Tuning of the Electronic and Magnetic Properties of GaN Monolayers via Doping with Lanthanide Atoms and by Applying Biaxial Strain
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
3.1. Ln-GaN Monolayer
3.2. Ln-GaN Systems at −6%~6% Biaxial Strain
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chung, K.; Lee, C.-H.; Yi, G.-C. Transferable Gan Layers Grown on ZnO-Coated Graphene Layers for Optoelectronic Devices. Science 2010, 330, 655–657. [Google Scholar] [CrossRef]
- Gupta, A.; Sakthivel, T.; Seal, S. Recent development in 2D materials beyond graphene. Prog. Mater. Sci. 2015, 73, 44–126. [Google Scholar] [CrossRef]
- Bhimanapati, G.-R.; Lin, Z.; Meunier, V.; Jung, Y.; Cha, J.; Das, S.; Xiao, D.; Son, Y.; Strano, M.-S.; Cooper, V.-R.; et al. Recent Advances in Two-Dimensional Materials beyond Graphene. ACS Nano 2015, 9, 11509–11539. [Google Scholar] [CrossRef]
- Şahin, H.; Cahangirov, S.; Topsakal, M.; Bekaroglu, E.; Akturk, E.; Senger, R.-T.; Ciraci, S. Monolayer honeycomb structures of group-iv elements and III-V binary compounds: First-principles calculations. Phys. Rev. B-Condens. Matter Mater. Phys. 2009, 80, 155453. [Google Scholar] [CrossRef]
- Al Balushi, Z.-Y.; Wang, K.; Ghosh, R.-K.; Vilá, R.-A.; Eichfeld, S.-M.; Caldwell, J.-D.; Qin, X.-Y.; Lin, Y.-C.; Desario, P.-A.; Stone, G.; et al. Two-dimensional gallium nitride realized via graphene encapsulation. Nat. Mater. 2016, 15, 1166–1171. [Google Scholar] [CrossRef]
