Applications of Nanoparticles in the Diagnosis and Treatment of Ovarian Cancer
Abstract
1. Introduction
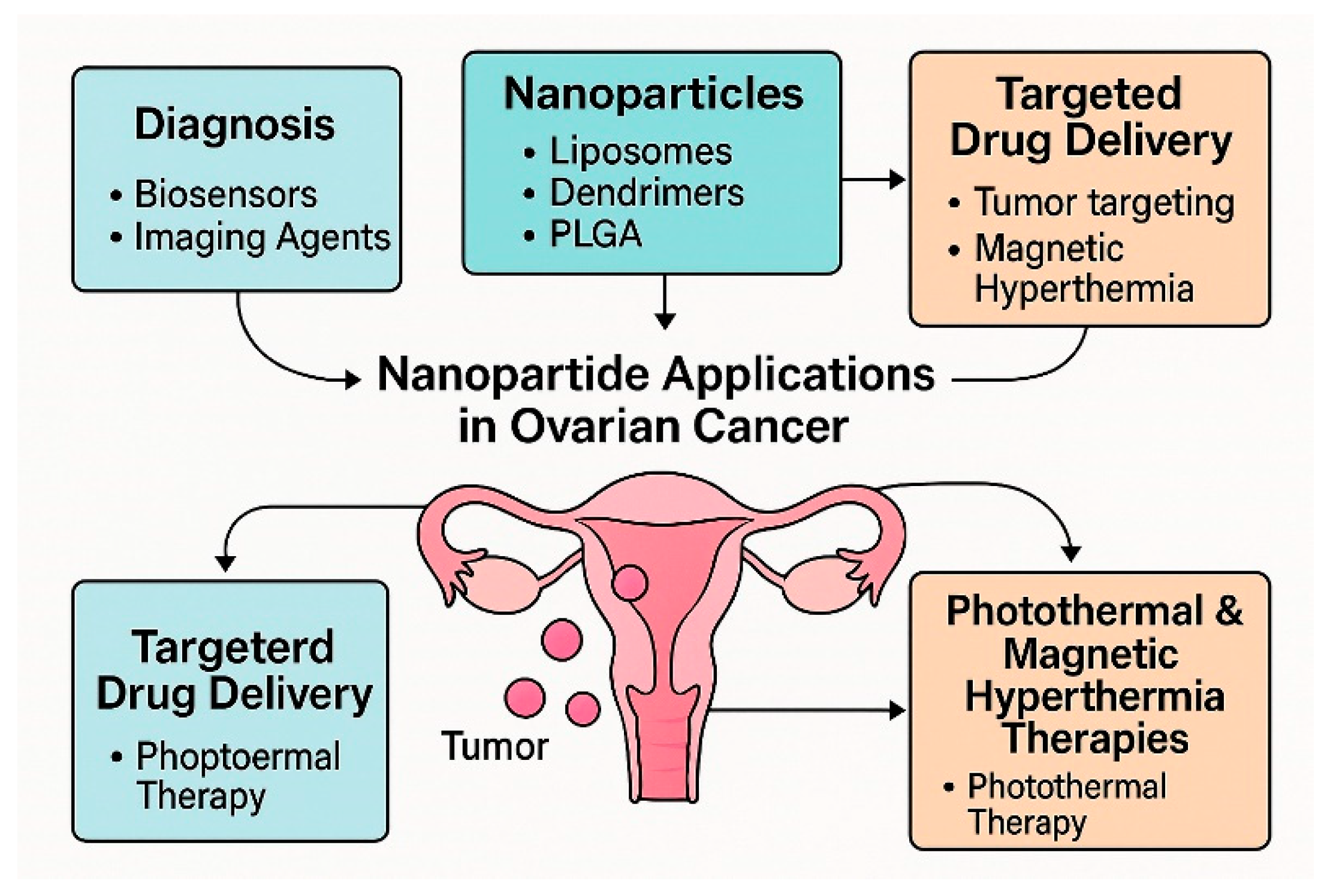
2. Types of Nanoparticles
3. Applications of Nanoparticles in Ovarian Cancer Diagnostics
4. Types of Nanoparticles Used in the Study on Ovarian Cancer Treatment
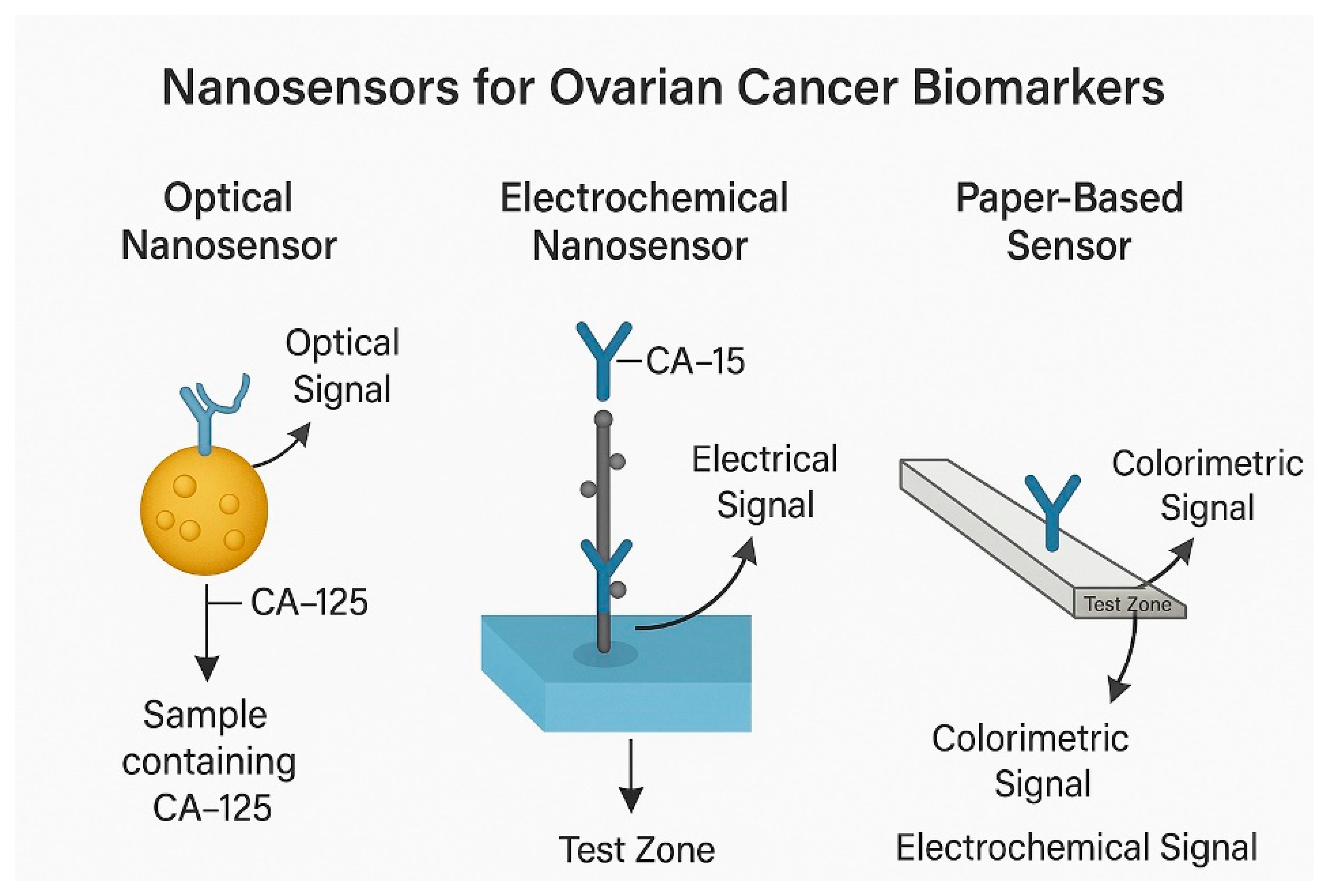
4.1. Optical Nanosensors
4.2. Electrochemical Nanosensors
4.3. Magnetoresistive and Paper-Based Biosensors
5. Nanotherapy-Drug Carriers Based on Nanoparticles
6. Benefits and Challenges of Nanoparticle Application (Figure 6)

7. Summary
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bray, F.; Laversanne, M.; Sung, H.; Ferlay, J.; Siegel, R.L.; Soerjomataram, I.; Jemal, A. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA A Cancer J. Clin. 2024, 74, 229–263. [Google Scholar] [CrossRef] [PubMed]
- Al-Azri, M.H. Delay in Cancer Diagnosis: Causes and Possible Solutions. Oman Med. J. 2016, 31, 325–326. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Tao, L.; Qiu, J.; Xu, J.; Yang, X.; Zhang, Y.; Tian, X.; Guan, X.; Cen, X.; Zhao, Y. Tumor biomarkers for diagnosis, prognosis and targeted therapy. Signal Transduct. Target. Ther. 2024, 9, 132. [Google Scholar] [CrossRef]
- Momenimovahed, Z.; Tiznobaik, A.; Taheri, S.; Salehiniya, H. Ovarian cancer in the world: Epidemiology and risk factors. Int. J. Womens Health 2019, 11, 287–299. [Google Scholar] [CrossRef]
- Zamwar, U.M.; Anjankar, A.P. Aetiology, Epidemiology, Histopathology, Classification, Detailed Evaluation, and Treatment of Ovarian Cancer. Cureus 2022, 14, e30561. [Google Scholar] [CrossRef]
- Ghisoni, E.; Imbimbo, M.; Zimmermann, S.; Valabrega, G. Ovarian Cancer Immunotherapy: Turning up the Heat. Int. J. Mol. Sci. 2019, 20, 2927. [Google Scholar] [CrossRef]
- Liberto, J.M.; Chen, S.Y.; Shih, I.M.; Wang, T.H.; Wang, T.L.; Pisanic, T.R., 2nd. Current and Emerging Methods for Ovarian Cancer Screening and Diagnostics: A Comprehensive Review. Cancers 2022, 14, 2885. [Google Scholar] [CrossRef]
- Mathieu, K.B.; Bedi, D.G.; Thrower, S.L.; Qayyum, A.; Bast, R.C., Jr. Screening for ovarian cancer: Imaging challenges and opportunities for improvement. Ultrasound Obs. Gynecol. 2018, 51, 293–303. [Google Scholar] [CrossRef]
- Li, X.; Li, Z.; Ma, H.; Li, X.; Zhai, H.; Li, X.; Cheng, X.; Zhao, X.; Zhao, Z.; Hao, Z. Ovarian cancer: Diagnosis and treatment strategies (Review). Oncol. Lett. 2024, 28, 441. [Google Scholar] [CrossRef] [PubMed]
- Tavares, V.; Marques, I.S.; Melo, I.G.; Assis, J.; Pereira, D.; Medeiros, R. Paradigm Shift: A Comprehensive Review of Ovarian Cancer Management in an Era of Advancements. Int. J. Mol. Sci. 2024, 25, 1845. [Google Scholar] [CrossRef]
- Dessale, M.; Mengistu, G.; Mengist, H.M. Nanotechnology: A Promising Approach for Cancer Diagnosis, Therapeutics and Theragnosis. Int. J. Nanomed. 2022, 17, 3735–3749. [Google Scholar] [CrossRef]
- Wang, B.; Hu, S.; Teng, Y.; Chen, J.; Wang, H.; Xu, Y.; Wang, K.; Xu, J.; Cheng, Y.; Gao, X. Current advance of nanotechnology in diagnosis and treatment for malignant tumors. Signal Transduct. Target. Ther. 2024, 9, 200. [Google Scholar] [CrossRef]
- Singh, S.; Arkoti, N.K.; Verma, V.; Pal, K. Nanomaterials and Their Distinguishing Features. In Nanomaterials for Advanced Technologies; Katiyar, J.K., Panwar, V., Ahlawat, N., Eds.; Springer Nature Singapore: Singapore, 2022; pp. 1–18. [Google Scholar]
- Baranwal, J.; Barse, B.; Di Petrillo, A.; Gatto, G.; Pilia, L.; Kumar, A. Nanoparticles in Cancer Diagnosis and Treatment. Materials 2023, 16, 5354. [Google Scholar] [CrossRef]
- Sun, L.; Liu, H.; Ye, Y.; Lei, Y.; Islam, R.; Tan, S.; Tong, R.; Miao, Y.-B.; Cai, L. Smart nanoparticles for cancer therapy. Signal Transduct. Target. Ther. 2023, 8, 418. [Google Scholar] [CrossRef]
- Henderson, E.; Huynh, G.; Wilson, K.; Plebanski, M.; Corrie, S. The Development of Nanoparticles for the Detection and Imaging of Ovarian Cancers. Biomedicines 2021, 9, 1554. [Google Scholar] [CrossRef] [PubMed]
- Tran, L.H.; Graulus, G.J.; Vincke, C.; Smiejkowska, N.; Kindt, A.; Devoogdt, N.; Muyldermans, S.; Adriaensens, P.; Guedens, W. Nanobodies for the Early Detection of Ovarian Cancer. Int. J. Mol. Sci. 2022, 23, 13687. [Google Scholar] [CrossRef]
- Joudeh, N.; Linke, D. Nanoparticle classification, physicochemical properties, characterization, and applications: A comprehensive review for biologists. J. Nanobiotechnol. 2022, 20, 262. [Google Scholar] [CrossRef] [PubMed]
- Khan, I.; Saeed, K.; Khan, I. Nanoparticles: Properties, applications and toxicities. Arab. J. Chem. 2019, 12, 908–931. [Google Scholar] [CrossRef]
- Yetisgin, A.A.; Cetinel, S.; Zuvin, M.; Kosar, A.; Kutlu, O. Therapeutic Nanoparticles and Their Targeted Delivery Applications. Molecules 2020, 25, 2193. [Google Scholar] [CrossRef] [PubMed]
- Edis, Z.; Wang, J.; Waqas, M.K.; Ijaz, M.; Ijaz, M. Nanocarriers-Mediated Drug Delivery Systems for Anticancer Agents: An Overview and Perspectives. Int. J. Nanomed. 2021, 16, 1313–1330. [Google Scholar] [CrossRef]
