Synthesis and Adsorption Property of SiO2@Co(OH)2 Core-Shell Nanoparticles
Abstract
:1. Introduction
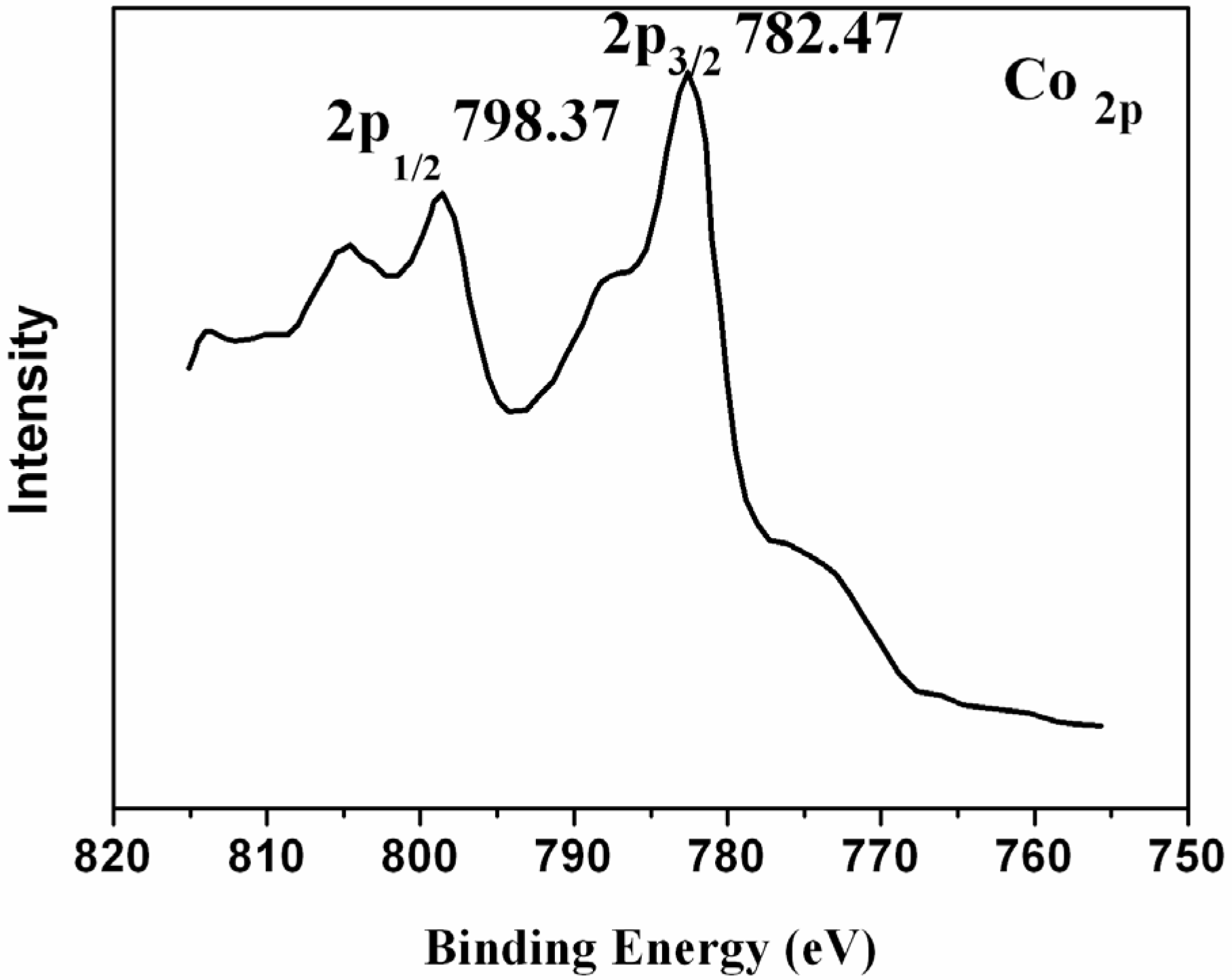
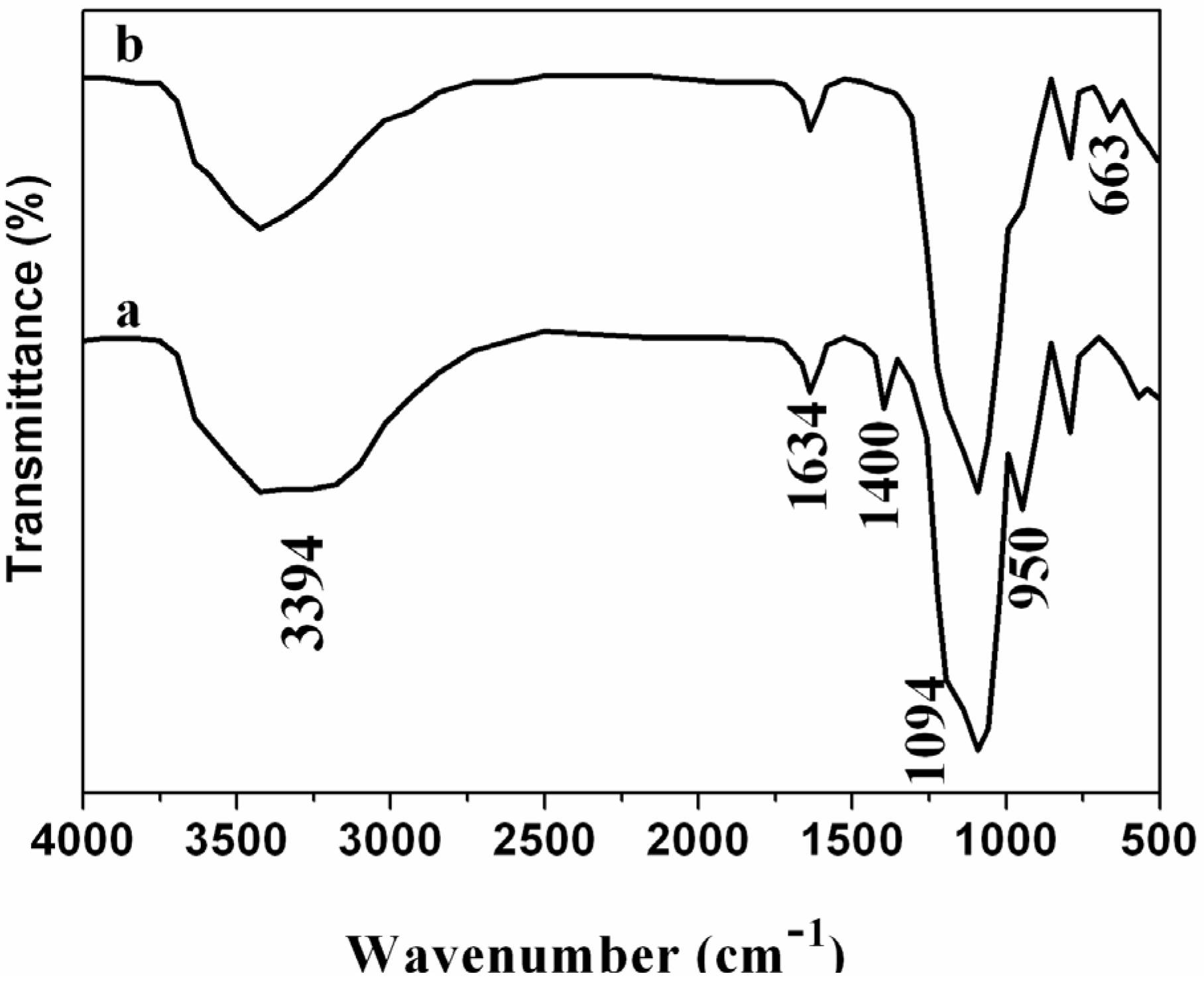
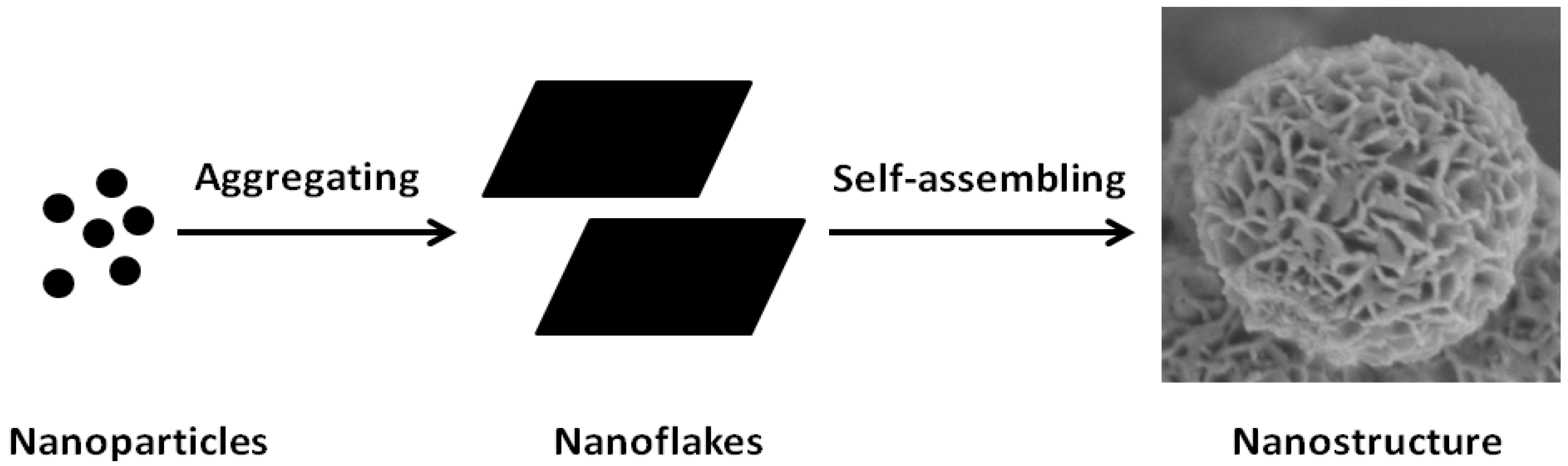
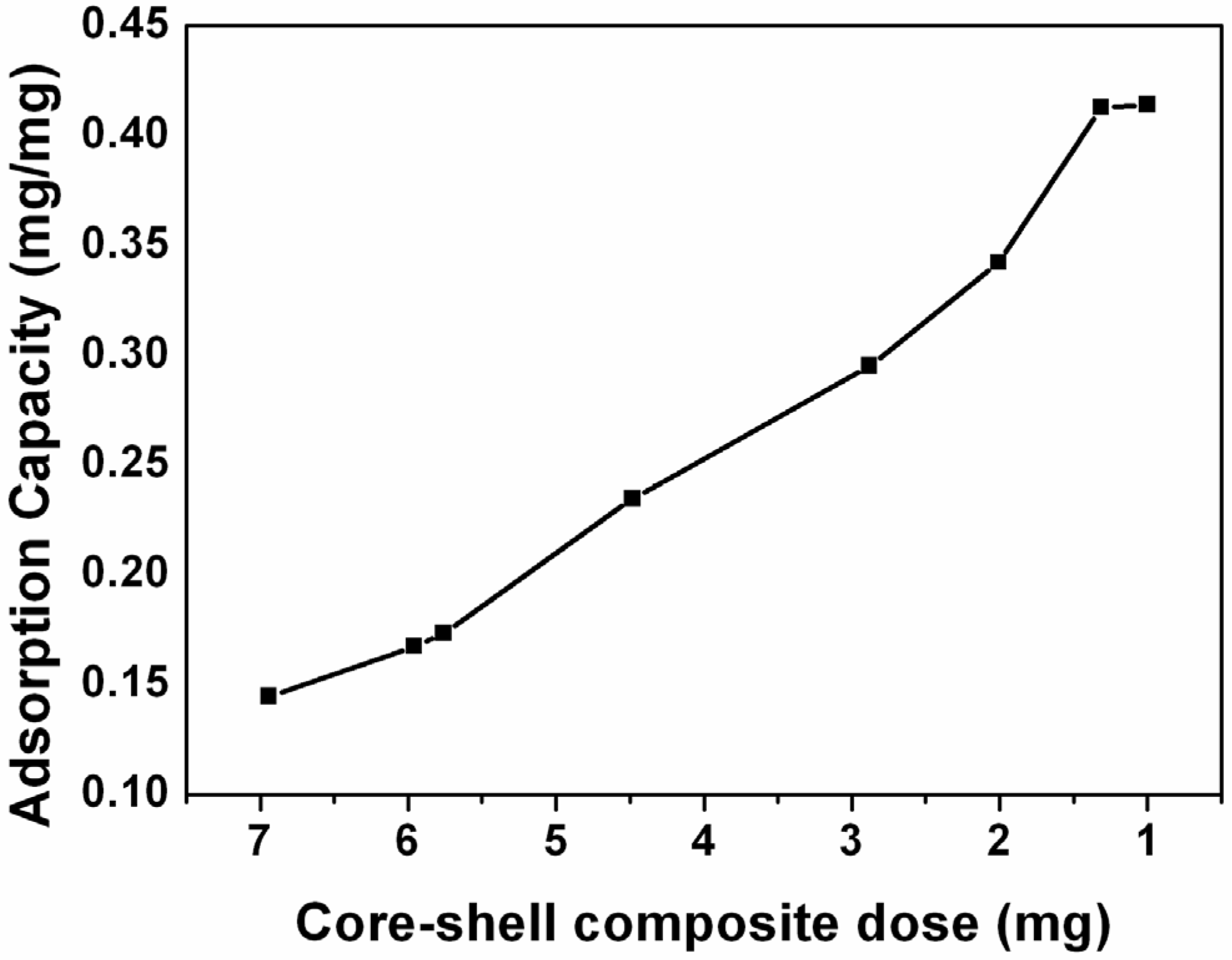
2. Results and Discussion


3. Experimental Section
3.1. Synthesis
3.2. Adsorption of RB
3.3. Characterization
4. Conclusions
Supplementary Materials
Acknowledgements
Conflicts of Interest
References
- Chen, G.; Ågren, H.; Ohulchanskyy, T.Y.; Prasad, P.N. Light upconverting core-shell nanostructures: Nanophotonic control for emerging applications. Chem. Soc. Rev. 2015, 44, 1680–1713. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Chen, H.; Zeng, D.; Tian, Y.; Chen, F.; Feng, J.; Shi, J. Core/shell structured hollow mesoporous nanocapsules: A potential platform for simultaneous cell imaging and anticancer drug delivery. ACS Nano 2010, 4, 6001–6013. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.; Yi, H.; Yuan, Y.; Jing, P.; Chai, Y.; Yuan, R.; Wilson, G.S. An electrochemical aptasensor for thrombin using synergetic catalysis of enzyme and porous Au@Pd core-shell nanostructures for signal amplification. Biosens. Bioelectron. 2015, 64, 423–428. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Jiang, P. Core-shell structured high-k polymer nanocomposites for energy storage and dielectric applications. Adv. Mater. 2015, 27, 546–554. [Google Scholar] [CrossRef] [PubMed]
