Recent Progress of 2D Layered Materials in Water-in-Salt/Deep Eutectic Solvent-Based Liquid Electrolytes for Supercapacitors
Abstract
1. Introduction
2. Types of Supercapacitors
2.1. Supercapacitors with EDLC Behavior
2.2. Pseudocapacitance-Based Supercapacitors
2.3. Hybrid Supercapacitors
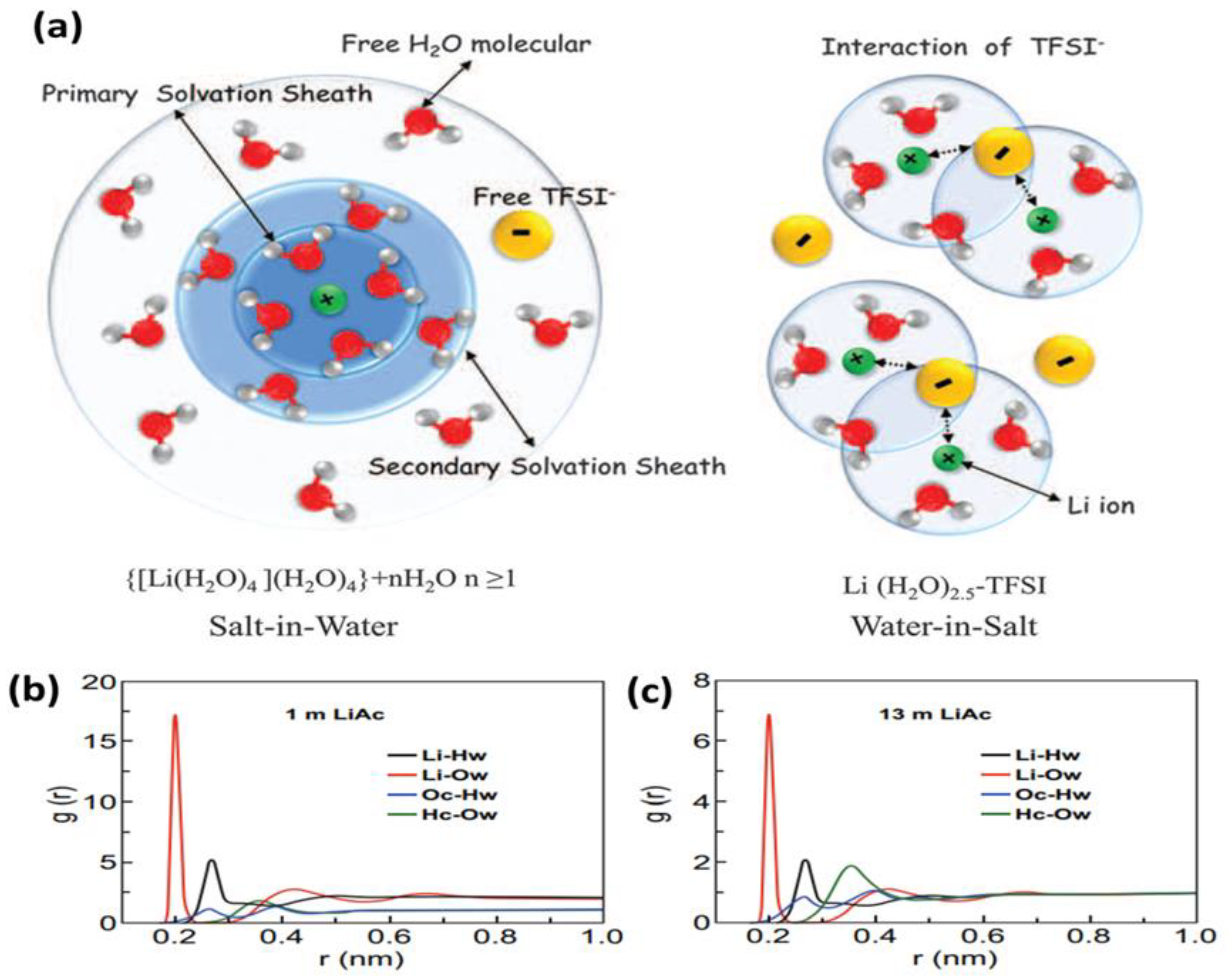
3. Unconventional Aqueous Electrolytes
3.1. Water-in-Salt Electrolytes
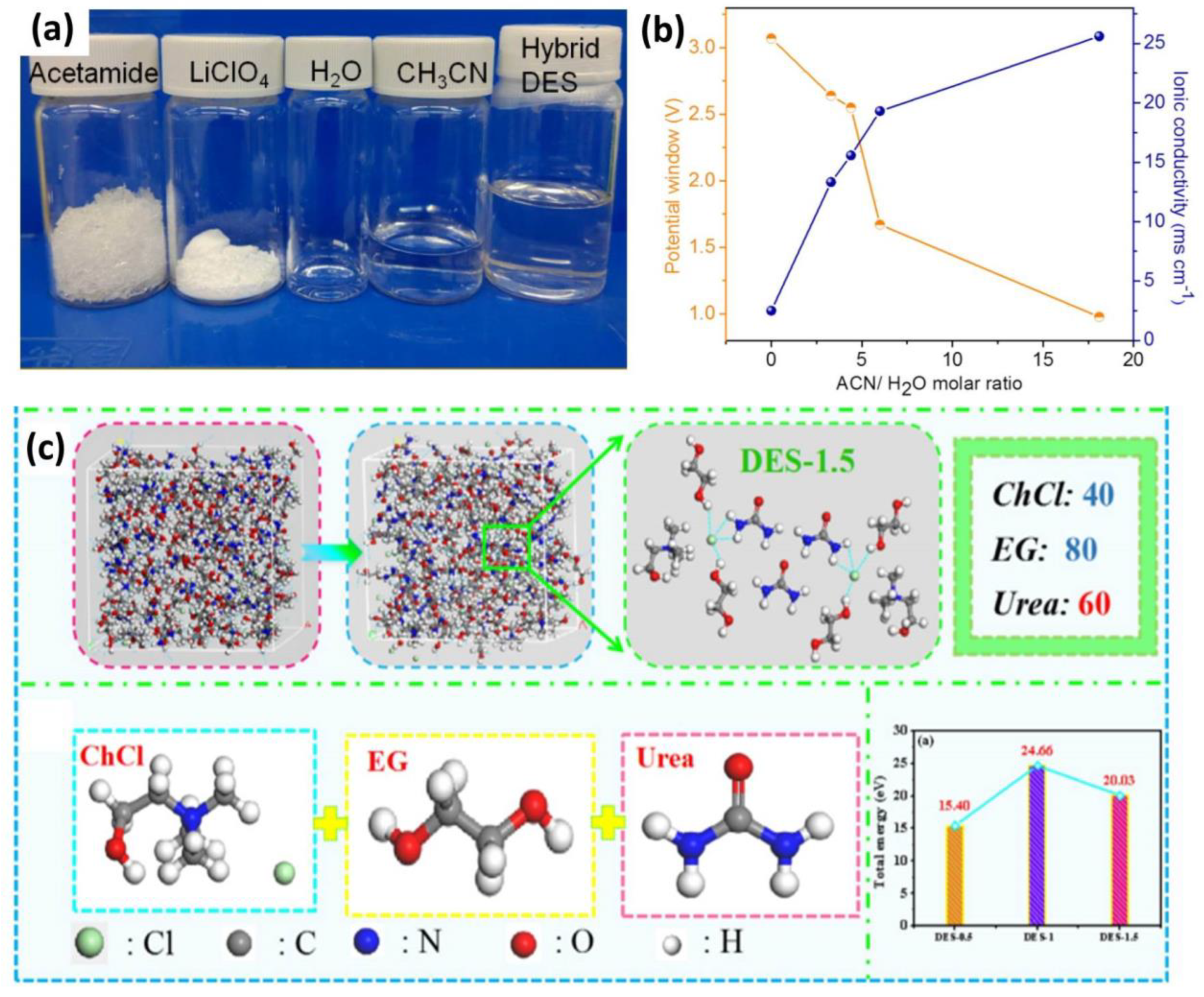
3.2. Deep Eutectic Solvents Electrolytes
4. Supercapacitor Performance of Two-Dimensional (2D) Electrode Material in Novel Aqueous Electrolytes
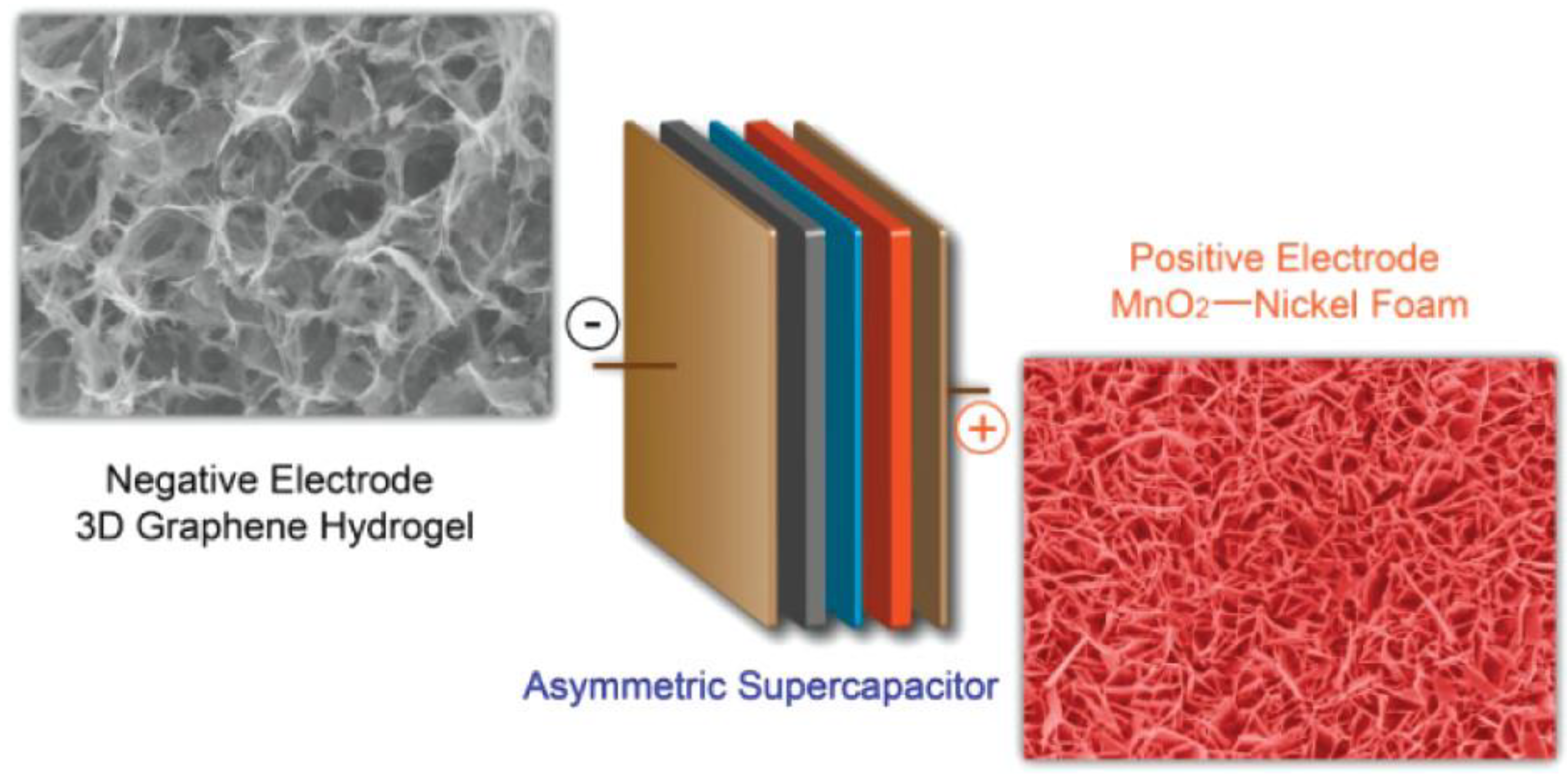
4.1. Graphene and Graphene Derivatives
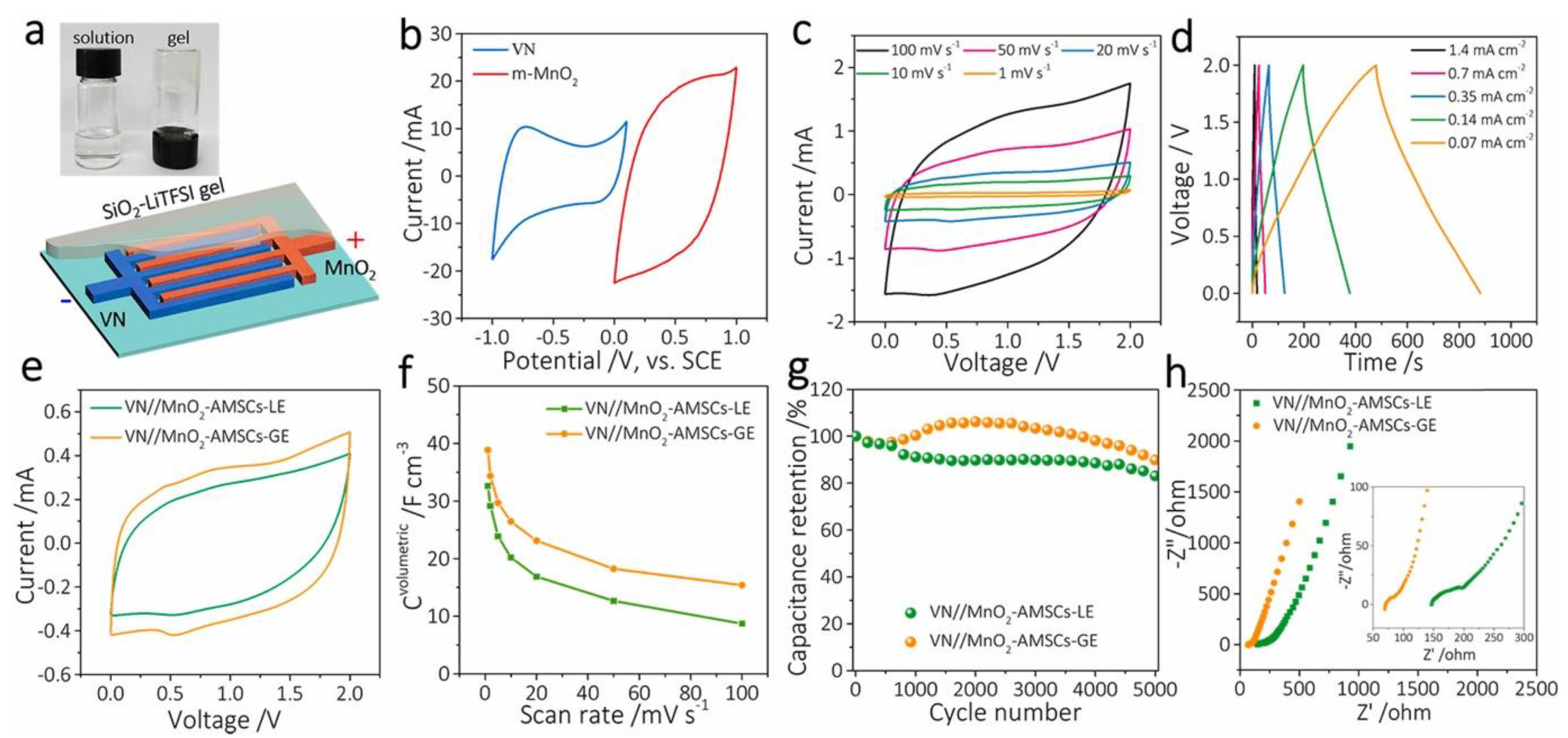
4.2. Metal Oxides and Sulfides
4.3. MXenes
4.4. Two-Dimensional Composite Materials
5. Conclusions and Future Perspectives
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Liu, Y.; Xu, X.; Shao, Z.; Jiang, S.P. Metal-organic frameworks derived porous carbon, metal oxides and metal sulfides-based compounds for supercapacitors application. Energy Storage Mater. 2020, 26, 1–22. [Google Scholar] [CrossRef]
- Koçak, B.; Fernandez, A.I.; Paksoy, H. Review on sensible thermal energy storage for industrial solar applications and sustainability aspects. Sol. Energy 2020, 209, 135–169. [Google Scholar] [CrossRef]
- Zhong, Y.; Xia, X.; Shi, F.; Zhan, J.; Tu, J.; Fan, H.J. Transition Metal Carbides and Nitrides in Energy Storage and Conversion. Adv. Sci. 2016, 3, 1500286. [Google Scholar] [CrossRef] [PubMed]
- Saleem, A.M.; Desmaris, V.; Enoksson, P. Performance Enhancement of Carbon Nanomaterials for Supercapacitors. J. Nanomater. 2016, 2016, 1537269. [Google Scholar] [CrossRef]
- Meng, C.; Gall, O.Z.; Irazoqui, P.P. A flexible super-capacitive solid-state power supply for miniature implantable medical devices. Biomed. Microdevices 2013, 15, 973–983. [Google Scholar] [CrossRef]
- dos Santos, R.B.; Rivelino, R.; Gueorguiev, G.K.; Kakanakova-Georgieva, A. Exploring 2D structures of indium oxide of different stoichiometry. CrystEngComm 2021, 23, 6661–6667. [Google Scholar] [CrossRef]
- Medeiros, P.V.C.; Gueorguiev, G.K.; Stafström, S. Bonding, charge rearrangement and interface dipoles of benzene, graphene, and PAH molecules on Au(111) and Cu(111). Carbon N. Y. 2015, 81, 620–628. [Google Scholar] [CrossRef]
- Dubey, R.; Guruviah, V. Review of carbon-based electrode materials for supercapacitor energy storage. Ionics 2019, 25, 1419–1445. [Google Scholar] [CrossRef]
- Lakshmi, K.C.S.; Vedhanarayanan, B.; Cheng, H.Y.; Ji, X.; Shen, H.H.; Lin, T.W. Molecularly engineered organic copolymers as high capacity cathode materials for aqueous proton battery operating at sub-zero temperatures. J. Colloid Interface Sci. 2022, 619, 123–131. [Google Scholar] [CrossRef]
- Vinodh, R.; Sasikumar, Y.; Kim, H.J.; Atchudan, R.; Yi, M. Chitin and chitosan based biopolymer derived electrode materials for supercapacitor applications: A critical review. J. Ind. Eng. Chem. 2021, 104, 155–171. [Google Scholar] [CrossRef]
- Vinodh, R.; Babu, R.S.; Sambasivam, S.; Muralee Gopi, C.V.V.; Alzahmi, S.; Kim, H.J.; de Barros, A.L.F.; Obaidat, I.M. Recent Advancements of Polyaniline/Metal Organic Framework (PANI/MOF) Composite Electrodes for Supercapacitor Applications: A Critical Review. Nanomaterials 2022, 12, 1511. [Google Scholar] [CrossRef] [PubMed]
- Winter, M.; Brodd, R.J. What are batteries, fuel cells, and supercapacitors? Chem. Rev. 2004, 104, 4245–4269. [Google Scholar] [CrossRef]
- Qi, D.; Liu, Y.; Liu, Z.; Zhang, L.; Chen, X. Design of Architectures and Materials in In-Plane Micro-supercapacitors: Current Status and Future Challenges. Adv. Mater. 2017, 29, 1602802. [Google Scholar] [CrossRef] [PubMed]
- Lim, J.M.; Jang, Y.S.; Van, T. Nguyen, H.; Kim, J.S.; Yoon, Y.; Park, B.J.; Seo, D.H.; Lee, K.K.; Han, Z.; Ostrikov, K.; et al. Advances in High-Voltage Supercapacitors for Energy Storage Systems: Materials and Electrolyte Tailoring to Implementation. Nanoscale Adv. 2023, 5, 615–626. [Google Scholar] [CrossRef]
