells. Nat. Commun. 2012, 3, 742. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Wang, H.; Zhu, Q.; Qin, W.; Han, G.; Shen, J.-R.; Zong, X.; Li, C. Spatially separated photosystem II and a silicon photoelectrochemical cell for overall water splitting: A natural–artificial photosynthetic hybrid. Angew. Chem. Int. Ed. 2016, 55, 9229–9233. [Google Scholar] [CrossRef] [PubMed]
- Ciesielski, P.N.; Hijazi, F.M.; Scott, A.M.; Faulkner, C.J.; Beard, L.; Emmett, K.; Rosenthal, S.J.; Cliffel, D.; Kane Jennings, G. Photosystem I—Based biohybrid photoelectrochemical cells. Bioresour. Technol. 2010, 101, 3047–3053. [Google Scholar] [CrossRef] [PubMed]
- LeBlanc, G.; Chen, G.; Gizzie, E.A.; Jennings, G.K.; Cliffel, D.E. Enhanced photocurrents of photosystem I films on p-doped silicon. Adv. Mater. 2012, 24, 5959–5962. [Google Scholar] [CrossRef] [PubMed]
- Gizzie, E.A.; Niezgoda, J.S.; Robinson, M.T.; Harris, A.G.; Jennings, G.K.; Rosenthal, S.J.; Cliffel, D.E. Photosystem I-polyaniline/TiO2 solid-state solar cells: Simple devices for biohybrid solar energy conversion. Energy Environ. Sci. 2015, 8, 3572–3576. [Google Scholar] [CrossRef]
- Ryu, W.; Bai, S.J.; Park, J.S.; Huang, Z.B.; Moseley, J.; Fabian, T.; Fasching, R.J.; Grossman, A.R.; Prinz, F.B. Direct extraction of photosynthetic electrons from single algal cells by nanoprobing system. Nano Lett. 2010, 10, 1137–1143. [Google Scholar] [CrossRef] [PubMed]
- Kim, L.H.; Kim, Y.J.; Hong, H.; Yang, D.; Han, M.; Yoo, G.; Song, H.W.; Chae, Y.; Pyun, J.C.; Grossman, A.R.; et al. Patterned nanowire electrode array for direct extraction of photosynthetic electrons from multiple living algal cells. Adv. Funct. Mater. 2016, 26, 7679–7689. [Google Scholar] [CrossRef]
- Calkins, J.O.; Umasankar, Y.; O’Neill, H.; Ramasamy, R.P. High photo-electrochemical activity of thylakoid–carbon nanotube composites for photosynthetic energy conversion. Energy Environ. Sci. 2013, 6, 1891–1900. [Google Scholar] [CrossRef]
- Sekar, N.; Umasankar, Y.; Ramasamy, R.P. Photocurrent generation by immobilized cyanobacteria via direct electron transport in photo-bioelectrochemical cells. Phys. Chem. Chem. Phys. 2014, 16, 7862–7871. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Zhang, P.-H.; Qiu, Y.; Zhang, J.; Yu, D.-Y. The photoelectrical properties of thylakoid membrane from two plants fabricated on Nano-ZnO. Mar. Sci. 2012, 2, 010. [Google Scholar]
- Yang, N.; Zhang, Y.; Halpert, J.E.; Zhai, J.; Wang, D.; Jiang, L. Granum-like stacking structures with TiO2–graphene nanosheets for improving photo-electric conversion. Small 2012, 8, 1762–1770. [Google Scholar] [CrossRef] [PubMed]