- He, Y.; Zhang, S.; She, Y.; Liu, Z.; Zhu, Y.; Cheng, Q.; Ji, X. Innovative utilization of cell membrane-coated nanoparticles in precision cancer therapy. Exploration 2024, 4, 20230164. [Google Scholar] [CrossRef]
- Peng, X.; Fang, J.; Lou, C.; Yang, L.; Shan, S.; Wang, Z.; Chen, Y.; Li, H.; Li, X. Engineered nanoparticles for precise targeted drug delivery and enhanced therapeutic efficacy in cancer immunotherapy. Acta Pharm. Sin. B 2024, 14, 3432–3456. [Google Scholar] [CrossRef]
- Kumari, M.; Acharya, A.; Krishnamurthy, P.T. Antibody-conjugated nanoparticles for target-specific drug delivery of chemotherapeutics. Beilstein J. Nanotechnol. 2023, 14, 912–926. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Zhou, H.; Tan, L.; Siu, K.T.H.; Guan, X.-Y. Exploring treatment options in cancer: Tumor treatment strategies. Signal Transduct. Target. Ther. 2024, 9, 175. [Google Scholar] [CrossRef] [PubMed]
- Debela, D.T.; Muzazu, S.G.; Heraro, K.D.; Ndalama, M.T.; Mesele, B.W.; Haile, D.C.; Kitui, S.K.; Manyazewal, T. New approaches and procedures for cancer treatment: Current perspectives. SAGE Open Med. 2021, 9, 20503121211034366. [Google Scholar] [CrossRef]
- Allen, T.M.; Cullis, P.R. Liposomal drug delivery systems: From concept to clinical applications. Adv. Drug Deliv. Rev. 2013, 65, 36–48. [Google Scholar] [CrossRef]
- Afzal, O.; Altamimi, A.S.A.; Nadeem, M.S.; Alzarea, S.I.; Almalki, W.H.; Tariq, A.; Mubeen, B.; Murtaza, B.N.; Iftikhar, S.; Riaz, N.; et al. Nanoparticles in Drug Delivery: From History to Therapeutic Applications. Nanomaterials 2022, 12, 4494. [Google Scholar] [CrossRef] [PubMed]
- Hoare, T.R.; Kohane, D.S. Hydrogels in drug delivery: Progress and challenges. Polymer 2008, 49, 1993–2007. [Google Scholar] [CrossRef]
- Namiot, E.D.; Sokolov, A.V.; Chubarev, V.N.; Tarasov, V.V.; Schiöth, H.B. Nanoparticles in Clinical Trials: Analysis of Clinical Trials, FDA Approvals and Use for COVID-19 Vaccines. Int. J. Mol. Sci. 2023, 24, 787. [Google Scholar] [CrossRef]
- Chehelgerdi, M.; Chehelgerdi, M.; Allela, O.Q.B.; Pecho, R.D.C.; Jayasankar, N.; Rao, D.P.; Thamaraikani, T.; Vasanthan, M.; Viktor, P.; Lakshmaiya, N.; et al. Progressing nanotechnology to improve targeted cancer treatment: Overcoming hurdles in its clinical implementation. Mol. Cancer 2023, 22, 169. [Google Scholar] [CrossRef]
- Ponnappan, N.; Chugh, A. Nanoparticle-Mediated Delivery of Therapeutic Drugs. Pharm. Med. 2015, 29, 155–167. [Google Scholar] [CrossRef]
- Dang, Y.; Guan, J. Nanoparticle-based drug delivery systems for cancer therapy. Smart Mater. Med. 2020, 1, 10–19. [Google Scholar] [CrossRef]
- Altammar, K.A. A review on nanoparticles: Characteristics, synthesis, applications, and challenges. Front. Microbiol. 2023, 14, 1155622. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Wang, Y.; Ran, F.; Cui, Y.; Liu, C.; Zhao, Q.; Gao, Y.; Wang, D.; Wang, S. A comparison between sphere and rod nanoparticles regarding their in vivo biological behavior and pharmacokinetics. Sci. Rep. 2017, 7, 4131. [Google Scholar] [CrossRef]
- Nguyen, M.D.; Tran, H.V.; Xu, S.; Lee, T.R. Fe3O4 Nanoparticles: Structures, Synthesis, Magnetic Properties, Surface Functionalization, and Emerging Applications. Appl. Sci. 2021, 11, 11301. [Google Scholar] [CrossRef]
- Shen, L.; Li, B.; Qiao, Y. Fe3O4 Nanoparticles in Targeted Drug/Gene Delivery Systems. Materials 2018, 11, 324. [Google Scholar] [CrossRef]
- Saha, S.; Ali, M.R.; Khaleque, M.A.; Bacchu, M.S.; Aly Saad Aly, M.; Khan, M.Z.H. Metal oxide nanocarrier for targeted drug delivery towards the treatment of global infectious diseases: A review. J. Drug Deliv. Sci. Technol. 2023, 86, 104728. [Google Scholar] [CrossRef]
- Kong, F.Y.; Zhang, J.W.; Li, R.F.; Wang, Z.X.; Wang, W.J.; Wang, W. Unique Roles of Gold Nanoparticles in Drug Delivery, Targeting and Imaging Applications. Molecules 2017, 22, 1445. [Google Scholar] [CrossRef] [PubMed]
- Han, X.; Xu, K.; Taratula, O.; Farsad, K. Applications of nanoparticles in biomedical imaging. Nanoscale 2019, 11, 799–819. [Google Scholar] [CrossRef]
- Bruna, T.; Maldonado-Bravo, F.; Jara, P.; Caro, N. Silver Nanoparticles and Their Antibacterial Applications. Int. J. Mol. Sci. 2021, 22, 7202. [Google Scholar] [CrossRef]
- de Andrade, L.R.M.; Andrade, L.N.; Bahú, J.O.; Cárdenas Concha, V.O.; Machado, A.T.; Pires, D.S.; Santos, R.; Cardoso, T.F.M.; Cardoso, J.C.; Albuquerque-Junior, R.L.C.; et al. Biomedical applications of carbon nanotubes: A systematic review of data and clinical trials. J. Drug Deliv. Sci. Technol. 2024, 99, 105932. [Google Scholar] [CrossRef]
- Zare, H.; Ahmadi, S.; Ghasemi, A.; Ghanbari, M.; Rabiee, N.; Bagherzadeh, M.; Karimi, M.; Webster, T.J.; Hamblin, M.R.; Mostafavi, E. Carbon Nanotubes: Smart Drug/Gene Delivery Carriers. Int. J. Nanomed. 2021, 16, 1681–1706. [Google Scholar] [CrossRef]
- Malik, S.; Singh, J.; Goyat, R.; Saharan, Y.; Chaudhry, V.; Umar, A.; Ibrahim, A.A.; Akbar, S.; Ameen, S.; Baskoutas, S. Nanomaterials-based biosensor and their applications: A review. Heliyon 2023, 9, e19929. [Google Scholar] [CrossRef]