- Chen, Y.-X.; Liu, K.-L.; Liu, J.-X.; Lv, T.-R.; Wei, B.; Zhang, T.; Zeng, M.-Q.; Wang, Z.-C.; Fu, L. Growth of 2D GaN Single Crystals on Liquid Metals. J. Am. Chem. Soc. 2018, 140, 16392–16395. [Google Scholar] [CrossRef] [PubMed]
- Shu, H.-B.; Niu, X.-H.; Ding, X.-J.; Wang, Y. Effects of strain and surface modification on stability, electronic and optical properties of GaN monolayer. Appl. Surf. Sci. 2019, 479, 475–481. [Google Scholar] [CrossRef]
- Ma, Y.-D.; Dai, Y.; Guo, M.; Niu, C.-W.; Yu, L.; Huang, B.-B. Strain-induced magnetic transitions in half-fluorinated single layers of bn, gan and graphene. Nanoscale 2011, 3, 2301–2306. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Xiong, Z.-H.; Qin, Z.-Z.; Chen, L.-L.; Wu, N.; Li, X.-X. Tuning magnetism of monolayer GaN by vacancy and nonmagnetic chemical doping. J. Phys. Chem. Solids 2016, 91, 1–6. [Google Scholar] [CrossRef]
- Wang, B.-Y.; Wang, D.; Ning, J.; Zhang, J.-C.; Hao, Y. Robust magnetic behavior in two-dimensional GaN caused by atomic vacancies. J. Mater. Sci. 2021, 56, 2311–2322. [Google Scholar] [CrossRef]
- Meng, X.-S.; Liu, H.-L.; Lin, L.-K.; Cheng, Y.-B.; Hou, X.; Zhao, S.-Y.; Lu, H.-M.; Meng, X.-K. Tuning the physical properties of two-dimensional GaN via adsorption by the groups IIIA-VIIA atoms. Appl. Surf. Sci. 2021, 539, 148302. [Google Scholar] [CrossRef]
