Antibacterial and Fluorescence Staining Properties of an Innovative GTR Membrane Containing 45S5BGs and AIE Molecules In Vitro
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials and Instruments
2.2. Synthesis of PLGA/BG and PLGA/BG/TPE-NIM+ Membranes
2.3. Characterization of PLGA/BG and PLGA/BG/TPE-NIM+ Membranes
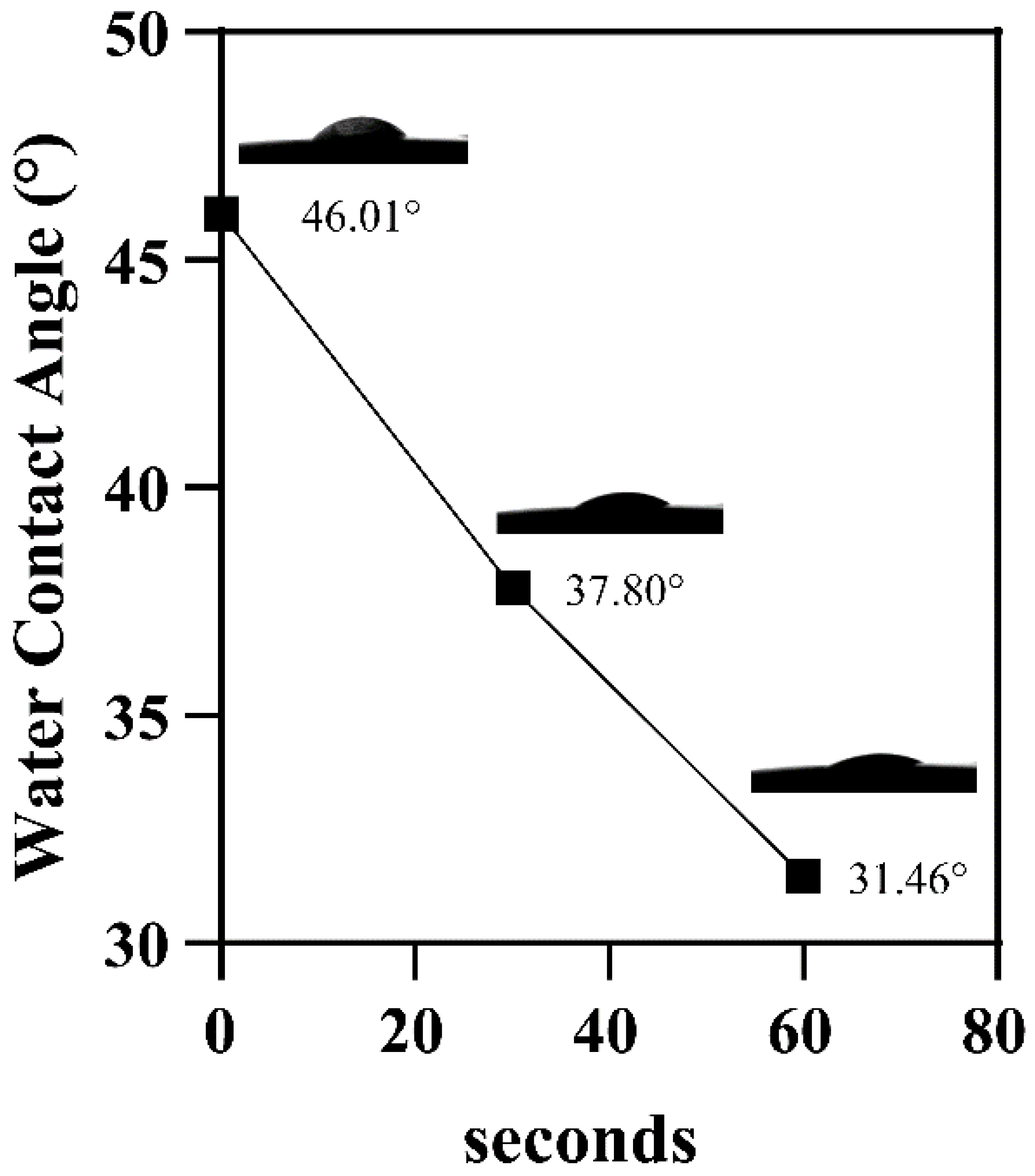
2.3.1. Water Contact Angle Measurement of the PLGA/BG/TPE-NIM+ Membrane
2.3.2. Scanning Electron Microscopy/Energy-Dispersive Spectrometry
2.4. Cell Culture
2.5. Cytotoxicity Test
2.6. Bacterial Culture
2.7. Antibacterial Activity Test
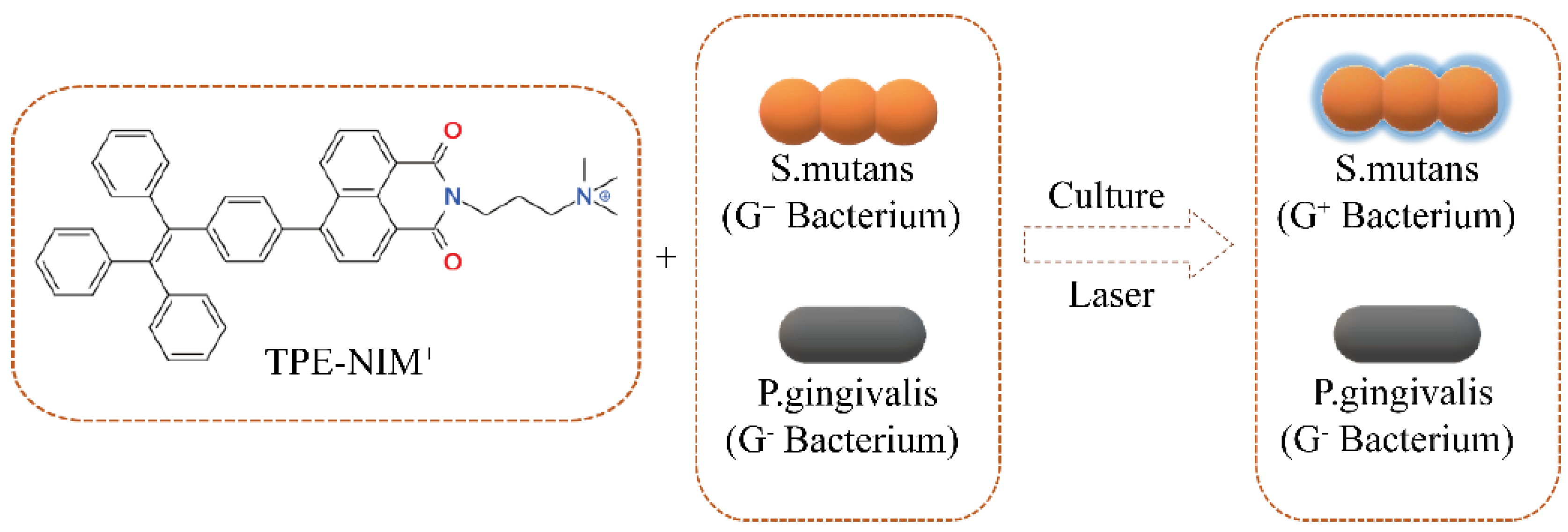
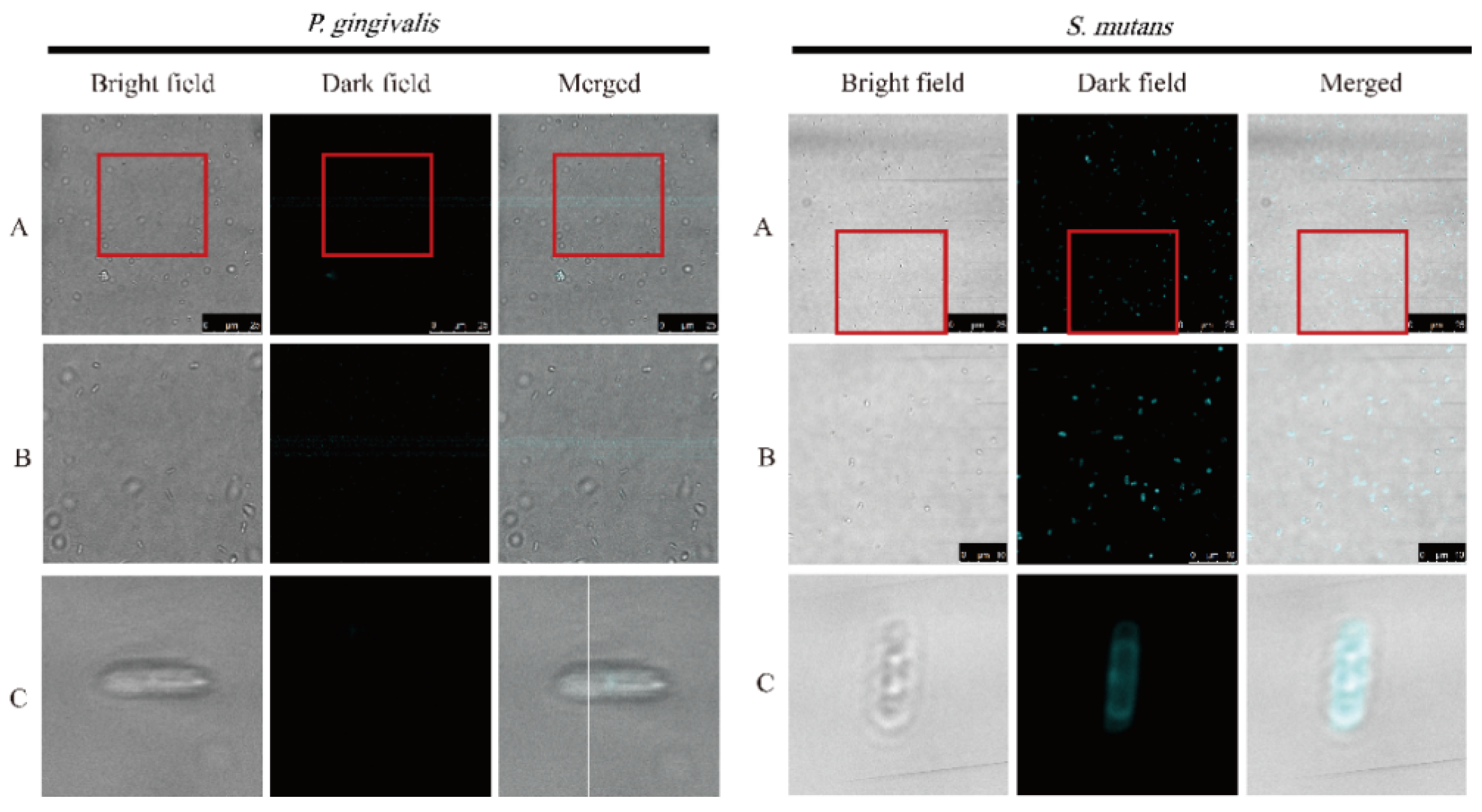
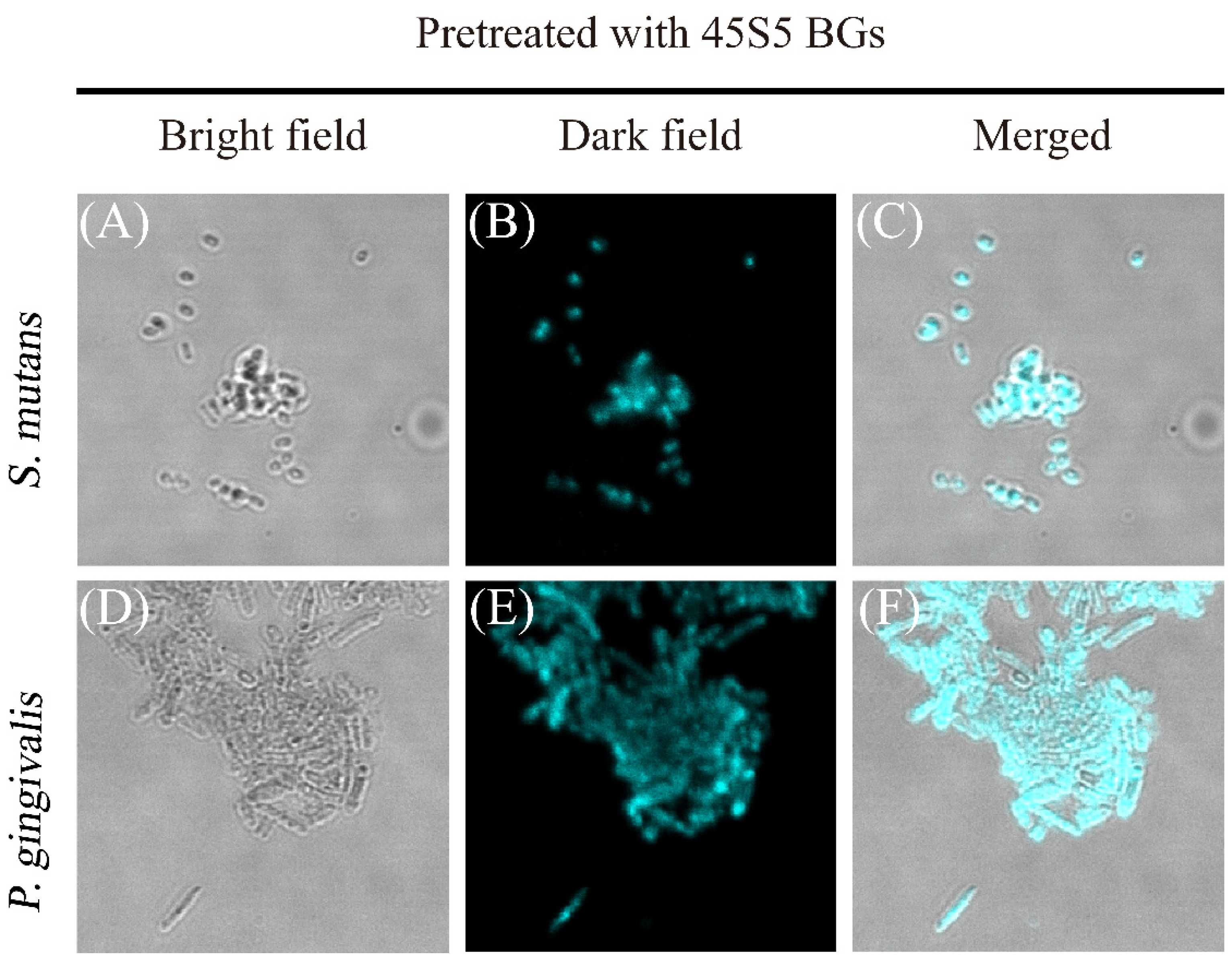
2.8. Staining Abilities of TPE-NIM+ toward Pathogens
2.9. Statistical Analysis
3. Results
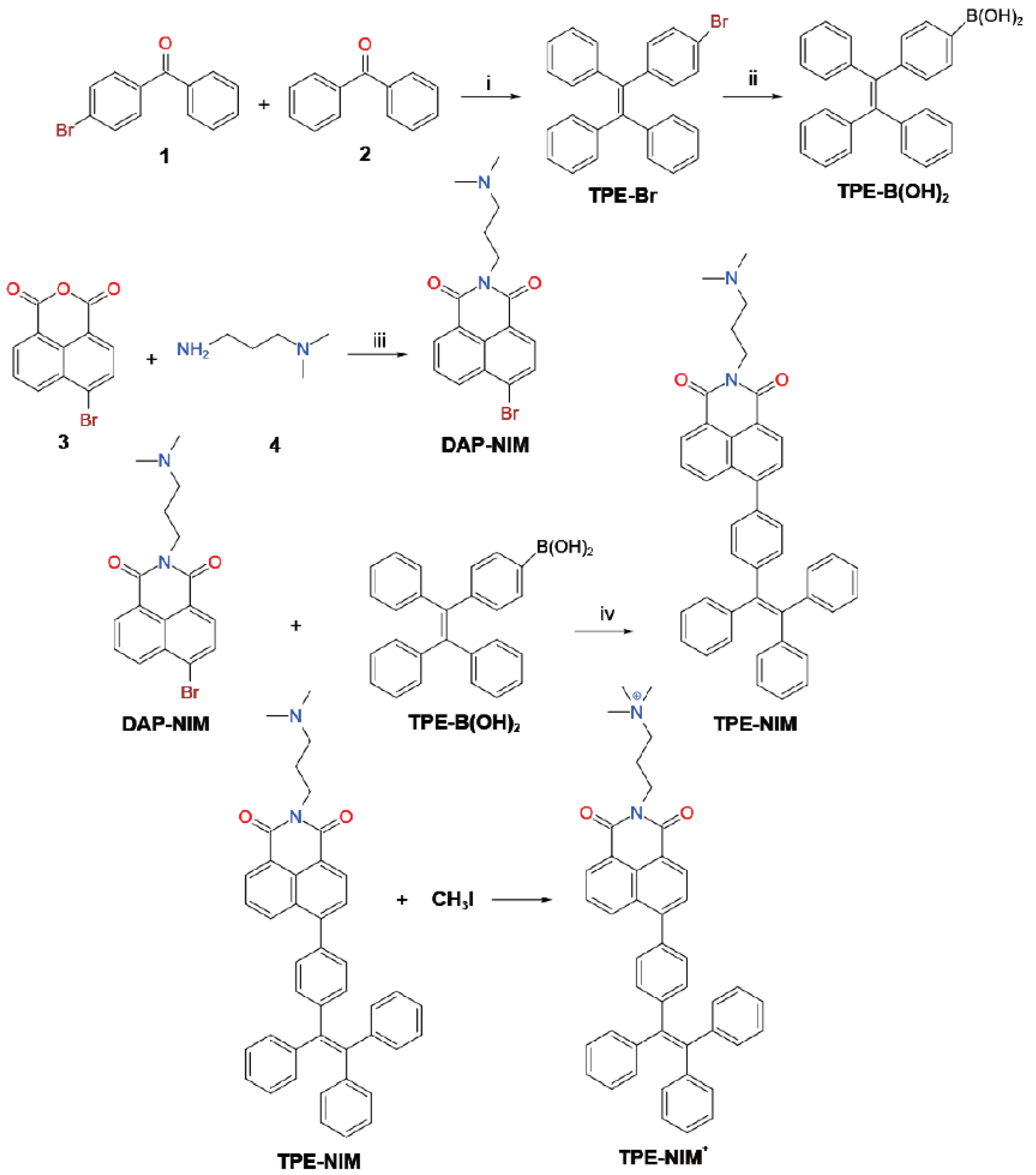
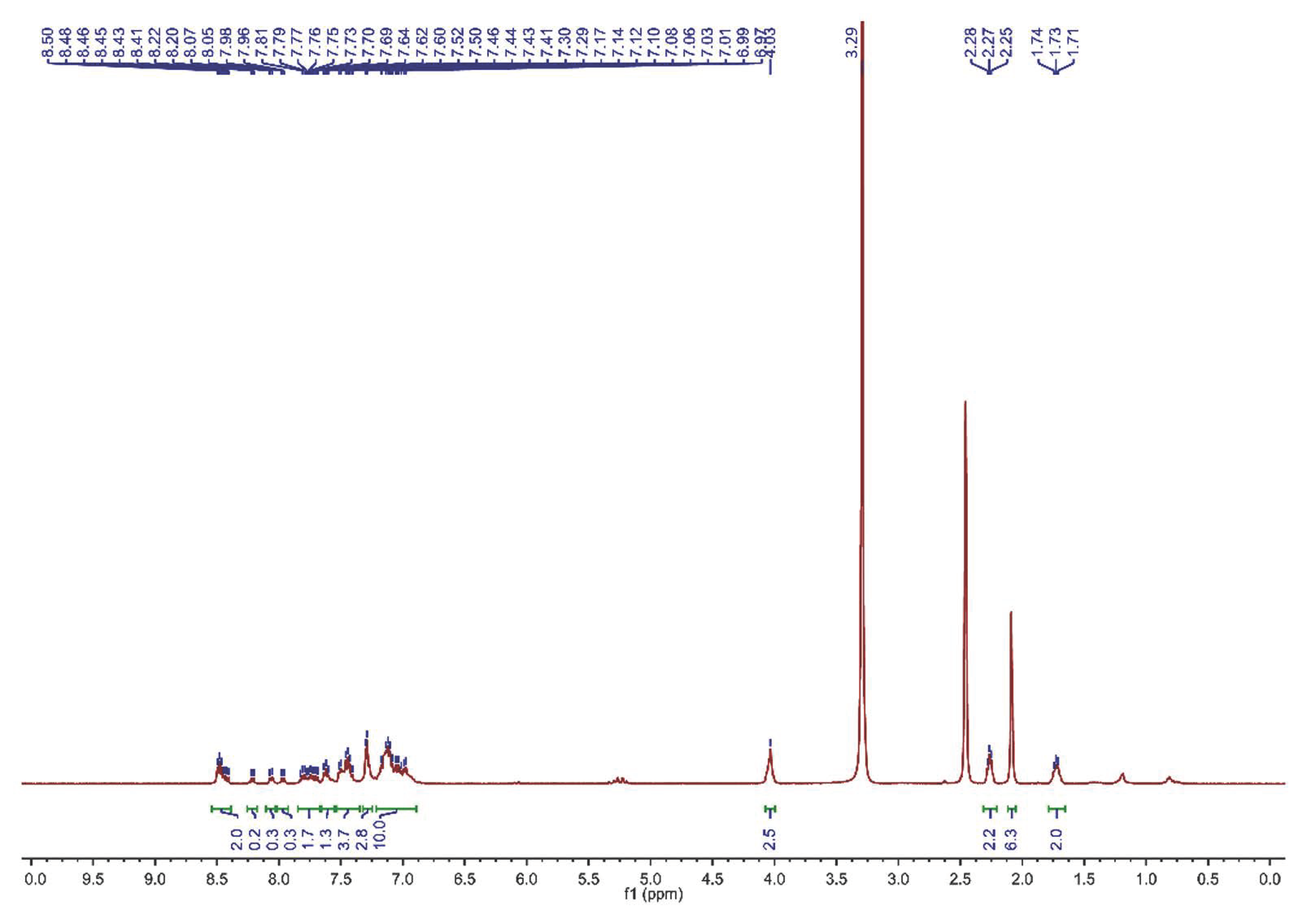
3.1. Synthesis and Characterization of TPE-NIM+
3.2. Preparation of PLGA/BG and PLGA/BG/TPE-NIM+ Membranes
3.3. Characterization of PLGA/BG and PLGA/BG/TPE-NIM+ Membranes
3.3.1. Water Contact Angle Measurement of the PLGA/BG/TPE-NIM+ Membrane
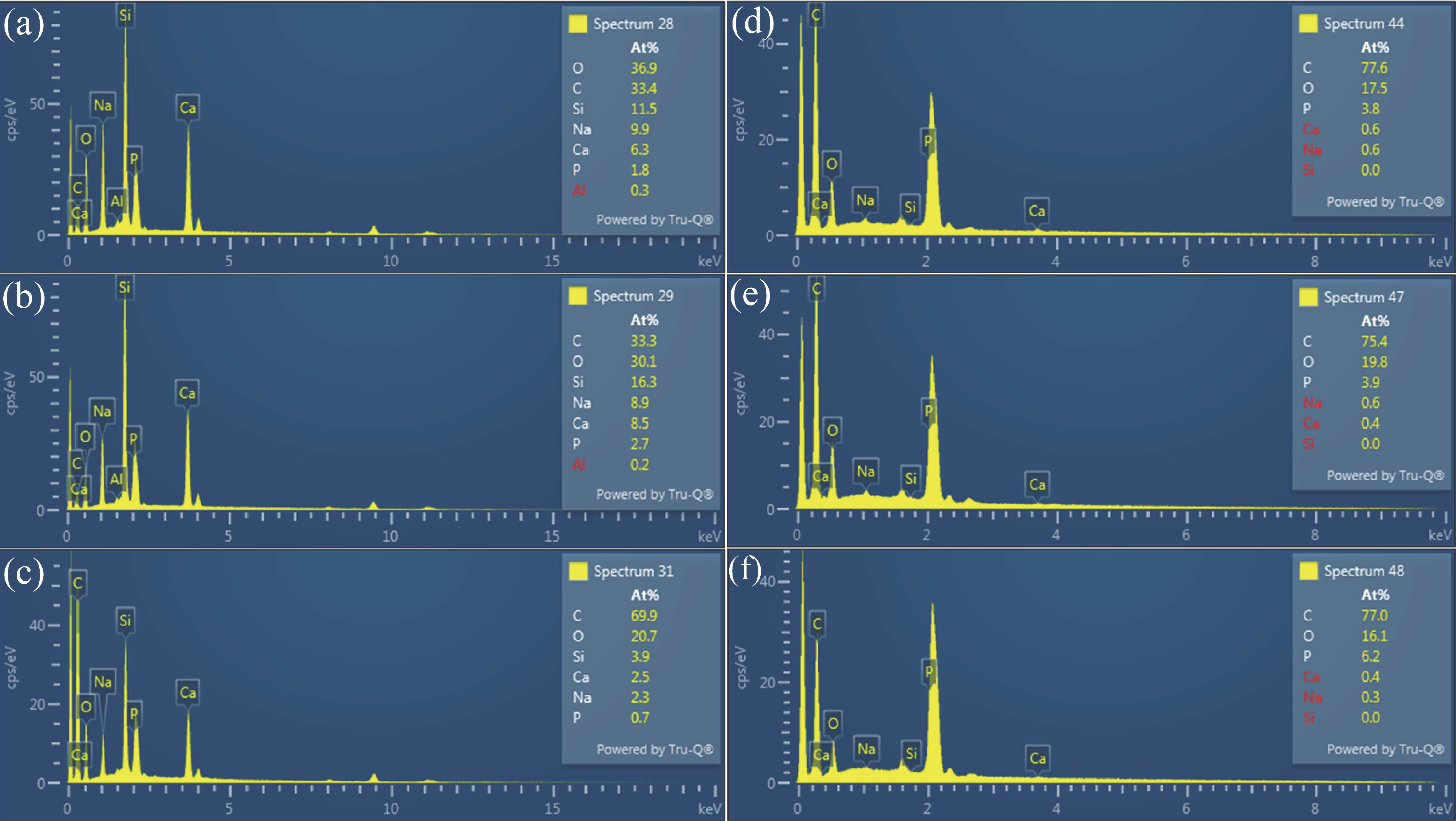
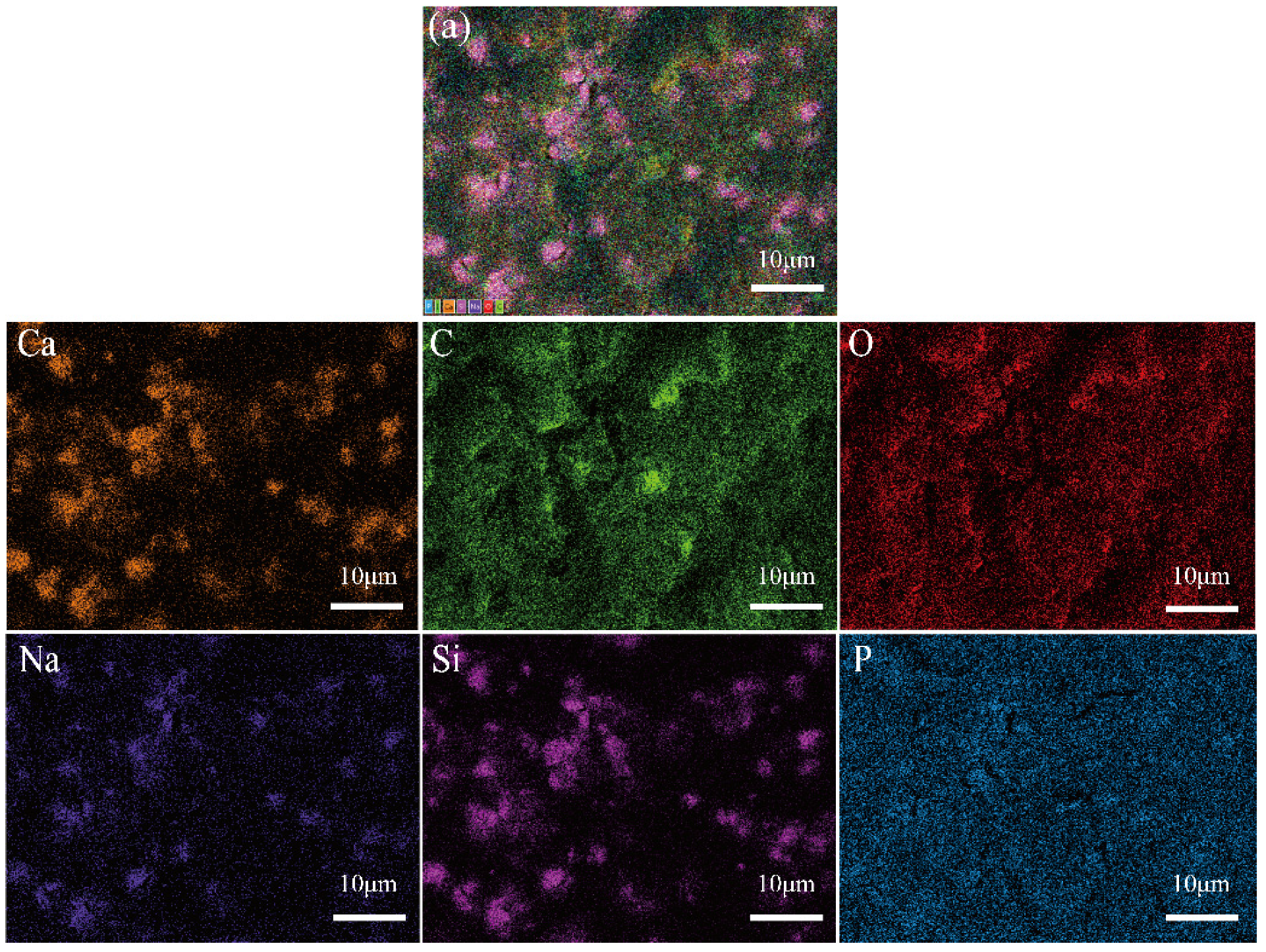
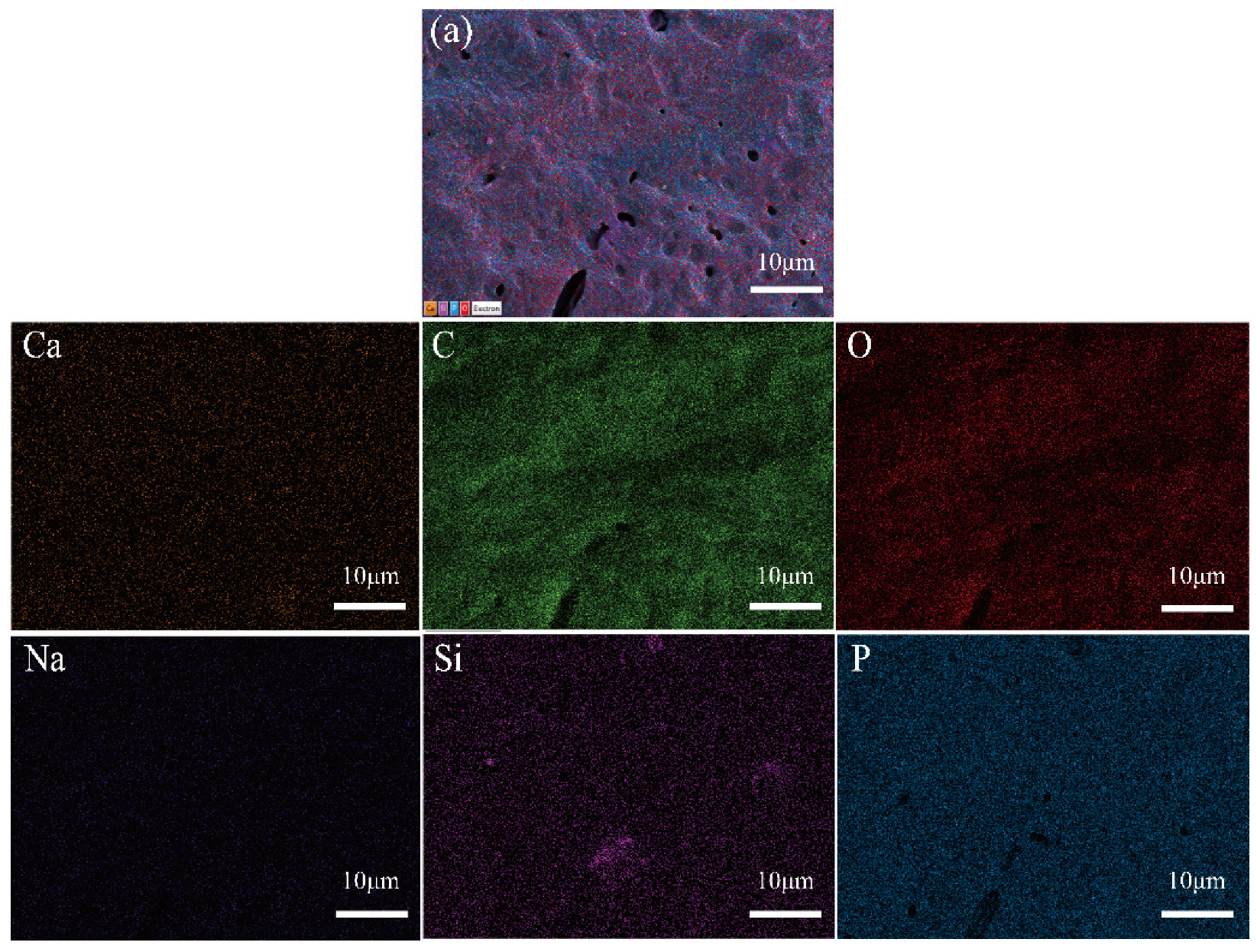
3.3.2. Scanning Electron Microscopy/Energy-Dispersive Spectrometry
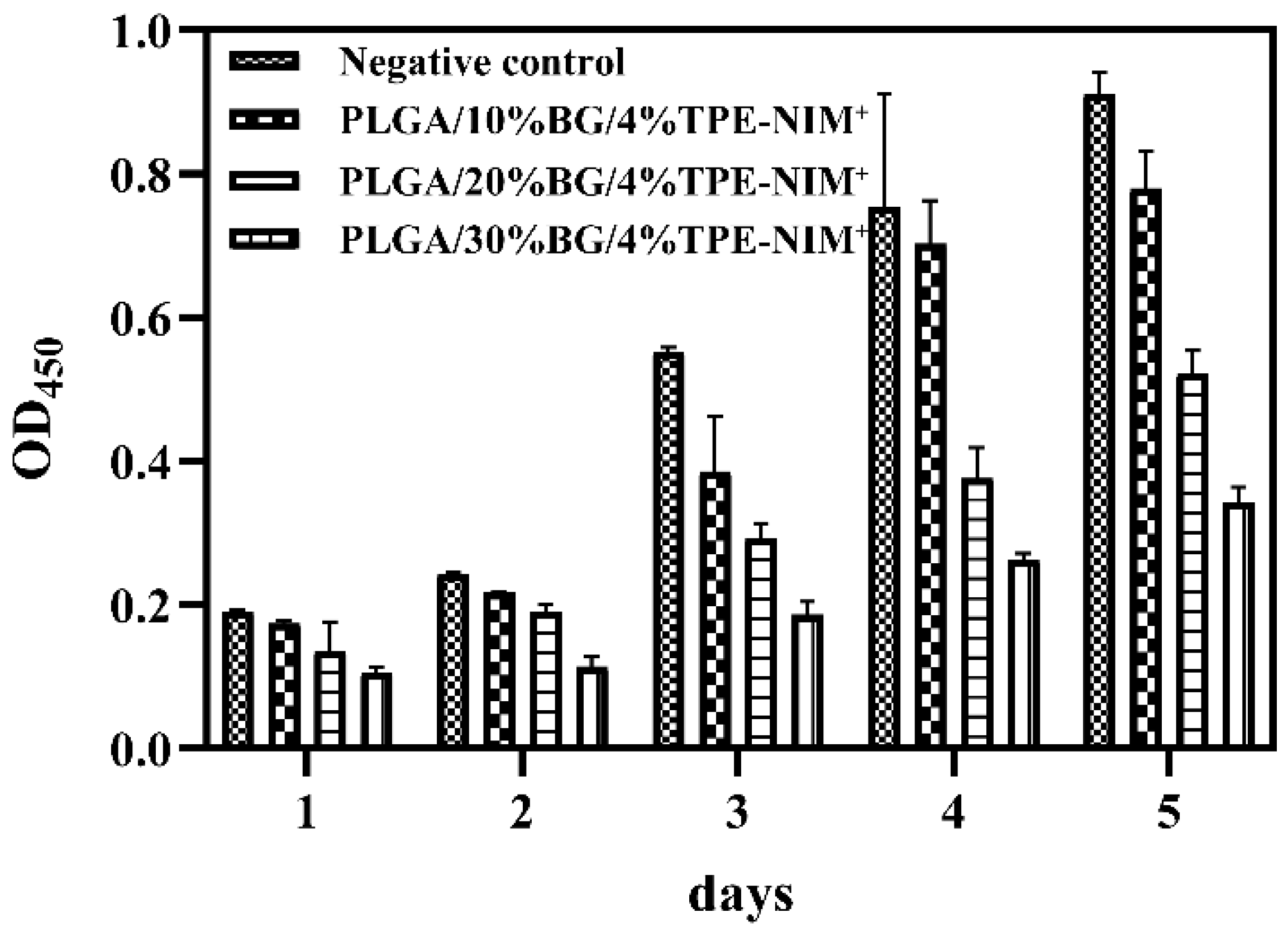
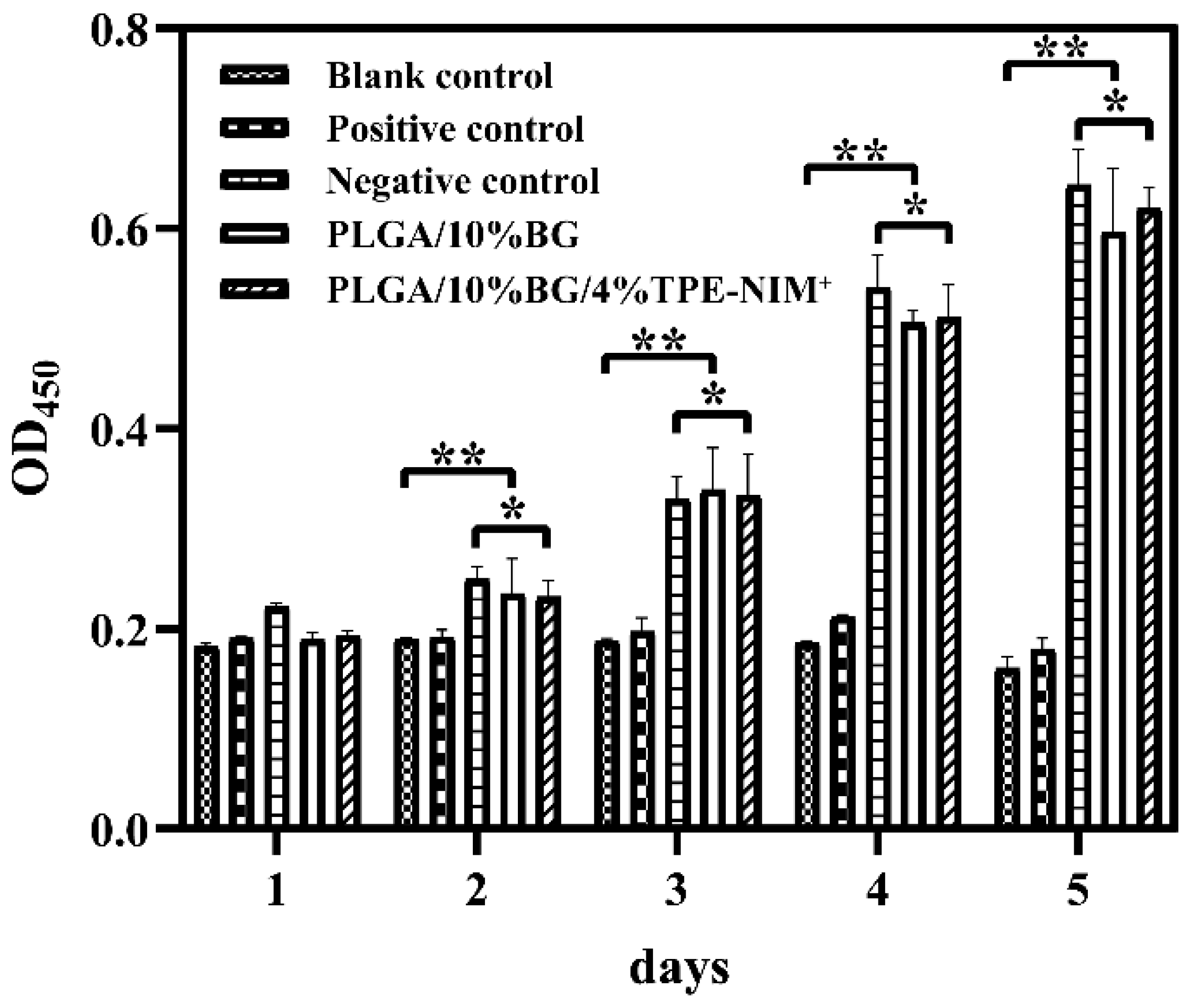
3.4. Cytotoxicity Assay
3.5. Antibacterial Activity Test
3.6. Staining Abilities of TPE-NIM+ toward Pathogens
