The Hydrogenation of Crotonaldehyde on PdCu Single Atom Alloy Catalysts
Abstract
1. Introduction
2. Materials and Methods
2.1. Catalyst Synthesis and Characterisation
2.2. Catalytic Testing
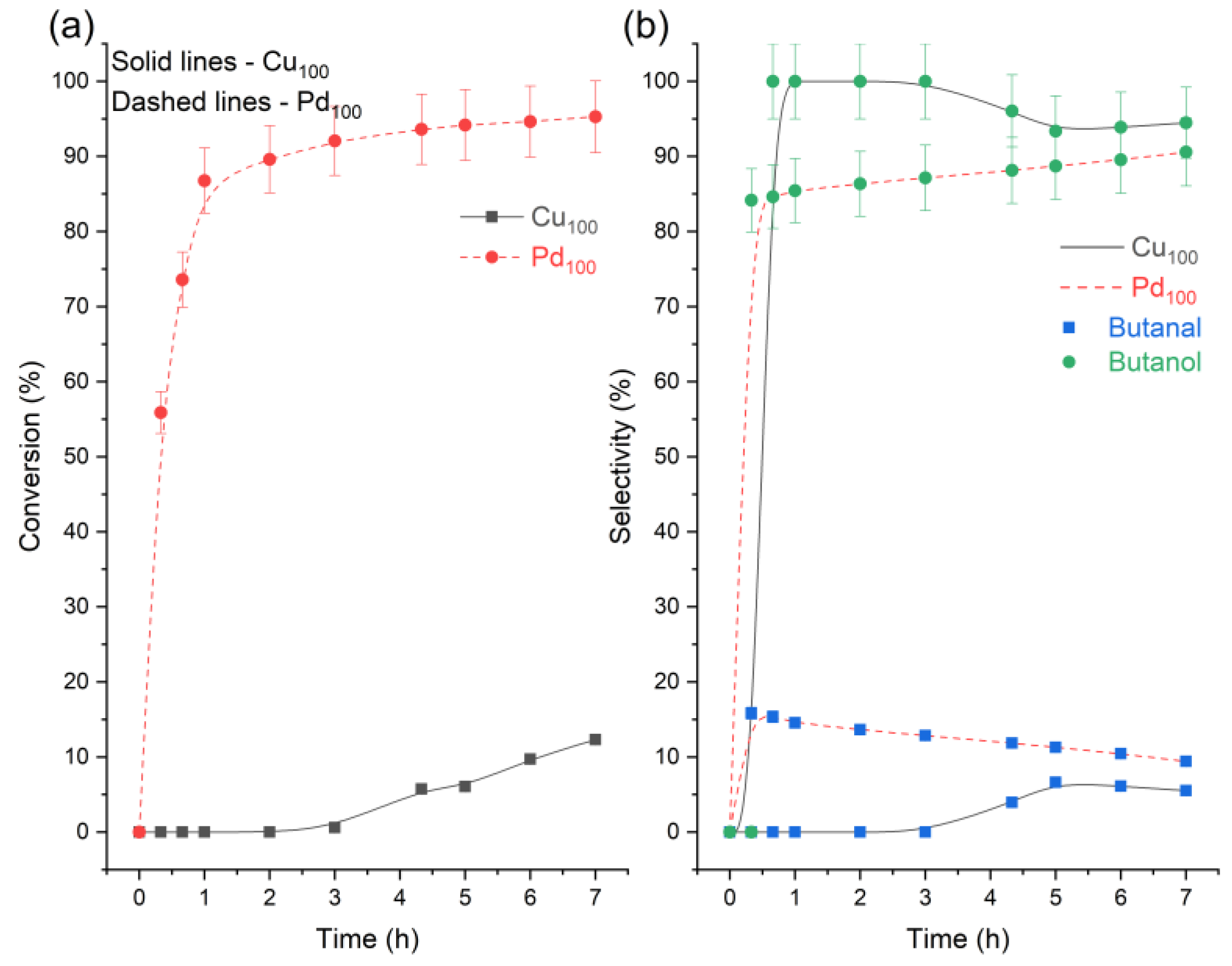
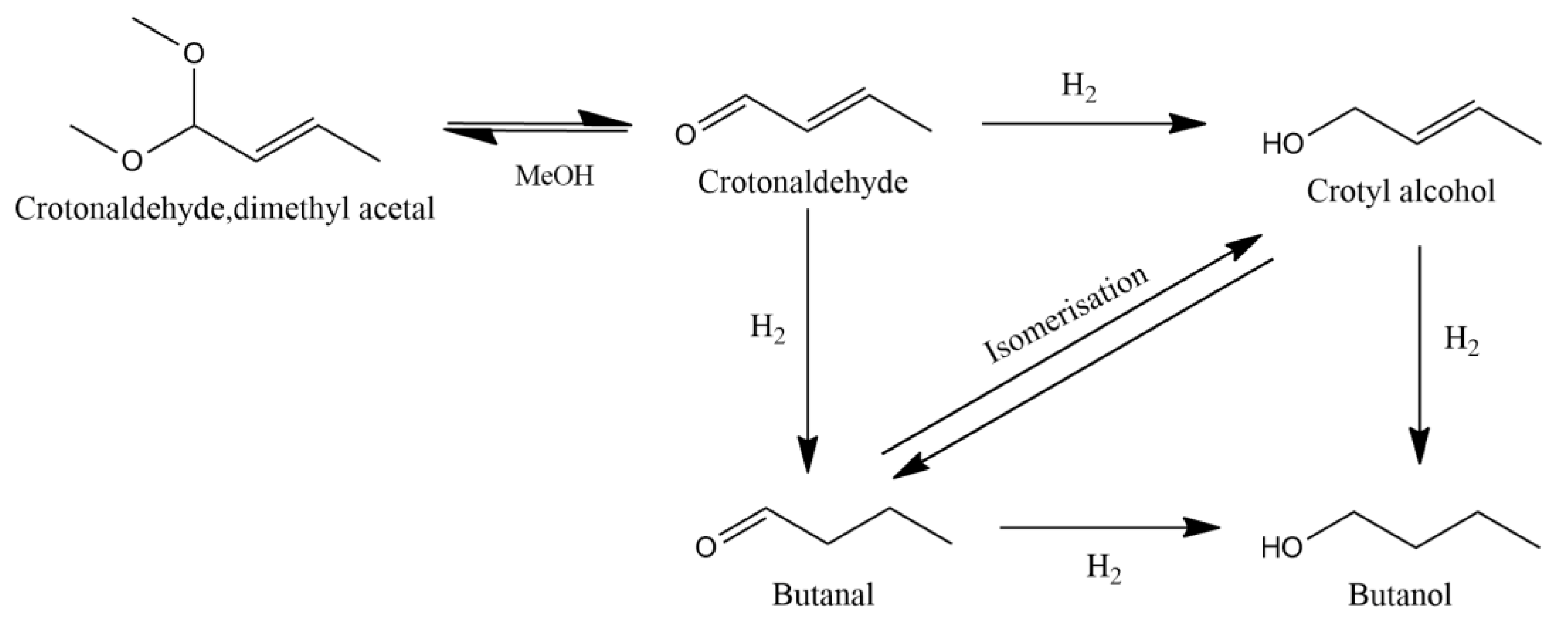
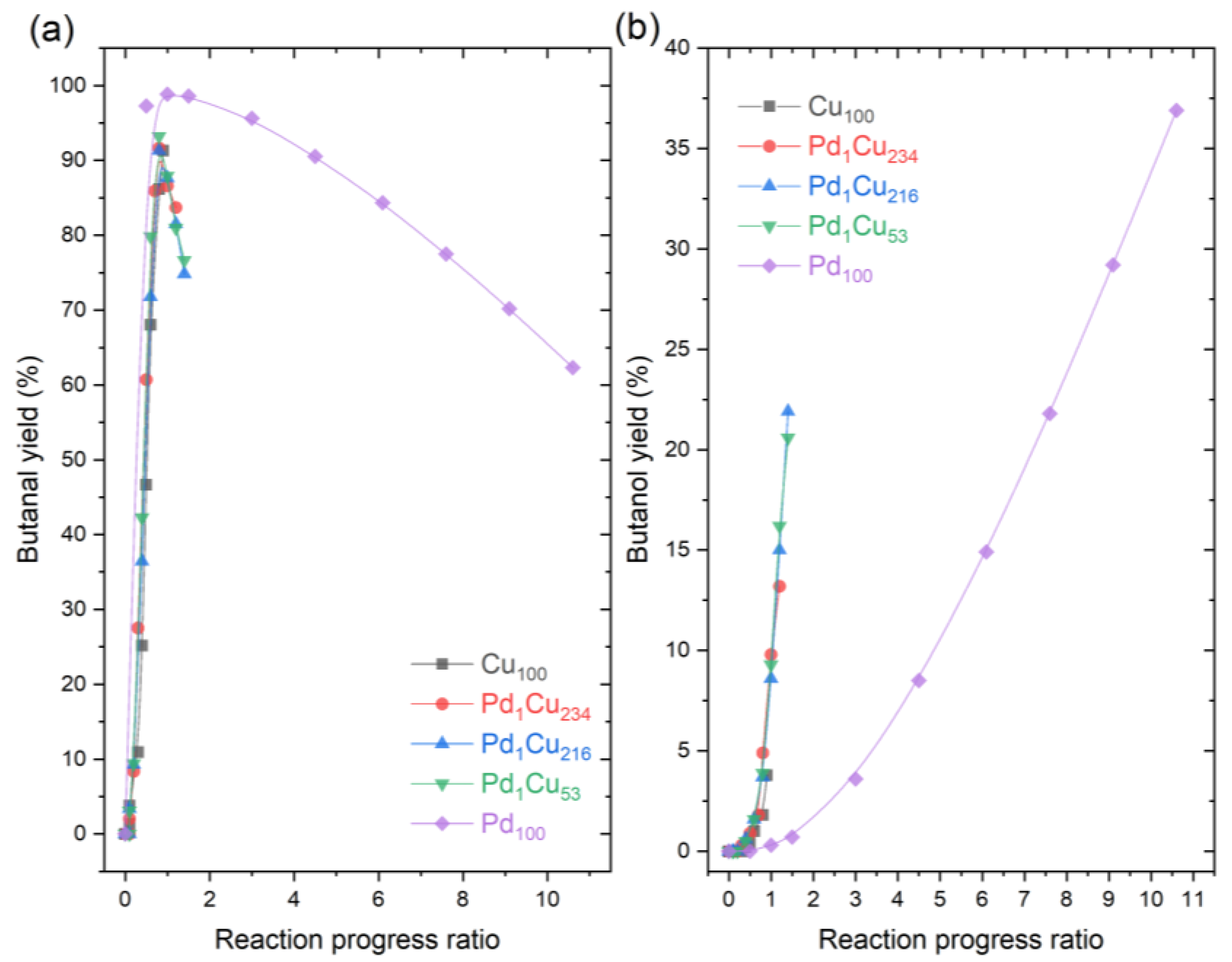
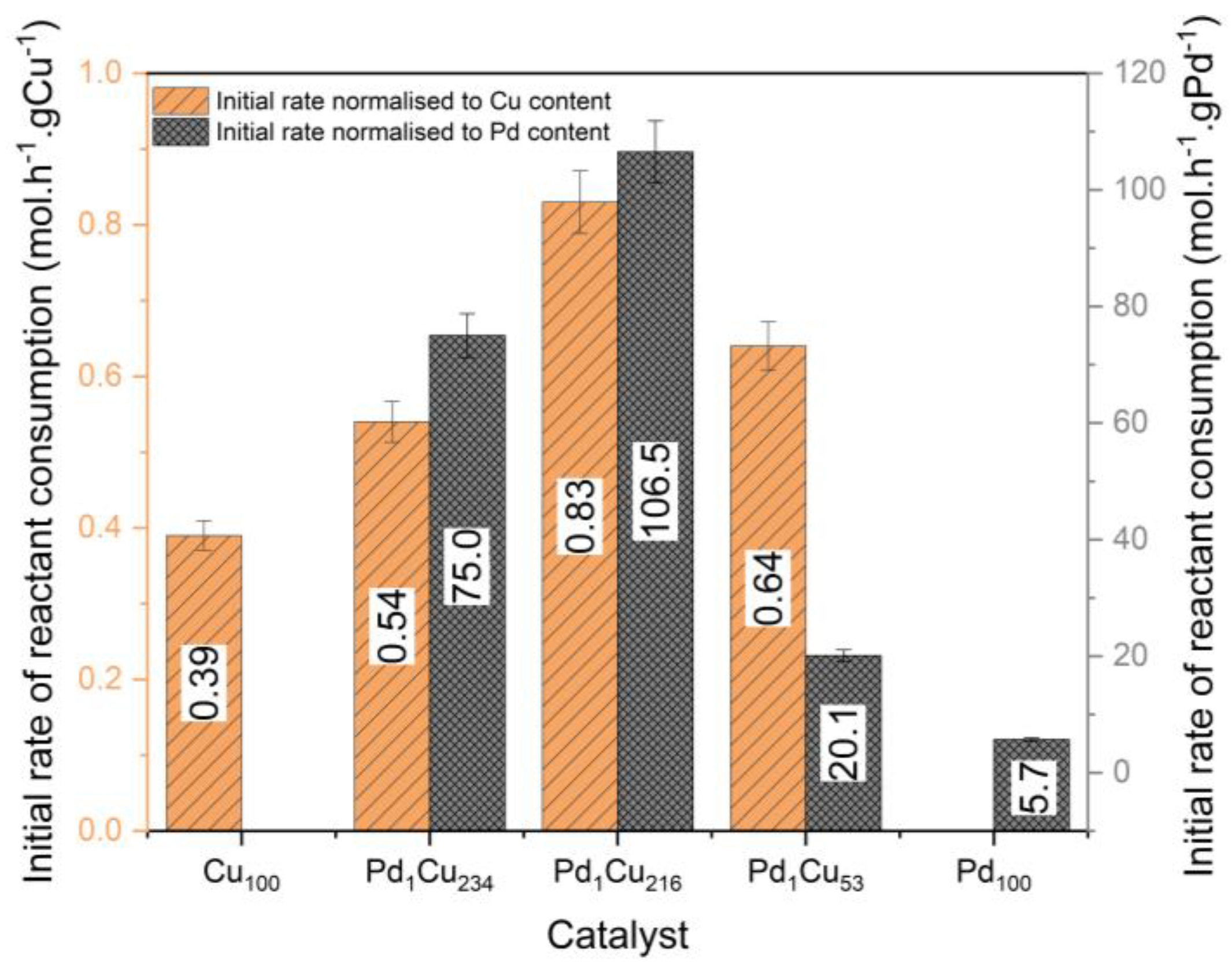
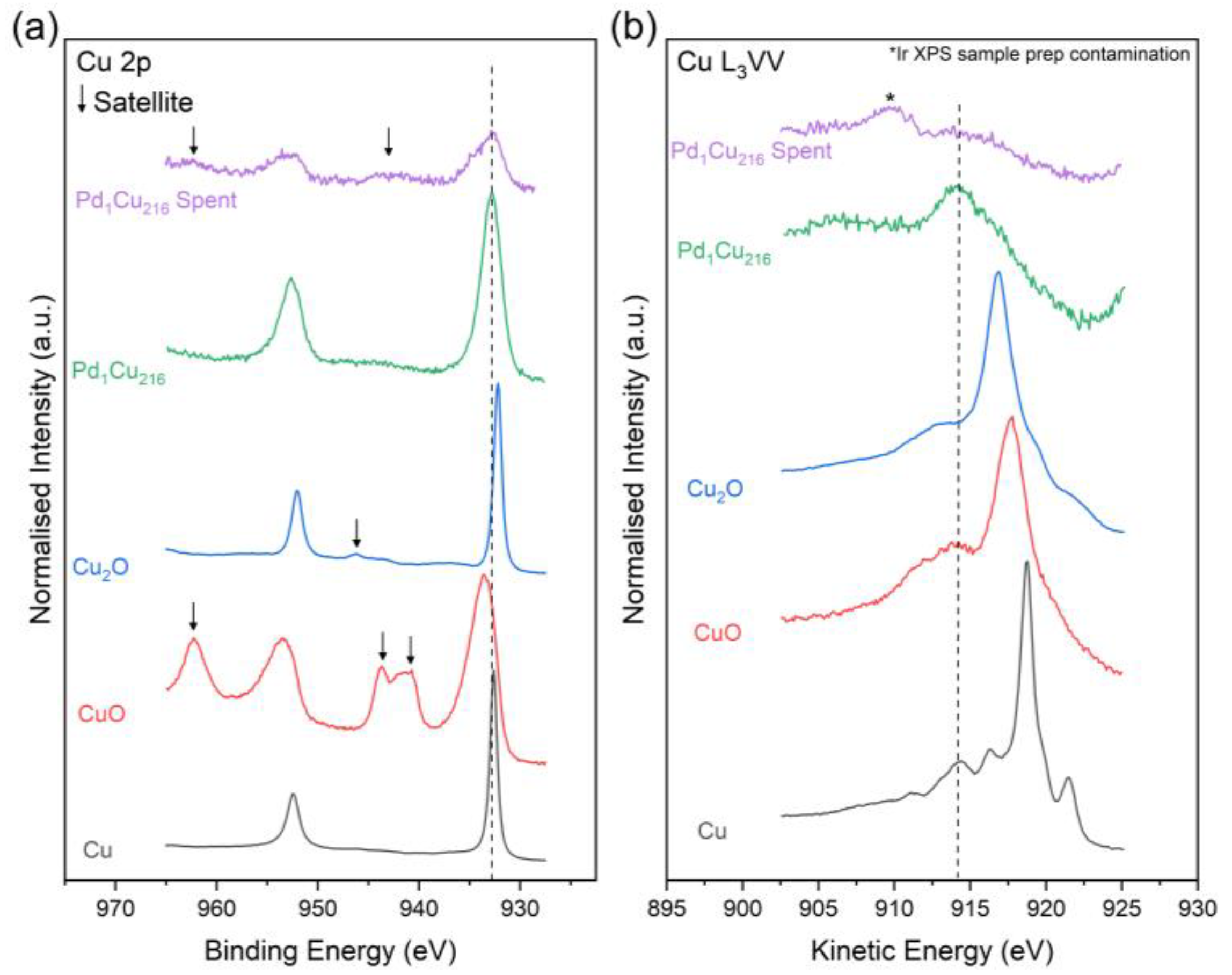
3. Results and Discussion

| Catalyst | Conversion (%) | Butanal S (%) | Butanol S (%) | Carbon Balance (%) |
|---|---|---|---|---|
| Cu100 | 12.3 ± 0.6 | 5.5 ± 0.3 | 94.5 ± 4.8 | 100 ± 5.0 |
| Pd100 | 95.3 ± 4.8 | 9.4 ± 0.5 | 90.6 ± 4.5 | 92.1± 4.6 |
| Sample | State | Cu 2p3/2 (eV) | L3VV (eV) | Cu0 + Cu+ (%) | Cu2+ (%) | Cu Dispersion (%) |
|---|---|---|---|---|---|---|
| Pd1Cu216 | Fresh | 932.83 | 914.24 | 97.6 | 2.4 | 80.3 ± 8.0 |
| Pd1Cu216 | Spent | 932.73 | N/A | 41.3 | 58.7 | 61.5 ± 6.2 |
| CuO | - | 933.62 | 917.78 | - | - | - |
| Cu2O | - | 932.29 | 916.70 | - | - | - |
| Cu * | - | 932.63 | 918.75 | - | - | - |
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Loy, A.C.M.; Teng, S.Y.; How, B.S.; Zhang, X.; Cheah, K.W.; Butera, V.; Leong, W.D.; Chin, B.L.F.; Yiin, C.L.; Taylor, M.J.; et al. Elucidation of single atom catalysts for energy and sustainable chemical production: Synthesis, characterization and frontier science. Prog. Energy Combust. Sci. 2023, 96, 101074. [Google Scholar] [CrossRef]
- Boucher, M.B.; Zugic, B.; Cladaras, G.; Kammert, J.; Marcinkowski, M.D.; Lawton, T.J.; Sykes, E.C.; Flytzani-Stephanopoulos, M. Single atom alloy surface analogs in Pd0.18Cu15 nanoparticles for selective hydrogenation reactions. Phys. Chem. Chem. Phys. 2013, 15, 12187–12196. [Google Scholar] [CrossRef] [PubMed]
- Hu, F.; Leng, L.; Zhang, M.; Chen, W.; Yu, Y.; Wang, J.; Horton, J.H.; Li, Z. Direct Synthesis of Atomically Dispersed Palladium Atoms Supported on Graphitic Carbon Nitride for Efficient Selective Hydrogenation Reactions. ACS Appl. Mater. Interfaces 2020, 12, 54146–54154. [Google Scholar] [CrossRef] [PubMed]