- González, A.; Goikolea, E.; Barrena, J.A.; Mysyk, R. Review on supercapacitors: Technologies and materials. Renew. Sustain. Energy Rev. 2016, 58, 1189–1206. [Google Scholar] [CrossRef]
- Lu, X.; Jiménez-Riobóo, R.J.; Leech, D.; Gutiérrez, M.C.; Ferrer, M.L.; Del Monte, F. Aqueous-Eutectic-in-Salt Electrolytes for High-Energy-Density Supercapacitors with an Operational Temperature Window of 100 °C, from-35 to +65 °C. ACS Appl. Mater. Interfaces 2020, 12, 29181–29193. [Google Scholar] [CrossRef] [PubMed]
- Huang, M.K.; Anuratha, K.S.; Xiao, Y.; Chen, Y.P.; Lin, J.Y. Co-solvent modified methylsulfonylmethane-based hybrid deep eutectic solvent electrolytes for high-voltage symmetric supercapacitors. Electrochim. Acta 2022, 424, 140612. [Google Scholar] [CrossRef]
- Jayalakshmi, M.; Balasubramanian, K. Simple capacitors to supercapacitors—An overview. Int. J. Electrochem. Sci. 2008, 3, 1196–1217. [Google Scholar]
- Wang, Y.; Song, Y.; Xia, Y. Electrochemical capacitors: Mechanism, materials, systems, characterization and applications. Chem. Soc. Rev. 2016, 45, 5925–5950. [Google Scholar] [CrossRef]
- Ratajczak, P.; Suss, M.E.; Kaasik, F.; Béguin, F. Carbon electrodes for capacitive technologies. Energy Storage Mater. 2019, 16, 126–145. [Google Scholar] [CrossRef]
- Fleischmann, S.; Mitchell, J.B.; Wang, R.; Zhan, C.; Jiang, D.E.; Presser, V.; Augustyn, V. Pseudocapacitance: From Fundamental Understanding to High Power Energy Storage Materials. Chem. Rev. 2020, 120, 6738–6782. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Zhang, L.; Zhang, J. A review of electrode materials for electrochemical supercapacitors. Chem. Soc. Rev. 2012, 41, 797–828. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Chang, H.; Zhang, M.; Chen, Y. Graphene-based materials for lithium-ion hybrid supercapacitors. Adv. Mater. 2015, 27, 5296–5308. [Google Scholar] [CrossRef]
- Chen, P.; Xiao, T.Y.; Qian, Y.H.; Li, S.S.; Yu, S.H. A nitrogen-doped graphene/carbon nanotube nanocomposite with synergistically enhanced electrochemical activity. Adv. Mater. 2013, 25, 3192–3196. [Google Scholar] [CrossRef]
- Wen, Z.; Wang, X.; Mao, S.; Bo, Z.; Kim, H.; Cui, S.; Lu, G.; Feng, X.; Chen, J. Crumpled nitrogen-doped graphene nanosheets with ultrahigh pore volume for high-performance supercapacitor. Adv. Mater. 2012, 24, 5610–5616. [Google Scholar] [CrossRef]
- Yang, J.; Zhou, X.Y.; Li, J.; Zou, Y.L.; Tang, J.J. Study of nano-porous hard carbons as anode materials for lithium ion batteries. Mater. Chem. Phys. 2012, 135, 445–450. [Google Scholar] [CrossRef]
- Subramanian, V.; Luo, C.; Stephan, A.M.; Nahm, K.S.; Thomas, S.; Wei, B. Supercapacitors from activated carbon derived from banana fibers. J. Phys. Chem. C 2007, 111, 7527–7531. [Google Scholar] [CrossRef]
- Li, J.G.; Ho, Y.F.; Ahmed, M.M.M.; Liang, H.C.; Kuo, S.W. Mesoporous Carbons Templated by PEO-PCL Block Copolymers as Electrode Materials for Supercapacitors. Chem.-A Eur. J. 2019, 25, 10456–10463. [Google Scholar] [CrossRef]
- Deng, T.; Zhang, W.; Arcelus, O.; Kim, J.G.; Carrasco, J.; Yoo, S.J.; Zheng, W.; Wang, J.; Tian, H.; Zhang, H.; et al. Atomic-level energy storage mechanism of cobalt hydroxide electrode for pseudocapacitors. Nat. Commun. 2017, 8, 15194. [Google Scholar] [CrossRef]
- Najib, S.; Erdem, E. Current progress achieved in novel materials for supercapacitor electrodes: Mini review. Nanoscale Adv. 2019, 1, 2817–2827. [Google Scholar] [CrossRef]
- Poudel, M.B.; Kim, H.J. Confinement of Zn-Mg-Al-layered double hydroxide and α-Fe2O3 nanorods on hollow porous carbon nanofibers: A free-standing electrode for solid-state symmetric supercapacitors. Chem. Eng. J. 2022, 429, 132345. [Google Scholar] [CrossRef]
- Chen, K.; Dong Noh, Y.; Li, K.; Komarneni, S.; Xue, D. Microwave-hydrothermal crystallization of polymorphic MnO2 for electrochemical energy storage. J. Phys. Chem. C 2013, 117, 10770–10779. [Google Scholar] [CrossRef]
- Jiang, Q.; Kurra, N.; Alhabeb, M.; Gogotsi, Y.; Alshareef, H.N. All Pseudocapacitive MXene-RuO2 Asymmetric Supercapacitors. Adv. Energy Mater. 2018, 8, 1703043. [Google Scholar] [CrossRef]
