Biomaterials Based on Carbon Nanotube Nanocomposites of Poly(styrene-b-isobutylene-b-styrene): The Effect of Nanotube Content on the Mechanical Properties, Biocompatibility and Hemocompatibility
Abstract
:1. Introduction
2. Materials and Methods
2.1. Synthesis and Characterization of SIBSs
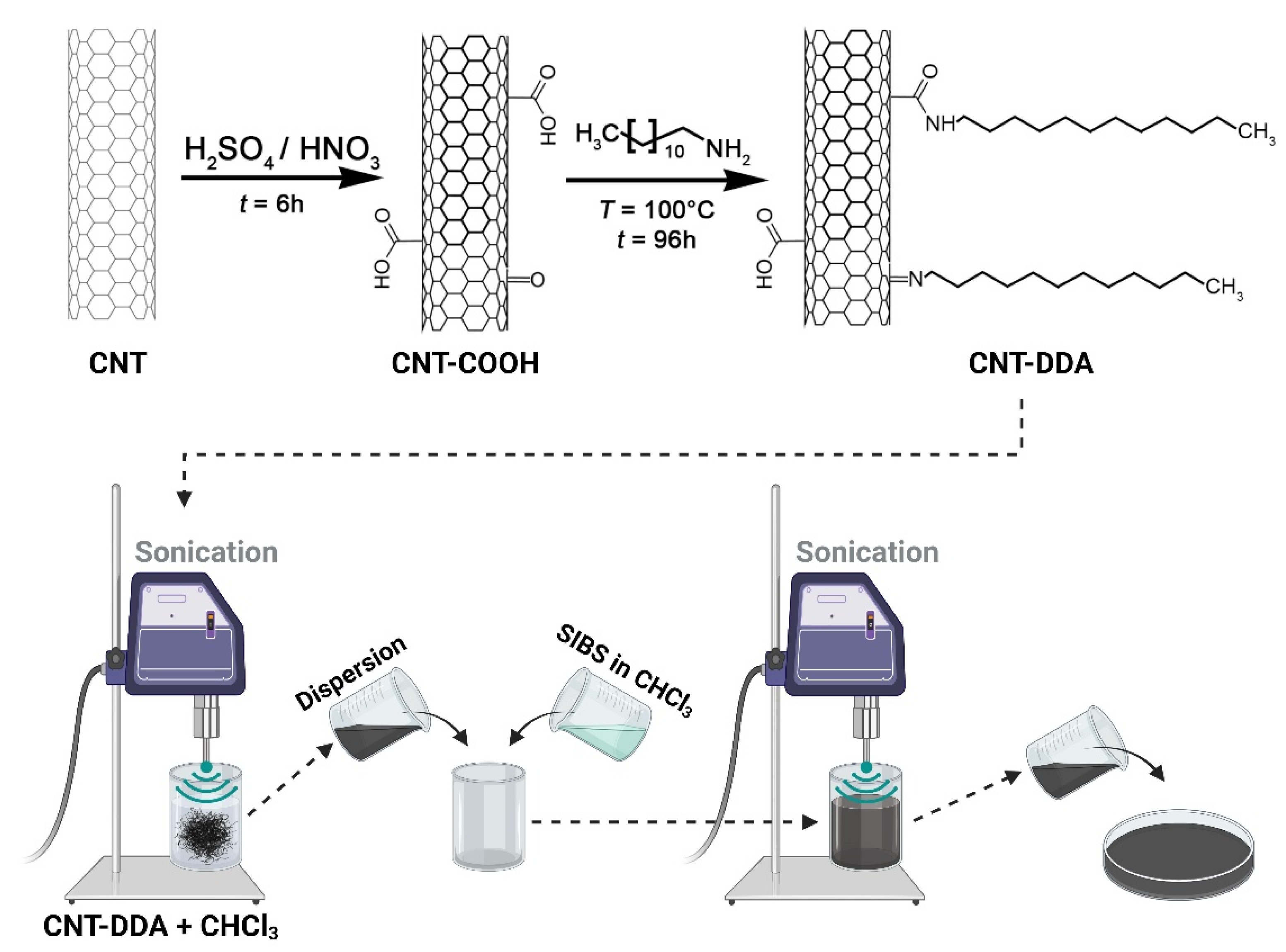
2.2. Modification of CNTs
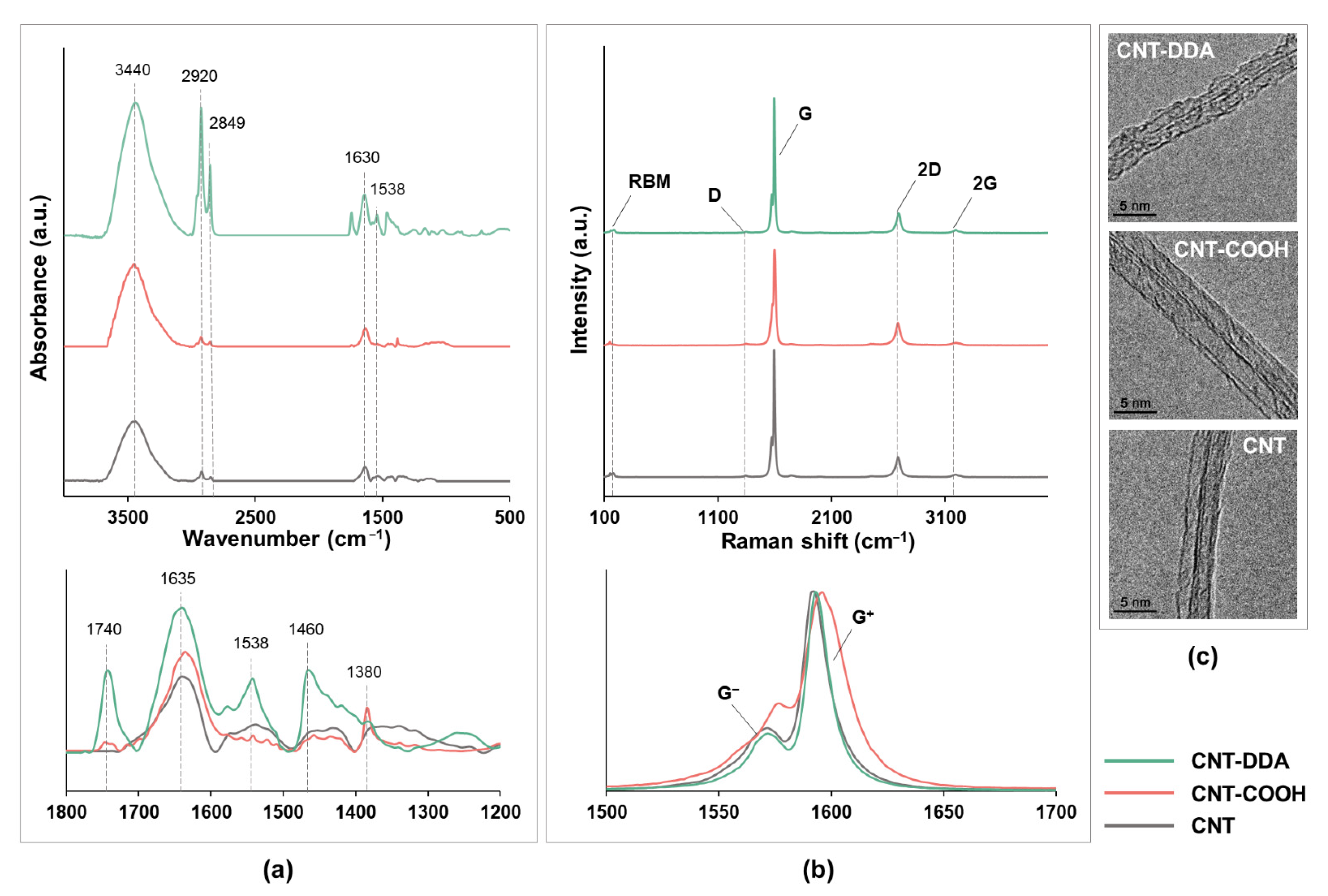
2.3. Characterization of the CNT Structure
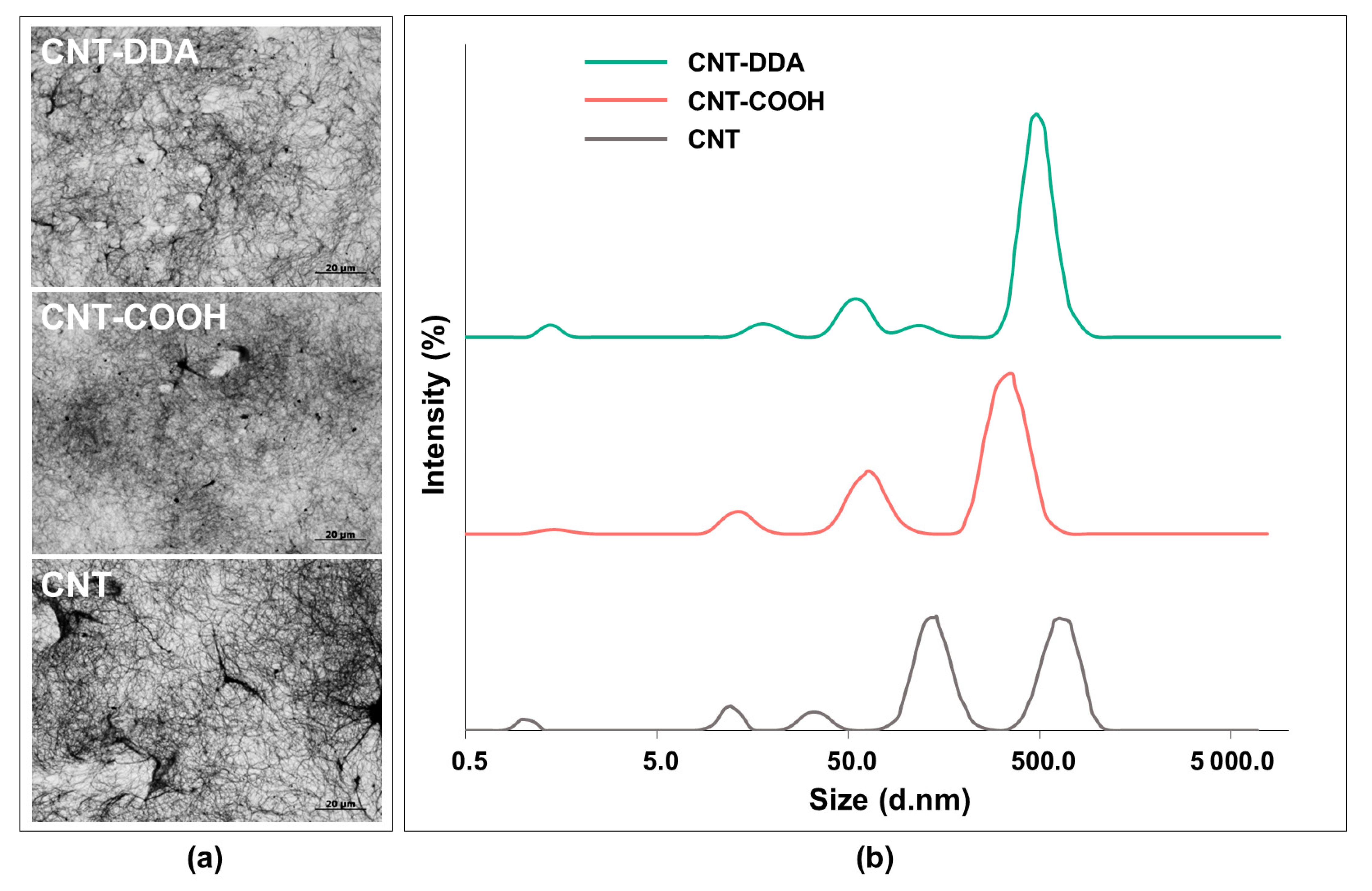
2.4. Synthesis and Analysis of CNT Dispersions in Chloroform
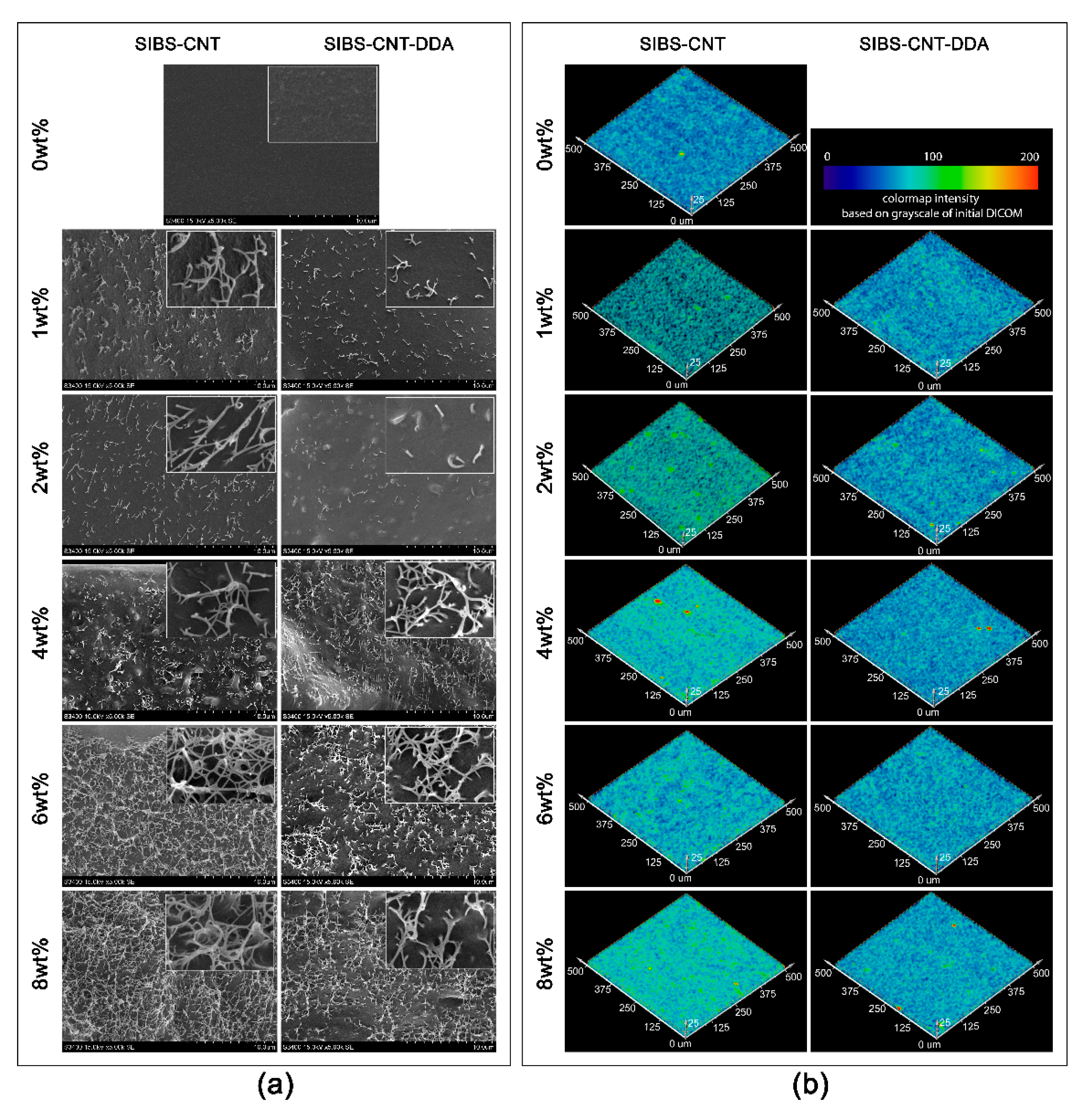
2.5. Preparation of SIBS-CNT Nanocomposites
2.6. Microscopy
2.7. X-ray Microtomography (Micro-CT)
2.8. Thermal Analysis
2.9. Mechanical Tests
2.10. Water Contact Angle Measurement
2.11. Cytotoxicity Assessment
2.12. Platelet Adhesion
- Disc-shaped platelet, no deformation;
- Large platelet with pseudopodia-like protrusions;
- Large irregularly shaped platelet with pseudopods, platelets accumulate in conglomerates;
- Spreading platelet, the cytoplasm spreads and fills the spaces between pseudopods;
- Platelet dense granules, pseudopodia cannot be found due to the proliferation of the cytoplasm.
