Potential Development of N-Doped Carbon Dots and Metal-Oxide Carbon Dot Composites for Chemical and Biosensing
Abstract
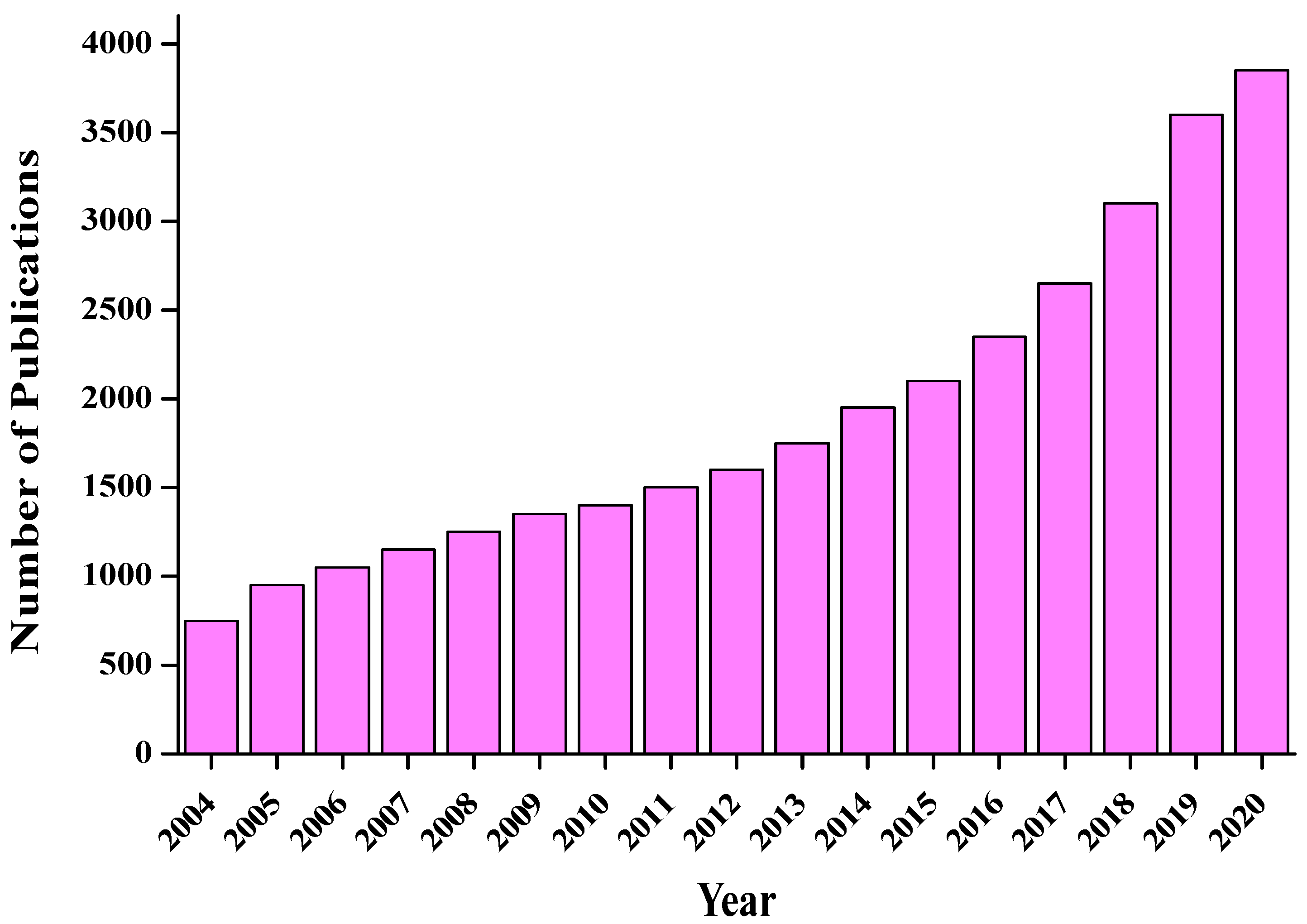
1. Introduction
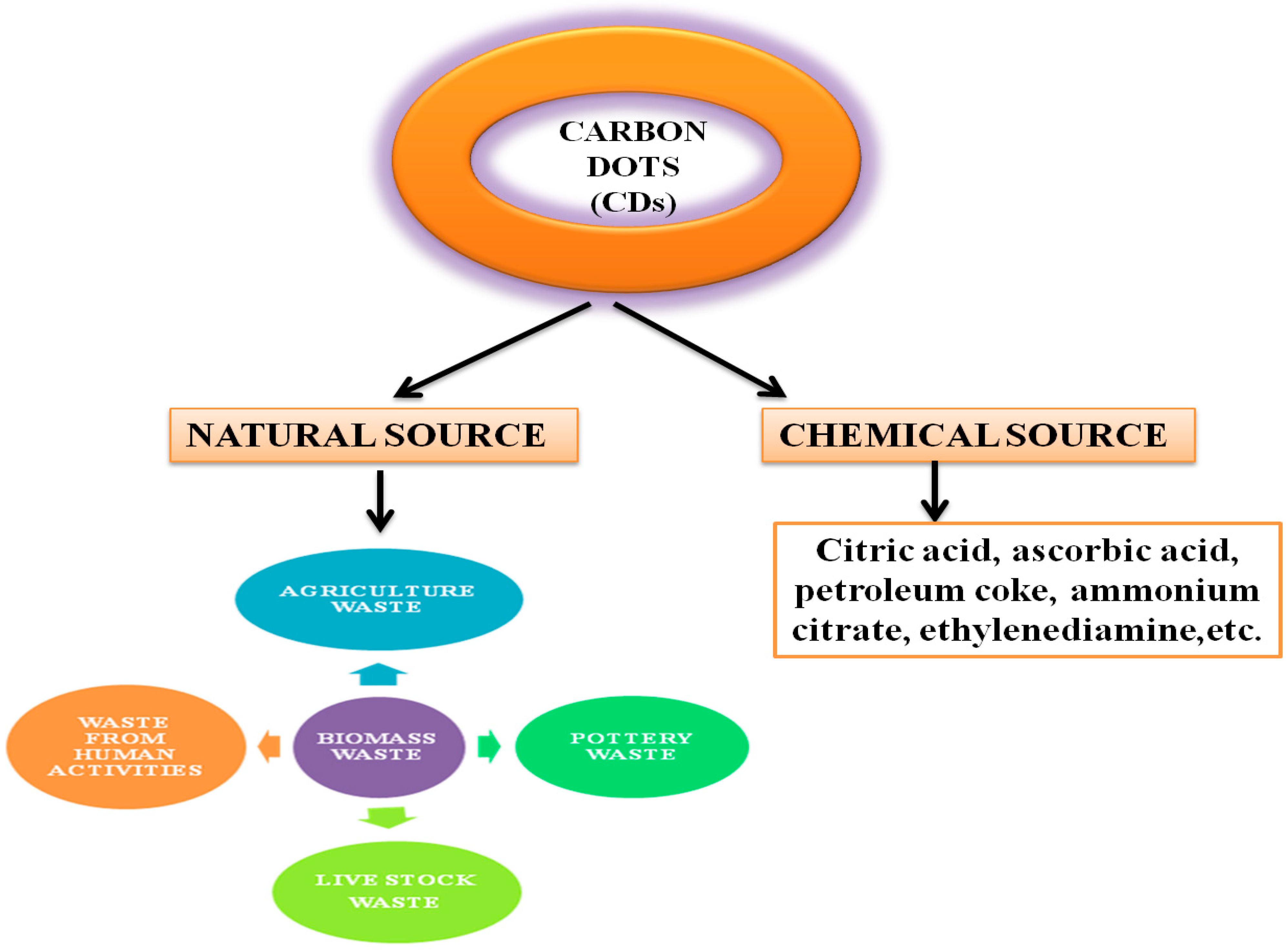
2. Carbon Sources for the Preparation of Carbon Dots
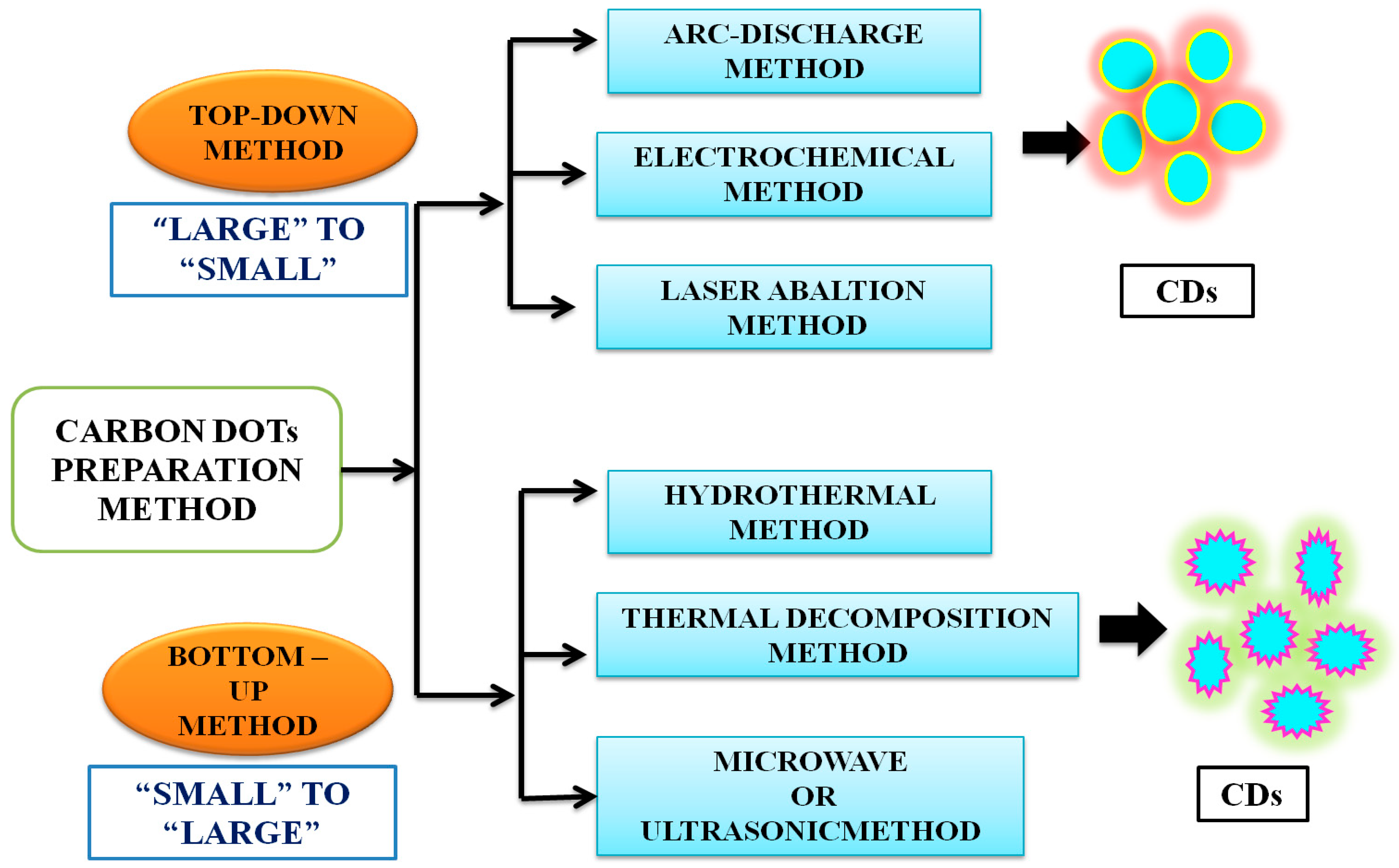
3. Methods of Preparation of CDs
4. Properties of Carbon Dots
4.1. Structural Properties
4.2. Optical Properties
4.2.1. Absorbance
4.2.2. Fluorescence Properties
4.2.3. Upconversion Photoluminescence
4.2.4. Electrochemiluminescence
4.3. Cytotoxicity and Biocompatibility
5. Surface Functionalization
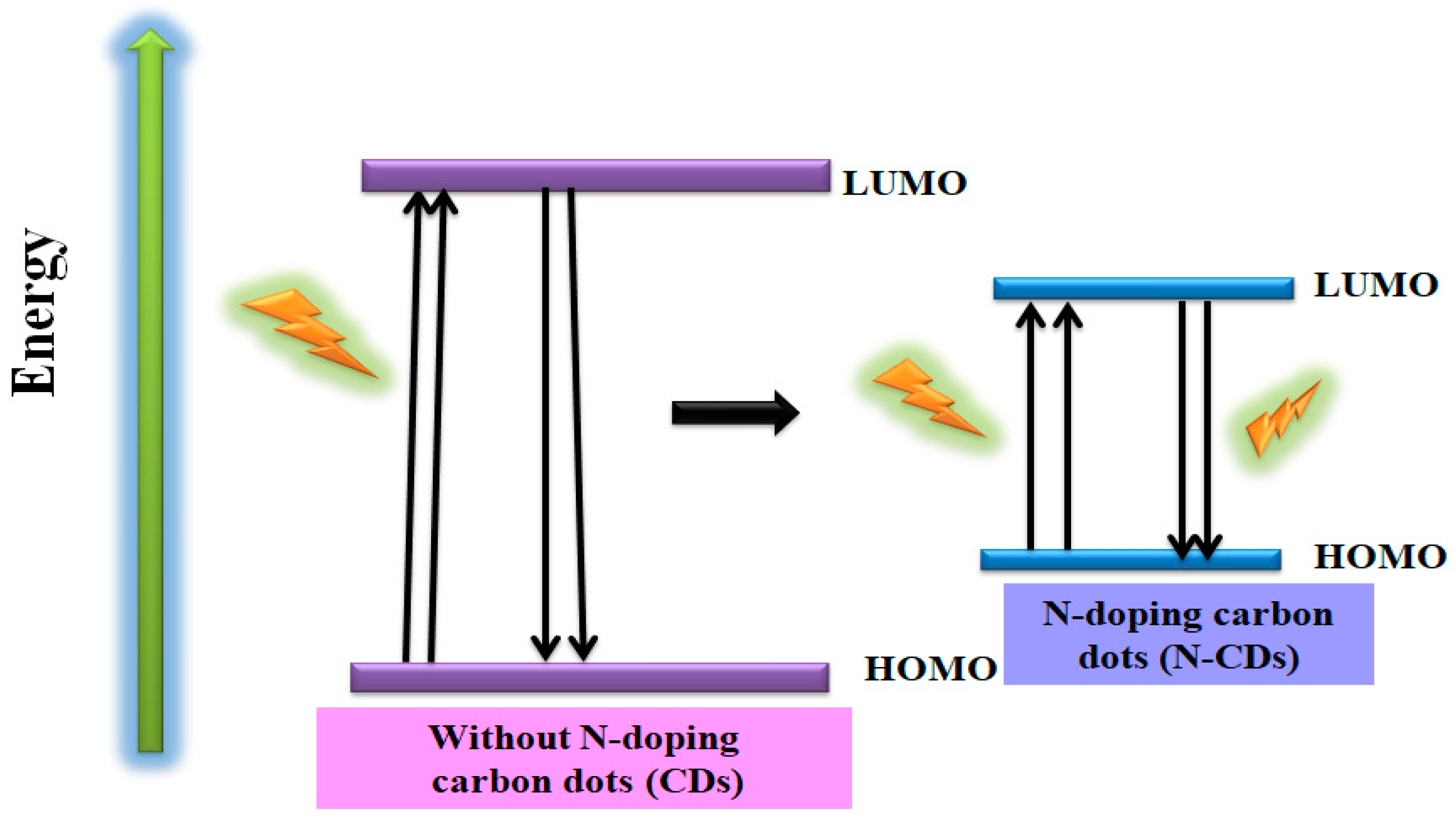
5.1. Doping of “N” Atom into CDs
5.1.1. N-Doped CDs in “Chemical Sensing”
5.1.2. N-Doped Carbon Dots in “Biosensing”
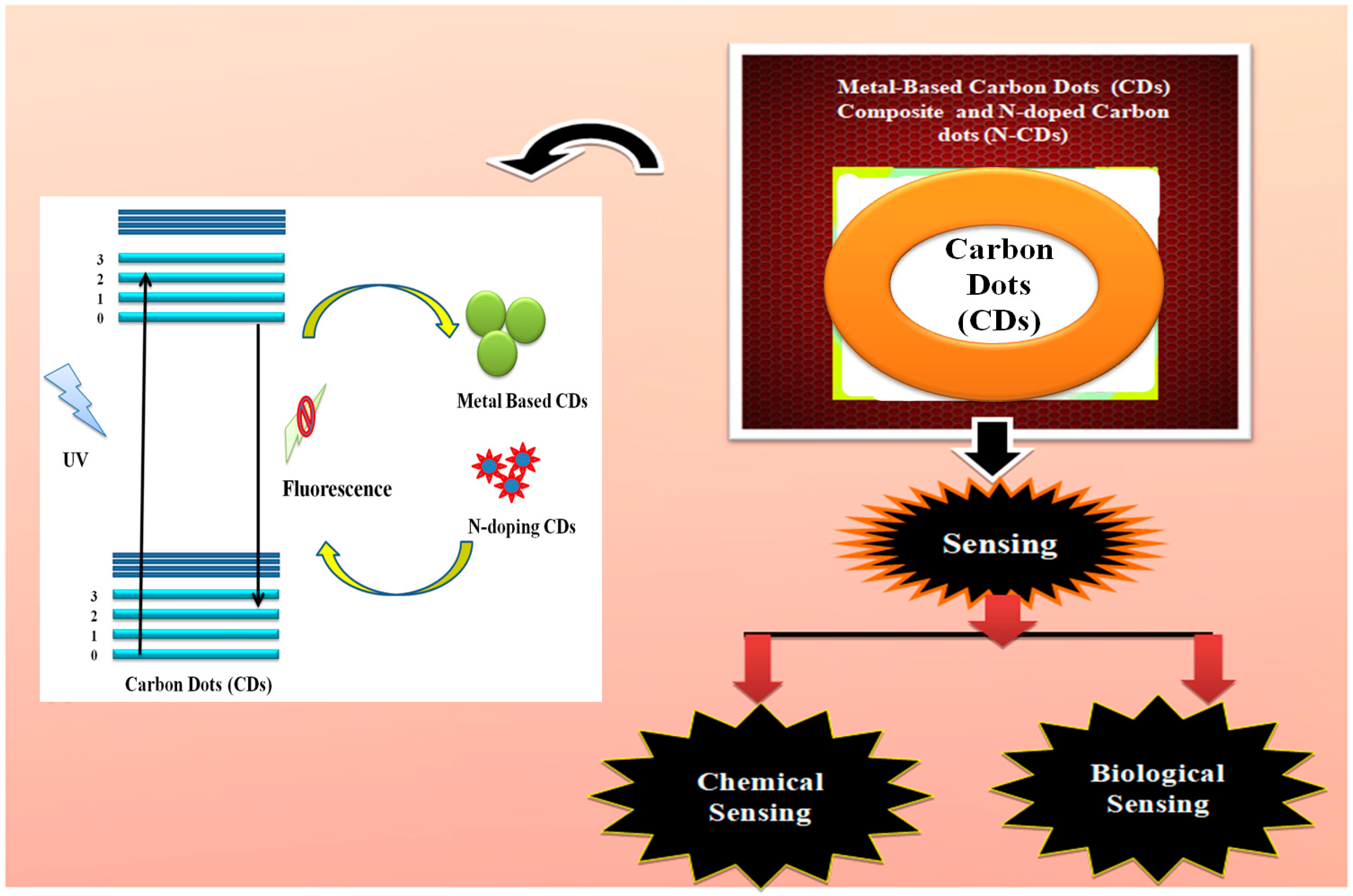
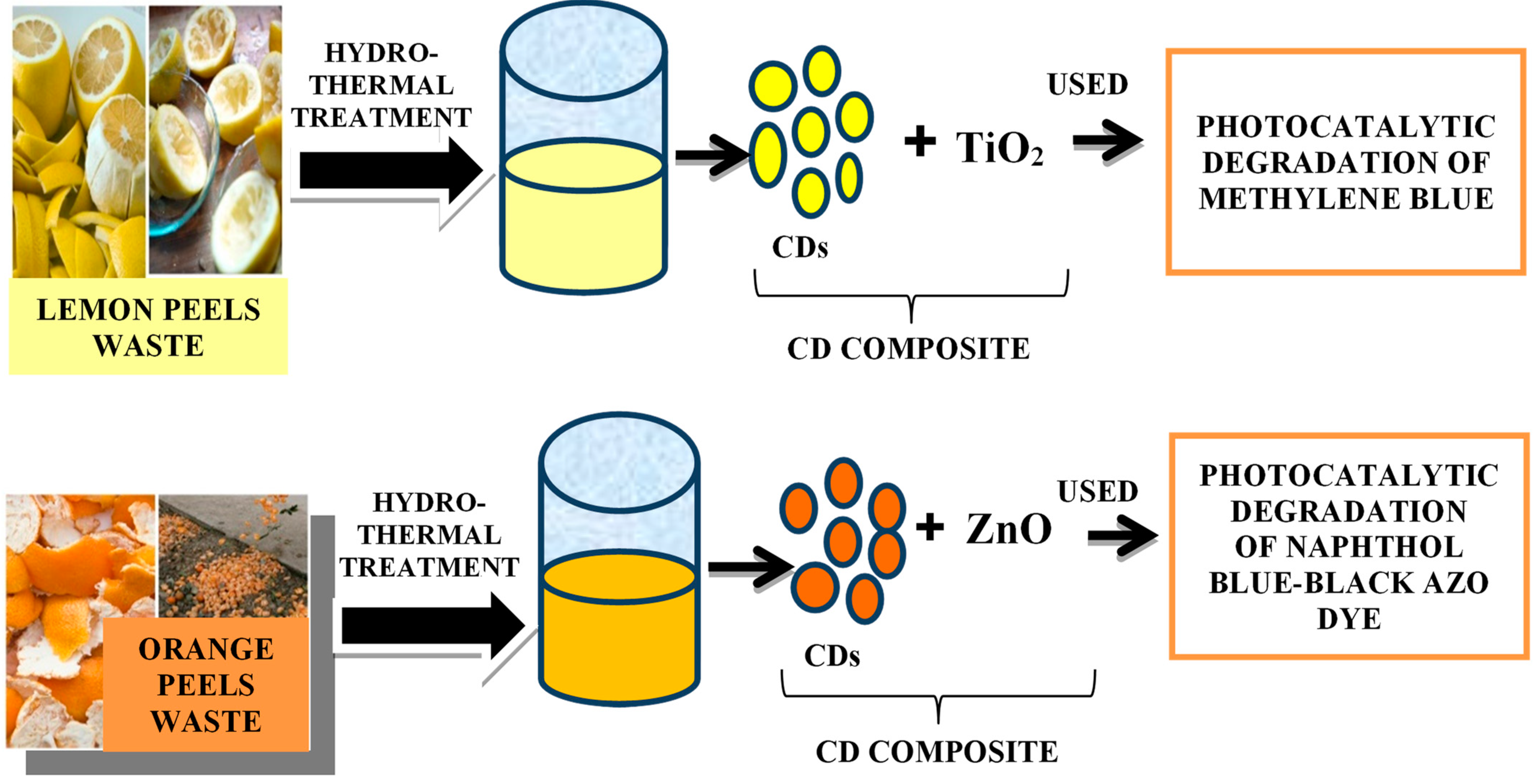
5.2. Carbon Dot Composites
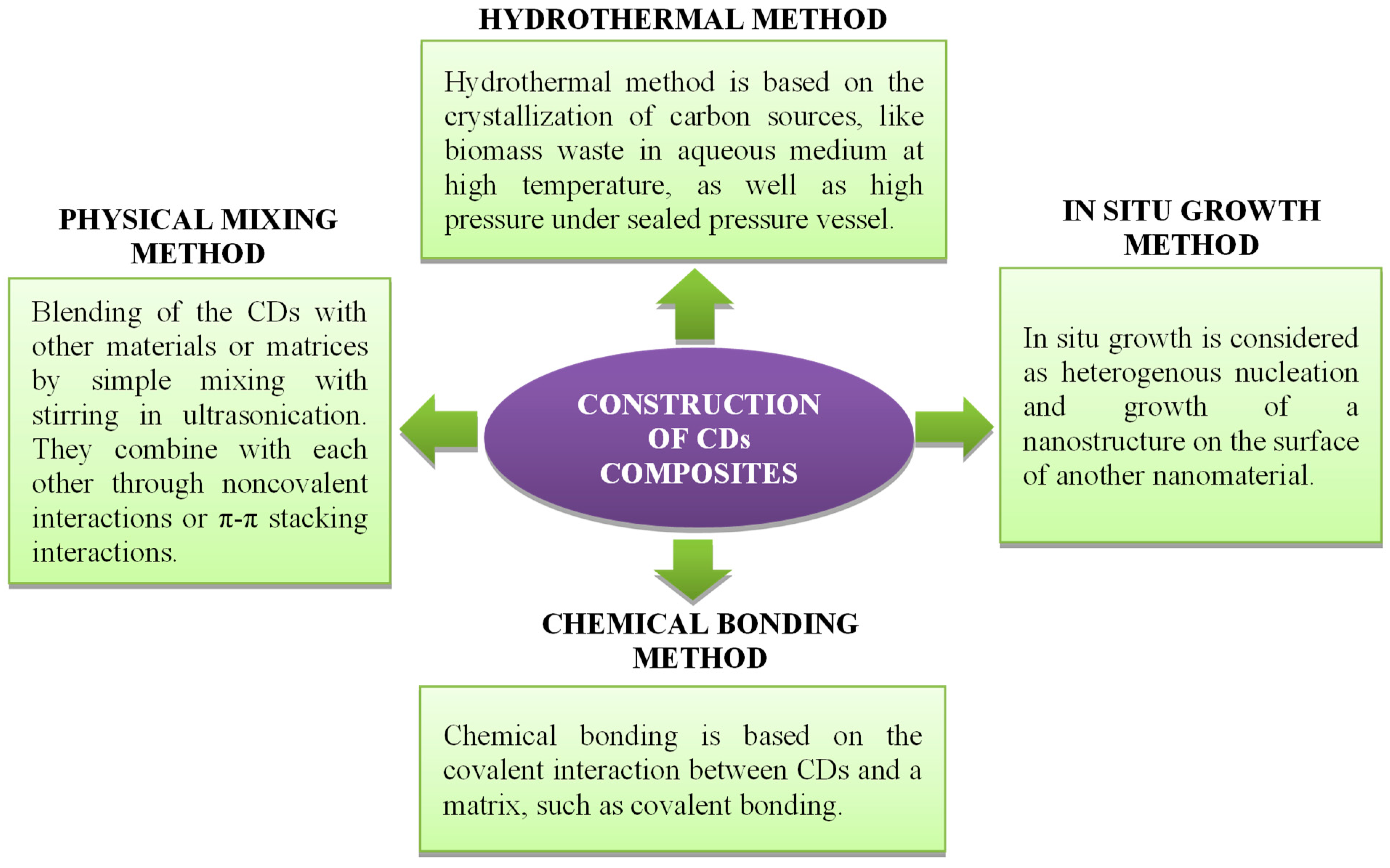
5.2.1. Synthesis of CD Composites
5.2.2. Metal-Based CD Composites
5.2.3. Metal-Based CD Composites Used in “Chemical Sensing”
5.2.4. Metal-Based CD Composites in “Biosensing”
6. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Schwarzenbach, R.P.; Egli, T.; Hofstetter, T.B.; Von Gunten, U.V.; Wehrli, B. Global Water Pollution and Human Health. Annu. Rev. Environ. Resour. 2010, 35, 109–136. [Google Scholar] [CrossRef]
- Jeevanantham, S.; Saravanan, A.; Hemavathy, R.; Kumar, P.S.; Yaashikaa, P.; Yuvaraj, D. Removal of toxic pollutants from water environment by phytoremediation: A survey on application and future prospects. Environ. Technol. Innov. 2019, 13, 264–276. [Google Scholar] [CrossRef]
- Lu, F.; Astruc, D. Nanocatalysts and other nanomaterials for water remediation from organic pollutants. Coord. Chem. Rev. 2020, 408, 213180. [Google Scholar] [CrossRef]
- Nuengmatcha, P.; Chanthai, S.; Mahachai, R.; Oh, W.C. Sonocatalytic performance of ZnO/graphene/TiO2 nanocomposite for degradation of dye pollutants (methylene blue texbrite BAC-L, texbrite BBU-L and texbrite NFW-L) under ultrasonic irradiation. Dyes Pigm. 2016, 134, 487–497. [Google Scholar] [CrossRef]
- Velez, N.S.P.; Gordillo, A.H.; Bizarro, M. Morphological effect of ZnO nanoflakes and nanobars on the photocatalytic dye degradation. Catal. Today 2017, 287, 106–112. [Google Scholar] [CrossRef]
- Hou, C.; Jiao, T.; Xing, R.; Chen, Y.; Zhou, J.; Zhang, L. Preparation of TiO2 nanoparticles modified electrospun nanocomposite membranes toward efficient dye degradation for wastewater treatment. J. Taiwan Inst. Chem. Eng. 2017, 78, 118–126. [Google Scholar] [CrossRef]
- Liu, J.; Mattiasson, B. Microbial BOD sensors for wastewater analysis. Water Res. 2002, 26, 3786–3802. [Google Scholar] [CrossRef]
- Mello, L.D.; Kubota, L.T. Review of the use of biosensors as analytical tools in the food and drink industries. Food Chem. 2002, 77, 237–256. [Google Scholar] [CrossRef]
- Sanz, V.C.; Mena, M.L.; González-Cortés, A.; Yáñez-Sedeño, P.; Pingarrón, J. Development of a tyrosinase biosensor based on gold nanoparticles-modified glassy carbon electrodes: Application to the measurement of a bioelectrochemical polyphenols index in wines. Anal. Chim. Acta 2005, 528, 1–8. [Google Scholar] [CrossRef]
- Kessler, A.; Ostfeld, A.; Sinai, G. Detecting Accidental Contaminations in Municipal Water Networks. J. Water Resour. Plan. Manag. 1998, 124, 192–198. [Google Scholar] [CrossRef]
- Zulkifli, S.N.; Rahim, H.A.; Lau, W.-J. Detection of contaminants in water supply: A review on state-of-the-art monitoring technologies and their applications. Sens. Actuators B Chem. 2018, 255, 2657–2689. [Google Scholar] [CrossRef] [PubMed]
- Khan, Z.G.; Patil, P.O. A comprehensive review on carbon dots and graphene quantum dots based fluorescent sensor for biothiols. Microchem. J. 2020, 157, 105011. [Google Scholar] [CrossRef]
- Das, P.; Maruthapandi, M.; Saravanan, A.; Natan, M.; Jacobi, J.; Banin, E.; Gedanken, A. Carbon dots for heavy metal sensing, pH-sensitive cargo delivery, and antibacterial applications. ACS Appl. Nano Mater. 2020, 3, 11777–11790. [Google Scholar] [CrossRef]
- Dakshayinia, B.S.; Reddy, K.R.; Mishrac, A.; Shettid, N.P.; Maloded, S.J.; Basuc, S.; Naveene, S.; Raghu, A.V. Role of conducting polymer and metal oxide-based hybrids for applications in ampereometric sensors and biosensors. Microchem. J. 2019, 147, 7–24. [Google Scholar] [CrossRef]
- Ijaz, I.; Gilani, E.; Nazir, A.; Bukhari, A. Detail review on chemical, physical and green synthesis, classification, characterizations and applications of nanoparticles. Green Chem. Lett. Rev. 2020, 13, 223–245. [Google Scholar] [CrossRef]
- Madkour, L.H. Introduction to Nanotechnology (NT) and Nanomaterials (NMs). J. Nano Electron. Mater. 2009, 116, 1–47. [Google Scholar] [CrossRef]
- Miklicanin, E.O.; Badnjevic, A.; Kazlagic, A.; Hajlova, M. Nanomaterials: A brief review. Health Technol. 2020, 10, 51–59. [Google Scholar] [CrossRef]
- Wang, R.; Lu, K.-Q.; Tang, Z.-R.; Xu, Y.-J. Recent progress in carbon quantum dots: Synthesis, properties and applications in photocatalysis. J. Mater. Chem. A 2017, 5, 3717–3734. [Google Scholar] [CrossRef]
- Zhou, Y.; Sharma, S.K.; Peng, Z.; Leblanc, R.M. Polymers in CDs: A review. Polym. J. 2017, 9, 67. [Google Scholar]
- Sakdaronnarong, C.; Sangjan, A.; Boonsith, S.; Kim, D.C.; Shin, H.S. Recent developments in synthesis and photocatalytic applications of carbon dots. Catalysts 2020, 10, 320. [Google Scholar] [CrossRef]
- Farshbaf, M.; Davaran, S.; Rahimi, F.; Annabi, N.; Salehi, R.; Akbarzadeh, A. Carbon quantum dots: Recent progresses on the synthesis, surface modification, and applications. Artif. Cell Nanomed. Biotechnol. 2018, 46, 1331–1348. [Google Scholar] [CrossRef] [PubMed]
- Part, F.; Zaba, C.; Bixner, O.; Grünewald, T.A.; Michor, H.; Küpcü, S.; Debreczeny, M.; De Vito Francesco, E.; Lassenberger, A.; Schrittwieser, S.; et al. Doping method determines para-or superparamagnetic properties of photostable and surface-modifiable quantum dots for multimodal bioimaging. Chem. Mater. 2018, 30, 4233–4241. [Google Scholar] [CrossRef]
- Azam, N.; Ali, M.N.; Khan, T.J. Carbon quantum dots for biomedical applications: Review and analysis. Front. Mater. 2021, 8, 700403. [Google Scholar] [CrossRef]
- Hasan, A.M.M.; Hasan, M.A.; Reza, A.; Islam, M.M.; Susan, M.A.B.H. Carbon dots as nano-modules for energy conservation and storage. Mater. Today Commun. 2021, 29, 102732. [Google Scholar] [CrossRef]