- Yu, F.; Pang, L.; Wang, H.X. Preparation of mulberry-like RuO2 electrode material for supercapacitors. Rare Met. 2021, 40, 440–447. [Google Scholar] [CrossRef]
- Kumar, A.; Thomas, A.; Garg, M.; Perumal, G.; Grewal, H.S.; Arora, H.S. High performance CuO@brass supercapacitor electrodes through surface activation. J. Mater. Chem. A 2021, 9, 9327–9336. [Google Scholar] [CrossRef]
- Rakhi, R.B.; Chen, W.; Cha, D.; Alshareef, H.N. Substrate dependent self-organization of mesoporous cobalt oxide nanowires with remarkable pseudocapacitance. Nano Lett. 2012, 12, 2559–2567. [Google Scholar] [CrossRef] [PubMed]
- Shakir, I.; Shahid, M.; Rana, U.A.; Al Nashef, I.M.; Hussain, R. Nickel–Cobalt Layered Double Hydroxide Anchored Zinc Oxide Nanowires grown on Carbon Fiber Cloth for High-Performance Flexible Pseudocapacitive Energy Storage Devices. Electrochim. Acta 2014, 129, 28–32. [Google Scholar] [CrossRef]
- Li, H.; Chen, J.; Zhang, L.; Wang, K.; Zhang, X.; Yang, B.; Liu, L.; Liu, W.; Yan, X. A metal-organic framework-derived pseudocapacitive titanium oxide/carbon core/shell heterostructure for high performance potassium ion hybrid capacitors. J. Mater. Chem. A 2020, 8, 16302–16311. [Google Scholar] [CrossRef]
- Seok, J.Y.; Lee, J.; Yang, M. Self-Generated Nanoporous Silver Framework for High-Performance Iron Oxide Pseudocapacitor Anodes. ACS Appl. Mater. Interfaces 2018, 10, 17223–17231. [Google Scholar] [CrossRef]
- Cong, S.; Tian, Y.; Li, Q.; Zhao, Z.; Geng, F. Single-crystalline tungsten oxide quantum dots for fast pseudocapacitor and electrochromic applications. Adv. Mater. 2014, 26, 4260–4267. [Google Scholar] [CrossRef]
- Wang, J.; Zheng, F.; Yu, Y.; Hu, P.; Li, M.; Wang, J.; Fu, J.; Zhen, Q.; Bashir, S.; Liu, J.L. Symmetric supercapacitors composed of ternary metal oxides (NiO/V2O5/MnO2) nanoribbon electrodes with high energy storage performance. Chem. Eng. J. 2021, 426, 131804. [Google Scholar] [CrossRef]
- Kumar M, S.; Yasoda K, Y.; Das, P.; Malik, S.; Kothurkar, N.K.; Batabyal, S.K. Urea-mediated synthesized carbon quantum dots to tune the electrochemical performance of polyaniline nanorods for supercapacitor device. J. Sci. Adv. Mater. Devices 2022, 7, 100403. [Google Scholar] [CrossRef]
- Kumar, M.S.; Das, P.; Yasoda, K.Y.; Kothurkar, N.K.; Malik, S.; Batabyal, S.K. Fabrication of organic nanocomposite of polyaniline for enhanced electrochemical performance. J. Energy Storage 2020, 31, 101700. [Google Scholar] [CrossRef]
- Dhawale, D.S.; Salunkhe, R.R.; Jamadade, V.S.; Dubal, D.P.; Pawar, S.M.; Lokhande, C.D. Hydrophilic polyaniline nanofibrous architecture using electrosynthesis method for supercapacitor application. Curr. Appl. Phys. 2010, 10, 904–909. [Google Scholar] [CrossRef]
- Gupta, V.; Miura, N. Electrochemically Deposited Polyaniline Nanowire’s Network. Electrochem. Solid-State Lett. 2005, 8, A630. [Google Scholar] [CrossRef]
- Bandyopadhyay, P.; Kuila, T.; Balamurugan, J.; Nguyen, T.T.; Kim, N.H.; Lee, J.H. Facile synthesis of novel sulfonated polyaniline functionalized graphene using m-aminobenzene sulfonic acid for asymmetric supercapacitor application. Chem. Eng. J. 2017, 308, 1174–1184. [Google Scholar] [CrossRef]
- Yang, Y.; Hao, Y.; Yuan, J.; Niu, L.; Xia, F. In situ preparation of caterpillar-like polyaniline/carbon nanotube hybrids with core shell structure for high performance supercapacitors. Carbon N. Y. 2014, 78, 279–287. [Google Scholar] [CrossRef]
- Bavio, M.A.; Acosta, G.G.; Kessler, T. Polyaniline and polyaniline-carbon black nanostructures as electrochemical capacitor electrode materials. Int. J. Hydrog. Energy 2014, 39, 8582–8589. [Google Scholar] [CrossRef]
- Zhang, D.; Dong, Q.-Q.; Wang, X.; Yan, W.; Deng, W.; Shi, L.-Y. Preparation of a Three-Dimensional Ordered Macroporous Carbon Nanotube/Polypyrrole Composite for Supercapacitors and Diffusion Modeling. J. Phys. Chem. C 2013, 117, 20446–20455. [Google Scholar] [CrossRef]
- Xu, R.; Wei, J.; Guo, F.; Cui, X.; Zhang, T.; Zhu, H.; Wang, K.; Wu, D. Highly conductive, twistable and bendable polypyrrole–carbon nanotube fiber for efficient supercapacitor electrodes. RSC Adv. 2015, 5, 22015–22021. [Google Scholar] [CrossRef]