- Boekhoven, J.; Zha, R.H.; Tantakitti, F.; Zhuang, E.; Zandi, R.; Newcomb, C.J.; Stupp, S.I. Alginate-peptide amphiphile core-shell microparticles as a targeted drug delivery system. RSC Adv. 2015, 5, 8753–8756. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Chen, H.; Guo, L.; He, Q.; Chen, F.; Zhou, J.; Shi, J. Hollow/rattle-type mesoporous nanostructures by a structural difference-based selective etching strategy. ACS Nano 2009, 4, 529–539. [Google Scholar] [CrossRef]
- Chen, Y.; Meng, Q.; Wu, M.; Wang, S.; Xu, P.; Chen, H.; Shi, J. Hollow mesoporous organosilica nanoparticles: a generic intelligent framework-hybridization approach for biomedicine. J. Am. Chem. Soc. 2014, 136, 16326–16334. [Google Scholar] [CrossRef] [PubMed]
- Salgueiriño-Maceira, V.; Correa-Duarte, M.A. Cobalt and silica based core–shell structured nanospheres. J. Mater. Chem. 2006, 16, 3593–3597. [Google Scholar] [CrossRef]
- Salgueiriño-Maceira, V.; Spasova, M.; Farle, M. Water-stable, magnetic silica–cobalt/cobalt oxide–silica multishell submicrometer spheres. Adv. Funct. Mater. 2005, 15, 1036–1040. [Google Scholar] [CrossRef]
- Claesson, E.; Philipse, A. Monodisperse magnetizable composite silica spheres with tunable dipolar interactions. Langmuir 2005, 21, 9412–9419. [Google Scholar] [CrossRef] [PubMed]
- Ocana, M.; Gonzalez-Elipe, A. Preparation and characterization of uniform spherical silica particles coated with Ni and Co compounds. Colloids Surf. A 1999, 157, 315–324. [Google Scholar] [CrossRef]
- Hou, Y.; Kondoh, H.; Shimojo, M.; Kogure, T.; Ohta, T. High-yield preparation of uniform cobalt hydroxide and oxide nanoplatelets and their characterization. J. Phys. Chem. B 2005, 109, 19094–19098. [Google Scholar] [CrossRef] [PubMed]
