Nanometric MIL-125-NH2 Metal–Organic Framework as a Potential Nerve Agent Antidote Carrier
Abstract
1. Introduction
2. Results and Discussion
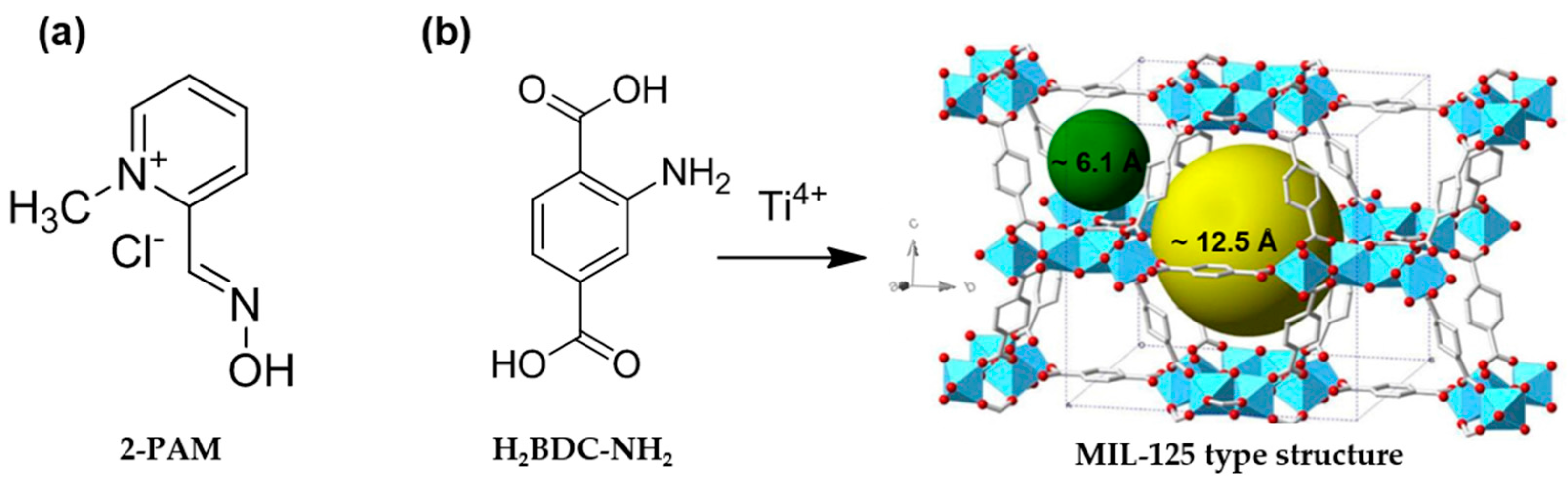
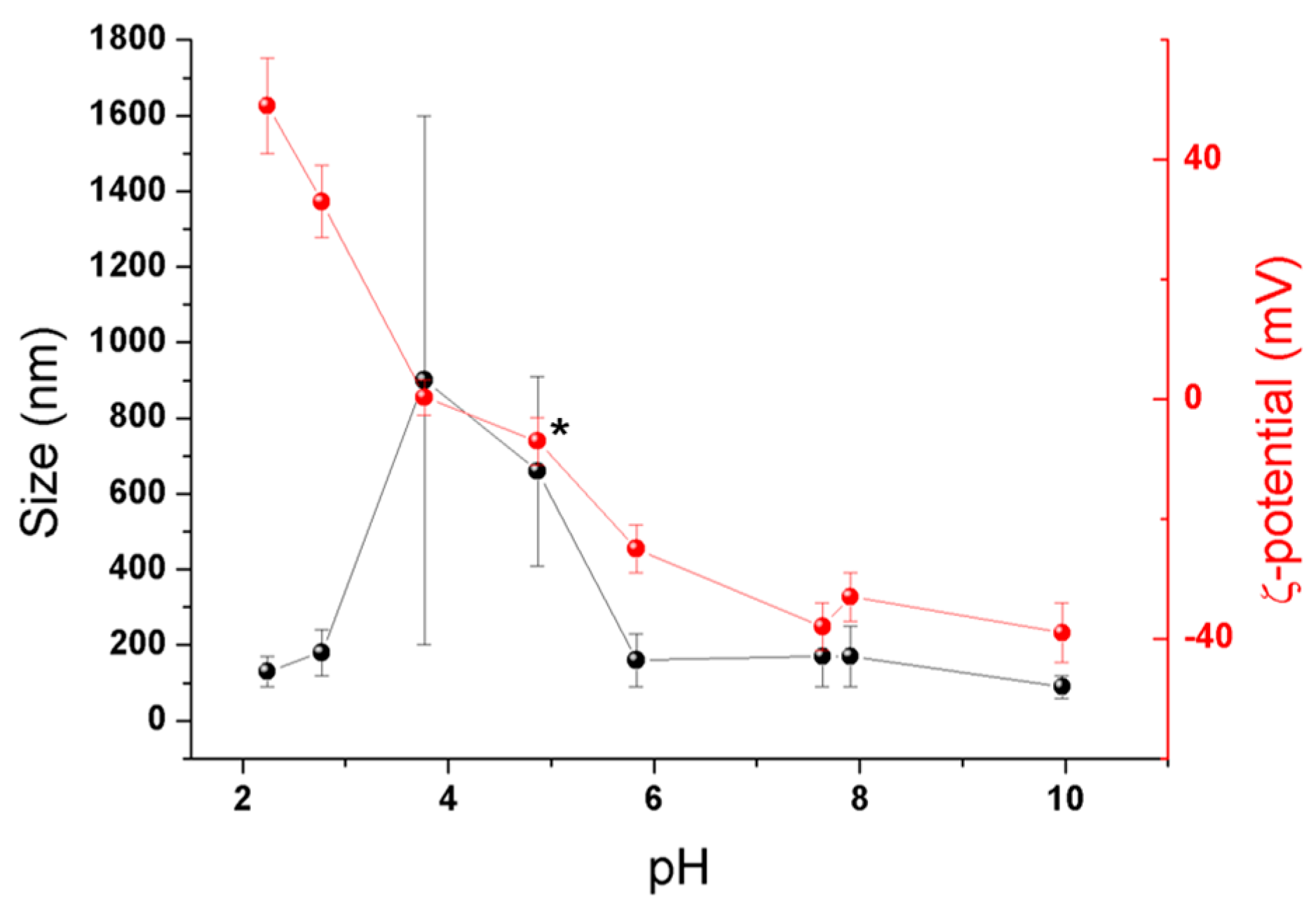
2.1. Synthesis and Colloidal Stability of MIL-125-NH2 Nanoparticles
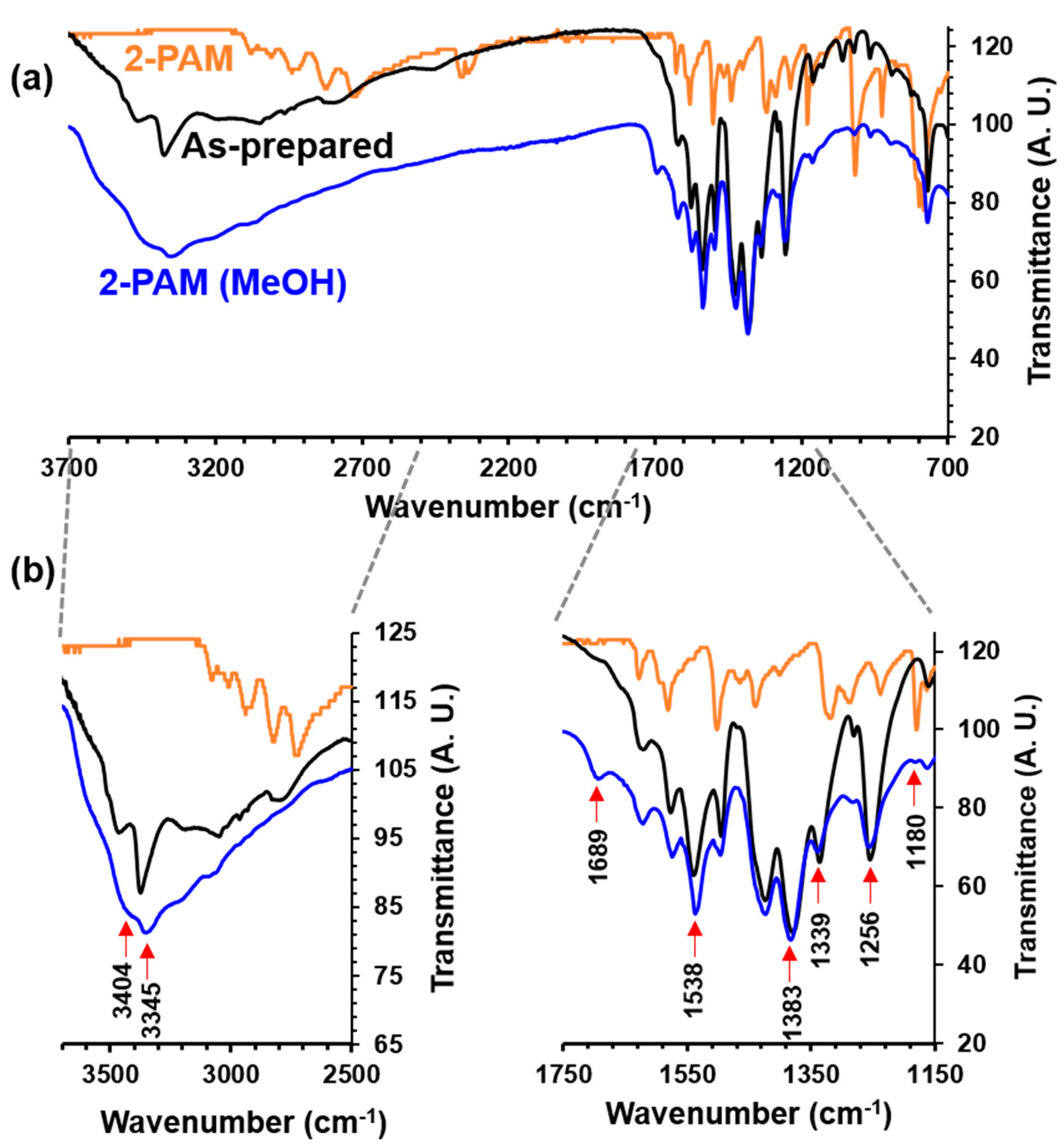
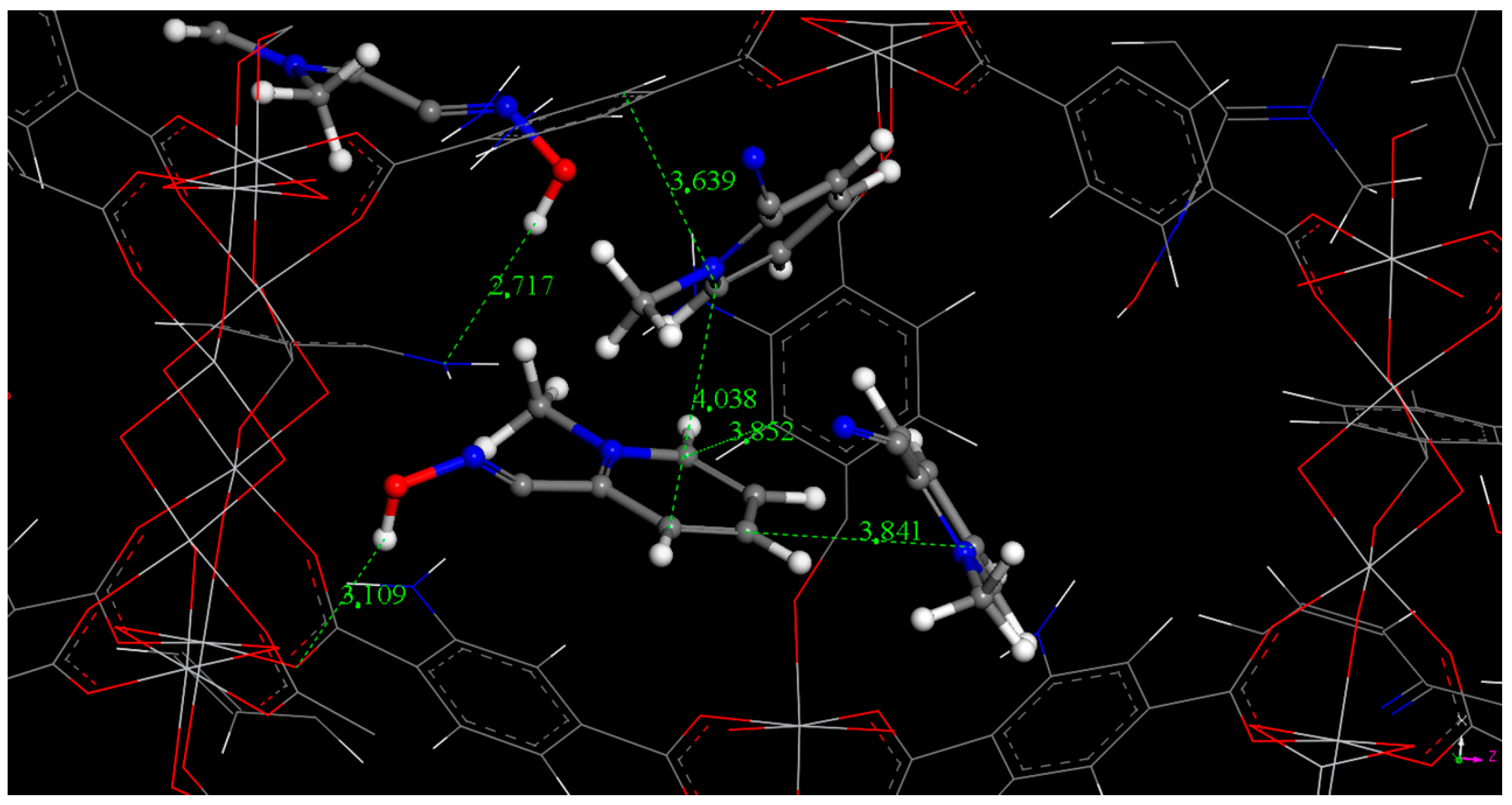
2.2. Encapsulation of 2-PAM
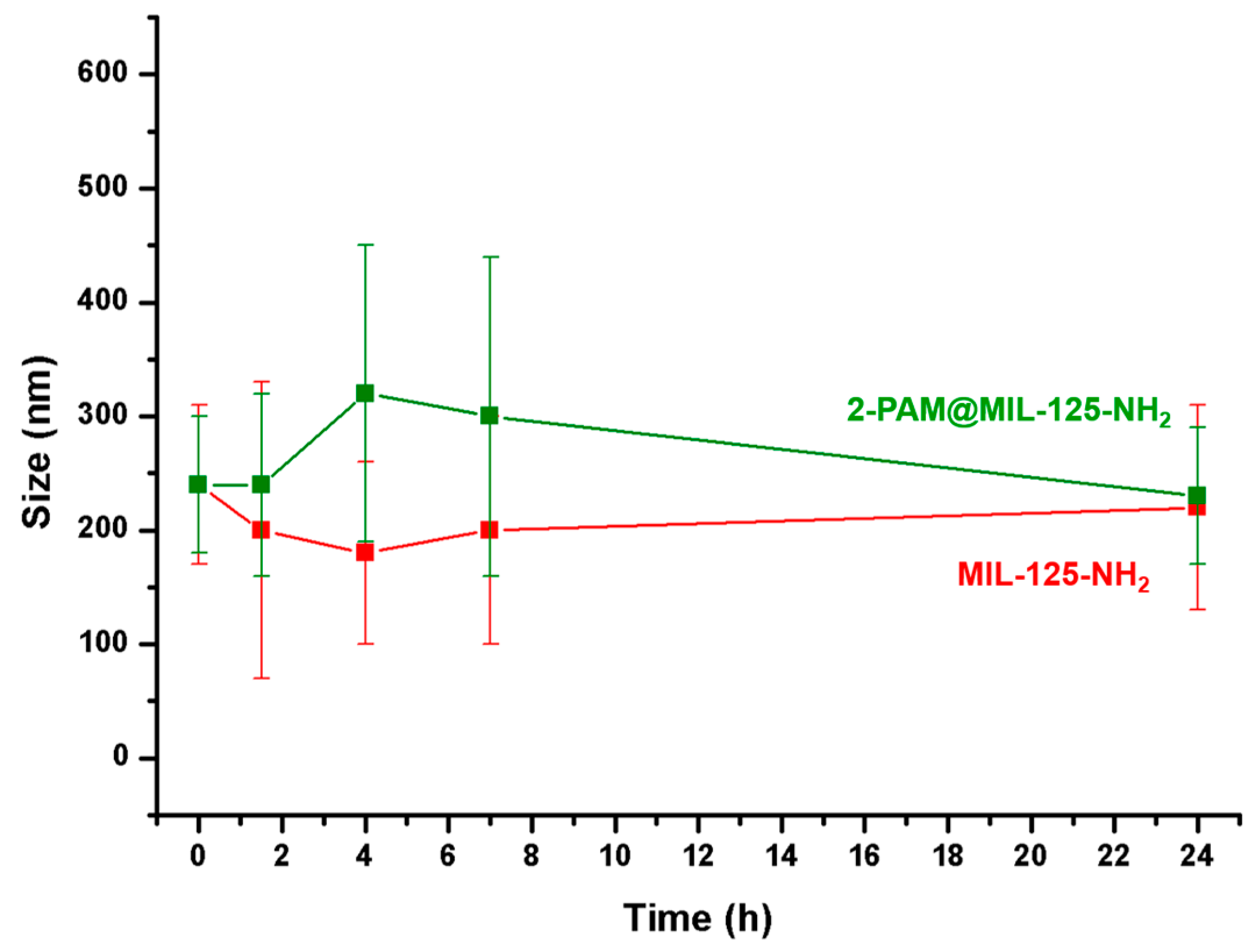
2.3. Colloidal Stability of 2-PAM@MIL-125-NH2
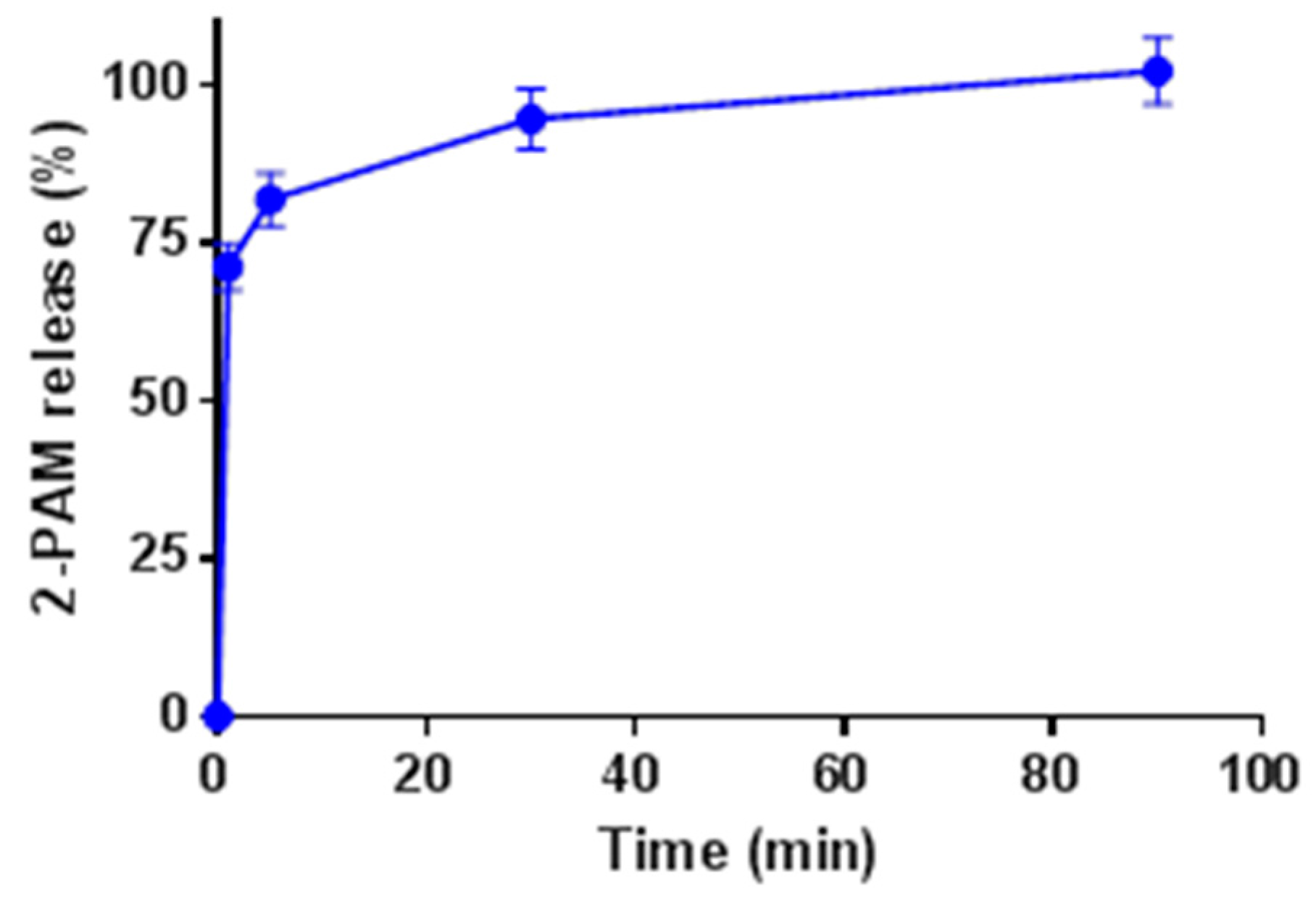
2.4. Release Kinetics of 2-PAM
3. Materials and Methods
3.1. General Instrumentation
3.2. Reagents and Solvents
3.3. Synthesis of MIL-125-NH2 Nanoparticles
3.4. Encapsulation of 2-PAM into the Nano-Sized MIL-125-NH2
3.5. Computational Simulation of 2-PAM Encapsulation
3.6. Colloidal Stability of MIL-125-NH2 nanoparticles and 2-PAM@MIL-125-NH2
3.7. 2-PAM Release Kinetics
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Marrs, T.C. Organophosphate Poisoning. Pharm. Ther. 1993, 58, 51–66. [Google Scholar] [CrossRef]
- Quinn, D.M. Acetylcholinesterase: Enzyme Structure, Reaction Dynamics, and Virtual Transition States. Chem. Rev. 1987, 87, 955–979. [Google Scholar] [CrossRef]
- Worek, F.; Eyer, P.; Aurbek, N.; Szinicz, L.; Thiermann, H. Recent advances in evaluation of oxime efficacy in nerve agent poisoning by in vitro analysis. Toxicol. Appl. Pharm. 2007, 219, 226–234. [Google Scholar] [CrossRef] [PubMed]
- Sakurada, K.; Matsubara, K.; Shimizu, K.; Shiono, H.; Seto, Y.; Tsuge, K.; Yoshino, M.; Sakai, I.; Mukoyama, H.; Takatori, T. Pralidoxime Iodide (2-PAM ) Penetrates across the Blood-Brain Barrier. Neurochem. Res. 2003, 28, 1401–1407. [Google Scholar] [CrossRef] [PubMed]
- Cai, W.; Chu, C.; Liu, G.; Wáng, Y.J. Metal–Organic Framework-Based Nanomedicine Platforms for Drug Delivery and Molecular Imaging. Small 2015, 11, 4806–4822. [Google Scholar] [CrossRef] [PubMed]
