The Development of Non-Enzymatic Glucose Biosensors Based on Electrochemically Prepared Polypyrrole–Chitosan–Titanium Dioxide Nanocomposite Films
Abstract
:1. Introduction
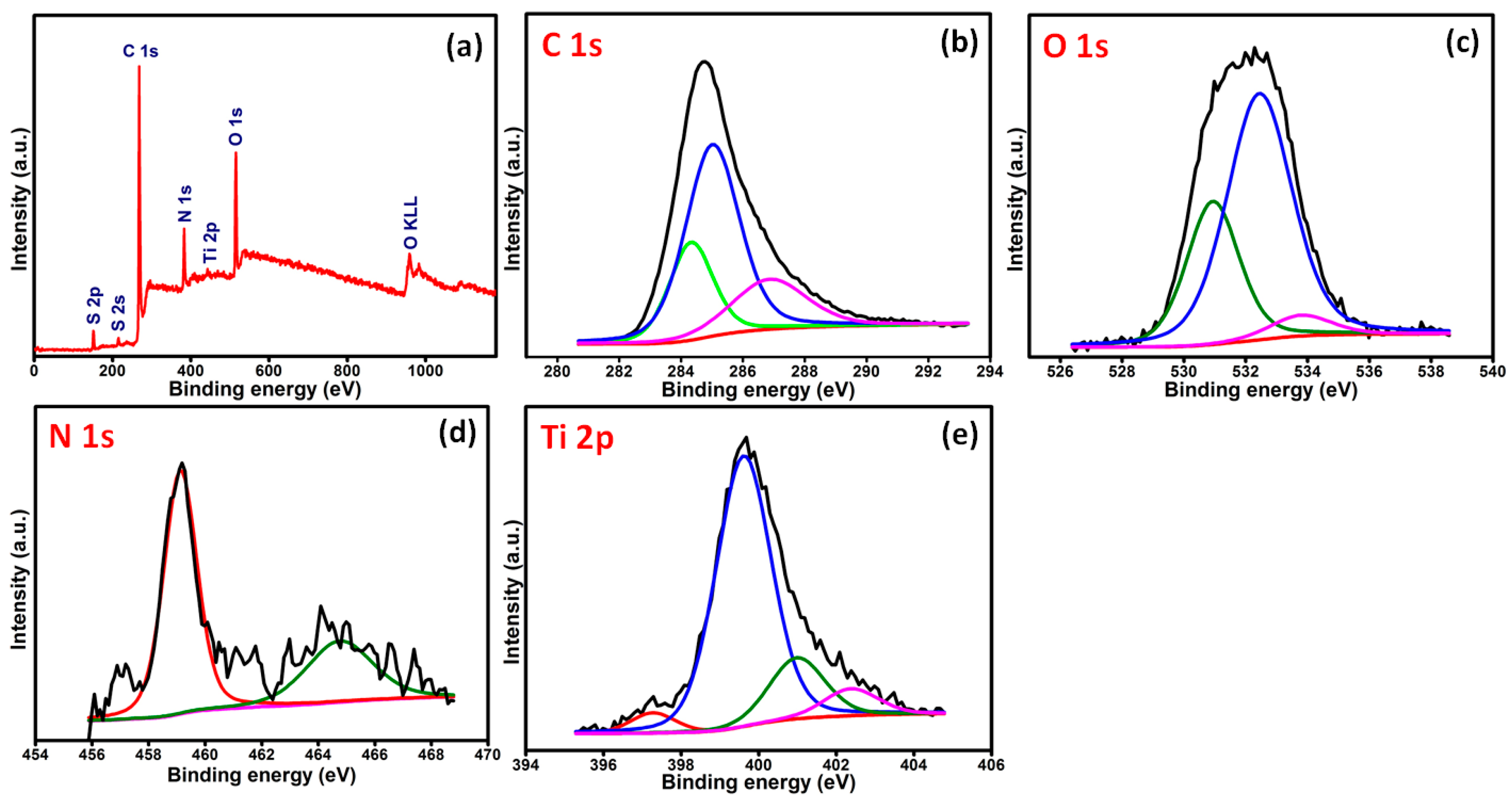
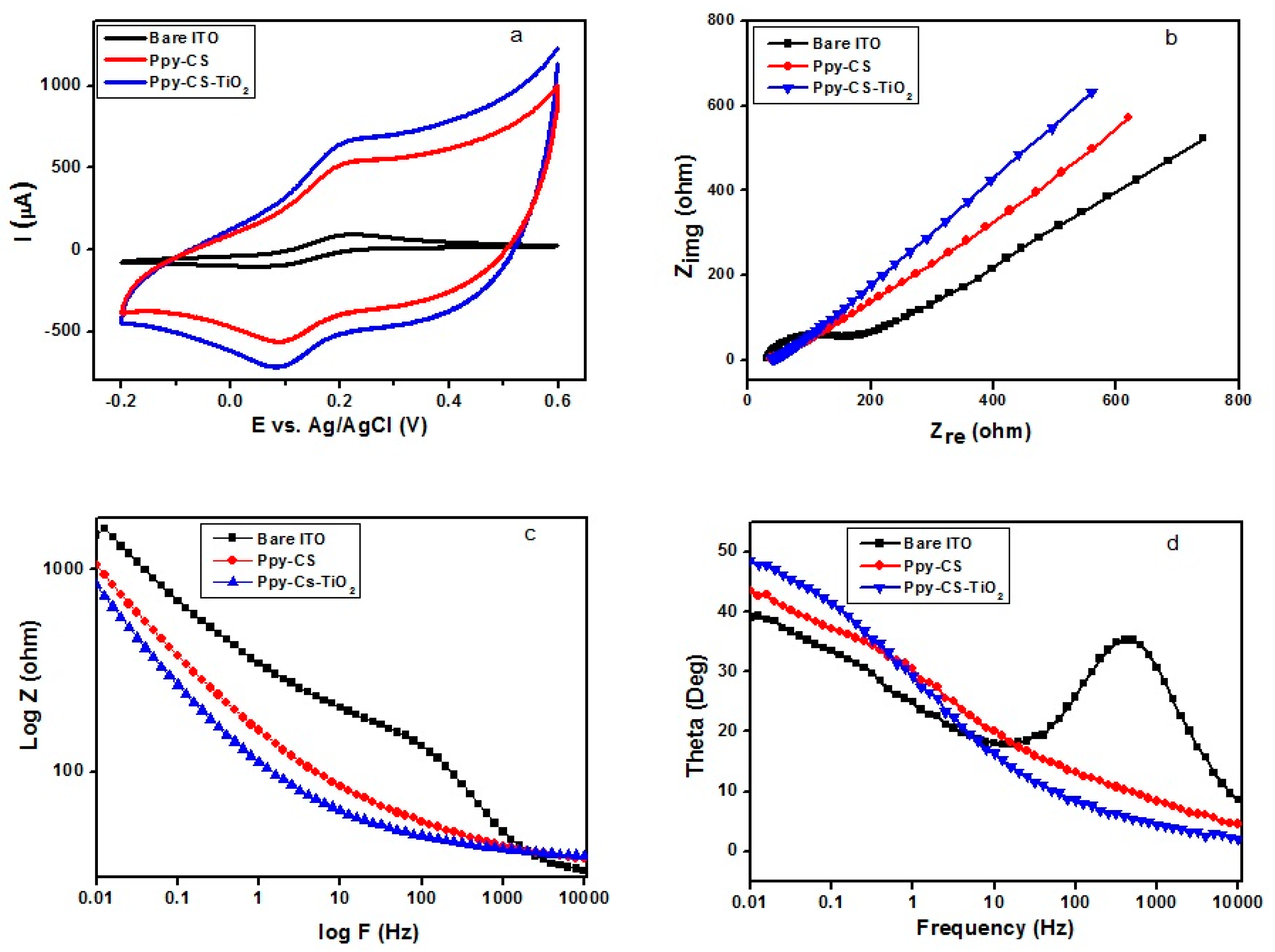
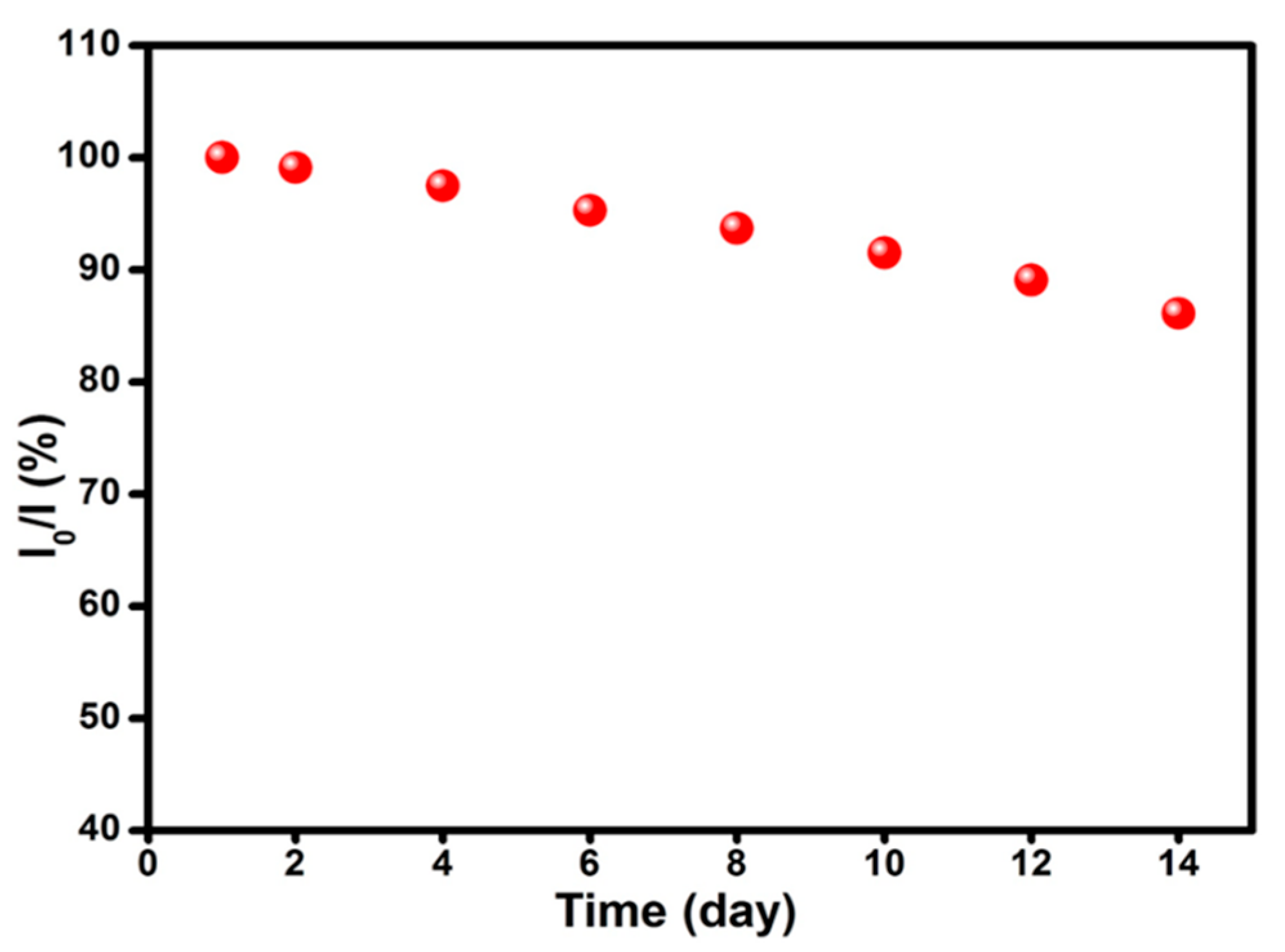
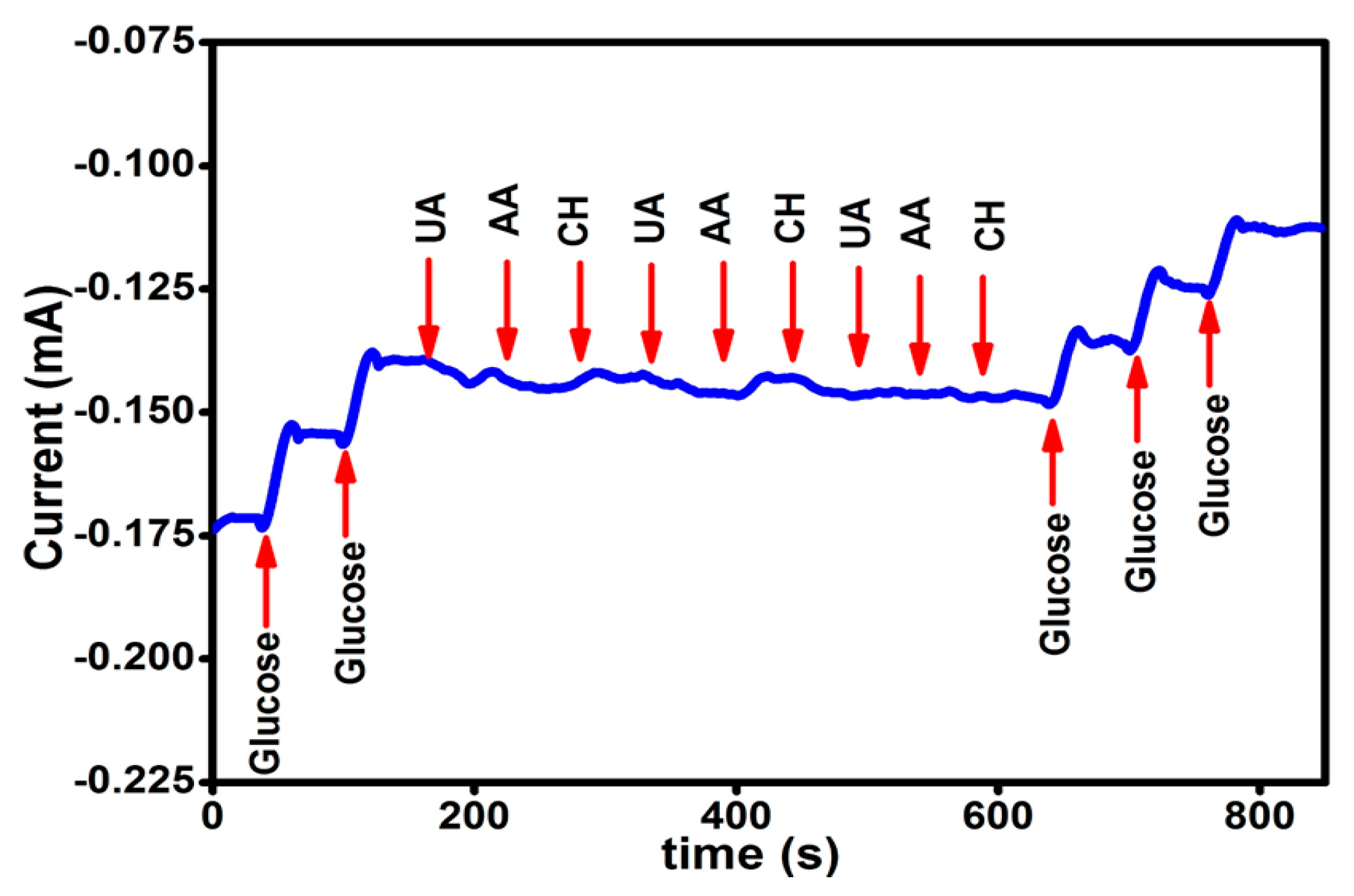
2. Results and Discussion
3. Materials and Methods
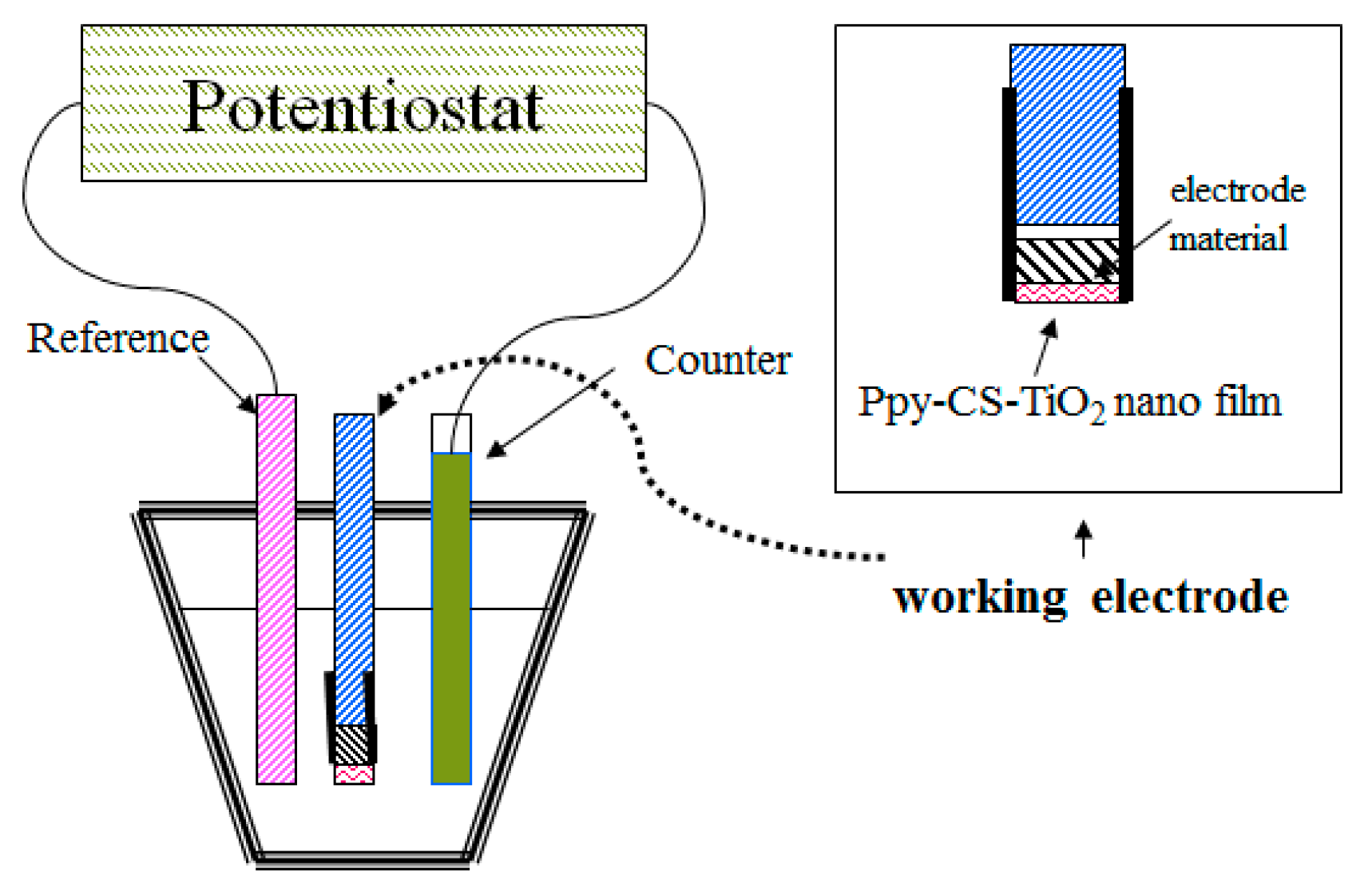
3.1. Instrument
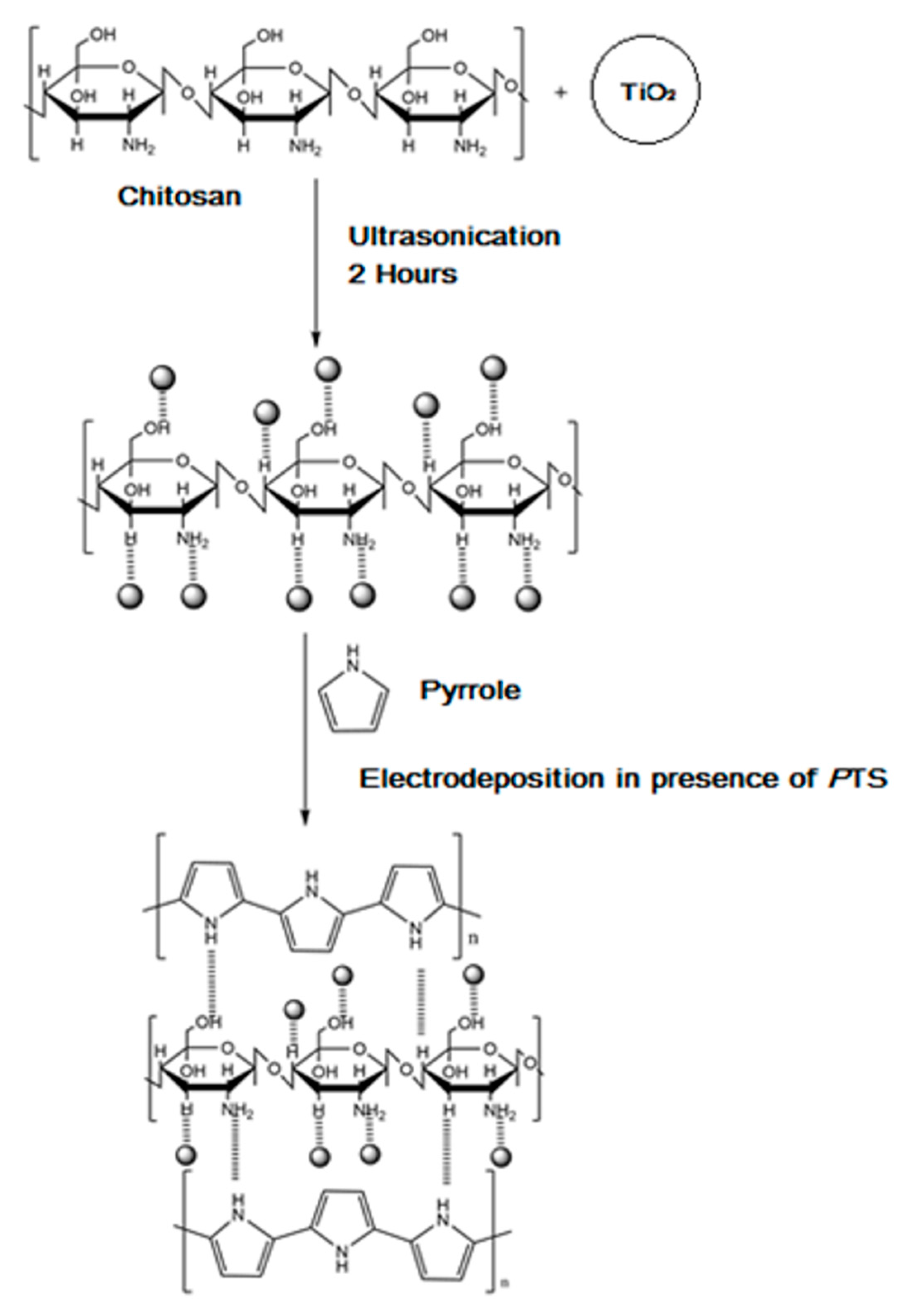
3.2. Preparation of Ppy-CS-TiO2 NP/ITO Nanocomposite Films
3.3. Mechanism for the Formation of Ppy-CS-TiO2 on ITO
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Sanchez, C.; Soler-Illia, G.D.A.; Ribot, F.; Lalot, T.; Mayer, C.R.; Cabuil, V. Designed hybrid organic–inorganic nanocomposites from functional nanobuilding blocks. Chem. Mater. 2001, 13, 3061–3083. [Google Scholar] [CrossRef]
- Mitzi, D.B. Thin-film deposition of organic–inorganic hybrid materials. Chem. Mater. 2001, 13, 3283–3298. [Google Scholar] [CrossRef]
- Gómez-Romero, P.; Chojak, M.; Cuentas-Gallegos, K.; Asensio, J.A.; Kulesza, P.J.; Casañ-Pastor, N.; Lira-Cantú, M. Hybrid organic–Inorganic nanocomposite materials for application in solid state electrochemical supercapacitors. Electrochem. Commun. 2003, 5, 149–153. [Google Scholar] [CrossRef]
- Coronado, E.; Galán-Mascarós, J.R.; Gómez-García, C.J.; Laukhin, V. Coexistence of ferromagnetism and metallic conductivity in a molecule-based layered compound. Nature 2000, 408, 447–449. [Google Scholar] [CrossRef] [PubMed]
