Design of Superhydrophobic CoFe2O4 Solar Seawater Desalination Device and Its Application in Organic Solvent Removal
Abstract
:1. Introduction
2. Experimental Section
2.1. Material
2.2. Preparation of Solar Evaporator
2.3. Characterization
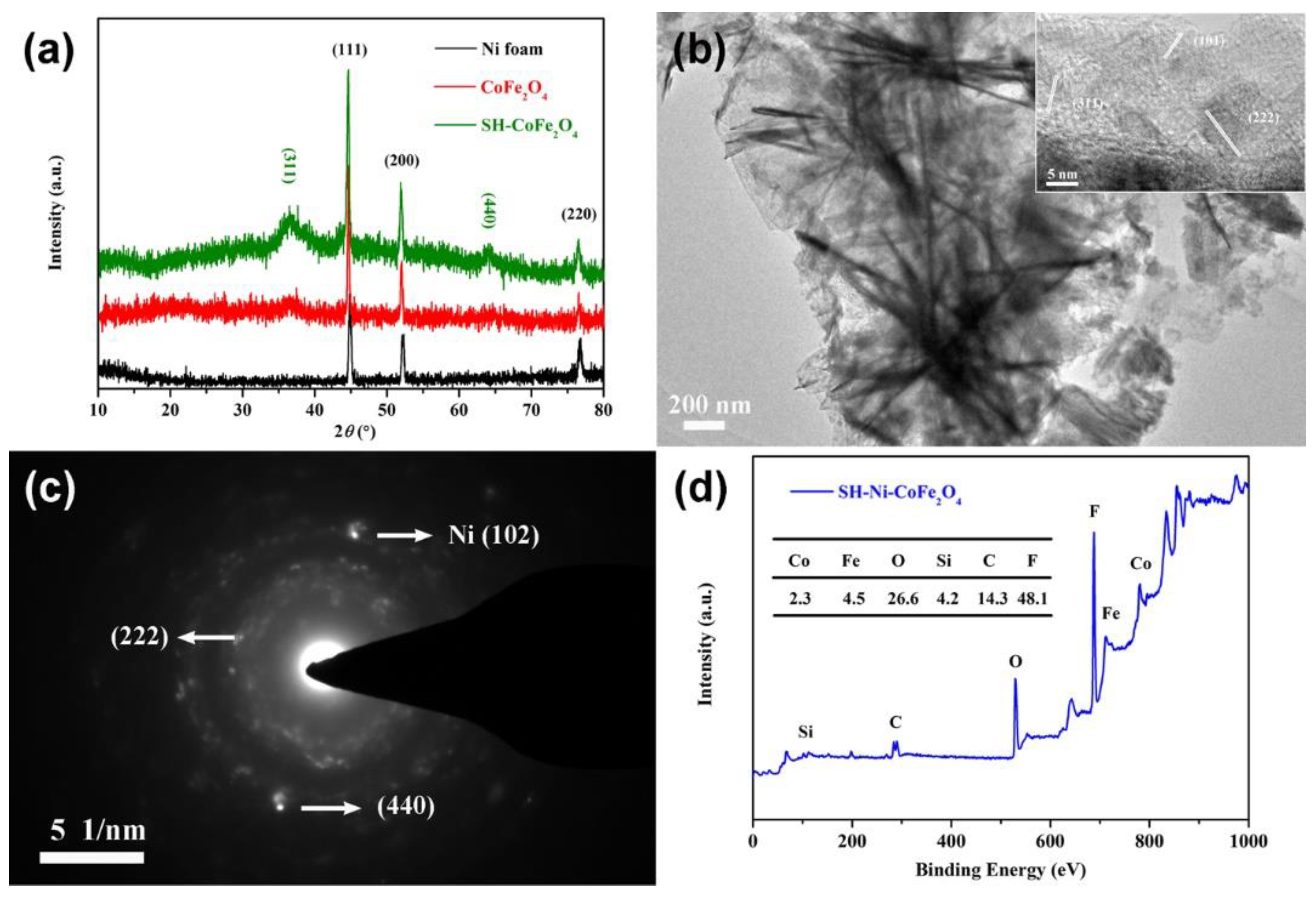
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jia, Y.; Liu, P.B.; Wang, Q.Y.; Wu, Y.; Cao, D.D.; Qiao, Q.A. Construction of Bi2S3-BiOBr nanosheets on TiO2 NTA as the effective photocatalysts: Pollutant removal, photoelectric conversion and hydrogen generation. J. Colloid Interf. Sci. 2021, 585, 459–469. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.S.; Wu, T.; Shen, H.; Yang, S.W.; Qin, Y.; Zhu, Z.M.; Zheng, L.; Wen, X.J.; Xia, M.G.; Yin, X.Z. Flexible MXene-based Janus porous fibrous membranes for sustainable solar-driven desalination and emulsions separation. J. Clean. Prod. 2022, 347, 131324. [Google Scholar] [CrossRef]
- Shahzad, M.W.; Burhan, M.; Ang, L.; Ng, K.C. Energy-water-environment nexus underpinning future desalination sustainability. Desalination 2017, 413, 52. [Google Scholar] [CrossRef]
- Shahzad, M.W.; Burhan, M.; Ng, K.C. Pushing desalination recovery to the maximum limit: Membrane and thermal processes integration. Desalination 2017, 416, 54. [Google Scholar] [CrossRef]
- Lu, X.L.; Eliemlech, M. Fabrication of desalination membranes by interfacial polymerization: History, current efforts, and future directions. Chem. Soc. Rev. 2021, 50, 6290. [Google Scholar] [CrossRef] [PubMed]
- Cheng, S.N.; Li, Y.H.; Jin, B.C.; Yu, Z.; Gu, R.N. Designing salt transmission channel of solar-driven multistage desalination device for efficient and stable freshwater production from seawater. Desalination 2022, 531, 115688. [Google Scholar] [CrossRef]
- Zheng, Z.H.; Liu, H.; Wu, D.Z.; Wang, X.D. Polyimide/MXene hybrid aerogel-based phase-change composites for solar-driven seawater desalination. Chem. Eng. J. 2022, 440, 135862. [Google Scholar] [CrossRef]
- Chen, Y.; Yang, J.; Zhu, L.; Jia, X.H.; Wang, S.Z.; Li, Y.; Song, H.J. An integrated highly hydrated cellulose network with a synergistic photothermal effect for efficient solar-driven water evaporation and salt resistance. J. Mater. Chem. A 2021, 9, 15482–15492. [Google Scholar] [CrossRef]