- Ju, J.; Zhang, R.; He, S.; Chen, W. Nitrogen-doped graphene quantum dots-based fluorescent probe for the sensitive turn-on detection of glutathione and its cellular imaging. RSC Adv. 2014, 4, 52583–52589. [Google Scholar] [CrossRef]
- Gao, X.; Du, C.; Zhuang, Z.; Chen, W. Carbon quantum dot-based nanoprobes for metal ion detection. J. Mater. Chem. C 2016, 4, 6927–6945. [Google Scholar] [CrossRef]
- You, J.; Kim, E. Fluorescent nanostructures from aromatic diblock copolymers via atom transfer radical polymerization. J. Nanosci. Nanotechnol. 2016, 16, 10927–10934. [Google Scholar] [CrossRef]
- Pakkath, S.A.R.; Chetty, S.S.; Selvarasu, P.; Murugan, A.V.; Kumar, Y.; Periyasamy, L.; Santhakumar, M.; Sadras, S.R.; Santhakumar, K. Transition metal ion (Mn2+, Fe2+, Co2+, and Ni2+)-doped carbon dots synthesized via microwave-assisted pyrolysis: A potential nanoprobe for magneto-fluorescent dual-modality bioimaging. ACS Biomater. Sci. Eng. 2018, 4, 2582–2596. [Google Scholar] [CrossRef]
- Ghann, W.; Sharma, V.; Kang, H.; Karim, F.; Richards, B.; Mobin, S.M.; Uddin, J.; Rahman, M.M.; Hossain, F.; Kabir, H.; et al. The synthesis and characterization of carbon dots and their application in dye sensitized solar cell. Int. J. Hydrog. Energy 2019, 44, 14580–14587. [Google Scholar] [CrossRef]
- Pramanik, S.; Chatterjee, S.; Kumar, G.S.; Devi, P.S. Egg-shell derived carbon dots for base pair selective DNA binding and recognition. Phys. Chem. Chem. Phys. 2018, 20, 20476–20488. [Google Scholar] [CrossRef]
- Yao, Y.-Y.; Gedda, G.; Girma, W.M.; Yen, C.-L.; Ling, Y.-C.; Chang, J.-Y. Magnetofluorescent Carbon Dots Derived from Crab Shell for Targeted Dual-Modality Bioimaging and Drug Delivery. ACS Appl. Mater. Interfaces 2017, 9, 13887–13899. [Google Scholar] [CrossRef] [PubMed]
- Hak, C.H.; Leong, K.H.; Chin, Y.H.; Saravanan, P.; Tan, S.T.; Chong, W.C.; Sim, L.C. Water hyacinth derived carbon quantum dots and g-C3N4 composites for sunlight driven photodegradation of 2,4-dichlorophenol. SN Appl. Sci. 2020, 2, 1030. [Google Scholar] [CrossRef]
- Tejwan, N.; Saha, S.K.; Das, J. Multifaceted applications of green carbon dots synthesized from renewable sources. Adv. Colloid Interface Sci. 2020, 275, 102046. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.; Luo, Y.; Tsai, P.; Wang, J.; Chen, X. Metal ions doped carbon quantum dots: Synthesis, physicochemical properties, and their applications. TrAC Trends Anal. Chem. 2018, 103, 87–101. [Google Scholar] [CrossRef]
- Vijesh, K.R.; Sebastian, M.; Nampoori, V.P.N.; Thomas, S. Carbon dots decorated graphene oxide nanosheets prepared by a novel technique with enhanced nonlinear optical properties. AIP Adv. 2019, 9, 015219. [Google Scholar] [CrossRef]
- AjKaur, A.; Pandey, K.; Kaur, R.; Vashishat, N. Nanocomposite of carbon quantum dots and graphene quantum dots: Environmental application as sensors. Chemosensors 2022, 10, 367. [Google Scholar] [CrossRef]
- Chahal, S.; Yousefi, N.; Tufenkji, N. Green synthesis of high quantum yield carbon dots from phenylalanine and citric acid: Role of stoichiometry and nitrogen doping. ACS Sustain. Chem. Eng. 2020, 8, 5566–5575. [Google Scholar] [CrossRef]
- Solanki, P.R.; Kaushik, A.; Agrawal, V.V.; Malhotra, B.D. Nanostructured metal oxide-based biosensors. NPG Asia Mater. 2011, 3, 17–24. [Google Scholar] [CrossRef]
- Joseph, J.; Anappara, A.A. White-light-emitting carbon dots prepared by the electrochemical exfoliation of graphite. ChemPhysChem 2017, 18, 292–298. [Google Scholar] [CrossRef]
- Yarur, F.; Macairan, J.-R.; Naccache, R. Ratiometric detection of heavy metal ions using fluorescent carbon dots. Environ. Sci. Nano 2019, 6, 1121–1130. [Google Scholar] [CrossRef]
- Xue, B.; Yang, Y.; Sun, Y.; Fan, J.; Li, X.; Zhang, Z. Photoluminescent lignin hybridized carbon quantum dots composites for bioimaging applications. Int. J. Biol. Macromol. 2019, 122, 954–961. [Google Scholar] [CrossRef] [PubMed]
- Queiroz, L.S.; de Souza, L.K.; Thomaz, K.T.C.; Lima, E.T.L.; Filho, G.N.D.R.; Nascimento, L.A.S.D.; Pires, L.H.D.O.; Faial, K.D.C.F.; da Costa, C.E. Activated carbon obtained from amazonian biomass tailings (acai seed): Modification, characterization, and use for removal of metal ions from water. J. Environ. Manag. 2020, 270, 110868. [Google Scholar] [CrossRef] [PubMed]
- Koupaie, E.H.; Dahadha, S.; Lakeh, A.A.B.; Azizi, A.; Elbeshbishy, E. Enzymatic pretreatment of lignocellulosic biomass for enhanced biomethane production—A review. J. Environ. Manag. 2019, 233, 774–784. [Google Scholar] [CrossRef]
- Kang, C.; Huang, Y.; Yang, H.; Yan, X.F.; Chen, Z.P. A review of carbon dots produced from biomass wastes. Nanomaterials 2020, 10, 2316. [Google Scholar] [CrossRef]
- Liu, M.; Xu, Y.; Niu, F.; Gooding, J.J.; Liu, J. Photoluminescent lignin hybridized carbon quantum dots composites for bioimaging applications. Analyst 2016, 141, 2657–2664. [Google Scholar] [CrossRef]
- Sun, Y.-P.; Zhou, B.; Lin, Y.; Wang, W.; Fernando, K.S.; Pathak, P.; Meziani, M.J.; Harruff, B.A.; Wang, X.; Wang, H. Quantum-Sized Carbon Dots for Bright and Colorful Photoluminescence. J. Am. Chem. Soc. 2006, 128, 7756–7757. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Ray, R.; Gu, Y.; Ploehn, H.J.; Gearheart, L.; Raker, K.; Scrivens, W.A. Electrophoretic analysis and purification of fluorescent single-walled carbon nanotube fragments. J. Am. Chem. Soc. 2004, 126, 12736–12737. [Google Scholar] [CrossRef] [PubMed]
- Sciortino, A.; Cayuela, A.; Soriano, M.L.; Gelardi, F.M.; Cannas, M.; Valcárcel, M.; Messina, F. Different natures of surface electronic transitions of carbon nanoparticles. Phys. Chem. Chem. Phys. 2017, 19, 22670–22677. [Google Scholar] [CrossRef]
- Wang, X.; Qu, K.; Xu, B.; Ren, J.; Qu, X. Microwave assisted one-step green synthesis of cell-permeable multicolor photoluminescent carbon dots without surface passivation reagents. J. Mater. Chem. 2011, 21, 2445–2450. [Google Scholar] [CrossRef]
- Zhuo, S.; Shao, M.; Lee, S.T. Upconversion and downconversion fluorescent graphene quantum dots: Ultrasonic preparation and photocatalysis. ACS Nano 2012, 6, 1059. [Google Scholar] [CrossRef]
- Xu, Q.; Liu, Y.; Gao, C.; Wei, J.; Zhou, H.; Chen, Y.; Dong, C.; Sreeprasad, T.S.; Li, N.; Xia, Z. Synthesis, mechanistic investigation, and application of photoluminescent sulfur and nitrogen co-doped carbon dots. J. Mater. Chem. C 2015, 3, 9885–9893. [Google Scholar] [CrossRef]
- Cao, L.; Wang, X.; Meziani, M.J.; Lu, F.; Wang, H.; Luo, P.G.; Lin, Y.; Harruff, B.A.; Veca, L.M.; Murray, D.; et al. Carbon Dots for Multiphoton Bioimaging. J. Am. Chem. Soc. 2007, 129, 11318–11319. [Google Scholar] [CrossRef] [PubMed]