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kinane, D.F.; Stathopoulou, P.G.; Papapanou, P.N. Periodontal diseases. Nat. Rev. Dis. Primers 2017, 3, 17038. [Google Scholar] [CrossRef] [PubMed]
- Schwarz, F.; Derks, J.; Monje, A.; Wang, H.L. Peri-implantitis. J. Periodontol. 2018, 89 (Suppl. 1), S267–S290. [Google Scholar] [CrossRef] [PubMed]
- Machtei, E.E.; Cho, M.I.; Dunford, R.; Norderyd, J.; Zambon, J.J.; Genco, R.J. Clinical, microbiological, and histological factors which influence the success of regenerative periodontal therapy. J. Periodontol. 1994, 65, 154–161. [Google Scholar] [CrossRef] [PubMed]
- Carcuac, O.; Derks, J.; Charalampakis, G.; Abrahamsson, I.; Wennström, J.; Berglundh, T. Adjunctive Systemic and Local Antimicrobial Therapy in the Surgical Treatment of Peri-implantitis: A Randomized Controlled Clinical Trial. J. Dent. Res. 2016, 95, 50–57. [Google Scholar] [CrossRef]
- Renvert, S.; Roos-Jansåker, A.M.; Claffey, N. Non-surgical treatment of peri-implant mucositis and peri-implantitis: A literature review. J. Clin. Periodontol. 2008, 35, 305–315. [Google Scholar] [CrossRef]
- Javed, F.; Alghamdi, A.S.; Ahmed, A.; Mikami, T.; Ahmed, H.B.; Tenenbaum, H.C. Clinical efficacy of antibiotics in the treatment of peri-implantitis. Int. Dent J. 2013, 63, 169–176. [Google Scholar] [CrossRef]
- Pietruska, M.; Dolińska, E.; Milewski, R.; Sculean, A. Effect of systemic antibiotics on the outcomes of regenerative periodontal surgery in intrabony defects: A randomized, controlled, clinical study. Clin. Oral. Investig. 2021, 25, 2959–2968. [Google Scholar] [CrossRef]