- Islam, M.J.; Granollers Mesa, M.; Osatiashtiani, A.; Taylor, M.J.; Manayil, J.C.; Parlett, C.M.A.; Isaacs, M.A.; Kyriakou, G. The effect of metal precursor on copper phase dispersion and nanoparticle formation for the catalytic transformations of furfural. Appl. Catal. B 2020, 273, 119062. [Google Scholar] [CrossRef]
- Lou, Y.; Zheng, Y.; Li, X.; Ta, N.; Xu, J.; Nie, Y.; Cho, K.; Liu, J. Pocketlike Active Site of Rh1/MoS2 Single-Atom Catalyst for Selective Crotonaldehyde Hydrogenation. J. Am. Chem. Soc. 2019, 141, 19289–19295. [Google Scholar] [CrossRef]
- Lucci, F.R.; Liu, J.; Marcinkowski, M.D.; Yang, M.; Allard, L.F.; Flytzani-Stephanopoulos, M.; Sykes, E.C.H. Selective hydrogenation of 1,3-butadiene on platinum–copper alloys at the single-atom limit. Nat. Commun. 2015, 6, 8550. [Google Scholar] [CrossRef]
- Wei, H.; Liu, X.; Wang, A.; Zhang, L.; Qiao, B.; Yang, X.; Huang, Y.; Miao, S.; Liu, J.; Zhang, T. FeOx-supported platinum single-atom and pseudo-single-atom catalysts for chemoselective hydrogenation of functionalized nitroarenes. Nat. Commun. 2014, 5, 5634. [Google Scholar] [CrossRef]
- Zhang, Z.; Liu, J.; Wang, J.; Wang, Q.; Wang, Y.; Wang, K.; Wang, Z.; Gu, M.; Tang, Z.; Lim, J.; et al. Single-atom catalyst for high-performance methanol oxidation. Nat. Commun. 2021, 12, 5235. [Google Scholar] [CrossRef]
- Flytzani-Stephanopoulos, M. Gold Atoms Stabilized on Various Supports Catalyze the Water–Gas Shift Reaction. Acc. Chem. Res. 2014, 47, 783–792. [Google Scholar] [CrossRef]
- Yang, M.; Liu, J.; Lee, S.; Zugic, B.; Huang, J.; Allard, L.F.; Flytzani-Stephanopoulos, M. A Common Single-Site Pt(II)–O(OH)x– Species Stabilized by Sodium on “Active” and “Inert” Supports Catalyzes the Water-Gas Shift Reaction. J. Am. Chem. Soc. 2015, 137, 3470–3473. [Google Scholar] [CrossRef]
