Effects of Copper Oxide Nanoparticles on Paddy Soil Properties and Components
Abstract
1. Introduction
2. Materials and Methods
2.1. Soils
2.2. CuO Particles
2.3. Soil Culture Experiment
2.4. Analysis of Soil Properties and Components
2.5. Analysis of Cu Bioavailability
2.6. Synchrotron Radiation X-ray Absorption Fine Structure (XAFS) Analysis
2.7. Statistical Analysis
3. Results
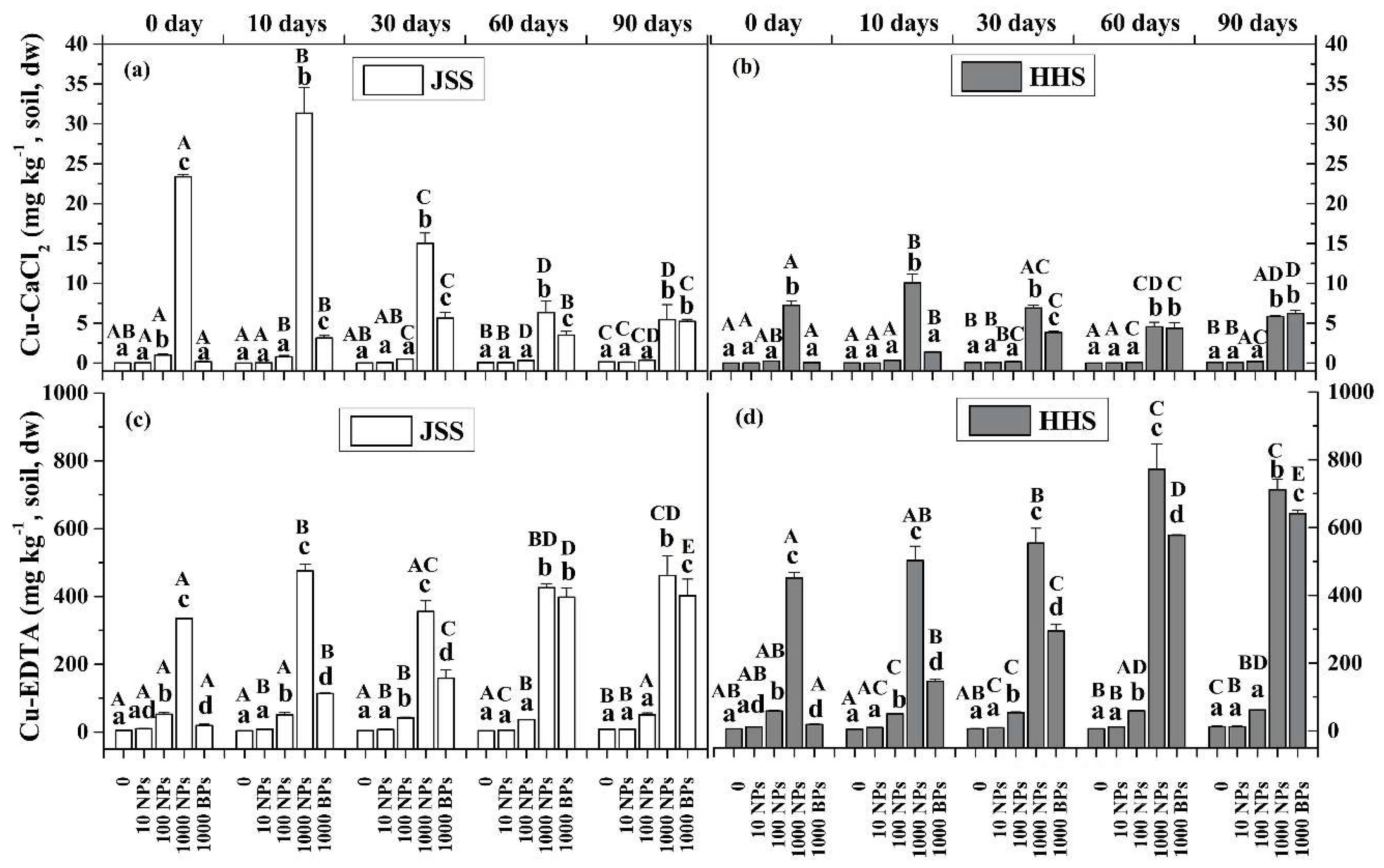
3.1. Transformation of CuO NPs in the Paddy Soil
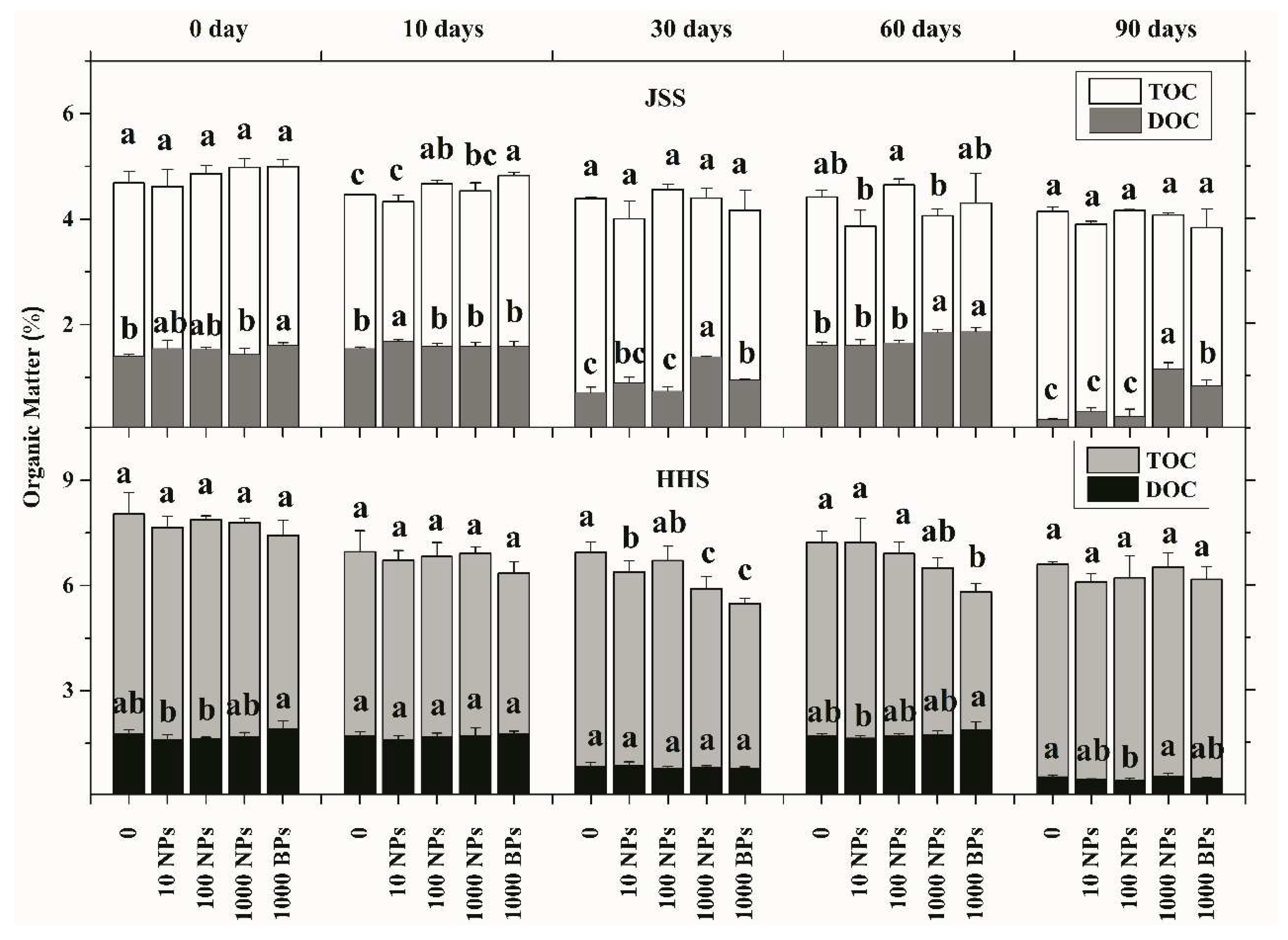
3.2. Effect of CuO NPs on Soil Organic Matter
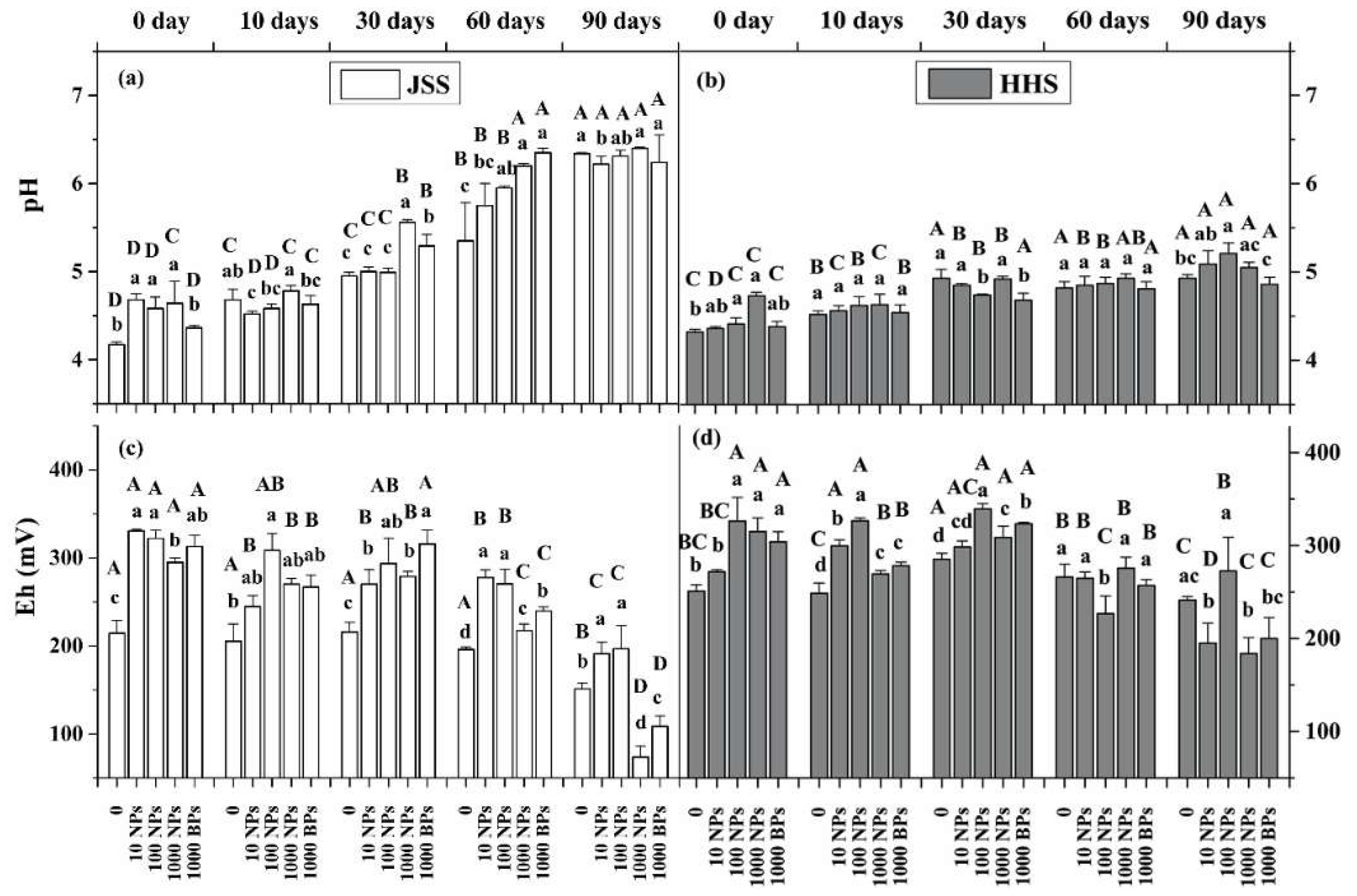
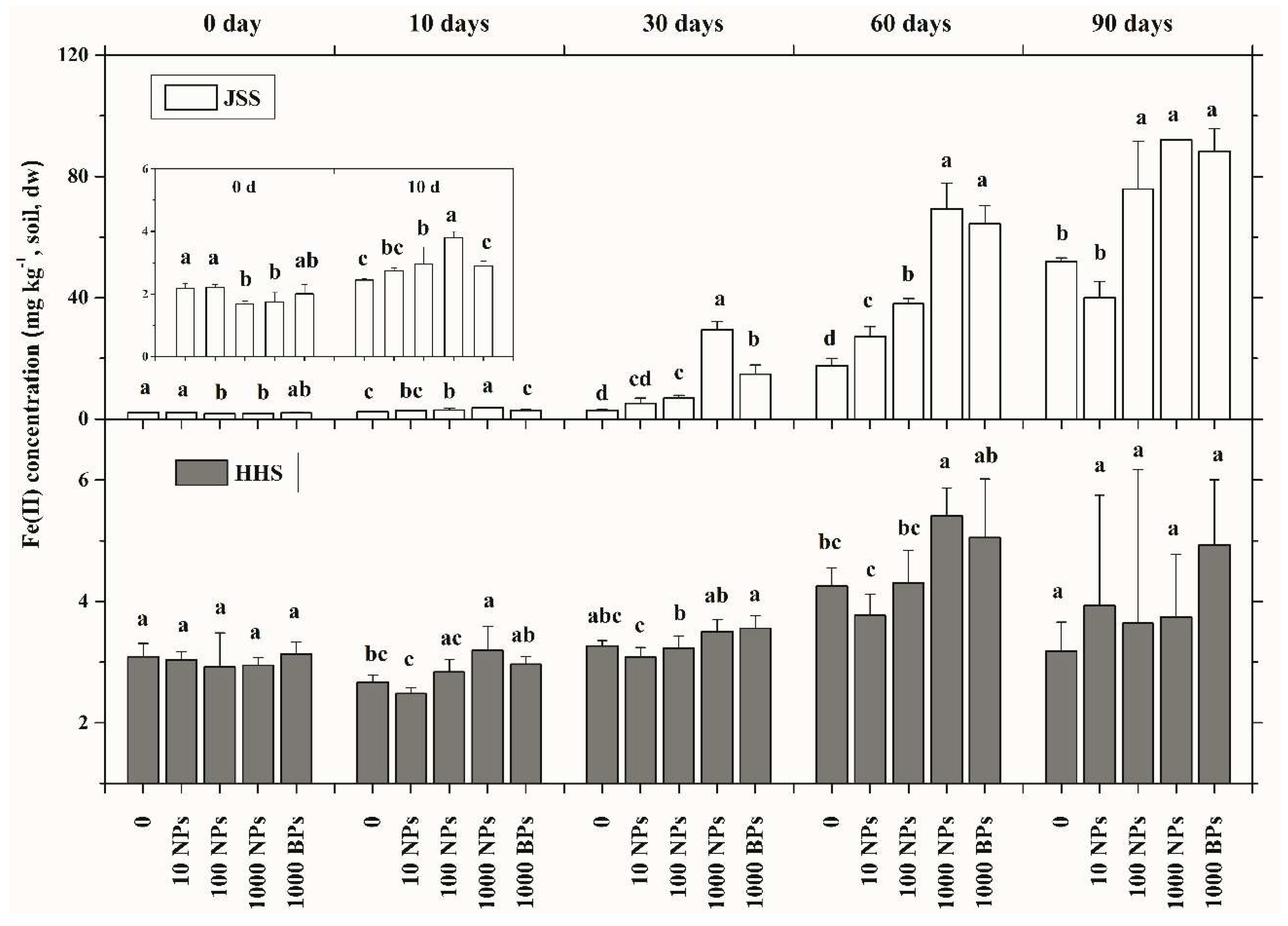
3.3. Effect of CuO NPs on Soil Fe(II) Content
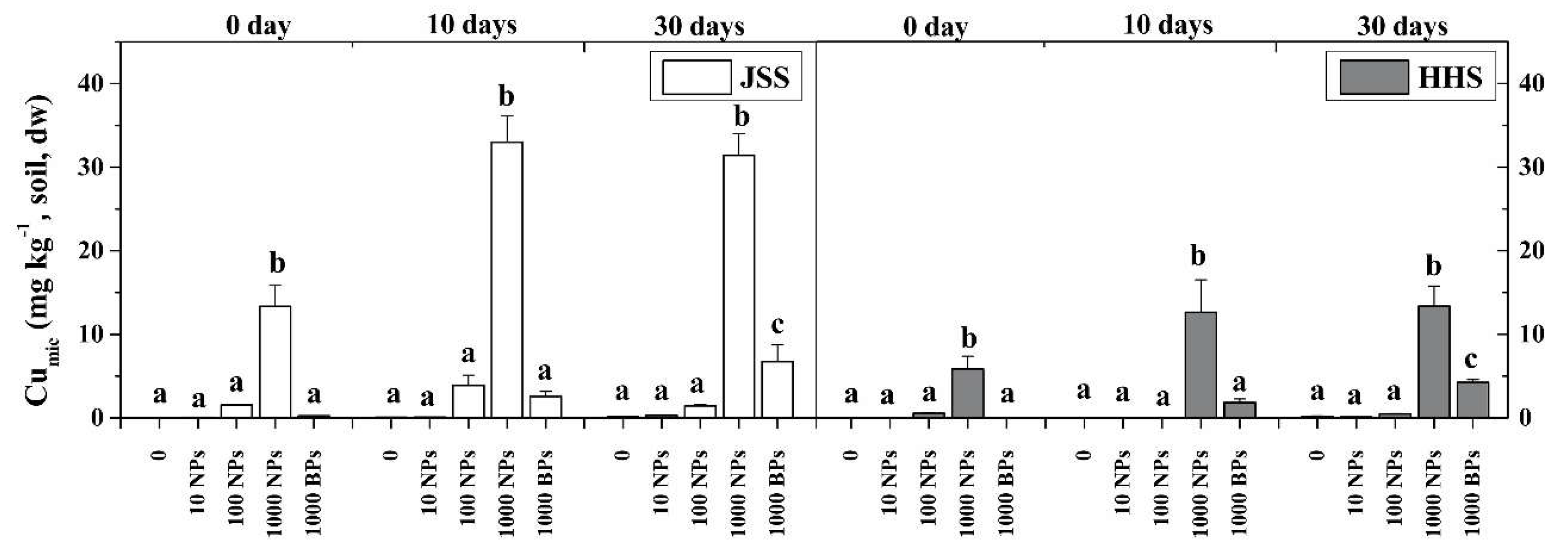
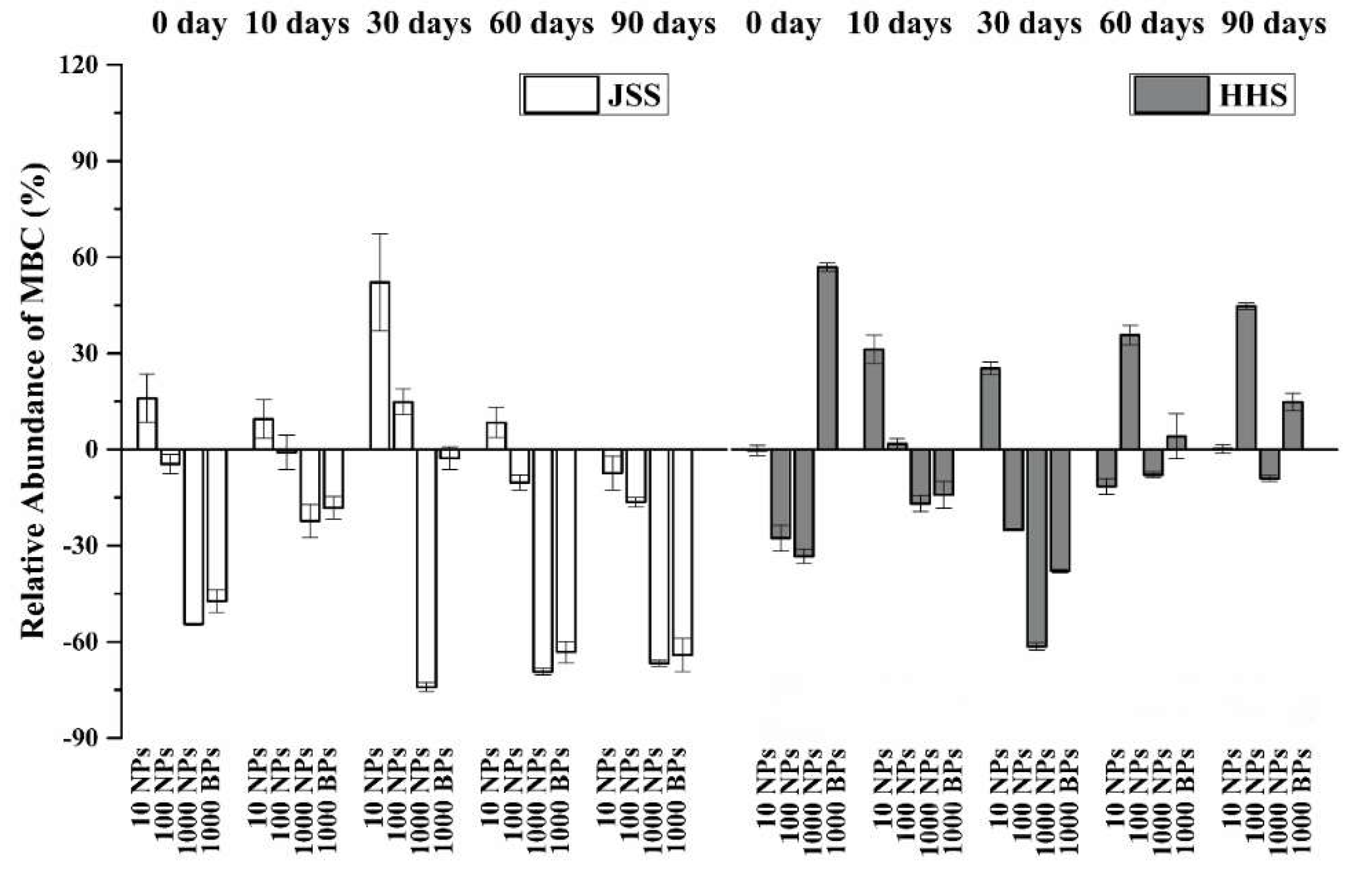
3.4. Effect of CuO NPs on Soil Microbial Biomass Carbon
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Suresh, A.K.; Pelletier, D.A.; Wang, W.; Moon, J.-W.; Gu, B.; Mortensen, N.P.; Allison, D.P.; Joy, D.C.; Phelps, T.J.; Doktycz, M.J. Silver nanocrystallites: Biofabrication using shewanella oneidensis, and an evaluation of their comparative toxicity on gram-negative and gram-positive bacteria. Environ. Sci. Technol. 2010, 44, 5210–5215. [Google Scholar] [CrossRef] [PubMed]
- Atha, D.H.; Wang, H.; Petersen, E.J.; Cleveland, D.; Holbrook, R.D.; Jaruga, P.; Dizdaroglu, M.; Xing, B.; Nelson, B.C. Copper oxide nanoparticle mediated DNA damage in terrestrial plant models. Environ. Sci. Technol. 2012, 46, 1819–1827. [Google Scholar] [CrossRef] [PubMed]
- Gottschalk, F.; Sonderer, T.; Scholz, R.W.; Nowack, B. Modeled environmental concentrations of engineered nanomaterials (TiO2, ZnO, Ag, CNT, fullerenes) for different regions. Environ. Sci. Technol. 2009, 43, 9216–9222. [Google Scholar] [CrossRef] [PubMed]
- Klaine, S.J.; Alvarez, P.J.; Batley, G.E.; Fernandes, T.F.; Handy, R.D.; Lyon, D.Y.; Mahendra, S.; McLaughlin, M.J.; Lead, J.R. Nanomaterials in the environment: Behavior, fate, bioavailability, and effects. Environ. Toxicol. Chem. 2008, 27, 1825–1851. [Google Scholar] [CrossRef] [PubMed]