- Zhuang, J.; Kuo, C.-H.; Chou, L.-Y.; Liu, D.-Y.; Weerapana, E.; Tsung, C.-K. Optimized Metal-Organic Framework Nanospheres for Drug Delivery: Evaluation of Small-Molecule Encapsulation. ACS Nano 2014, 8, 2812–2819. [Google Scholar] [CrossRef] [PubMed]
- Rahoui, N.; Jiang, B.; Taloub, N.; Huang, Y.D. Spatio-temporal control strategy of drug delivery systems based nano structures. J. Control. Release 2017, 255, 176–201. [Google Scholar] [CrossRef] [PubMed]
- Porcaro, F.; Carlini, L.; Ugolini, A.; Visaggio, D.; Visca, P.; Fratoddi, I.; Venditti, I.; Meneghini, C.; Simonelli, L.; Marini, C.; et al. Synthesis and Structural Characterization of Silver Nanoparticles Stabilized with 3-Mercapto-1-Propansulfonate and 1-Thioglucose Mixed Thiols for Antibacterial Applications. Materials 2016, 9, 1028. [Google Scholar] [CrossRef] [PubMed]
- Venditti, I.; Hassanein, T.F.; Fratoddi, I.; Fontana, L.; Battocchio, C.; Rinaldi, F.; Carafa, M.; Marianecci, C.; Diociaiuti, M.; Agostinelli, E.; et al. Bioconjugation of gold-polymer core–shell nanoparticles with bovine serum amine oxidase for biomedical applications. Colloids Surf. B 2015, 134, 314–321. [Google Scholar] [CrossRef] [PubMed]
- Dhand, C.; Prabhakaran, M.P.; Beuerman, R.W.; Lakshminarayanan, R.; Dwivedi, N.; Ramakrishna, S. Role of size of drug delivery carriers for pulmonary and intravenous administration with emphasis on cancer therapeutics and lug-targeted drug delivery. RSC Adv. 2014, 4, 32673–32689. [Google Scholar] [CrossRef]
- Venditti, I.; Cartoni, A.; Fontana, L.; Testa, G.; Scaramuzzo, F.A.; Faccini, R.; Terracciano, C.M.; Camillocci, E.S.; Morganti, S.; Giordano, A.; et al. Y3+ embedded in polymeric nanoparticles: Morphology, dimension and stability of composite colloidal system. Colloids Surf. A 2017, 532, 125–131. [Google Scholar] [CrossRef]
- Pujari-palmer, S.; Lu, X.; Ott, M.K. The Influence of Hydroxyapatite Nanoparticle Morphology on Embryonic Development in a Zebrafish Exposure Model. Nanomaterials 2017, 7, 89. [Google Scholar] [CrossRef] [PubMed]
