Effect of the Microstructure of Carbon Supports on the Oxygen Reduction Properties of the Loaded Non-Noble Metal Catalysts
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of Carbon Precursors
2.2. Preparation of Catalysts
2.3. Physical and Chemical Property Analysis
2.4. Evaluation of the Electrochemical Performance
3. Results and Discussion
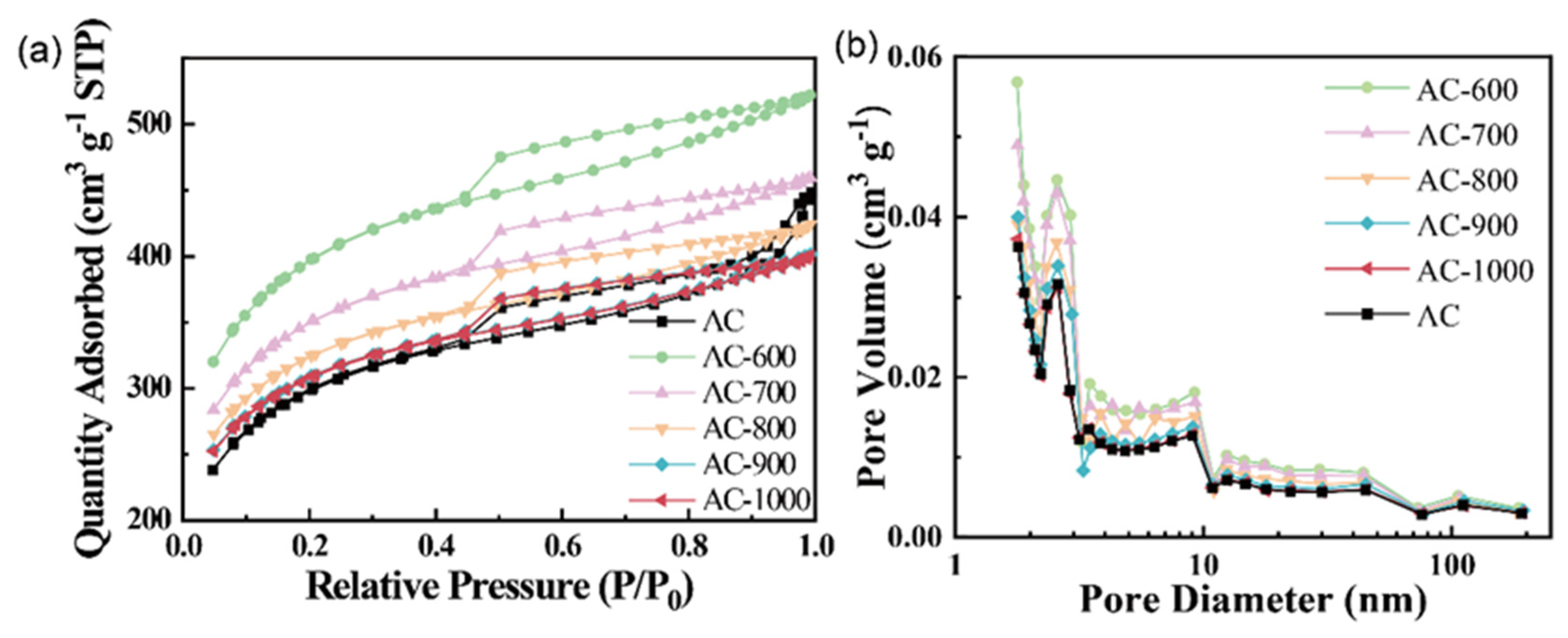
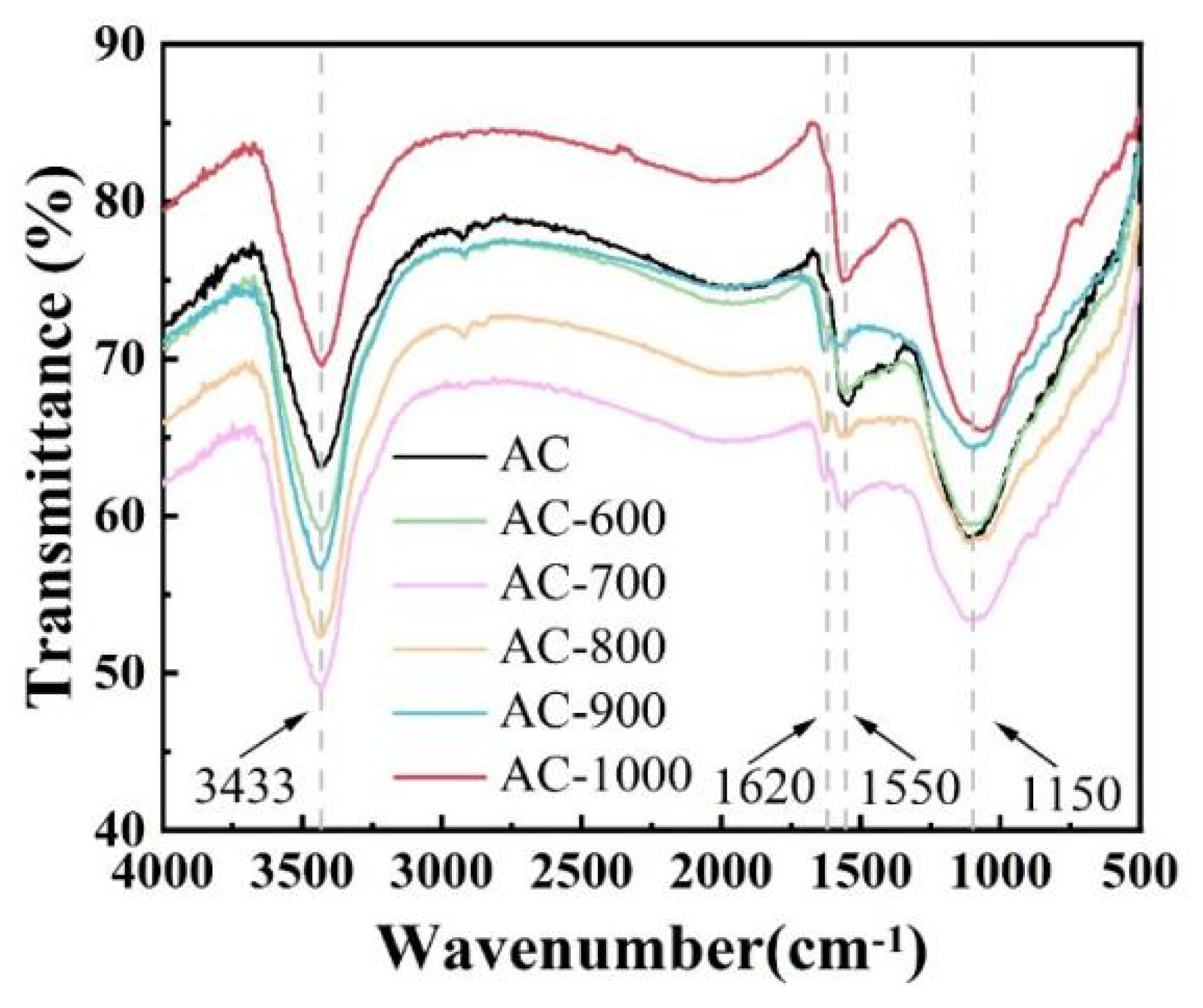
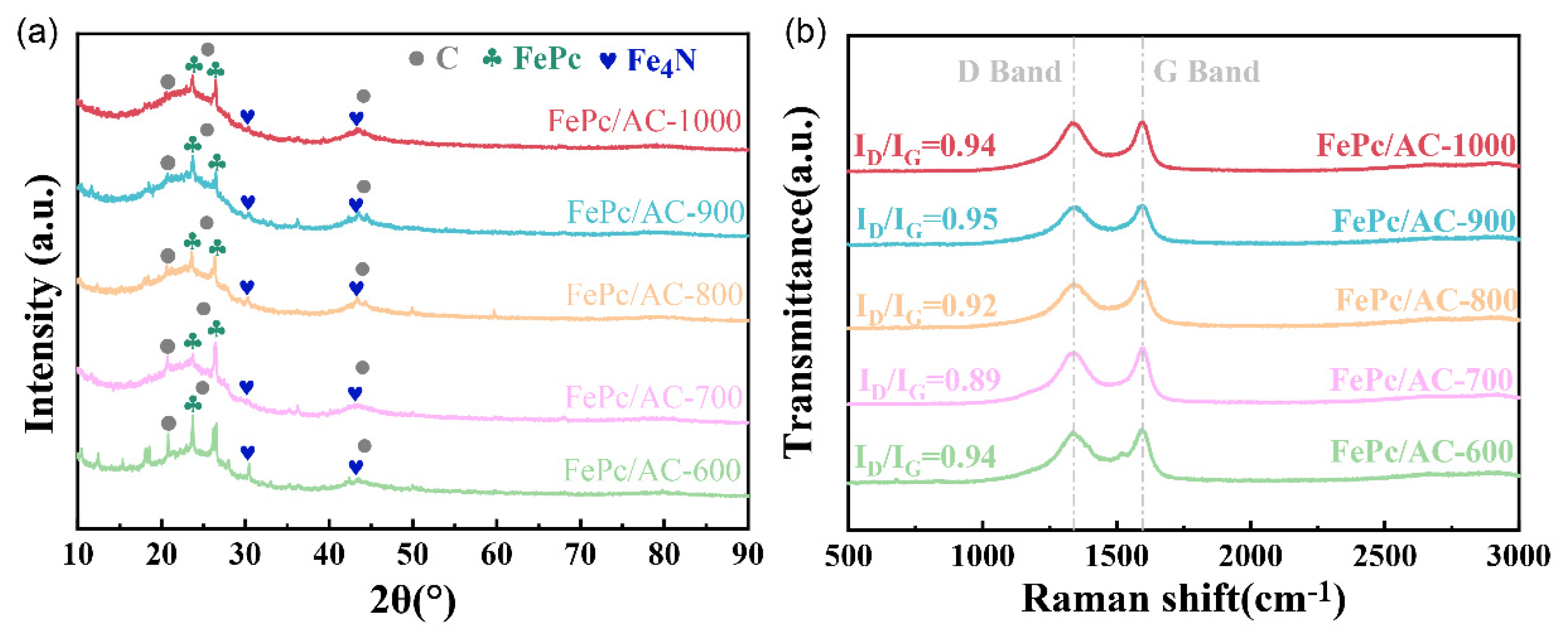
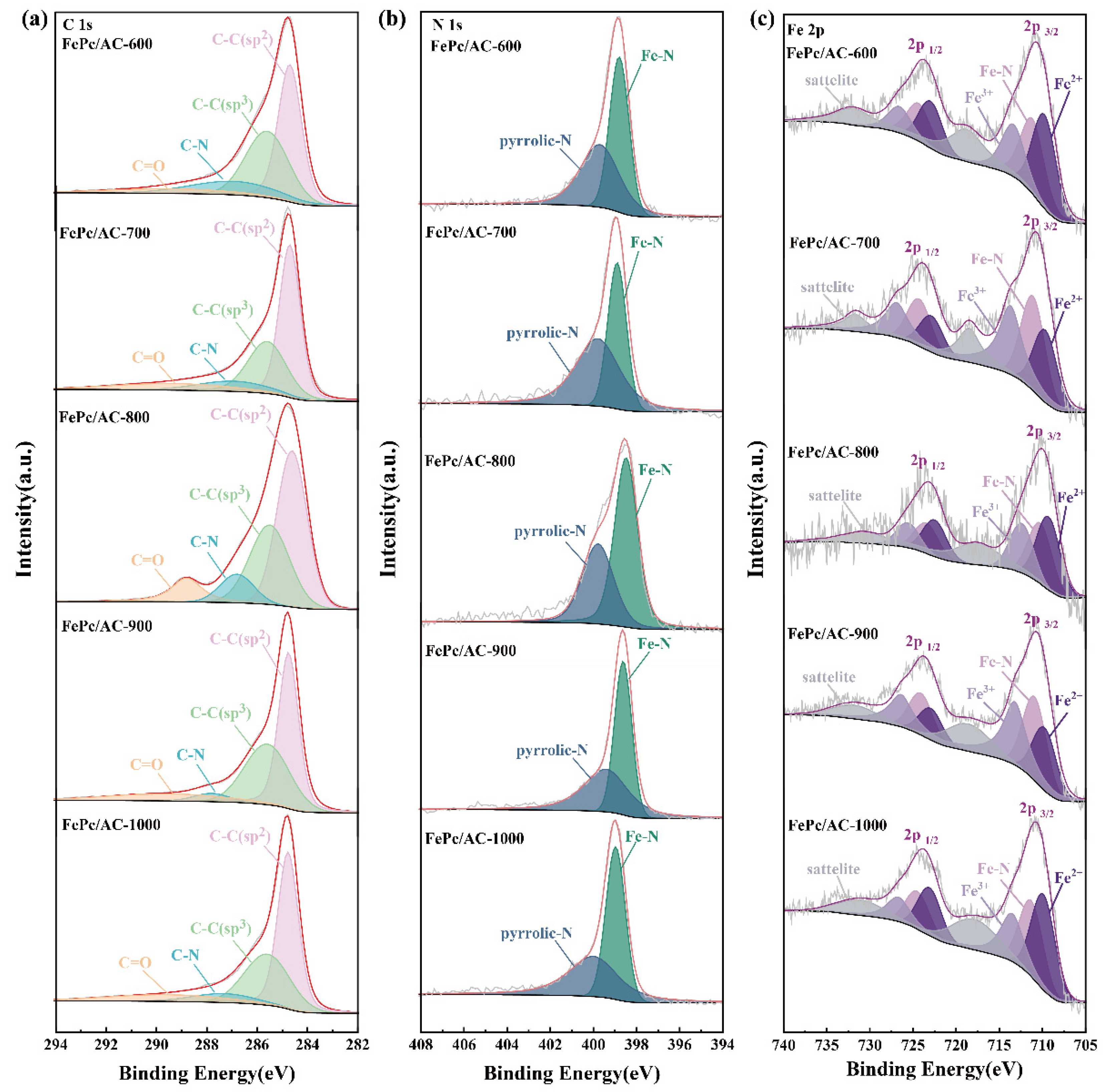
3.1. Physical and Chemical Properties of the Carbon Precursors and Catalysts
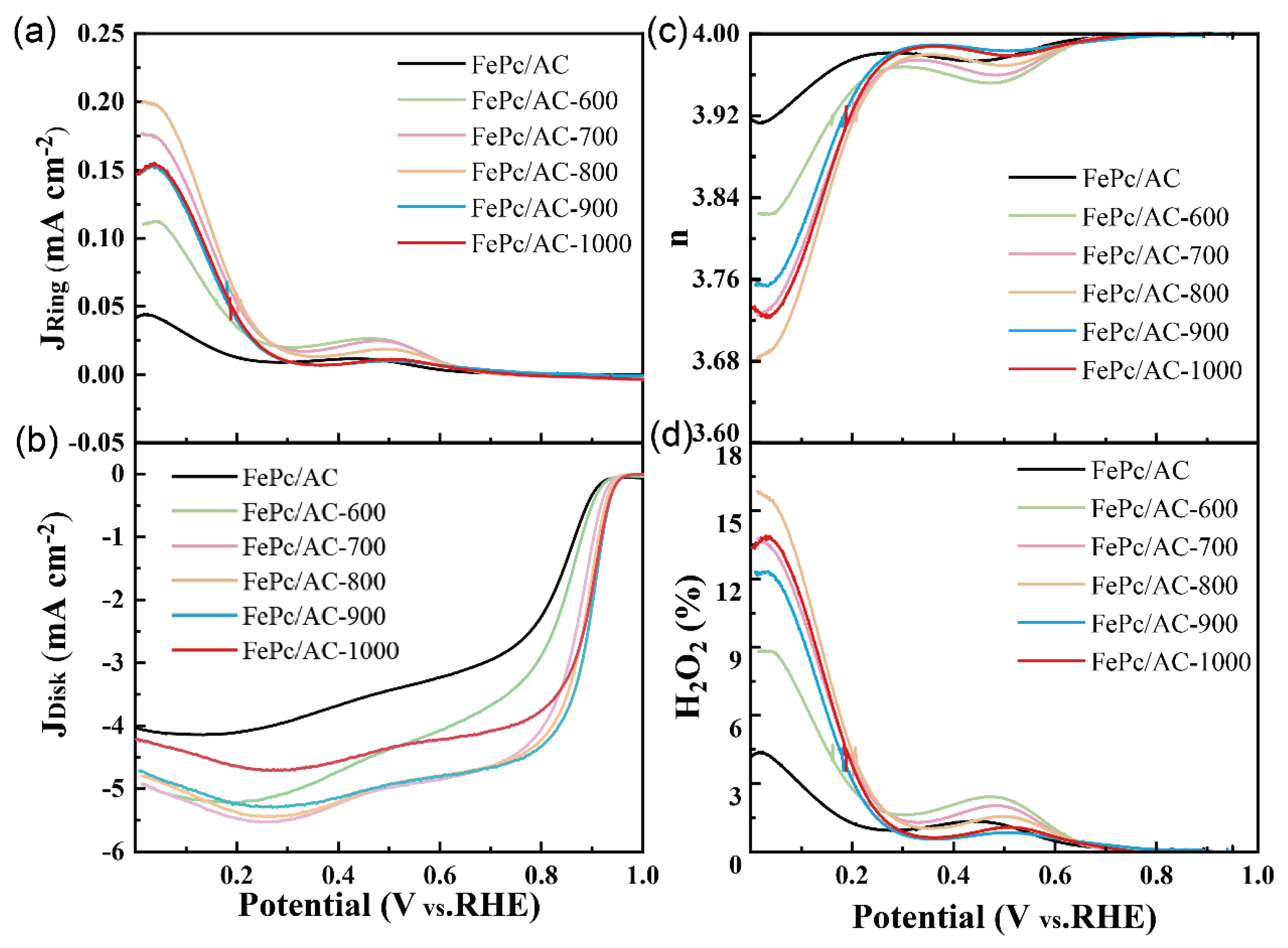
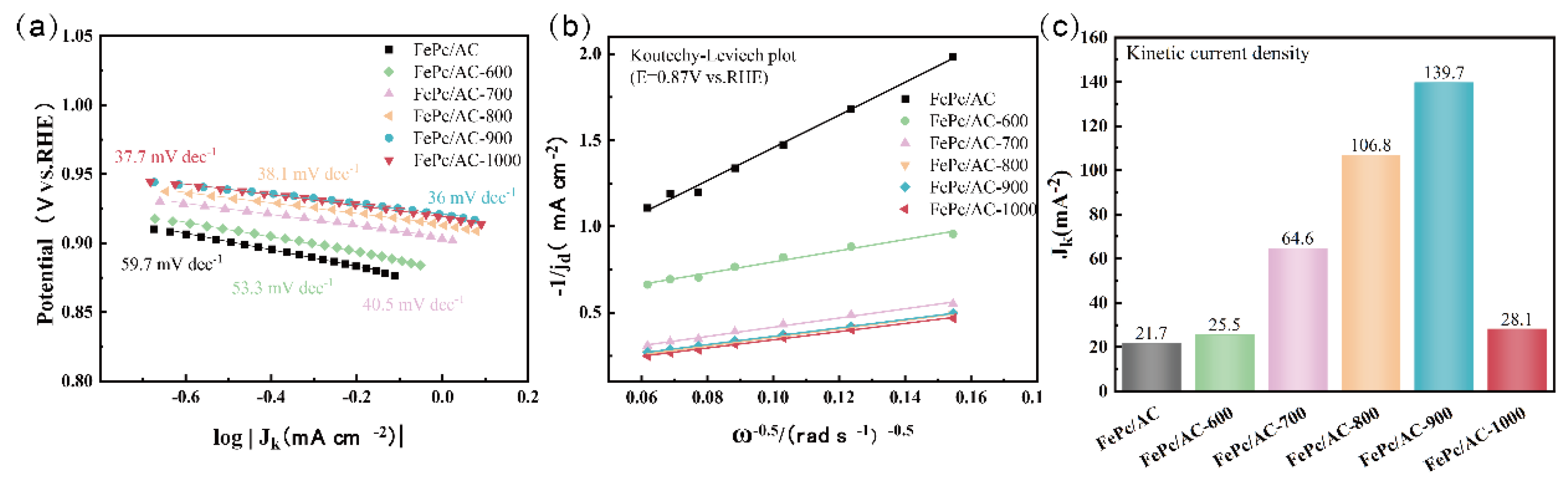
3.2. Electrochemical Properties of the Catalysts
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Tariq, A.H.; Kazmi, S.A.A.; Hassan, M.; Ali, S.; Anwar, M. Analysis of fuel cell integration with hybrid microgrid systems for clean energy: A comparative review. Int. J. Hydrogen Energy 2024, 52, 1005–1034. [Google Scholar] [CrossRef]
- Meda, U.S.; Rajyaguru, Y.V.; Pandey, A. Generation of green hydrogen using self-sustained regenerative fuel cells: Opportunities and challenges. Int. J. Hydrogen Energy 2023, 48, 28289–28314. [Google Scholar] [CrossRef]
- Janssen, M.; Weber, P.; Oezaslan, M. Recent advances of various Pt-based catalysts for oxygen reduction reaction (ORR) in polymer electrolyte membrane fuel cells (PEMFCs). Curr. Opin. Electrochem. 2023, 40, 101337. [Google Scholar] [CrossRef]
- Wang, H.; Gao, J.; Chen, C.; Zhao, W.; Zhang, Z.; Li, D.; Chen, Y.; Wang, C.; Zhu, C.; Ke, X. PtNi-W/C with atomically dispersed tungsten sites toward boosted ORR in proton exchange membrane fuel cell devices. Nano-Micro Lett. 2023, 15, 143. [Google Scholar] [CrossRef]
- Zhang, M.; Li, H.; Chen, J.; Ma, F.X.; Zhen, L.; Wen, Z.; Xu, C.Y. A Low-Cost, Durable Bifunctional Electrocatalyst Containing Atomic Co and Pt Species for Flow Alkali-Al/Acid Hybrid Fuel Cell and Zn–Air Battery. Adv. Funct. Mater. 2023, 33, 2303189. [Google Scholar] [CrossRef]
- Chen, Y.; Zhang, S.; Jung, J.C.-Y.; Zhang, J. Carbons as low-platinum catalyst supports and non-noble catalysts for polymer electrolyte fuel cells. Prog. Energy Combust. Sci. 2023, 98, 101101. [Google Scholar] [CrossRef]