- Xiao, C.; Li, C.; Hu, J.; Zhu, L. The Application of Carbon Nanomaterials in Sensing, Imaging, Drug Delivery and Therapy for Gynecologic Cancers: An Overview. Molecules 2022, 27, 4465. [Google Scholar] [CrossRef] [PubMed]
- Bitar, A.; Ahmad, N.M.; Fessi, H.; Elaissari, A. Silica-based nanoparticles for biomedical applications. Drug Discov. Today 2012, 17, 1147–1154. [Google Scholar] [CrossRef] [PubMed]
- Pesado-Gómez, C.; Serrano-García, J.S.; Amaya-Flórez, A.; Pesado-Gómez, G.; Soto-Contreras, A.; Morales-Morales, D.; Colorado-Peralta, R. Fullerenes: Historical background, novel biological activities versus possible health risks. Coord. Chem. Rev. 2024, 501, 215550. [Google Scholar] [CrossRef]
- Fernandes, N.B.; Shenoy, R.U.K.; Kajampady, M.K.; CEM, D.C.; Shirodkar, R.K.; Kumar, L.; Verma, R. Fullerenes for the treatment of cancer: An emerging tool. Environ. Sci. Pollut. Res. Int. 2022, 29, 58607–58627. [Google Scholar] [CrossRef] [PubMed]
- Bhandari, M.M.; Raj, S.; Kumar, A.; Kaur, D.P. Bibliometric Analysis on Exploitation of Biogenic Gold and Silver Nanoparticles in Breast, Ovarian and Cervical Cancer Therapy. Front. Pharmacol. 2022, 13, 1035769. [Google Scholar] [CrossRef]
- Hossen, M.N.; Wang, L.; Chinthalapally, H.R.; Robertson, J.D.; Fung, K.M.; Wilhelm, S.; Bieniasz, M.; Bhattacharya, R.; Mukherjee, P. Switching the Intracellular Pathway and Enhancing the Therapeutic Efficacy of Small Interfering RNA by Auroliposome. Sci. Adv. 2020, 6, eaba5379. [Google Scholar] [CrossRef]
- Hong, S.; Zhang, X.; Chen, J.; Zhou, J.; Zheng, Y.; Xu, C. Targeted Gene Silencing Using a Follicle-Stimulating Hormone Peptide-Conjugated Nanoparticle System Improves Its Specificity and Efficacy in Ovarian Clear Cell Carcinoma in Vitro. J. Ovarian Res. 2013, 6, 80. [Google Scholar] [CrossRef]
- Zhang, X.; Chen, J.; Zheng, Y.; Gao, X.; Kang, Y.; Liu, J.-c.; Cheng, M.J.; Sun, H.; Xu, C. Follicle-Stimulating Hormone Peptide Can Facilitate Paclitaxel Nanoparticles to Target Ovarian Carcinoma In Vivo. Cancer Res. 2009, 69, 6506–6514. [Google Scholar] [CrossRef] [PubMed]
- Selopal, G.S.; Mohammadnezhad, M.; Besteiro, L.V.; Cavuslar, O.; Liu, J.; Zhang, H.; Navarro-Pardo, F.; Liu, G.; Wang, M.; Durmusoglu, E.G.; et al. Synergistic Effect of Plasmonic Gold Nanoparticles Decorated Carbon Nanotubes in Quantum Dots/TiO(2) for Optoelectronic Devices. Adv. Sci. 2020, 7, 2001864. [Google Scholar] [CrossRef]
- Hong, S.S.; Zhang, M.X.; Zhang, M.; Yu, Y.; Chen, J.; Zhang, X.Y.; Xu, C.J. Follicle-stimulating hormone peptide-conjugated nanoparticles for targeted shRNA delivery lead to effective gro-α silencing and antitumor activity against ovarian cancer. Drug Deliv. 2018, 25, 576–584. [Google Scholar] [CrossRef] [PubMed]
- Gong, X.; Chi, H.; Strohmer, D.F.; Teíchmann, A.; Xia, Z.; Wang, Q. Exosomes: A Potential Tool for Immunotherapy of Ovarian Cancer. Front. Immunol. 2023, 13, 1089410. [Google Scholar] [CrossRef]
- Liu, W.; Nie, L.; Li, F.; Aguilar, Z.P.; Xu, H.; Xiong, Y.; Fu, F.; Xu, H. Folic Acid Conjugated Magnetic Iron Oxide Nanoparticles for Nondestructive Separation and Detection of Ovarian Cancer Cells From Whole Blood. Biomater. Sci. 2016, 4, 159–166. [Google Scholar] [CrossRef]
- Anchordoquy, T.J.; Barenholz, Y.; Boraschi, D.; Chorny, M.; Decuzzi, P.; Dobrovolskaia, M.A.; Farhangrazi, Z.S.; Farrell, D.; Gabizón, A.; Ghandehari, H.; et al. Mechanisms and Barriers in Cancer Nanomedicine: Addressing Challenges, Looking for Solutions. Acs Nano 2017, 11, 12–18. [Google Scholar] [CrossRef]
- Lavudi, K.; Kokkanti, R.R.; Patnaik, S.; Penchalaneni, J. Chemotherapeutic Potential of AgNP Orchestrated Semecarpus Anacardium Nut Extracts Against Ovarian Cancer Cell Line, PA-1. Eur. J. Med Heal. Res. 2024, 2, 51–62. [Google Scholar] [CrossRef]
- Brain, K.E.; Smits, S.; Simon, A.E.; Forbes, L.J.; Roberts, C.; Robbé, I.J.; Steward, J.; White, C.; Neal, R.D.; Hanson, J. Ovarian cancer symptom awareness and anticipated delayed presentation in a population sample. BMC Cancer 2014, 14, 171. [Google Scholar] [CrossRef]
- Stewart, C.; Ralyea, C.; Lockwood, S. Ovarian Cancer: An Integrated Review. Semin. Oncol. Nurs. 2019, 35, 151–156. [Google Scholar] [CrossRef]
- Dilley, J.; Burnell, M.; Gentry-Maharaj, A.; Ryan, A.; Neophytou, C.; Apostolidou, S.; Karpinskyj, C.; Kalsi, J.; Mould, T.; Woolas, R.; et al. Ovarian cancer symptoms, routes to diagnosis and survival—Population cohort study in the ‘no screen’ arm of the UK Collaborative Trial of Ovarian Cancer Screening (UKCTOCS). Gynecol. Oncol. 2020, 158, 316–322. [Google Scholar] [CrossRef] [PubMed]
- Kurman, R.J.; Visvanathan, K.; Roden, R.; Wu, T.C.; Shih Ie, M. Early detection and treatment of ovarian cancer: Shifting from early stage to minimal volume of disease based on a new model of carcinogenesis. Am. J. Obs. Gynecol. 2008, 198, 351–356. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, M.; Gao, X.; Chen, Y.; Liu, T. Nanotechnology in cancer diagnosis: Progress, challenges and opportunities. J. Hematol. Oncol. 2019, 12, 137. [Google Scholar] [CrossRef]
