RETRACTED: Enhanced Adsorption of Zn(II) onto Graphene Oxides Investigated Using Batch and Modeling Techniques
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Synthesis and Characterization of GO
2.3. Batch Adsorption Experiments
2.4. Surface Complexation Modeling
3. Results and Discussion
3.1. Characterization
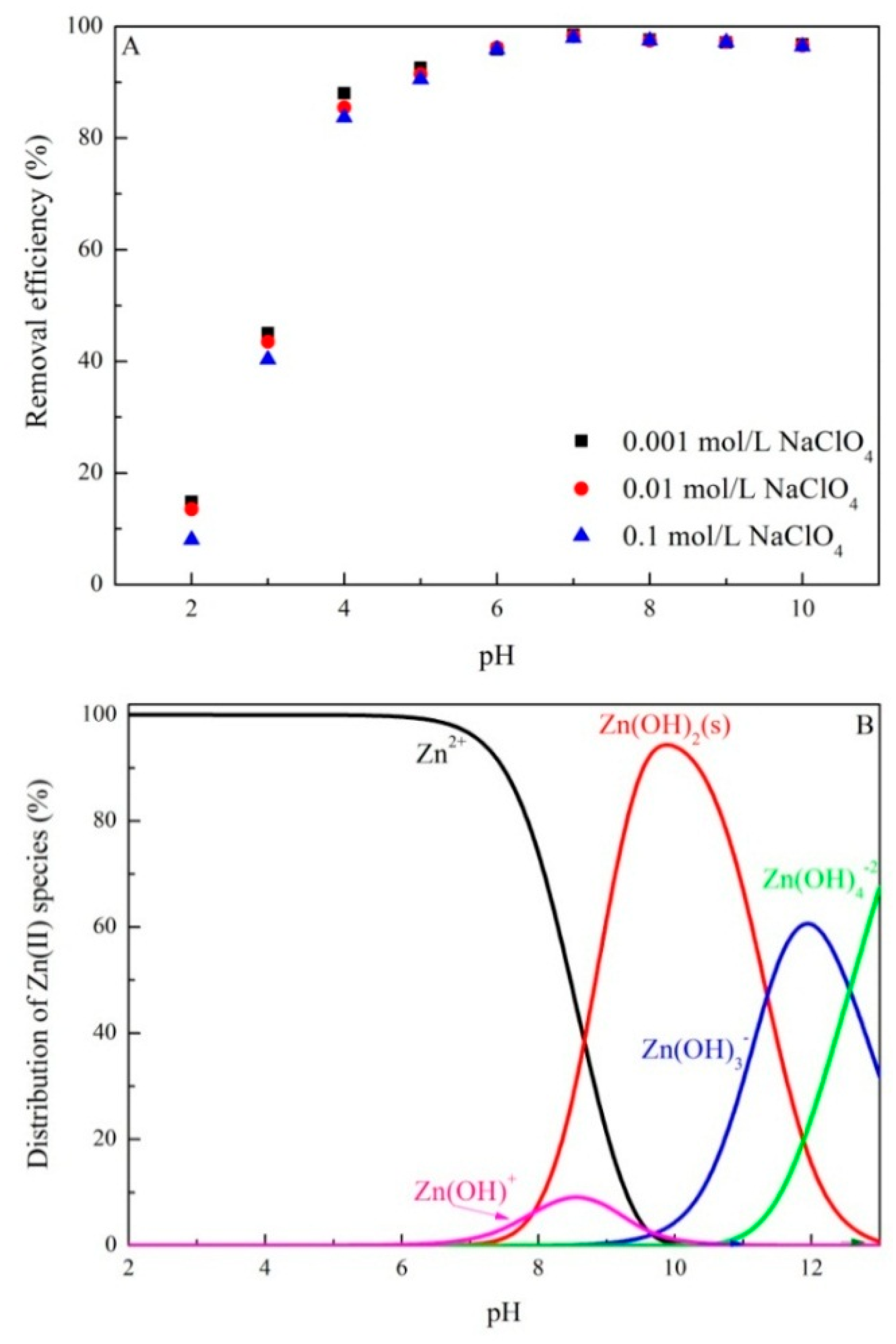
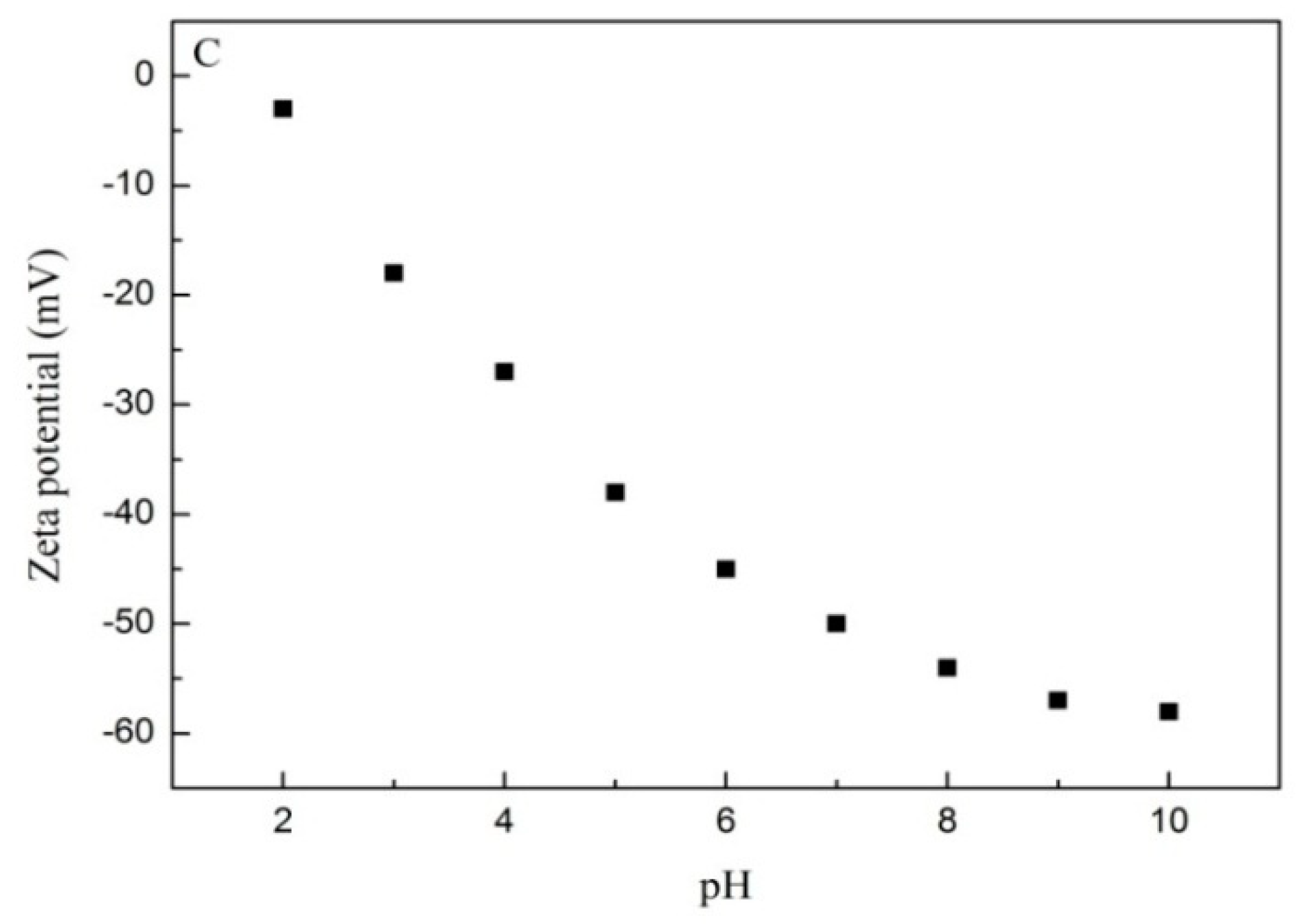
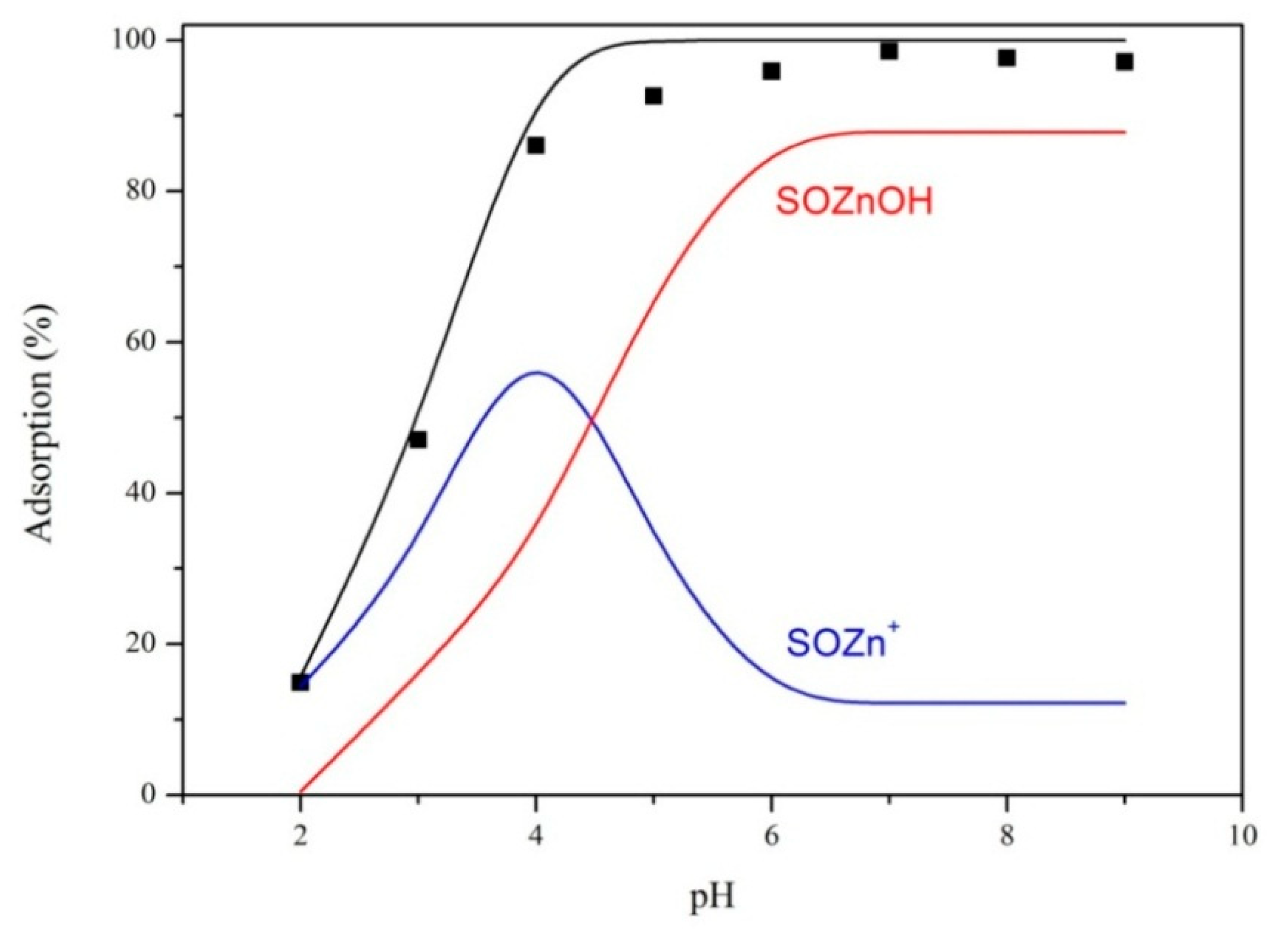
3.2. Effect of pH and Ionic Strength
3.3. Adsorption Isotherms
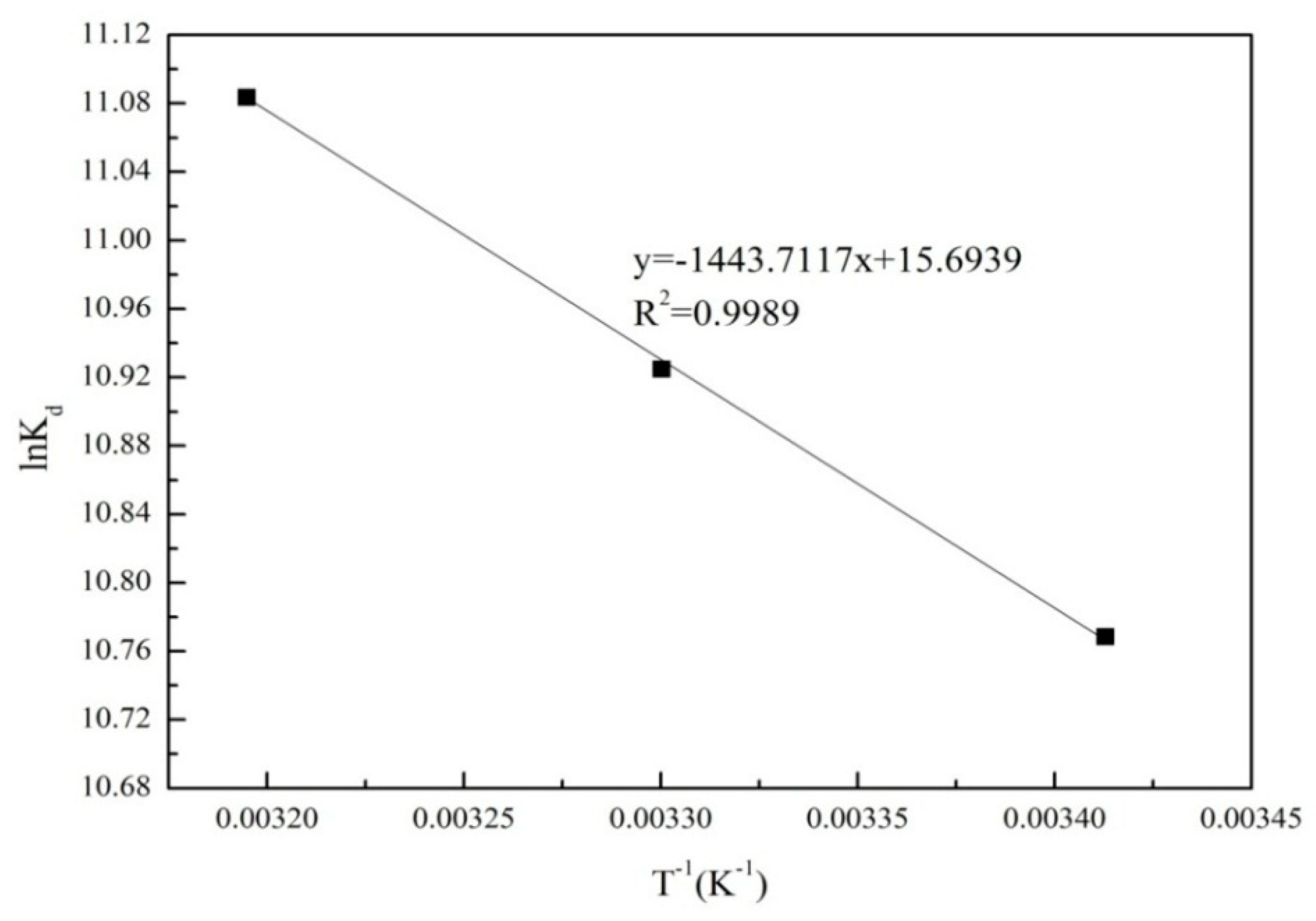
3.4. Thermodynamic Parameters
3.5. Adsorption Kinetics
3.6. Surface Complexation Modeling
3.7. Regeneration and Reusability
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Lingamdinne, L.P.; Koduru, J.R.; Choi, Y.-L.; Chang, Y.-Y.; Yang, J.-K. Studies on removal of Pb(II) and Cr(III) using graphene oxide based inverse spinel nickel ferrite nano-composite as sorbent. Hydrometallurgy 2016, 165, 64–72. [Google Scholar] [CrossRef]
- Wu, S.; Kong, L.; Liu, J. Removal of mercury and fluoride from aqueous solutions by three-dimensional reduced-graphene oxide aerogel. Res. Chem. Intermed. 2015, 42, 4513–4530. [Google Scholar] [CrossRef]
- Wang, H.; Yuan, X.; Wu, Y.; Huang, H.; Zeng, G.; Liu, Y.; Wang, X.; Lin, N.; Qi, Y. Adsorption characteristics and behaviors of graphene oxide for Zn(II) removal from aqueous solution. Appl. Surf. Sci. 2013, 279, 432–440. [Google Scholar] [CrossRef]