- Wang, Y.; Hao, J.; Liu, Y.; Liu, M.; Sheng, K.; Wang, Y.; Yang, J.; Li, J.; Li, W. Recent advances in regulating the performance of acid oxygen reduction reaction on carbon-supported non-precious metal single atom catalysts. J. Energy Chem. 2023, 76, 601–616. [Google Scholar] [CrossRef]
- SBhoyate, D.; Kim, J.; de Souza, F.M.; Lin, J.; Lee, E.; Kumar, A.; Gupta, R.K. Science and engineering for non-noble-metal-based electrocatalysts to boost their ORR performance: A critical review. Coord. Chem. Rev. 2023, 474, 214854. [Google Scholar] [CrossRef]
- Sivaraman, R.; Opulencia, M.J.C.; Majdi, A.; Patra, I.; Abid, M.K.; Hammid, A.T.; Derakhshandeh, M. Design a promising non-precious electro-catalyst for oxygen reduction reaction in fuel cells. Int. J. Hydrogen Energy 2023, 48, 6308–6316. [Google Scholar] [CrossRef]
- Akula, S.; Mooste, M.; Kozlova, J.; Käärik, M.; Treshchalov, A.; Kikas, A.; Kisand, V.; Aruväli, J.; Paiste, P.; Tamm, A. Transition metal (Fe, Co, Mn, Cu) containing nitrogen-doped porous carbon as efficient oxygen reduction electrocatalysts for anion exchange membrane fuel cells. Chem. Eng. J. 2023, 458, 141468. [Google Scholar] [CrossRef]
- Shi, Z.; Yang, W.; Gu, Y.; Liao, T.; Sun, Z. Metal-nitrogen-doped carbon materials as highly efficient catalysts: Progress and rational design. Adv. Sci. 2020, 7, 2001069. [Google Scholar] [CrossRef]
- Yan, L.; Liu, A.; Ma, R.; Guo, C.; Ding, X.; Feng, P.; Jia, D.; Xu, M.; Ai, L.; Guo, N. Regulating the specific surface area and porous structure of carbon for high performance supercapacitors. Appl. Surf. Sci. 2023, 615, 156267. [Google Scholar] [CrossRef]
- Zhang, L.; Jiao, X.; He, G.; Shen, Z.; Wang, W. Iron phthalocyanine decorated porous biomass-derived carbon as highly effective electrocatalyst for oxygen reduction reaction. J. Environ. Chem. Eng. 2023, 11, 109676. [Google Scholar] [CrossRef]
- Chai, L.; Hu, Z.; Wang, X.; Zhang, L.; Li, T.-T.; Hu, Y.; Pan, J.; Qian, J.; Huang, S. Fe7C3 nanoparticles with in situ grown CNT on nitrogen doped hollow carbon cube with greatly enhanced conductivity and ORR performance for alkaline fuel cell. Carbon 2021, 174, 531–539. [Google Scholar] [CrossRef]
