Mechanical Dispersion of Nanoparticles and Its Effect on the Specific Heat Capacity of Impure Binary Nitrate Salt Mixtures
Abstract
:1. Introduction
2. Experimental Section
2.1. Salt Production and Nanoparticles Dispersion
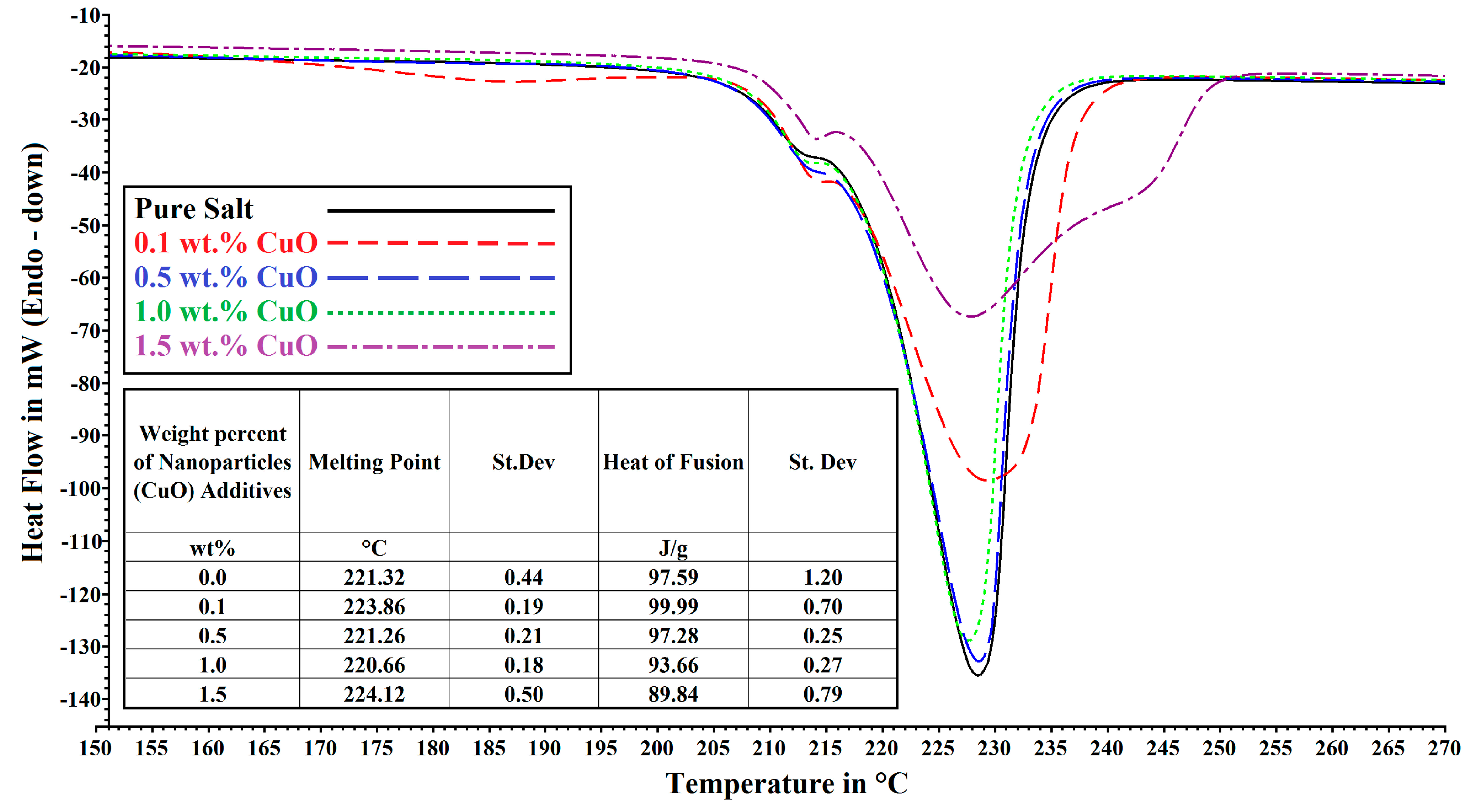
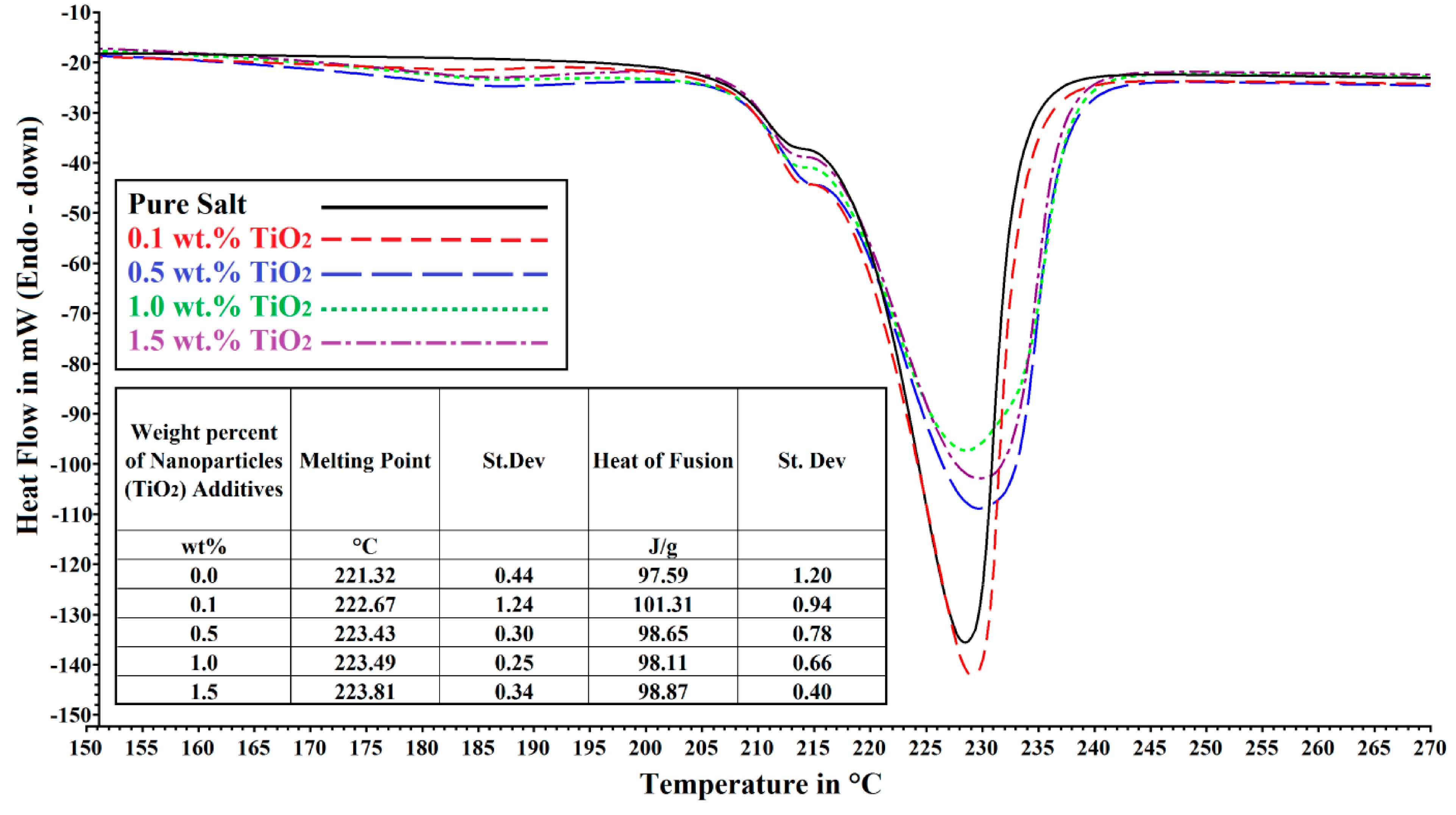