- Grover, S.; Shekhar, S.; Sharma, R.K.; Singh, G. Multiwalled carbon nanotube supported polypyrrole manganese oxide composite supercapacitor electrode: Role of manganese oxide dispersion in performance evolution. Electrochim. Acta 2014, 116, 137–145. [Google Scholar] [CrossRef]
- Wang, B.; Qiu, J.; Feng, H.; Sakai, E. Preparation of graphene oxide/polypyrrole/multi-walled carbon nanotube composite and its application in supercapacitors. Electrochim. Acta 2015, 151, 230–239. [Google Scholar] [CrossRef]
- Liu, T.; Finn, L.; Yu, M.; Wang, H.; Zhai, T.; Lu, X.; Tong, Y.; Li, Y. Polyaniline and polypyrrole pseudocapacitor electrodes with excellent cycling stability. Nano Lett. 2014, 14, 2522–2527. [Google Scholar] [CrossRef] [PubMed]
- Lu, Z.; Chang, Z.; Zhu, W.; Sun, X. Beta-phased Ni(OH)2 nanowall film with reversible capacitance higher than theoretical Faradic capacitance. Chem. Commun. 2011, 47, 9651–9653. [Google Scholar] [CrossRef] [PubMed]
- Dubal, D.P.; Ayyad, O.; Ruiz, V.; Gómez-Romero, P. Hybrid Energy Storage: The Merging of Battery and Supercapacitor Chemistries. Chem. Soc. Rev. 2015, 44, 1777–1790. [Google Scholar] [CrossRef]
- Borenstein, A.; Hanna, O.; Attias, R.; Luski, S.; Brousse, T.; Aurbach, D. Carbon-based composite materials for supercapacitor electrodes: A review. J. Mater. Chem. A 2017, 5, 12653–12672. [Google Scholar] [CrossRef]
- Poudel, M.B.; Kim, A.A.; Lohani, P.C.; Yoo, D.J.; Kim, H.J. Assembling zinc cobalt hydroxide/ternary sulfides heterostructure and iron oxide nanorods on three-dimensional hollow porous carbon nanofiber as high energy density hybrid supercapacitor. J. Energy Storage 2023, 60, 106713. [Google Scholar] [CrossRef]
- Gao, H.; Xiao, F.; Ching, C.B.; Duan, H. High-performance asymmetric supercapacitor based on graphene hydrogel and nanostructured MnO 2. ACS Appl. Mater. Interfaces 2012, 4, 2801–2810. [Google Scholar] [CrossRef]
- Prakash, D.; Manivannan, S. Unusual battery type pseudocapacitive behaviour of graphene oxynitride electrode: High energy solid-state asymmetric supercapacitor. J. Alloy. Compd. 2021, 854, 156853. [Google Scholar] [CrossRef]
- Yang, J.; Xu, X.; Zhou, X.; Jiang, S.; Chen, W.; Shi, S.; Wang, D.; Liu, Z. Ultrasmall Co3O4 Nanoparticles Confined in P, N-Doped Carbon Matrices for High-Performance Supercapacitors. J. Phys. Chem. C 2020, 124, 9225–9232. [Google Scholar] [CrossRef]
- Jiang, W.; Hu, F.; Yan, Q.; Wu, X. Investigation on electrochemical behaviors of NiCo2O4 battery-type supercapacitor electrodes: The role of an aqueous electrolyte. Inorg. Chem. Front. 2017, 4, 1642–1648. [Google Scholar] [CrossRef]
- Redondo, E.; Goikolea, E.; Mysyk, R. The decisive role of electrolyte concentration in the performance of aqueous chloride-based carbon/carbon supercapacitors with extended voltage window. Electrochim. Acta 2016, 221, 177–183. [Google Scholar] [CrossRef]
- Kurzweil, P.; Chwistek, M. Electrochemical stability of organic electrolytes in supercapacitors: Spectroscopy and gas analysis of decomposition products. J. Power Sources 2008, 176, 555–567. [Google Scholar] [CrossRef]
- Ivol, F.; Porcher, M.; Ghosh, A.; Jacquemin, J. Phenylacetonitrile (C6H5CH2CN) Ionic Liquid Blends as Alternative Electrolytes for Safe and High-Performance Supercapacitors. Molecules 2020, 25, 2697. [Google Scholar] [CrossRef] [PubMed]
- Reece, R.; Lekakou, C.; Smith, P.A. A structural supercapacitor based on activated carbon fabric and a solid electrolyte. Mater. Sci. Technol. 2019, 35, 368–375. [Google Scholar] [CrossRef]
- Moon, W.G.; Kim, G.P.; Lee, M.; Song, H.D.; Yi, J. A biodegradable gel electrolyte for use in high-performance flexible supercapacitors. ACS Appl. Mater. Interfaces 2015, 7, 3503–3511. [Google Scholar] [CrossRef]
- Jayaramulu, K.; Dubal, D.P.; Nagar, B.; Ranc, V.; Tomanec, O.; Petr, M.; Datta, K.K.R.; Zboril, R.; Gómez-Romero, P.; Fischer, R.A. Ultrathin Hierarchical Porous Carbon Nanosheets for High-Performance Supercapacitors and Redox Electrolyte Energy Storage. Adv. Mater. 2018, 30, 1705789. [Google Scholar] [CrossRef]