- Tiede, K.; Hassellöv, M.; Breitbarth, E.; Chaudhry, Q.; Boxall, A. Considerations for environmental fate and ecotoxicity testing to support environmental risk assessments for engineered nanoparticles. J. Chromatogr. A 2009, 1216, 503–509. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, W.; Bowman, B.; Drury, C.; Tan, C.; Lu, X. Indicators of good soil physical quality: Density and storage parameters. Geoderma 2002, 110, 131–146. [Google Scholar] [CrossRef]
- Peňa, N.; Antón, A.; Kamilaris, A.; Fantke, P. Modeling ecotoxicity impacts in vineyard production: Addressing spatial differentiation for copper fungicides. Sci. Total Environ. 2018, 616–617, 796–804. [Google Scholar] [CrossRef] [PubMed]
- Rinklebe, J.; Shaheen, S.M.; Yu, K. Release of As, Ba, Cd, Cu, Pb, and Sr under pre-definite redox conditions in different rice paddy soils originating from the U.S.A. and Asia. Geoderma 2016, 270, 21–32. [Google Scholar] [CrossRef]
- Martinez, R.E.; Marquez, J.E.; Hòa, H.; Gieré, R. Open-pit coal-mining effects on rice paddy soil composition and metal bioavailability to Oryza sativa L. plants in Cam Pha, northeastern Vietnam. Environ. Sci. Pollut. Res. 2013, 20, 7686–7698. [Google Scholar] [CrossRef] [PubMed]
- Dror, I.; Yaron, B.; Berkowitz, B. Abiotic soil changes induced by engineered nanomaterials: A critical review. J. Contam. Hydrol. 2015, 181, 3–16. [Google Scholar] [CrossRef] [PubMed]
- Pan, B.; Xing, B. Applications and implications of manufactured nanoparticles in soils: A review. Eur. J. Soil Sci. 2012, 63, 437–456. [Google Scholar] [CrossRef]
- Fang, J.; Shan, X.; Wen, B.; Lin, J.; Owens, G. Stability of titania nanoparticles in soil suspensions and transport in saturated homogeneous soil columns. Environ. Pollut. 2009, 157, 1101–1109. [Google Scholar] [CrossRef] [PubMed]
- Hyung, H.; Fortner, J.D.; Hughes, J.B.; Kim, J.-H. Natural organic matter stabilizes carbon nanotubes in the aqueous phase. Environ. Sci. Technol. 2007, 41, 179–184. [Google Scholar] [CrossRef] [PubMed]
