The Effect of Ni Addition onto a Cu-Based Ternary Support on the H2 Production over Glycerol Steam Reforming Reaction
Abstract
1. Introduction
2. Materials and Methods
2.1. Catalyst Preparation
2.2. Catalyst Characterization
2.3 Catalytic Tests
2.4. Reaction Metrics
3. Results and Discussion
3.1. Characterization Results
3.1.1. Microstructural Characterization
3.1.2. Textural and Morphological Studies
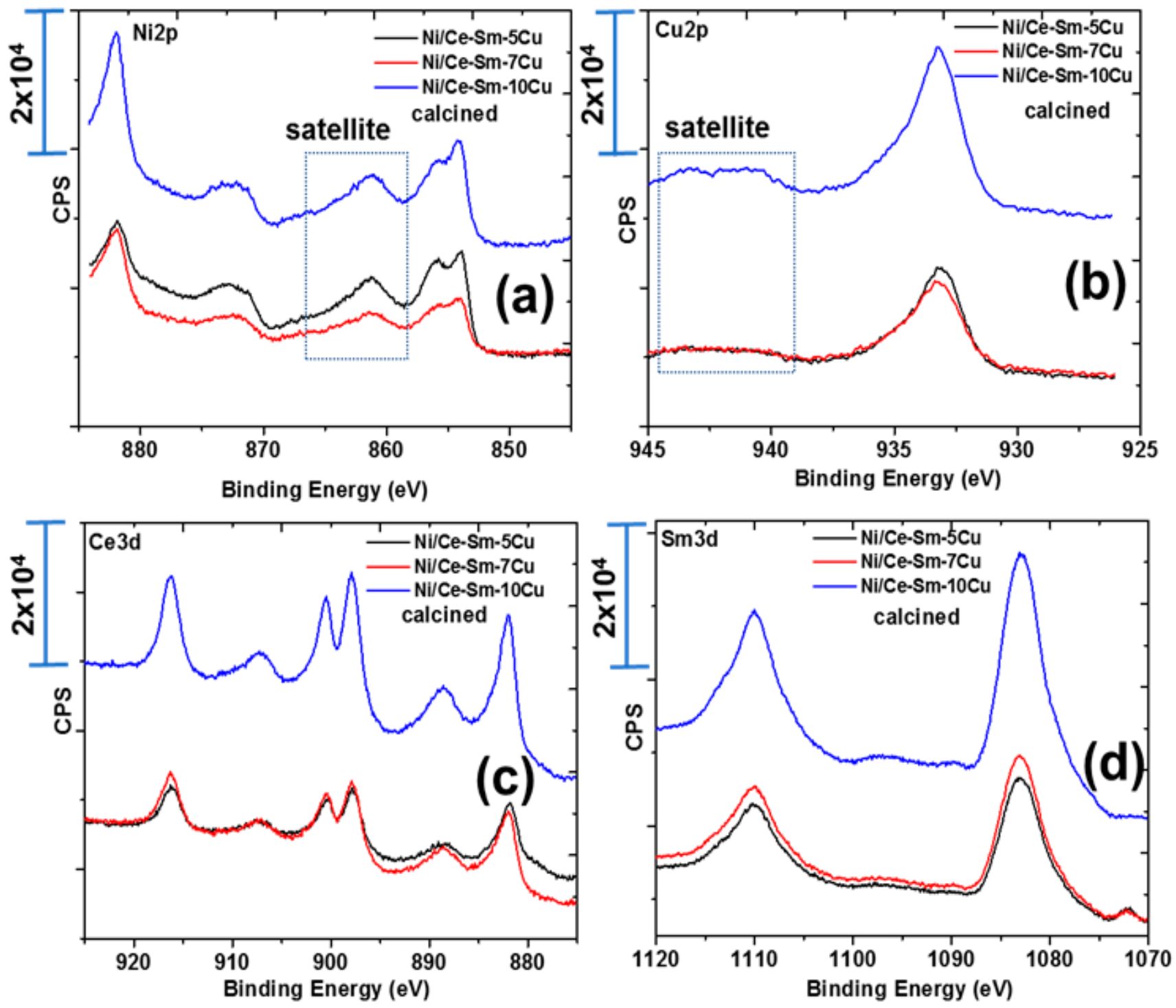
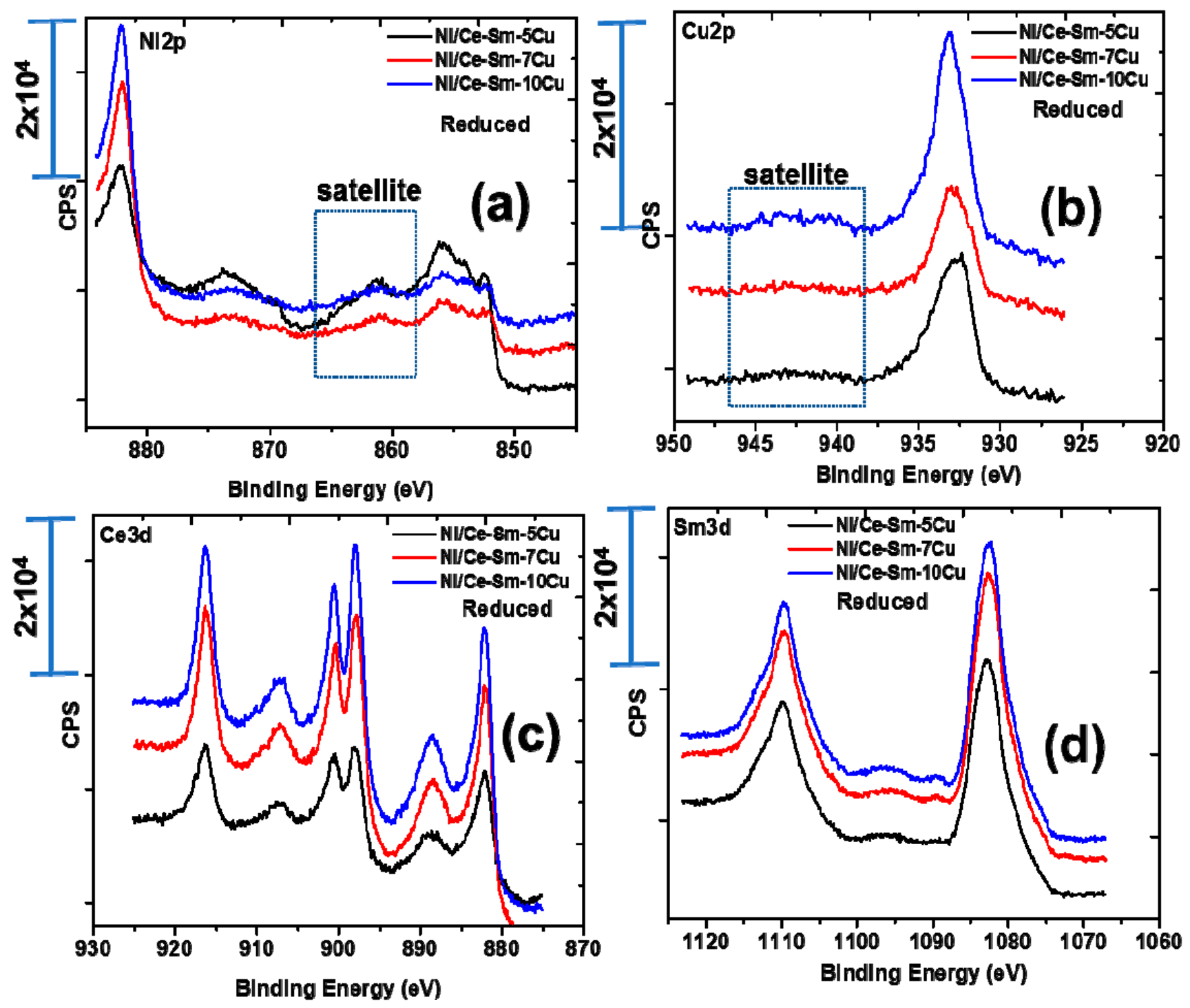
3.1.3. Surface and Redox Properties
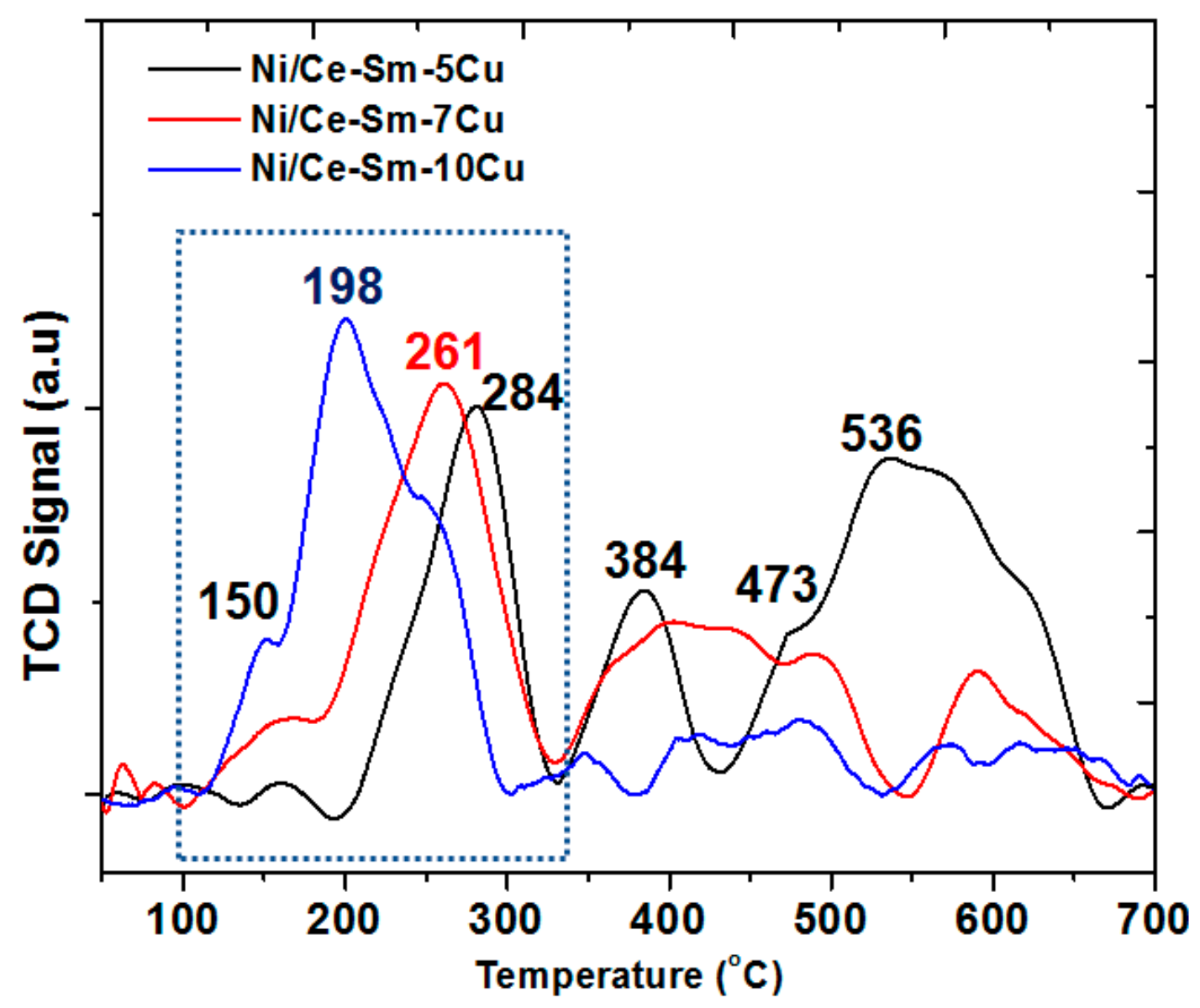
- In the case of 10Ni/CeO2 catalyst, two reduction peaks were found at higher temperatures, compared to the 10Cu/CeO2 catalyst, namely at 271 °C and 357 °C. Also, for the 10Ni/CeO2 catalyst, the Ni2+ to metallic Ni0 reduction took place at 357 °C. In the case where some of the Ni2+ ions have been incorporated into ceria lattice, then their reduction took place at higher temperatures (425–450 °C). Herein, this broad peak was noticed only in the case of Ni-Ce-Sm-7Cu and a Ni/Ce-Sm-10Cu catalyst, which implies that in these cases, incorporation of Ni into the ceria fluorite structure, might have happened to a greater extent compared to the Ni/Ce-Sm-5Cu catalyst.