- Cui, X.; Liu, Y.; Han, G.; Cao, M.; Han, L.; Zhou, B.; Mehdi, S.; Wu, X.; Li, B.; Jiang, J. Wood-Derived Integral Air Electrode for Enhanced Interfacial Electrocatalysis in Rechargeable Zinc–Air Battery. Small 2021, 17, 2101607. [Google Scholar] [CrossRef]
- Meng, X.; Zhang, X.; Rageloa, J.; Liu, Z.; Wang, W. Coordination strategy to prepare high-performance Fe-Nx catalysts for Al-air batteries. J. Power Sources 2023, 567, 232988. [Google Scholar] [CrossRef]
- Kostuch, A.; Jarczewski, S.; Surówka, M.K.; Kuśtrowski, P.; Sojka, Z.; Kruczała, K. The joint effect of electrical conductivity and surface oxygen functionalities of carbon supports on the oxygen reduction reaction studied over bare supports and Mn–Co spinel/carbon catalysts in alkaline media. Catal. Sci. Technol. 2021, 11, 7578–7591. [Google Scholar] [CrossRef]
- Ibrahim, F.O.; Kisand, K.; Douglin, J.C.; Sarapuu, A.; Kikas, A.; Käärik, M.; Kozlova, J.; Aruväli, J.; Treshchalov, A.; Leis, J.; et al. Ionothermal synthesis of mesoporous FeNC electrocatalysts for high-performance anion-exchange membrane fuel cells. Chem. Eng. J. 2025, 510, 161560. [Google Scholar] [CrossRef]
- Sanetuntikul, J.; Shanmugam, S. High pressure pyrolyzed non-precious metal oxygen reduction catalysts for alkaline polymer electrolyte membrane fuel cells. Nanoscale 2015, 7, 7644–7650. [Google Scholar] [CrossRef] [PubMed]
- Moura, J.P.C.; Luchetti, L.E.; Fernandes, C.M.; Trench, A.B.; Lange, C.N.; Batista, B.L.; Almeida, J.M.; Santos, M.C. Experimental and theoretical studies of WO3/Vulcan XC-72 electrocatalyst enhanced H2O2 yield ORR performed in acid and alkaline medium. J. Environ. Chem. Eng. 2024, 12, 113182. [Google Scholar] [CrossRef]
- Mei, X.; Zhao, X.; Chen, Y.; Deng, B.; Geng, Q.; Cao, Y.; Li, Y.; Dong, F. Highly Efficient H2O2 Production via Two-Electron Electrochemical Oxygen Reduction over Fe-Doped CeO2. ACS Sustain. Chem. Eng. 2023, 11, 15609–15619. [Google Scholar] [CrossRef]
- Zhao, K.-M.; Wu, D.-X.; Wu, W.-K.; Nie, J.-B.; Geng, F.-S.; Li, G.; Shi, H.-Y.; Huang, S.-C.; Huang, H.; Zhang, J.; et al. Identifying high-spin hydroxyl-coordinated Fe3+N4 as the active centre for acidic oxygen reduction using molecular model catalysts. Nat. Catal. 2025, 8, 422–435. [Google Scholar] [CrossRef]