- Sidorov, S.N.; Volkov, I.V.; Davankov, V.A.; Tsyurupa, M.P.; Valetsky, P.M.; Bronstein, L.M.; Lakina, N.V. Platinum-containing hyper-cross-linked polystyrene as a modifier-free selective catalyst for l-sorbose oxidation. J. Am. Chem. Soc. 2001, 123, 10502–10510. [Google Scholar] [CrossRef] [PubMed]
- Merkel, T.C.; Freeman, B.D.; Spontak, R.J.; He, Z.; Pinnau, I.; Meakin, P.; Hill, A. JUltrapermeable, reverse-selective nanocomposite membranes. Science 2002, 296, 519–522. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Wang, J.; Kang, X.; Wang, C.; Wang, D.; Liu, J.; Lin, Y. Glucose biosensor based on immobilization of glucose oxidase in platinum nanoparticles/graphene/chitosan nanocomposite film. Talanta 2009, 80, 403–406. [Google Scholar] [CrossRef] [PubMed]
- Tsai, Y.C.; Li, S.C.; Liao, S.W. Electrodeposition of polypyrrole–multiwalled carbon nanotube—Glucose oxidase nanobiocomposite film for the detection of glucose. Biosens. Bioelectron. 2006, 22, 495–500. [Google Scholar] [CrossRef] [PubMed]
- Butterworth, M.D.; Corradi, R.; Johal, J.; Lascelles, S.F.; Maeda, S.; Armes, S.P. Zeta potential measurements on conducting polymer–inorganic oxide nanocomposite particles. J. Colloid Interface Sci. 1995, 174, 510–517. [Google Scholar] [CrossRef]
- Hajji, P.; David, L.; Gerard, J.F.; Pascault, J.P.; Vigier, G. Synthesis, structure, and morphology of polymer—Silica hybrid nanocomposites based on hydroxyethyl methacrylate. J. Polym. Sci. Part B-Polym. Phys. 1999, 37, 3172–3187. [Google Scholar] [CrossRef]