- Cheng, P.; Chen, X.; Qinglin, L.; Lijuan, S.; Yongming, L.; Jiyan, S. Fate and transformation of CuO nanoparticles in the soil-rice system during the life cycle of rice plants. Environ. Sci. Technol. 2017, 51, 4907–4917. [Google Scholar] [CrossRef]
- Xu, C.; Peng, C.; Sun, L.; Zhang, S.; Huang, H.; Chen, Y.; Shi, J. Distinctive effects of TiO2 and CuO nanoparticles on soil microbes and their community structures in flooded paddy soil. Soil Biol. Biochem. 2015, 86, 24–33. [Google Scholar] [CrossRef]
- Wiesner, M.R.; Lowry, G.V.; Alvarez, P.; Dionysiou, D.; Biswas, P. Assessing the risks of manufactured nanomaterials. Environ. Sci. Technol. 2006, 40, 4336–4345. [Google Scholar] [CrossRef] [PubMed]
- Lamparter, A.; Bachmann, J.; Goebel, M.-O.; Woche, S. Carbon mineralization in soil: Impact of wetting–drying, aggregation and water repellency. Geoderma 2009, 150, 324–333. [Google Scholar] [CrossRef]
- Shi, J.; Peng, C.; Yang, Y.; Yang, J.; Zhang, H.; Yuan, X.; Chen, Y.; Hu, T. Phytotoxicity and accumulation of copper oxide nanoparticles to the Cu-tolerant plant Elsholtzia splendens. Nanotoxicology 2014, 8, 179–188. [Google Scholar] [CrossRef] [PubMed]
- Brookes, P. The use of microbial parameters in monitoring soil pollution by heavy metals. Biol. Fertil. Soils 1995, 19, 269–279. [Google Scholar] [CrossRef]
- Vance, E.; Brookes, P.; Jenkinson, D. An extraction method for measuring soil microbial biomass C. Soil Biol. Biochem. 1987, 19, 703–707. [Google Scholar] [CrossRef]
- Jaenicke, S.; Ander, C.; Bekel, T.; Bisdorf, R.; Dröge, M.; Gartemann, K.-H.; Jünemann, S.; Kaiser, O.; Krause, L.; Tille, F. Comparative and joint analysis of two metagenomic datasets from a biogas fermenter obtained by 454-pyrosequencing. PLoS ONE 2011, 6, e14519. [Google Scholar] [CrossRef] [PubMed]
- Lakanen, E.; Erviö, R. A comparison of eight extractants for the determination of plant available micronutrients in soils. Suomen Maataloustieteellisen Seuran Julkaisuja 1971, 123, 223–232. [Google Scholar]
- Lindsay, W.; Norvell, W.A. Development of a DTPA soil test for zinc, iron, manganese, and copper. Soil Sci. Soc. Am. J. 1978, 42, 421–428. [Google Scholar] [CrossRef]