- Lanone, S.; Rogerieux, F.; Geys, J.; Dupont, A.; Maillot-Marechal, E.; Boczkowski, J.; Lacroix, G.; Hoet, P. Comparative toxicity of 24 manufactured nanoparticles in human alveolar epithelial and macrophage cell lines. Part. Fibre Toxicol. 2009, 6, 14. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Chen, J.; Wei, K.; Chen, J.; Huang, C. Release of Doxorubicin by a Folate-Grafted, Chitosan-Coated Magnetic Nanoparticle. Nanomaterials 2017, 7, 85. [Google Scholar] [CrossRef] [PubMed]
- Dadparvar, M.; Wagner, S.; Wien, S.; Worek, F.; von Briesen, H.; Kreuter, J. Freeze-drying of HI-6-loaded recombinant human serum albumin nanoparticles for improved storage stability. Eur. J. Pharm. Biopharm. 2014, 88, 510–517. [Google Scholar] [CrossRef] [PubMed]
- Orbesteanu, A.M.; Cojocaru, V.; Ailiesei, I.; Mircioiu, C.; Cinteza, L.O. Studies on the formulation of nanostructured carriers for increasing the bioavailability of pralidoxime chloride. Stud. Univ. Vasile Goldis Arad Ser. Stiint. Vietii 2014, 24, 357–361. [Google Scholar]
- Chigumira, W.; Maposa, P.; Gadaga, L.L.; Dube, A.; Tagwireyi, D.; Maponga, C.C. Preparation and Evaluation of Pralidoxime-Loaded PLGA Nanoparticles as Potential Carriers of the Drug across the Blood Brain Barrier. J. Nanopart. 2015, 2015. [Google Scholar] [CrossRef]
- Pashirova, T.N.; Zueva, I.V.; Petrov, K.A.; Babaev, V.M.; Lukashenko, S.S.; Rizvanov, I.K.; Souto, E.B.; Nikolsky, E.E.; Zakharova, L.Y.; Masson, P.; et al. Nanoparticle-Delivered 2-PAM for Rat Brain Protection against Paraoxon Central Toxicity. ACS Appl. Mater. Interfaces 2017, 9, 16922–16932. [Google Scholar] [CrossRef] [PubMed]
- Joosen, M.J.A.; van der Schans, M.J.; van Helden, H.P.M. Percutaneous exposure to the nerve agent VX: Efficacy of combined atropine, obidoxime and diazepam treatment. Chem-Biol. Interact. 2010, 188, 255–263. [Google Scholar] [CrossRef] [PubMed]