- Yoon, H. Current trends in sensors based on conducting polymer nanomaterials. Nanomaterials 2013, 3, 524–549. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Ma, J.; Zhang, S.; Wang, W.; Chen, Z. A highly sensitive nonenzymatic glucose sensor based on CuO nanoparticles decorated carbon spheres. Sens. Actuator B Chem. 2015, 211, 385–391. [Google Scholar] [CrossRef]
- Bao, S.J.; Li, C.M.; Zang, J.F.; Cui, X.Q.; Qiao, Y.; Guo, J. New nanostructured TiO2 for direct electrochemistry and glucose sensor applications. Adv. Funct. Mater. 2008, 18, 591–599. [Google Scholar] [CrossRef]
- Wei, H.; Wang, E. Fe3O4 magnetic nanoparticles as peroxidase mimetics and their applications in H2O2 and glucose detection. Anal. Chem. 2008, 80, 2250–2254. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Gao, X.; Jin, L.; Wu, Q.; Chen, Z.; Lin, X. Amperometric glucose biosensor based on silver nanowires and glucose oxidase. Sens. Actuator B Chem. 2013, 176, 9–14. [Google Scholar] [CrossRef]
- Ramanavicius, A.; Kausaite, A.; Ramanaviciene, A.; Acaite, J.; Malinauskas, A. Redox enzyme–glucose oxidase–initiated synthesis of polypyrrole. Synth. Met. 2006, 156, 409–413. [Google Scholar] [CrossRef]
- Welch, C.M.; Compton, R.G. The use of nanoparticles in electroanalysis: A review. Anal. Bioanal. Chem. 2006, 384, 601–619. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Dong, Z.; Li, Y.; Li, J.; Wang, J.; Yang, H.; Li, R. High performance non-enzymatic glucose biosensor based on copper nanowires–carbon nanotubes hybrid for intracellular glucose study. Sens. Actuator B Chem. 2013, 182, 618–624. [Google Scholar] [CrossRef]