- Quevauviller, P. Operationally defined extraction procedures for soil and sediment analysis I. Standardization. TrAC Trends Anal. Chem. 1998, 17, 289–298. [Google Scholar] [CrossRef]
- Khan, K.S.; Heinze, S.; Joergensen, R.G. Simultaneous measurement of S, macronutrients, and heavy metals in the soil microbial biomass with CHCl3 fumigation and NH4NO3 extraction. Soil Biol. Biochem. 2009, 41, 309–314. [Google Scholar] [CrossRef]
- Borm, P.; Klaessig, F.C.; Landry, T.D.; Moudgil, B.; Pauluhn, J.; Thomas, K.; Trottier, R.; Wood, S. Research strategies for safety evaluation of nanomaterials, part V: Role of dissolution in biological fate and effects of nanoscale particles. Toxicol. Sci. 2006, 90, 23–32. [Google Scholar] [CrossRef] [PubMed]
- Voegelin, A.; Pfister, S.; Scheinost, A.C.; Marcus, M.A.; Kretzschmar, R. Changes in zinc speciation in field soil after contamination with zinc oxide. Environ. Sci. Technol. 2005, 39, 6616–6623. [Google Scholar] [CrossRef] [PubMed]
- de Santiago-Martín, A.; Constantin, B.; Guesdon, G.; Kagambega, N.; Raymond, S.; Cloutier, R.G. Bioavailability of engineered nanoparticles in soil systems. J. Hazard. Toxic Radioact. Waste 2016, 20, B4015001. [Google Scholar] [CrossRef]
- Deonarine, A.; Lau, B.L.; Aiken, G.R.; Ryan, J.N.; Hsu-Kim, H. Effects of humic substances on precipitation and aggregation of zinc sulfide nanoparticles. Environ. Sci. Technol. 2011, 45, 3217–3223. [Google Scholar] [CrossRef] [PubMed]
- Rathnayake, S.; Unrine, J.M.; Judy, J.; Miller, A.F.; Rao, W.; Bertsch, P.M. Multitechnique investigation of the pH dependence of phosphate induced transformations of ZnO nanoparticles. Environ. Sci. Technol. 2014, 48, 4757–4764. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Von Dem Bussche, A.; Kabadi, P.K.; Kane, A.B.; Hurt, R.H. Biological and environmental transformations of copper-based nanomaterials. ACS Nano 2013, 7, 8715–8727. [Google Scholar] [CrossRef] [PubMed]
- Deng, R.; Lin, D.; Zhu, L.; Majumdar, S.; White, J.C.; Gardea-Torresdey, J.L.; Xing, B. Nanoparticle interactions with co-existing contaminants: Joint toxicity, bioaccumulation and risk. Nanotoxicology 2017, 11, 591–612. [Google Scholar] [CrossRef] [PubMed]