- Hu, S.; Chang, J.; Liu, M.; Ning, C. Study on antibacterial effect of 45S5 Bioglass. J. Mater. Sci. Mater. Med. 2009, 20, 281–286. [Google Scholar] [CrossRef]
- Porwal, H.; Grasso, S.; Cordero-Arias, L.; Li, C.; Boccaccini, A.R.; Reece, M.J. Processing and bioactivity of 45S5 Bioglass((R))-graphene nanoplatelets composites. J. Mater. Sci. Mater. Med. 2014, 25, 1403–1413. [Google Scholar] [CrossRef]
- Hench, L.L. Bioactive materials: The potential for tissue regeneration. J. BioMed. Mater. Res. 1998, 41, 511–518. [Google Scholar] [CrossRef]
- Stoor, P.; Söderling, E.; Salonen, J.I. Antibacterial effects of a bioactive glass paste on oral microorganisms. Acta Odontol. Scand. 1998, 56, 161–165. [Google Scholar] [CrossRef] [PubMed]
- Verrier, S.; Blaker, J.J.; Maquet, V.; Hench, L.L.; Boccaccini, A.R. PDLLA/Bioglass composites for soft-tissue and hard-tissue engineering: An in vitro cell biology assessment. Biomaterials 2004, 25, 3013–3021. [Google Scholar] [CrossRef] [PubMed]
- Kokubo, T.; Takadama, H. How useful is SBF in predicting in vivo bone bioactivity? Biomaterials 2006, 27, 2907–2915. [Google Scholar] [CrossRef] [PubMed]
- Hench, L.L. Bioceramics: From Concept to Clinic. J. Am. Ceram. Soc. 1991, 74, 1487–1510. [Google Scholar] [CrossRef]
- Hench, L.L.; Jones, J.R. Bioactive Glasses: Frontiers and Challenges. Front. Bioeng Biotechnol. 2015, 3, 194. [Google Scholar] [CrossRef] [Green Version]
- Hench, L.L.; Splinter, R.J.; Allen, W.C.; Greenlee, T.K. Bonding mechanisms at the interface of ceramic prosthetic materials. J. Biomed. Mater. Res. 1971, 5, 117–141. [Google Scholar] [CrossRef]