- Das, R.; Bandyopadhyay, R.; Pramanik, P. Carbon quantum dots from natural resource: A review. Mater. Today Chem. 2018, 8, 96–109. [Google Scholar] [CrossRef]
- Anuar, N.K.K.; Tan, H.L.; Lim, Y.P.; So’Aib, M.S.; Abu Bakar, N.F. A Review on Multifunctional Carbon-Dots Synthesized From Biomass Waste: Design/ Fabrication, Characterization and Applications. Front. Energy Res. 2021, 9, 626549. [Google Scholar] [CrossRef]
- Sima, M.; Vrbova, K.; Zavodna, T.; Honkova, K.; Chvojkova, I.; Ambroz, A.; Klema, J.; Rossnerova, A.; Polakova, K.; Malina, T.; et al. The Differential Effect of Carbon Dots on Gene Expression and DNA Methylation of Human Embryonic Lung Fibroblasts as a Function of Surface Charge and Dose. Int. J. Mol. Sci. 2020, 21, 4763. [Google Scholar] [CrossRef] [PubMed]
- Hawrylak, P.; Peeters, F.; Ensslin, K. Carbononics—Integrating electronics, photonics and spintronics with graphene quantum dots. Phys. Status Solidi RRL Rapid Res. Lett. 2016, 10, 11–12. [Google Scholar] [CrossRef]
- Hai, X.; Feng, J.; Chen, X.; Wang, J. Tuning the optical properties of graphene quantum dots for biosensing and bioimaging. J. Mater. Chem. B 2018, 6, 3219–3234. [Google Scholar] [CrossRef]
- Qian, Z.; Shan, X.; Chai, L.; Ma, J.; Chen, J.; Feng, H. Sidoped carbon quantum dots: A facile and general preparation strategy, bioimaging application, and multifunctional sensor. ACS Appl. Mater. Interfaces 2014, 6, 6797–6805. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Jia, X.-H.; Yin, X.-B.; He, X.-W.; Zhang, Y.-K. Carbon quantum dot stabilized gadolinium nanoprobe prepared via a one-pot hydrothermal approach for magnetic resonance and fluorescence dual-modality bioimaging. Anal. Chem. 2014, 86, 12122–12129. [Google Scholar] [CrossRef]
- Zuo, J.; Jiang, T.; Zhao, X.; Xiong, X.; Xiao, S.; Zhu, Z. Preparation and Application of Fluorescent Carbon Dots. J. Nanomater. 2015, 2015, 787862. [Google Scholar] [CrossRef]
- Yoo, D.; Park, Y.; Cheon, B.; Park, M.-H. Carbon dots as an effective fluorescent sensing platform for metal ion detection. Nanoscale Res. Lett. 2019, 14, 272. [Google Scholar] [CrossRef] [PubMed]
- Freire, R.; Le, N.D.; Jiang, Z.; Kim, C.S.; Rotello, V.M.; Fechine, P. NH2-rich carbon quantum dots: A protein-responsive probe for detection and identification. Sens. Actuators B Chem. 2018, 255, 2725–2732. [Google Scholar] [CrossRef]
- Sarkar, T.; Bohidar, H.; Solanki, P.R. Carbon dots-modified chitosan based electrochemical biosensing platform for detection of vitamin D. Int. J. Biol. Macromol. 2018, 109, 687–697. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Shi, R.; Gao, M.; Zhang, K.; Deng, L.; Fu, Q.; Wang, L.; Gao, D. Sensitivity fluorescent switching sensor for Cr (VI) and ascorbic acid detection based on orange peels-derived carbon dots modified with EDTA. Food Chem. 2020, 318, 126506. [Google Scholar] [CrossRef] [PubMed]
- Cai, L.; Fu, Z.; Cui, F. Synthesis of carbon dots and their application as Turn Off–On fluorescent sensor for mercury (II) and glutathione. J. Fluoresc. 2020, 30, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Ren, J.; Malfatti, L.; Innocenzi, P. Citric acid derived carbon dots, the challenge of understanding the synthesis-structure relationship. Carbon 2021, 7, 2. [Google Scholar] [CrossRef]
- De Medeiros, T.V.; Manioudakis, J.; Noun, F.; Macairan, J.-R.; Victoria, F.; Naccache, R. Microwave-assisted synthesis of carbon dots and their applications. J. Mater. Chem. C 2019, 7, 7175–7195. [Google Scholar] [CrossRef]
- Meng, W.; Bai, X.; Wang, B.; Liu, Z.; Lu, S.; Yang, B. Biomass-Derived Carbon Dots and Their Applications. Energy Environ. Mater. 2019, 2, 172–192. [Google Scholar] [CrossRef]
- Li, Y.; Hu, Y.; Zhao, Y.; Shi, G.; Deng, L.; Hou, Y.; Qu, L. An electrochemical avenue to green-luminescent graphene quantum dots as potential electron-acceptors for photovoltaics. Adv. Mater. 2011, 23, 776–780. [Google Scholar] [CrossRef]
- Shenderova, O.; Hens, S.; Vlasov, I.; Turner, S.; Lu, Y.-G.; Van Tendeloo, G.; Schrand, A.; Burikov, S.A.; Dolenko, T.A. Carbon-Dot-Decorated Nanodiamonds. Part. Part. Syst. Charact. 2014, 31, 580–590. [Google Scholar] [CrossRef]
- Dimos, K. Carbon Quantum Dots: Surface passivation and functionalization. Curr. Org. Chem. 2016, 20, 682–695. [Google Scholar] [CrossRef]
- Li, L.; Dong, T. Correction: Photoluminescence tuning in carbon dots: Surface passivation or/and functionalization, heteroatom doping. J. Mater. Chem. C 2018, 6, 7944–7970. [Google Scholar] [CrossRef]
- Zhang, M.; Su, R.; Zhong, J.; Fei, L.; Cai, W.; Guan, Q.; Li, W.; Li, N.; Chen, Y.; Cai, L.; et al. Red/orange dual-emissive carbon dots for pH sensing and cell imaging. Nano Res. 2019, 12, 815–821. [Google Scholar] [CrossRef]
- Konar, S.; Kumar, B.P.; Mahto, M.K.; Samanta, D.; Shaik, A.S.; Shaw, M.; Mandal, M.; Pathak, A. N-doped carbon dot as fluorescent probe for detection of cysteamine and multicolor cell imaging. Sens. Actuators B Chem. 2019, 286, 77–85. [Google Scholar] [CrossRef]
- Song, P.; Zhang, L.; Long, H.; Meng, M.; Liu, T.; Yin, Y.; Xi, R. A multianalyte fluorescent carbon dots sensing system constructed based on specific recognition of Fe(iii) ions. RSC Adv. 2017, 7, 28637–28646. [Google Scholar] [CrossRef]
- Feng, J.; Wang, W.-J.; Hai, X.; Yu, Y.-L.; Wang, J.-H. Green preparation of nitrogen-doped carbon dots derived from silkworm chrysalis for cell imaging. J. Mater. Chem. B 2016, 4, 387–393. [Google Scholar] [CrossRef] [PubMed]
- Li, H.T.; Kang, Z.H.; Liu, Y.; Lee, S.T. Carbon Nanodots: Synthesis, Properties and Applications. J. Mater. Chem. 2012, 22, 24230–24253. [Google Scholar] [CrossRef]
- Zhang, R.; Liu, Y.; Yu, L.; Li, Z.; Sun, S. Preparation of high-quality biocompatible carbon dots by extraction, with new thoughts on the luminescence mechanisms. Nanotechnology 2013, 24, 225601. [Google Scholar] [CrossRef]
- Gan, Z.X.; Wu, X.L.; Zhou, G.X.; Shen, J.C.; Chu, P.K. Is there real upconversion photoluminescence from grapheme quantum dots? Adv. Opt. Mater. 2013, 1, 554–558. [Google Scholar] [CrossRef]
- Yuan, Y.H.; Liu, Z.X.; Li, R.S.; Zou, H.Y.; Lin, M.; Liu, H.; Huang, C.Z. Synthesis of nitrogen-doping carbon dots with different photoluminescence properties by controlling the surface states. Nanoscale 2016, 8, 6770–6776. [Google Scholar] [CrossRef]