- Lin, Z.X.; Wu, T.T.; Feng, Y.F.; Shi, J.; Zhou, B.; Zhu, C.H.; Wang, Y.Y.; Liang, R.L.; Mizuno, M. Poly(N-phenylglycine)/MoS2 nanohybrid with synergistic solar-thermal conversion for efficient water purification and thermoelectric power generation. ACS Appl. Mater. Interfaces 2022, 14, 1034–1044. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Ma, R.R.; Zhan, J.L.; Shi, W.S.; Zhu, Y.Y.; Tian, Y.Q. Superhydrophobic/superoleophilic starch-based cryogels coated by silylated porous starch/Fe3O4 hybrid micro/nanoparticles for removing discrete oil patches from water. Sep. Purif. Technol. 2022, 291, 120872. [Google Scholar] [CrossRef]
- Miao, X.; Liu, S.; Wang, M.; Ge, B.; Zhu, C.Q. Fabrication of interface heating composite evaporator for rapid sewage treatment and water purification. Sci. China Technol. Sci. 2021, 64, 2292–2299. [Google Scholar] [CrossRef]
- Liu, Y.D.; Liu, J.H.; Wang, Z.B.; Yuan, Y.K.; Hua, J.; Liu, K. Robust and durable superhydrophobic and oil-absorbent silica particles with ultrahigh separation efficiency and recyclability. Micropor. Mesopor. Mat. 2022, 335, 111772. [Google Scholar] [CrossRef]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ge, X.; Zhou, Z.; Tan, Z.; Wang, S.; Zhao, X.; Ren, G.; Ge, B.; Li, W. Design of Superhydrophobic CoFe2O4 Solar Seawater Desalination Device and Its Application in Organic Solvent Removal. Nanomaterials 2022, 12, 1531. https://doi.org/10.3390/nano12091531
Ge X, Zhou Z, Tan Z, Wang S, Zhao X, Ren G, Ge B, Li W. Design of Superhydrophobic CoFe2O4 Solar Seawater Desalination Device and Its Application in Organic Solvent Removal. Nanomaterials. 2022; 12(9):1531. https://doi.org/10.3390/nano12091531
Chicago/Turabian StyleGe, Xiangcai, Zhijun Zhou, Zheng Tan, Shoufei Wang, Xingchuan Zhao, Guina Ren, Bo Ge, and Wei Li. 2022. "Design of Superhydrophobic CoFe2O4 Solar Seawater Desalination Device and Its Application in Organic Solvent Removal" Nanomaterials 12, no. 9: 1531. https://doi.org/10.3390/nano12091531
APA StyleGe, X., Zhou, Z., Tan, Z., Wang, S., Zhao, X., Ren, G., Ge, B., & Li, W. (2022). Design of Superhydrophobic CoFe2O4 Solar Seawater Desalination Device and Its Application in Organic Solvent Removal. Nanomaterials, 12(9), 1531. https://doi.org/10.3390/nano12091531