- Munukka, E.; Leppäranta, O.; Korkeamäki, M.; Vaahtio, M.; Peltola, T.; Zhang, D.; Hupa, L.; Ylänen, H.; Salonen, J.I.; Viljanen, M.K.; et al. Bactericidal effects of bioactive glasses on clinically important aerobic bacteria. J. Mater. Sci. Mater. Med. 2008, 19, 27–32. [Google Scholar] [CrossRef]
- Drago, L.; Toscano, M.; Bottagisio, M. Recent Evidence on Bioactive Glass Antimicrobial and Antibiofilm Activity: A Mini-Review. Materials 2018, 11, 326. [Google Scholar] [CrossRef] [Green Version]
- Allan, I.; Newman, H.; Wilson, M. Antibacterial activity of particulate bioglass against supra- and subgingival bacteria. Biomaterials 2001, 22, 1683–1687. [Google Scholar] [CrossRef]
- Mohammadi, Z.; Dummer, P.M. Properties and applications of calcium hydroxide in endodontics and dental traumatology. Int. Endod. J. 2011, 44, 697–730. [Google Scholar] [CrossRef]
- Xu, Y.T.; Wang, M.T.; Wu, Q.; Chen, Y.M.; Li, C.H. Study on the antibacterial effect of bioactive glass on cariogenic bacteria and supragingival plaque in vitro. Stomatology 2014, 34, 167–170. (In Chinese) [Google Scholar] [CrossRef]
- Drago, L.; De Vecchi, E.; Bortolin, M.; Toscano, M.; Mattina, R.; Romanò, C.L. Antimicrobial activity and resistance selection of different bioglass S53P4 formulations against multidrug resistant strains. Future Microbiol. 2015, 10, 1293–1299. [Google Scholar] [CrossRef] [PubMed]
- Lafaurie, G.I.; Sabogal, M.A.; Castillo, D.M.; Rincón, M.V.; Gómez, L.A.; Lesmes, Y.A.; Chambrone, L. Microbiome and Microbial Biofilm Profiles of Peri-Implantitis: A Systematic Review. J. Periodontol. 2017, 88, 1066–1089. [Google Scholar] [CrossRef] [PubMed]