- Ghasemzadeh, F.; Kanjouri, F. Strain effect on the electronic properties of iii-nitride nanosheets: Ab-initio study. Sci. China Technol. Sci. 2018, 61, 535–541. [Google Scholar] [CrossRef]
- Alaal, N.; Roqan, I.-S. Tuning the Electronic Properties of Hexagonal Two-Dimensional GaN Monolayers via Doping for Enhanced Optoelectronic Applications. ACS Appl. Nano Mater. 2018, 2, 202–213. [Google Scholar] [CrossRef]
- Chen, G.-X.; Fan, X.-B.; Li, S.-Q.; Zhang, J.-M. First-principles study of magnetic properties of alkali metals and alkaline earth metals doped two-dimensional GaN materials. Acta Phys. Sin. 2019, 68, 237303. [Google Scholar] [CrossRef]
- Li, J.; Liu, H. Magnetism investigation of GaN monolayer doped with group VIII B transition metals. J. Mater. Sci. 2018, 53, 15986–15994. [Google Scholar] [CrossRef]
- Su, B.; Li, N. Lanthanide atom substitutionally doped blue phosphorene: Electronic and magnetic behaviors. Phys. Chem. Chem. Phys. 2018, 20, 11003–11012. [Google Scholar] [CrossRef]
- Li, Y.-J.; Wang, M.; Tang, M.-Y.; Tian, X.; Gao, S.; He, Z.; Li, Y.; Zhou, T.-G. Spin and orbital magnetic moments and spin anisotropy energies of light rare earth atoms embedded in graphene: A first-principles study. Phys. E Low-Dimens. Syst. Nanostruct. 2016, 75, 169–173. [Google Scholar] [CrossRef]