- Naasz, S.; Altenburger, R.; Kühenl, D. Environmental mixtures of nanomaterials and chemicals: The Trojan-horse phenomenon and its relevance for ecotoxicity. Sci. Total Environ. 2018, 635, 1170–1181. [Google Scholar] [CrossRef] [PubMed]
- Misra, S.K.; Dybowska, A.; Berhanu, D.; Luoma, S.N.; Valsami-Jones, E. The complexity of nanoparticle dissolution and its importance in nanotoxicological studies. Sci. Total Environ. 2012, 438, 225–232. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Kim, K.; Shon, H.; Kim, S.D.; Cho, J. Biotoxicity of nanoparticles: Effect of natural organic matter. J. Nanoparticles Res. 2011, 13, 3051–3061. [Google Scholar] [CrossRef]
- Nurmi, J.T.; Tratnyek, P.G.; Sarathy, V.; Baer, D.R.; Amonette, J.E.; Pecher, K.; Wang, C.; Linehan, J.C.; Matson, D.W.; Penn, R.L. Characterization and properties of metallic iron nanoparticles: Spectroscopy, electrochemistry, and kinetics. Environ. Sci. Technol. 2005, 39, 1221–1230. [Google Scholar] [CrossRef] [PubMed]
- Cullen, L.G.; Tilston, E.L.; Mitchell, G.R.; Collins, C.D.; Shaw, L.J. Assessing the impact of nano-and micro-scale zerovalent iron particles on soil microbial activities: Particle reactivity interferes with assay conditions and interpretation of genuine microbial effects. Chemosphere 2011, 82, 1675–1682. [Google Scholar] [CrossRef] [PubMed]
- Dinesh, R.; Anandaraj, M.; Srinivasan, V.; Hamza, S. Engineered nanoparticles in the soil and their potential implications to microbial activity. Geoderma 2012, 173, 19–27. [Google Scholar] [CrossRef]
- Niedermeier, A.; Robinson, J. Hydrological controls on soil redox dynamics in a peat-based, restored wetland. Geoderma 2007, 137, 318–326. [Google Scholar] [CrossRef]
- Frenk, S.; Ben-Moshe, T.; Dror, I.; Berkowitz, B.; Minz, D. Effect of metal oxide nanoparticles on microbial community structure and function in two different soil types. PLoS ONE 2013, 8, e84441. [Google Scholar] [CrossRef] [PubMed]