- Mark Welch, J.L.; Ramirez-Puebla, S.T.; Borisy, G.G. Oral Microbiome Geography: Micron-Scale Habitat and Niche. Cell Host Microbe 2020, 28, 160–168. [Google Scholar] [CrossRef]
- Sayed, S.M.; Xu, K.F.; Jia, H.R.; Yin, F.F.; Ma, L.; Zhang, X.; Khan, A.; Ma, Q.; Wu, F.G.; Lu, X. Naphthalimide-based multifunctional AIEgens: Selective, fast, and wash-free fluorescence tracking and identification of Gram-positive bacteria. Anal Chim. Acta 2021, 1146, 41–52. [Google Scholar] [CrossRef]
- Sayed, S.M.; Jia, H.R.; Jiang, Y.W.; Zhu, Y.X.; Ma, L.; Yin, F.; Hussain, I.; Khan, A.; Ma, Q.; Wu, F.G.; et al. Photostable AIE probes for wash-free, ultrafast, and high-quality plasma membrane staining. J. Mater. Chem. B 2021, 9, 4303–4308. [Google Scholar] [CrossRef]
- He, X.; Xiong, L.H.; Zhao, Z.; Wang, Z.; Luo, L.; Lam, J.W.Y.; Kwok, R.T.K.; Tang, B.Z. AIE-based theranostic systems for detection and killing of pathogens. Theranostics 2019, 9, 3223–3248. [Google Scholar] [CrossRef]
- Kobayashi, H.; Ogawa, M.; Alford, R.; Choyke, P.L.; Urano, Y. New strategies for fluorescent probe design in medical diagnostic imaging. Chem. Rev. 2010, 110, 2620–2640. [Google Scholar] [CrossRef] [Green Version]
- Hu, R.; Zhou, F.; Zhou, T.; Shen, J.; Wang, Z.; Zhao, Z.; Qin, A.; Tang, B.Z. Specific discrimination of gram-positive bacteria and direct visualization of its infection towards mammalian cells by a DPAN-based AIEgen. Biomaterials 2018, 187, 47–54. [Google Scholar] [CrossRef]