- Ostroski, I.C.; Barros, M.A.; Silva, E.A.; Dantas, J.H.; Arroyo, P.A.; Lima, O.C. A comparative study for the ion exchange of Fe(III) and Zn(II) on zeolite NaY. J. Hazard. Mater. 2009, 161, 1404–1412. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, L.S.; Rodrigues, M.S.; de Carvalho, J.C.M.; Lodi, A.; Finocchio, E.; Perego, P.; Converti, A. Adsorption of Ni2+, Zn2+ and Pb2+ onto dry biomass of Arthrospira (Spirulina) platensis and Chlorella vulgaris. I. Single metal systems. Chem. Eng. J. 2011, 173, 326–333. [Google Scholar] [CrossRef]
- Lu, C.; Chiu, H. Chemical modification of multiwalled carbon nanotubes for sorption of Zn2+ from aqueous solution. Chem. Eng. J. 2008, 139, 462–468. [Google Scholar] [CrossRef]
- Khademi, Z.; Ramavandi, B.; Ghaneian, M.T. The behaviors and characteristics of a mesoporous activated carbon prepared from Tamarix hispida for Zn(II) adsorption from wastewater. J. Environl. Chem. Eng. 2015, 3, 2057–2067. [Google Scholar] [CrossRef]
- Liu, H.; Zhu, Y.; Xu, B.; Li, P.; Sun, Y.; Chen, T. Mechanical investigation of U(VI) on pyrrhotite by batch, EXAFS and modeling techniques. J. Hazard. Mater. 2017, 322, 488–498. [Google Scholar] [CrossRef] [PubMed]
- Ghasemi, N.; Ghasemi, M.; Moazeni, S.; Ghasemi, P.; Alharbi, N.S.; Gupta, V.K.; Agarwal, S.; Burakova, I.V.; Tkachev, A.G. Zn(II) removal by amino-functionalized magnetic nanoparticles: Kinetics, isotherm, and thermodynamic aspects of adsorption. J. Ind. Eng. Chem. 2018, 62, 302–310. [Google Scholar] [CrossRef]
- Krzisnik, N.; Mladenovic, A.; Skapin, A.S.; Skrlep, L.; Scancar, J.; Milacic, R. Nanoscale zero-valent iron for the removal of Zn2+, Zn(II)-EDTA and Zn(II)-citrate from aqueous solutions. Sci. Total Environ. 2014, 476–477, 20–28. [Google Scholar] [CrossRef] [PubMed]