- Obodo, K.-O.; Ouma, C.-N.-M.; Gebreyesus, G.; Obodo, J.-T.; Ezeonu, S.-O.; Bouhafs, B. DFT+ U studies of the electronic and optical properties of ReS2 mono-layer doped with lanthanide atoms. Mater. Res. Express 2019, 6, 106307. [Google Scholar] [CrossRef]
- Obodo, K.-O.; Gebreyesus, G.; Ouma, C.-N.-M.; Obodo, J.-T.; Ezeonu, S.-O.; Rai, D.-P.; Bouhafs, B. Controlling the electronic and optical properties of HfS2 mono-layers via lanthanide substitutional doping: A DFT+ U study. RSC Adv. 2020, 10, 15670–15676. [Google Scholar] [CrossRef]
- He, W.-Y.; Zhang, S.-H.; Luo, Y.; Wang, S.-K. Exploring Monolayer GaN Doped with Transition Metals: Insights from First-Principles Studies. J. Supercond. Nov. Magn. 2024, 37, 157–163. [Google Scholar] [CrossRef]
- Filho, M.-A.-M.; Farmer, W.; Hsiao, C.-L.; dos Santos, R.-B.; Hultman, L.; Birch, J.; Ankit, K.; Gueorguiev, G.-K. Density Functional Theory-Fed Phase Field Model for Semiconductor Nanostructures: The Case of Self-Induced Core−Shell InAlN Nanorods. Cryst. Growth Des 2024, 24, 4717–4727. [Google Scholar] [CrossRef]
- Pela, R.-R.; Hsiao, C.-L.; Hultman, L.; Birch, J.; Gueorguiev, G.-K. Electronic and Optical Properties of core–shell InAlN nanorods: A comparative study via LDA, LDA-1/2, mBJ, HSE06, G0W0 and BSE methods. Phys. Chem. Chem. Phys 2024, 26, 7504–7514. [Google Scholar] [CrossRef] [PubMed]
- Chhetri, S.-K.; Acharya, G.; Graf, D.; Basnet, R.; Rahman, S.; Sharma, M.-M.; Upreti, D.; Nabi, M.-R.-U.; Kryvyi, S.; Sakon, J.; et al. Large Negative Magnetoresistance in Antiferromagnetic Gd2Se3. Phys. Rev. B 2025, 111, 014431. [Google Scholar] [CrossRef]