- Pankratova, G.; Pankratov, D.; Di Bari, C.; Goñi-Urtiaga, A.; Toscano, M.D.; Chi, Q.; Pita, M.; Gorton, L.; De Lacey, A.L. 3D graphene matrix supported and thylakoid membranes based high-performance bioelectrochemical solar cell. ACS Appl. Energy Mater. 2018, 1, 319–323. [Google Scholar] [CrossRef]
- Carpentier, R. Photosynthesis Research Protocols; Humana Press: New York, NY, USA, 2004; Volume 274. [Google Scholar]
- Arnon, D.I. Copper enzymes in isolated chloroplasts. Polyphenoloxidase in beta vulgaris. Plant Physiol. 1949, 24, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Granick, S.; Porter, K. The structure of the spinach chloroplast as interpreted with the electron microscope. Am. J. Bot. 1947, 545–550. [Google Scholar] [CrossRef]
- Dekker, J.P.; Boekema, E.J. Supramolecular organization of thylakoid membrane proteins in green plants. Biochim. Biophys. Acta 2005, 1706, 12–39. [Google Scholar] [CrossRef] [PubMed]
- Austin, J.R.; Staehelin, L.A. Three-dimensional architecture of grana and stroma thylakoids of higher plants as determined by electron tomography. Plant Physiol. 2011, 155, 1601–1611. [Google Scholar] [CrossRef] [PubMed]
- Bard, A.; Faulkner, L. Electrochemical Methods: Fundamentals and Applications, 2nd ed.; Wiley: New York, NY, USA, 2001. [Google Scholar]
- Sekar, N.; Ramasamy, R.P. Electrochemical impedance spectroscopy for microbial fuel cell characterization. J. Microb. Biochem. Technol. 2013, S6, 4. [Google Scholar] [CrossRef]
- O’hayre, R.; Cha, S.-W.; Prinz, F.B.; Colella, W. Fuel Cell Fundamentals; John Wiley & Sons: Hoboken, NJ, USA, 2016. [Google Scholar]
- Huang, Z.P.; Geyer, N.; Werner, P.; de Boor, J.; Gosele, U. Metal-assisted chemical etching of silicon: A review. Adv. Mater. 2011, 23, 285–308. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.L.; Peng, K.Q.; Fan, X.; Jie, J.S.; Zhang, R.Q.; Lee, S.T.; Wong, N.B. Preparation of large-area uniform silicon nanowires arrays through metal-assisted chemical etching. J. Phys. Chem. C 2008, 112, 4444–4450. [Google Scholar] [CrossRef]
- Li, X. Metal assisted chemical etching for high aspect ratio nanostructures: A review of characteristics and applications in photovoltaics. Curr. Opin. Solid State Mater. Sci. 2012, 16, 71–81. [Google Scholar] [CrossRef]
- Sjoholm, K.H.; Rasmussen, M.; Minteer, S.D. Bio-solar cells incorporating catalase for stabilization of thylakoid bioelectrodes during direct photoelectrocatalysis. ECS Electrochem. Lett. 2012, 1, G7–G9. [Google Scholar] [CrossRef]
- Lee, J.; Im, J.; Kim, S. Mediatorless solar energy conversion by covalently bonded thylakoid monolayer on the glassy carbon electrode. Bioelectrochemistry 2016, 108, 21–27. [Google Scholar] [CrossRef] [PubMed]