2.2. Differential Scanning Calorimeter (DSC)
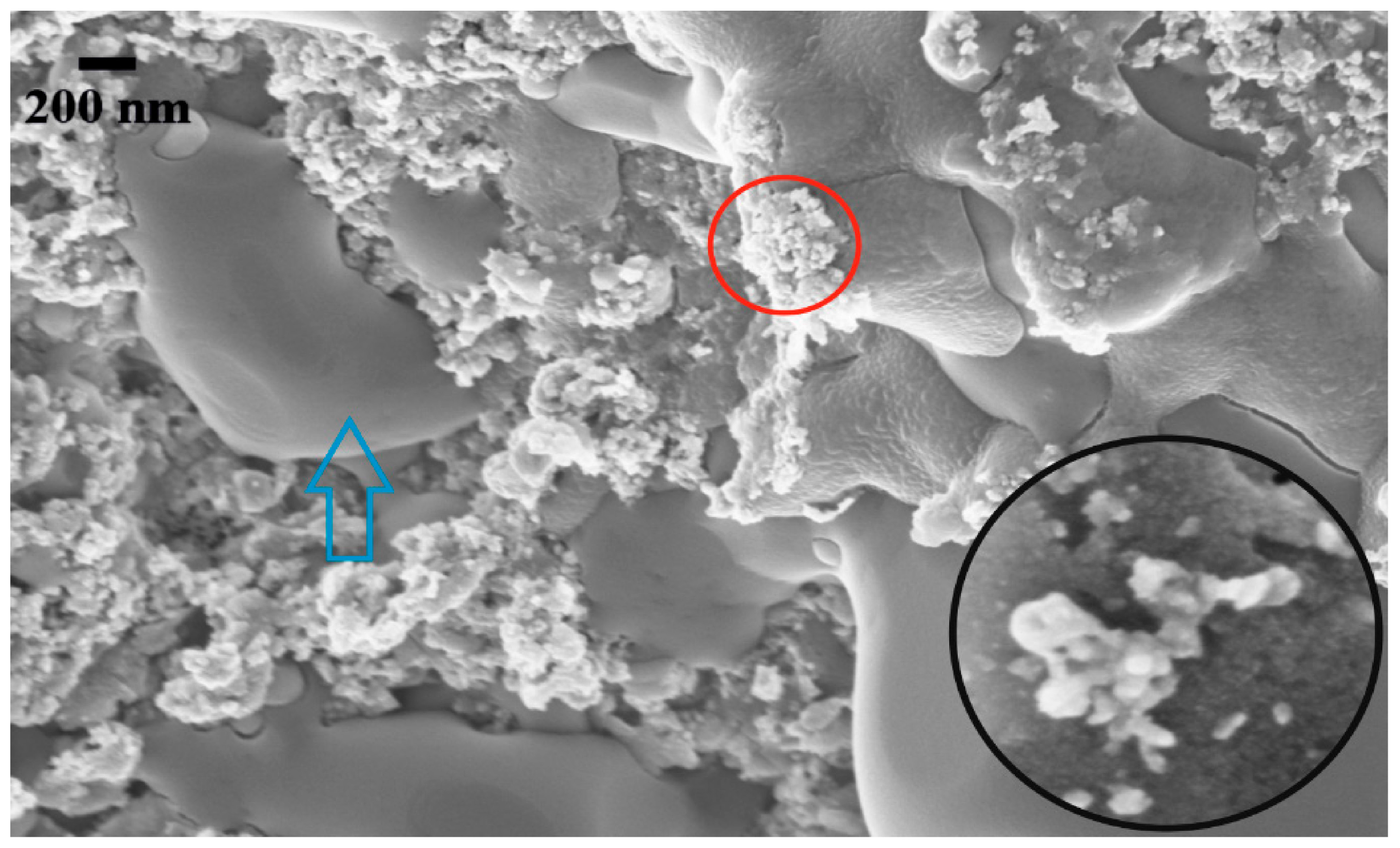
2.3. Scanning Electron Microscopy (SEM) and Energy Dispersive X-Ray (EDX)
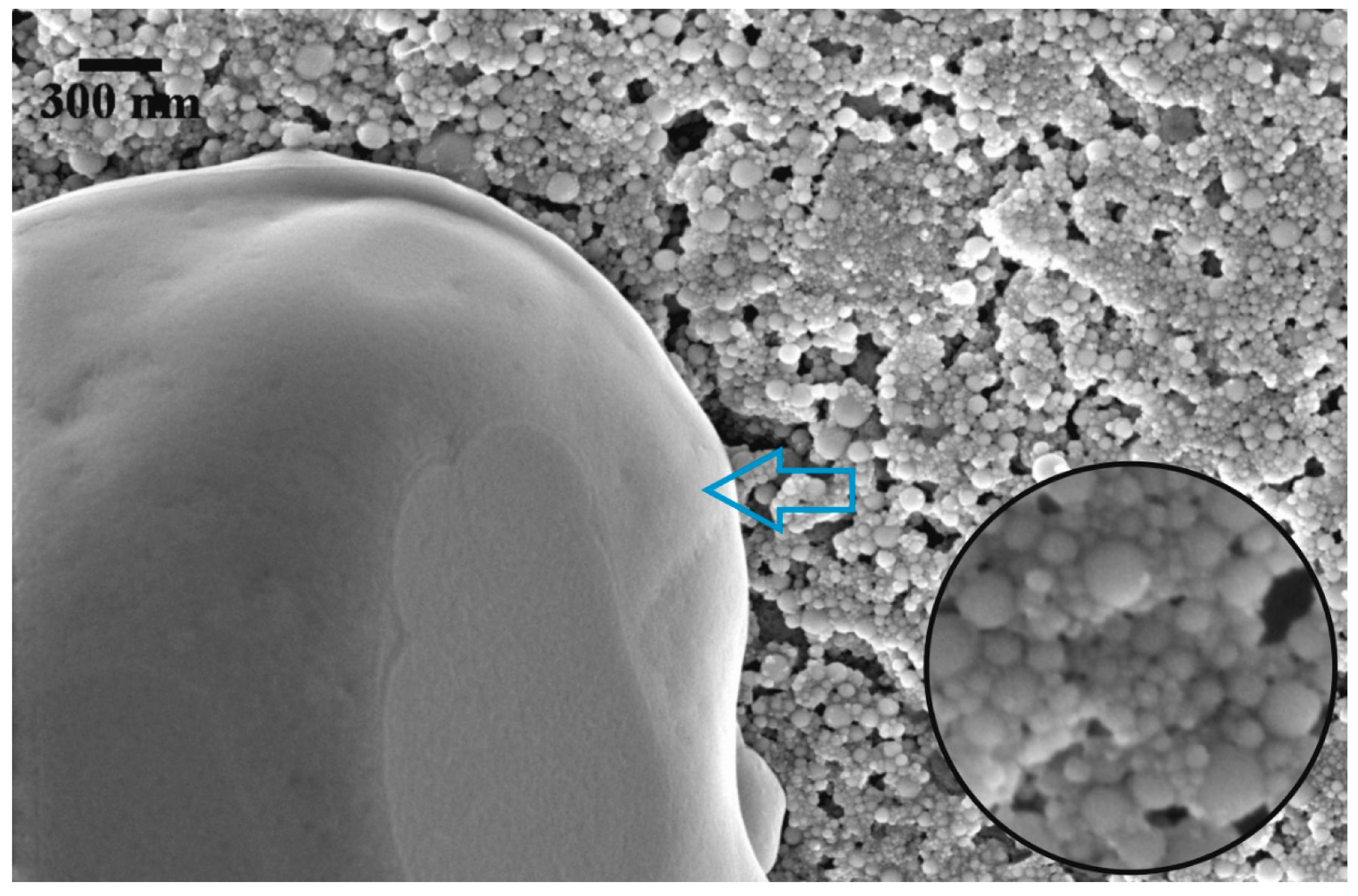
3. Results and Discussions
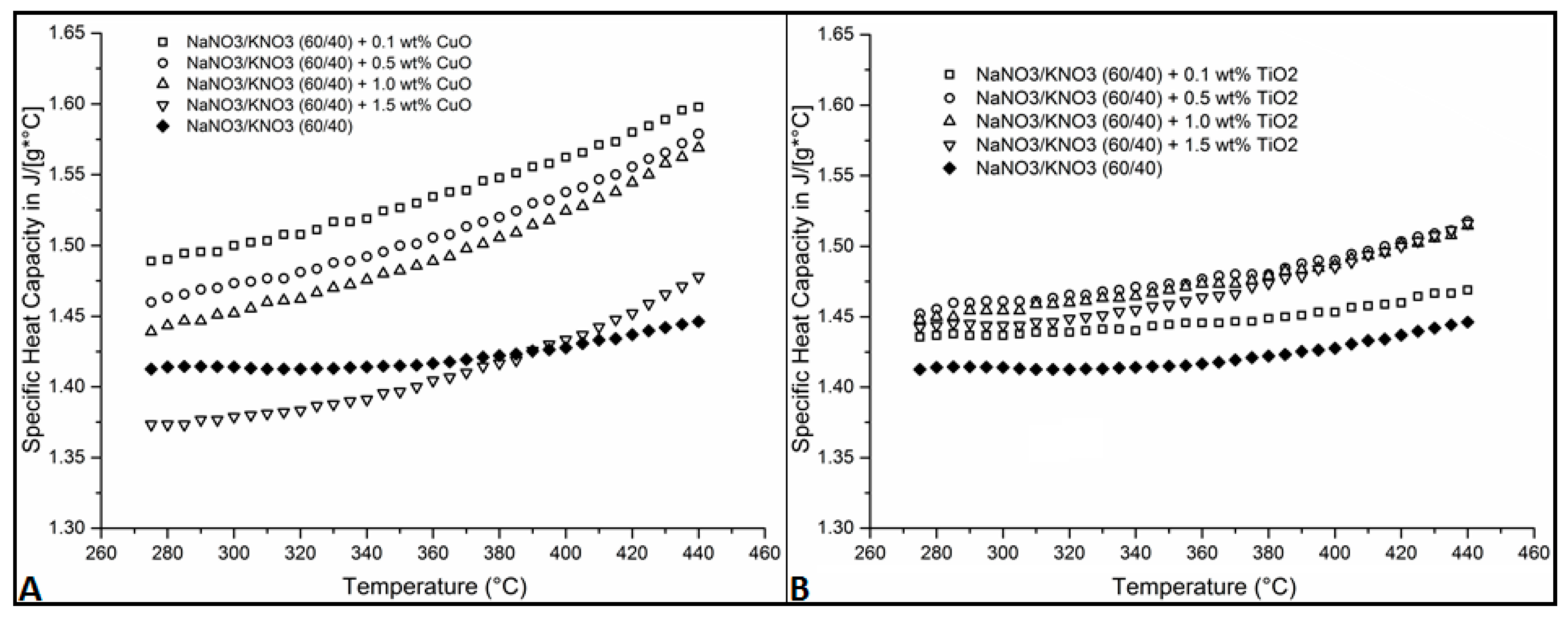