- Khazaei, M.; Hosseini, M.S.; Haghighi, A.M.; Misaghi, M. Nanosensors and their applications in early diagnosis of cancer. Sens. Bio-Sens. Res. 2023, 41, 100569. [Google Scholar] [CrossRef]
- Yang, Y.; Huang, Q.; Xiao, Z.; Liu, M.; Zhu, Y.; Chen, Q.; Li, Y.; Ai, K. Nanomaterial-based biosensor developing as a route toward in vitro diagnosis of early ovarian cancer. Mater. Today Bio 2022, 13, 100218. [Google Scholar] [CrossRef]
- Ahmed Taha, B.; Kadhim, A.C.; Addie, A.J.; Haider, A.J.; Azzahrani, A.S.; Raizada, P.; Rustagi, S.; Chaudhary, V.; Arsad, N. Advancing cancer diagnostics through multifaceted optical biosensors supported by nanomaterials and artificial intelligence: A panoramic outlook. Microchem. J. 2024, 205, 111307. [Google Scholar] [CrossRef]
- Kim, P.S.; Djazayeri, S.; Zeineldin, R. Novel nanotechnology approaches to diagnosis and therapy of ovarian cancer. Gynecol. Oncol. 2011, 120, 393–403. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Chen, C.; Wang, P.; Mulvey, J.J.; Yang, Y.; Wun, C.; Antman-Passig, M.; Luo, H.B.; Cho, S.; Long-Roche, K.; et al. Detection of ovarian cancer via the spectral fingerprinting of quantum-defect-modified carbon nanotubes in serum by machine learning. Nat. Biomed. Eng. 2022, 6, 267–275. [Google Scholar] [CrossRef] [PubMed]
- Alim, S.; Vejayan, J.; Yusoff, M.M.; Kafi, A.K.M. Recent uses of carbon nanotubes & gold nanoparticles in electrochemistry with application in biosensing: A review. Biosens. Bioelectron. 2018, 121, 125–136. [Google Scholar] [CrossRef] [PubMed]
- Elhami, A.; Mobed, A.; Soleimany, R.; Yazdani, Y.; Kazemi, E.S.; Mohammadi, M.; Saffarfar, H. Sensitive and Cost-Effective Tools in the Detection of Ovarian Cancer Biomarkers. Anal. Sci. Adv. 2024, 5, e202400029. [Google Scholar] [CrossRef]
- Ge, F.; Ding, W.; Han, C.; Zhang, L.; Liu, Q.; Zhao, J.; Luo, Z.; Jia, C.; Qu, P.; Zhang, L. Electrochemical Sensor for the Detection and Accurate Early Diagnosis of Ovarian Cancer. ACS Sens. 2024, 9, 2897–2906. [Google Scholar] [CrossRef]
- Shanbhag, M.M.; Manasa, G.; Mascarenhas, R.J.; Mondal, K.; Shetti, N.P. Fundamentals of bio-electrochemical sensing. Chem. Eng. J. Adv. 2023, 16, 100516. [Google Scholar] [CrossRef]
- Baranwal, J.; Barse, B.; Gatto, G.; Broncova, G.; Kumar, A. Electrochemical Sensors and Their Applications: A Review. Chemosensors 2022, 10, 363. [Google Scholar] [CrossRef]
- El-Gammal, M.A.; Sayed, F.E.; Allam, N.K. Comprehensive analysis of electrochemical biosensors for early ovarian cancer detection. RSC Adv. 2024, 14, 37580–37597. [Google Scholar] [CrossRef]
- Fu, L.; Karimi-Maleh, H. Leveraging electrochemical sensors to improve efficiency of cancer detection. World J. Clin. Oncol. 2024, 15, 360–366. [Google Scholar] [CrossRef]
- Zhang, J.; Ding, H.; Zhang, F.; Xu, Y.; Liang, W.; Huang, L. New trends in diagnosing and treating ovarian cancer using nanotechnology. Front. Bioeng. Biotechnol. 2023, 11, 1160985. [Google Scholar] [CrossRef] [PubMed]
- Klein, T.; Wang, W.; Yu, L.; Wu, K.; Boylan, K.L.M.; Vogel, R.I.; Skubitz, A.P.N.; Wang, J.-P. Development of a multiplexed giant magnetoresistive biosensor array prototype to quantify ovarian cancer biomarkers. Biosens. Bioelectron. 2019, 126, 301–307. [Google Scholar] [CrossRef] [PubMed]
- Wu, K.; Tonini, D.; Liang, S.; Saha, R.; Chugh, V.K.; Wang, J.P. Giant Magnetoresistance Biosensors in Biomedical Applications. ACS Appl. Mater. Interfaces 2022, 14, 9945–9969. [Google Scholar] [CrossRef]
- Kumari, R.; Singh, A.; Azad, U.P.; Chandra, P. Insights into the Fabrication and Electrochemical Aspects of Paper Microfluidics-Based Biosensor Module. Biosensors 2023, 13, 891. [Google Scholar] [CrossRef]
- Mustafa, F.; Finny, A.S.; Kirk, K.A.; Andreescu, S. Chapter Three—Printed paper-based (bio)sensors: Design, fabrication and applications. In Comprehensive Analytical Chemistry; Merkoçi, A., Ed.; Elsevier: Amsterdam, The Netherlands, 2020; Volume 89, pp. 63–89. [Google Scholar]
- Martins, G.V.; Marques, A.C.; Fortunato, E.; Sales, M.G.F. Wax-printed paper-based device for direct electrochemical detection of 3-nitrotyrosine. Electrochim. Acta 2018, 284, 60–68. [Google Scholar] [CrossRef]
- Ding, H.; Zhang, J.; Zhang, F.; Xu, Y.; Liang, W.; Yu, Y. Nanotechnological approaches for diagnosis and treatment of ovarian cancer: A review of recent trends. Drug Deliv. 2022, 29, 3218–3232. [Google Scholar] [CrossRef]
- Hasanzadeh, M.; Sahmani, R.; Solhi, E.; Mokhtarzadeh, A.; Shadjou, N.; Mahboob, S. Ultrasensitive immunoassay of carcinoma antigen 125 in untreated human plasma samples using gold nanoparticles with flower like morphology: A new platform in early stage diagnosis of ovarian cancer and efficient management. Int. J. Biol. Macromol. 2018, 119, 913–925. [Google Scholar] [CrossRef]
- Rebelo, T.S.C.R.; Costa, R.; Brandão, A.T.S.C.; Silva, A.F.; Sales, M.G.F.; Pereira, C.M. Molecularly imprinted polymer SPE sensor for analysis of CA-125 on serum. Anal. Chim. Acta 2019, 1082, 126–135. [Google Scholar] [CrossRef]