- Zang, X.; Shen, C.; Sanghadasa, M.; Lin, L. High-Voltage Supercapacitors Based on Aqueous Electrolytes. ChemElectroChem 2019, 6, 976–988. [Google Scholar] [CrossRef]
- Zhao, C.; Zheng, W. A review for aqueous electrochemical supercapacitors. Front. Energy Res. 2015, 3, 23. [Google Scholar] [CrossRef]
- Wu, J.; Liang, Q.; Yu, X.; Qiu-Feng, L.; Ma, L.; Qin, X.; Chen, G.; Li, B. Deep Eutectic Solvents for Boosting Electrochemical Energy Storage and Conversion: A Review and Perspective. Adv. Funct. Mater. 2021, 31, 2011102. [Google Scholar] [CrossRef]
- Zaidi, W.; Boisset, A.; Jacquemin, J.; Timperman, L.; Anouti, M. Deep eutectic solvents based on N-methylacetamide and a lithium salt as electrolytes at elevated temperature for activated carbon-based supercapacitors. J. Phys. Chem. C 2014, 118, 4033–4042. [Google Scholar] [CrossRef]
- Suo, L.; Borodin, O.; Gao, T.; Olguin, M.; Ho, J.; Fan, X.; Luo, C.; Wang, C.; Xu, K. “Water-in-salt” electrolyte enables high-voltage aqueous lithium-ion chemistries. Science 2015, 350, 938–943. [Google Scholar] [CrossRef]
- Da Silva, D.A.C.; Pinzón C., M. J.; Messias, A.; Fileti, E.E.; Pascon, A.; Franco, D.V.; Da Silva, L.M.; Zanin, H.G. Effect of conductivity, viscosity, and density of water-in-salt electrolytes on the electrochemical behavior of supercapacitors: Molecular dynamics simulations and: In situ characterization studies. Mater. Adv. 2022, 3, 611–623. [Google Scholar] [CrossRef]
- Swallow, J.E.N.; Fraser, M.W.; Kneusels, N.J.H.; Charlton, J.F.; Sole, C.G.; Phelan, C.M.E.; Björklund, E.; Bencok, P.; Escudero, C.; Pérez-Dieste, V.; et al. Revealing solid electrolyte interphase formation through interface-sensitive Operando X-ray absorption spectroscopy. Nat. Commun. 2022, 13, 6070. [Google Scholar] [CrossRef] [PubMed]
- Xie, J.; Guan, Y.; Huang, Y.; Lu, Y.C. Solid-Electrolyte Interphase of Molecular Crowding Electrolytes. Chem. Mater. 2022, 34, 5176–5183. [Google Scholar] [CrossRef]
- Shen, Y.; Liu, B.; Liu, X.; Liu, J.; Ding, J.; Zhong, C.; Hu, W. Water-in-salt electrolyte for safe and high-energy aqueous battery. Energy Storage Mater. 2021, 34, 461–474. [Google Scholar] [CrossRef]
- Amiri, M.; Bélanger, D. Physicochemical and Electrochemical Properties of Water-in-Salt Electrolytes. ChemSusChem 2021, 14, 2487–2500. [Google Scholar] [CrossRef]
- Lakshmi, K.C.S.; Ji, X.; Chen, T.Y.; Vedhanarayanan, B.; Lin, T.W. Pseudocapacitive and battery-type organic polymer electrodes for a 1.9 V hybrid supercapacitor with a record concentration of ammonium acetate. J. Power Sources 2021, 511, 230434. [Google Scholar] [CrossRef]
- Thareja, S.; Kumar, A. “Water-In-Salt” Electrolyte-Based High-Voltage (2.7 V) Sustainable Symmetric Supercapacitor with Superb Electrochemical Performance—An Analysis of the Role of Electrolytic Ions in Extending the Cell Voltage. ACS Sustain. Chem. Eng. 2021, 9, 2338–2347. [Google Scholar] [CrossRef]
- Seetha Lakshmi, K.C.; Ji, X.; Shao, L.D.; Vedhanarayanan, B.; Lin, T.W. Tailor-made organic polymers towards high voltage aqueous ammonium/potassium-ion asymmetric supercapacitors. Appl. Surf. Sci. 2022, 577, 151918. [Google Scholar] [CrossRef]
- Meng, C.; Zhou, F.; Liu, H.; Zhu, Y.; Fu, Q.; Wu, Z.S. Water-in-Salt Ambipolar Redox Electrolyte Extraordinarily Boosting High Pseudocapacitive Performance of Micro-supercapacitors. ACS Energy Lett. 2022, 7, 1706–1711. [Google Scholar] [CrossRef]
- Dong, S.; Wang, Y.; Chen, C.; Shen, L.; Zhang, X. Niobium Tungsten Oxide in a Green Water-in-Salt Electrolyte Enables Ultra-Stable Aqueous Lithium-Ion Capacitors. Nano-Micro Lett. 2020, 12, 168. [Google Scholar] [CrossRef] [PubMed]
- Navarro-Suárez, A.M.; Johansson, P. Perspective—Semi-Solid Electrolytes Based on Deep Eutectic Solvents: Opportunities and Future Directions. J. Electrochem. Soc. 2020, 167, 070511. [Google Scholar] [CrossRef]
- Tomé, L.I.N.; Baião, V.; da Silva, W.; Brett, C.M.A. Deep eutectic solvents for the production and application of new materials. Appl. Mater. Today 2018, 10, 30–50. [Google Scholar] [CrossRef]