2.13. Statistical Analysis
3. Results
3.1. Synthesis of SIBS Copolymers
3.2. Characterization of the CNT Structure
3.3. Analysis of CNTs Dispersions in Chloroform
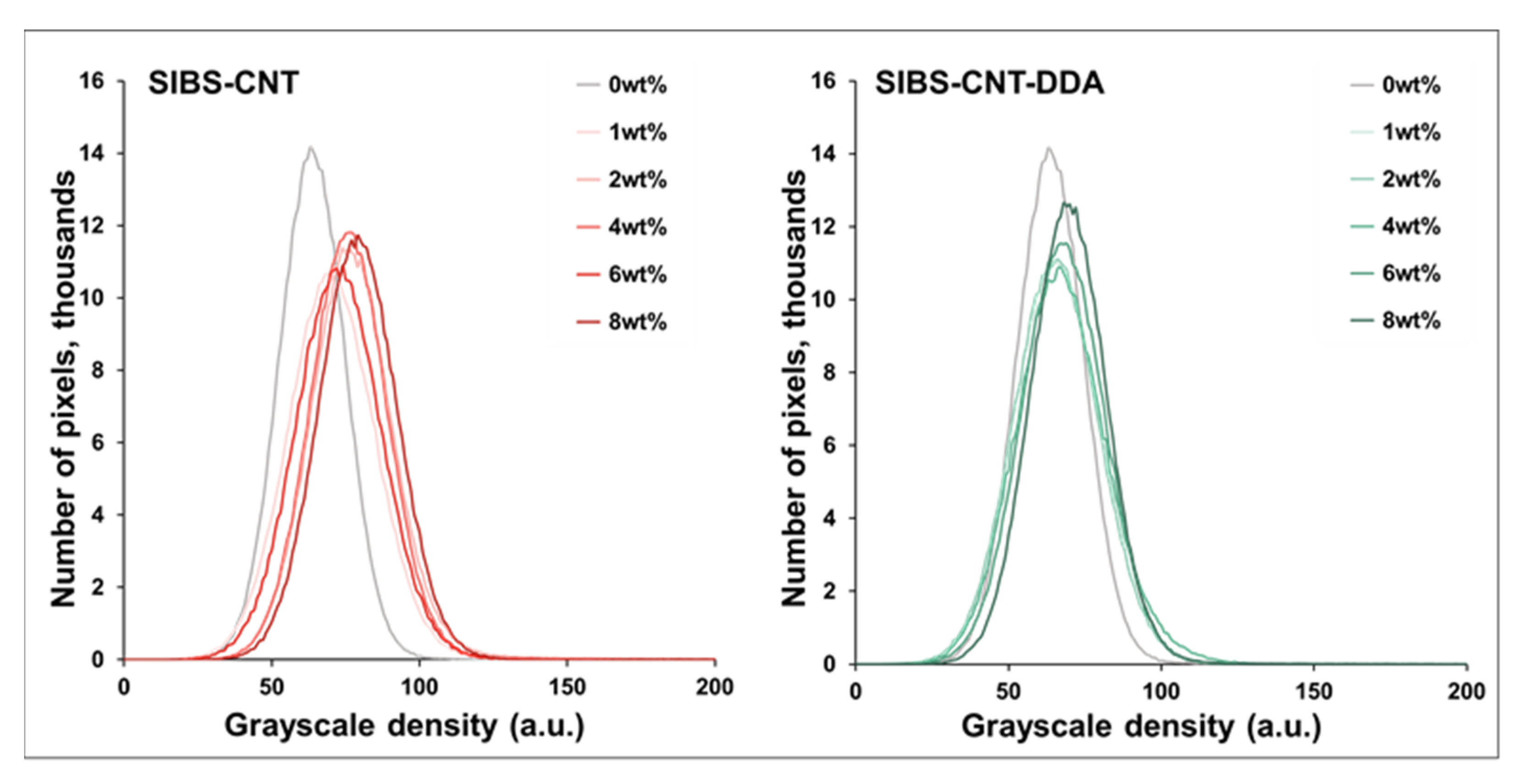
3.4. Analysis of the Structure of the Obtained Nanocomposites
3.5. Thermal Stability Evaluation
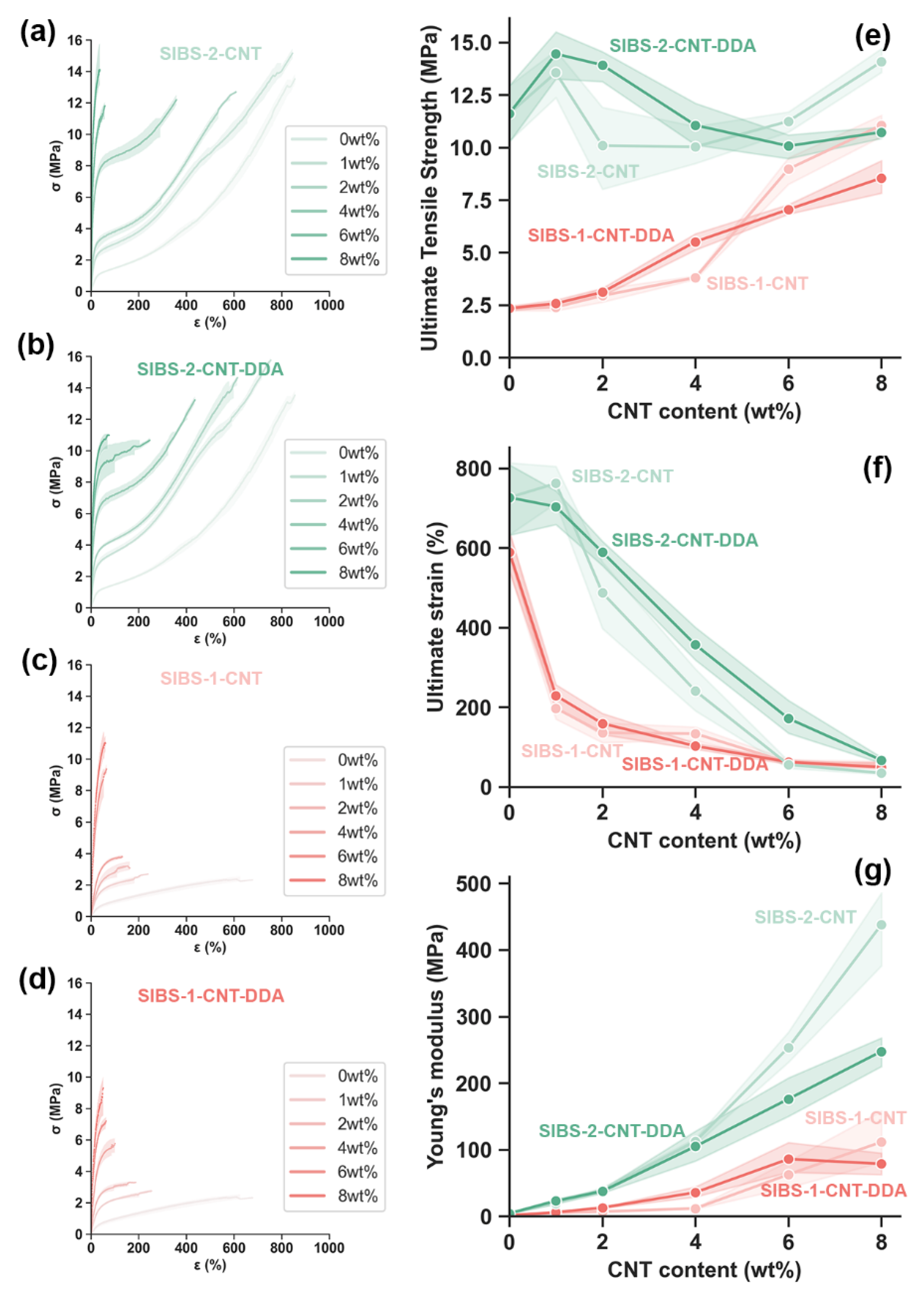
3.6. Mechanical Tests
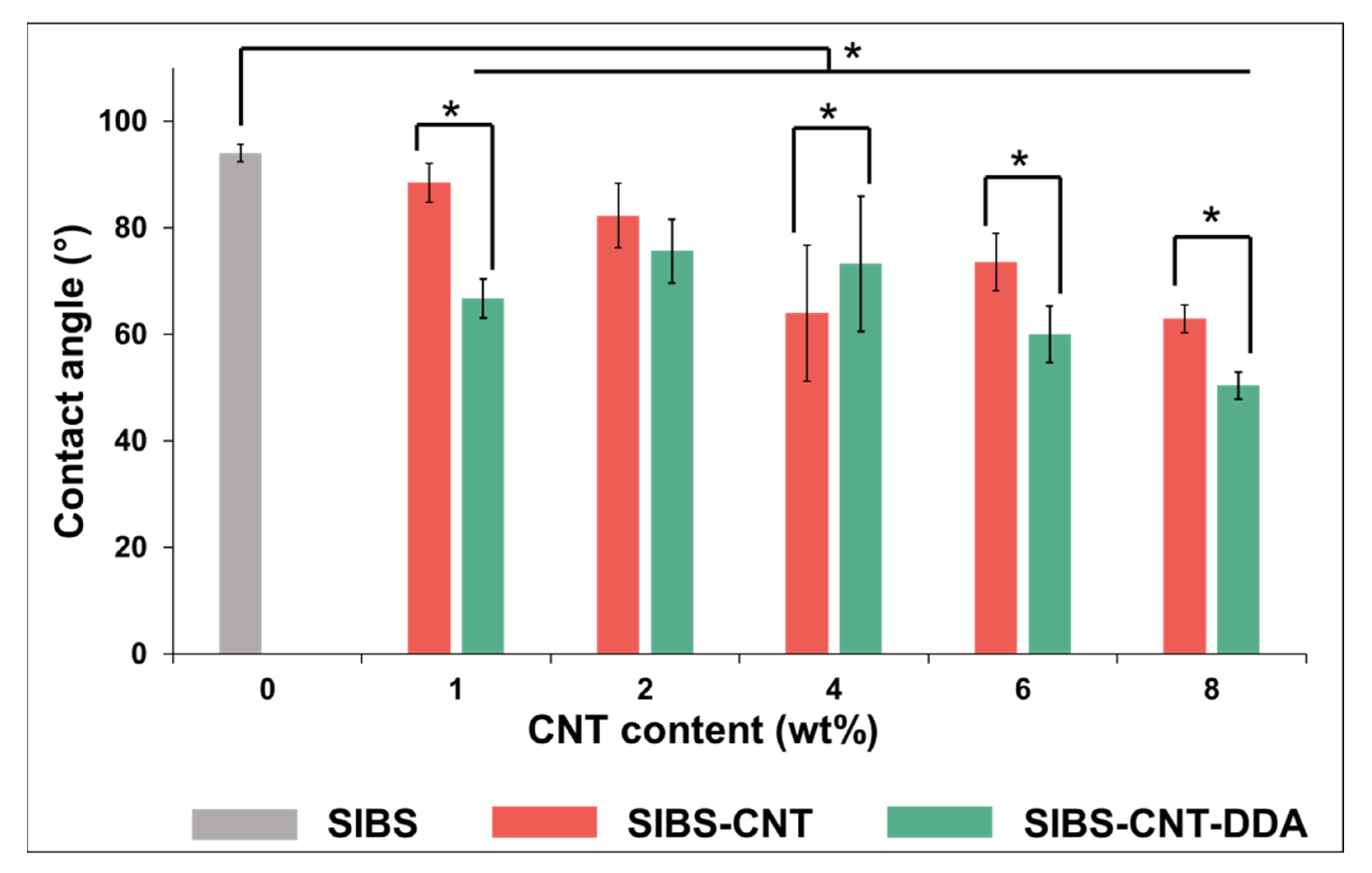
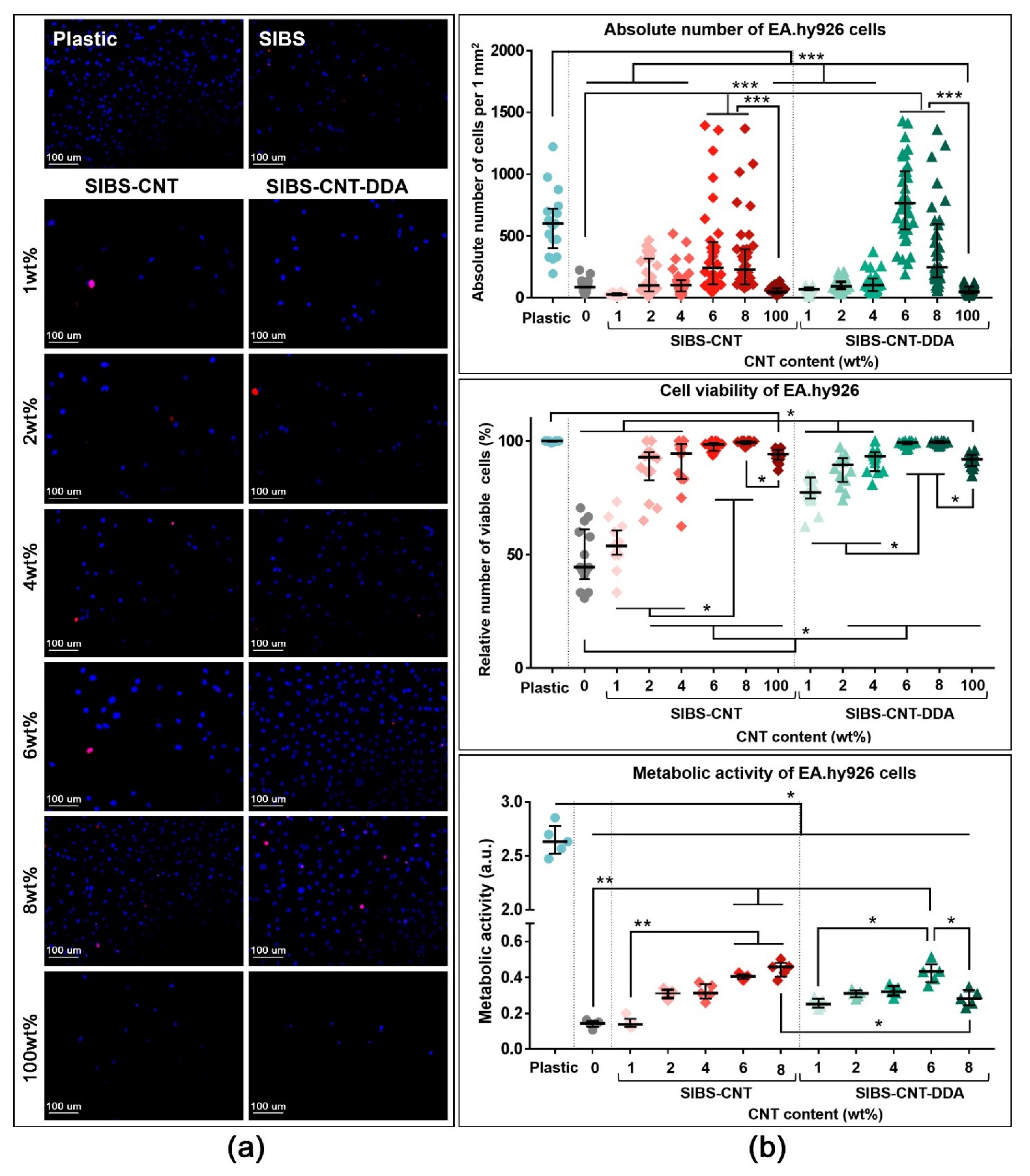
3.7. Evaluation of Cytotoxicity and Wettability
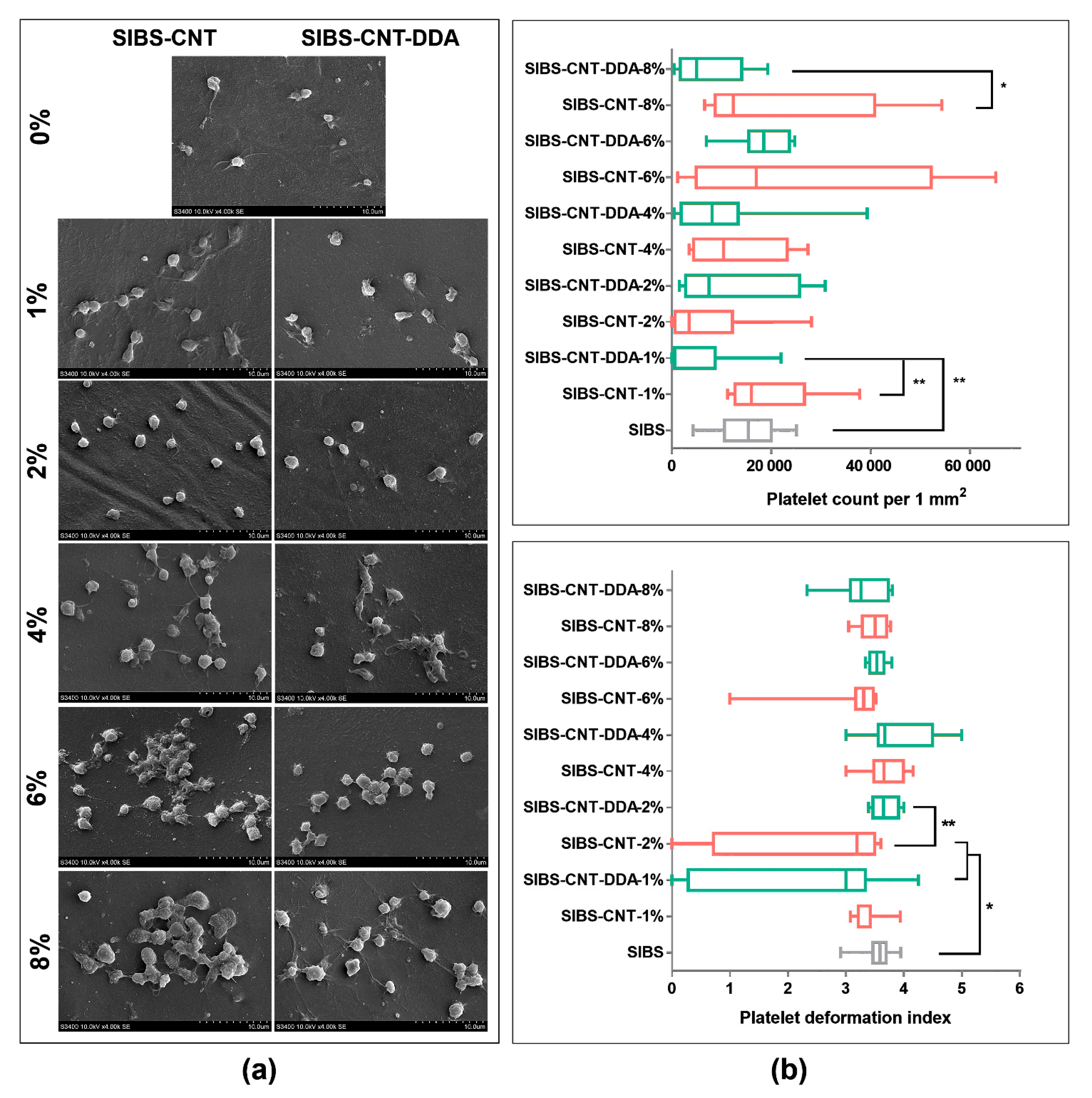
3.8. Platelet Adhesion
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Cui, Z.; Yang, B.; Li, R.-K. Application of biomaterials in cardiac repair and regeneration. Engineering 2016, 2, 141–148. [Google Scholar] [CrossRef] [Green Version]
- Wang, L.; Wu, S.; Cao, G.; Fan, Y.; Dunne, N.; Li, X. Biomechanical studies on biomaterial degradation and co-cultured cells: Mechanisms, potential applications, challenges and prospects. J. Mater. Chem. B 2019, 7, 7439–7459. [Google Scholar] [CrossRef] [PubMed]
- Maurer, E.; Barcikowski, S.; Gökce, B. Process chain for the fabrication of nanoparticle polymer composites by laser ablation synthesis. Chem. Eng. Technol. 2017, 40, 1535–1543. [Google Scholar] [CrossRef]
- Ates, M.; Eker, A.A.; Eker, B. Carbon nanotube-based nanocomposites and their applications. Adhes. Sci. Technol. 2017, 31, 1977–1997. [Google Scholar] [CrossRef]
- Moses, J.C.; Gangrade, A.; Mandal, B.B. Carbon nanotubes and their polymer nanocomposites. In Nanomaterials and Polymer Nanocomposites, 1st ed.; Karak, N., Ed.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 145–175. [Google Scholar] [CrossRef]
- Eatemadi, A.; Daraee, H.; Karimkhanloo, H.; Kouhi, M.; Zarghami, N.; Akbarzadeh, A.; Abasi, M.; Hanifehpour, Y.; Joo, S.W. Carbon nanotubes: Properties, synthesis, purification, and medical applications. Nanoscale Res. Lett. 2014, 9, 393. [Google Scholar] [CrossRef] [Green Version]
- Sheheri, S.Z.; Amshany, Z.M.; Sulami, Q.A.; Tashkandi, N.Y.; Hussein, M.A.; Shishtawy, R.M. The preparation of carbon nanofillers and their role on the performance of variable polymer nanocomposites. Des. Monomers Polym. 2019, 22, 8–53. [Google Scholar] [CrossRef] [Green Version]
- Kalakonda, P.; Banne, S.; Kalakonda, P. Enhanced mechanical properties of multiwalled carbon nanotubes/thermoplastic polyurethane nanocomposites. Nanomater. Nanotechnol. 2019, 9, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Donchak, V.; Stetsyshyn, Y.; Bratychak, M.; Broza, G.; Harhay, K.; Stepina, N.; Kostenko, M.; Voronov, S. Nanoarchitectonics at surfaces using multifunctional initiators of surface-initiated radical polymerization for fabrication of the nanocomposites. Appl. Surf. Sci. Adv. 2021, 5, 100104. [Google Scholar] [CrossRef]
- Oveissi, F.; Naficy, S.; Lee, A.; Winlaw, D.S.; Dehghani, F. Materials and manufacturing perspectives in engineering heart valves: A review. Mater. Today Bio 2019, 5, 100038. [Google Scholar] [CrossRef]
- Raja, I.S.; Song, S.-J.; Kang, M.S.; Lee, Y.B.; Kim, B.; Hong, S.W.; Jeong, S.J.; Lee, J.-C.; Han, D.-W. Toxicity of zero- and one-dimensional carbon nanomaterials. Nanomaterials 2019, 9, 1214. [Google Scholar] [CrossRef] [Green Version]
- Arash, B.; Wang, Q.; Varadan, V. Mechanical properties of carbon nanotube/polymer composites. Sci. Rep. 2014, 4, 6479. [Google Scholar] [CrossRef] [PubMed]
- Hussain, F.; Hojjati, M.; Okamoto, M.; Gorga, R.E. Review article: Polymer-matrix nanocomposites, processing, manufacturing, and application: An overview. J. Compos. Mater. 2006, 40, 1511–1575. [Google Scholar] [CrossRef]