- Acharya, G.; Pandey, K.; Sharma, M.-M.; Wang, J.; Chhetri, S.-K.; Nabi, M.-R.-U.; Upreti, D.; Basnet, R.; Sakon, J.; Hu, J. Large negative magnetoresistance in the off-stoichiometric topological semimetal PrSbxTe2−x Phys. Rev. B 2025, 111, 024421. [Google Scholar] [CrossRef]
- Li, R.; Kam, P.-Y. Averages of the product of two Gaussian Q-functions over fading statistics and applications. IEEE Commun. Lett 2007, 11, 58–60. [Google Scholar] [CrossRef]
- Perdew, J.-P.; Burke, K.; Ernzerhof, M. Generalized Gradient Approximation Made Simple. Phys. Rev. Lett. 1996, 77, 3865. [Google Scholar] [CrossRef]
- Yuan, Z.-Y.; Li, N. Manipulating the magnetic moment in phosphorene by lanthanide atom doping: A first-principle study. RSC Adv. 2016, 6, 92048–92056. [Google Scholar] [CrossRef]
- Cen, W.-L.; Liu, Y.; Wu, Z.-B.; Wang, H.-Q.; Weng, X.-L. A theoretic insight into the catalytic activity promotion of CeO2 surfaces by Mn doping. Phys. Chem. Chem. Phys. 2012, 14, 5769–5777. [Google Scholar] [CrossRef]
- Ma, C.-L.; Picozzi, S.; Wang, X.; Yang, Z.-Q. First principles study of the electronic structures of erbium silicides with non-frozen 4f treatment. Eur. Phys. J. B 2007, 59, 297–303. [Google Scholar] [CrossRef]