- Cheng, C.; Shi, Y.; Li, M.; Xing, M.; Wu, Q. Carbon quantum dots from carbonized walnut shells: Structural evolution, fluorescence characteristics, and intracellular bioimaging. Mater. Sci. Eng. C 2017, 79, 473–480. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, C.V.; Corrêa, J.R.; Aiube, C.M.; Andrade, L.P.; Galvão, P.M.; Costa, P.A.; Campos, A.L.; Pereira, A.J.; Ghesti, G.F.; Felix, J.F.; et al. Down- and Up-Conversion Photoluminescence of Carbon-Dots from Brewing Industry Waste: Application in Live Cell-Imaging Experiments. J. Braz. Chem. Soc. 2015, 26, 2623–2628. [Google Scholar] [CrossRef]
- Wen, X.; Yu, P.; Toh, Y.-R.; Ma, X.; Tang, J. On the upconversion fluorescence in carbon nanodots and graphene quantum dots. Chem. Commun. 2014, 50, 4703–4706. [Google Scholar] [CrossRef] [PubMed]
- Nair, A.; Haponiuk, J.T.; Thomas, S.; Gopi, S. Natural carbon-based quantum dots and their applications in drug delivery: A review. Biomed. Pharmacother. 2020, 132, 110834. [Google Scholar] [CrossRef] [PubMed]
- Sun, D.; Ban, R.; Zhang, P.-H.; Wu, G.-H.; Zhang, J.-R.; Zhu, J.-J. Hair fiber as a precursor for synthesizing of sulfur- and nitrogen-co-doped carbon dots with tunable luminescence properties. Carbon 2013, 64, 424–434. [Google Scholar] [CrossRef]
- Wu, Z.L.; Liu, Z.X.; Yuan, Y.H. Carbon dots: Materials, synthesis, properties and approaches to long-wavelength and multicolor emission. J. Mater. Chem. B 2015, 5, 3794–3809. [Google Scholar] [CrossRef]
- Gao, H.; Xia, S.; Zhang, R.; Zhao, Y.; Wang, W.; An, Z.; Qi, H. Efficient green electrogenerated chemiluminescence from cyclometalated iridium(III) complex. J. Electroanal. Chem. 2015, 755, 71–76. [Google Scholar] [CrossRef]
- He, Y.; Hu, X.; Gong, Z.; Chen, S.; Yuan, R. A novel electrochemiluminescence biosensor based on the self-ECL emission of conjugated polymer dots for lead ion detection. Mikrochim. Acta 2020, 187, 237. [Google Scholar] [CrossRef]
- Swanick, K.N.; Ladouceur, S.; Colman, E.Z.; Ding, Z.F. Panchromic cationic iridium (III) complexes. Angew. Chem. Int. Ed. 2012, 51, 11079–11082. [Google Scholar] [CrossRef]
- Hou, J.; Wang, L.; Zhang, P.; Xu, Y.; Ding, L. Facile synthesis of carbon dots in an immiscible system with excitation-independent emission and thermally activated delayed fluorescence. Chem. Commun. 2015, 51, 17768–17771. [Google Scholar] [CrossRef]
- Islam, A.; Ahmad, H.; Zaidi, N.; Kumar, S. Graphene oxide sheets immobilized polystyrene for column preconcentration and sensitive determination of lead by flame atomic absorption spectrometry. ACS Appl. Mater. Interfaces 2014, 6, 13257–13265. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Y.; Rowe, A.A.A.; Plaxco, K.W. Electrochemical detection of parts-per-billion lead via an electrode-bound DNAzyme assembly. J. Am. Chem. Soc. 2007, 129, 262–263. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Dong, S.; Wang, E. A Lead(II)-driven DNA molecular device for turn-on fluorescence detection of lead(II) ion with high selectivity and sensitivity. J. Am. Chem. Soc. 2010, 132, 13156–13157. [Google Scholar] [CrossRef] [PubMed]
- Vercauteren, J.; Pérès, C.; Devos, C.; Sandra, P.; Vanhaecke, F.; Moens, L. Stir Bar Sorptive Extraction for the Determination of ppq-Level Traces of Organotin Compounds in Environmental Samples with Thermal Desorption-Capillary Gas Chromatography−ICP Mass Spectrometry. Anal. Chem. 2001, 73, 1509–1514. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.J.; Xue, Z.J.; Luo, D.; Yu, W.; Guo, Z.H.; Wang, T. Green synthesis of luminescent nitrogen-doped carbon dots from milk and its imaging application. Anal. Chem. 2014, 86, 5620. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.W.; Ge, L.; Zhang, Y.; Song, X.R.; Li, N.Q.; Ge, S.G.; Yu, J.H. Battery-triggered microfluidic paper-based multiplex electrochemiluminescence immune device based on potential-resolution strategy. Lab Chip 2012, 12, 4489–4498. [Google Scholar] [CrossRef]
- Li, L.-L.; Ji, J.; Fei, R.; Wang, C.-Z.; Lu, Q.; Zhang, J.-R.; Jiang, L.-P.; Zhu, J.-J. A facile microwave avenue to electrochemiluminescent two-color graphene quantum dots. Adv. Funct. Mater. 2012, 22, 2971–2979. [Google Scholar] [CrossRef]
- Wang, Z.-X.; Zheng, C.-L.; Li, Q.-L.; Ding, S.-N. Electrochemiluminescence of a nanoAg–carbon nanodot composite and its application to detect sulfide ions. Analyst 2014, 139, 1751–1755. [Google Scholar] [CrossRef]
- Bozetine, H.; Wang, Q.; Barras, A.; Li, M.; Hadjersi, T.; Szunerits, S.; Boukherroub, R. Green chemistry approach for the synthesis of ZnO-carbon dots nanocomposites with good photocatalytic properties under visible light. J. Colloid Interface Sci. 2016, 465, 286–294. [Google Scholar] [CrossRef]
- Iqbal, A.; Tian, Y.; Wang, X.; Gong, D.; Guo, Y.; Iqbal, K.; Wang, Z.; Liu, W.; Qin, W. Carbon dots prepared by solid state method via citric acid and 1,10- phenanthroline for selective and sensing detection of Fe2þ and Fe3þ. Sens. Actuators B Chem. 2016, 237, 408–415. [Google Scholar] [CrossRef]
- Edison, T.N.J.I.; Atchudan, R.; Sethuraman, M.G.; Shim, J.-J.; Lee, Y.R. Microwave assisted green synthesis of fluorescent N-doped carbon dots: Cytotoxicity and bio-imaging applications. J. Photochem. Photobiol. B Biol. 2016, 161, 154–161. [Google Scholar] [CrossRef] [PubMed]
- Jhonsi, M.A.; Ananth, D.A.; Nambirajan, G.; Sivasudha, T.; Yamini, R.; Bera, S.; Kathiravan, A. Antimicrobial activity, cytotoxicity and DNA binding studies of carbon dots. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2018, 196, 295–302. [Google Scholar] [CrossRef] [PubMed]
- Arul, V.; Sethuraman, M.G. Facile green synthesis of fluorescent N-doped carbon dots from Actinidia deliciosa and their catalytic activity and cytotoxicity applications. Opt. Mater. 2018, 78, 181–190. [Google Scholar] [CrossRef]
- Park, S.Y.; Lee, H.U.; Park, E.S.; Lee, S.C.; Lee, J.W.; Jeong, S.W.; Kim, C.H.; Lee, Y.C.; Huh, Y.S.; Lee, J. Photoluminescent green carbon nanodots from food-waste derived sources: Large-scale synthesis, properties, and biomedical applications. ACS Appl. Mater. Interfaces 2014, 6, 3365–3370. [Google Scholar] [CrossRef] [PubMed]
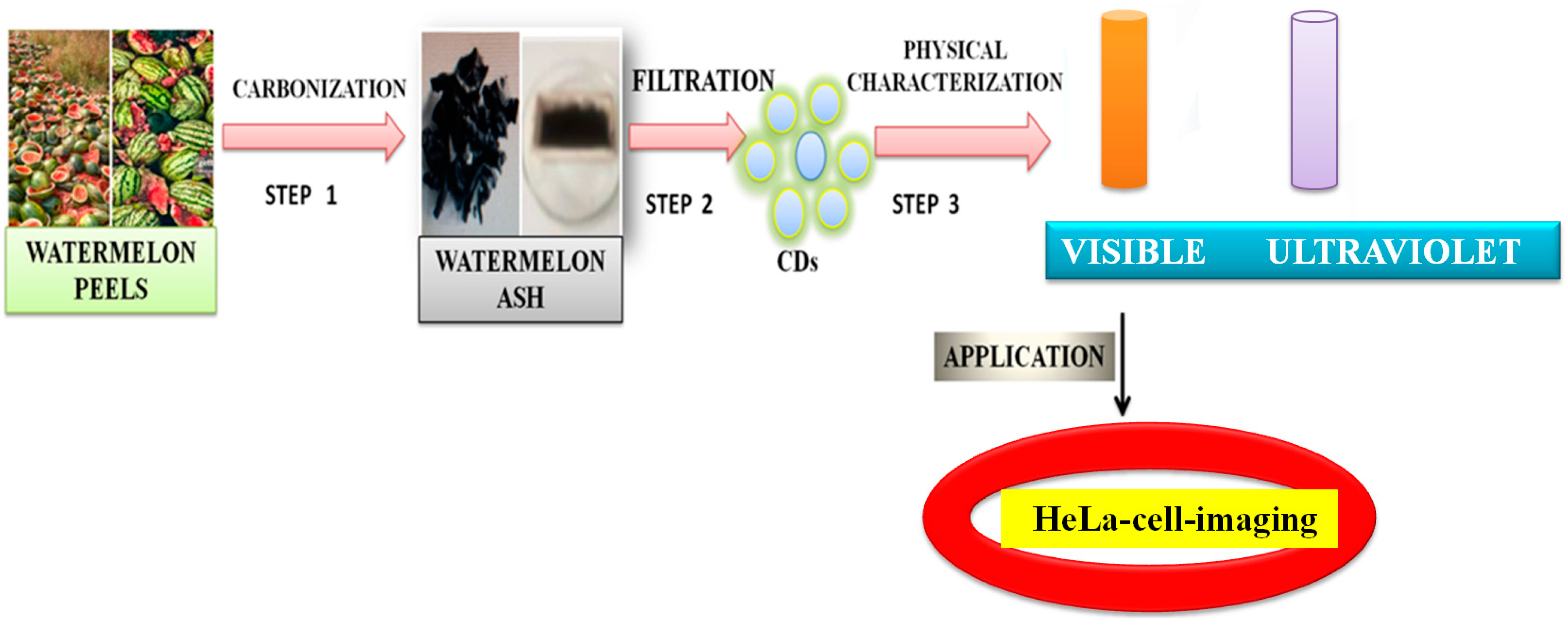
- Zhou, J.; Sheng, Z.; Han, H.; Zou, M.; Li, C. Facile synthesis of fluorescent carbon dots using watermelon peel as a carbon source. Mater. Lett. 2012, 66, 222–224. [Google Scholar] [CrossRef]
- Xue, M.; Zou, M.; Zhao, J.; Zhan, Z.; Zhao, S. Green preparation of fluorescent carbon dots from lychee seeds and their application for the selective detection of methylene blue and imaging in living cells. J. Mater. Chem. B 2015, 3, 6783–6789. [Google Scholar] [CrossRef] [PubMed]
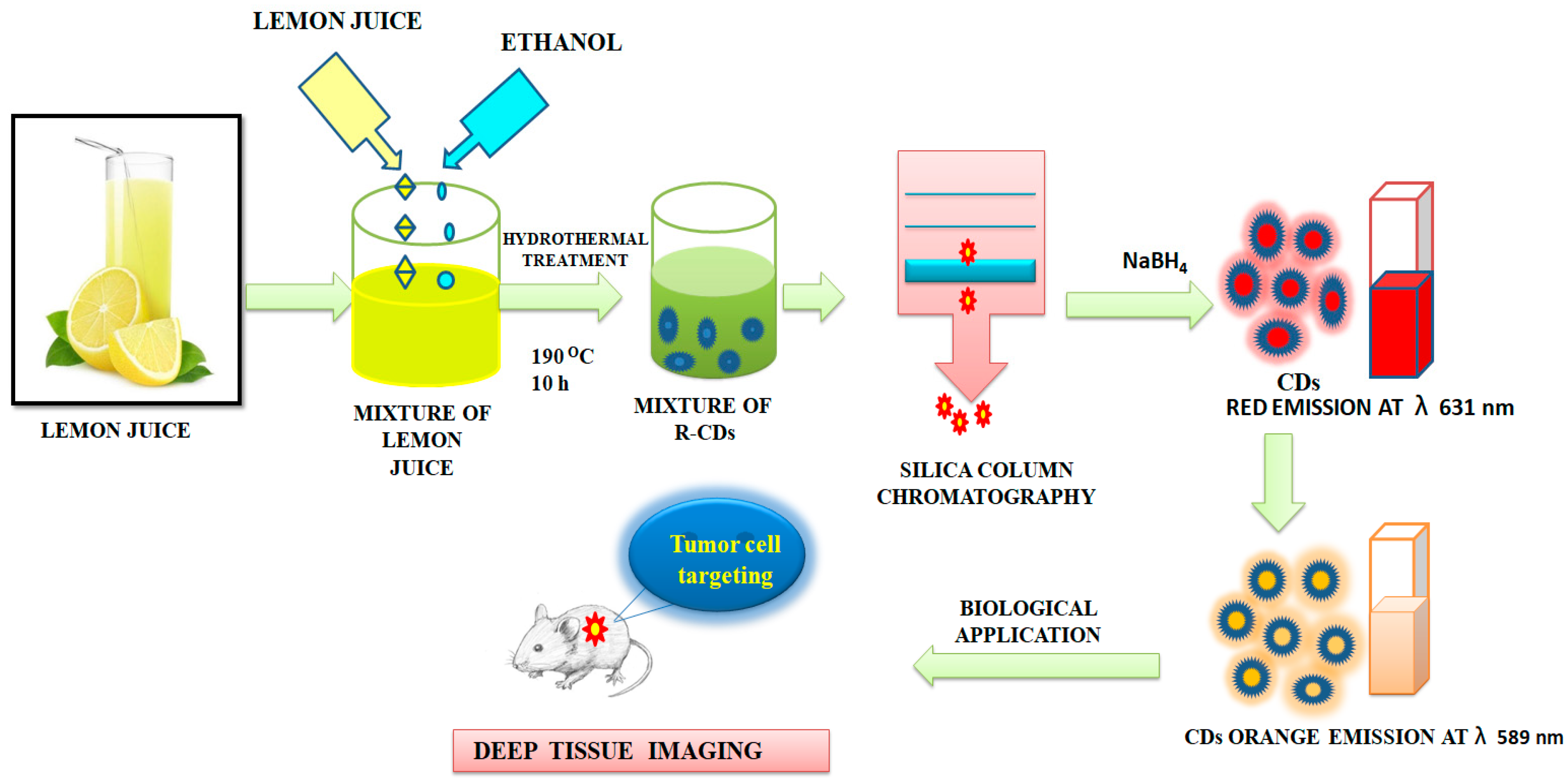