- Xiao, X.; Li, H.; Pan, Y.; Si, P. Non-enzymatic glucose sensors based on controllable nanporous gold/copper oxide nanohybrids. Talanta 2014, 125, 366–371. [Google Scholar] [CrossRef] [PubMed]
- Zaidi, S.A.; Shin, J.H. Recent developments in nanostructure based electrochemical glucose sensors. Talanta 2016, 149, 30–42. [Google Scholar] [CrossRef] [PubMed]
- Guan, P.; Li, Y.; Zhang, J.; Li, W. Non-Enzymatic Glucose Biosensor Based on CuO-Decorated CeO2 Nanoparticles. Nanomaterials 2016, 6, 159. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.; Long, M.; Tan, L.; Zhang, Y.; Ouyang, J.; Liu, P.; Tang, A. Helical TiO2 nanotube arrays modified by Cu–Cu2O with ultrahigh sensitivity for the nonenzymatic electro-oxidation of glucose. ACS Appl. Mater. Interfaces 2015, 7, 12719–12730. [Google Scholar] [CrossRef] [PubMed]
- He, M.Q.; Bao, L.L.; Sun, K.Y.; Zhao, D.X.; Li, W.B.; Xia, J.X.; Li, H.M. Synthesis of molecularly imprinted polypyrrole/titanium dioxide nanocomposites and its selective photocatalytic degradation of rhodamine B under visible light irradiation. Express Polym. Lett. 2014, 8, 850–861. [Google Scholar] [CrossRef]
- Si, P.; Ding, S.; Yuan, J.; Lou, X.W.; Kim, D.H. Hierarchically structured one-dimensional TiO2 for protein immobilization, direct electrochemistry, and mediator-free glucose sensing. ACS Nano 2011, 5, 7617–7626. [Google Scholar] [CrossRef] [PubMed]