- Liang, C.; Das, K.; McClendon, R. The influence of temperature and moisture contents regimes on the aerobic microbial activity of a biosolids composting blend. Bioresour. Technol. 2003, 86, 131–137. [Google Scholar] [CrossRef]
- Li, F.; Wang, X.; Li, Y.; Liu, C.; Zeng, F.; Zhang, L.; Hao, M.; Ruan, H. Enhancement of the reductive transformation of pentachlorophenol by polycarboxylic acids at the iron oxide–water interface. J. Colloid Interface Sci. 2008, 321, 332–341. [Google Scholar] [CrossRef] [PubMed]
- Vargas, M.; Kashefi, K.; Blunt-Harris, E.L.; Lovley, D.R. Microbiological evidence for Fe (III) reduction on early Earth. Nature 1998, 395, 65–67. [Google Scholar] [CrossRef] [PubMed]
- Liesack, W.; Schnell, S.; Revsbech, N.P. Microbiology of flooded rice paddies. FEMS Microbiol. Rev. 2000, 24, 625–645. [Google Scholar] [CrossRef] [PubMed]
- Weber, K.A.; Achenbach, L.A.; Coates, J.D. Microorganisms pumping iron: Anaerobic microbial iron oxidation and reduction. Nat. Rev. Microbiol. 2006, 4, 752–764. [Google Scholar] [CrossRef] [PubMed]
- Maithreepala, R.; Doong, R.-A. Synergistic effect of copper ion on the reductive dechlorination of carbon tetrachloride by surface-bound Fe (II) associated with goethite. Environ. Sci. Technol. 2004, 38, 260–268. [Google Scholar] [CrossRef] [PubMed]
- Maithreepala, R.; Doong, R.-A. Reductive dechlorination of carbon tetrachloride in aqueous solutions containing ferrous and copper ions. Environ. Sci. Technol. 2004, 38, 6676–6684. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.-K.; Tao, L.; Chen, M.-J.; Li, F.-B. Effects of the FeII/CuII interaction on copper aging enhancement and pentachlorophenol reductive transformation in paddy soil. J. Agric. Food Chem. 2012, 60, 630–638. [Google Scholar] [CrossRef] [PubMed]
- Bosch, J.; Heister, K.; Hofmann, T.; Meckenstock, R.U. Nanosized iron oxide colloids strongly enhance microbial iron reduction. Appl. Environ. Microbiol. 2010, 76, 184–189. [Google Scholar] [CrossRef] [PubMed]
- Kerisit, S.; Rosso, K.M.; Dupuis, M.; Valiev, M. Molecular computational investigation of electron-transfer kinetics across cytochrome-iron oxide interfaces. J. Physi. Chem. C 2007, 111, 11363–11375. [Google Scholar] [CrossRef]