- Islam, M.J.; Granollers Mesa, M.; Osatiashtiani, A.; Manayil, J.C.; Isaacs, M.A.; Taylor, M.J.; Tsatsos, S.; Kyriakou, G. PdCu single atom alloys supported on alumina for the selective hydrogenation of furfural. Appl. Catal. B Environ. 2021, 299, 120652. [Google Scholar] [CrossRef]
- Mäki-Arvela, P.; Tiainen, L.-P.; Neyestanaki, A.K.; Sjöholm, R.; Rantakylä, T.-K.; Laine, E.; Salmi, T.; Murzin, D.Y. Liquid phase hydrogenation of citral: Suppression of side reactions. Appl. Catal. A Gen. 2002, 237, 181–200. [Google Scholar] [CrossRef]
- Gallezot, P.; Richard, D. Selective Hydrogenation of α,β-Unsaturated Aldehydes. Catal. Rev. Sci. Eng. 1998, 40, 81–126. [Google Scholar] [CrossRef]
- McInroy, A.R.; Uhl, A.; Lear, T.; Klapötke, T.M.; Shaikhutdinov, S.; Schauermann, S.; Rupprechter, G.; Freund, H.-J.; Lennon, D. Morphological and chemical influences on alumina-supported palladium catalysts active for the gas phase hydrogenation of crotonaldehyde. J. Chem. Phys. 2011, 134, 214704. [Google Scholar] [CrossRef]
- Shi, W.; Zhang, L.; Ni, Z.; Xiao, X.; Xia, S. Adsorption and hydrogenation mechanism of crotonaldehyde on a Pd(111) surface by periodic DFT calculations. RSC Adv. 2014, 4, 27003–27012. [Google Scholar] [CrossRef]
- Luneau, M.; Lim, J.S.; Patel, D.A.; Sykes, E.C.H.; Friend, C.M.; Sautet, P. Guidelines to Achieving High Selectivity for the Hydrogenation of α,β-Unsaturated Aldehydes with Bimetallic and Dilute Alloy Catalysts: A Review. Chem. Rev. 2020, 120, 12834–12872. [Google Scholar] [CrossRef]
- Chiu, M.E.; Watson, D.J.; Kyriakou, G.; Tikhov, M.S.; Lambert, R.M. Tilt the Molecule and Change the Chemistry: Mechanism of S-Promoted Chemoselective Catalytic Hydrogenation of Crotonaldehyde on Cu(111). Angew. Chem. Int. Ed. 2006, 45, 7530–7534. [Google Scholar] [CrossRef]