- According to many literature reports, the reduction of single ceria takes place at ≈500 and 830 °C for the surface and the bulk oxygen species, respectively [52].
- The low temperature reduction peak at 150 °C can be linked to the reduction of amorphous CuO, in weak interaction with the support, whereas in the 150–200 °C range, the reduction of CuO in strong interaction with the support and the partial reduction of surface CeO2 at the metal support interface takes place [53,65,66]. The peak above 200 °C can be assigned to highly dispersed NiO [53,65,66]. Among the catalysts presented in Figure 7, the Ni/Ce-Sm-5Cu has the highest reduction temperature (284 °C) compared to the Ni-Ce-Sm-7Cu (261 °C) and Ni-Ce-Sm-10Cu (198 °C) and this can be due to the Cu-rich character of this catalyst (Ni/Cu = 0.37) corroborating for some incorporation of the Ni into the ceria fluorite structure, and thus, suppressing its reduction. However, such an Ni incorporation is expected to diminish the sintering likelihood for this catalyst.
- It has been reported by Lin et al. [67] that the TPR of the CuNi bimetallic catalysts had five TPR peaks, demonstrating the complexity of the reduction process. It was also suggested that the presence of metallic Cu enhanced the reduction of the Ni. This effect might be due to the competitive growth of the two oxide phases (NiO, CuO) that leads to a reduction in their crystallite size. It is worthwhile to recall here that in Figure 1 (XRD data) only traces of NiO and CuO phases were found. In agreement with Lin et al. [67], easiest reduction took place in the case of Ni/Ce-Sm-10Cu.
3.1.4. Surface Acidity/Basicity Studies
3.2. Catalytic Performance
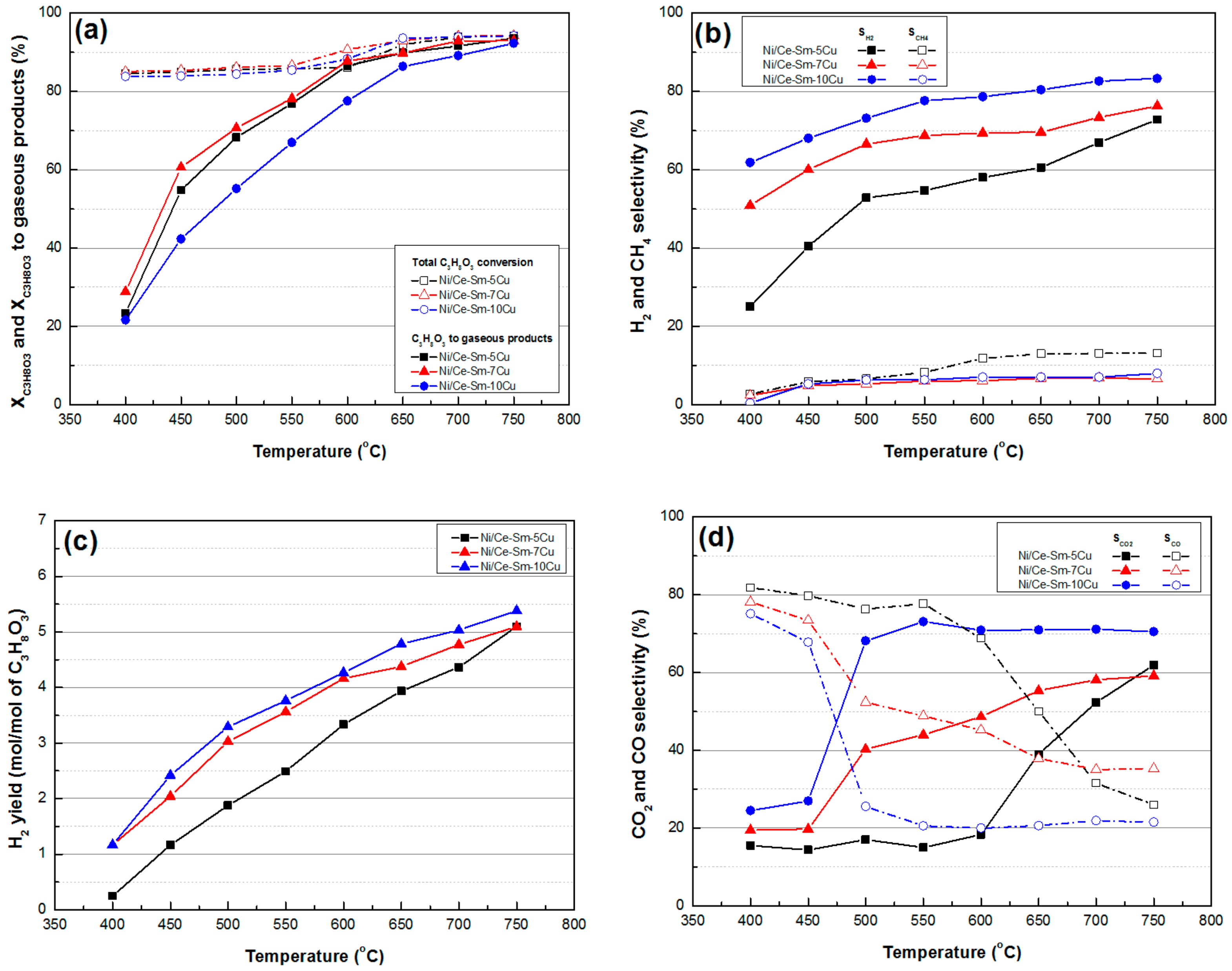
3.2.1. Glycerol Conversion and Selectivity to Gaseous Products
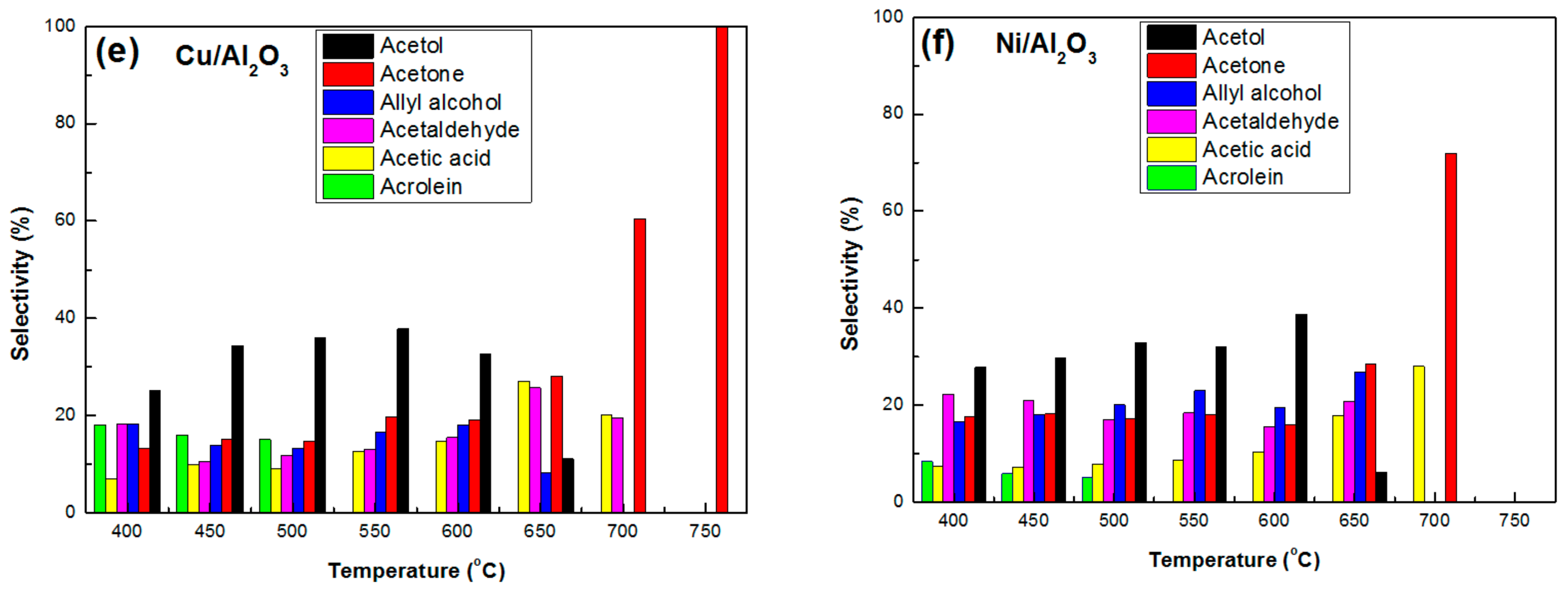
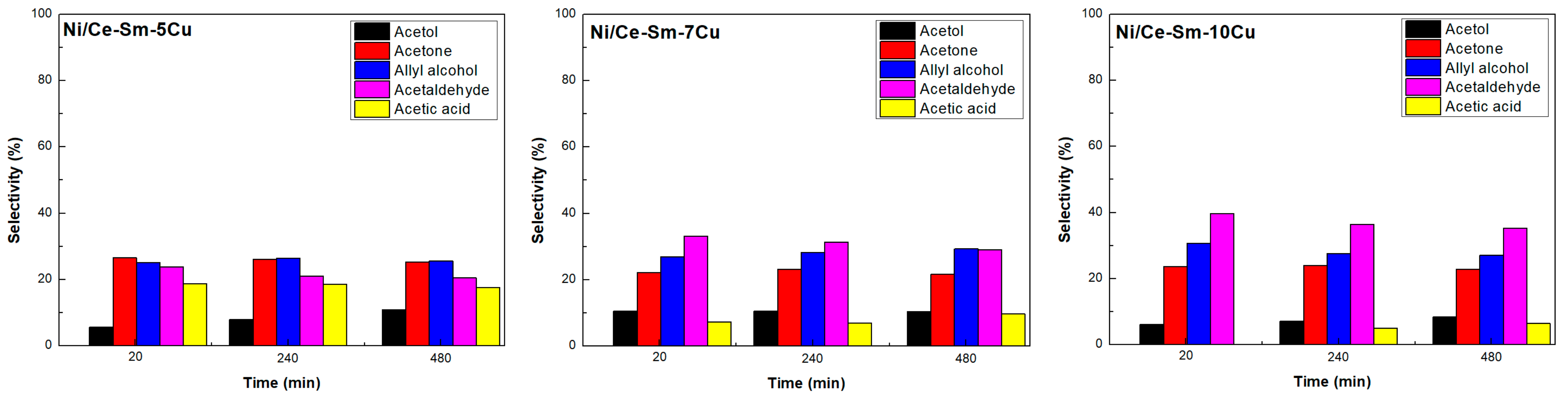
3.2.2. Selectivity to Liquid Products
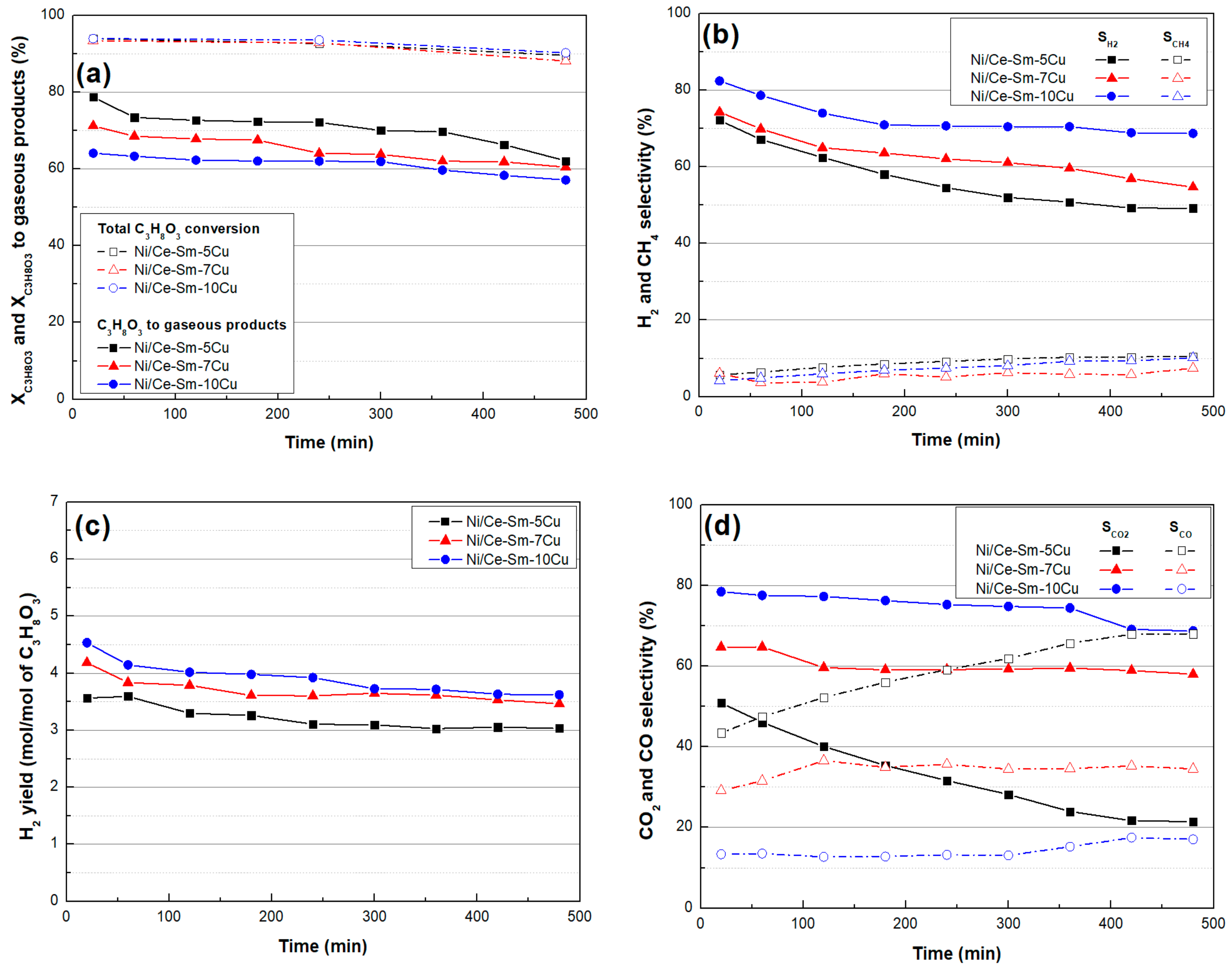
3.3. Catalytic Stability
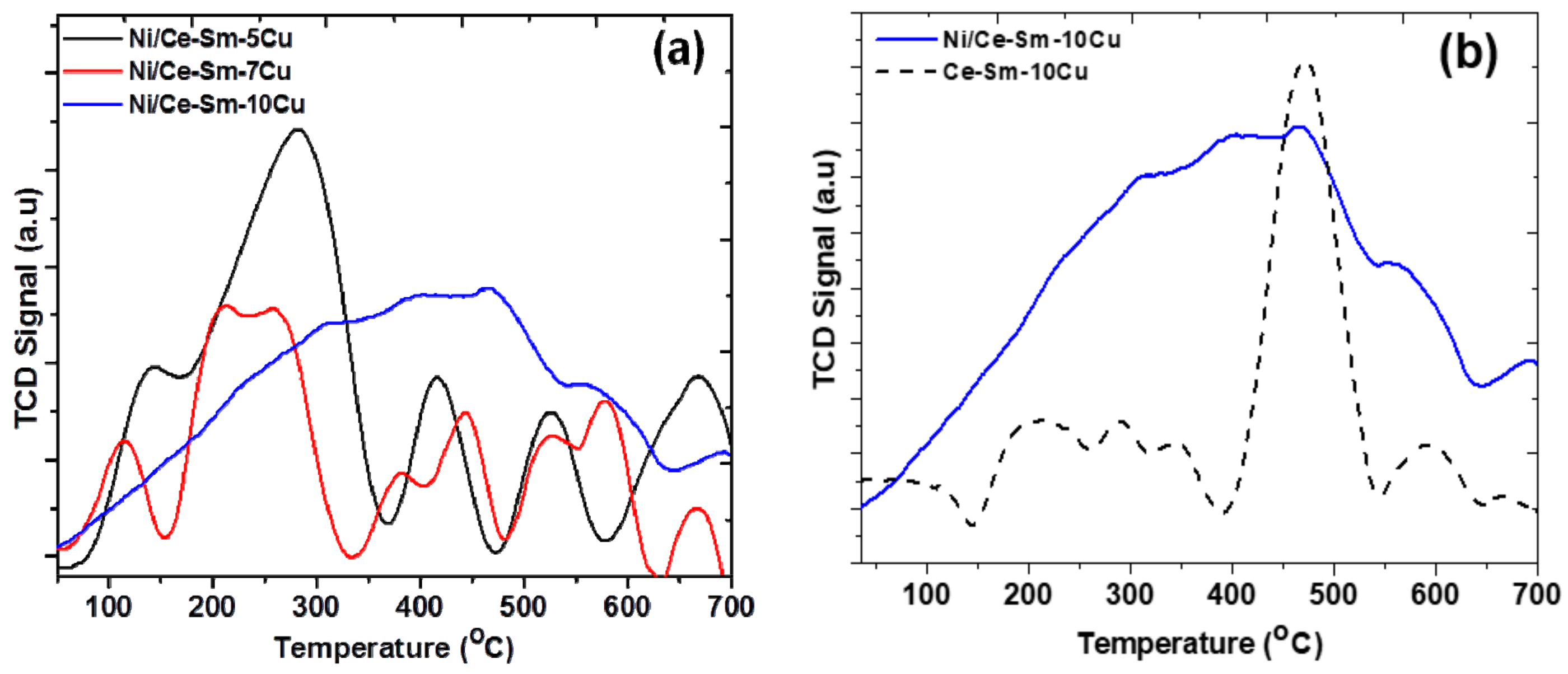
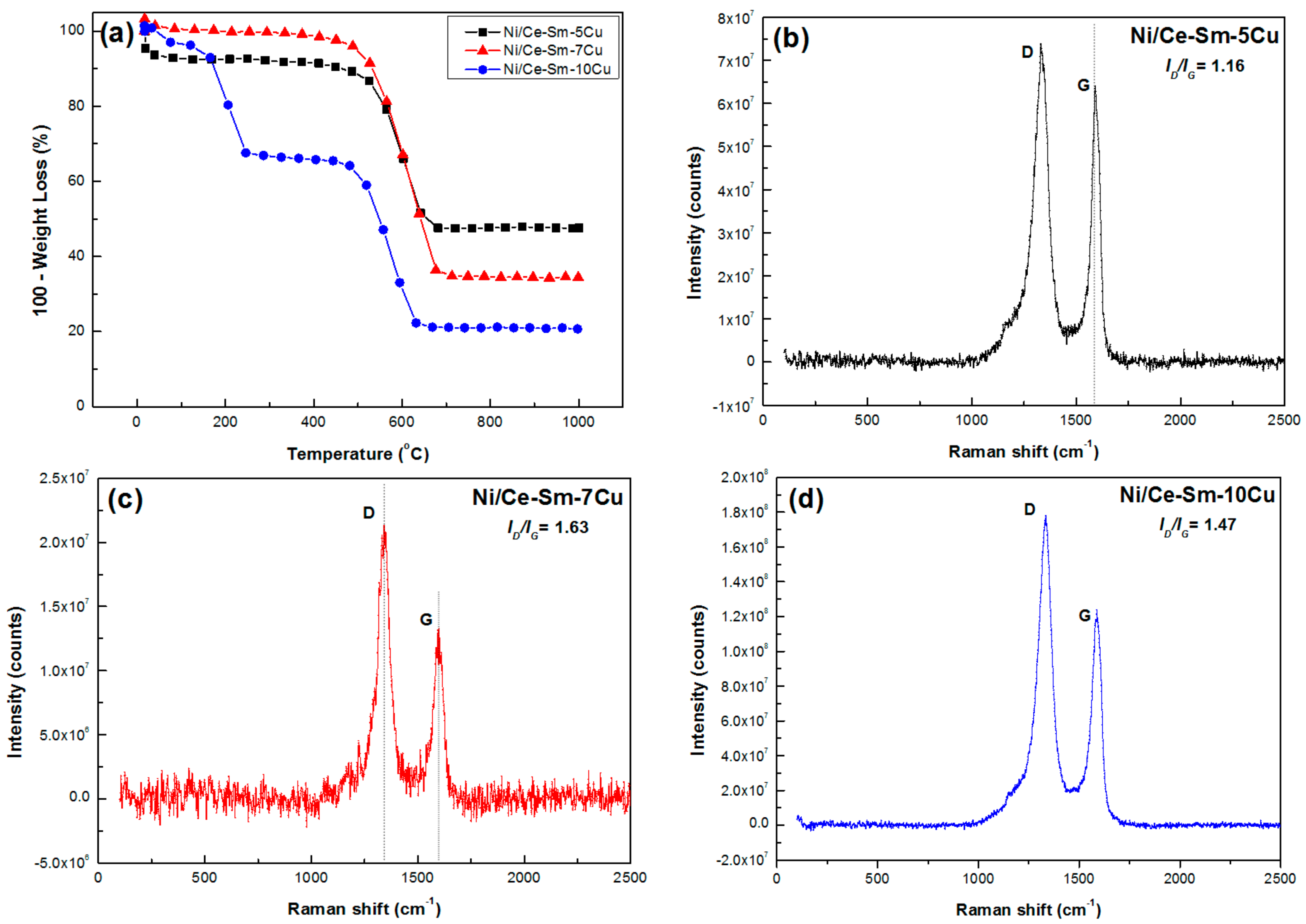
3.4. Characterization of Used Catalysts
4. Conclusions
- The catalysts are mainly composed of ceria type cubic lattice with traces of CuO and NiO being rather non-porous or macroporous materials with a spongy morphology due to the evaporation of gases originating from the decomposition of organic compounds used in the synthesis. They also present a rich population of mobile oxygen species both in surface and in the bulk. The increase in Cu content seems to facilitate the reducibility of the catalyst. Furthermore, all catalysts present weak, medium and strong acid and basic sites, a key feature towards the tailoring of the liquid products of this reaction.