- Chen, W.-G.; Yuan, P.-F.; Zhang, S.; Sun, Q.; Liang, E.-J.; Jia, Y. Electronic properties of anatase TiO2 doped by lanthanides: A DFT+ U study. Phys. B Condens. Matter 2012, 407, 1038–1043. [Google Scholar] [CrossRef]
- Dudarev, S.-L.; Botton, G.-A.; Savrasov, S.-Y.; Humphreys, C.-J.; Sutton, A.-P. Electron-energy-loss spectra and the structural stability of nickel oxide: An LSDA+U study. Phys. Rev. B 1998, 57, 1505. [Google Scholar] [CrossRef]
- Liu, W.-Q.; Tong, J.-W.; Deng, L.; Yang, B.; Xie, G.-M.; Qin, G.-W.; Tian, F.-B.; Zhang, X.-M. Two-dimensional ferromagnetic semiconductors of rare-earth monolayer GdX2 (x= Cl, Br, I) with large perpendicular magnetic anisotropy and high curie temperature. Mater. Today Phys. 2021, 21, 100514. [Google Scholar] [CrossRef]
- Casadei, M.; Ren, X.-G.; Rinke, P.; Rubio, A.; Scheffler, M. Density-functional theory for f-electron systems: The α-γ phase transition in cerium. Phys. Rev. Lett. 2012, 109, 146402. [Google Scholar] [CrossRef] [PubMed]
- Henkelman, G.; Arnaldsson, A.; Jónsson, H. A fast and robust algorithm for bader decomposition of charge density. Comput. Mater. Sci. 2006, 36, 354–360. [Google Scholar] [CrossRef]
- Goesten, M.-G.; Hoffmann, R. Mirrors of Bonding in Metal Halide Perovskites. J. Am. Chem. Soc. 2018, 140, 12996–13010. [Google Scholar] [CrossRef] [PubMed]
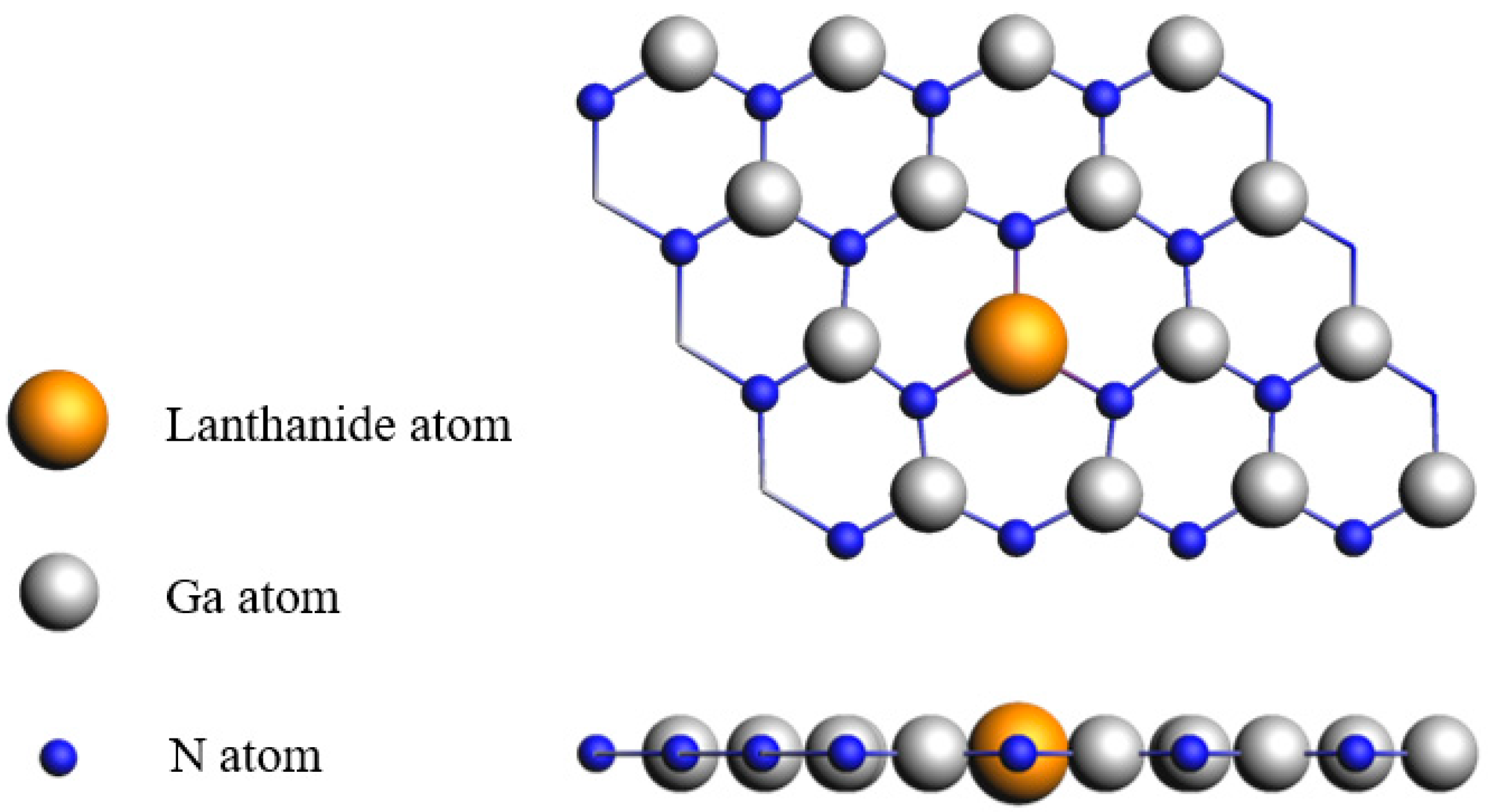
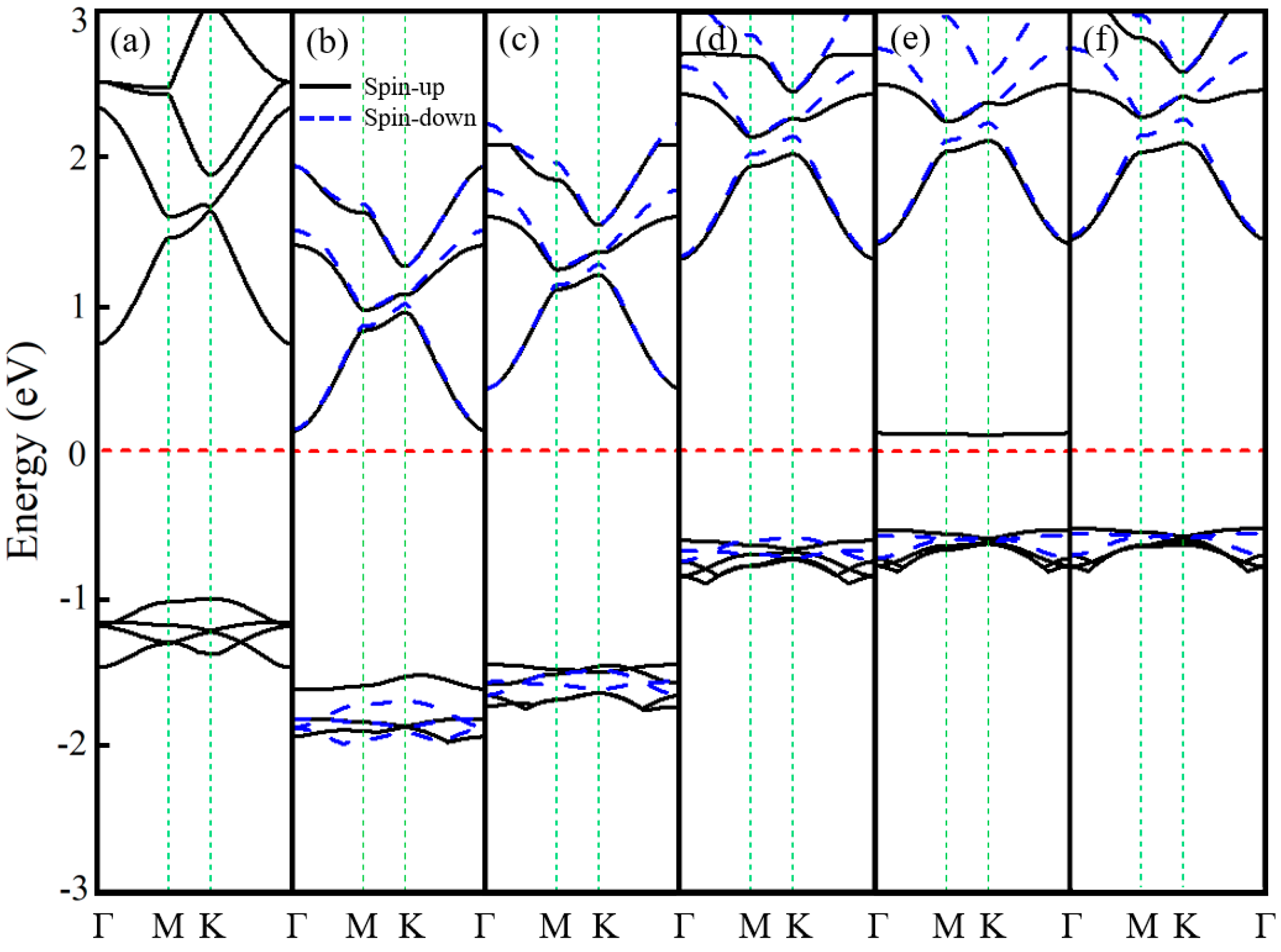
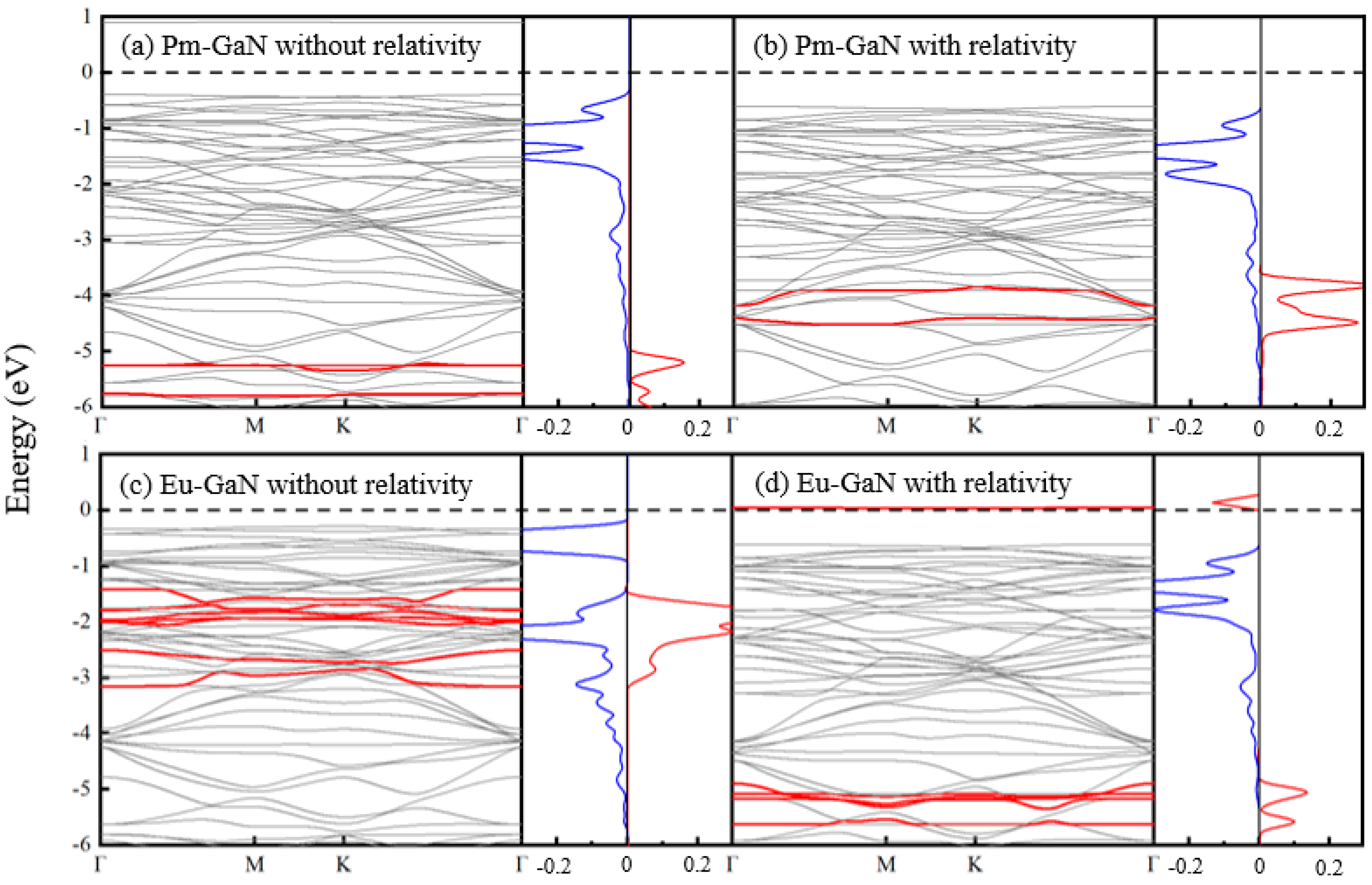
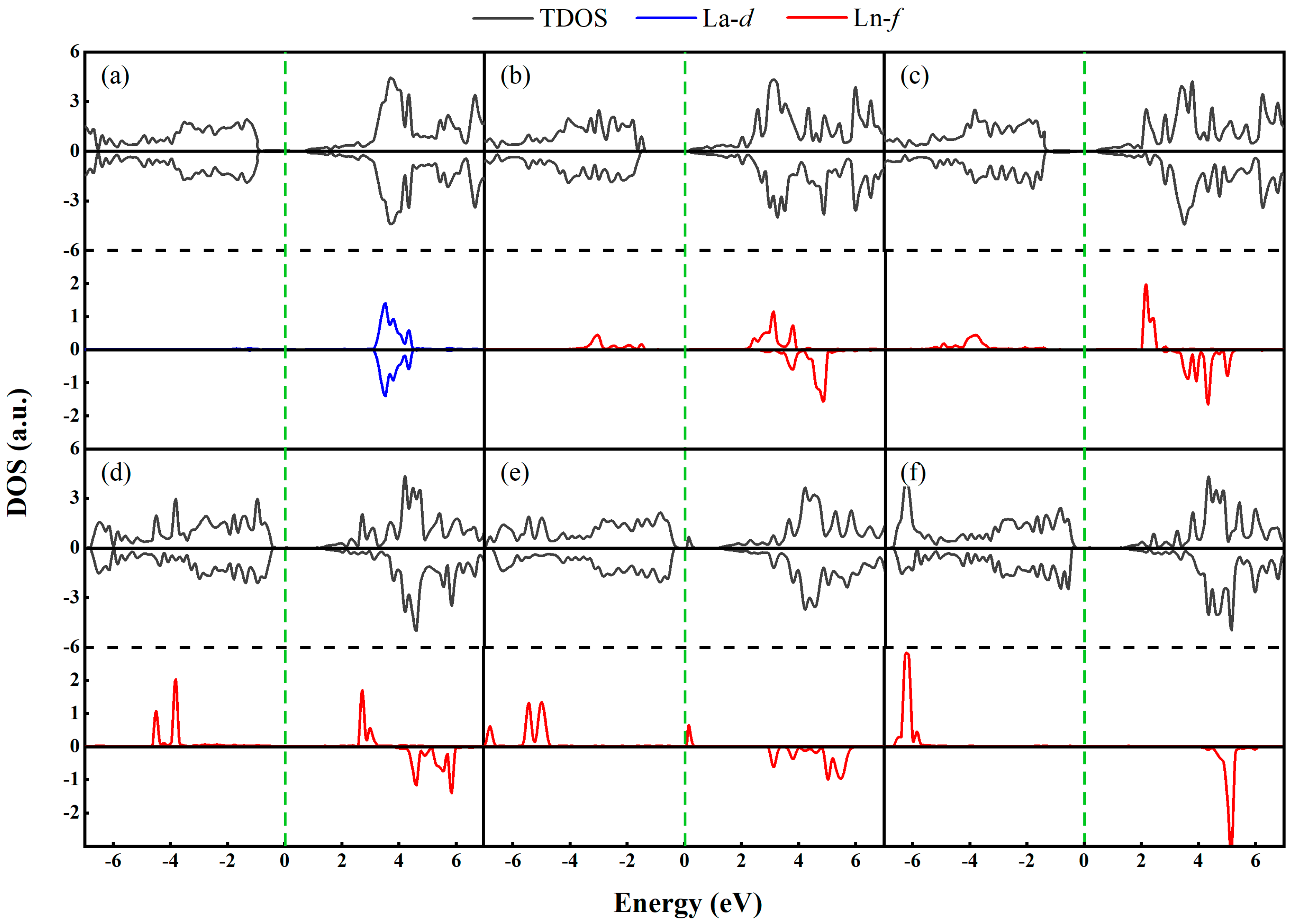