- Bai, J.; Zhou, B. Titanium dioxide nanomaterials for sensor applications. Chem. Rev. 2014, 114, 10131–10176. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Xu, L.; Xing, R.; Song, J.; Song, H.; Liu, D.; Zhou, J. Electrospun three-dimensional porous CuO/TiO2 hierarchical nanocomposites electrode for nonenzymatic glucose biosensing. Electrochem. Commun. 2012, 20, 75–78. [Google Scholar] [CrossRef]
- Lawrie, G.; Keen, I.; Drew, B.; Chandler-Temple, A.; Rintoul, L.; Fredericks, P.; Grøndahl, L. Interactions between alginate and chitosan biopolymers characterized using FTIR and XPS. Biomacromolecules 2007, 8, 2533–2541. [Google Scholar] [CrossRef] [PubMed]
- Kang, E.T.; Neoh, K.G.; Tan, K.L. X-ray photoelectron spectroscopic studies of electroactive polymers. In Polymer Characteristics; Springer: Berlin/Heidelberg, Germany, 1993; Volume 106, pp. 135–190. [Google Scholar]
- Wagner, C.D.; Naumkin, A.V.; Kraut-Vass, A.; Allison, J.W.; Powell, C.J.; Rumble, J.R., Jr. NIST X-ray Photoelectron Spectroscopy Database. Available online: http:/srdata.nist.gov/xps/ (accessed on 20 February 2017).
- Jensen, H.; Soloviev, A.; Li, Z.; Søgaard, E.G. XPS and FTIR investigation of the surface properties of different prepared titania nano-powders. Appl. Surf. Sci. 2005, 246, 239–249. [Google Scholar] [CrossRef]