| Temperature | 0.1 wt % CuO | 0.5 wt % CuO | 1.0 wt % CuO | 1.5 wt % CuO | 0.1 wt % TiO2 | 0.5 wt % TiO2 | 1.0 wt % TiO2 | 1.5 wt % TiO2 |
|---|---|---|---|---|---|---|---|---|
| °C | % Increase | % Increase | % Increase | % Increase | % Increase | % Increase | % Increase | % Increase |
| 265 | 4.87 | 2.97 | 1.47 | −2.79 | 1.39 | 2.66 | 2.03 | 2.07 |
| 300 | 6.08 | 4.19 | 2.70 | −2.49 | 1.60 | 3.33 | 2.86 | 2.10 |
| 350 | 7.90 | 6.02 | 4.76 | −1.29 | 2.09 | 4.13 | 3.82 | 3.10 |
| 400 | 9.43 | 7.71 | 6.78 | 0.40 | 1.80 | 4.37 | 4.13 | 4.02 |
| 440 | 10.48 | 9.18 | 8.49 | 2.19 | 1.57 | 4.95 | 4.72 | 4.85 |


4. Conclusions
Acknowledgment
Author Contributions
Conflicts of Interest
References
- Choi, S.U.S.; Eastman, J.A. Enhancing Thermal Conductivity of Fluids with Nano-Particles. In Proceedings of the International Mechanical Engineering Congress and Exposition, San Francisco, CA, USA, 12–17 November 1995.