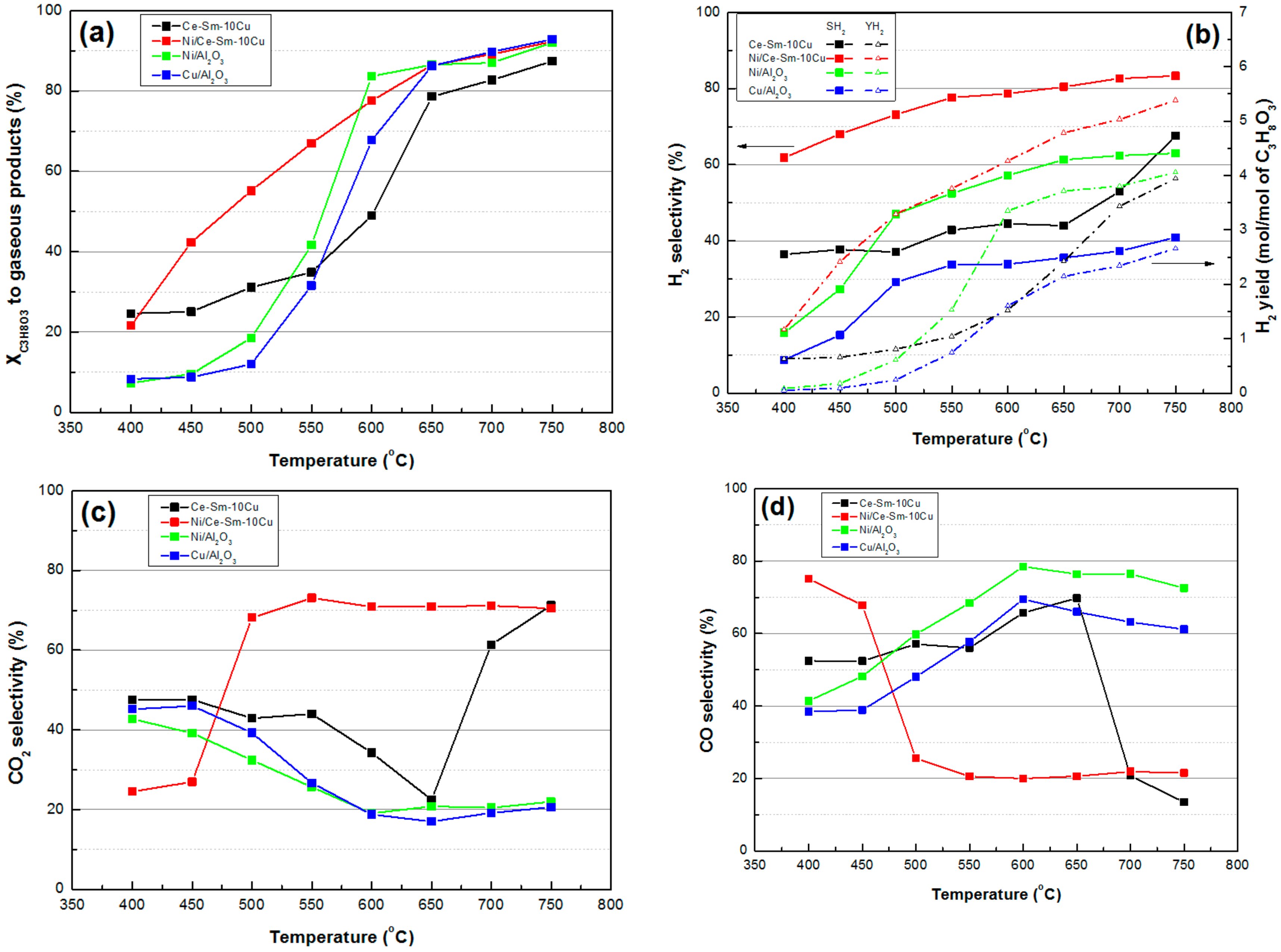
- In terms of catalytic activity, all of the catalysts had very high XC3H8O3 for the entire temperature range; from ≈84% at 400 °C to ≈94% at 750 °C. Ni/Ce-Sm-10Cu catalyst showed lower X C3H8O3-gas implying the increased Cu content had a detrimental effect on performance, especially below 650 °C. In terms of SH2 and YH2, both appeared to vary in the following order Ni/Ce-Sm-10Cu > Ni/Ce-Sm-7Cu > Ni/Ce-Sm-5Cu, where the high impact of Cu content is demonstrated. Moreover, the catalysts with the higher Cu content (Ni/Ce-Sm-7Cu and Ni/Ce-Sm-10Cu) had low, stable values of SCH4, for the entire temperature range. In contrast, for the Ni/Ce-Sm-5Cu catalyst, these values increased with temperature from ≈3% at 400 °C to 13% at 750 °C. At low reaction temperatures, all catalysts were more selective towards CO and less selective towards CO2. A variety of liquid products were detected, however, all catalysts stopped producing effluents over 650 °C.
- The stability testing experiments showed that the catalysts were quite stable, exhibiting high glycerol conversion (~90%) after 8 h of operation, whereas SH2 gradually decreased with time for all samples, with Ni/Ce-Sm-10Cu exhibiting the highest value (~70%) among them.
- All catalysts accumulated high amounts of carbon, following the order Ni/Ce-Sm-5Cu < Ni/Ce-Sm-7Cu < Ni/Ce-Sm-10Cu (52, 65 and 79 wt.%, respectively); however, the majority combusted at temperatures below 600 °C, which corroborates with the excellent catalytic performance of all samples. Raman studies over the used catalysts indicate that the incorporation of Cu in the support matrix helped control the graphitisation degree of the carbon deposited during the reaction at hand.