- Sathe, A.; Peck, M.A.; Balasanthiran, C.; Langell, M.A.; Rioux, R.M.; Hoefelmeyer, J.D. X-ray photoelectron spectroscopy of transition metal ions attached to the surface of rod-shape anatase TiO2 nanocrystals. Inorg. Chim. Acta 2014, 422, 8–13. [Google Scholar] [CrossRef]
- Di Valentin, C.; Finazzi, E.; Pacchioni, G.; Selloni, A.; Livraghi, S.; Paganini, M.C.; Giamello, E. N-Doped TiO2: Theory and Experiment. Met. Oxides 2007, 339, 44–56. [Google Scholar] [CrossRef]
- Colón, G.; Maicu, M.; Hidalgo, M.C.; Navío, J.A. Cu-Doped TiO2 Systems with Improved Photocatalytic Activity. Appl. Catal. B Environ. 2006, 67, 41–51. [Google Scholar] [CrossRef]
- Morita, K.; Shibuya, T.; Yasuoka, K. Stability of Excess Electrons Introduced by Ti Interstitial in Rutile TiO2(110) Surface. J. Phys. Chem. C 2017, 121, 1602–1607. [Google Scholar] [CrossRef]
- Toghill, K.E.; Compton, R.G. Electrochemical non-enzymatic glucose sensors: A perspective and an evaluation. Int. J. Electrochem. Sci. 2010, 5, 1246–1301. [Google Scholar]
- Si, P.; Huang, Y.; Wang, T.; Ma, J. Nanomaterials for electrochemical non-enzymatic glucose biosensors. RSC Adv. 2013, 3, 3487–3502. [Google Scholar] [CrossRef]
- Park, S.; Boo, H.; Chung, T.D. Electrochemical non-enzymatic glucose sensors. Anal. Chim. Acta 2006, 556, 46–57. [Google Scholar] [CrossRef] [PubMed]
- Surareungchai, W.D.; Tasakorn, P. Quadruple-pulsed amperometric detection for simultaneous flow injection determination of glucose and fructose. Anal. Chim. Acta 2001, 448, 215–220. [Google Scholar] [CrossRef]