- Gogoi, H.; Leiviska, T.; Heiderscheidt, E.; Postila, H.; Tanskanen, J. Removal of metals from industrial wastewater and urban runoff by mineral and bio-based sorbents. J. Environ. Manag. 2018, 209, 316–327. [Google Scholar] [CrossRef] [PubMed]
- Peng, W.; Li, H.; Liu, Y.; Song, S. A review on heavy metal ions adsorption from water by graphene oxide and its composites. J. Mol. Liq. 2017, 230, 496–504. [Google Scholar] [CrossRef]
- Sun, Y.; Li, J.; Wang, X. The retention of uranium and europium onto sepiolite investigated by macroscopic, spectroscopic and modeling techniques. Geochim. Cosmochim. Acta 2014, 140, 621–643. [Google Scholar] [CrossRef]
- Sun, Y.; Wang, X.; Ai, Y.; Yu, Z.; Huang, W.; Chen, C.; Hayat, T.; Alsaedi, A.; Wang, X. Interaction of sulfonated graphene oxide with U(VI) studied by spectroscopic analysis and theoretical calculations. Chem. Eng. J. 2017, 310, 292–299. [Google Scholar] [CrossRef]
- Tang, J.; Huang, Y.; Gong, Y.; Lyu, H.; Wang, Q.; Ma, J. Preparation of a novel graphene oxide/Fe-Mn composite and its application for aqueous Hg(II) removal. J. Hazard. Mater. 2016, 316, 151–158. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhang, S.; Gao, J.; Chung, T.-S. Layer-by-layer construction of graphene oxide (GO) framework composite membranes for highly efficient heavy metal removal. J. Membrane Sci. 2016, 515, 230–237. [Google Scholar] [CrossRef]
- Tan, P.; Bi, Q.; Hu, Y.; Fang, Z.; Chen, Y.; Cheng, J. Effect of the degree of oxidation and defects of graphene oxide on adsorption of Cu2+ from aqueous solution. Appl. Surf. Sci. 2017, 423, 1141–1151. [Google Scholar] [CrossRef]