- Sharifzadeh, Z.; Morsali, A. Defect engineering enables synergistic action of hierarchical porosity and multifunctionality for efficient catalysis of diverse reactions. Appl. Mater. Today 2023, 35, 101971. [Google Scholar] [CrossRef]
- Islam, M.M.U.; Li, J.; Roychand, R.; Saberian, M. Microstructure, thermal conductivity and carbonation resistance properties of sustainable structural lightweight concrete incorporating 100% coarser rubber particles. Constr. Build. Mater. 2023, 408, 133658. [Google Scholar] [CrossRef]
- Ding, R.; Wang, Y.Q.; Zeng, F.R.; Liu, B.W.; Wang, Y.Z.; Zhao, H.B. One-Step Self-Flowering Method toward Programmable Ultrathin Porous Carbon-Based Materials for Microwave Absorption and Hydrogen Evolution. Small 2023, 19, 2302132. [Google Scholar] [CrossRef] [PubMed]
- Calzaferri, G.; Gallagher, S.H.; Lustenberger, S.; Walther, F.; Brühwiler, D. Multiple equilibria description of type H1 hysteresis in gas sorption isotherms of mesoporous materials. Mater. Chem. Phys. 2023, 296, 127121. [Google Scholar] [CrossRef]
- Shao, Z.; Tan, B.; Su, C.; Zan, X.; Song, X.; Wang, H. Study on the variation in key functional groups of coal and the mechanism of CO and CO2 generation under the thermal effect of lean-oxygen conditions. Fuel 2024, 357, 130061. [Google Scholar] [CrossRef]
- Chen, B.; Wu, D.; Wang, T.; Yuan, F.; Jia, D. Rapid preparation of porous carbon by flame burning carbonization method for supercapacitor. Chem. Eng. J. 2023, 462, 142163. [Google Scholar] [CrossRef]
- Li, M.; Ye, Q.; Hou, S.; Yang, J.; Chi, B.; Deng, Y.; Tian, X. Fluorine and phosphorus atoms cooperated on an N-doped 3D porous carbon network for enhanced ORR performance toward the zinc–air batteries. J. Mater. Chem. A 2023, 11, 8730–8738. [Google Scholar] [CrossRef]
- Qiang, H.; Shi, M.; Wang, F.; Xia, M. Green synthesis of high N-doped hierarchical porous carbon nanogranules with ultra-high specific surface area and porosity for capacitive deionization. Sep. Purif. Technol. 2023, 308, 122918. [Google Scholar] [CrossRef]