- Fulton, M.D.; Najahi-Missaoui, W. Liposomes in Cancer Therapy: How Did We Start and Where Are We Now. Int. J. Mol. Sci. 2023, 24, 6615. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Hu, S.; Sun, M.; Shi, J.; Zhang, H.; Yu, H.; Yang, Z. Recent advances and clinical translation of liposomal delivery systems in cancer therapy. Eur. J. Pharm. Sci. 2024, 193, 106688. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.; Chen, G.; Zhang, J. A Review of Liposomes as a Drug Delivery System: Current Status of Approved Products, Regulatory Environments, and Future Perspectives. Molecules 2022, 27, 1372. [Google Scholar] [CrossRef]
- Wang, S.; Chen, Y.; Guo, J.; Huang, Q. Liposomes for Tumor Targeted Therapy: A Review. Int. J. Mol. Sci. 2023, 24, 2643. [Google Scholar] [CrossRef]
- Chis, A.A.; Dobrea, C.; Morgovan, C.; Arseniu, A.M.; Rus, L.L.; Butuca, A.; Juncan, A.M.; Totan, M.; Vonica-Tincu, A.L.; Cormos, G.; et al. Applications and Limitations of Dendrimers in Biomedicine. Molecules 2020, 25, 3982. [Google Scholar] [CrossRef]
- Cai, L.; Xu, G.; Shi, C.; Guo, D.; Wang, X.; Luo, J. Telodendrimer nanocarrier for co-delivery of paclitaxel and cisplatin: A synergistic combination nanotherapy for ovarian cancer treatment. Biomaterials 2015, 37, 456–468. [Google Scholar] [CrossRef]
- Sampathkumar, S.-G.; Yarema, K.J. Targeting Cancer Cells with Dendrimers. Chem. Biol. 2005, 12, 5–6. [Google Scholar] [CrossRef]
- Ma, P.; Mumper, R.J. Paclitaxel Nano-Delivery Systems: A Comprehensive Review. J. Nanomed. Nanotechnol. 2013, 4, 1000164. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Qiao, X.; Yang, X.; Yuan, M.; Xian, S.; Zhang, L.; Yang, D.; Liu, S.; Dai, F.; Tan, Z.; et al. The role of a drug-loaded poly (lactic co-glycolic acid) (PLGA) copolymer stent in the treatment of ovarian cancer. Cancer Biol. Med. 2020, 17, 237–250. [Google Scholar] [CrossRef]
- Dinarvand, R.; Sepehri, N.; Manoochehri, S.; Rouhani, H.; Atyabi, F. Polylactide-co-glycolide nanoparticles for controlled delivery of anticancer agents. Int. J. Nanomed. 2011, 6, 877–895. [Google Scholar] [CrossRef] [PubMed]
- Vines, J.B.; Yoon, J.H.; Ryu, N.E.; Lim, D.J.; Park, H. Gold Nanoparticles for Photothermal Cancer Therapy. Front. Chem. 2019, 7, 167. [Google Scholar] [CrossRef] [PubMed]
- Georgeous, J.; AlSawaftah, N.; Abuwatfa, W.H.; Husseini, G.A. Review of Gold Nanoparticles: Synthesis, Properties, Shapes, Cellular Uptake, Targeting, Release Mechanisms and Applications in Drug Delivery and Therapy. Pharmaceutics 2024, 16, 1332. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Y.G.; Peng, Q.L.; Gurunathan, S. Silver nanoparticles enhance the apoptotic potential of gemcitabine in human ovarian cancer cells: Combination therapy for effective cancer treatment. Int. J. Nanomed. 2017, 12, 6487–6502. [Google Scholar] [CrossRef]
- Takáč, P.; Michalková, R.; Čižmáriková, M.; Bedlovičová, Z.; Balážová, Ľ.; Takáčová, G. The Role of Silver Nanoparticles in the Diagnosis and Treatment of Cancer: Are There Any Perspectives for the Future? Life 2023, 13, 466. [Google Scholar] [CrossRef]
- Wu, M.; Huang, S. Magnetic nanoparticles in cancer diagnosis, drug delivery and treatment. Mol. Clin. Oncol. 2017, 7, 738–746. [Google Scholar] [CrossRef]
- Nowak-Jary, J.; Płóciennik, A.; Machnicka, B. Functionalized Magnetic Fe(3)O(4) Nanoparticles for Targeted Methotrexate Delivery in Ovarian Cancer Therapy. Int. J. Mol. Sci. 2024, 25, 9098. [Google Scholar] [CrossRef]
- Giustini, A.J.; Petryk, A.A.; Cassim, S.M.; Tate, J.A.; Baker, I.; Hoopes, P.J. Magnetic Nanoparticle Hyperthermia in Cancer Treatment. Nano Life 2010, 1, 17–32. [Google Scholar] [CrossRef]
- Bañobre-López, M.; Teijeiro, A.; Rivas, J. Magnetic nanoparticle-based hyperthermia for cancer treatment. Rep. Pract. Oncol. Radiother. 2013, 18, 397–400. [Google Scholar] [CrossRef]
- Montazersaheb, P.; Pishgahzadeh, E.; Jahani, V.B.; Farahzadi, R.; Montazersaheb, S. Magnetic nanoparticle-based hyperthermia: A prospect in cancer stem cell tracking and therapy. Life Sci. 2023, 323, 121714. [Google Scholar] [CrossRef]
- Yao, Y.; Zhou, Y.; Liu, L.; Xu, Y.; Chen, Q.; Wang, Y.; Wu, S.; Deng, Y.; Zhang, J.; Shao, A. Nanoparticle-Based Drug Delivery in Cancer Therapy and Its Role in Overcoming Drug Resistance. Front. Mol. Biosci. 2020, 7, 193. [Google Scholar] [CrossRef]
- Elumalai, K.; Srinivasan, S.; Shanmugam, A. Review of the efficacy of nanoparticle-based drug delivery systems for cancer treatment. Biomed. Technol. 2024, 5, 109–122. [Google Scholar] [CrossRef]
- Kim, M.S.; Haney, M.J.; Zhao, Y.; Mahajan, V.; Deygen, I.; Klyachko, N.L.; Inskoe, E.; Piroyan, A.; Sokolsky, M.; Okolie, O.; et al. Development of exosome-encapsulated paclitaxel to overcome MDR in cancer cells. Nanomedicine 2016, 12, 655–664. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Liu, Y.; Zheng, S.; Zhang, T.; Wu, J.; Sun, Y.; Zhang, J.; Liu, G. Role of exosomes in the immune microenvironment of ovarian cancer. Oncol. Lett. 2021, 21, 377. [Google Scholar] [CrossRef]
- Zhou, X.; Jin, W.; Ma, J. Lung inflammation perturbation by engineered nanoparticles. Front. Bioeng. Biotechnol. 2023, 11, 1199230. [Google Scholar] [CrossRef]