- Huang, Y.Y.; Terentjev, E.M. Dispersion of carbon nanotubes: Mixing, sonication, stabilization, and composite properties. Polymers 2012, 4, 275–295. [Google Scholar] [CrossRef] [Green Version]
- Mallakpour, S.; Soltanian, S. Surface functionalization of carbon nanotubes: Fabrication and applications. RSC Adv. 2016, 6, 109916–109935. [Google Scholar] [CrossRef]
- Arrigo, R.; Teresi, R.; Gambarotti, C.; Parisi, F.; Lazzara, G.; Dintcheva, N.T. Sonication-induced modification of carbon nanotubes: Effect on the rheological and thermo-oxidative behaviour of polymer-based nanocomposites. Materials 2018, 11, 383. [Google Scholar] [CrossRef] [Green Version]
- Zhao, Y.-L.; Stoddart, J.F. Noncovalent functionalization of single-walled carbon nanotubes. Acc. Chem. Res. 2009, 42, 1161–1171. [Google Scholar] [CrossRef]
- Inácio, Â.S.; Mesquita, K.A.; Baptista, M.; Ramalho-Santos, J.; Vaz, W.L.C.; Vieira, O.V. In vitro surfactant structure-toxicity relationships: Implications for surfactant Use in Sexually Transmitted Infection Prophylaxis and Contraception. PLoS ONE 2011, 6, e19850. [Google Scholar] [CrossRef] [Green Version]
- Zhang, J.; Zou, H.; Qing, Q.; Yang, Y.; Li, Q.; Liu, Z.; Guo, X.; Du, Z. Effect of chemical oxidation on the structure of single-walled carbon nanotubes. J. Phys. Chem. B 2003, 107, 3712–3718. [Google Scholar] [CrossRef]
- Avilés, F.; Rodríguez, J.C.; Estay, P.T.; Zobeiri, M.; Bolados, H.A. Improving carbon nanotube/polymer interactions in nanocomposites. In Carbon Nanotube-Reinforced Polymers, 1st ed.; Rafiee, R., Ed.; Elsevier: Amsterdam, The Netherlands, 2018; pp. 145–175. [Google Scholar] [CrossRef]
- Ham, H.T.; Koo, C.M.; Kim, S.O.; Choi, Y.S.; Chung, I.J. Chemical modification of carbon nanotubes and preparation of polystyrene/carbon nanotubes composites. Macromol. Res. 2004, 12, 384–390. [Google Scholar] [CrossRef]
- Ferreira, F.V.; Francisco, W.; De Menezes, B.R.C.; Cividanes, L.D.S.; Coutinho, A.D.R.; Thim, G.P. Carbon nanotube functionalized with dodecylamine for the effective dispersion in solvents. Appl. Surf. Sci. 2015, 357B, 2154–2159. [Google Scholar] [CrossRef]
- De Menezes, B.R.C.; Ferreira, F.V.; Silva, B.C.; Simonetti, E.A.N.; Bastos, T.M.; Cividanes, L.S.; Thim, G.P. Effects of octadecylamine functionalization of carbon nanotubes on dispersion, polarity, and mechanical properties of CNT/HDPE nanocomposites. J. Mater. Sci. 2018, 53, 14311–14327. [Google Scholar] [CrossRef]
- Bernard, M.; Jubeli, E.; Pungente, M.D.; Yagoubi, N. Biocompatibility of polymer-based biomaterials and medical devices—Regulations, in vitro screening and risk-management. Biomater. Sci. 2018, 24, 2025–2053. [Google Scholar] [CrossRef] [PubMed]
- Nezami, F.R.; Athanasiou, L.S.; Edelman, E.R. Endovascular drug-delivery and drug-elution systems. In Biomechanics of Living Organs, 1st ed.; Ohayon, J., Finet, G., Pettigrew, R.I., Eds.; Elsevier: Amsterdam, The Netherlands, 2021; Volume 4, pp. 595–631. [Google Scholar] [CrossRef]
- Ovcharenko, E.; Rezvova, M.; Nikishau, P.; Kostjuk, S.; Glushkova, T.; Antonova, L.; Trebushat, D.; Akentieva, T.; Shishkova, D.; Krivikina, E.; et al. Polyisobutylene-based thermoplastic elastomers for manufacturing polymeric heart valve leaflets: In vitro and in vivo results. Appl. Sci. 2019, 9, 4773. [Google Scholar] [CrossRef] [Green Version]
- Pinchuk, L.; Wilson, G.J.; Barry, J.J.; Schoephoerster, R.T.; Parele, J.-M.; Kennedy, J.P. Medical applications of poly(styrene-block-isobutylene-block-styrene) (“SIBS”). Biomaterials 2008, 29, 448–460. [Google Scholar] [CrossRef] [PubMed]