- Wan, S.; He, F.; Wu, J.; Wan, W.; Gu, Y.; Gao, B. Rapid and highly selective removal of lead from water using graphene oxide-hydrated manganese oxide nanocomposites. J. Hazard. Mater. 2016, 314, 32–40. [Google Scholar] [CrossRef] [PubMed]
- Yari, M.; Rajabi, M.; Moradi, O.; Yari, A.; Asif, M.; Agarwal, S.; Gupta, V.K. Kinetics of the adsorption of Pb(II) ions from aqueous solutions by graphene oxide and thiol functionalized graphene oxide. J. Mol. Liq. 2015, 209, 50–57. [Google Scholar] [CrossRef]
- Zhang, J.; Gong, J.-L.; Zenga, G.-M.; Ou, X.-M.; Jiang, Y.; Chang, Y.-N.; Guo, M.; Zhang, C.; Liu, H.-Y. Simultaneous removal of humic acid/fulvic acid and lead from landfill leachate using magnetic graphene oxide. Appl. Surf. Sci. 2016, 370, 335–350. [Google Scholar] [CrossRef]
- Sun, Y.; Shao, D.; Chen, C.; Yang, S.; Wang, X. Highly efficient enrichment of radionuclides on graphene oxide-supported polyaniline. Environ. Sci. Technol. 2013, 47, 9904–9910. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Chen, C.; Tan, X.; Shao, D.; Li, J.; Zhao, G.; Yang, S.; Wang, Q.; Wang, X. Enhanced adsorption of Eu(III) on mesoporous Al2O3/expanded graphite composites investigated by macroscopic and microscopic techniques. Dalton Trans. 2012, 41, 13388–13394. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Yang, S.; Chen, Y.; Ding, C.; Cheng, W.; Wang, X. Adsorption and desorption of U(VI) on functionalized graphene oxides: A combined experimental and theoretical study. Environ. Sci. Technol. 2015, 49, 4255–4262. [Google Scholar] [CrossRef] [PubMed]
- Yang, P.; Liu, Q.; Liu, J.; Zhang, H.; Li, Z.; Li, R.; Liu, L.; Wang, J. Bovine serum albumin-coated graphene oxide for effective adsorption of Uranium(VI) from aqueous solutions. Ind. Eng. Chem. Res. 2017, 56, 3588–3598. [Google Scholar] [CrossRef]