- Ao, L.-H.; Wei, Y.-G.; Tian, H.-R.; Zhao, H.; Li, J.; Ban, J.-Q. Advances in the study of silica nanoparticles in lung diseases. Sci. Total Environ. 2024, 912, 169352. [Google Scholar] [CrossRef]
- Zhuo, L.-B.; Liu, Y.-M.; Jiang, Y.; Yan, Z. Zinc oxide nanoparticles induce acute lung injury via oxidative stress-mediated mitochondrial damage and NLRP3 inflammasome activation: In vitro and in vivo studies. Environ. Pollut. 2024, 341, 122950. [Google Scholar] [CrossRef] [PubMed]
- Hammond, J.; Maher, B.A.; Gonet, T.; Bautista, F.; Allsop, D. Oxidative Stress, Cytotoxic and Inflammatory Effects of Urban Ultrafine Road-Deposited Dust from the UK and Mexico in Human Epithelial Lung (Calu-3) Cells. Antioxidants 2022, 11, 1814. [Google Scholar] [CrossRef] [PubMed]
- Khanna, P.; Ong, C.; Bay, B.H.; Baeg, G.H. Nanotoxicity: An Interplay of Oxidative Stress, Inflammation and Cell Death. Nanomaterials 2015, 5, 1163–1180. [Google Scholar] [CrossRef]
- Zhu, L.; Luo, M.; Zhang, Y.; Fang, F.; Li, M.; An, F.; Zhao, D.; Zhang, J. Free radical as a double-edged sword in disease: Deriving strategic opportunities for nanotherapeutics. Coord. Chem. Rev. 2023, 475, 214875. [Google Scholar] [CrossRef]
- kazemi, S.; Hosseingholian, A.; Gohari, S.D.; Feirahi, F.; Moammeri, F.; Mesbahian, G.; Moghaddam, Z.S.; Ren, Q. Recent advances in green synthesized nanoparticles: From production to application. Mater. Today Sustain. 2023, 24, 100500. [Google Scholar] [CrossRef]
- Najahi-Missaoui, W.; Arnold, R.D.; Cummings, B.S. Safe Nanoparticles: Are We There Yet? Int. J. Mol. Sci. 2020, 22, 385. [Google Scholar] [CrossRef] [PubMed]
- Satchanska, G.; Davidova, S.; Petrov, P.D. Natural and Synthetic Polymers for Biomedical and Environmental Applications. Polymers 2024, 16, 1159. [Google Scholar] [CrossRef] [PubMed]
- Kalpana Manivannan, R.; Sharma, N.; Kumar, V.; Jayaraj, I.; Vimal, S.; Umesh, M. A comprehensive review on natural macromolecular biopolymers for biomedical applications: Recent advancements, current challenges, and future outlooks. Carbohydr. Polym. Technol. Appl. 2024, 8, 100536. [Google Scholar] [CrossRef]
- Santhanakrishnan, K.R.; Koilpillai, J.; Narayanasamy, D. PEGylation in Pharmaceutical Development: Current Status and Emerging Trends in Macromolecular and Immunotherapeutic Drugs. Cureus 2024, 16, e66669. [Google Scholar] [CrossRef]
- Serna-Gallén, P.; Mužina, K. Metallic nanoparticles at the forefront of research: Novel trends in catalysis and plasmonics. Nano Mater. Sci. 2024. [Google Scholar] [CrossRef]
- Mansouri, E.; Mesbahi, A.; Hamishehkar, H.; Montazersaheb, S.; Hosseini, V.; Rajabpour, S. The effect of nanoparticle coating on biological, chemical and biophysical parameters influencing radiosensitization in nanoparticle-aided radiation therapy. BMC Chem. 2023, 17, 180. [Google Scholar] [CrossRef]
- Al-Thani, A.N.; Jan, A.G.; Abbas, M.; Geetha, M.; Sadasivuni, K.K. Nanoparticles in cancer theragnostic and drug delivery: A comprehensive review. Life Sci. 2024, 352, 122899. [Google Scholar] [CrossRef]
- Abdelkawi, A.; Slim, A.; Zinoune, Z.; Pathak, Y. Surface Modification of Metallic Nanoparticles for Targeting Drugs. Coatings 2023, 13, 1660. [Google Scholar] [CrossRef]
- Seidu, T.A.; Kutoka, P.T.; Asante, D.O.; Farooq, M.A.; Alolga, R.N.; Bo, W. Functionalization of Nanoparticulate Drug Delivery Systems and Its Influence in Cancer Therapy. Pharmaceutics 2022, 14, 1113. [Google Scholar] [CrossRef]
- Adepu, S.; Ramakrishna, S. Controlled Drug Delivery Systems: Current Status and Future Directions. Molecules 2021, 26, 5905. [Google Scholar] [CrossRef]
- Ezike, T.C.; Okpala, U.S.; Onoja, U.L.; Nwike, C.P.; Ezeako, E.C.; Okpara, O.J.; Okoroafor, C.C.; Eze, S.C.; Kalu, O.L.; Odoh, E.C.; et al. Advances in drug delivery systems, challenges and future directions. Heliyon 2023, 9, e17488. [Google Scholar] [CrossRef]
- Levit, S.L.; Tang, C. Polymeric Nanoparticle Delivery of Combination Therapy With Synergistic Effects in Ovarian Cancer. Nanomaterials 2021, 11, 1048. [Google Scholar] [CrossRef]
- Nie, L.; Li, F.; Huang, X.; Aguilar, Z.P.; Wang, Y.A.; Xiong, Y.; Fu, F.; Xu, H. Folic Acid Targeting for Efficient Isolation and Detection of Ovarian Cancer CTCs From Human Whole Blood Based on Two-Step Binding Strategy. ACS Appl. Mater. Interfaces 2018, 10, 14055–14062. [Google Scholar] [CrossRef]
- Wang, Q.; Wang, D.; Li, D.; Lü, J.; Wei, Q. Folate Modified Nanoparticles for Targeted Co-Delivery Chemotherapeutic Drugs and Imaging Probes for Ovarian Cancer. Biomed. Phys. Eng. Express 2015, 1, 045009. [Google Scholar] [CrossRef]
- Patel, A.; Londhe, S.; Tripathy, S.; Nagchowdhury, P.; Patra, C.R. Folic Acid–encapsulated Silver Nitroprusside Nanoparticles for Targeted Therapy in Ovarian Cancer. Biomed. Mater. 2025, 20, 045007. [Google Scholar] [CrossRef]
- Gralewska, P.; Gajek, A.; Marczak, A.; Rogalska, A. Targeted Nanocarrier-Based Drug Delivery Strategies for Improving the Therapeutic Efficacy of PARP Inhibitors Against Ovarian Cancer. Int. J. Mol. Sci. 2024, 25, 8304. [Google Scholar] [CrossRef]