- Kong, L.-B.; Lang, J.-W.; Liu, M.; Luo, Y.-C.; Kang, L. Facile approach to prepare loose-packed cobalt hydroxide nano-flakes materials for electrochemical capacitors. J. Power Sour. 2009, 194, 1194–1201. [Google Scholar] [CrossRef]
- Song, D.; Wang, Y.; Wang, Q.; Wang, Y.; Jiao, L.; Yuan, H. Effect and function mechanism of amorphous sulfur on the electrochemical properties of cobalt hydroxide electrode. J. Power Sour. 2010, 195, 7115–7119. [Google Scholar] [CrossRef]
- Qin, H.; Lao, S.; Liu, Z.; Zhu, J.; Li, Z. Effects of heat treatment on the structure, morphology and electrocatalytic activity of cobalt hydroxide electrocatalyst. Int. J. Hydrog. Energy 2010, 35, 1872–1878. [Google Scholar] [CrossRef]
- Liu, X.; Ma, R.; Bando, Y.; Sasaki, T. Layered cobalt hydroxide nanocones: Micro–assisted synthesis, exfoliation, and structure modification. Angew. Chem. Int. Ed. 2010, 49, 8253–8256. [Google Scholar] [CrossRef]
- Wang, W.Z.; Zhou, Q.; Wang, L.; Yang, T.; Zhang, G. A room temperature solution-phase process to synthesize pure phase single–crystalline hexagonal cobalt hydroxide nanoplates. J. Cryst. Growth 2010, 312, 3845–3849. [Google Scholar]
- Yang, J.; Liu, H.; Martens, W.N.; Frost, R.L. Synthesis and characterization of cobalt hydroxide, cobalt oxyhydroxide, and cobalt oxide nanodiscs. J. Phys. Chem. C 2009, 114, 111–119. [Google Scholar] [CrossRef]
- Hosono, E.; Fujihara, S.; Honma, I.; Zhou, H. Fabrication of morphology and crystal structure controlled nanorod and nanosheet cobalt hydroxide based on the difference of oxygen-solubility between water and methanol, and conversion into Co3O4. J. Mater. Chem. 2005, 15, 1938–1945. [Google Scholar] [CrossRef]