- Lower, B.H.; Shi, L.; Yongsunthon, R.; Droubay, T.C.; McCready, D.E.; Lower, S.K. Specific bonds between an iron oxide surface and outer membrane cytochromes MtrC and OmcA from Shewanella oneidensis MR-1. J. Bacteriol. 2007, 189, 4944–4952. [Google Scholar] [CrossRef] [PubMed]
- Ben-Moshe, T.; Frenk, S.; Dror, I.; Minz, D.; Berkowitz, B. Effects of metal oxide nanoparticles on soil properties. Chemosphere 2013, 90, 640–646. [Google Scholar] [CrossRef] [PubMed]
- Ben-Moshe, T.; Dror, I.; Berkowitz, B. Oxidation of organic pollutants in aqueous solutions by nanosized copper oxide catalysts. Appl. Catal. B Environ. 2009, 85, 207–211. [Google Scholar] [CrossRef]
- Fink, L.; Dror, I.; Berkowitz, B. Enrofloxacin oxidative degradation facilitated by metal oxide nanoparticles. Chemosphere 2012, 86, 144–149. [Google Scholar] [CrossRef] [PubMed]
- Zhou, D.-M.; Jin, S.-Y.; Wang, Y.-J.; Wang, P.; Weng, N.-Y.; Wang, Y. Assessing the impact of iron-based nanoparticles on pH, dissolved organic carbon, and nutrient availability in soils. Soil Sediment Contam. 2012, 21, 101–114. [Google Scholar] [CrossRef]
- Chen, S.; Liu, Y. Study on the photocatalytic degradation of glyphosate by TiO2 photocatalyst. Chemosphere 2007, 67, 1010–1017. [Google Scholar] [CrossRef] [PubMed]
- Stebbing, A. Hormesis—The stimulation of growth by low levels of inhibitors. Sci. Total Environ. 1982, 22, 213–234. [Google Scholar] [CrossRef]
- Vittori Antisari, L.; Carbone, S.; Gatti, A.; Vianello, G.; Nannipieri, P. Toxicity of metal oxide (CeO2, Fe3O4, SnO2) engineered nanoparticles on soil microbial biomass and their distribution in soil. Soil Biol. Biochem. 2013, 60, 87–94. [Google Scholar] [CrossRef]
- Tourinho, P.S.; van Gestel, C.A.M.; Lofts, S.; Svendsen, C.; Soares, A.M.V.M.; Loureiro, S. Metal-based nanoparticles in soil: Fate, behavior, and effects on soil invertebrates. Environ. Toxicol. Chem. 2012, 31, 1679–1692. [Google Scholar] [CrossRef] [PubMed]