- Risnayanti, C.; Jang, Y.S.; Lee, J.; Ahn, H.J. PLGA Nanoparticles Co-Delivering MDR1 and BCL2 siRNA for Overcoming Resistance of Paclitaxel and Cisplatin in Recurrent or Advanced Ovarian Cancer. Sci. Rep. 2018, 8, 7498. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; lyer, A.; Singh, A.; Choy, E.; Hornicek, F.J.; Amiji, M.M.; Duan, Z. MDR1 siRNA Loaded Hyaluronic Acid-Based CD44 Targeted Nanoparticle Systems Circumvent Paclitaxel Resistance in Ovarian Cancer. Sci. Rep. 2015, 5, 8509. [Google Scholar] [CrossRef] [PubMed]
- Shahin, S.A.; Wang, R.; Simargi, S.; Contreras, A.; Echavarria, L.; Qu, L.; Wen, W.; Dellinger, T.H.; Unternaehrer, J.; Tamanoi, F.; et al. Hyaluronic Acid Conjugated Nanoparticle Delivery of siRNA Against TWIST Reduces Tumor Burden and Enhances Sensitivity to Cisplatin in Ovarian Cancer. Nanomed. Nanotechnol. Biol. Med. 2018, 14, 1381–1394. [Google Scholar] [CrossRef] [PubMed]
- Talekar, M.; Ouyang, Q.; Goldberg, M.S.; Amiji, M.M. Cosilencing of PKM-2 and MDR-1 Sensitizes Multidrug-Resistant Ovarian Cancer Cells to Paclitaxel in a Murine Model of Ovarian Cancer. Mol. Cancer Ther. 2015, 14, 1521–1531. [Google Scholar] [CrossRef]
- You, T.; Zhang, S. Recent Advances in PLGA Polymer Nanocarriers for Ovarian Cancer Therapy. Front. Oncol. 2025, 15, 1526718. [Google Scholar] [CrossRef]
- Weigelt, J.; Petrosyan, M.O.; Oliveira-Ferrer, L.; Schmalfeldt, B.; Bartmann, C.; Dietl, J.; Stürken, C.; Schumacher, U. Ovarian Cancer Cells Regulate Their Mitochondrial Content and High Mitochondrial Content Is Associated With a Poor Prognosis. BMC Cancer 2024, 24, 43. [Google Scholar] [CrossRef]
- Satpathy, M.; Wang, L.Y.; Zieliński, R.; Qian, W.; Wang, Y.A.; Mohs, A.M.; Kairdolf, B.A.; Ji, X.; Capala, J.; Lipowska, M.; et al. Targeted Drug Delivery and Image-Guided Therapy of Heterogeneous Ovarian Cancer Using HER2-Targeted Theranostic Nanoparticles. Theranostics 2019, 9, 778–795. [Google Scholar] [CrossRef]
- Werner, M.E.; Karve, S.; Sukumar, R.; Cummings, N.D.; Copp, J.A.; Chen, R.C.; Zhang, T.; Wang, A.Z. Folate-Targeted Nanoparticle Delivery of Chemo- And Radiotherapeutics for the Treatment of Ovarian Cancer Peritoneal Metastasis. Biomaterials 2011, 32, 8548–8554. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Liu, Q.; Zhang, M.; Cao, C.; Liu, X.; Zhang, M.; Li, G.; Xu, C.; Zhang, X. Enhanced Antitumor Effects of Follicle-Stimulating Hormone Receptor-Mediated Hexokinase-2 Depletion on Ovarian Cancer Mediated by a Shift in Glucose Metabolism. J. Nanobiotechnology 2020, 18, 161. [Google Scholar] [CrossRef] [PubMed]
- Liang, S.; Liu, Y.-Y.; He, J.; Gao, T.M.; Li, L.; He, S. Family With Sequence Similarity 46 Member a Confers Chemo-Resistance to Ovarian Carcinoma via TGF-β/Smad2 Signaling. Bioengineered 2022, 13, 10629–10639. [Google Scholar] [CrossRef]
- Leung, C.S.; Yeung, T.-L.; Yip, K.P.; Pradeep, S.; Balasubramanian, L.; Liu, J.; Wong, K.K.; Mangala, L.S.; Armaiz-Peña, G.N.; López-Berestein, G.; et al. Calcium-Dependent FAK/CREB/TNNC1 Signalling Mediates the Effect of Stromal MFAP5 on Ovarian Cancer Metastatic Potential. Nat. Commun. 2014, 5, 5092. [Google Scholar] [CrossRef]
- Poley, M.; Mora-Raimundo, P.; Shammai, Y.; Kaduri, M.; Koren, L.; Adir, O.; Shklover, J.; Shainsky-Roitman, J.; Ramishetti, S.; Man, F.; et al. Nanoparticles Accumulate in the Female Reproductive System During Ovulation Affecting Cancer Treatment and Fertility. ACS Nano 2022, 16, 5246–5257. [Google Scholar] [CrossRef]
- Poley, M.; Shammai, Y.; Kaduri, M.; Koren, L.; Adir, O.; Shklover, J.; Shainsky, J.; Ben-Aharon, I.; Zinger, A.; Schroeder, A. Chemotherapeutic Nanoparticles Accumulate in the Female Reproductive System During Ovulation Affecting Fertility and Anticancer Activity. BioRxiv 2020. [Google Scholar] [CrossRef]
- Tiwari, H.; Gupta, P.; Verma, A.; Singh, S.; Kumar, R.; Gautam, H.K.; Gautam, V. Advancing Era and Rising Concerns in Nanotechnology-Based Cancer Treatment. ACS Chem. Health Saf. 2024, 31, 153–161. [Google Scholar] [CrossRef]
- Wang, K. The Promise of Nanomedicine in Ovarian Cancer Treatment: A Review and Outlook. In Proceedings of the 3rd International Conference on Biological Engineering and Medical Science (ICBioMed2023), Online, 2–9 September 2023; p. 120. [Google Scholar] [CrossRef]
- Morris, S.A.; Gaheen, S.; Lijowski, M.; Heiskanen, M.; Klemm, J. Experiences in Supporting the Structured Collection of Cancer Nanotechnology Data Using caNanoLab. Beilstein J. Nanotechnol. 2015, 6, 1580–1593. [Google Scholar] [CrossRef]
- Schneider, J.A.; Grodzinski, P.; Liang, X.J. Cancer Nanotechnology Research in the United States and China: Cooperation to Promote Innovation. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2011, 3, 441–448. [Google Scholar] [CrossRef]
- Bregoli, L.; Movia, D.; Gavigan-Imedio, J.D.; Lysaght, J.; Reynolds, J.; Prina-Mello, A. Nanomedicine Applied to Translational Oncology: A Future Perspective on Cancer Treatment. Nanomed. Nanotechnol. Biol. Med. 2016, 12, 81–103. [Google Scholar] [CrossRef]