- Ding, H.; Ji, Y.; Wei, J.-S.; Gao, Q.-Y.; Zhou, Z.-Y.; Xiong, H.-M. Facile synthesis of red-emitting carbon dots from pulp-free lemon juice for bioimaging. J. Mater. Chem. B 2017, 5, 5272–5277. [Google Scholar] [CrossRef]
- Oh, E.; Liu, R.; Nel, A.; Gemill, K.B.; Bihal, M.; Cohen, Y.; Medintz, I.L. Meta-analysis of cellular toxicity for cadmium-containing quantum dots. Nat. Nanotechnol. 2016, 11, 479–486. [Google Scholar] [CrossRef]
- Bilal, M.; Oh, E.; Liu, R.; Breger, J.C.; Medintz, I.L.; Cohen, Y. Bayesian Network Resource for Meta-Analysis: Cellular Toxicity of Quantum Dots. Small 2019, 15, e1900510. [Google Scholar] [CrossRef]
- O’Hara, T.; Seddon, B.; O’Connor, A.; McClean, S.; Singh, B.; Iwuoha, E.; Fuku, X.; Dempsey, E. Quantum dot nanotoxicity investigations using juman lung cells and TOXOR electrochemical enzyme assay methodology. ACS Sens. 2017, 2, 165–171. [Google Scholar] [CrossRef]
- Allocca, M.; Mattera, L.; Bauduin, A.; Miedziak, B.; Moros, M.; De Trizio, L.; Tino, A.; Reiss, P.; Ambrosone, A.; Tortiglione, C. An integrated multilevel analysis profiling biosafety and toxicity onduced by indium- and cadmium-based quantum dots in vivo. Environ. Sci. Technol. 2019, 53, 3938–3947. [Google Scholar] [CrossRef] [PubMed]
- Yaghini, E.; Turner, H.; Pilling, A.; Naasani, I.; MacRobert, A.J. In vivo biodistribution and toxicology studies of cadmium-free indium-based quantum dot nanoparticles in a rat model. Nanomed. Nanotechnol. Biol. Med. 2018, 14, 2644–2655. [Google Scholar] [CrossRef] [PubMed]
- Tarantini, A.; Wegner, K.D.; Dussert, F.; Sarret, G.; Beal, D.; Mattera, L.; Lincheneau, C.; Proux, O.; Truffier-Boutry, D.; Moriscot, C.; et al. Physicochemical alterations and toxicity of InP alloyed quantum dots aged in environmental conditions: A safer by design evaluation. NanoImpact 2019, 14, 100168. [Google Scholar] [CrossRef]
- Bandi, R.; Gangapurama, B.R.; Dadigalaa, R.; Eslavathb, R.; Singhb, S.S.; Guttena, V. Facile and green synthesis of fluorescent carbon dots from Onion waste and their potential applications as sensor and multicolour imaging agents. RSC Adv. 2016, 6, 28633–28639. [Google Scholar] [CrossRef]
- Yang, J.-M.; Hu, X.-W.; Liu, Y.-X.; Zhang, W. Fabrication of a carbon quantum dots-immobilized zirconium-based metal-organic framework composite fluorescence sensor for highly sensitive detection of 4-nitrophenol. Microporous Mesoporous Mater. 2019, 274, 149–154. [Google Scholar] [CrossRef]
- Ren, X.; Wei, J.; Ren, J.; Qiang, L.; Tang, F.; Meng, X. A sensitive biosensor for the fluorescence detection of the acetylcholinesterase reaction system based on carbon dots. Colloids Surf. B Biointerfaces 2014, 125, 90–95. [Google Scholar] [CrossRef]
- Jiang, G.; Jiang, T.; Zhou, H.; Yao, J.; Kong, X. Preparation of N-doped carbon quantum dots for highly sensitive detection of dopamine by an electrochemical method. RSC Adv. 2015, 5, 9064–9068. [Google Scholar] [CrossRef]
- Jiang, Y.; Wang, B.; Meng, F.; Cheng, Y.; Zhu, C. Microwave-assisted preparation of N-doped carbon dots as a biosensor for electrochemical dopamine detection. J. Colloid Interface Sci. 2015, 452, 199–202. [Google Scholar] [CrossRef]
- Li, G.; Fu, H.; Chen, X.; Gong, P.; Chen, G.; Xia, L.; Wang, H.; You, J.; Wu, Y. Facile and Sensitive Fluorescence Sensing of Alkaline Phosphatase Activity with Photoluminescent Carbon Dots Based on Inner Filter Effect. Anal. Chem. 2016, 88, 2720–2726. [Google Scholar] [CrossRef]
- Wang, H.; Lu, Q.; Hou, Y.; Liu, Y.; Zhang, Y. High fluorescence S, N co-doped carbon dots as an ultra-sensitive fluorescent probe for the determination of uric acid. Talanta 2016, 155, 62–69. [Google Scholar] [CrossRef]
- Liu, H.; Ding, J.; Zhang, K.; Ding, L. Construction of biomass carbon dots based fluorescence sensors and their applications in chemical and biological analysis. TrAC Trends Anal. Chem. 2019, 118, 315–337. [Google Scholar] [CrossRef]
- Edison, T.N.J.I.; Atchudan, R.; Shim, J.-J.; Kalimuthu, S.; Ahn, B.-C.; Lee, Y.R. Turn-off fluorescence sensor for the detection of ferric ion in water using green synthesized N-doped carbon dots and its bio-imaging. J. Photochem. Photobiol. B Biol. 2016, 158, 235–242. [Google Scholar] [CrossRef] [PubMed]
- Yi, Z.; Li, X.; Zhang, H.; Ji, X.; Sun, W.; Yu, Y.; Liu, Y.; Huang, J.; Sarshar, Z.; Sain, M. High quantum yield photoluminescent N-doped carbon dots for switch sensing and imaging. Talanta 2021, 222, 121663. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Luo, S.; Wu, P.; Ma, C.; Wu, X.; Xu, M.; Li, W.; Liu, S. Hydrothermal synthesis of green fluorescent nitrogen doped carbon dots for the detection of nitrite and multicolor cellular imaging. Anal. Chim. Acta 2019, 1090, 133–142. [Google Scholar] [CrossRef]
- Liu, Y.; Liu, Y.; Park, S.-J.; Zhang, Y.; Kim, T.; Chae, S.; Park, M.; Kim, H.-Y. One-step synthesis of robust nitrogen-doped carbon dots: Acid-evoked fluorescence enhancement and their application in Fe3+ detection. J. Mater. Chem. A 2015, 3, 17747–17754. [Google Scholar] [CrossRef]
- Chena, Y.; Wua, Y.; Wenga, B.; Wangab, B.; Lia, C. Facile synthesis of nitrogen and sulfur co-doped carbon dots and application for Fe(III) ions detection and cCell imaging. Sens. Actuators B Chem. 2016, 223, 689–696. [Google Scholar] [CrossRef]
- Yuan, H.; Yub, J.; Fenga, S.; Gonga, Y. Highly photoluminescent pH-independent nitrogen-doped carbon dots for sensitive and selective sensing of p-nitrophenol. RSC Adv. 2016, 6, 15192–15200. [Google Scholar] [CrossRef]
- Shamsipur, M.; Molaei, K.; Molaabasi, F.; Hosseinkhani, S.; Alizadeh, N.; Alipour, M.; Moassess, S. One-step synthesis and characterization of highly luminescent nitrogen and phosphorus co-doped carbon dots and their application as highly selective and sensitive nanoprobes for low level detection of uranyl ion in hair and water samples and application to cellular imaging. Sens. Actuators B Chem. 2018, 257, 772–782. [Google Scholar] [CrossRef]
- Lu, M.; Zhou, L. One-step sonochemical synthesis of versatile nitrogen-doped carbon quantum dots for sensitive detection of Fe2+ ions and temperature in vitro. Mater. Sci. Eng. C 2019, 101, 352–359. [Google Scholar] [CrossRef]
- Xie, Y.; Cheng, D.; Liu, X.; Han, A. Green Hydrothermal Synthesis of N-doped Carbon Dots from Biomass Highland Barley for the Detection of Hg2+. Sensors 2019, 19, 3169. [Google Scholar] [CrossRef]
- Latha, M.; Aruna-Devi, R.; Bogireddy, N.K.R.; Rios, S.E.S.; Mochan, W.L.; Castrellon-Uribe, J.; Agarwal, V. N-doped oxidized carbon dots for methanol sensing in alcoholic beverages. RSC Adv. 2020, 10, 22522–22532. [Google Scholar] [CrossRef] [PubMed]