- Fittipaldi, M.; Garcia, C.; Rodriguez, L.A.; Grace, L.R. Optimization of injection molding parameters for poly(styrene-isobutylene-styrene) block copolymer. AIP Conf. Proc. 2016, 1713, 040004. [Google Scholar] [CrossRef] [Green Version]
- Rezvova, M.A.; Yuzhalin, A.E.; Glushkova, T.V.; Makarevich, M.I.; Nikishau, P.A.; Kostjuk, S.V.; Klyshnikov, K.Y.; Matveeva, V.G.; Khanova, M.Y.; Ovcharenko, E.A. Biocompatible nanocomposites based on poly(styrene-block-isobutylene-block-styrene) and carbon nanotubes for biomedical application. Polymers 2020, 12, 2158. [Google Scholar] [CrossRef]
- Makarevich, M.I.; Nikishau, P.A.; Berezianko, I.A.; Glushkova, T.V.; Rezvova, M.A.; Ovcharenko, E.A.; Bekmukhamedov, G.E.; Yakhvarov, D.G.; Kostjuk, S.V. Aspects of the synthesis of poly(styrene-block-isobutylene-block-styrene) by TiCl4-co-initiated cationic polymerization in open conditions. Macromol 2021, 1, 243–255. [Google Scholar] [CrossRef]
- Afshari, R.; Mazinani, S.; Abdouss, M. Nanohybrid nanoparticles based on chitosan/functionalized carbon nanotubes as anti-HIV Nanocarrier. Nano. 2015, 10, 1550010. [Google Scholar] [CrossRef]
- Rahimpour, A.; Jahanshahi, M.; Khalili, S.; Mollahosseini, A.; Zirepour, A.; Rajaeian, B. Novel functionalized carbon nanotubes for improving the surface properties and performance of polyethersulfone (PES) membrane. Desalination 2012, 286, 99–107. [Google Scholar] [CrossRef]
- Vinayan, B.P.; Nagar, R.; Raman, V.; Rajalakshmi, N.; Dhathathreyan, K.S.; Ramaprabhu, S. Synthesis of graphene-multiwalled carbon nanotubes hybrid nanostructure by strengthened electrostatic interaction and its lithium ion battery application. J. Mater. Chem. 2012, 22, 9949. [Google Scholar] [CrossRef]
- Zhao, Z.; Yanga, Z.; Hu, Y. Multiple functionalization of multi-walled carbon nanotubes with carboxyl and amino groups. Appl. Surf. Sci. 2013, 276, 476. [Google Scholar] [CrossRef]
- Hedin, U. Long-term results of PTFE grafts. J. Vasc. Access 2015, 16, S87–S92. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Y.; Gao, X.; Wei, Y.; Wang, X.; Wang, J.; Zhang, Y.; Gao, C. Enhanced desalination performance of carboxyl functionalized graphene oxide nanofiltration membranes. Desalination 2017, 405, 29–39. [Google Scholar] [CrossRef]
- Vukovic, G.; Marinkovic, A.; Obradovic, M.; Radmilović, V.; Čolić, M.; Aleksić, R.; Uskoković, P.S. Synthesis, characterization and cytotoxicity of surface amino-functionalized water-dispersible multi-walled carbon nanotubes. Appl. Surf. Sci. 2009, 55, 8067. [Google Scholar] [CrossRef]
- Forfang, K.; Zimmermann, B.; Kosa, G.; Kohler, A.; Shapaval, V. FTIR spectroscopy for evaluation and monitoring of lipid extraction efficiency for oleaginous fungi. PLoS ONE 2017, 12, e0170611. [Google Scholar] [CrossRef] [Green Version]
- Md Salim, R.; Asik, J.; Sarjadi, M.S. Chemical functional groups of extractives, cellulose and lignin extracted from native Leucaena leucocephala bark. Wood Sci. Technol. 2021, 55, 295–313. [Google Scholar] [CrossRef]
- Rodriguez, R.D.; Toader, M.; Hermann, S.; Sheremet, E.; Müller, S.; Gordan, O.D.; Yu, H.; Schulz, E.S.; Hietschold, M.; Zahn, D.R.T. Nanoscale optical and electrical characterization of horizontally aligned single-walled carbon nanotubes. Nanoscale Res. Lett. 2012, 7, 682. [Google Scholar] [CrossRef] [Green Version]
- Schöttner, S.; Hossain, R.; Rüttiger, C.; Gallei, M. Ferrocene-modified block copolymers for the preparation of smart porous membranes. Polymers 2017, 9, 491. [Google Scholar] [CrossRef] [Green Version]