- Kurniawan, F.; Tsakova, V.; Mirsky, V.M. Gold Nanoparticles in nonenzymatic electrochemical detection of sugars. Electroanalysis 2006, 18, 1937–1942. [Google Scholar] [CrossRef]
- Feng, D.; Wang, F.; Chen, Z.L. Electrochemical glucose sensor based on one-step construction of gold nanoparticle chitosan composite film. Sens. Actuators B 2009, 138, 539–544. [Google Scholar] [CrossRef]
- Li, C.; Su, Y.; Lv, X.; Xia, H.; Shi, H.; Yang, X.; Zhang, J.; Wang, Y. Controllable anchoring of gold nanoparticles to polypyrrole nanofibers by hydrogen bonding and their application in nonenzymatic glucose sensors. Biosens. Bioelectron. 2012, 38, 402–406. [Google Scholar] [CrossRef] [PubMed]
- AL-Mokaram, A.M.A.A.; Yahya, R.; Abdi, M.M.; Mahmud, H.N.M.E. One-step electrochemical deposition of Polypyrrole–Chitosan–Iron oxide nanocomposite films for non-enzymatic glucose biosensor. Mater. Lett. 2016, 183, 90–93. [Google Scholar] [CrossRef]
- Romero-Arcos, M.; Garnica-Romo, M.G.; Martínez-Flores, H.E. Electrochemical Study and Characterization of an Amperometric Biosensor Based on the Immobilization of Laccase in a Nanostructure of TiO2 Synthesized by the Sol–Gel Method. Materials 2016, 9, 543. [Google Scholar] [CrossRef]


| Electrode Material | Technique Methods | Electrolyte | Linear Range (mM) | Detection Limit (μM) | Reference |
|---|---|---|---|---|---|
| Au/Nafion | Amperometry | 0.1 M NaOH | 5.0–60 | 1200 | [38] |
| Layer-by-layer Au NPs/Au E | Nil | 0.1 M NaOH | Up to 8 | 500 | [39] |
| Au NPs/chitosan/GCE | Nil | PBS | 0.4–10.7 | 370 | [40] |