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Child, M.; Koskinen, O.; Linnanen, L.; Breyer, C. Sustainability guardrails for energy scenarios of the global energy transition. Renew. Sustain. Energy Rev. 2018, 91, 321–334. [Google Scholar] [CrossRef]
- Oh, Y.K.; Hwang, K.-R.; Kim, C.; Kim, J.R.; Lee, J.S. Recent developments and key barriers to advanced biofuels: A short review. Bioresour. Technol. 2018, 257, 320–333. [Google Scholar] [CrossRef] [PubMed]
- Hao, H.; Geng, Y.; Sarkis, J. Carbon footprint of global passenger cars: Scenarios through 2050. Energy 2016, 101, 121–131. [Google Scholar] [CrossRef]
- Charisiou, N.D.; Papageridis, K.N.; Siakavelas, G.; Tzounis, L.; Goula, M.A. Effect of active metal supported on SiO2 for selective hydrogen production from the glycerol steam reforming reaction. Bioresources 2016, 11, 10173–10189. [Google Scholar] [CrossRef]
- Ambat, I.; Srivastava, V.; Sillanpaa, M. Recent advancement in biodiesel production methodologies using various feedstock: A review. Renew. Sustain. Energy Rev. 2018, 90, 356–369. [Google Scholar] [CrossRef]
- Goula, M.A.; Charisiou, N.D.; Papageridis, K.N.; Siakavelas, G. Influence of the synthesis method parameters used to prepare nickel-based catalysts on the catalytic performance for the glycerol steam reforming reaction. Chin. J. Catal. 2016, 37, 1949–1965. [Google Scholar] [CrossRef]
- Dou, B.; Song, Y.; Wang, C.; Chen, H.; Xu, Y. Hydrogen production from catalytic steam reforming of biodiesel byproduct glycerol: Issues and challenges. Renew. Sustain. Energy Rev. 2014, 30, 950–960. [Google Scholar] [CrossRef]
- Goula, M.A.; Charisiou, N.D.; Pandis, P.K.; Stathopoulos, V.N. Ni/apatite-type lanthanum silicate supported catalyst for the glycerol steam reforming reaction. RSC Adv. 2016, 6, 78954–78958. [Google Scholar] [CrossRef]
- Dobosz, J.; Cichy, M.; Zawadzki, M.; Borowiecki, T. Glycerol steam reforming over calcium hydroxyapatite supported cobalt and cobalt-cerium catalysts. J. Energy Chem. 2018, 27, 404–412. [Google Scholar] [CrossRef]
- Chen, H.; Ding, Y.; Cong, N.T.; Dou, B.; Dupont, V.; Ghadiri, M.; Williams, P.T. A comparative study on hydrogen production from steam-glycerol reforming: Thermodynamics and experimental. Renew. Energy 2011, 36, 779–788. [Google Scholar] [CrossRef]
- Silva, J.M.; Soria, M.A.; Madeira, L.M. Challenges and strategies for optimization of glycerol steam reforming process. Renew. Sustain. Energy Rev. 2015, 42, 1187–1213. [Google Scholar] [CrossRef]
- Sad, M.E.; Duarte, H.A.; Vignatti, C.H.; Padro, C.L.; Apesteguıa, C.R. Steam reforming of glycerol: Hydrogen production optimization. Int. J. Hydrogen Energy 2015, 40, 6097–6106. [Google Scholar] [CrossRef]
- Aman, D.; Radwan, D.; Emaid, M.; Mikhail, S.; van Steen, E. Comparing nickel and cobalt perovskites for steam reforming of glycerol. Mol. Catal. 2018, 452, 60–67. [Google Scholar] [CrossRef]
- Charisiou, N.D.; Siakavelas, G.; Papageridis, K.N.; Baklavaridis, A.; Tzounis, L.; Polychronopoulou, K.; Goula, M.A. Hydrogen production via the glycerol steam reforming reaction over nickel supported on alumina and lanthana-alumina catalysts. Int. J. Hydrogen Energy 2017, 42, 13039–13060. [Google Scholar] [CrossRef]
- Charisiou, N.D.; Papageridis, K.N.; Siakavelas, G.; Tzounis, L.; Kousi, K.; Baker, M.A.; Hinder, S.J.; Sebastian, V.; Polychronopoulou, K.; Goula, M.A. Glycerol steam reforming for hydrogen production over nickel supported on alumina, zirconia and silica catalysts. Top. Catal. 2017, 60, 1226–1250. [Google Scholar] [CrossRef]
- Charisiou, N.D.; Siakavelas, G.; Papageridis, K.N.; Sebastian, V.; Hinder, S.J.; Baker, M.A.; Polychronopoulou, K.; Goula, M.A. The influence of SiO2 doping on the Ni/ZrO2 supported catalyst for hydrogen production through the glycerol steam reforming reaction. Catal. Today 2019, 319, 206–219. [Google Scholar] [CrossRef]
- Bepari, S.; Pradhan, N.C.; Dalai, A.K. Selective production of hydrogen by steam reforming of glycerol over Ni/Fly ash catalyst. Catal. Today 2017, 291, 36–46. [Google Scholar] [CrossRef]
- Menor, M.; Sayas, S.; Chica, A. Natural sepiolite promoted with Ni as new and efficient catalyst for the sustainable production of hydrogen by steam reforming of the biodiesel by-products glycerol. Fuel 2017, 193, 351–358. [Google Scholar] [CrossRef]
- Yan, Z.; Liu, S.; Zhang, Y.; Wang, T.; Luo, S.; Chu, W.; Jing, F. The role of Zr in NiZrAl oxides catalyst and the evaluation on steam reforming of glycerol for hydrogen product. Catal. Today 2019, 319, 229–238. [Google Scholar] [CrossRef]
- Martinez, T.L.M.; Araque, M.; Centeno, M.A.; Roger, A.C. Role of ruthenium on the catalytic properties of CeZr and CeZrCo mixed oxides for glycerol steam reforming reaction toward H2 production. Catal. Today 2015, 242, 80–90. [Google Scholar]
- Gallegos-Suarez, E.; Guerrero-Ruiz, A.; Rodriguez-Ramos, I. Efficient hydrogen production from glycerol by steam reforming with carbon supported ruthenium catalysts. Carbon 2016, 96, 578–587. [Google Scholar] [CrossRef]
- Wu, G.; Li, S.; Zhang, C.; Wang, T.; Gong, J. Glycerol steam reforming over perovskite-derived nickel-based catalysts. Appl. Catal. B-Environ. 2014, 144, 277–285. [Google Scholar] [CrossRef]
- Yurdakul, M.; Ayas, N.; Bizkarra, K.; El Doukkali, M.; Cambra, J.F. Preparation of Ni-based catalysts to produce hydrogen from glycerol by steam reforming process. Int. J. Hydrogen Energy 2016, 41, 8084–8091. [Google Scholar] [CrossRef]
- Zamzuri, N.H.; Mat, R.; Amin, N.A.S.; Talebian-Kiakalaieh, A. Hydrogen production from catalytic steam reforming of glycerol over various supported nickel catalysts. Int. J. Hydrogen Energy 2017, 42, 9087–9098. [Google Scholar] [CrossRef]
- Dieuzeide, M.L.; Laborde, M.; Amadeo, N.; Cannilla, C.; Bonura, G.; Frusteri, F. Hydrogen production by glycerol steam reforming: How Mg doping affects the catalytic behaviour of Ni/Al2O3 catalysts. Int. J. Hydrogen Energy 2016, 41, 157–166. [Google Scholar] [CrossRef]
- Senseni, A.Z.; Fattahi, S.M.S.; Rezaei, M.; Meshkani, F. A comparative study of experimental investigation and response surface optimization of steam reforming of glycerol over nickel nano-catalysts. Int. J. Hydrogen Energy 2016, 41, 10178–10192. [Google Scholar] [CrossRef]
- Charisiou, N.D.; Papageridis, K.N.; Tzounis, L.; Sebastian, V.; Baker, M.A.; Hinder, S.J.; AlKetbi, M.; Polychronopoulou, K.; Goula, M.A. Ni supported on CaO-MgO-Al2O3 as a highly selective and stable catalyst for H2 production via the glycerol steam reforming reaction. Int. J. Hydrogen Energy 2018. [Google Scholar] [CrossRef]
- Senseni, A.Z.; Meshkani, F.; Rezaei, M. Steam reforming of glycerol on mesoporous nanocrystalline Ni/Al2O3 catalysts for H2 production. Int. J. Hydrogen Energy 2016, 41, 20137–20146. [Google Scholar] [CrossRef]
- Demsash, H.D.; Kondamudi, K.V.K.; Upadhyayula, S.; Mohan, R. Ruthenium doped nickel-alumina-ceria catalyst in glycerol steam reforming. Fuel Process. Technol. 2018, 169, 150–156. [Google Scholar] [CrossRef]
- Papageridis, K.N.; Charisiou, N.D.; Siakavelas, G.; Avraam, D.G.; Tzounis, L.; Kousi, K.; Goula, M.A. Comparative study of Ni, Co, Cu supported on γ-alumina catalysts for hydrogen production via the glycerol steam reforming reaction. Fuel Process. Technol. 2016, 152, 156–175. [Google Scholar] [CrossRef]