- Lou, X.W.; Deng, D.; Lee, J.Y.; Feng, J.; Archer, L.A. Self-supported formation of needlelike Co3O4 nanotubes and their application as lithium-ion battery electrodes. Adv. Mater. 2008, 20, 258–262. [Google Scholar] [CrossRef]
- Zhang, H.; Wu, J.; Zhai, C.; Ma, X.; Du, N.; Tu, J.; Yang, D. From cobalt nitrate carbonate hydroxide hydrate nanowires to porous nanorods for high performance lithium-ion battery electrodes. Nanotechnology 2008, 19. [Google Scholar] [CrossRef]
- Oaki, Y.; Imai, H. Biomimetic morphological design for manganese oxide and cobalt hydroxide nanoflakes with a mosaic interior. J. Mater. Chem. 2007, 17, 316–321. [Google Scholar] [CrossRef]
- Yan, C.; Jiang, H.; Zhao, T.; Li, C.; Ma, J.; Lee, P.S. Binder-free Co(OH)2 nanoflake-ITO nanowire heterostructured electrodes for electrochemical energy storage with improved high-rate capabilities. J. Mater. Chem. 2011, 21, 10482–10488. [Google Scholar] [CrossRef]
- Wang, M.; Ma, J.; Chen, C.; Zheng, X.; Du, Z.; Xu, J. Preparation of self-assembled cobalt hydroxide nanoflowers and the catalytic decomposition of cyclohexyl hydroperoxide. J. Mater. Chem. 2011, 21, 12609–12612. [Google Scholar] [CrossRef]
- Zhang, Y.; Xia, X.; Kang, J.; Tu, J. Hydrothermal synthesized porous Co(OH)2 nanoflake film for supercapacitor application. Chin. Sci. Bull. 2012, 57, 4215–4219. [Google Scholar] [CrossRef]