- Liu, W.; Zhang, H.; Ye, W.; Xiao, B.; Sun, Z.; Cheng, Y.; Wang, M.S. Regulating the wettability of hard carbon through open mesochannels for enhanced K+ storage. Small 2023, 19, 2300605. [Google Scholar] [CrossRef]
- Hu, C.; Liang, Q.; Yang, Y.; Peng, Q.; Luo, Z.; Dong, J.; Isimjan, T.T.; Yang, X. Conductivity-enhanced porous N/P co-doped metal-free carbon significantly enhances oxygen reduction kinetics for aqueous/flexible zinc-air batteries. J. Colloid Interface Sci. 2023, 633, 500–510. [Google Scholar] [CrossRef]
- Elkady, M.; Shokry, H.; Hamad, H. New activated carbon from mine coal for adsorption of dye in simulated water or multiple heavy metals in real wastewater. Materials 2020, 13, 2498. [Google Scholar] [CrossRef]
- Errico, S.; Moggio, M.; Diano, N.; Portaccio, M.; Lepore, M. Different experimental approaches for Fourier-transform infrared spectroscopy applications in biology and biotechnology: A selected choice of representative results. Biotechnol. Appl. Biochem. 2023, 70, 937–961. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Cheon, J.Y.; Shin, T.J.; Park, J.Y.; Joo, S.H. Effect of surface oxygen functionalization of carbon support on the activity and durability of Pt/C catalysts for the oxygen reduction reaction. Carbon 2016, 101, 449–457. [Google Scholar] [CrossRef]
- Luo, X.; Zheng, H.; Lai, W.; Yuan, P.; Li, S.; Li, D.; Chen, Y. Defect engineering of carbons for energy conversion and storage applications. Energy Environ. Mater. 2023, 6, e12402. [Google Scholar] [CrossRef]
- Rocha, R.P.; Pereira, M.F.R.; Figueiredo, J.L. Characterisation of the surface chemistry of carbon materials by temperature-programmed desorption: An assessment. Catal. Today 2023, 418, 114136. [Google Scholar] [CrossRef]
- Long, J.; He, W.; Liao, H.; Ye, W.; Dou, H.; Zhang, X. In Situ Prepared Three-Dimensional Covalent and Hydrogen Bond Synergistic Binder to Boost the Performance of SiO x Anodes for Lithium-Ion Batteries. ACS Appl. Mater. Interfaces 2023, 15, 10726–10734. [Google Scholar] [CrossRef] [PubMed]
- Liang, B.; Guo, S.; Zhao, Y.; Khan, I.U.; Zhang, X.; Li, K.; Lv, C. Single iron atoms anchored on activated carbon as active centres for highly efficient oxygen reduction reaction in air-cathode microbial fuel cell. J. Power Sources 2020, 450, 227683. [Google Scholar] [CrossRef]