- Hamidi, H.; Hasan, K.; Emek, S.C.; Dilgin, Y.; Akerlund, H.E.; Albertsson, P.A.; Leech, D.; Gorton, L. Photocurrent generation from thylakoid membranes on osmium-redox-polymer-modified electrodes. Chemsuschem 2015, 8, 990–993. [Google Scholar] [CrossRef] [PubMed]
- Dewi, H.A.; Meng, F.B.; Sana, B.; Guo, C.X.; Norling, B.; Chen, X.D.; Lim, S.R. Investigation of electron transfer from isolated spinach thylakoids to indium tin oxide. RSC Adv. 2014, 4, 48815–48820. [Google Scholar] [CrossRef]
- Asada, K. Production and scavenging of reactive oxygen species in chloroplasts and their functions. Plant Physiol. 2006, 141, 391–396. [Google Scholar] [CrossRef] [PubMed]
- Yeung, C.H.; de Geyter, C.; de Geyter, M.; Nieschlag, E. Production of reactive oxygen species by and hydrogen peroxide scavenging activity of spermatozoa in an IVF program. J. Assist. Reprod. Genet. 1996, 13, 495–500. [Google Scholar] [CrossRef] [PubMed]
- Rasmussen, M.; Wingersky, A.; Minteer, S.D. Improved performance of a thylakoid bio-solar cell by incorporation of carbon quantum dots. ECS Electrochem. Lett. 2014, 3, H1–H3. [Google Scholar] [CrossRef]

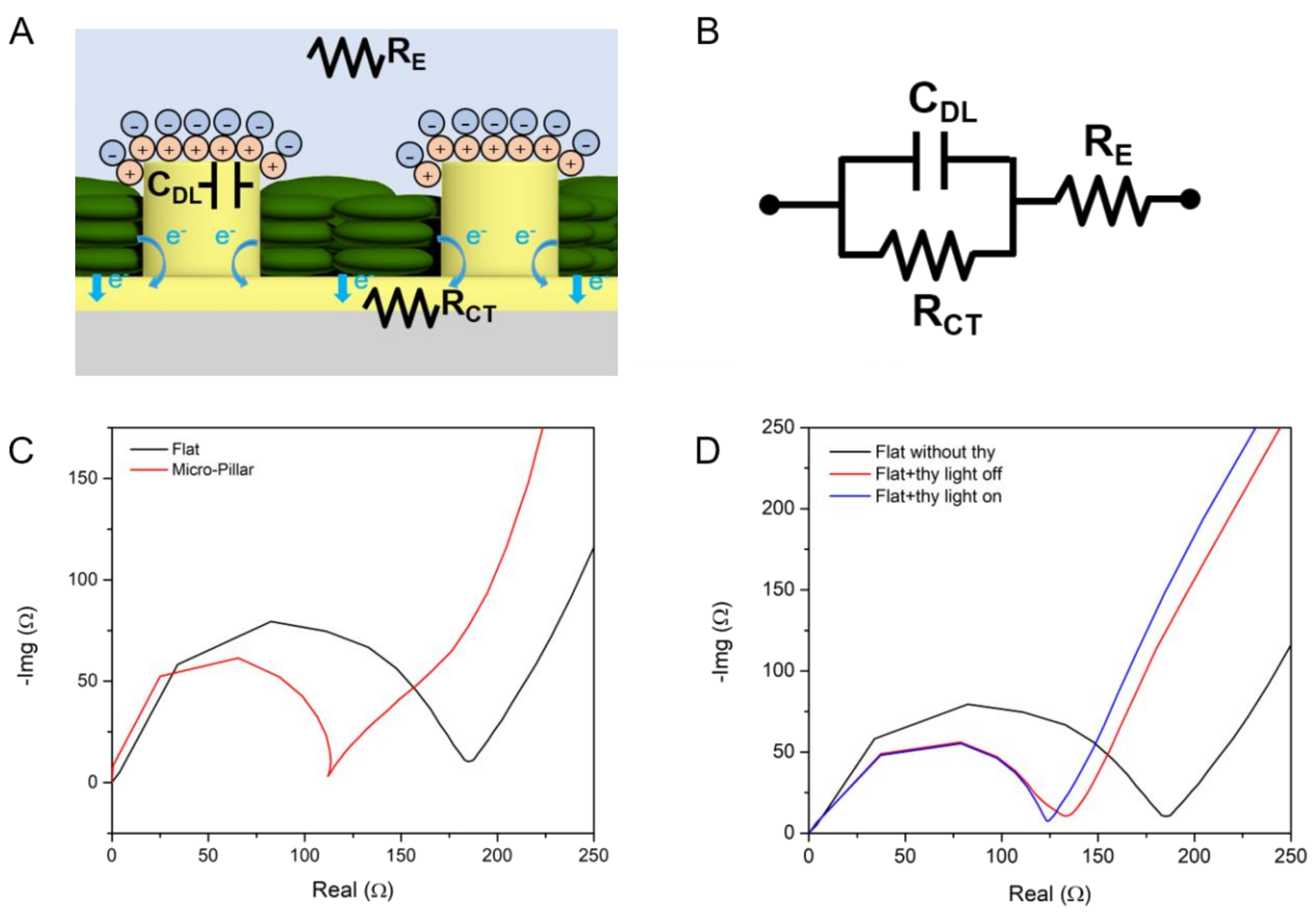
| RE (Ω) | RCT (Ω) | RINT (Ω) | CDL (F) | |
|---|---|---|---|---|
| Flat electrode | 10.3 | 165.8 | 176.1 | 1.32 × 10−9 |
| MP electrode | 4.0 | 109.9 | 113.9 | 1.36 × 10−9 |
| RE (Ω) | RCT (Ω) | RINT (Ω) | |
|---|---|---|---|
| No thylakoid | 10.2 | 165.8 | 176.1 |
| With thylakoid (light off) | 9.5 | 117 | 126.5 |
| With thylakoid (light on) | 9.0 | 112.7 | 121.7 |
| Surface Area Ratio | Charge Generated (1st Cycle) | Charge Generated (2nd Cycle) | |
|---|---|---|---|
| Flat | 100% | 6443 C | 3877 C |
| MACE 1 | 133% | 8014 C | 5136 C |
| MACE 2 | 236% | 15,545 C | 8609 C |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ryu, D.; Kim, Y.J.; Kim, S.I.; Hong, H.; Ahn, H.S.; Kim, K.; Ryu, W. Thylakoid-Deposited Micro-Pillar Electrodes for Enhanced Direct Extraction of Photosynthetic Electrons. Nanomaterials 2018, 8, 189. https://doi.org/10.3390/nano8040189
Ryu D, Kim YJ, Kim SI, Hong H, Ahn HS, Kim K, Ryu W. Thylakoid-Deposited Micro-Pillar Electrodes for Enhanced Direct Extraction of Photosynthetic Electrons. Nanomaterials. 2018; 8(4):189. https://doi.org/10.3390/nano8040189
Chicago/Turabian StyleRyu, DongHyun, Yong Jae Kim, Seon Il Kim, Hyeonaug Hong, Hyun S. Ahn, Kyunghoon Kim, and WonHyoung Ryu. 2018. "Thylakoid-Deposited Micro-Pillar Electrodes for Enhanced Direct Extraction of Photosynthetic Electrons" Nanomaterials 8, no. 4: 189. https://doi.org/10.3390/nano8040189
APA StyleRyu, D., Kim, Y. J., Kim, S. I., Hong, H., Ahn, H. S., Kim, K., & Ryu, W. (2018). Thylakoid-Deposited Micro-Pillar Electrodes for Enhanced Direct Extraction of Photosynthetic Electrons. Nanomaterials, 8(4), 189. https://doi.org/10.3390/nano8040189