- Russo, P.; D’Urso, L.; Hu, A.; Zhou, N.; Compagnini, G. In liquid laser treated graphene oxide for dye removal. Appl. Surf. Sci. 2015, 348, 85–91. [Google Scholar] [CrossRef]
- Parmar, K.R.; Murthy, Z.V.P. Synthesis of acetone reduced graphene oxide/Fe3O4 composite through simple and efficient chemical eduction of exfoliated graphene oxide for removal of dye from aqueous solution. J. Mater. Sci. 2014, 49, 6772–6783. [Google Scholar] [CrossRef]
- Peng, Y.; Li, J. Ammonia adsorption on graphene and graphene oxide: A first-principles study. Front. Environ. Sci. Eng. 2013, 7, 403–411. [Google Scholar] [CrossRef]
- Huang, X.; Pan, M. The highly efficient adsorption of Pb(II) on graphene oxides: A process combined by batch experiments and modeling techniques. J. Mol. Liq. 2016, 215, 410–416. [Google Scholar] [CrossRef]
- Ding, C.; Cheng, W.; Sun, Y.; Wang, X. Determination of chemical affinity of graphene oxide nanosheets with radionuclides investigated by macroscopic, spectroscopic and modeling techniques. Dalton Trans. 2014, 43, 3888–3896. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Chen, T.; Zou, X.; Zhu, M.; Chen, D.; Pan, M. The Adsorption of Cd(II) on Manganese Oxide Investigated by Batch and Modeling Techniques. Int. J. Environ. Res. Public Health 2017, 14, 1145. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Zhang, S.; Chen, C.; Zhao, G.; Yang, X.; Li, J.; Wang, X. Removal of Cu(II) and fulvic acid by graphene oxide nanosheets decorated with Fe3O4 nanoparticles. ACS Appl. Mater. Interfaces 2012, 4, 4991–5000. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Gao, H.; Wang, Y. Uptake of Ni(II) from aqueous solution onto graphene oxide: Investigated by batch and modeling techniques. J. Mol. Liq. 2017, 227, 303–308. [Google Scholar] [CrossRef]