- Ünlü, A.E.; Arıkaya, A.; Takaç, S. Use of deep eutectic solvents as catalyst: A mini-review. Green Process. Synth. 2019, 8, 355–372. [Google Scholar] [CrossRef]
- Smith, E.L.; Abbott, A.P.; Ryder, K.S. Deep Eutectic Solvents (DESs) and Their Applications. Chem. Rev. 2014, 114, 11060–11082. [Google Scholar] [CrossRef]
- Zainal-Abidin, M.H.; Hayyan, M.; Hayyan, A.; Jayakumar, N.S. New horizons in the extraction of bioactive compounds using deep eutectic solvents: A review. Anal. Chim. Acta 2017, 979, 1–23. [Google Scholar] [CrossRef]
- Di Pietro, M.E.; Mele, A. Deep eutectics and analogues as electrolytes in batteries. J. Mol. Liq. 2021, 338, 116597. [Google Scholar] [CrossRef]
- Zhang, C.; Zhang, L.; Yu, G. Eutectic Electrolytes as a Promising Platform for Next-Generation Electrochemical Energy Storage. Acc. Chem. Res. 2020, 53, 1648–1659. [Google Scholar] [CrossRef]
- Lien, C.W.; Vedhanarayanan, B.; Chen, J.H.; Lin, J.Y.; Tsai, H.H.; Shao, L.D.; Lin, T.W. Optimization of acetonitrile/water content in hybrid deep eutectic solvent for graphene/MoS2 hydrogel-based supercapacitors. Chem. Eng. J. 2021, 405, 126706. [Google Scholar] [CrossRef]
- Zhong, M.; Tang, Q.F.; Qiu, Z.G.; Wang, W.P.; Chen, X.Y.; Zhang, Z.J. A novel electrolyte of ternary deep eutectic solvent for wide temperature region supercapacitor with superior performance. J. Energy Storage 2020, 32, 101904. [Google Scholar] [CrossRef]
- Tran, K.T.; Truong, T.T.T.; Nguyen, H.V.; Nguyen, Q.D.; Phung, Q.; Le, P.M.L.; Tran, M.V. Hybrid Deep Eutectic Solvent of LiTFSI-Ethylene Glycol Organic Electrolyte for Activated Carbon-Based Supercapacitors. J. Chem. 2021, 2021, 9940750. [Google Scholar] [CrossRef]
- Forouzandeh, P.; Pillai, S.C. Two-dimensional (2D) electrode materials for supercapacitors. Mater. Today Proc. 2021, 41, 498–505. [Google Scholar] [CrossRef]
- Tatlisu, A.; Huang, Z.; Chen, R. High-Voltage and Low-Temperature Aqueous Supercapacitor Enabled by “Water-in-Imidazolium Chloride” Electrolytes. ChemSusChem 2018, 11, 3899–3904. [Google Scholar] [CrossRef] [PubMed]
- Khademi, B.; Nateghi, M.R.; Shayesteh, M.R.; Nasirizadeh, N. High voltage binder free hybrid supercapacitor based on reduced graphene oxide/graphene oxide electrodes and “water in salt” electrolyte. J. Energy Storage 2021, 43, 103164. [Google Scholar] [CrossRef]
- Choudhury, B.J.; Ingtipi, K.; Moholkar, V.S. Improved energy density of reduced graphene oxide based aqueous symmetric supercapacitors in redox-active and “water-in-salt” electrolytes. J. Energy Storage 2022, 52, 105006. [Google Scholar] [CrossRef]
- Zhang, M.; Li, Y.; Shen, Z. “Water-in-salt” electrolyte enhanced high voltage aqueous supercapacitor with all-pseudocapacitive metal-oxide electrodes. J. Power Sources 2019, 414, 479–485. [Google Scholar] [CrossRef]
- Zhang, M.; Makino, S.; Mochizuki, D.; Sugimoto, W. High-performance hybrid supercapacitors enabled by protected lithium negative electrode and “water-in-salt” electrolyte. J. Power Sources 2018, 396, 498–505. [Google Scholar] [CrossRef]
- Zhu, Y.; Zheng, S.; Lu, P.; Ma, J.; Das, P.; Su, F.; Cheng, H.M.; Wu, Z.S. Kinetic regulation of MXene with water-in-LiCl electrolyte for high-voltage micro-supercapacitors. Natl. Sci. Rev. 2022, 9, nwac024. [Google Scholar] [CrossRef]
- Avireddy, H.; Byles, B.W.; Pinto, D.; Delgado Galindo, J.M.; Biendicho, J.J.; Wang, X.; Flox, C.; Crosnier, O.; Brousse, T.; Pomerantseva, E.; et al. Stable high-voltage aqueous pseudocapacitive energy storage device with slow self-discharge. Nano Energy 2019, 64, 103961. [Google Scholar] [CrossRef]
- Wang, X.; Mathis, T.S.; Sun, Y.; Tsai, W.Y.; Shpigel, N.; Shao, H.; Zhang, D.; Hantanasirisakul, K.; Malchik, F.; Balke, N.; et al. Titanium Carbide MXene Shows an Electrochemical Anomaly in Water-in-Salt Electrolytes. ACS Nano 2021, 15, 15274–15284. [Google Scholar] [CrossRef]
- Tsai, H.H.; Lin, T.J.; Vedhanarayanan, B.; Tsai, C.C.; Chen, T.Y.; Ji, X.; Lin, T.W. A 1.9-V all-organic battery-supercapacitor hybrid device with high rate capability and wide temperature tolerance in a metal-free water-in-salt electrolyte. J. Colloid Interface Sci. 2022, 612, 76–87. [Google Scholar] [CrossRef]