- Kitagawa, S. Metal-organic frameworks. Chem. Soc. Rev. 2014, 43, 5415–5418. [Google Scholar]
- Farha, O.K.; Eryazici, I.; Jeong, N.C.; Hauser, B.G.; Wilmer, C.E.; Sarjeant, A.A.; Snurr, R.Q.; Nguyen, S.T.; Yazaydin, A.O.; Hupp, J.T. Metal–Organic Framework Materials with Ultrahigh Surface Areas: Is the Sky the Limit? J. Am. Chem. Soc. 2012, 134, 15016–15021. [Google Scholar] [CrossRef] [PubMed]
- Furukawa, H.; Cordova, K.E.; O’Keeffe, M.; Yaghi, O.M. The Chemistry and Applications of Metal-Organic Frameworks. Science 2013, 341, 974–986. [Google Scholar] [CrossRef] [PubMed]
- Hidalgo, T.; Serre, C.; Horcajada, P. Nanostructured metal–organic frameworks and their bio-related applications. Coord. Chem. Rev. 2016, 307, 342–360. [Google Scholar]
- Horcajada, P.; Gref, R.; Baati, T.; Allan, P.K.; Maurin, G.; Couvreur, P.; Férey, G.; Morris, R.E.; Serre, C. Metal-Organic Frameworks in Biomedicine. Chem. Rev. 2012, 112, 1232–1268. [Google Scholar] [CrossRef] [PubMed]
- Mondloch, J.E.; Katz, M.J.; Iii, W.C.I.; Ghosh, P.; Liao, P.; Bury, W.; Wagner, G.W.; Hall, M.G.; Decoste, J.B.; Peterson, G.W.; et al. Destruction of chemical warfare agents using metal–organic frameworks. Nat. Mater. 2015, 14, 512–516. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Moon, S.-Y.; Guelta, M.A.; Lin, L.; Gómez-Gualdrón, D.A.; Snurr, R.Q.; Harvey, S.P.; Hupp, J.T.; Farha, O.K. Nanosizing a Metal–Organic Framework Enzyme Carrier for Accelerating Nerve Agent Hydrolysis. ACS Nano 2016, 10, 9174–9182. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Moon, S.Y.; Guelta, M.A.; Harvey, S.P.; Hupp, J.T.; Farha, O.K. Encapsulation of a Nerve Agent Detoxifying Enzyme by a Mesoporous Zirconium Metal-Organic Framework Engenders Thermal and Long-Term Stability. J. Am. Chem. Soc. 2016, 138, 8052–8055. [Google Scholar] [CrossRef] [PubMed]