- Zhai, Z.; Xu, J.; Gong, T.; Cao, B.; Cui, K.; Hou, L.; Yuan, C. Sustainable fabrication of N-doped carbon quantum dots and their application in fluorescent ink, Fe(III) detection and fluorescent films. Inorg. Chem. Commun. 2022, 140, 109387. [Google Scholar] [CrossRef]
- Gu, L.; Zhang, J.; Yang, G.; Tang, Y.; Zhang, X.; Huang, X.; Zhai, W.; Fodjo, E.K.; Kong, C. Green preparation of carbon quantum dots with wolfberry as on-off-on nanosensors for the detection of Fe3+ and l-ascorbic acid. Food Chem. 2022, 376, 131898. [Google Scholar] [CrossRef] [PubMed]
- John, B.K.; John, N.; Korah, B.K.; Thara, C.; Abraham, T.; Mathew, B. Nitrogen-doped carbon quantum dots as a highly selective fluorescent and electrochemical sensor for tetracycline. J. Photochem. Photobiol. 2022, 432, 114060. [Google Scholar] [CrossRef]
- Rong, M.; Feng, Y.; Wang, Y.; Chen, X. One-pot solid phase pyrolysis synthesis of nitrogen-doped carbon dots for Fe3+ sensing and bioimaging. Sens. Actuators B Chem. 2017, 245, 868–874. [Google Scholar] [CrossRef]
- Chai, C.; Qiao, X.; Zheng, L.; Duan, H.; Bian, W.; Choi, M.M.F. Nitrogen-doped carbon dots a fluorescent probe for the detection of p-hydroxybenzaldehyde and cell imaging, Fullerenes. Fuller. Nanotub. Carbon Nanostruct. 2022, 30, 535–542. [Google Scholar] [CrossRef]
- Jia, J.; Lin, B.; Gao, Y.; Jiao, Y.; Li, L.; Dong, C.; Shuang, S. Highly luminescent N-doped carbon dots from black soya beans for free radical scavenging, Fe3+ sensing and cellular imaging. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2019, 211, 363–372. [Google Scholar] [CrossRef]
- Li, J.; Zuo, G.; Pan, X.; Wei, W.; Qi, X.; Su, T.; Dong, W. Nitrogen-doped carbon dots as a fluorescent probe for the highly sensitive detection of Ag+ and cell imaging. Luminescence 2018, 33, 243–248. [Google Scholar] [CrossRef]
- Dang, V.D.; Ganganboina, A.B.; Doong, R.-A. Bipyridine- and Copper-Functionalized N-doped Carbon Dots for Fluorescence Turn Off–On Detection of Ciprofloxacin. ACS Appl. Mater. Interfaces 2020, 12, 32247–32258. [Google Scholar] [CrossRef]
- Rao, L.; Tang, Y.; Lu, H.; Yu, S.; Ding, X.; Xu, K.; Li, Z.; Zhang, J.Z. Highly Photoluminescent and Stable N-Doped Carbon Dots as Nanoprobes for Hg2+ Detection. Nanomaterials 2018, 8, 900. [Google Scholar] [CrossRef]
- Fu, L.; Wang, A.; Lai, G.; Lin, C.-T.; Yu, J.; Yu, A.; Liu, Z.; Xie, K.; Su, W. A glassy carbon electrode modified with N-doped carbon dots for improved detection of hydrogen peroxide and paracetamol. Mikrochim. Acta 2018, 185, 87. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Fan, H.; Huang, L.; Xia, J.; Huang, J.; Li, M.; Ding, H.; Huang, K.; Li, S. Eosinophilic nitrogen-doped carbon dots derived from tribute chrysanthemum for label-free detection of Fe3+ ions and hydrazine. J. Taiwan Inst. Chem. Eng. 2017, 78, 247–253. [Google Scholar] [CrossRef]
- Sun, Z.; Chen, Z.; Luo, J.; Zhu, Z.; Zhang, X.; Liu, R.; Wu, Z.-C. A yellow-emitting nitrogen-doped carbon dots for sensing of vitamin B12 and their cell-imaging. Dye. Pigment. 2020, 176, 108227. [Google Scholar] [CrossRef]
- Yu, Y.; Li, C.; Chen, C.; Huang, H.; Liang, C.; Lou, Y.; Chen, X.-B.; Shi, Z.; Feng, S. Saccharomyces-derived carbon dots for biosensing pH and vitamin B 12. Talanta 2019, 195, 117–126. [Google Scholar] [CrossRef] [PubMed]
- Simões, E.; Leitão, J.M.; da Silva, J.E. Sulfur and nitrogen co-doped carbon dots sensors for nitric oxide fluorescence quantification. Anal. Chim. Acta 2017, 960, 117–122. [Google Scholar] [CrossRef] [PubMed]
- Tammina, S.K.; Yang, D.; Koppala, S.; Cheng, C.; Yang, Y. Highly photoluminescent N, P doped carbon quantum dots as a fluorescent sensor for the detection of dopamine and temperature. J. Photochem. Photobiol. B Biol. 2019, 194, 61–70. [Google Scholar] [CrossRef]
- Yola, M.L.; Atar, N. Development of molecular imprinted sensor including graphitic carbon nitride/N-doped carbon dots composite for novel recognition of epinephrine. Compos. Part B Eng. 2019, 175, 107113. [Google Scholar] [CrossRef]
- Barua, S.; Raul, P.K.; Gopalakrishnan, R.; Das, B.; Vanlalhmuaka; Veer, V. Zero-Dimensional and Highly Oxygenated Graphene Oxide for Multifunctional Poly(lactic acid) Bionanocomposites. ACS Sustain. Chem. Eng. 2016, 4, 2345–2350. [Google Scholar] [CrossRef]
- Mirtchev, P.; Henderson, E.J.; Soheilnia, N.; Yipc, C.M.; Ozin, G.A. Carbon nanodots: Synthesis, properties and applications. J. Mater. Chem. 2012, 22, 1265. [Google Scholar] [CrossRef]
- Wang, H.; Dai, H. Strongly coupled inorganic–nano-carbon hybrid materials for energy storage. Chem. Soc. Rev. 2013, 42, 3088–3113. [Google Scholar] [CrossRef]
- Zhen, S.J.; Fu, W.L.; Chen, B.B.; Zhan, L.; Zou, H.Y.; Gao, M.X.; Huang, C.Z. Facile exfoliation of MoS2 nanosheets by protein as a photothermal-triggered drug delivery system for synergistic tumor therapy. RSC Adv. 2016, 6, 93645–93648. [Google Scholar] [CrossRef]
- Rai, M.; Ingle, A. Role of nanotechnology in agriculture with special reference to management of insect pests. Appl. Microbiol. Biotechnol. 2012, 94, 287–293. [Google Scholar] [CrossRef] [PubMed]
- Černík, M.; Padil, V.V.T. Green synthesis of copper oxide nanoparticles using gum karaya as a biotemplate and their antibacterial application. Int. J. Nanomed. 2013, 8, 889–898. [Google Scholar] [CrossRef]
- Gunalan, S.; Sivaraj, R.; Rajendran, V. Green synthesized ZnO nanoparticles against bacterial and fungal pathogens. Prog. Nat. Sci. 2012, 22, 693–700. [Google Scholar] [CrossRef]
- Stankic, S.; Suman, S.; Haque, F.; Vidic, J. Pure and multi metal oxide nanoparticles: Synthesis, antibacterial and cytotoxic properties. J. Nanobiotechnol. 2016, 14, 73. [Google Scholar] [CrossRef] [PubMed]
- Fierro, J.L.G. Metal Oxides: Chemistry and Applications; CRC Press: Boca Raton, FL, USA, 2005. [Google Scholar]
- Prasannan, A.; Imae, T. One-Pot Synthesis of Fluorescent Carbon Dots from Orange Waste Peels. Ind. Eng. Chem. Res. 2013, 52, 15673–15678. [Google Scholar] [CrossRef]
- Tyagi, A.; Tripathi, K.M.; Singh, N.; Choudhary, S.; Gupta, R.K. Green synthesis of carbon quantum dots from lemon peel waste: Applications in sensing and photocatalysis. RSC Adv. 2016, 6, 72423–72432. [Google Scholar] [CrossRef]
- Hammi, N.; Marcotte, N.; Marinova, M.; Draoui, K.; Royer, S.; El Kadib, A. Nanostructured metal oxide@carbon dots through sequential chitosan templating and carbonisation route. Carbohydr. Polym. Technol. Appl. 2021, 2, 100043. [Google Scholar] [CrossRef]