- Raghubanshi, H.; Ngobeni, S.M.; Osikoya, S.O.; Shooto, N.E.; Dikio, C.W.; Naidoo, E.B.; Dikio, E.D.; Pandey, R.K.; Prakash, R. Synthesis of graphene oxide and its application for the adsorption pf Pb2+ from aqueous solution. J. Ind. Eng. Chem. 2017, 47, 169–178. [Google Scholar] [CrossRef]
- Jin, Z.; Sheng, J.; Sun, Y. Characterization of radioactive cobalt on graphene oxide by macroscopic and spectroscopic techniques. J. Radioanal. Nucl. Chem. 2014, 299, 1979–1986. [Google Scholar] [CrossRef]
- Qi, Y.; Yang, M.; Xu, W.; He, S.; Men, Y. Natural polyaccharides-modified graphene oxide for adsorption of organic dyes from aqueous solutions. J. Colloid Interf. Sci. 2017, 486, 84–96. [Google Scholar] [CrossRef] [PubMed]
- Weng, C.-H.; Huang, C.P. Adsorption characteristics of Zn(II) from dilute aqueous solution by fly ash. Colloid Surf. A 2004, 247, 137–143. [Google Scholar] [CrossRef]
- Liu, Z.; Chen, L.; Zhang, Z.; Li, Y.; Dong, Y.; Sun, Y. Synthesis of multi-walled carbon nanotube–hydroxyapatite composites and its application in the sorption of Co(II) from aqueous solutions. J. Mol. Liq. 2013, 179, 46–53. [Google Scholar] [CrossRef]
- Pan, M.; Lin, X.; Xie, J.; Huang, X. Kinetic, equilibrium and thermodynamic studies for phosphate adsorption on aluminum hydroxide modified palygorskite nano-composites. RSC Adv. 2017, 7, 4492–4500. [Google Scholar] [CrossRef]