- Martinez, T.L.M.; Araque, M.; Vargas, J.C.; Roger, A.C. Effect of Ce/Zr ratio in CeZr-CoRh catalysts on the hydrogen production by glycerol steam reforming. Appl. Catal. B-Environ. 2013, 132–133, 499–510. [Google Scholar] [CrossRef]
- Bobadilla, L.F.; Romero-Sarria, F.; Centeno, M.A.; Odriozola, J.A. Promoting effect of Sn on supported Ni catalyst during steam reforming of glycerol. Int. J. Hydrogen Energy 2016, 41, 9234–9244. [Google Scholar] [CrossRef]
- Sanchez, E.A.; Comelli, R.A. Hydrogen production by glycerol steam-reforming over nickel and nickel-cobalt impregnated on alumina. Int. J. Hydrogen Energy 2014, 39, 8650–8655. [Google Scholar] [CrossRef]
- Wang, C.; Dou, B.; Chen, H.; Son, Y.; Xu, Y.; Du, X.; Zhang, L.; Luo, T.; Tan, C. Renewable hydrogen production from steam reforming of glycerol by Ni–Cu–Al, Ni–Cu–Mg, Ni–Mg catalysts. Int. J. Hydrogen Energy 2013, 38, 3562–3571. [Google Scholar] [CrossRef]
- Dou, B.; Wang, C.; Song, Y.; Chen, H.; Xu, Y. Activity of Ni–Cu–Al based catalyst for renewable hydrogen production from steam reforming of glycerol. Energy Convers. Manag. 2014, 78, 253–259. [Google Scholar] [CrossRef]
- Ramesh, S.; Yang, E.H.; Jung, J.S.; Moon, D.J. Copper decorated perovskite an efficient catalyst for low temperature hydrogen production by steam reforming of glycerol. Int. J. Hydrogen Energy 2015, 40, 11428–11435. [Google Scholar] [CrossRef]
- Polychronopoulou, K.; Jaoude, M.A. Nano-architectural advancement of CeO2-driven catalysis via electrospinning. Surf. Coat. Technol. 2018, 350, 245–280. [Google Scholar] [CrossRef]
- Polychronopoulou, K.; Efstathiou, A.M. Effects of sol−gel synthesis on 5Fe−15Mn−40Zn−40Ti−O mixed oxide structure and its H2S removal efficiency from industrial gas streams. Environ. Sci. Technol. 2009, 43, 4367–4372. [Google Scholar] [CrossRef] [PubMed]
- Petallidou, K.C.; Polychronopoulou, K.; Boghosian, S.; Carcia-Rodriguez, S.; Efstathiou, A.M. Water–gas shift reaction on Pt/Ce1–xTixO2−δ: The effect of Ce/Ti ratio. J. Phys. Chem. C 2013, 117, 25467–25477. [Google Scholar] [CrossRef]
- Moletti, A.; Coluccini, C.; Pasini, D.; Taglietti, A. A chiral probe for the detection of Cu(II) by UV, CD and emission spectroscopies. Dalton Trans. 2007, 16, 1588–1592. [Google Scholar] [CrossRef] [PubMed]
- Pasini, D.; Righetti, P.P.; Rossi, V. Malonate crown ethers as building blocks for novel D-π-A chromophores. Org. Lett. 2002, 4, 23–26. [Google Scholar] [CrossRef] [PubMed]
- Goula, M.A.; Charisiou, N.D.; Siakavelas, G.; Tzounis, L.; Tsiaoussis, I.; Panagiotopoulou, P.; Goula, G.; Yentekakis, I.V. Syngas production via the biogas dry reforming reaction over Ni supported on zirconia modified with CeO2 or La2O3 catalysts. Int. J. Hydrogen Energy 2017, 42, 13724–13740. [Google Scholar] [CrossRef]
- Charisiou, N.D.; Siakavelas, G.; Tzounis, L.; Sebastian, V.; Monzon, A.; Baker, M.A.; Hinder, S.J.; Polychronopoulou, K.; Yentekakis, I.V.; Goula, M.A. An in depth investigation of deactivation through carbon formation during the biogas dry reforming reaction for Ni supported on modified with CeO2 and La2O3 zirconia catalysts. Int. J. Hydrogen Energy 2018, 43, 18955–18976. [Google Scholar] [CrossRef]
- Jiang, B.; Li, L.; Bian, Z.; Li, Z.; Sun, Z.; Tang, D.; Kawi, S.; Dou, B.; Goula, M.A. Chemical looping glycerol reforming for hydrogen production by Ni@ZrO2 nano-composite oxygen carriers. Int. J. Hydrogen Energy 2018, 43, 13200–13211. [Google Scholar] [CrossRef]
- Demsash, H.D.; Mohan, R. Steam reforming of glycerol to hydrogen over ceria promoted nickel–alumina catalysts. Int. J. Hydrogen Energy 2016, 41, 22732–22742. [Google Scholar] [CrossRef]
- Adhikari, S.; Fernando, S.D.; To, S.D.F.; Bricka, R.M.; Steele, R.H.; Haryanto, A. Conversion of glycerol to hydrogen via a steam reforming process over nickel catalysts. Energy Fuel 2008, 22, 1220–1226. [Google Scholar] [CrossRef]
- Pant, K.K.; Jain, R.; Jain, S. Renewable hydrogen production by steam reforming of glycerol over Ni/CeO2 catalyst prepared by precipitation deposition method. Korean J. Chem. Eng. 2011, 28, 1859–1866. [Google Scholar] [CrossRef]
- Liu, Y.; Hayakawa, T.; Suzuki, K.; Hamakawa, S.; Tsunodo, T.; Ishii, T.; Kumagai, M. Highly active copper/ceria catalysts for steam reforming of methanol. Appl. Catal. A-Gen. 2002, 223, 137–145. [Google Scholar] [CrossRef]
- Lei, Y.; Luo, Y.; Li, X.; Lu, J.; Mei, Z.; Peng, W.; Chen, R.; Chen, K.; Chen, D.; He, D. The role of samarium on Cu/Al2O3 catalyst in the methanol steam reforming for hydrogen production. Catal. Today 2018, 307, 162–168. [Google Scholar] [CrossRef]
- Osasuwa, O.U.; Khan, M.R.; Lam, S.S.; Assabumrungrat, A.; Cheng, C.K. An assessment of the longevity of samarium cobalt trioxide perovskite catalyst during the conversion of greenhouse gases into syngas. J. Clean. Prod. 2018, 185, 576–587. [Google Scholar] [CrossRef]
- Polychronopoulou, K.; Zedan, A.F.; Katsiotis, M.S.; Baker, M.A.; AlKhoori, A.A.; AlQaradawi, S.Y.; Hinder, S.J.; AlHassan, S. Rapid microwave assisted sol-gel synthesis of CeO2 and CexSm1-xO2 nanoparticle catalysts for CO oxidation. Mol. Catal. 2017, 428, 41–55. [Google Scholar] [CrossRef]
- Polychronopoulou, K.; Zedan, A.F.; AlKetbi, M.; Stephen, S.; Ather, M.; Katsiotis, M.S.; Arvanitidis, J.; Christofilos, D.; Isakovich, A.F.; AlHassan, S. Tailoring the efficiency of an active catalyst for CO abatement through oxidation reaction: The case study of samarium-doped ceria. J. Environ. Chem. Eng. 2018, 6, 266–280. [Google Scholar] [CrossRef]
- AlKetbi, M.; Polychronopoulou, K.; Zedan, A.F.; Sebastian, V.; Baker, M.A.; AlKhoori, A.; Jaoude, M.A.; Alnuaimi, O.; Hinder, S.J.; Tharalekshmy, A.; et al. Tuning the activity of Cu-containing rare earth oxide catalysts for CO oxidation reaction: Cooling while heating paradigm in microwave-assisted synthesis. Mater. Res. Bull. 2018, 108, 142–150. [Google Scholar] [CrossRef]
- Polychronopoulou, K.; Costa, C.N.; Efstathiou, A.M. The role of oxygen and hydroxyl support species on the mechanism of H2 production in the steam reforming of phenol over metal oxide-supported-Rh and -Fe catalysts. Catal. Today 2006, 112, 89–93. [Google Scholar] [CrossRef]
- Polychronopoulou, K.; Efstathiou, A.M. Spillover of labile OH, H, and O species in the H2 production by steam reforming of phenol over supported-Rh catalysts. Catal. Today 2006, 116, 341–347. [Google Scholar] [CrossRef]
- Polychronopoulou, K.; Giannakopoulos, K.; Efstathiou, A.M. Tailoring MgO-based supported Rh catalysts for purification of gas streams from phenol. Appl. Catal. B-Environ. 2012, 111, 360–375. [Google Scholar] [CrossRef]
- Petallidou, K.C.; Polychronopoulou, K.; Fierro, J.L.G.; Efstathiou, A.M. Low-temperature water-gas shift on Pt/Ce0.8La0.2O2−δ–CNT: The effect of Ce0.8La0.2O2−δ/CNT ratio. Appl. Catal. A-Gen. 2015, 504, 585–598. [Google Scholar] [CrossRef]
- Sing, K.S.W.; Everett, D.H.; Hall, R.A.W.; Moscou, L.; Pierotti, R.A.; Rouquerol, J.; Siemieniewska, T. Reporting physisorption data for gas/solid systems with special reference to the determination of surface area and porosity. IUPAC 1985, 57, 603–619. [Google Scholar] [CrossRef]
- Rouquerol, J.; Rouquerol, F.; Llewellyn, P.; Maurin, G.; Sing, K.S. Adsorption by Powders and Porous Solids: Principles, Methodology and Applications; Academic Press: Cambridge, MA, USA, 1999. [Google Scholar]