- Hutchings, G.J.; King, F.; Okoye, I.P.; Rochester, C.H. Influence of sulphur poisoning of copper/alumina catalyst on the selective hydrogenation of crotonaldehyde. Appl. Catal. A Gen. 1992, 83, L7–L13. [Google Scholar] [CrossRef]
- Mäki-Arvela, P.; Hájek, J.; Salmi, T.; Murzin, D.Y. Chemoselective hydrogenation of carbonyl compounds over heterogeneous catalysts. Appl. Catal. A Gen. 2005, 292, 1–49. [Google Scholar] [CrossRef]
- Tamura, M.; Tokonami, K.; Nakagawa, Y.; Tomishige, K. Selective Hydrogenation of Crotonaldehyde to Crotyl Alcohol over Metal Oxide Modified Ir Catalysts and Mechanistic Insight. ACS Catal. 2016, 6, 3600–3609. [Google Scholar] [CrossRef]
- Taylor, M.J.; Durndell, L.J.; Isaacs, M.A.; Parlett, C.M.A.; Wilson, K.; Lee, A.F.; Kyriakou, G. Highly selective hydrogenation of furfural over supported Pt nanoparticles under mild conditions. Appl. Catal. B Environ. 2016, 180, 580–585. [Google Scholar] [CrossRef]
- Budroni, G.; Kondrat, S.A.; Taylor, S.H.; Morgan, D.J.; Carley, A.F.; Williams, P.B.; Hutchings, G.J. Selective deposition of palladium onto supported nickel—Bimetallic catalysts for the hydrogenation of crotonaldehyde. Catal. Sci. Technol. 2013, 3, 2746–2754. [Google Scholar] [CrossRef]
- Campo, B.C.; Volpe, M.A.; Gigola, C.E. Liquid-Phase Hydrogenation of Crotonaldehyde over Platinum- and Palladium-Based Catalysts. Ind. Eng. Chem. 2009, 48, 10234–10239. [Google Scholar] [CrossRef]
- Chiu, M.E.; Kyriakou, G.; Williams, F.J.; Watson, D.J.; Tikhov, M.S.; Lambert, R.M. Sulfur, normally a poison, strongly promotes chemoselective catalytic hydrogenation: Stereochemistry and reactivity of crotonaldehyde on clean and S-modified Cu(111). Chem. Commun. 2006, 12, 1283–1285. [Google Scholar] [CrossRef]
- Harraz, F.A.; El-Hout, S.E.; Killa, H.M.; Ibrahim, I.A. Catalytic hydrogenation of crotonaldehyde and oxidation of benzene over active and recyclable palladium nanoparticles stabilized by polyethylene glycol. J. Mol. Catal. A Chem. 2013, 370, 182–188. [Google Scholar] [CrossRef]