- Yang, Q.; Yang, G.; Peng, W.; Song, S. Adsorption of Zn(II) on graphene oxide prepared from low-purity of amorphous graphite. Surf. Interface Anal. 2017, 49, 398–404. [Google Scholar] [CrossRef]
- Najafi, F. Removal of zinc(II) ion by graphene oxide(GO) and functionalized graphene oxide-glycine(GO-G) as adsorbents from aqueous solution: Kinetics studies. Int. Nano Lett. 2015, 5, 171–178. [Google Scholar] [CrossRef]
- Huang, J.; Wu, Z.; Chen, L.; Sun, Y. Surface complexation modeling of adsorption of Cd(II) on graphene oxides. J. Mol. Liq. 2015, 209, 753–758. [Google Scholar] [CrossRef]
- Guo, S.; Jiao, P.; Dan, Z.; Duan, N.; Chen, G.; Zhang, J. Preparation of l-arginine modified magnetics adsorbent by one-step method for removal of Zn(II) and Cd(II) from aqueous solution. Chem. Eng. J. 2017, 317, 999–1011. [Google Scholar] [CrossRef]
- Liu, L.; Qiu, G.; Suib, S.L.; Liu, F.; Zhen, L.; Tan, W.; Qin, L. Enhancement of Zn2+ and Ni2+ removal performance using a deionization pseudocapacitor with nanostructured birnessite and its carbon nanotube composite electrodes. Chem. Eng. J. 2017, 328, 464–473. [Google Scholar] [CrossRef]

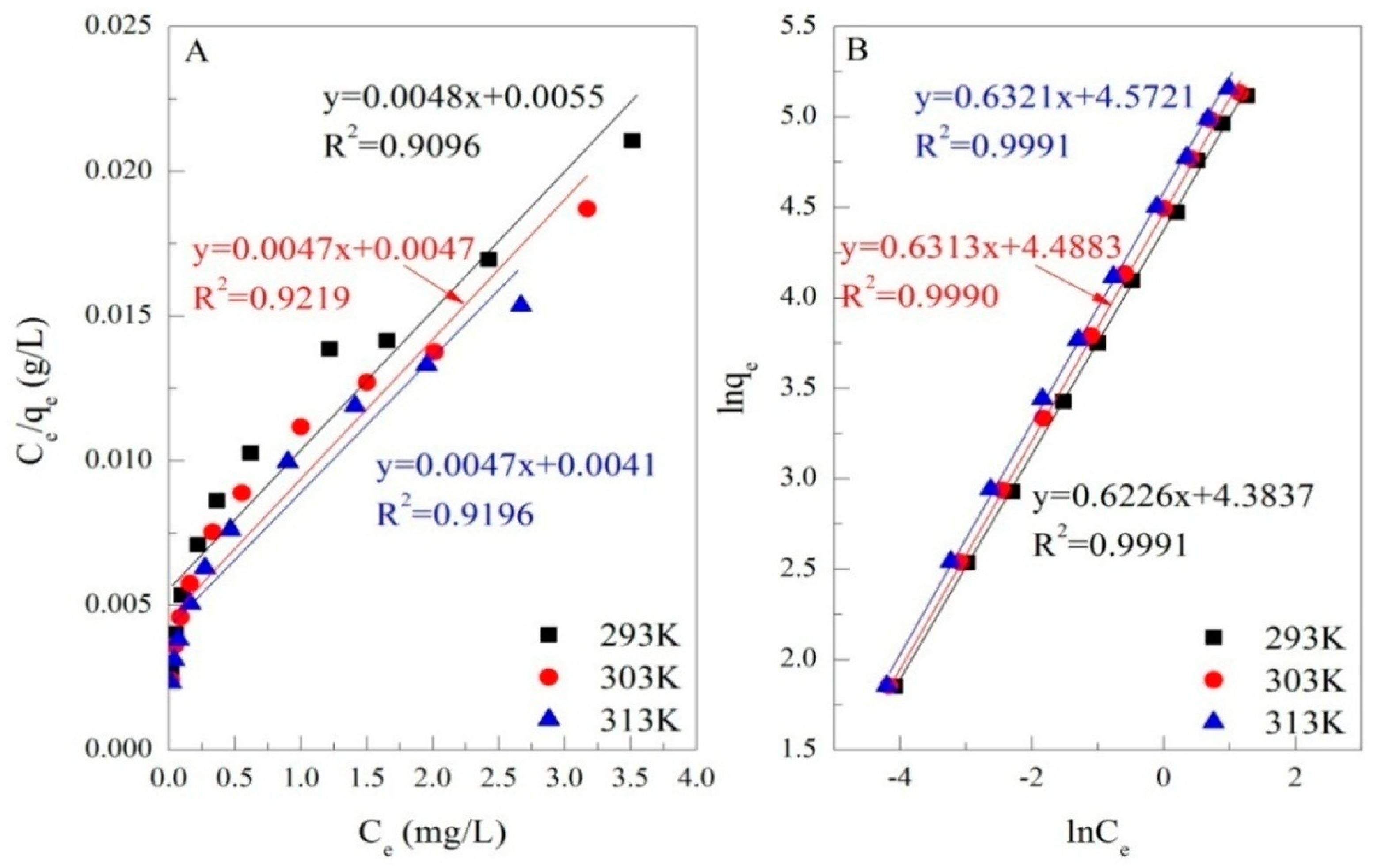
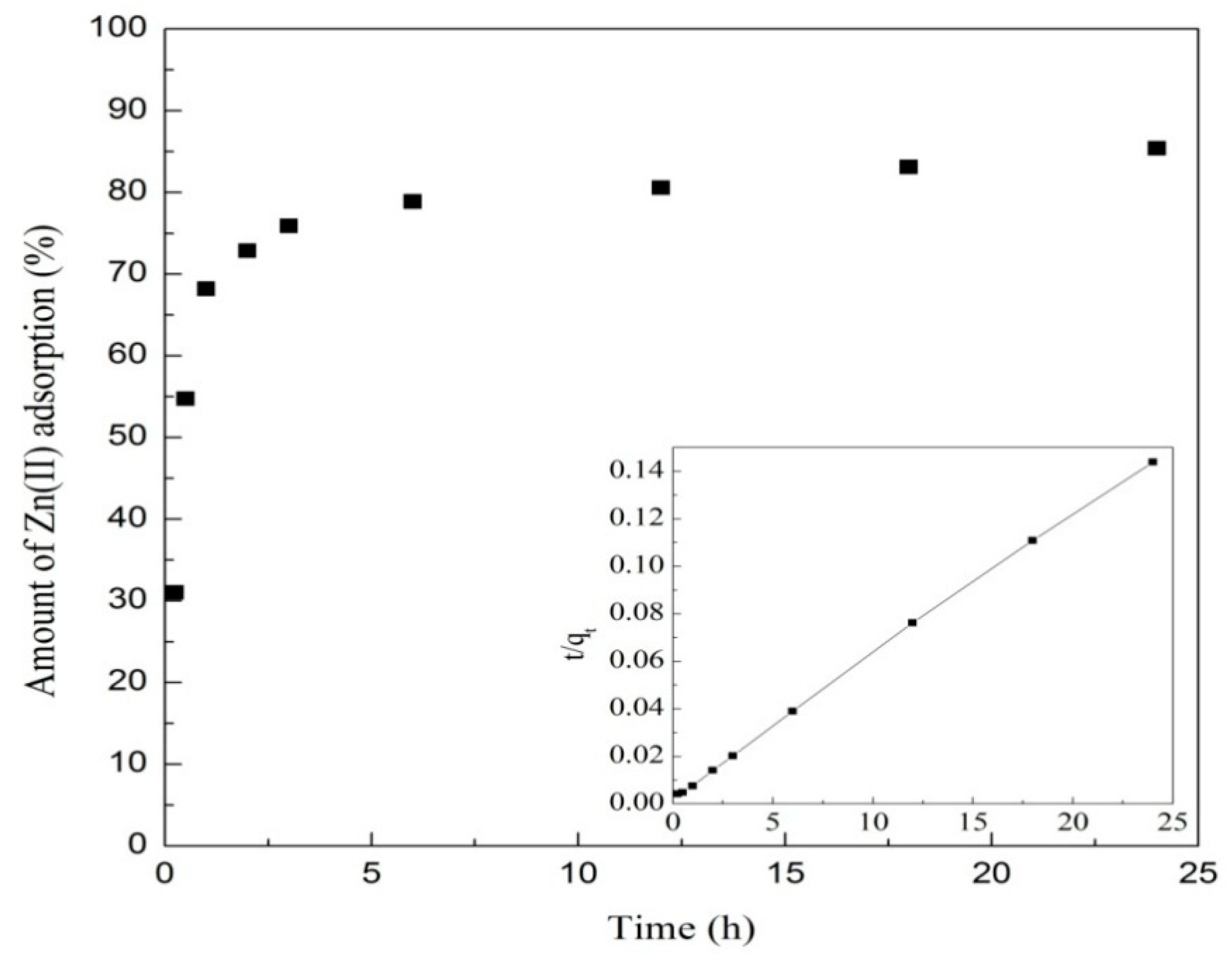

| Isotherm Models | Parameters | T = 293 K | T = 303 K | T = 313 K |
|---|---|---|---|---|
| Langmuir | qm (mg/g) | 208.33 | 210.97 | 211.42 |
| K (L/mg) | 0.8727 | 1.0085 | 1.1537 | |
| R2 | 0.9096 | 0.9219 | 0.9196 | |
| Frenudlich | lnKf (mg1−n·Ln/g) | 4.3837 | 4.4883 | 4.5721 |
| 1/n | 0.6226 | 0.6313 | 0.6321 | |
| R2 | 0.9991 | 0.9990 | 0.9991 |
| T (K) | ΔG0 (kJ/mol) | ΔS0 (J/mol/K) | ΔH0 (kJ/mol) |
|---|---|---|---|
| 293 | −26.23 | 130 | 12.00 |