- Damyanova, S.; Pawelec, B.; Palcheva, R.; Karakirova, Y.; Capel Sanchez, M.C.; Tyuliev, G.; Gaigneaux, E.; Fierro, J.L.G. Structure and surface properties of ceria-modified Ni-based catalysts for hydrogen production. Appl. Catal. B-Environ. 2018, 225, 340–353. [Google Scholar] [CrossRef]
- Liu, H.; Huang, Z.; Kang, H.; Li, X.; Xia, C.; Chen, J.; Liu, H. Efficient bimetallic NiCu-SiO2 catalysts for selective hydrogenolysis of xylitol to ethylene glycol and propylene glycol. Appl. Catal. B-Environ. 2018, 220, 251–263. [Google Scholar] [CrossRef]
- Polychronopoulou, K.; Kalamaras, C.M.; Efstathiou, A.M. Ceria-based materials for hydrogen production via hydrocarbon steam reforming and water-gas shift reactions. Recent Patents Mater. Sci. 2011, 4, 122–145. [Google Scholar]
- Charisiou, N.D.; Polychronopoulou, K.; Asif, A.; Goula, M.A. The potential of glycerol and phenol towards H2 production using steam reforming reaction: A review. Surf. Coat. Technol. 2018, 352, 92–111. [Google Scholar] [CrossRef]
- Saw, E.T.; Oemar, U.; Tan, X.R.; Du, Y.; Borgna, A.; Hidajat, K.; Kawi, S. Bimetallic Ni–Cu catalyst supported on CeO2 for high-temperature water–gas shift reaction: Methane suppression via enhanced CO adsorption. J. Catal. 2014, 314, 32–46. [Google Scholar] [CrossRef]
- Avgouropoulos, G.; Ioannides, T. Selective CO oxidation over CuO-CeO2 catalysts prepared via the urea-nitrate combustion method. Appl. Catal. A-Gen. 2003, 244, 155–167. [Google Scholar] [CrossRef]
- Liu, Z.; Zhou, R.; Zheng, X. Influence of preparation methods on CuO-CeO2 catalysts in the preferential oxidation of CO in excess hydrogen. J. Nat. Gas Chem. 2008, 17, 125–129. [Google Scholar] [CrossRef]
- Lin, J.-H.; Biswas, P.; Guliants, V.V.; Misture, S. Hydrogen production by water–gas shift reaction over bimetallic Cu–Ni catalysts supported on La-doped mesoporous ceria. Appl. Catal. A-Gen. 2010, 387, 87–94. [Google Scholar] [CrossRef]
- Simonetti, D.A.; Dumesic, J.A. Catalytic production of liquid fuels from biomass-derived oxygenated hydrocarbons: Catalytic coupling at multiple length scales. Cat. Rev. Sci. Eng. 2009, 51, 441–484. [Google Scholar] [CrossRef]
- Sanchez-Sanchez, M.C.; Navarro, R.M.; Fierro, J.L.G. Ethanol steam reforming over Ni/MxOy–Al2O3 (M=Ce, La, Zr and Mg) catalysts: Influence of support on the hydrogen production. Int. J. Hydrogen Energy 2007, 32, 1462–1471. [Google Scholar] [CrossRef]
- Benbenek, S.; Fedorynska, E.; Winiarek, P. Investigation of the acidity of Ni/Al2O3 and Ni/SiO2−Al2O3 catalysts. React. Kinet. Catal. Lett. 1993, 51, 189–195. [Google Scholar] [CrossRef]
- Liu, C.; Hou, R.; Wang, T. Role of acid sites and surface hydroxyl groups in isophthalonitrile hydrogenation catalyzed by supported Ni-Co catalysts. RSC Adv. 2015, 5, 26465–26474. [Google Scholar] [CrossRef]
- Lu, H.; Yin, H.; Liu, Y.; Jiang, T.; Yu, L. Influence of support on catalytic activity of Ni catalysts in p-nitrophenol hydrogenation to p-aminophenol. Catal. Commun. 2008, 10, 313–316. [Google Scholar] [CrossRef]
- Iriondo, A.; Barrio, V.L.; Cambra, J.F.; Arias, P.L.; Guemez, M.B.; Navarro, R.M.; Sachez-Sanchez, M.C.; Fiero, J.L.G. Hydrogen production from glycerol over nickel catalysts supported on Al2O3 modified by Mg, Zr, Ce or La. Top. Catal. 2008, 49, 46–58. [Google Scholar] [CrossRef]
- Dang, C.; Wang, H.; Yu, H.; Peng, F. Sorption-enhanced steam reforming of glycerol over NiCuCaAl catalysts for producing fuel-cell grade hydrogen. Int. J. Hydrogen Energy 2017, 42, 17446–17456. [Google Scholar] [CrossRef]
- Chen, L.C.; Cheng, H.; Chiang, C.W.; Lin, S.D. Sustainable hydrogen production by ethanol steam reforming using a partially reduced copper-nickel oxide catalyst. ChemSusChem 2015, 10, 1787–1793. [Google Scholar] [CrossRef] [PubMed]
- Khzouz, M.; Gkanas, E.I.; Du, S.; Wood, J. Catalytic performance of Ni-Cu/Al2O3 for effective syngas production by methanol steam reforming. Fuel 2018, 232, 672–683. [Google Scholar] [CrossRef]
- Corma, A.; Huber, G.W.; Sauvanaud, L.; O’Connor, P. Biomass to chemicals: Catalytic conversion of glycerol/water mixtures into acrolein reaction network. J. Catal. 2018, 257, 163–171. [Google Scholar] [CrossRef]
- Chai, S.-H.; Wang, H.-P.; Liang, Y.; Xu, B.-Q. Sustainable production of acrolein: Investigation of solid acid–base catalysts for gas-phase dehydration of glycerol. Green Chem. 2007, 9, 1130–1136. [Google Scholar] [CrossRef]
- Nakagawa, Y.; Tomishige, K. Heterogeneous catalysis of the glycerol hydrogenolysis. Catal. Sci. Technol. 2011, 1, 179–190. [Google Scholar] [CrossRef]
- Yuan, Z.; Wang, L.; Wang, J.; Xia, S.; Chen, P.; Hou, Z.; Zheng, X. Hydrogenolysis of glycerol over homogenously dispersed copper on solid base catalysts. Appl. Catal. B-Environ. 2011, 101, 431–440. [Google Scholar] [CrossRef]
- Miyazawa, T.; Kusunoki, Y.; Kunimori, K.; Tomishige, K. Glycerol conversion in the aqueous solution under hydrogen over Ru/C + an ion-exchange resin and its reaction mechanism. J. Catal. 2006, 240, 213–221. [Google Scholar] [CrossRef]
- Delgado, S.N.; Yap, D.; Vivier, L.; Especel, C. Influence of the nature of the support on the catalytic properties of Pt-based catalysts for hydrogenolysis of glycerol. J. Mol. Catal. A Chem. 2013, 367, 89–98. [Google Scholar] [CrossRef]
- Maglinao, R.; He, B. Verification of propylene glycol preparation from glycerol via the acetol pathway by in situ hydrogenolysis. Biofuels 2012, 3, 675–682. [Google Scholar] [CrossRef]
- Guo, L.; Zhou, J.; Mao, J.; Guo, X.; Zhang, S. Supported Cu catalysts for the selective hydrogenolysis of glycerol to propanediols. Appl. Catal. A-Gen. 2009, 367, 93–98. [Google Scholar] [CrossRef]
- Sato, S.; Akiyama, M.; Takahashi, R.; Hara, T.; Inui, K.; Yokota, M. Vapor-phase reaction of polyols over copper catalysts. Appl. Catal. A-Gen. 2008, 347, 186–191. [Google Scholar] [CrossRef]
- Rodriguez, J.A.; Goodman, D.W. The nature of the metal-metal bond in bimetallic surfaces. Science 1992, 257, 897–903. [Google Scholar] [CrossRef] [PubMed]
- Besenbacher, F.; Chorkendorff, I.; Clausen, B.S.; Hammer, B.; Molenbroek, A.M.; Norskov, J.K.; Stensgaard, I. Design of a surface alloy catalyst for steam reforming. Science 1998, 279, 1913–1915. [Google Scholar] [CrossRef] [PubMed]
- Ungureanu, A.; Dragoi, B.; Chirieac, A.; Royer, S.; Duprez, D.; Dumitriu, E. Synthesis of highly thermostable copper-nickel nanoparticles confined in the channels of ordered mesoporous SBA-15 silica. J. Mater. Chem. 2011, 21, 12529–12541. [Google Scholar] [CrossRef]
- Huang, T.J.; Jhao, S.Y. Ni-Cu/samaria-doped ceria catalysts for steam reforming of methane in the presence of carbon dioxide. Appl. Catal. A-Gen. 2006, 302, 325–332. [Google Scholar] [CrossRef]
- Noller, H.; Lin, W.M. Activity and selectivity of Ni-CuAl2O3 catalysts for hydrogenation of crotonaldehyde and mechanism of hydrogenation. J. Catal. 1984, 85, 25–30. [Google Scholar] [CrossRef]
- Yin, A.; Wen, C.; Guo, X.; Dai, W.L.; Fan, K. Influence of Ni species on the structural evolution of Cu/SiO2 catalyst for the chemoselective hydrogenation of dimethyl oxalate. J. Catal. 2011, 280, 77–88. [Google Scholar] [CrossRef]
- Studt, F.; Abild-Pedersen, F.; Wu, Q.; Jensen, A.D.; Temel, B.; Grunwaldt, J.D.; Norskov, J.K. CO hydrogenation to methanol on Cu-Ni catalysts: Theory and experiment. J. Catal. 2012, 293, 5160. [Google Scholar] [CrossRef]
- De Rogatis, L.; Montini, T.; Lorenzut, B.; Fornasiero, P. NixCuy/Al2O3 based catalysts for hydrogen production. Energy Environ. Sci. 2008, 1, 501–509. [Google Scholar] [CrossRef]