- Zhang, J.; Kong, L.B.; Cai, J.J.; Luo, Y.C.; Kang, L. Nanoflake-like cobalt hydroxide/ordered mesoporous cabon composite for electrochemical capacitors. J. Solid State Electrochem. 2010, 14, 2065–2075. [Google Scholar] [CrossRef]
- Sampanthar, J.T.; Zeng, H.C. Arresting butterfly-like intermediate nanocrystals of β-Co(OH)2 via ethylenediamine-mediated synthesis. J. Am. Chem. Soc. 2002, 124, 6668–6675. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Qian, H.; Ren, J. CdTe@Co(OH)2 (core-shell) nanoparticles: Aqueous synthesis and characterization. Chem. Commun. 2005, 2005, 4083–4085. [Google Scholar] [CrossRef]
- Xu, R.; Zeng, H.C. Mechanistic investigation on self-redox decompositions of cobalt-hydroxide-nitrate compounds with different nitrate anion configurations in interlayer space. Chem. Mater. 2003, 15, 2040–2048. [Google Scholar] [CrossRef]
- Burneau, A.; Barres, O.; Gallas, J.P.; Lavalley, J.C. Comparative study of the surface hydroxyl groups of fumed and precipitated silicas. 2. Characterization by infrared spectroscopy of the interactions with water. Langmuir 1990, 6, 1364–1372. [Google Scholar] [CrossRef]
- Alvarez-Puebla, R.A.; Garrido, J.J.; Valenzuela-Calahorro, C.; Goulet, P.J.G. Retention and induced aggregation of Co(II) on a humic substance: Sorption isotherms, infrared absorption, and molecular modeling. J. Surf. Sci. 2005, 575, 136–146. [Google Scholar] [CrossRef]
- Díaz, R.; Lazo, M.F. Spectroscopic study of CuO/CoO catalysts supported by Si-Al-Y zeolite matrices prepared by two sol-gel methods. J. Sol-Gel Sci. Technol. 2000, 17, 137–144. [Google Scholar] [CrossRef]