- Ranade, S.V.; Miller, K.M.; Richard, R.E.; Chan, A.K.; Allen, M.J.; Helmus, M.N. Physical characterization of controlled release of paclitaxel from the TAXUS Express2 drug-eluting stent. J. Biomed. Mater. Res. 2004, 71A, 625–634. [Google Scholar] [CrossRef]
- Gyor, M.; Fodor, Z.; Wang, H.C.; Faust, R. Polyisobutylene-based thermoplastic elastomers. I. Synthesis and characterization of polystyrene-polyisobutylene-polystyrene triblock copolymers. J. Macromol. Sci. Part A Pure Appl. Chem. 1994, A31, 2055–2056. [Google Scholar] [CrossRef]
- Raphey, V.R.; Henna, T.K.; Nivitha, K.P.; Mufeedha, P.; Sabu, C.; Pramod, K. Advanced biomedical applications of carbon nanotube. Mater. Sci. Eng. C 2019, 100, 616–630. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Liu, B.; Gao, X.; Xu, D. A review of the interfacial characteristics of polymer nanocomposites containing carbon nanotubes. RSC Adv. 2018, 8, 28048–28085. [Google Scholar] [CrossRef] [Green Version]
- Zhou, Y.; Fang, Y.; Ramasamy, R.P. Non-covalent functionalization of carbon nanotubes for electrochemical biosensor development. Sensors 2019, 19, 392. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Richard, C. Supramolecular self-assembly of lipid derivatives on carbon nanotubes. Science 2003, 300, 775–778. [Google Scholar] [CrossRef]
- Li, H.; Qiu, Y. Dispersion, sedimentation and aggregation of multi-walled carbon nanotubes as affected by single and binary mixed surfactants. R. Soc. Open Sci. 2019, 6, 190241. [Google Scholar] [CrossRef] [Green Version]
- Tkalya, E.E.; Ghislandi, M.; de With, G.; Koning, C.E. The use of surfactants for dispersing carbon nanotubes and graphene to make conductive nanocomposites. Curr. Opin. Colloid Interface Sci. 2012, 17, 225–232. [Google Scholar] [CrossRef]
- Ferreira, F.V.; Franceschi, W.; Menezes, B.R.C.; Brito, F.S.; Lozano, K.; Coutinho, A.R.; Thim, G.P. Dodecylamine functionalization of carbon nanotubes to improve dispersion, thermal and mechanical properties of polyethylene based nanocomposites. Appl. Surf. Sci. 2017, 410, 267–277. [Google Scholar] [CrossRef]
- Branca, C.; Frusteri, F.; Magazù, V.; Mangione, A. Characterization of carbon nanotubes by TEM and infrared spectroscopy. J. Phys. Chem. B 2004, 108, 3469–3473. [Google Scholar] [CrossRef]
- Tavares, M.I.B.; Silva, E.O.; Silva, P.R.C.; Menezes, L.R. Polymer nanocomposites. In Nanostructured Materials—Fabrication to Applications, 1st ed.; Seehra, M., Ed.; IntechOpen: London, UK, 2017. [Google Scholar] [CrossRef] [Green Version]
- Liu, C.X.; Choi, J.W. Improved dispersion of carbon nanotubes in polymers at high concentrations. Nanomaterials 2012, 2, 329–347. [Google Scholar] [CrossRef] [Green Version]
- Sadek, E.M.; El-Nashar, D.E.; Ward, A.A.; Ahmed, S.M. Study on the properties of multi-walled carbon nanotubes reinforced poly (vinyl alcohol) composites. J. Polym. Res. 2018, 25, 249. [Google Scholar] [CrossRef]
- Ramasubramaniam, R.; Chen, J.; Liu, H. Homogeneous carbon nanotube/polymer composites for electrical applications. Appl. Phys. Lett. 2003, 83, 2928–2930. [Google Scholar] [CrossRef]
- Rashidi, A.; Olfatbakhsh, T.; Crawford, B.; Milani, A.S. A review of current challenges and case study toward optimizing micro-computed X-ray tomography of carbon fabric composites. Materials 2020, 13, 3606. [Google Scholar] [CrossRef]
- Mehdikhani, M.; Gorbatikh, L.; Verpoest, I.; Lomov, S.V. Voids in fiber-reinforced polymer composites: A review on their formation, characteristics, and effects on mechanical performance. J. Compos. Mater. 2019, 53, 1579–1669. [Google Scholar] [CrossRef]