- Wang, H.; Agarwal, P.; Zhao, S.; Xu, R.X.; Yu, J.; Lu, X.; He, X. Hyaluronic acid-decorated dual responsive nanoparticles of Pluronic F127, PLGA, and chitosan for targeted co-delivery of doxorubicin and irinotecan to eliminate cancer stem-like cells. Biomaterials 2015, 72, 74–89. [Google Scholar] [CrossRef] [Green Version]
- Wang, N.; Wu, X.S.; Li, C.; Feng, M.F. Synthesis, characterization, biodegradation, and drug delivery application of biodegradable lactic/glycolic acid polymers: I. Synthesis and characterization. J. BioMater. Sci. Polym Ed 2000, 11, 301–318. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, X.S. Synthesis, characterization, biodegradation, and drug delivery application of biodegradable lactic/glycolic acid polymers: Part III. Drug delivery application. Artif. Cells Blood Substit Immobil. Biotechnol. 2004, 32, 575–591. [Google Scholar] [CrossRef] [PubMed]
- Zadpoor, A.A. Relationship between in vitro apatite-forming ability measured using simulated body fluid and in vivo bioactivity of biomaterials. Mater. Sci. Eng. C Mater. Biol. Appl. 2014, 35, 134–143. [Google Scholar] [CrossRef] [PubMed]
- Hu, R.; Maldonado, J.-L.; Rodriguez, M.; Deng, C.; Jim, C.; Jacky, W.; Yuen, M.; Ramos-Ortiz, G.; Tang, B. Luminogenic materials constructed from tetraphenylethene building blocks: Synthesis, aggregation-induced emission, two-photon absorption, light refraction, and explosive detection. J. Mater. Chem. 2011, 22, 232–240. [Google Scholar] [CrossRef] [Green Version]
- Baier, R.E. Surface behaviour of biomaterials: The theta surface for biocompatibility. J. Mater. Sci. Mater. Med. 2006, 17, 1057–1062. [Google Scholar] [CrossRef]