- Lee, S.; Choi, S.U.S.; Li, S.; Eastman, J.A. Measuring thermal conductivity of fluids containing oxide nanoparticles. J. Heat Transf.-Trans. Asme 1999, 121, 280–289. [Google Scholar] [CrossRef]
- Das, S.K.; Putra, N.; Thiesen, P.; Roetzel, W. Temperature dependence of thermal conductivity enhancement for nanofluids. J. Heat Transf.-Trans. Asme 2003, 125, 567–574. [Google Scholar] [CrossRef]
- Patel, H.E.; Das, S.K.; Sundararajan, T.; Nair, A.S.; George, B.; Pradeep, T. Thermal conductivities of naked and monolayer protected metal nanoparticle based nanofluids: Manifestation of anomalous enhancement and chemical effects. Appl. Phys. Lett. 2003, 83, 2931–2933. [Google Scholar] [CrossRef]
- Wen, D.S.; Ding, Y.L. Experimental investigation into convective heat transfer of nanofluids at the entrance region under laminar flow conditions. Int. J. Heat Mass Transf. 2004, 47, 5181–5188. [Google Scholar] [CrossRef]
- Wen, D.S.; Ding, Y.L. Formulation of nanofluids for natural convective heat transfer applications. Int. J. Heat Fluid Flow 2005, 26, 855–864. [Google Scholar] [CrossRef]
- Xie, H.Q.; Wang, J.C.; Xi, T.G.; Liu, Y.; Ai, F. Dependence of the thermal conductivity of nanoparticle-fluid mixture on the base fluid. J. Mater. Sci. Lett. 2002, 21, 1469–1471. [Google Scholar] [CrossRef]
- Zhang, X.; Gu, H.; Fujii, M. Effective thermal conductivity and thermal diffusivity of nanofluids containing spherical and cylindrical nanoparticles. Exp. Therm. Fluid Sci. 2007, 31, 593–599. [Google Scholar] [CrossRef]
- Eastman, J.A.; Choi, S.U.S.; Li, S.; Yu, W.; Thompson, L.J. Anomalously increased effective thermal conductivities of ethylene glycol-based nanofluids containing copper nanoparticles. Appl. Phys. Lett. 2001, 78, 718–720. [Google Scholar] [CrossRef]
- He, Y.; Jin, Y.; Chen, H.; Ding, Y.; Cang, D.; Lu, H. Heat transfer and flow behaviour of aqueous suspensions of TiO2 nanoparticles (nanofluids) flowing upward through a vertical pipe. Int. J. Heat Mass Transf. 2007, 50, 2272–2281. [Google Scholar] [CrossRef]
- Ding, Y.L.; Alias, H.; Wen, D.S.; Williams, R.A. Heat transfer of aqueous suspensions of carbon nanotubes (CNT nanofluids). Int. J. Heat Mass Transf. 2006, 49, 240–250. [Google Scholar] [CrossRef]
- Ding, Y.L.; Chen, H.; Wang, L.; Yang, C.-Y.; He, Y.; Yang, W.; Lee, W.P.; Zhang, L.; Huo, R. Heat transfer intensification using nanofluids. Kona-Powder Part. 2007, 25, 23–38. [Google Scholar] [CrossRef]
- Xie, H.Q.; Wang, J.; Xi, T.; Liu, Y.; Ai, F.; Wu, Q. Thermal conductivity enhancement of suspensions containing nanosized alumina particles. J. Appl. Phys. 2002, 91, 4568–4572. [Google Scholar] [CrossRef]
- Trisaksri, V.; Wongwises, S. Critical review of heat transfer characteristics of nanofluids. Renew. Sustain. Energy Rev. 2007, 11, 512–523. [Google Scholar] [CrossRef]