- Kim, J.Y.; Choi, H.J.; Kang, C.S.; Kim, S.H. Influence of modified carbon nanotube on physical properties and crystallization behavior of poly(ethylene terephthalate) nanocomposite. Polym. Compos. 2010, 31, 858–869. [Google Scholar] [CrossRef]
- Zhou, T.Y.; Tsui, G.C.P.; Liang, J.Z.; Zou, S.Y.; Tang, C.Y.; Mišković-Stanković, V. Thermal properties and thermal stability of PP/MWCNT composites. Compos. Part B Eng. 2016, 90, 107–114. [Google Scholar] [CrossRef]
- Corcione, C.E.; Frigione, M. Characterization of nanocomposites by thermal analysis. Materials 2012, 5, 2960–2980. [Google Scholar] [CrossRef] [Green Version]
- Huxtable, S.T.; Cahill, D.G.; Shenogin, S.; Xue, L.; Ozisik, R.; Barone, P.; Usrey, M.; Strano, M.S.; Siddons, G.; Shim, M.; et al. Interfacial heat flow in carbon nanotube suspensions. Nature Mater. 2003, 2, 731–734. [Google Scholar] [CrossRef]
- Zadeh, Z.E.; Solouk, A.; Shafieian, M.; Nazarpak, M.H. Electrospun polyurethane/carbon nanotube composites with different amounts of carbon nanotubes and almost the same fiber diameter for biomedical applications. Mater. Sci. Eng. C 2020, 118, 111403. [Google Scholar] [CrossRef]
- Salah, N.; Alfawzan, A.M.; Saeed, A.; Alshahrie, A.; Allafi, W. Effective reinforcements for thermoplastics based on carbon nanotubes of oil fly ash. Sci. Rep. 2019, 9, 20288. [Google Scholar] [CrossRef] [Green Version]
- Yang, X.; Sun, L.; Zhang, C.; Huang, B.; Chu, Y.; Zhan, B. Modulating the sensing behaviors of poly(styrene-ethylene-butylene-styrene)/carbon nanotubes with low-dimensional fillers for large deformation sensors. Compos. Part B. Eng. 2019, 160, 605–614. [Google Scholar] [CrossRef]
- Svensson, S.; Åkesson, D.; Bohlén, M. Reprocessing of high-density polyethylene reinforced with carbon nanotubes. J. Polym. Environ. 2020, 28, 1967–1973. [Google Scholar] [CrossRef]
- Paul, J.; Sindhu, S.; Nurmawati, M.H.; Valiyaveettil, S. Mechanics of prestressed polydimethylsiloxane-carbon nanotube composite. Appl. Phys. Lett. 2006, 89, 184101–184103. [Google Scholar] [CrossRef]
- Babal, A.S.; Gupta, R.; Singh, B.P.; Singh, V.N.; Dhakate, S.R.; Mathur, R.B. Mechanical and electrical properties of high performance MWCNT/polycarbonate composites prepared by an industrial viable twin screw extruder with back flow channel. RSC Adv. 2014, 4, 64649–64658. [Google Scholar] [CrossRef] [Green Version]
- Tian, F.; Cui, D.; Schwarz, H.; Estrada, G.G.; Kobayashi, H. Cytotoxicity of single-wall carbon nanotubes on human fibroblasts. Toxicol. In Vitro 2006, 20, 1202–1212. [Google Scholar] [CrossRef] [PubMed]
- Mohammadi, E.; Zeinali, M.; Mohammadi-Sardoo, M.; Iranpour, M.; Behnam, B.; Mandegary, A. The effects of functionalization of carbon nanotubes on toxicological parameters in mice. Hum. Exp. Toxicol. 2020, 096032711989998. [Google Scholar] [CrossRef] [PubMed]
- Gilmore, K.J.; Moulton, S.E.; Wallace, G.G. Incorporation of carbon nanotubes into the biomedical polymer poly(styrene-isobutylene-styrene). Carbon 2007, 45, 402–410. [Google Scholar] [CrossRef]
- Gaharwar, A.K.; Patel, A.; Dolatshahi-Pirouz, A.; Zhang, H.; Rangarajan, K.; Iviglia, G.; Shin, S.-R.; Hussain, M.A.; Khademhosseini, A. Elastomeric nanocomposite scaffolds made from poly(glycerol sebacate) chemically crosslinked with carbon nanotubes. Biomater. Sci. 2015, 3, 46–58. [Google Scholar] [CrossRef] [Green Version]
- Hebbar, R.S.; Isloor, A.M.; Ismail, A.F. Contact angle measurements. In Membrane Characterization, 1st ed.; Hilal, N., Ismail, A., Matsuura, T., Oatley-Radcliffe, D., Eds.; Elsevier: Amsterdam, The Netherlands, 2017; pp. 219–255. [Google Scholar] [CrossRef]