- Yoshino, T.; Baba, N. Characterization and properties of electrochromic cobalt oxide thin film prepared by electrodeposition. Sol. Energy Mater. Sol. Cells 1995, 39, 391–397. [Google Scholar] [CrossRef]
- Graf, C.; Vossen, D.L.; Imhof, A.; van Blaaderen, A. A general method to coat colloidal particles with silica. Langmuir 2003, 19, 6693–6700. [Google Scholar] [CrossRef]
- Jiang, H.; Zhao, T.; Li, C.; Ma, J. Hierarchical self-assembly of ultrathin nickel hydroxide nanoflakes for high-performance supercapacitors. J. Mater. Chem. 2011, 21, 3818–3823. [Google Scholar] [CrossRef]
- Xu, R.; Zeng, H.C. Dimensional control of cobalt-hydroxide-carbonate nanorods and their thermal conversion to one-dimensional arrays of Co3O4 nanoparticles. J. Phys. Chem. B 2003, 107, 12643–12649. [Google Scholar] [CrossRef]
- Mustafa, S.; Nawab, G.; Naeem, A.; Rehana, N.; Safdar, M. Sorption of phosphate on cobalt hydroxide. J. Chem. Soc. Pak. 2003, 25, 25–28. [Google Scholar]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meng, Y. Synthesis and Adsorption Property of SiO2@Co(OH)2 Core-Shell Nanoparticles. Nanomaterials 2015, 5, 554-564. https://doi.org/10.3390/nano5020554
Meng Y. Synthesis and Adsorption Property of SiO2@Co(OH)2 Core-Shell Nanoparticles. Nanomaterials. 2015; 5(2):554-564. https://doi.org/10.3390/nano5020554
Chicago/Turabian StyleMeng, Yongde. 2015. "Synthesis and Adsorption Property of SiO2@Co(OH)2 Core-Shell Nanoparticles" Nanomaterials 5, no. 2: 554-564. https://doi.org/10.3390/nano5020554
APA StyleMeng, Y. (2015). Synthesis and Adsorption Property of SiO2@Co(OH)2 Core-Shell Nanoparticles. Nanomaterials, 5(2), 554-564. https://doi.org/10.3390/nano5020554