- Tsai, Y.R.; Vedhanarayanan, B.; Chen, T.Y.; Lin, Y.C.; Lin, J.Y.; Ji, X.; Lin, T.W. A tailor-made deep eutectic solvent for 2.2 V wide temperature-tolerant supercapacitors via optimization of N,N-dimethylformamide/water co-solvents. J. Power Sources 2022, 521, 230954. [Google Scholar] [CrossRef]
- Qin, J.; Wang, S.; Zhou, F.; Das, P.; Zheng, S.; Sun, C.; Bao, X.; Wu, Z.S. 2D mesoporous MnO2 nanosheets for high-energy asymmetric micro-supercapacitors in water-in-salt gel electrolyte. Energy Storage Mater. 2019, 18, 397–404. [Google Scholar] [CrossRef]
- Sahoo, R.; Lee, T.H.; Pham, D.T.; Luu, T.H.T.; Lee, Y.H. Fast-Charging High-Energy Battery-Supercapacitor Hybrid: Anodic Reduced Graphene Oxide-Vanadium(IV) Oxide Sheet-on-Sheet Heterostructure. ACS Nano 2019, 13, 10776–10786. [Google Scholar] [CrossRef]
- Long, C.; Miao, L.; Zhu, D.; Duan, H.; Lv, Y.; Li, L.; Liu, M.; Gan, L. Adapting a Kinetics-Enhanced Carbon Nanostructure to Li/Na Hybrid Water-in-Salt Electrolyte for High-Energy Aqueous Supercapacitors. ACS Appl. Energy Mater. 2021, 4, 5727–5737. [Google Scholar] [CrossRef]
- Tsai, H.-Y.; Kumar, M.S.; Vedhanarayanan, B.; Shen, H.-H.; Lin, T.-W. Urea-Based Deep Eutectic Solvent with Magnesium/Lithium Dual Ions as an Aqueous Electrolyte for High-Performance Battery-Supercapacitor Hybrid Devices. Batteries 2023, 9, 69. [Google Scholar] [CrossRef]
- Song, X.F.; Hu, J. Two-dimensional semiconductors: Recent progress and future perspectives. J. Mater. Chem. C 2013, 1, 2952–2969. [Google Scholar] [CrossRef]
- Lai, L.; Chen, L.; Zhan, D.; Sun, L.; Liu, J.; Lim, S.H.; Poh, C.K.; Shen, Z.; Lin, J. One-step synthesis of NH2-graphene from in situ graphene-oxide reduction and its improved electrochemical properties. Carbon N. Y. 2011, 49, 3250–3257. [Google Scholar] [CrossRef]
- Chen, Z.; Ren, W.; Gao, L.; Liu, B.; Pei, S.; Cheng, H.M. Three-dimensional flexible and conductive interconnected graphene networks grown by chemical vapour deposition. Nat. Mater. 2011, 10, 424–428. [Google Scholar] [CrossRef]
- Ossonon, B.D.; Bélanger, D. Synthesis and characterization of sulfophenyl-functionalized reduced graphene oxide sheets. RSC Adv. 2017, 7, 27224–27234. [Google Scholar] [CrossRef]
- Jayasena, B.; Subbiah, S. A novel mechanical cleavage method for synthesizing few-layer graphenes. Nanoscale Res. Lett. 2011, 6, 95. [Google Scholar] [CrossRef] [PubMed]
- Štengl, V.; Henych, J.; Bludská, J.; Ecorchard, P.; Kormunda, M. A green method of graphene preparation in an alkaline environment. Ultrason. Sonochem. 2015, 24, 65–71. [Google Scholar] [CrossRef] [PubMed]
- Konios, D.; Stylianakis, M.M.; Stratakis, E.; Kymakis, E. Dispersion behaviour of graphene oxide and reduced graphene oxide. J. Colloid Interface Sci. 2014, 430, 108–112. [Google Scholar] [CrossRef]
- Tkachev, S.V.; Buslaeva, E.Y.; Naumkin, A.V.; Kotova, S.L.; Laure, I.V.; Gubin, S.P. Reduced Graphene Oxide. Inorg. Mater. 2012, 48, 796–802. [Google Scholar] [CrossRef]
- Sun, Z.; Yan, Z.; Yao, J.; Beitler, E.; Zhu, Y.; Tour, J.M. Growth of graphene from solid carbon sources. Nature 2010, 468, 549–552. [Google Scholar] [CrossRef]
- Narula, U.; Tan, C.M. Determining the parameters of importance of a graphene synthesis process using design-of-experiments method. Appl. Sci. 2016, 6, 204. [Google Scholar] [CrossRef]
- Tan, Y.B.; Lee, J.M. Graphene for supercapacitor applications. J. Mater. Chem. A 2013, 1, 14814–14843. [Google Scholar] [CrossRef]
- Chhowalla, M.; Shin, H.S.; Eda, G.; Li, L.J.; Loh, K.P.; Zhang, H. The chemistry of two-dimensional layered transition metal dichalcogenide nanosheets. Nat. Chem. 2013, 5, 263–275. [Google Scholar] [CrossRef]
- Tanwar, S.; Arya, A.; Gaur, A.; Sharma, A.L. Transition metal dichalcogenide (TMDs) electrodes for supercapacitors: A comprehensive review. J. Phys. Condens. Matter 2021, 33, 303002. [Google Scholar] [CrossRef]