- Molaei, M.J. Principles, mechanisms, and application of carbon quantum dots in sensors: A review. Anal. Meth. 2020, 12, 1266–1287. [Google Scholar] [CrossRef]
- Tejwan, N.; Saini, A.K.; Sharma, A.; Singh, T.A.; Kumar, N.; Das, J. Metal-doped and hybrid carbon dots: A comprehensive review on their synthesis and biomedical applications. J. Control. Release 2021, 330, 132–150. [Google Scholar] [CrossRef]
- Sohal, N.; Maity, B.; Basu, S. Carbon Dot-MnO2 nanosphere composite sensors for selective detection of glutathione. ACS Appl. Nano Mater. 2020, 3, 5955–5964. [Google Scholar] [CrossRef]
- El-Shamy, A.G. New nano-composite based on carbon dots (CDs) decorated magnesium oxide (MgO) nano-particles (CDots@MgO) sensor for high H2S gas sensitivity performance. Sens. Actuators B Chem. 2020, 329, 129154. [Google Scholar] [CrossRef]
- Woo, J.; Song, Y.; Ahn, J.; Kim, H. Green one-pot preparation of carbon dots (CD)-embedded cellulose transparent film for Fe3+ indicator using ionic liquid. Cellulose 2020, 27, 4609–4621. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, W.; Yue, X.; Yang, Q.; Liu, F.; Wang, Y.; Zhang, D.; Li, Z.; Wang, J. One-pot synthesis of multifunctional magnetic ferrite-MoS2 carbon dots nanohybrid adsorbent for efficient Pb(II) removal. J. Mater. Chem. A 2016, 4, 3893. [Google Scholar] [CrossRef]
- Yu, L.; Yue, X.; Yang, R.; Jing, S.; Qu, L. A sensitive and low toxicity electrochemical sensor for 2,4-dichlorophenol based on the nanocomposite of carbon dots, hexadecyltrimethyl ammonium bromide and chitosan. Sens. Actuators B Chem. 2016, 224, 241–247. [Google Scholar] [CrossRef]
- Ma, Y.; Xu, G.; Wei, F.; Cen, Y.; Ma, Y.; Song, Y.; Xu, X.; Shi, M.; Muhammad, S.; Hu, Q. A dual-emissive fluorescent sensor fabricated by encapsulating quantum dots and carbon dots into metal–organic frameworks for the ratiometric detection of Cu2+ in tap water. J. Mater. Chem. C 2017, 5, 8566–8571. [Google Scholar] [CrossRef]
- Cheng, M.; Wu, Z.; Liu, G.; Zhao, L.; Gao, Y.; Li, S.; Zhang, B.; Yan, X.; Geyu, L. Carbon dots decorated hierarchical litchi-like In2O3 nanospheres for highly sensitive and selective NO2 detection. Sens. Actuators B Chem. 2020, 304, 127272. [Google Scholar] [CrossRef]
- Guo, H.; Wang, X.; Wu, N.; Xu, M.; Wang, M.; Zhang, L.; Yang, W. In-situ synthesis of carbon dots-embedded europium metal-organic frameworks for ratiometric fluorescence detection of Hg2+ in aqueous environment. Anal. Chim. Acta 2021, 1141, 13–20. [Google Scholar] [CrossRef]
- Lin, X.; Gao, G.; Zheng, L.; Chi, Y.; Chen, G. Encapsulation of Strongly Fluorescent Carbon Quantum Dots in Metal–Organic Frameworks for Enhancing Chemical Sensing. Anal. Chem. 2014, 86, 1223–1228. [Google Scholar] [CrossRef]
- Liu, P.; Liu, J.; Xu, Y. Ratiometric fluorescence determination of hydrogen peroxide using carbon dot-embedded Ag@EuWO4(OH) nanocomposites. Microchim. Acta 2020, 187, 369. [Google Scholar] [CrossRef]
- Mehta, A.; Mishra, A.; Kainth, S.; Basu, S. Carbon quantum dots/TiO2 nanocomposite for sensing of toxic metals and photodetoxification of dyes with kill waste by waste concept. Mater. Des. 2018, 155, 485–493. [Google Scholar] [CrossRef]
- Rao, H.; Liu, W.; Lu, Z.; Wang, Y.; Ge, H.; Zou, P.; Wang, X.; He, H.; Zeng, X.; Wang, Y. Silica-coated carbon dots conjugated to CdTe quantum dots: A ratiometric fluorescent probe for copper(II). Mikrochim. Acta 2016, 183, 581–588. [Google Scholar] [CrossRef]
- Qin, S.J.; Yan, B. Dual-emissive ratiometric fluorescent probe based on Eu3+/C-dots@MOF hybrids for the biomarker diaminotoluene sensing. Sens. Actuators B Chem. 2018, 272, 510–517. [Google Scholar] [CrossRef]
- He, C.; Peng, L.; Lv, L.; Cao, Y.; Tu, J.; Huang, W.; Zhang, K. In situ growth of carbon dots on TiO2 nanotube arrays for PEC enzyme biosensors with visible light response. RSC Adv. 2019, 9, 15084–15091. [Google Scholar] [CrossRef]
- Huang, Q.; Lin, X.; Zhu, J.-J.; Tong, Q.-X. Pd-Au@carbon dots nanocomposite: Facile synthesis and application as an ultrasensitive electrochemical biosensor for determination of colitoxin DNA in human serum. Biosens. Bioelectron. 2017, 94, 507–512. [Google Scholar] [CrossRef]
- Yang, L.; Huang, N.; Lu, Q.; Liu, M.; Li, H.; Zhang, Y.; Yao, S. A quadruplet electrochemical platform for ultrasensitive and simultaneous detection of ascorbic acid, dopamine, uric acid and acetaminophen based on a ferrocene derivative functional Au NPs/carbon dots nanocomposite and graphene. Anal. Chim. Acta 2016, 903, 69–80. [Google Scholar] [CrossRef]
- Perelshtein, I.; Perkas, N.; Rahimipour, S.; Gedanken, A. Bifunctional Carbon Dots—Magnetic and Fluorescent Hybrid Nanoparticles for Diagnostic Applications. Nanomaterials 2020, 10, 1384. [Google Scholar] [CrossRef]
- Atchudan, R.; Nesakumar, T.; Edison, J.I.; Perumal, S.; Karthik, N.; Karthikeyan, D.; Shanmugam, M.; Lee, Y.R. Concurrent synthesis of nitrogen-doped carbon dots for cell imaging and ZnO@nitrogen-doped carbon sheets for photocatalytic degradation of methylene blue. J. Photochem. Photobiol. A Chem. 2018, 350, 75–85. [Google Scholar] [CrossRef]
- Huang, Q.; Lin, X.; Lin, C.; Zhang, Y.; Hu, S.; Wei, C. A high performance electrochemical biosensor based on Cu2O–carbon dots for selective and sensitive determination of dopamine in human serum. RSC Adv. 2015, 5, 54102–54108. [Google Scholar] [CrossRef]
- Zhai, Y.; Shen, F.; Zhang, X.; Jing, P.; Li, D.; Yang, X.; Zhou, D.; Xu, X.; Qu, S. Synthesis of green emissive carbon dots@montmorillonite composites and their application for fabrication of light-emitting diodes and latent fingerprints markers. J. Colloid Interface Sci. 2019, 554, 344–352. [Google Scholar] [CrossRef]
- Wang, A.; Guan, C.; Shan, G.; Chen, Y.; Wang, C.; Liu, Y. A nanocomposite prepared from silver nanoparticles and carbon dots with peroxidase mimicking activity for colorimetric and SERS-based determination of uric acid. Mikrochim. Acta 2019, 186, 644. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Jiang, K.; Zhu, J.; Zhang, L.; Lin, H. A FRET-based carbon dots-MnO2 nanosheets architecture for glutathione sensing in human whole blood samples. Chem. Commun. 2015, 51, 12748–12751. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Pan, M.; Fang, G.; Wang, S. Carbon dots embedded metal-organic framework@molecularly imprinted nanoparticles for highly sensitive and selective detection of quercetin. Sens. Actuators B Chem. 2019, 286, 321–327. [Google Scholar] [CrossRef]