- Zlotea, C.; Phanon, D.; Mazaj, M.; Heurtaux, D.; Guillerm, V.; Serre, C.; Horcajada, P.; Devic, T.; Magnier, E.; Cuevas, F.; et al. Effect of NH2 and CF3 functionalization on the hydrogen sorption properties of MOFs. Dalton Trans. 2011, 40, 4879–4881. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Kim, S.-N.; Kim, J.; Ahn, W. Liquid phase adsorption of selected chloroaromatic compounds over metal organic frameworks. Mater. Res. Bull. 2013, 48, 4499–4505. [Google Scholar] [CrossRef]
- Rada, Z.H.; Abid, H.R.; Shang, J.; He, Y.; Webley, P.; Liu, S.; Sun, H.; Wang, S. Effects of amino functionality on uptake of CO2, CH4 and selectivity of CO2/CH4 on titanium based MOFs. Fuel 2015, 160, 318–327. [Google Scholar] [CrossRef]
- Kim, S.-N.; Kim, J.; Kim, H.; Cho, H.; Ahn, W. Adsorption/catalytic properties of MIL-125 and NH2-MIL-125. Catal. Today 2013, 204, 85–93. [Google Scholar] [CrossRef]
- Mounfield, W.P., III; Han, C.; Pang, S.H.; Tumuluri, U.; Jiao, Y.; Bhattacharyya, S.; Dutzer, M.R.; Nair, S.; Wu, Z.; Lively, R.P.; et al. Synergistic Effects of Water and SO2 on Degradation of MIL-125 in the Presence of Acid Gases. J. Phys. Chem. C 2016, 120, 27230–27240. [Google Scholar] [CrossRef]
- The Micro/Nano Fabrication Center. Available online: http://mfc.engr.arizona.edu/safety/MSDSFOLDER/Titanium tetrachlorideMSDS.pdf (accessed on 10 October 2017).
- Acros Organics. Available online: https://www.ch.ntu.edu.tw/~genchem99/msds/exp27/TiO2.pdf (accessed on 10 October 2017).
- Tamames-Tabar, C.; Cunha, D.; Imbuluzqueta, E.; Ragon, F.; Serre, C.; Blanco-Prieto, M.J.; Horcajada, P. Cytotoxicity of nanoscaled metal–organic frameworks. J. Mater. Chem. B 2014, 2, 262–271. [Google Scholar] [CrossRef]