- Lu, X.; Yang, P.; Xu, H.; Xiao, L.; Liu, L.; Li, R.; Alekseeva, E.; Zhang, J.; Levin, O.; An, M. Biomass derived robust Fe4N active sites supported on porous carbons as oxygen reduction reaction catalysts for durable Zn–air batteries. J. Mater. Chem. A 2023, 11, 3725–3734. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, J.; Yang, W.; Zhang, J.; Fu, Q.; Song, Y.-C.; Wei, Z.; Liao, Q.; Zhu, X. Fe–N-doped carbon nanoparticles from coal tar soot and its novel application as a high performance air-cathode catalyst for microbial fuel cells. Electrochim. Acta 2020, 363, 137177. [Google Scholar] [CrossRef]
- Schmidt, K.; Laumer, J.; O’Leary, S.K. The D-and G-branches associated with the Raman spectrum of thin-film carbon. Solid State Commun. 2023, 360, 115041. [Google Scholar] [CrossRef]
- Hu, S.; Ni, W.; Yang, D.; Ma, C.; Zhang, J.; Duan, J.; Gao, Y.; Zhang, S. Fe3O4 nanoparticles encapsulated in single-atom Fe–N–C towards efficient oxygen reduction reaction: Effect of the micro and macro pores. Carbon 2020, 162, 245–255. [Google Scholar] [CrossRef]
- Santos, A.C.; Badenhorst, C.; Białecka, B.; Cameán, I.; Guedes, A.; Moreira, K.; Predeanu, G.; Suárez-Ruiz, I.; Wagner, N.; Valentim, B. Graphitization: Microstructural and microtextural transformations of residual char from international coal combustion ash. Int. J. Coal Geol. 2024, 285, 104470. [Google Scholar] [CrossRef]
- Ruggiero, B.N.; Gutierrez, K.M.S.; George, J.D.; Mangan, N.M.; Notestein, J.M.; Seitz, L.C. Probing the relationship between bulk and local environments to understand impacts on electrocatalytic oxygen reduction reaction. J. Catal. 2022, 414, 33–43. [Google Scholar] [CrossRef]
- Zhu, S.; Huang, Y.; Yu, T.; Lei, Y.; Zhu, X.; Yang, T.; Gu, J.; Wang, C. Enhance the durability of the oxygen reduction reaction catalyst through the synergy of improved graphitization and nitrogen doping of carbon carrier. Int. J. Hydrogen Energy 2024, 51, 956–965. [Google Scholar] [CrossRef]