| 303 | −27.52 | ||
| 313 | −28.84 |
| Kinetic Models | Parameters | T = 293 K | T = 303 K | T = 313 K |
|---|---|---|---|---|
| Pseudo first-order | qe (mg/g) | 78.16 | 96.75 | 92.12 |
| k1 (h−1) | 0.2947 | 0.3328 | 0.292 | |
| R2 | 0.7486 | 0.6888 | 0.7688 | |
| Pseudo second-order | qe (mg/g) | 167.50 | 169.78 | 174.83 |
| k2 (g/mg h) | 0.015 | 0.014 | 0.018 | |
| R2 | 0.9994 | 0.9991 | 0.9992 |
| Equations | LogK |
|---|---|
| Protonation and deprotonation | |
| SOH + H+ = SOH2+ | 4.52 |
| SOH = SO− + H+ | −7.88 |
| Surface complexation modeling | |
| SOH + Zn2+ = SOZn+ + H+ | 3.48 |
| SOH + Zn2+ + H2O = SOZnOH + 2H+ | −8.64 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pan, M.; Wu, G.; Liu, C.; Lin, X.; Huang, X. RETRACTED: Enhanced Adsorption of Zn(II) onto Graphene Oxides Investigated Using Batch and Modeling Techniques. Nanomaterials 2018, 8, 806. https://doi.org/10.3390/nano8100806
Pan M, Wu G, Liu C, Lin X, Huang X. RETRACTED: Enhanced Adsorption of Zn(II) onto Graphene Oxides Investigated Using Batch and Modeling Techniques. Nanomaterials. 2018; 8(10):806. https://doi.org/10.3390/nano8100806
Chicago/Turabian StylePan, Min, Guangxue Wu, Chang Liu, Xinxin Lin, and Xiaoming Huang. 2018. "RETRACTED: Enhanced Adsorption of Zn(II) onto Graphene Oxides Investigated Using Batch and Modeling Techniques" Nanomaterials 8, no. 10: 806. https://doi.org/10.3390/nano8100806
APA StylePan, M., Wu, G., Liu, C., Lin, X., & Huang, X. (2018). RETRACTED: Enhanced Adsorption of Zn(II) onto Graphene Oxides Investigated Using Batch and Modeling Techniques. Nanomaterials, 8(10), 806. https://doi.org/10.3390/nano8100806