- Wang, W.; Wang, L.; Huang, Y.; Xie, Z.; Jing, X. Nanoscale Metal–Organic Framework–Hemoglobin Conjugates. Chem. Asian J. 2016, 11, 750–756. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.; Sun, D.; Chen, Y.; Huang, R.; Ding, Z.; Fu, X. An Amine-Functionalized Titanium Metal–Organic Framework Photocatalyst with Visible-Light-Induced Activity for CO2 Reduction. Angew. Chem. Int. Ed. 2012, 51, 3364–3367. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Yuan, X.; Wu, Y.; Zeng, G.; Chen, X.; Leng, L.; Wu, Z.; Jiang, L.; Li, H. Facile synthesis of amino-functionalized titanium metal-organic frameworks and their superior visible-light photocatalytic activity for Cr(VI) reduction. J. Hazard. Mater. 2015, 286, 187–194. [Google Scholar] [CrossRef] [PubMed]
- Barea, E.; Montoro, C.; Navarro, J.A.R. Toxic gas removal – metal-organic frameworks for the capture and degradation of toxic gases and vapours. Chem. Soc. Rev. 2014, 43, 5419–5430. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Chen, Y.; Zhang, Y.; Cong, H.; Fu, B.; Wen, S.; Ruan, S. A novel humidity sensor based on NH2-MIL-125(Ti) metal organic framework with high responsiveness. J. Nanopart. Res. 2013, 125, 1–6. [Google Scholar] [CrossRef]
- Su, Z.; Chen, H.; Sun, X.; Huang, Y.; Dong, X. Amine-functionalized metal organic framework (NH2-MIL-125(Ti)) incorporated sodium alginate mixed matrix membranes for dehydration of acetic acid by pervaporation. RSC Adv. 2015, 5, 99008–99017. [Google Scholar] [CrossRef]
- Jin, D.; Gong, A.; Zhou, H. Visible-light-activated photoelectrochemical biosensor for the detection of the pesticide acetochlor in vegetables and fruit based on its inhibition of glucose oxidase. RSC Adv. 2017, 7, 17489–17496. [Google Scholar] [CrossRef]
- Luo, J.; Xu, F.; Tu, J.; Wu, X.; Hou, X. Amine-functionalized titanium metal organic framework for photochemical vapor generation for determination of selenium by inductively coupled plasma optical emission spectrometry. Microchem. J. 2017, 132, 245–250. [Google Scholar] [CrossRef]