- Xu, W.; Dang, R.; Zhou, L.; Yang, Y.; Lin, T.; Guo, Q.; Xie, F.; Hu, Z.; Ding, F.; Liu, Y. Conversion of surface residual alkali to solid electrolyte to enable Na-ion full cells with robust interfaces. Adv. Mater. 2023, 35, 2301314. [Google Scholar] [CrossRef]
- de Lima Tinoco, M.V.; Fujii, L.R.; Nicoliche, C.Y.; Giordano, G.F.; Barbosa, J.A.; da Rocha, J.F.; Santos, G.T.D.; Bettini, J.; Santhiago, M.; Strauss, M.; et al. Scalable and green formation of graphitic nanolayers produces highly conductive pyrolyzed paper toward sensitive electrochemical sensors. Nanoscale 2023, 15, 6201–6214. [Google Scholar] [CrossRef]
- Gu, Y.; Fan, C.; Hao, X.; Hu, F.; Zhang, C.; Yang, H.; Li, C.M.; Guo, C. Oxidase mimic graphdiyne for efficient superoxide generation in wide pH ranges. Adv. Funct. Mater. 2022, 32, 2110192. [Google Scholar] [CrossRef]
- Zhong, D.; Liu, P.; Liu, Y.; Xu, Y. Nitrogen-doped Zn/Fe@ PCN derived from metal–organic frameworks activating persulfate to efficiently degrade rhodamine B. Environ. Sci. Pollut. Res. 2024, 31, 2228–2242. [Google Scholar] [CrossRef]
- Wu, L.; Zhao, R.; Du, G.; Wang, H.; Hou, M.; Zhang, W.; Sun, P.; Chen, T. Hierarchically porous Fe/N/S/C nanospheres with high-content of Fe-Nx for enhanced ORR and Zn-air battery performance. Green Energy Environ. 2023, 8, 1693–1702. [Google Scholar] [CrossRef]
- Niu, L.; Zhao, J.; Chen, X.; Wang, G.; Zhang, W.; Wang, X. P-doping modulates the local electronic structure of Pt atoms to enhance intrinsic ORR activity. Mol. Catal. 2024, 557, 113997. [Google Scholar] [CrossRef]
- Zou, Y.; Li, J.; Peng, Q.; Liu, Z.; Fu, Q.; Zhang, L.; Liao, Q.; Zhu, X. Tuning the wettability of advanced mesoporous FeNC catalysts for optimizing the construction of the gas/liquid/solid three-phase interface in air-cathodes. Chem. Eng. J. 2022, 450, 138342. [Google Scholar] [CrossRef]
- Wang, Q.; Yang, Y.; Sun, F.; Chen, G.; Wang, J.; Peng, L.; Chen, W.-T.; Shang, L.; Zhao, J.; Sun-Waterhouse, D.; et al. Molten NaCl-assisted synthesis of porous Fe-N-C electrocatalysts with a high density of catalytically accessible FeN4 active sites and outstanding oxygen reduction reaction performance. Adv. Energy Mater. 2021, 11, 2100219. [Google Scholar] [CrossRef]