- Yu, W.; Xie, H. A review on nanofluids: Preparation, stability mechanisms, and applications. J. Nanomater. 2012, 2012. [Google Scholar] [CrossRef]
- Yang, X.; Liu, Z. A kind of nanofluid consisting of surface-functionalized nanoparticles. Nanoscale Res. Lett. 2010, 5, 1324–1328. [Google Scholar] [CrossRef] [PubMed]
- Prasher, R.; Phelan, P.E.; Bhattacharya, P. Effect of aggregation kinetics on the thermal conductivity of nanoscale colloidal solutions (nanofluid). Nano Lett. 2006, 6, 1529–1534. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.; Kim, J.W.; Kim, B.G. A new parameter to control heat transport in nanofluids: Surface charge state of the particle in suspension. J. Phys. Chem. B 2006, 110, 4323–4328. [Google Scholar] [CrossRef] [PubMed]
- Das, S.K.; Choi, S.U.S.; Patel, H.E. Heat transfer in nanofluids—A review. Heat Transf. Eng. 2006, 27, 3–19. [Google Scholar] [CrossRef]
- Xuan, Y.M.; Li, Q. Heat transfer enhancement of nanofluids. Int. J. Heat Fluid Flow 2000, 21, 58–64. [Google Scholar] [CrossRef]
- Keblinski, P.; Prasher, R.; Eapen, J. Thermal conductance of nanofluids: Is the controversy over? J. Nanoparticle Res. 2008, 10, 1089–1097. [Google Scholar] [CrossRef]
- Buongiorno, J.; Venerus, D.C.; Prabhat, N.; McKrell, T.; Townsend, J.; Christianson, R.; Tolmachev, Y.V.; Keblinski, P.; Hu, L.-W.; Alvarado, J.L.; et al. A benchmark study on the thermal conductivity of nanofluids. J. Appl. Phys. 2009, 106. [Google Scholar] [CrossRef]
- Zhou, S.-Q.; Ni, R. Measurement of the specific heat capacity of water-based Al(2)O(3) nanofluid. Appl. Phys. Lett. 2008, 92. [Google Scholar] [CrossRef]
- Pantzali, M.N.; Kanaris, A.G.; Antoniadis, K.D.; Mouza, A.A.; Paras, S.V. Effect of nanofluids on the performance of a miniature plate heat exchanger with modulated surface. Int. J. Heat Fluid Flow 2009, 30, 691–699. [Google Scholar] [CrossRef]
- Lu, M.C.; Huang, C.H. Specific heat capacity of molten salt-based alumina nanofluid. Nanoscale Res. Lett. 2013, 8. [Google Scholar] [CrossRef] [PubMed]
- Shin, D.; Banerjee, D. Effects of silica nanoparticles on enhancing the specific heat capacity of carbonate salt eutectic (work in progress). Int. J. Struct. Chang. Solids 2010, 2, 25–31. [Google Scholar]
- Shin, D.; Banerjee, D. Enhancement of specific heat capacity of high-temperature silica-nanofluids synthesized in alkali chloride salt eutectics for solar thermal-energy storage applications. Int. J. Heat Mass Transf. 2011, 54, 1064–1070. [Google Scholar] [CrossRef]
- Shin, D.H.; Banerjee, D. Enhanced specific heat of silica nanofluid. J. Heat Transf.-Trans. Asme 2011, 133. [Google Scholar] [CrossRef]