- Sohail, M.; Yun, Y.-N.; Lee, E.; Kim, S.K.; Cho, K.; Kim, J.-N.; Kim, T.W.; Moon, J.-H.; Kim, H. Synthesis of Highly Crystalline NH2-MIL-125 (Ti) with S-Shaped Water Isotherms for Adsorption Heat Transformation. Cryst. Growth Des. 2017, 17, 1208–1213. [Google Scholar] [CrossRef]
- Ragon, F.; Horcajada, P.; Chevreau, H.; Hwang, Y.K.; Lee, U.; Miller, S.R.; Devic, T.; Chang, J.-S.; Serre, C. In Situ Energy-Dispersive X-ray Diffraction for the Synthesis Optimization and Scale-up of the Porous Zirconium Terephthalate UiO-66. Inorg. Chem. 2014, 53, 2491–2500. [Google Scholar] [CrossRef] [PubMed]
- Socrates, G. Infrared Characteristic Group Frequencies: Tables and Charts, 2nd ed.; John Wiley Sons: Chichester, UK, 1994. [Google Scholar]
- Hunter, R.J. Zeta Potential in Colloid Science: Principles and Applications; Academic Press: London, UK, 1988. [Google Scholar]
- Peula, J.M.; de las Nieves, F.J. Adsorption of monomeric bovine serum albumin on sulfonated polystyrene model colloids 3. Colloidal stability of latex-protein complexes. Colloids Surf. A Physicochem. Eng. Asp. 1994, 90, 55–62. [Google Scholar] [CrossRef]
- Bellido, E.; Guillevic, M.; Hidalgo, T.; Santander-ortega, M.J.; Serre, C.; Horcajada, P. Understanding the Colloidal Stability of the Mesoporous MIL-100(Fe) Nanoparticles in Physiological Media. Langmuir 2014, 30, 5911–5920. [Google Scholar] [CrossRef] [PubMed]
- Jain, K.K. Nanobiotechnology-based strategies for crossing the blood—Brain barrier Review. Nanomedicine 2012, 7, 1225–1233. [Google Scholar] [CrossRef] [PubMed]
- Simon-Yarza, M.T.; Baati, T.; Paci, A.; Lesueur, L.L.; Seck, A.; Chiper, M.; Gref, R.; Serre, C.; Couvreur, P.; Horcajada, P. Antineoplastic busulfan encapsulated in a metal organic framework nanocarrier: First in vivo. J. Mater. Chem. B 2016, 4, 585–588. [Google Scholar] [CrossRef]
- Rappé, A.K.; Casewit, C.J.; Colwell, K.S.; Goddard, W.A.; Skiff, W.M. UFF, a Full Periodic Table Force Field for Molecular Mechanics and Molecular Dynamics Simulations. J. Am. Chem. Soc. 1992, 114, 10024–10035. [Google Scholar] [CrossRef]