- Li, J.; Chen, S.; Yang, N.; Deng, M.; Ibraheem, S.; Deng, J.; Li, J.; Li, L.; Wei, Z. Ultrahigh-loading zinc single-atom catalyst for highly efficient oxygen reduction in both acidic and alkaline media. Angew. Chem. 2019, 131, 7109–7113. [Google Scholar] [CrossRef]



| 1 MPa | 2 MPa | 3 MPa | ||
|---|---|---|---|---|
| AC | 8.87 × 10−4 | 1.36 × 10−3 | 1.62 × 10−3 | |
| Electrical conductivity S/m | AC-600 | 1.87 × 10−2 | 2.62 × 10−2 | 3.01 × 10−2 |
| AC-700 | 2.64 × 10 | 3.47 × 102 | 4.22 × 102 | |
| AC-800 | 5.59 × 103 | 8.23 × 103 | 9.84 × 103 | |
| AC-900 | 1.52 × 104 | 2.37 × 104 | 2.86 × 104 | |
| AC-1000 | 2.44 × 104 | 3.28 × 104 | 3.68 × 104 | |
| Sample | Eon (V vs. RHE) | E1/2 (V vs. RHE) | JL at 0.4 V (mA cm−2) | H2O2% at 0.4 V (%) | n at 0.4 V |
|---|---|---|---|---|---|
| FePc/AC | 0.92 | 0.81 | 3.67 | 1.25 | 3.972 |
| FePc/AC-600 | 0.93 | 0.83 | 4.72 | 1.72 | 3.958 |
| FePc/AC-700 | 0.94 | 0.87 | 5.23 | 1.51 | 3.965 |
| FePc/AC-800 | 0.95 | 0.88 | 5.20 | 1.22 | 3.975 |
| FePc/AC-900 | 0.95 | 0.89 | 5.13 | 0.73 | 3.991 |
| FePc/AC-1000 | 0.95 | 0.89 | 4.56 | 0.75 | 3.988 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ma, D.; Zhang, Y.; Liang, M.; Niu, R.; Ge, Y.; Zou, Y.; Dong, X. Effect of the Microstructure of Carbon Supports on the Oxygen Reduction Properties of the Loaded Non-Noble Metal Catalysts. Nanomaterials 2025, 15, 1327. https://doi.org/10.3390/nano15171327
Ma D, Zhang Y, Liang M, Niu R, Ge Y, Zou Y, Dong X. Effect of the Microstructure of Carbon Supports on the Oxygen Reduction Properties of the Loaded Non-Noble Metal Catalysts. Nanomaterials. 2025; 15(17):1327. https://doi.org/10.3390/nano15171327
Chicago/Turabian StyleMa, Dan, Yudong Zhang, Menghan Liang, Runyu Niu, Yao Ge, Yanan Zou, and Xiaorui Dong. 2025. "Effect of the Microstructure of Carbon Supports on the Oxygen Reduction Properties of the Loaded Non-Noble Metal Catalysts" Nanomaterials 15, no. 17: 1327. https://doi.org/10.3390/nano15171327
APA StyleMa, D., Zhang, Y., Liang, M., Niu, R., Ge, Y., Zou, Y., & Dong, X. (2025). Effect of the Microstructure of Carbon Supports on the Oxygen Reduction Properties of the Loaded Non-Noble Metal Catalysts. Nanomaterials, 15(17), 1327. https://doi.org/10.3390/nano15171327