- Li, Y.; Pham, J.Q.; Johnston, K.P.; Green, P.F. Contact angle of water on polystyrene thin films: Effects of CO2 environment and film thickness. Langmuir 2007, 23, 9785–9793. [Google Scholar] [CrossRef]
- Du, Y.; Li, C.; Jin, J.; Li, C.; Jiang, W. Surface modification of polyisobutylene via grafting amino acid-based poly (acryloyl-6-aminocaproic acid) as multifunctional material. Colloids Surf. B Biointerfaces 2018, 161, 73–82. [Google Scholar] [CrossRef]
- Owens, A.P.; Mackman, N. Tissue factor and thrombosis: The clot starts here. Thromb. Haemost. 2010, 104, 432–439. [Google Scholar] [CrossRef]
- De Paoli Lacerda, S.H.; Semberova, J.; Holada, K.; Simakova, O.; Hudson, S.D.; Simak, J. Carbon nanotubes activate store-operated calcium entry in human blood platelets. ACS Nano 2011, 5, 5808–5813. [Google Scholar] [CrossRef] [PubMed]
- Koh, L.B.; Rodriguez, I.; Zhou, J. Platelet adhesion studies on nanostructured poly(lactic-co-glycolic-acid)–carbon nanotube composite. J. Biomed. Mater. Res. A 2008, 86, 394–401. [Google Scholar] [CrossRef] [PubMed]
- Meng, J.; Kong, H.; Xu, H.Y.; Song, L.; Wang, C.Y.; Xie, S.S. Improving the blood compatibility of polyurethane using carbon nanotubes as fillers and its implications to cardiovascular surgery. J. Biomed. Mater. Res. A 2005, 74, 208–214. [Google Scholar] [CrossRef] [PubMed]

| Parameter | SIBS-1 | SIBS-2 |
|---|---|---|
| Mn of the middle PIB block a | 33 200 | 50 700 |
| Mn(SIBS) a | 39 900 | 70 900 |
| Mw/Mn a | 1.47 | 1.57 |
| w(St), % (NMR) b | 27 | 28 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rezvova, M.A.; Nikishau, P.A.; Makarevich, M.I.; Glushkova, T.V.; Klyshnikov, K.Y.; Akentieva, T.N.; Efimova, O.S.; Nikitin, A.P.; Malysheva, V.Y.; Matveeva, V.G.; et al. Biomaterials Based on Carbon Nanotube Nanocomposites of Poly(styrene-b-isobutylene-b-styrene): The Effect of Nanotube Content on the Mechanical Properties, Biocompatibility and Hemocompatibility. Nanomaterials 2022, 12, 733. https://doi.org/10.3390/nano12050733
Rezvova MA, Nikishau PA, Makarevich MI, Glushkova TV, Klyshnikov KY, Akentieva TN, Efimova OS, Nikitin AP, Malysheva VY, Matveeva VG, et al. Biomaterials Based on Carbon Nanotube Nanocomposites of Poly(styrene-b-isobutylene-b-styrene): The Effect of Nanotube Content on the Mechanical Properties, Biocompatibility and Hemocompatibility. Nanomaterials. 2022; 12(5):733. https://doi.org/10.3390/nano12050733
Chicago/Turabian StyleRezvova, Maria A., Pavel A. Nikishau, Miraslau I. Makarevich, Tatiana V. Glushkova, Kirill Yu. Klyshnikov, Tatiana N. Akentieva, Olga S. Efimova, Andrey P. Nikitin, Valentina Yu. Malysheva, Vera G. Matveeva, and et al. 2022. "Biomaterials Based on Carbon Nanotube Nanocomposites of Poly(styrene-b-isobutylene-b-styrene): The Effect of Nanotube Content on the Mechanical Properties, Biocompatibility and Hemocompatibility" Nanomaterials 12, no. 5: 733. https://doi.org/10.3390/nano12050733
APA StyleRezvova, M. A., Nikishau, P. A., Makarevich, M. I., Glushkova, T. V., Klyshnikov, K. Y., Akentieva, T. N., Efimova, O. S., Nikitin, A. P., Malysheva, V. Y., Matveeva, V. G., Senokosova, E. A., Khanova, M. Y., Danilov, V. V., Russakov, D. M., Ismagilov, Z. R., Kostjuk, S. V., & Ovcharenko, E. A. (2022). Biomaterials Based on Carbon Nanotube Nanocomposites of Poly(styrene-b-isobutylene-b-styrene): The Effect of Nanotube Content on the Mechanical Properties, Biocompatibility and Hemocompatibility. Nanomaterials, 12(5), 733. https://doi.org/10.3390/nano12050733