- Chen, L.C.; Lin, S.D. The ethanol steam reforming over Cu-Ni/SiO2 catalysts: Effect of Cu/Ni ratio. Appl. Catal. B-Environ. 2011, 106, 639–649. [Google Scholar] [CrossRef]
- Liang, T.; Wang, Y.; Chen, M.; Yang, Z.; Liu, S.; Zhou, Z.; Xiaojing, L. Steam reforming of phenol-ethanol to produce hydrogen over bimetallic Ni-Cu catalysts supported on sepiolite. Int. J. Hydrogen Energy 2017, 42, 28233–28246. [Google Scholar] [CrossRef]
- Pu, J.; Nishikado, K.; Wang, N.; Nguyen, T.T.; Maki, T.; Qian, E.W. Core-shell nickel catalysts for the steam reforming of acetic acid. Appl. Catal. B-Environ. 2018, 224, 69–79. [Google Scholar] [CrossRef]
- Goula, M.A.; Lemonidou, A.A.; Efstathiou, A.M. Characterization of carbonaceous species formed during reforming of CH4 with CO2 over Ni/CaO-Al2O3 catalysts studied by various transient techniques. J. Catal. 1996, 161, 626–640. [Google Scholar] [CrossRef]
- Savutto, E.; Navarro, R.M.; Mota, N.; Di Carlo, A.; Bocci, E.; Carlini, M.; Fierro, J.L.G. Steam reforming of tar model compounds over Ni/Mayenite catalysts: Effect of Ce addition. Fuel 2018, 224, 676–686. [Google Scholar] [CrossRef]
- Gandarias, I.; Arias, P.L.; Requies, J.; El Doukkali, M.; Güemez, M.B. Liquid-phase glycerol hydrogenolysis to 1,2-propanediol under nitrogen pressure using 2-propanol as hydrogen source. J. Catal. 2011, 282, 237–247. [Google Scholar] [CrossRef]
- Charisiou, N.D.; Tzounis, L.; Sebastian, V.; Baker, M.A.; Hinder, S.J.; Polychronopoulou, K.; Goula, M.A. Investigating the correlation between deactivation and the carbon deposited on the surface of Ni/Al2O3 and Ni/La2O3-Al2O3 catalysts during the biogas reforming reaction. Appl. Surf. Sci. 2018, in press. [Google Scholar] [CrossRef]
- Yu, Z.; Hu, X.; Jia, P.; Zhang, Z.; Dong, D.; Hu, G.; Hu, S.; Wang, Y.; Xiang, J. Steam reforming of acetic acid over nickel-based catalysts: The intrinsic effects of nickel precursors on behaviors of nickel catalysts. Appl. Catal. B-Environ. 2018, 237, 538–553. [Google Scholar] [CrossRef]
- Charisiou, N.D.; Siakavelas, G.; Papageridis, K.N.; Baklavaridis, A.; Tzounis, L.; Avraam, D.G.; Goula, M.A. Syngas production via the biogas dry reforming reaction over nickel supported on modified with CeO2 and/or La2O3 alumina catalysts. J. Nat. Gas Sci. Eng. 2016, 31, 164–183. [Google Scholar] [CrossRef]
- Zhang, Z.; Hu, X.; Li, J.; Gao, G.; Dong, D.; Westerhof, R.; Hu, S.; Xiang, J.; Wang, Y. Steam reforming of acetic acid over Ni/Al2O3 catalysts: Correlation of nickel loading with properties and catalytic behaviors of the catalysts. Fuel 2018, 217, 389. [Google Scholar] [CrossRef]
- Zhang, C.; Zhang, P.; Li, S.; Wu, G.; Ma, X.; Gong, J. Superior reactivity of skeletal Ni based catalysts for low-temperature steam reforming to produce CO-free hydrogen. Phys. Chem. Chem. Phys. 2012, 14, 3295–3298. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Cai, W.; Li, Y.; Xu, Y.; Shen, W. Hydrogen production by steam reforming of ethanol over an Ir/CeO2 catalyst: Reaction mechanism and stability of the catalyst. Int. J. Hydrogen Energy 2008, 33, 4377–4386. [Google Scholar] [CrossRef]
- Fierro, V.; Akdim, O.; Mirodatos, C. On-board hydrogen production in a hybrid electric vehicle by bio-ethanol oxidative steam reforming over Ni and noble metal based catalysts. Green Chem. 2003, 5, 20–24. [Google Scholar] [CrossRef]
- Vizcaino, A.; Carrero, A.; Calles, J. Hydrogen production by ethanol steam reforming over Cu-Ni supported catalysts. Int. J. Hydrogen Energy 2007, 32, 1450–1461. [Google Scholar] [CrossRef]
- Li, Y.; Fu, Q.; Flytzani-Stephanopoulos, M. Low-temperature water-gas shift reaction over Cu- and Ni-loaded cerium oxide catalysts. Appl. Catal. B-Environ. 2000, 27, 179–191. [Google Scholar] [CrossRef]
- Greeley, J.; Mavrikakis, M. Surface and subsurface hydrogen: Adsorption properties on transition metals and near-surface alloys. J. Phys. Chem. B 2008, 109, 3460–3471. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; Mao, Y.; Wang, X.; Zhang, M. Preparation of a Cu-Ru/carbon nanotube catalyst for hydrogenolysis of glycerol to 1,2-propanediolviahydrogen spillover. Green Chem. 2011, 13, 1311–1316. [Google Scholar] [CrossRef]
- Cepeda, E.A.; Iriarte-Velasco, U.; Calvo, B.; Sierra, I. Hydrogenation of sunflower oil over M/SiO2 and M/Al2O3 (M = Ni, Pd, Pt, Co, Cu) catalysts. Oil Chem. J. Am. Soc. 2016, 93, 731–741. [Google Scholar] [CrossRef]
- Trasarti, A.F.; Bertero, N.M.; Apesteguía, C.R.; Marchi, A.J. Liquid-phase hydrogenation of acetophenone over silica-supported Ni, Co and Cu catalysts: Influence of metal and solvent. Appl. Catal. A-Gen. 2014, 475, 282–291. [Google Scholar] [CrossRef]
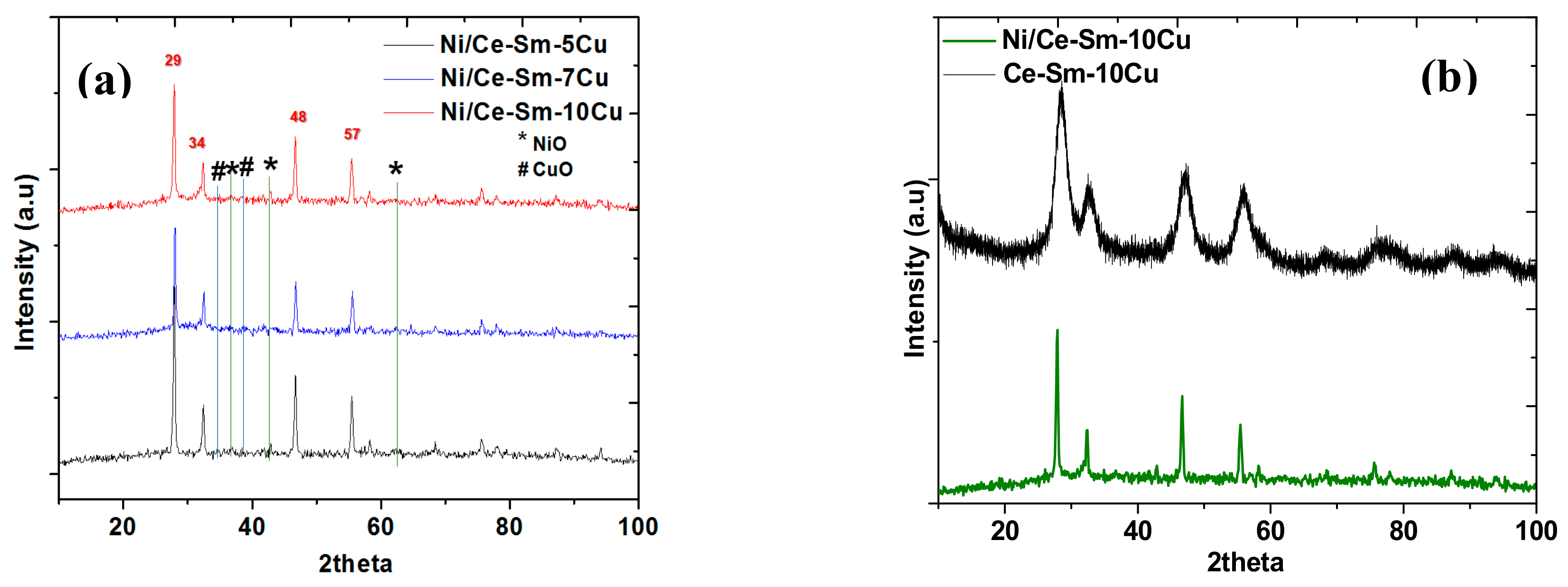


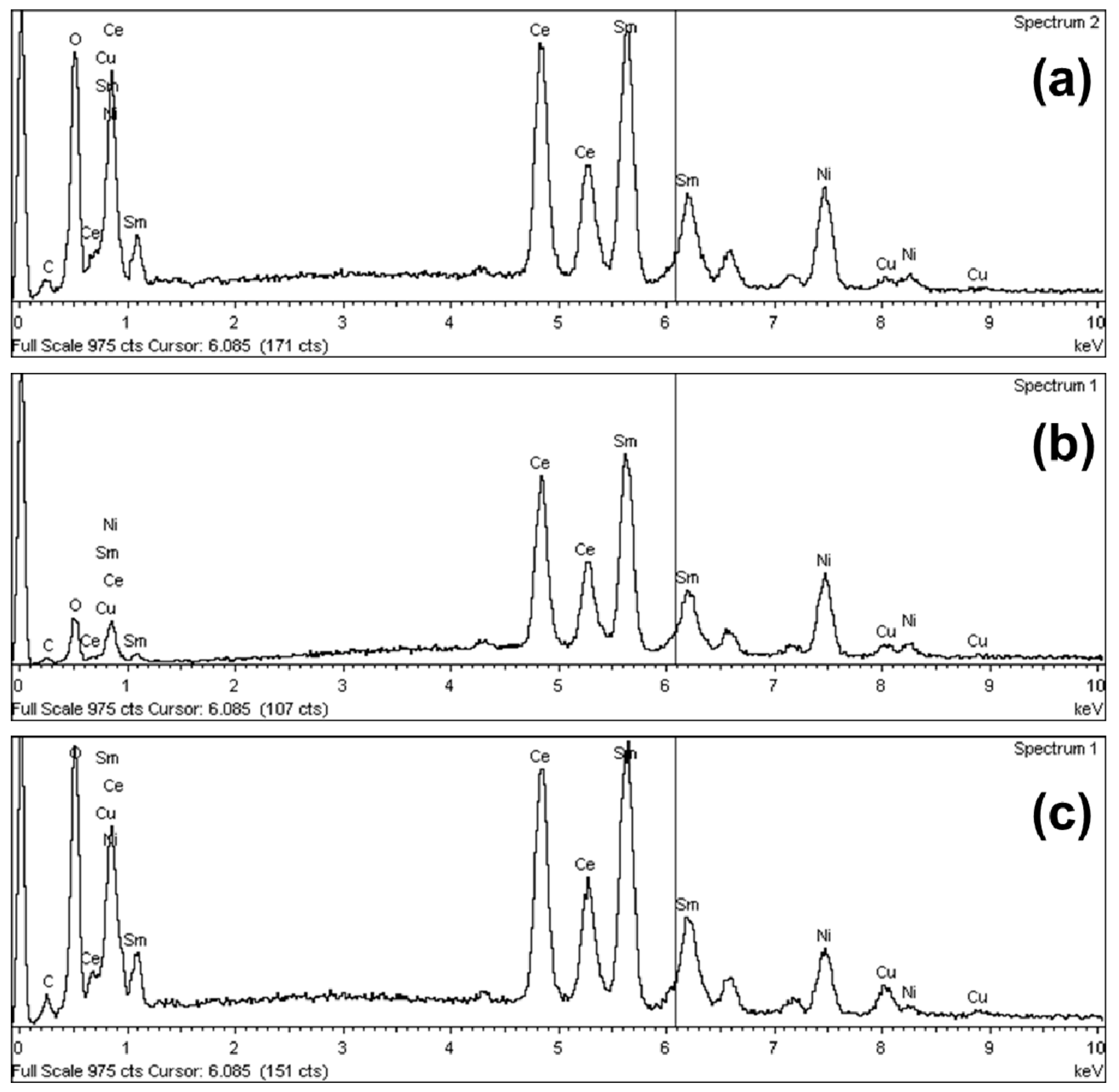


| Catalyst | Brunauer-Emmet-Teller(BET) | D | Pore Size 1 | Lattice Parameter 2 | X-ray Spectroscopy (EDS) at.% (Ratios) | |||
|---|---|---|---|---|---|---|---|---|
| (m2/g) | (nm) | (nm) | (Å) | Ce/Sm | Ni/Ce | Ni/Cu | Ni:Ce:Sm:Cu | |
| Ni/Ce-Sm-5Cu | 0.68 | 63.9 | 40 | 5.52 | 1.10 | 0.56 | 5.6 | 0.56(22%):1(39%):0.90(35%):0.10(4%) |
| Ni/Ce-Sm-7Cu | 0.1 | 62.1 | 198 | 5.51 | 1.04 | 0.86 | 3.23 | 0.86(29%):1(33%):0.96(32%):0.19(6%) |
| Ni/Ce-Sm-10Cu | 0.7 | 62.3 | 178 | 5.52 | 1.07 | 0.84 | 2.63 | 0.84(27%):1(33%):0.92(30%):0.32(10%) |
| Ce-Sm-10Cu (support) | 3.69 | 9.3 | 51.8 | 5.42 | n/a | n/a | ||
| Catalyst | Ni2p3/2 | Cu2p | Ce3d5/2 | Ni/Cu | Ni/Ce | Ce/Sm | |
|---|---|---|---|---|---|---|---|
| Ni0 | Ni2+ | Ratio | |||||
| Ni/Ce-Sm-5Cu-c 1 | n/a | 854.08/856.08 | 933.28 | 881.78 | 2.81 | 2.66 | 1.15 |
| Ni/Ce-Sm-7Cu-c 1 | n/a | 854.18/856.08 | 933.38 | 882.08 | 1.29 | 0.58 | 0.35 |
| Ni/Ce-Sm-10Cu-c 1 | n/a | 854.18/855.88 | 933.28 | 881.88 | 0.58 | 0.40 | 0.24 |
| Ni/Ce-Sm-5Cu-r 2 | 852.68 | 856.28 | 932.78 | 882.18 | 2.40 | 3.82 | 1.53 |
| Ni/Ce-Sm-7Cu-r 2 | 852.48 | 856.08 | 933.08 | 882.18 | 1.50 | 1.70 | 0.71 |
| Ni/Ce-Sm-10Cu-r 2 | 852.38 | 855.98 | 933.08 | 882.18 | 1.32 | 1.69 | 0.83 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Polychronopoulou, K.; Charisiou, N.; Papageridis, K.; Sebastian, V.; Hinder, S.; Dabbawala, A.; AlKhoori, A.; Baker, M.; Goula, M. The Effect of Ni Addition onto a Cu-Based Ternary Support on the H2 Production over Glycerol Steam Reforming Reaction. Nanomaterials 2018, 8, 931. https://doi.org/10.3390/nano8110931
Polychronopoulou K, Charisiou N, Papageridis K, Sebastian V, Hinder S, Dabbawala A, AlKhoori A, Baker M, Goula M. The Effect of Ni Addition onto a Cu-Based Ternary Support on the H2 Production over Glycerol Steam Reforming Reaction. Nanomaterials. 2018; 8(11):931. https://doi.org/10.3390/nano8110931
Chicago/Turabian StylePolychronopoulou, Kyriaki, Nikolaos Charisiou, Kyriakos Papageridis, Victor Sebastian, Steven Hinder, Aasif Dabbawala, Ayesha AlKhoori, Mark Baker, and Maria Goula. 2018. "The Effect of Ni Addition onto a Cu-Based Ternary Support on the H2 Production over Glycerol Steam Reforming Reaction" Nanomaterials 8, no. 11: 931. https://doi.org/10.3390/nano8110931
APA StylePolychronopoulou, K., Charisiou, N., Papageridis, K., Sebastian, V., Hinder, S., Dabbawala, A., AlKhoori, A., Baker, M., & Goula, M. (2018). The Effect of Ni Addition onto a Cu-Based Ternary Support on the H2 Production over Glycerol Steam Reforming Reaction. Nanomaterials, 8(11), 931. https://doi.org/10.3390/nano8110931