- Kun, I.; Szöllösi, G.; Bartók, M. Crotonaldehyde hydrogenation over clay-supported platinum catalysts. J. Mol. Catal. A Chem. 2001, 169, 235–246. [Google Scholar] [CrossRef]
- Tian, L.; Yang, Q.; Jiang, Z.; Zhu, Y.; Pei, Y.; Qiao, M.; Fan, K. Highly chemoselective hydrogenation of crotonaldehyde over Ag–In/SBA-15 fabricated by a modified “two solvents” strategy. Chem. Commun. 2011, 47, 6168–6170. [Google Scholar] [CrossRef]
- Wang, X.-X.; Zheng, H.-Y.; Liu, X.-J.; Xie, G.-Q.; Lu, J.-Q.; Jin, L.-Y.; Luo, M.-F. Effects of NaCl on Pt/ZrO2 catalysts for selective hydrogenation of crotonaldehyde. Appl. Catal. A Gen. 2010, 388, 134–140. [Google Scholar] [CrossRef]
- Yu, J.; Yang, Y.; Chen, L.; Li, Z.; Liu, W.; Xu, E.; Zhang, Y.; Hong, S.; Zhang, X.; Wei, M. NiBi intermetallic compounds catalyst toward selective hydrogenation of unsaturated aldehydes. Appl. Catal. B 2020, 277, 119273. [Google Scholar] [CrossRef]
- Hutchings, G.J.; King, F.; Okoye, I.P.; Padley, M.B.; Rochester, C.H. Modification of Selectivity in the Hydrogenation of Crotonaldehyde Using Cu/Al2O3 Catalysts Modified with Sulfur Compounds: Effect of Sulfur Source. J. Catal. 1994, 148, 464–469. [Google Scholar] [CrossRef]
- Urquhart, A.J.; Williams, F.J.; Vaughan, O.P.H.; Cropley, R.L.; Lambert, R.M. Adsorbate conformation determines catalytic chemoselectivity: Crotonaldehyde on the Pt(111) surface. Chem. Commun. 2005, 15, 1977–1979. [Google Scholar] [CrossRef]
- Noller, H.; Lin, W.M. Activity and selectivity of Ni-CuAl2O3 catalysts for hydrogenation of crotonaldehyde and mechanism of hydrogenation. J. Catal. 1984, 85, 25–30. [Google Scholar] [CrossRef]
- Rao, R.; Dandekar, A.; Baker, R.T.K.; Vannice, M.A. Properties of Copper Chromite Catalysts in Hydrogenation Reactions. J. Catal. 1997, 171, 406–419. [Google Scholar] [CrossRef]