- Fabrega, J.; Fawcett, S.R.; Renshaw, J.C.; Lead, J.R. Silver nanoparticle impact on bacterial growth: Effect of pH, concentration, and organic matter. Environ. Sci. Technol. 2009, 43, 7285–7290. [Google Scholar] [CrossRef] [PubMed]
- Rousk, J.; Ackermann, K.; Curling, S.F.; Jones, D.L. Comparative toxicity of nanoparticulate CuO and ZnO to soil bacterial communities. PLoS ONE 2012, 7, e34197. [Google Scholar] [CrossRef] [PubMed]
- Josko, I.; Oleszczuk, P.; Futa, B. The effect of inorganic nanoparticles (ZnO, Cr2O3, CuO and Ni) and their bulk counterparts on enzyme activities in different soils. Geoderma 2014, 232, 528–537. [Google Scholar] [CrossRef]
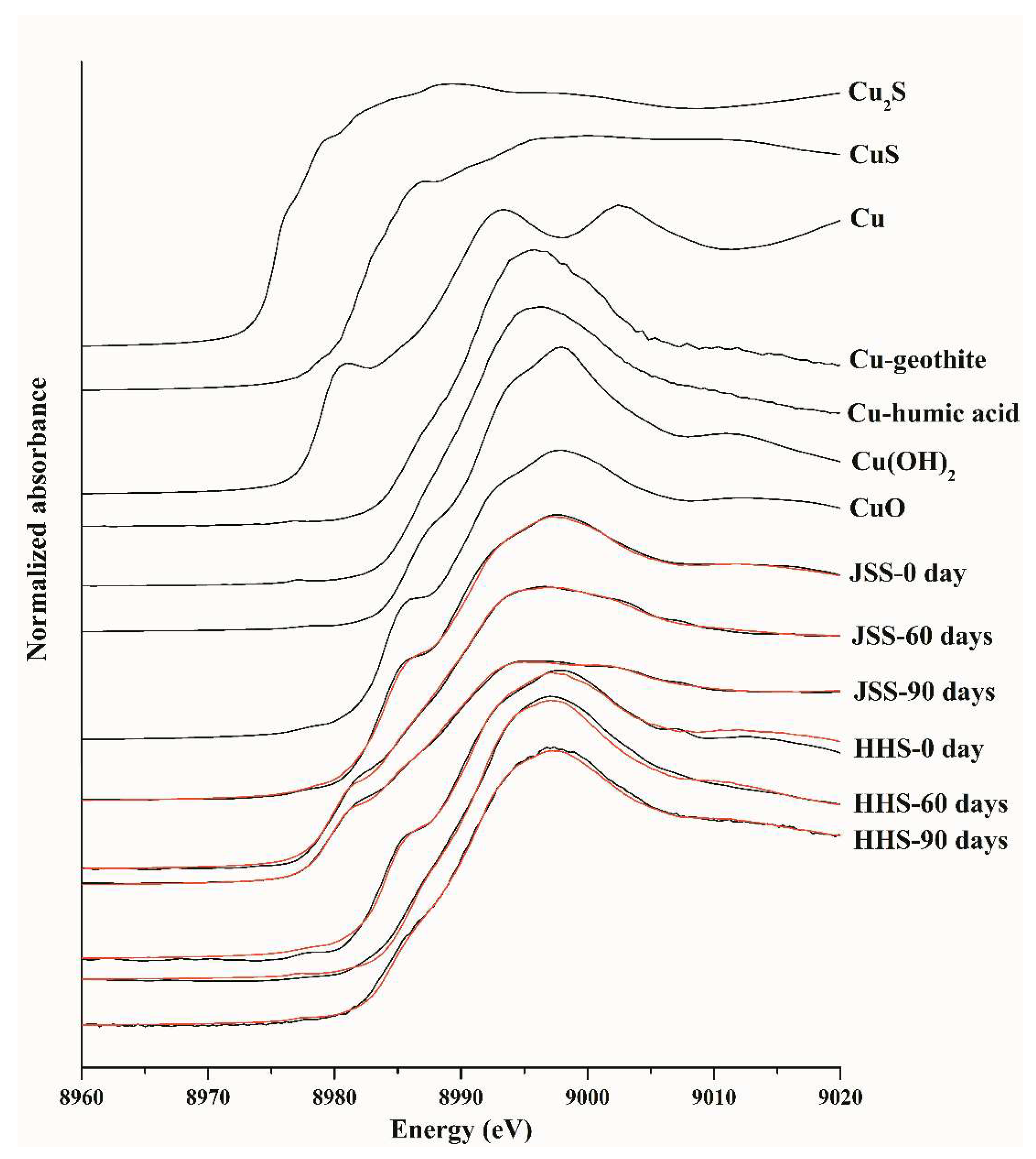
| Reference Compounds | Percentages of Targeted Components (%) | |||||
|---|---|---|---|---|---|---|
| JSS-0 Day | JSS-60 Days | JSS-90 Days | HHS-0 Days | HHS-60 Days | HHS-90 Days | |
| CuO | 69.3 | 0.0 | 0.0 | 88.4 | 0.0 | 41.9 |
| Cu-goethite | 15.2 | 0.0 | 0.0 | 24.3 | 0.0 | 0.0 |
| CuS | 19.9 | 0.0 | 0.0 | 2.5 | 0.0 | 0.0 |
| Cu-humic acid | 0.0 | 24.9 | 25.6 | 0.0 | 64.1 | 43.7 |
| Cu(OH)2 | 0.0 | 11.1 | 0.0 | 0.0 | 39.9 | 19.0 |
| Cu2S | 0.0 | 38.7 | 40.3 | 0.0 | 2.6 | 0.0 |
| Cu | 0.0 | 27.5 | 35.4 | 0.0 | 0.0 | 0.0 |
| R factor | 0.0002 | 0.0001 | 0.0001 | 0.0005 | 0.0006 | 0.0003 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shi, J.; Ye, J.; Fang, H.; Zhang, S.; Xu, C. Effects of Copper Oxide Nanoparticles on Paddy Soil Properties and Components. Nanomaterials 2018, 8, 839. https://doi.org/10.3390/nano8100839
Shi J, Ye J, Fang H, Zhang S, Xu C. Effects of Copper Oxide Nanoparticles on Paddy Soil Properties and Components. Nanomaterials. 2018; 8(10):839. https://doi.org/10.3390/nano8100839
Chicago/Turabian StyleShi, Jiyan, Jien Ye, Huaxiang Fang, Shu Zhang, and Chen Xu. 2018. "Effects of Copper Oxide Nanoparticles on Paddy Soil Properties and Components" Nanomaterials 8, no. 10: 839. https://doi.org/10.3390/nano8100839
APA StyleShi, J., Ye, J., Fang, H., Zhang, S., & Xu, C. (2018). Effects of Copper Oxide Nanoparticles on Paddy Soil Properties and Components. Nanomaterials, 8(10), 839. https://doi.org/10.3390/nano8100839