| GCE/GNPs/PpyNFs | Amperometry | 0.1 M NaOH | 0.2–13 | - | [41] |
| Ppy-CS-Fe3O4NP/ITO | Amperometry | 0.1 M NaOH | 1–16 | 234 | [42] |
| TiO2/LAC | Amperometry | 0.1 M NaOH | 3.75–150 | 3.75 | [43] |
| Ppy-CS-TiO2NP/ITO | Amperometry | 0.1 M NaOH | 1–14 | 614 | This work |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
AL-Mokaram, A.M.A.A.A.; Yahya, R.; Abdi, M.M.; Mahmud, H.N.M.E. The Development of Non-Enzymatic Glucose Biosensors Based on Electrochemically Prepared Polypyrrole–Chitosan–Titanium Dioxide Nanocomposite Films. Nanomaterials 2017, 7, 129. https://doi.org/10.3390/nano7060129
AL-Mokaram AMAAA, Yahya R, Abdi MM, Mahmud HNME. The Development of Non-Enzymatic Glucose Biosensors Based on Electrochemically Prepared Polypyrrole–Chitosan–Titanium Dioxide Nanocomposite Films. Nanomaterials. 2017; 7(6):129. https://doi.org/10.3390/nano7060129
Chicago/Turabian StyleAL-Mokaram, Ali M. A. Abdul Amir, Rosiyah Yahya, Mahnaz M. Abdi, and Habibun Nabi Muhammad Ekramul Mahmud. 2017. "The Development of Non-Enzymatic Glucose Biosensors Based on Electrochemically Prepared Polypyrrole–Chitosan–Titanium Dioxide Nanocomposite Films" Nanomaterials 7, no. 6: 129. https://doi.org/10.3390/nano7060129
APA StyleAL-Mokaram, A. M. A. A. A., Yahya, R., Abdi, M. M., & Mahmud, H. N. M. E. (2017). The Development of Non-Enzymatic Glucose Biosensors Based on Electrochemically Prepared Polypyrrole–Chitosan–Titanium Dioxide Nanocomposite Films. Nanomaterials, 7(6), 129. https://doi.org/10.3390/nano7060129