- Šimoník, J.; Beránek, L. Mono and bimolecular mechanisms in the catalytic isomerization of crotyl alcohol to butyraldehyde. J. Catal. 1972, 24, 348–351. [Google Scholar] [CrossRef]
- Bi, Q.; Yuan, X.; Lu, Y.; Wang, D.; Huang, J.; Si, R.; Sui, M.; Huang, F. One-Step High-Temperature-Synthesized Single-Atom Platinum Catalyst for Efficient Selective Hydrogenation. Research 2020, 2020, 9140841. [Google Scholar] [CrossRef]
- Kanzaki, M.; Kawaguchi, Y.; Kawasaki, H. Fabrication of Conductive Copper Films on Flexible Polymer Substrates by Low-Temperature Sintering of Composite Cu Ink in Air. ACS Appl. Mater. Interfaces 2017, 9, 20852–20858. [Google Scholar] [CrossRef]
- Koebel, M.M.; Jones, L.C.; Somorjai, G.A. Preparation of size-tunable, highly monodisperse PVP-protected Pt-nanoparticles by seed-mediated growth. J. Nanopart. Res. 2008, 10, 1063–1069. [Google Scholar] [CrossRef]
- Taylor, M.J.; Beaumont, S.K.; Islam, M.J.; Tsatsos, S.; Parlett, C.A.M.; Issacs, M.A.; Kyriakou, G. Atom efficient PtCu bimetallic catalysts and ultra dilute alloys for the selective hydrogenation of furfural. Appl. Catal. B 2021, 284, 119737. [Google Scholar] [CrossRef]
- Sitthisa, S.; Sooknoi, T.; Ma, Y.; Balbuena, P.B.; Resasco, D.E. Kinetics and mechanism of hydrogenation of furfural on Cu/SiO2 catalysts. J. Catal. 2011, 277, 1–13. [Google Scholar] [CrossRef]
- Lesiak, M.; Binczarski, M.; Karski, S.; Maniukiewicz, W.; Rogowski, J.; Szubiakiewicz, E.; Berlowska, J.; Dziugan, P.; Witońska, I. Hydrogenation of furfural over Pd–Cu/Al2O3 catalysts. The role of interaction between palladium and copper on determining catalytic properties. J. Mol. Catal. A Chem. 2014, 395, 337–348. [Google Scholar] [CrossRef]
- Batista, J.; Pintar, A.; Mandrino, D.; Jenko, M.; Martin, V. XPS and TPR examinations of γ-alumina-supported Pd-Cu catalysts. Appl. Catal. A Gen. 2001, 206, 113–124. [Google Scholar] [CrossRef]