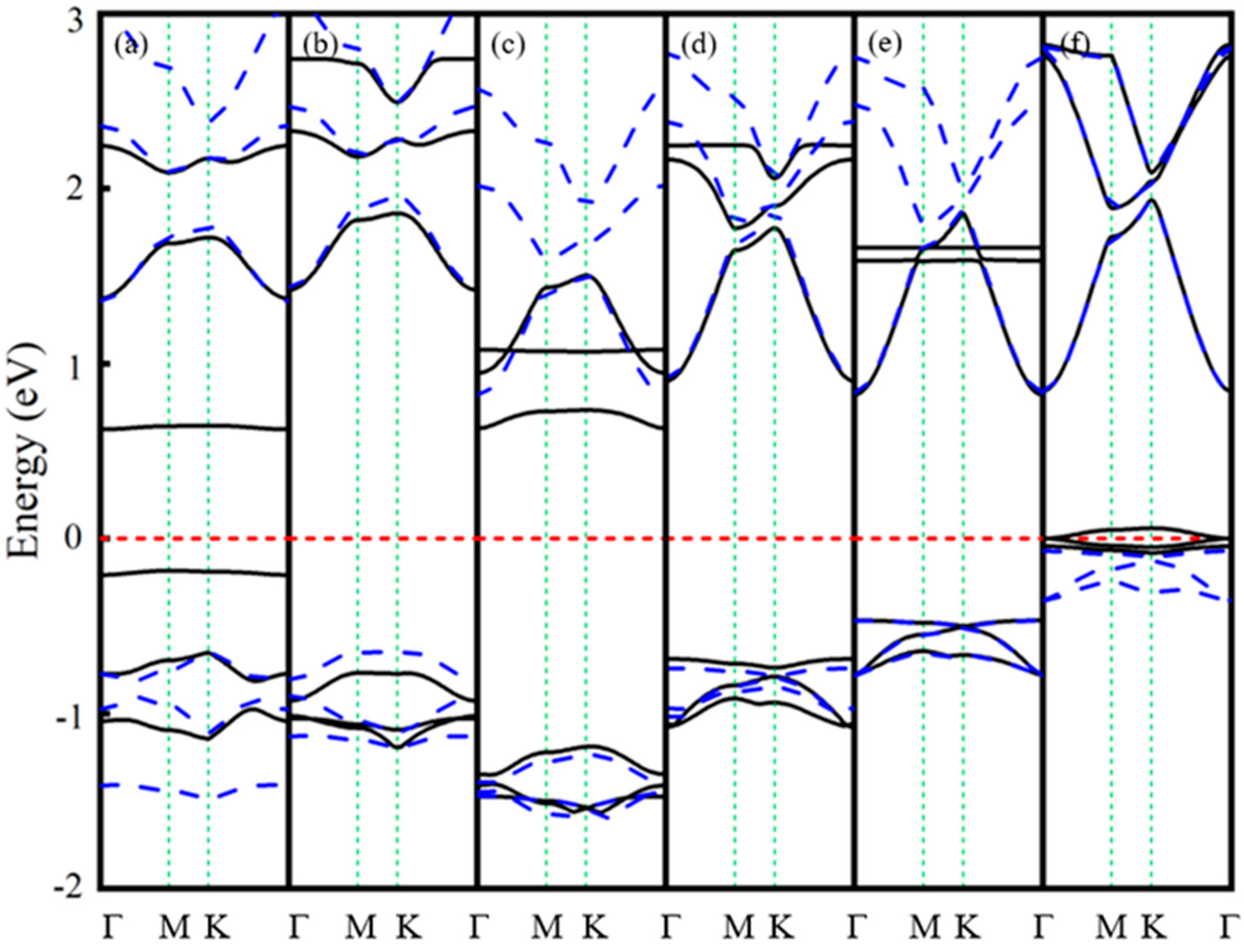
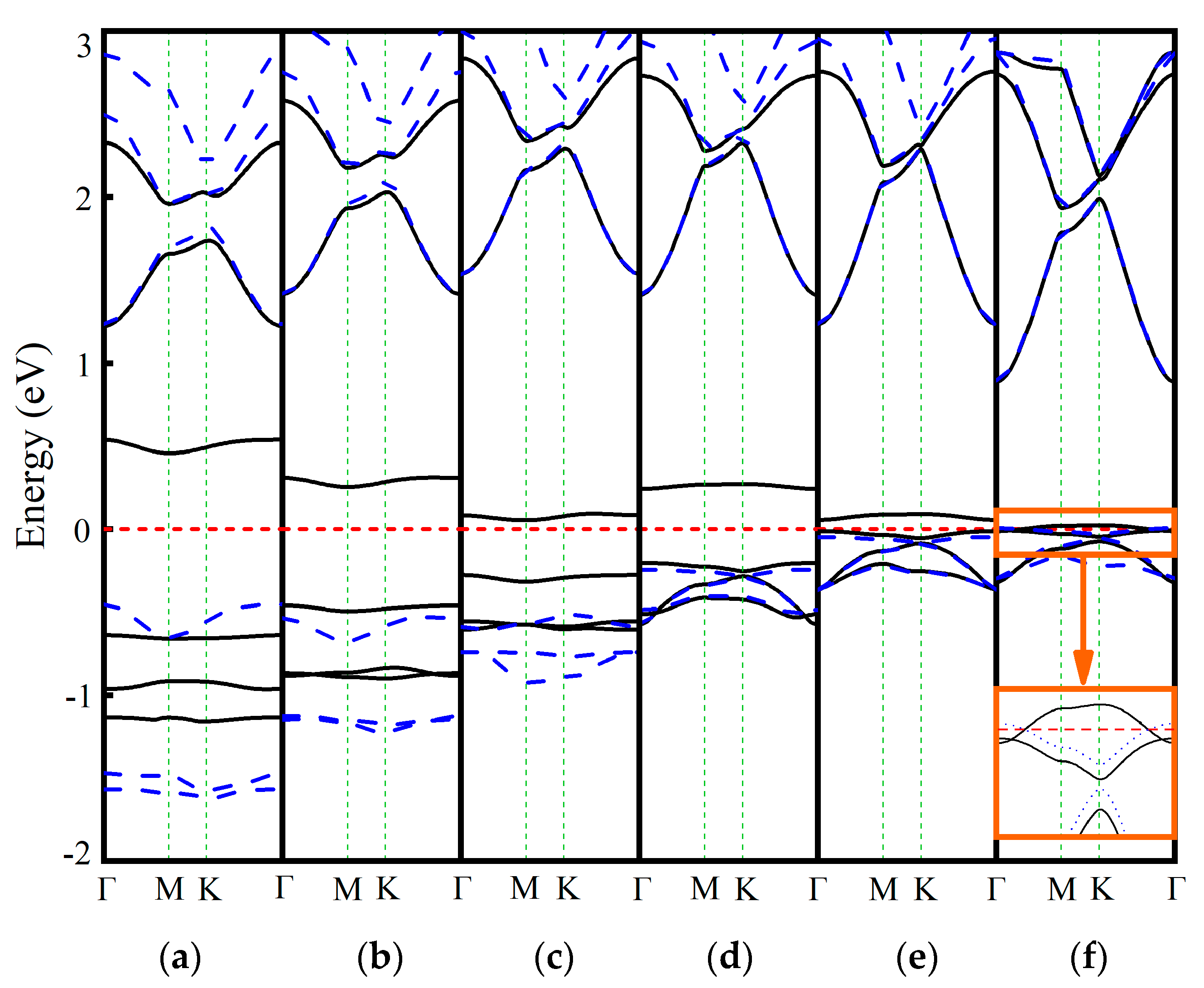
| Dopant | a (Å) | lLn-N (Å) | Fermi Level (eV) | Eb (eV) | Mtot (μB) | MLn (μB) | MN (μB) |
|---|---|---|---|---|---|---|---|
| None | 12.880 | 1.865 | −4.370 | / | 0 | 0 | 0 |
| La | 13.070 | 2.203 | −3.882 | −11.450 | 0 | 0 | 0 |
| Pr | 13.046 | 2.154 | −3.312 | −9.773 | 2.0 | 2.017 | −0.008 |
| Nd | 13.042 | 2.152 | −3.582 | −9.296 | 3.0 | 3.007 | −0.009 |
| Pm | 13.037 | 2.147 | −4.484 | −9.477 | 4.0 | 4.028 | −0.012 |
| Eu | 13.036 | 2.153 | −4.608 | −7.700 | 6.0 | 6.051 | −0.017 |
| Gd | 13.028 | 2.126 | −4.631 | −13.680 | 7.0 | 7.029 | −0.013 |
| Dopant | Ln | N1 | Ga1 | N2 |
|---|---|---|---|---|
| None | / | 1.457 | −1.457 | / |
| La | −1.946 | 1.561 | −1.433 | 1.457 |
| Pr | −1.820 | 1.526 | −1.440 | 1.457 |
| Nd | −1.818 | 1.534 | −1.444 | 1.455 |
| Pm | −1.828 | 1.536 | −1.442 | 1.458 |
| Eu | −1.870 | 1.566 | −1.450 | 1.457 |
| Gd | −1.861 | 1.551 | −1.442 | 1.458 |
| Dopant | −6% | −4% | −2% | 0 | 2% | 4% | 6% |
|---|---|---|---|---|---|---|---|
| La | 1.881 | 1.895 | 1.839 | 1.728 | 1.559 | 1.213 | 0.860 |
| Pr | 1.694 | 1.893 | 1.856 | 1.662 | 1.609 | 1.248 | 0.881 |
| Nd | 1.770 | 2.034 | 1.930 | 1.871 | 1.678 | 1.284 | 0.861 |
| Pm | 0.808 | 2.070 | 1.812 | 1.892 | 1.589 | 1.291 | / |
| Eu | 0.840 | 0.701 | 0.317 | 0.649 | 0.445 | 0.069 | / |
| Gd | 2.090 | 2.144 | 2.117 | 1.972 | 1.640 | 1.281 | 0.909 |