- Hu, M.; Zhang, H.; Hu, T.; Fan, B.; Wang, X.; Li, Z. Emerging 2D MXenes for supercapacitors: Status, challenges and prospects. Chem. Soc. Rev. 2020, 49, 6666–6693. [Google Scholar] [CrossRef] [PubMed]
- Panda, S.; Deshmukh, K.; Khadheer Pasha, S.K.; Theerthagiri, J.; Manickam, S.; Choi, M.Y. MXene based emerging materials for supercapacitor applications: Recent advances, challenges, and future perspectives. Coord. Chem. Rev. 2022, 462, 214518. [Google Scholar] [CrossRef]

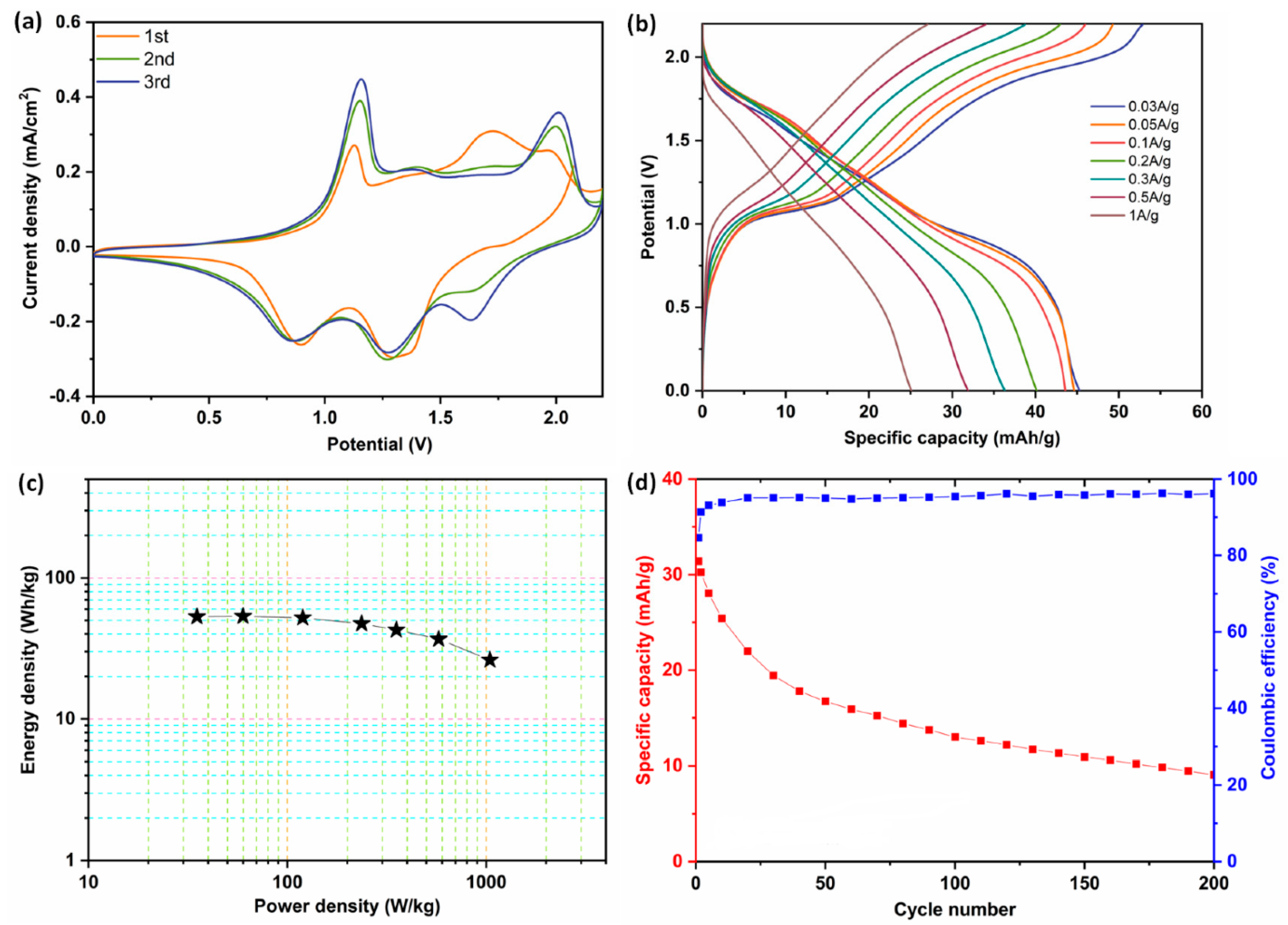
| Materials | Electrolyte | Specific Capacitance | Energy Density | Power Density | Cyclic Stability | Ref. |
|---|---|---|---|---|---|---|
| graphene nanoplatelets-coated carbon paper | 20 M 1-butyl-3-methylimidazolium chloride + 0.1 M 4-hydroxy-2,2,6,6-tetramethyl piperidin-1-oxyl | 480 F g−1 at 20 mV s−1 | 110 Wh kg−1 | 10 kW kg−1 | 2.56% capacitance loss after 500 cycles | [94] |
| reduced graphene oxide and graphene oxide | 17 m NaClO4 | 59.7 F g−1 | 43.8 Wh kg−1 | 115.6 W kg−1 | 84% after 10,000 cycles | [95] |
| N-doped RGO | 17 m NaClO4 | 138 F g−1 at 1 A g−1 | 140 Wh kg−1 | 10,125 W kg−1 | 98% after 10,000 cycles | [79] |
| rGO | 11 M NaNO3 | 149.4 F g−1 | 22.87 Wh kg−1 | 210 W kg−1 | 98.1% after 5000 cycles | [96] |
| MnO2 (cathode) and Fe3O4 (anode) | 21 m LiTFSI | 35.5 Wh kg−1 | 2692 W kg−1 | 87% after 3000 cycles | [97] | |
| Nb18W16O93 | 13 m LiAc | 54 mAh g−1 | 41.9 Wh kg−1 | 20 kW kg−1 | 85% after 50,000 cycles | [82] |
| MnO2 | 21 m LiTFSI | 303 F g−1 | 405 Wh kg−1 | 16.7 kW kg−1 | 90% after 3000 cycles | [98] |
| 1T-MoS2 | acetone and water added acetamide and lithium perchlorate | 42.4 F g−1 at 1 A g−1 | 31.2 Wh kg−1 | 5.7 kW kg−1 | 91% after 20,000 cycles | [90] |
| Ti3C2Tx | 20 M LiCl | 89.2 F cm−3 | 33.6 mWh cm−3 | 25 W cm−3 | nearly no capacity decay after 10,000 cycles | [99] |
| Ti3C2 (anode) and MnO2 (cathode) | 21 M potassium acetate | 25 F cm−3 | 16.80 mWh cm−3 | 137 mW cm−3 | 93% after 10,000 cycles | [100] |
| Ti3C2Tx | 19.8 m LiCl | 26 F g−1 | 9.2 Wh kg−1 | 41 W kg−1 | Coulombic efficiency above 95% | [101] |
| PTCDI–rGO | 32 m Ammonium acetate | 165 mAh g−1 | 12.9 Wh kg−1 | 827 W kg−1 | 74% after 3000 cycles | [102] |
| PANI–rGO | N-methyl acetamide and lithium perchlorate in water and DMF | 41.9 F g−1 | 28.2 Wh kg−1 | 5.6 kW kg−1 | 60% after 3000 cycles | [103] |
| VN//MnO2–AMSCs–GE | 5 M LiTFSI | 243 F g−1 | 21.6 mW cm−3 | 1539 mW cm−3 | 90% after 5000 cycles | [104] |
| rGO@ VO2 | 1 M LiPF6 in a 1:1 (v/v) ratio combining ethylene carbonate and diethyl carbonate | 1214 mAh g−1 | 126.7 Wh kg−1 | 10,000 W kg−1 | 80% after 10,000 cycles | [105] |
| carbon nanostructures | LiOTf/NaOTf | 284 F g−1 | 39.2 Wh kg−1 | 22 kW kg−1 | 85.5% after 10,000 cycles | [106] |
| PTCDI/rGO | DES (urea: magnesium chloride: lithium perchlorate: water) | 76.5 mAh g−1 at 0.03 A g−1 | 53 Wh kg−1 | 1042 W kg−1 | Coulombic efficiency is 99% after 200 cycles | [107] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Melethil, K.; Kumar, M.S.; Wu, C.-M.; Shen, H.-H.; Vedhanarayanan, B.; Lin, T.-W. Recent Progress of 2D Layered Materials in Water-in-Salt/Deep Eutectic Solvent-Based Liquid Electrolytes for Supercapacitors. Nanomaterials 2023, 13, 1257. https://doi.org/10.3390/nano13071257
Melethil K, Kumar MS, Wu C-M, Shen H-H, Vedhanarayanan B, Lin T-W. Recent Progress of 2D Layered Materials in Water-in-Salt/Deep Eutectic Solvent-Based Liquid Electrolytes for Supercapacitors. Nanomaterials. 2023; 13(7):1257. https://doi.org/10.3390/nano13071257
Chicago/Turabian StyleMelethil, Krishnakumar, Munusamy Sathish Kumar, Chun-Ming Wu, Hsin-Hui Shen, Balaraman Vedhanarayanan, and Tsung-Wu Lin. 2023. "Recent Progress of 2D Layered Materials in Water-in-Salt/Deep Eutectic Solvent-Based Liquid Electrolytes for Supercapacitors" Nanomaterials 13, no. 7: 1257. https://doi.org/10.3390/nano13071257
APA StyleMelethil, K., Kumar, M. S., Wu, C.-M., Shen, H.-H., Vedhanarayanan, B., & Lin, T.-W. (2023). Recent Progress of 2D Layered Materials in Water-in-Salt/Deep Eutectic Solvent-Based Liquid Electrolytes for Supercapacitors. Nanomaterials, 13(7), 1257. https://doi.org/10.3390/nano13071257