- Cárdenas-Lizana, F.; Gómez-Quero, S.; Amorim, C.; Keane, M.A. Gas phase hydrogenation of p-chloronitrobenzene over Pd–Ni/Al2O3. Appl. Catal. A Gen. 2014, 473, 41–50. [Google Scholar] [CrossRef]
- Parker, S.F.; Walker, H.C.; Callear, S.K.; Grünewald, E.; Petzold, T.; Wolf, D.; Möbus, K.; Adam, J.; Wieland, S.D.; Jiménez-Ruiz, M.; et al. The effect of particle size, morphology and support on the formation of palladium hydride in commercial catalysts. Chem. Sci. 2019, 10, 480–489. [Google Scholar] [CrossRef] [PubMed]
- Kyriakou, G.; Boucher, M.B.; Jewell, A.D.; Lewis, E.A.; Lawton, T.J.; Baber, A.E.; Tierney, H.L.; Flytzani-Stephanopoulos, M.; Sykes, E.C.H. Isolated metal atom geometries as a strategy for selective heterogeneous hydrogenations. Science 2012, 335, 1209–1212. [Google Scholar] [CrossRef]

| Catalyst | Pd Loading (wt.%) | Cu Loading (wt.%) | Pd:Cu Atomic Ratio | Particle Size (nm) | Cu Dispersion (%) | Pd Dispersion (%) | EXAFS Designation |
|---|---|---|---|---|---|---|---|
| Cu100 | - | 0.9403 ± 0.0267 | - | 2.7 ± 0.7 | 71.0 ± 7.1 | - | - |
| Pd1Cu234 | 0.0064 ± 0.0006 | 0.8947 ± 0.0253 | 1: 234 | 2.6 ± 0.7 | 79.9 ± 8.0 | N/A | - |
| Pd1Cu216 | 0.0067 ± 0.0006 | 0.8599 ± 0.0262 | 1: 216 | 2.0 ± 0.6 | 80.3 ± 8.0 | 90.9 ± 9.1 | SAA |
| Pd1Cu53 | 0.0296 ± 0.0022 | 0.9296 ± 0.0232 | 1: 53 | 7.0 ± 4.4 | 68.9 ± 6.9 | 41.7 ± 4.2 | SAA |
| Pd100 | 0.8882 ± 0.0529 | - | - | 5.1 ± 2.7 | - | 22.9 ± 2.3 | - |
| Catalyst | Conversion (%) | Butanal S (%) | Butanol S (%) | Crotyl Alcohol S (%) | Acetal S (%) |
|---|---|---|---|---|---|
| Cu100 | 98.1 ± 4.9 | 93.1 ± 4.7 | 3.9 ± 0.2 | 1.5 ± 0.1 | 1.6 ± 0.1 |
| Pd1Cu234 | 99.3 ± 5.0 | 84.3 ± 4.4 | 13.3 ± 0.5 | 1.5 ± 0.1 | 1.0 ± 0.1 |
| Pd1Cu216 | 99.7 ± 5.0 | 75.1 ± 3.8 | 22.0 ± 1.1 | 1.6 ± 0.1 | 1.3 ± 0.1 |
| Pd1Cu53 | 99.0 ± 5.0 | 77.4 ± 4.1 | 20.8 ± 0.8 | 1.2 ± 0.1 | 0.6 ± 0.1 |
| Pd100 | 99.2 ± 5.0 | 62.8 ± 3.1 | 37.2 ± 1.9 | 0.0 | 0.0 |