| Dopant | EPauli (eV) | Eelstat (eV) | Eorb (eV) | Eint (eV) |
|---|---|---|---|---|
| La | 32.258 | −21.639 | −23.743 | −13.124 |
| Pr | 29.557 | −20.334 | −21.188 | −11.965 |
| Nd | 28.984 | −20.199 | −19.992 | −11.208 |
| Pm | 28.478 | −19.858 | −19.371 | −10.751 |
| Eu | 26.670 | −18.653 | −17.274 | −9.257 |
| Gd | 29.930 | −20.424 | −22.308 | −12.802 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wen, X.; Lei, B.; Zhang, L.; Lu, H. Tuning of the Electronic and Magnetic Properties of GaN Monolayers via Doping with Lanthanide Atoms and by Applying Biaxial Strain. Nanomaterials 2025, 15, 1331. https://doi.org/10.3390/nano15171331
Wen X, Lei B, Zhang L, Lu H. Tuning of the Electronic and Magnetic Properties of GaN Monolayers via Doping with Lanthanide Atoms and by Applying Biaxial Strain. Nanomaterials. 2025; 15(17):1331. https://doi.org/10.3390/nano15171331
Chicago/Turabian StyleWen, Xue, Bocheng Lei, Lili Zhang, and Haiming Lu. 2025. "Tuning of the Electronic and Magnetic Properties of GaN Monolayers via Doping with Lanthanide Atoms and by Applying Biaxial Strain" Nanomaterials 15, no. 17: 1331. https://doi.org/10.3390/nano15171331
APA StyleWen, X., Lei, B., Zhang, L., & Lu, H. (2025). Tuning of the Electronic and Magnetic Properties of GaN Monolayers via Doping with Lanthanide Atoms and by Applying Biaxial Strain. Nanomaterials, 15(17), 1331. https://doi.org/10.3390/nano15171331