- Tiznobaik, H.; Shin, D. Enhanced specific heat capacity of high-temperature molten salt-based nanofluids. Int. J. Heat Mass Transf. 2013, 57, 542–548. [Google Scholar] [CrossRef]
- Tiznobaik, H.; Shin, D. Experimental validation of enhanced heat capacity of ionic liquid-based nanomaterial. Appl. Phys. Lett. 2013, 102. [Google Scholar] [CrossRef]
- Jo, B.; Banerjee, D. Enhanced specific heat capacity of molten salt-based nanomaterials: Effects of nanoparticle dispersion and solvent material. Acta Mater. 2014, 75, 80–91. [Google Scholar] [CrossRef]
- Chieruzzi, M.; Cerritelli, G.; Miliozzi, A.; Kenny, J. Effect of nanoparticles on heat capacity of nanofluids based on molten salts as PCM for thermal energy storage. Nanoscale Res. Lett. 2013, 448. [Google Scholar] [CrossRef] [PubMed]
- Andreu-Cabedo, P.; Mondragon, R.; Hernandez, L.; Martinez-Cuenca, R.; Cabedo, L.; Julia, J.E. Increment of specific heat capacity of solar salt with SiO2 nanoparticles. Nanoscale Res. Lett. 2014, 9, 582. [Google Scholar] [CrossRef] [PubMed]
- Dudda, B.; Shin, D. Effect of nanoparticle dispersion on specific heat capacity of a binary nitrate salt eutectic for concentrated solar power applications. Int. J. Therm. Sci. 2013, 69, 37–42. [Google Scholar] [CrossRef]
- Tan, Z.C.; Wang, L.; Shi, Q. Study of heat capacity enhancement in some nanostructured materials. Pure Appl. Chem. 2009, 81, 1871–1880. [Google Scholar] [CrossRef]
- Oh, S.H.; Kauffmann, Y.; Scheu, C.; Kaplan, W.D.; Ruhle, M. Ordered liquid aluminum at the interface with sapphire. Science 2005, 310, 661–663. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Tan, Z.C.; Meng, S.G.; Liang, D.B.; Li, G.G. Enhancement of molar heat capacity of nanostructured Al2O3. J. Nanoparticle Res. 2001, 3, 483–487. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lasfargues, M.; Geng, Q.; Cao, H.; Ding, Y. Mechanical Dispersion of Nanoparticles and Its Effect on the Specific Heat Capacity of Impure Binary Nitrate Salt Mixtures. Nanomaterials 2015, 5, 1136-1146. https://doi.org/10.3390/nano5031136
Lasfargues M, Geng Q, Cao H, Ding Y. Mechanical Dispersion of Nanoparticles and Its Effect on the Specific Heat Capacity of Impure Binary Nitrate Salt Mixtures. Nanomaterials. 2015; 5(3):1136-1146. https://doi.org/10.3390/nano5031136
Chicago/Turabian StyleLasfargues, Mathieu, Qiao Geng, Hui Cao, and Yulong Ding. 2015. "Mechanical Dispersion of Nanoparticles and Its Effect on the Specific Heat Capacity of Impure Binary Nitrate Salt Mixtures" Nanomaterials 5, no. 3: 1136-1146. https://doi.org/10.3390/nano5031136
APA StyleLasfargues, M., Geng, Q., Cao, H., & Ding, Y. (2015). Mechanical Dispersion of Nanoparticles and Its Effect on the Specific Heat Capacity of Impure Binary Nitrate Salt Mixtures. Nanomaterials, 5(3), 1136-1146. https://doi.org/10.3390/nano5031136