- Ponnaiah, S.K.; Prakash, P.; Vellaichamy, B.; Paulmony, T.; Selvanathan, R. Picomolar-level electrochemical detection of thiocyanate in the saliva samples of smokers and non-smokers of tobacco using carbon dots doped Fe3O4 nanocomposite embedded on g-C3N4 nanosheets. Electrochim. Acta 2018, 283, 914–921. [Google Scholar] [CrossRef]
- Mandani, S.; Sharma, B.; Dey, D.; Sarma, T.K. Carbon nanodots as ligand exchange probes in Au@C-dot nanobeacons for fluorescent turn-on detection of biothiols. Nanoscale 2015, 7, 1802–1808. [Google Scholar] [CrossRef] [PubMed]
- Hou, J.; Zhou, T.; Wang, L.; Zhang, P.; Ding, L. Template-free microwave-assisted fabrication of carbon dots/Zn(OH)2 composites for separation and enhancing chemical sensing. Sens. Actuators B Chem. 2016, 230, 615–622. [Google Scholar] [CrossRef]
- Wang, L.; Chen, X.; Liu, C.; Yang, W. Non-enzymatic acetylcholine electrochemical biosensor based on flower-like NiAl layered double hydroxides decorated with carbon dots. Sens. Actuators B Chem. 2016, 233, 199–205. [Google Scholar] [CrossRef]
- Wu, L.; Li, M.; Zhang, M.; Yan, M.; Ge, S.; Yu, J. Ultrasensitive electrochemiluminescence immunosensor for tumor marker detection based on nanoporous silver@carbon dots as labels. Sens. Actuators B Chem. 2015, 186, 761–767. [Google Scholar] [CrossRef]
- Li, X.; Zhang, S.; Kulinich, S.; Liu, Y.; Zeng, H. Engineering surface states of carbon dots to achieve controllable luminescence for solid-luminescent composites and sensitive Be2+ detection. Sci. Rep. 2014, 4, 4976. [Google Scholar] [CrossRef]
- Moradi, S.; Sadrjavadi, K.; Farhadian, N.; Hosseinzadeh, L.; Shahlaei, M. Easy synthesis, characterization and cell cytotoxicity of green nano carbon dots using hydrothermal carbonization of Gum Tragacanth and chitosan bio-polymers for bioimaging. J. Mol. Liq. 2018, 259, 284–290. [Google Scholar] [CrossRef]
- Singh, D.P.; Thakur, A.; Kumar, P. Green synthesis of glowing carbon dots from Carica papaya waste pulp and their application as label-free chemo probe for chromium detection in water. Sens. Actuat. B 2019, 283, 363–372. [Google Scholar]
- Yu, S.; Chen, K.; Wang, F.; Zhu, Y.; Zhang, X. Polymer composite fluorescent hydrogel film based on nitrogen-doped carbon dots and their application in the detection of Hg2+ ions. Luminescence 2017, 32, 970–977. [Google Scholar] [CrossRef] [PubMed]
- Ngo, Y.-L.T.; Nguyen, P.L.; Jana, J.; Choi, W.M.; Chung, J.S.; Hur, S.H. Simple paper-based colorimetric and fluorescent glucose sensor using N-doped carbon dots and metal oxide hybrid structures. Anal. Chim. Acta 2021, 1147, 187–198. [Google Scholar] [CrossRef] [PubMed]
- Alexpandi, R.; Gopi, C.V.V.M.; Durgadevi, R.; Kim, H.-J.; Pandian, S.K.; Ravi, A.V. Metal sensing-carbon dots loaded TiO2-nanocomposite for photocatalytic bacterial deactivation and application in aquaculture. Sci. Rep. 2020, 10, 12883. [Google Scholar] [CrossRef]



| Quantum Dots vs. Carbon Dots | Source | Method | Cytotoxicity and Detection-Limit Range | Reference |
|---|---|---|---|---|
| CdTe QDs | CdCl2, NaBH4, tellurium powder, phosphate buffered saline tablets | Electrochemical method | 118 ± 49 μg/mL with the help of electrochemical assay 157 ± 31 μg/mL by MTT cytotoxicity assay | [110] |
| InPZnS QDs | InPZnS alloy core and a thin ZnS shell | Heating Up Method | 70 nM of QDs responsible for cytotoxic effect on environmental health on 72 h exposure | [111] |
| Indium-based QDs | In and Zn content and implementing a more robust outer shell | Heating Up Method | 12.5 mg/kg and 50 mg/kg QDs generally collected in the liver as well as the spleen | [112] |
| InZnP and InZnPS QD | Indium myristate, ZnSt2 | Precipitation Method | 6.25–200 nM upon 24 h exposure | [113] |
| CDs | Onion waste | Hydrothermal method | 0.31 μM | [114] |
| CDs | ZrCl2, trinitrophenol (TNP), 2,4-dinitrophenol, 4-nitrophenol, phenol, Polyvinylpyrrolidone, acetic acid, citric acid | Hydrothermal Method | 0.01–20.0 μM at a low-detection limit of 3.5 nM | [115] |
| CDs | Gelatin | Hydrothermal Method | 1−75 μmol/L | [116] |
| CDs | Carbohydrate | Hydrothermal Method | 0 to 1 × 103 | [117] |
| CDs | DEA | Microwave Method | 5.0 × 10−2 to 8.0 | [118] |
| CDs | Catechol | Hydrothermal Method | 1 × 10−2 to 25 | [119] |
| CDs | Citric acid | Hydrothermal Method | 8.0 × 10−2 to 50 | [120] |
| CDs | Source | Preparation Method | Cytotoxicity/ Detection Limit/ Sensor Response | Quantum Yield | Detection Element | Reference |
|---|---|---|---|---|---|---|
| CDs | Citric acid and urea | Hydrothermal method | 23 mM | 44.7% | Toxic Be2+ ions | [190] |
| CDs | Free Gum Tragacanth and Chitosan | Hydrothermal method | 50 µg/mL | - | Cellular Biosensing | [191] |
| CDs | Carica papaya waste pulp | Hydrothermal Method | 0.708–2.4 ppb | 23.7% | Cr ions detection | [192] |
| N-CDs | Citric acid and ethylenediamine | Microwave-assisted solvothermal method | 0.089 µmol/L | 79.6% | Hg2+ | [193] |
| N-CDs | Citric acid and ethanediamine | Hydrothermal method | 0.076 nM | 84% | Cu2+ ions | [194] |
| N-CDs | Citric acid and ethanediamine | Hydrothermal method | 84 nM | - | Fe3+ in aqueous solution | [139] |
| CDs@MgO nanocomposite | Magnesium nitrate dehydrate, KOH, HF, ethanol and D-glucose | Hydrothermal method | 120 ppm | ~74 ± 3% | H2S | [163] |
| Carbon dots loadedTiO2 nanocomposite | Diplocyclos palmatus leaf extract | Hydrothermal method | 12.5 mM | 54% | Fe 3+ and acute-hepatopancreatic necrosis disease (AHPND) | [195] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sahu, Y.; Hashmi, A.; Patel, R.; Singh, A.K.; Susan, M.A.B.H.; Carabineiro, S.A.C. Potential Development of N-Doped Carbon Dots and Metal-Oxide Carbon Dot Composites for Chemical and Biosensing. Nanomaterials 2022, 12, 3434. https://doi.org/10.3390/nano12193434
Sahu Y, Hashmi A, Patel R, Singh AK, Susan MABH, Carabineiro SAC. Potential Development of N-Doped Carbon Dots and Metal-Oxide Carbon Dot Composites for Chemical and Biosensing. Nanomaterials. 2022; 12(19):3434. https://doi.org/10.3390/nano12193434
Chicago/Turabian StyleSahu, Yogita, Ayesha Hashmi, Rajmani Patel, Ajaya K. Singh, Md. Abu Bin Hasan Susan, and Sónia A. C. Carabineiro. 2022. "Potential Development of N-Doped Carbon Dots and Metal-Oxide Carbon Dot Composites for Chemical and Biosensing" Nanomaterials 12, no. 19: 3434. https://doi.org/10.3390/nano12193434
APA StyleSahu, Y., Hashmi, A., Patel, R., Singh, A. K., Susan, M. A. B. H., & Carabineiro, S. A. C. (2022). Potential Development of N-Doped Carbon Dots and Metal-Oxide Carbon Dot Composites for Chemical and Biosensing. Nanomaterials, 12(19), 3434. https://doi.org/10.3390/nano12193434