| Blank | 4.7 ± 0.2 | 0.0 | 0.0 | 0.0 | 100 ± 5.0 |
| Al2O3 | 1.3 ± 0.1 | 0.0 | 0.0 | 0.0 | 100 ± 5.0 |
| Catalyst | Conversion (%) | Butanal S (%) | Butanol S (%) | Crotyl Alcohol S (%) | Acetal S (%) |
|---|---|---|---|---|---|
| Cu100 1 | 98.1 ± 4.9 | 93.1 ± 4.7 | 3.9 ± 0.2 | 1.5 ± 0.1 | 1.6 ± 0.1 |
| Cu100 2 | 98.1 ± 4.9 | 94.4 ± 4.7 | 1.9 ± 0.1 | 1.6 ± 0.1 | 2.1 ± 0.1 |
| Pd1Cu216 1 | 99.7 ± 5.0 | 75.1 ± 3.8 | 22.0 ± 1.1 | 1.6 ± 0.1 | 1.3 ± 0.1 |
| Pd1Cu216 2 | 99.2 ± 5.0 | 73.7 ± 3.7 | 23.7 ± 1.2 | 1.5 ± 0.1 | 1.2 ± 0.1 |
| Pd1Cu53 1 | 99.0 ± 5.0 | 77.4 ± 4.1 | 20.8 ± 0.8 | 1.2 ± 0.1 | 0.6 ± 0.1 |
| Pd1Cu53 2 | 99.0 ± 5.0 | 77.8 ± 4.2 | 18.2 ± 0.6 | 1.6 ± 0.1 | 2.3 ± 0.1 |
| Pd100 1 | 99.7 ± 5.0 | 62.8 ± 3.1 | 37.2 ± 1.9 | 0.0 | 0.0 |
| Pd100 2 | 100.0 ± 5.0 | 65.3 ± 3.3 | 34.7 ± 1.7 | 0.0 | 0.0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Islam, M.J.; Granollers Mesa, M.; Osatiashtiani, A.; Taylor, M.J.; Isaacs, M.A.; Kyriakou, G. The Hydrogenation of Crotonaldehyde on PdCu Single Atom Alloy Catalysts. Nanomaterials 2023, 13, 1434. https://doi.org/10.3390/nano13081434
Islam MJ, Granollers Mesa M, Osatiashtiani A, Taylor MJ, Isaacs MA, Kyriakou G. The Hydrogenation of Crotonaldehyde on PdCu Single Atom Alloy Catalysts. Nanomaterials. 2023; 13(8):1434. https://doi.org/10.3390/nano13081434
Chicago/Turabian StyleIslam, Mohammed J., Marta Granollers Mesa, Amin Osatiashtiani, Martin J. Taylor, Mark A. Isaacs, and Georgios Kyriakou. 2023. "The Hydrogenation of Crotonaldehyde on PdCu Single Atom Alloy Catalysts" Nanomaterials 13, no. 8: 1434. https://doi.org/10.3390/nano13081434
APA StyleIslam, M. J., Granollers Mesa, M., Osatiashtiani, A., Taylor, M. J., Isaacs, M. A., & Kyriakou, G. (2023). The Hydrogenation of Crotonaldehyde on PdCu Single Atom Alloy Catalysts. Nanomaterials, 13(8), 1434. https://doi.org/10.3390/nano13081434