- López-Estévez, A.M.; Zhang, Y.; Medel, M.; Arriaga, I.; Sanjurjo, L.; Huck-Iriart, C.; Abrescia, N.G.A.; Vicent, M.a.J.; Ouyang, D.; Torres, D.; et al. Engineering Hyaluronic Acid-Based Nanoassemblies for Monoclonal Antibody Delivery—Design, Characterization, and Biological Insights. Nano Res. 2024, 17, 9111–9125. [Google Scholar] [CrossRef]
- Engelberth, S.A.; Hempel, N.; Bergkvist, M. Development of Nanoscale Approaches for Ovarian Cancer Therapeutics and Diagnostics. Crit. Rev.™ Oncog. 2014, 19, 281–315. [Google Scholar] [CrossRef] [PubMed]
- Matei, D. Pegylated Liposomal Doxorubicin in Ovarian Cancer. Ther. Clin. Risk Manag. 2009, 5, 639–650. [Google Scholar] [CrossRef]
- Jiang, J.; Cui, X.; Huang, Y.; Yan, D.; Wang, B.; Yang, Z.; Chen, M.; Wang, J.; Zhang, Y.; Liu, G.; et al. Advances and Prospects in Integrated Nano-Oncology. Nano Biomed. Eng. 2024, 16, 152–187. [Google Scholar] [CrossRef]
- Grau-Béjar, J.F.; Oaknin, A.; Williamson, C.W.; Mayadev, J.; Peters, P.; Secord, A.A.; Wield, A.; Coffman, L. Novel Therapies in Gynecologic Cancer. Am. Soc. Clin. Oncol. Educ. Book. 2022, 42, 483–499. [Google Scholar] [CrossRef] [PubMed]
- Ejeta, B.M.; Das, M.K.; Das, S. Advancements in Nanotechnology-Based Paclitaxel Delivery Systems: Systematic Review on Overcoming Solubility, Toxicity, and Drug Resistance Challenges in Cancer Therapy. J. Angiother. 2024, 8, 1–7. [Google Scholar] [CrossRef]
- Yallapu, M.M.; Maher, D.M.; Sundram, V.; Bell, M.C.; Jaggi, M.; Chauhan, S.C. Curcumin Induces Chemo/Radio-Sensitization in Ovarian Cancer Cells and Curcumin Nanoparticles Inhibit Ovarian Cancer Cell Growth. J. Ovarian Res. 2010, 3, 11. [Google Scholar] [CrossRef]
- Mekradh, H.A.; Hameed, F.M. Gold Nanoparticles as Anti-Ovarian Cancer Therapy. Al-Qadisiyah Med. J. 2019, 15, 78–83. [Google Scholar] [CrossRef]
- Mannoush, S.H.; Thaker, A.A.; Jabir, M.S. Inhibition of Ovarian Cancer Cells Growth Using Gold Nanoparticles and Silica Coated Gold Nanoparticles: In-Vitro Study. J. Pharm. Negat. Results 2022, 13, 727–733. [Google Scholar] [CrossRef]
- Yang, X.; Iyer, A.K.; Singh, A.; Milane, L.; Choy, E.; Hornicek, F.J.; Amiji, M.M.; Duan, Z. Cluster of Differentiation 44 Targeted Hyaluronic Acid Based Nanoparticles for MDR1 siRNA Delivery to Overcome Drug Resistance in Ovarian Cancer. Pharm. Res. 2014, 32, 2097–2109. [Google Scholar] [CrossRef] [PubMed]



| Feature | Liposomes | Polymeric NPs | Dendrimers | Gold NPs | Iron Oxide NPs |
|---|---|---|---|---|---|
| Structure | Phospholipid bilayer vesicles | Biodegradable polymer matrix | Highly branched synthetic polymers | Inorganic metallic core | Superparamagnetic iron oxide core |
| Drug Loading | Hydrophilic (core) + lipophilic (bilayer) | Hydrophobic/hydrophilic (tunable) | Surface or internal cavity loading | Surface adsorption/conjugation | Surface or encapsulated drugs |
| Targeting Potential | High (PEGylation, ligand-conjugation) | High (modifiable surface) | Very high (multiple functional end-groups) | High (easy to conjugate targeting ligands) | High (can be guided magnetically or with ligands) |
| Biocompatibility | Excellent | Good to excellent | Moderate to good | Variable (depends on size/surface modification) | Good (FDA-approved for imaging) |
| Imaging Capability | Limited | Limited | Limited | Excellent (CT, photoacoustic) | Excellent (MRI contrast agent) |
| Theranostic Use | Moderate (drug + limited imaging) | Moderate | Moderate | High (therapy + imaging) | High (hyperthermia + imaging + drug delivery) |
| Clinical Trials | Several in advanced stages | Some ongoing trials | Preclinical/early stage | Mostly preclinical | Some in imaging, early therapy trials |
| Challenges | Stability, RES uptake | Burst release, scaling production | Toxicity at higher generations, synthesis complexity | Long-term toxicity, accumulation in organs | Heating control, clearance from the body |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
El-Mallul, A.; Tomasiuk, R.; Pieńkowski, T.; Kowalska, M.; Hasan, D.; Kostrzewa, M.; Czerwonka, D.; Sado, A.; Rogowska, W.; Zubrzycki, I.Z.; et al. Applications of Nanoparticles in the Diagnosis and Treatment of Ovarian Cancer. Nanomaterials 2025, 15, 1200. https://doi.org/10.3390/nano15151200
El-Mallul A, Tomasiuk R, Pieńkowski T, Kowalska M, Hasan D, Kostrzewa M, Czerwonka D, Sado A, Rogowska W, Zubrzycki IZ, et al. Applications of Nanoparticles in the Diagnosis and Treatment of Ovarian Cancer. Nanomaterials. 2025; 15(15):1200. https://doi.org/10.3390/nano15151200
Chicago/Turabian StyleEl-Mallul, Ahmed, Ryszard Tomasiuk, Tadeusz Pieńkowski, Małgorzata Kowalska, Dilawar Hasan, Marcin Kostrzewa, Dominik Czerwonka, Aleksandra Sado, Wiktoria Rogowska, Igor Z. Zubrzycki, and et al. 2025. "Applications of Nanoparticles in the Diagnosis and Treatment of Ovarian Cancer" Nanomaterials 15, no. 15: 1200. https://doi.org/10.3390/nano15151200
APA StyleEl-Mallul, A., Tomasiuk, R., Pieńkowski, T., Kowalska, M., Hasan, D., Kostrzewa, M., Czerwonka, D., Sado, A., Rogowska, W., Zubrzycki, I. Z., & Wiacek, M. (2025). Applications of Nanoparticles in the Diagnosis and Treatment of Ovarian Cancer. Nanomaterials, 15(15), 1200. https://doi.org/10.3390/nano15151200