- Rappé, A.K.; Goddard, W.A. Charge Equilibration for Molecular Simulations. J. Phys. Chem. 1991, 95, 3358–3363. [Google Scholar] [CrossRef]
| NPs | Medium | Size (nm) | PdI | ζ-Potential (mV) |
|---|---|---|---|---|
| MIL-125-NH2 | DMF | 240 ± 70 | 0.16 ± 0.01 | * |
| Water | 660 ± 250 | 0.263 ± 0.003 | −7 ± 4 | |
| Methanol | 320 ± 100 | 0.10 ± 0.02 | −40 ± 7 | |
| Ethanol | 220 ± 60 | 0.06 ± 0.03 | −47 ± 19 | |
| PBS | 230 ± 60 | 0.18 ± 0.03 | −23 ± 5 | |
| PBS FBS | 210 ± 100 | 0.220 ± 0.007 | −10 ± 4 | |
| 2-PAM@MIL-125-NH2 | Methanol | 630 ± 120 | 0.5 ± 0.1 | −10 ± 7 |
| PBS | 290 ± 130 | 0.3 ± 0.1 | −18 ± 4 | |
| PBS FBS | 240 ± 40 | 0.4 ± 0.1 | −9 ± 5 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vilela, S.M.F.; Salcedo-Abraira, P.; Colinet, I.; Salles, F.; De Koning, M.C.; Joosen, M.J.A.; Serre, C.; Horcajada, P. Nanometric MIL-125-NH2 Metal–Organic Framework as a Potential Nerve Agent Antidote Carrier. Nanomaterials 2017, 7, 321. https://doi.org/10.3390/nano7100321
Vilela SMF, Salcedo-Abraira P, Colinet I, Salles F, De Koning MC, Joosen MJA, Serre C, Horcajada P. Nanometric MIL-125-NH2 Metal–Organic Framework as a Potential Nerve Agent Antidote Carrier. Nanomaterials. 2017; 7(10):321. https://doi.org/10.3390/nano7100321
Chicago/Turabian StyleVilela, Sérgio M. F., Pablo Salcedo-Abraira, Isabelle Colinet, Fabrice Salles, Martijn C. De Koning, Marloes J. A. Joosen, Christian Serre, and Patricia Horcajada. 2017. "Nanometric MIL-125-NH2 Metal–Organic Framework as a Potential Nerve Agent Antidote Carrier" Nanomaterials 7, no. 10: 321. https://doi.org/10.3390/nano7100321
APA StyleVilela, S. M. F., Salcedo-Abraira, P., Colinet, I., Salles, F., De Koning, M. C., Joosen, M. J. A., Serre, C., & Horcajada, P. (2017). Nanometric MIL-125-NH2 Metal–Organic Framework as a Potential Nerve Agent Antidote Carrier. Nanomaterials, 7(10), 321. https://doi.org/10.3390/nano7100321