- Poh, P.S.P.; Hutmacher, D.W.; Holzapfel, B.M.; Solanki, A.K.; Stevens, M.M.; Woodruff, M.A. In vitro and in vivo bone formation potential of surface calcium phosphate-coated polycaprolactone and polycaprolactone/bioactive glass composite scaffolds. Acta BioMater. 2016, 30, 319–333. [Google Scholar] [CrossRef]
- Rupp, F.; Liang, L.; Geis-Gerstorfer, J.; Scheideler, L.; Hüttig, F. Surface characteristics of dental implants: A review. Dent Mater. 2018, 34, 40–57. [Google Scholar] [CrossRef]
- Arima, Y.; Iwata, H. Effect of wettability and surface functional groups on protein adsorption and cell adhesion using well-defined mixed self-assembled monolayers. Biomaterials 2007, 28, 3074–3082. [Google Scholar] [CrossRef]
- Berglundh, T.; Zitzmann, N.U.; Donati, M. Are peri-implantitis lesions different from periodontitis lesions? J. Clin Periodontol 2011, 38 (Suppl. 11), 188–202. [Google Scholar] [CrossRef]
- Larsen, T.; Fiehn, N.E. Dental biofilm infections—An update. Apmis 2017, 125, 376–384. [Google Scholar] [CrossRef]
- Mysak, J.; Podzimek, S.; Sommerova, P.; Lyuya-Mi, Y.; Bartova, J.; Janatova, T.; Prochazkova, J.; Duskova, J. Porphyromonas gingivalis: Major periodontopathic pathogen overview. J. Immunol. Res. 2014, 2014, 476068. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhuang, Y.; Lin, K.; Yu, H. Advance of Nano-Composite Electrospun Fibers in Periodontal Regeneration. Front Chem. 2019, 7, 495. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Dong, Y.; Zhang, N.; Shi, J.; Zhang, X.; Qi, C.; Midgley, A.C.; Wang, S. Potentials of sandwich-like chitosan/polycaprolactone/gelatin scaffolds for guided tissue regeneration membrane. Mater. Sci. Eng C Mater. Biol. Appl. 2020, 109, 110618. [Google Scholar] [CrossRef] [PubMed]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wei, Y.-W.; Sayed, S.M.; Zhu, W.-W.; Xu, K.-F.; Wu, F.-G.; Xu, J.; Nie, H.-P.; Wang, Y.-L.; Lu, X.-L.; Ma, Q. Antibacterial and Fluorescence Staining Properties of an Innovative GTR Membrane Containing 45S5BGs and AIE Molecules In Vitro. Nanomaterials 2022, 12, 641. https://doi.org/10.3390/nano12040641
Wei Y-W, Sayed SM, Zhu W-W, Xu K-F, Wu F-G, Xu J, Nie H-P, Wang Y-L, Lu X-L, Ma Q. Antibacterial and Fluorescence Staining Properties of an Innovative GTR Membrane Containing 45S5BGs and AIE Molecules In Vitro. Nanomaterials. 2022; 12(4):641. https://doi.org/10.3390/nano12040641
Chicago/Turabian StyleWei, Yu-Wen, Sayed Mir Sayed, Wei-Wen Zhu, Ke-Fei Xu, Fu-Gen Wu, Jing Xu, He-Peng Nie, Yu-Li Wang, Xiao-Lin Lu, and Qian Ma. 2022. "Antibacterial and Fluorescence Staining Properties of an Innovative GTR Membrane Containing 45S5BGs and AIE Molecules In Vitro" Nanomaterials 12, no. 4: 641. https://doi.org/10.3390/nano12040641
APA StyleWei, Y.-W., Sayed, S. M., Zhu, W.-W., Xu, K.-F., Wu, F.-G., Xu, J., Nie, H.-P., Wang, Y.-L., Lu, X.-L., & Ma, Q. (2022). Antibacterial and Fluorescence Staining Properties of an Innovative GTR Membrane Containing 45S5BGs and AIE Molecules In Vitro. Nanomaterials, 12(4), 641. https://doi.org/10.3390/nano12040641







