Size-Dependent Phonon-Assisted Anti-Stokes Photoluminescence in Nanocrystals of Organometal Perovskites
Abstract
:1. Introduction
2. Materials and Methods
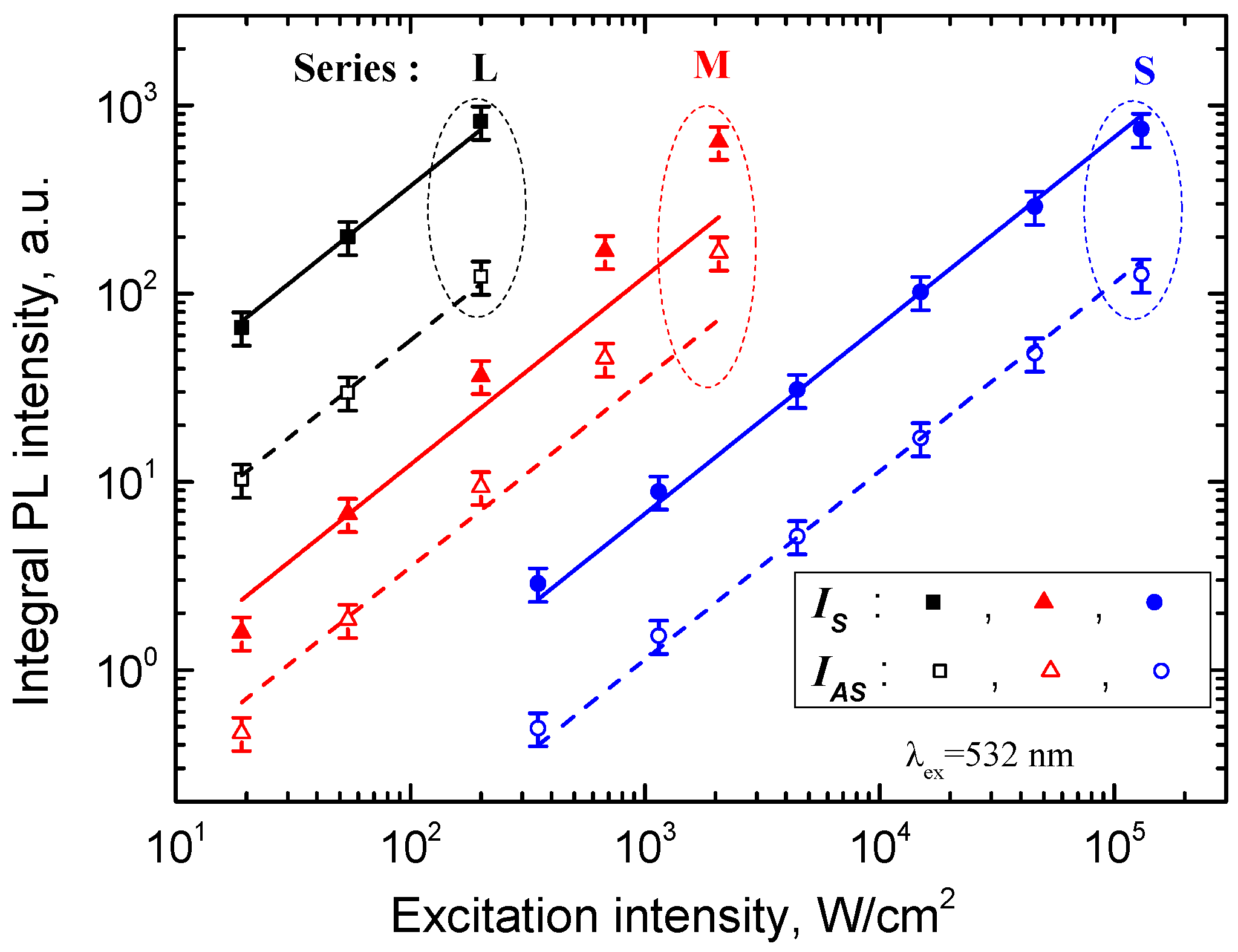
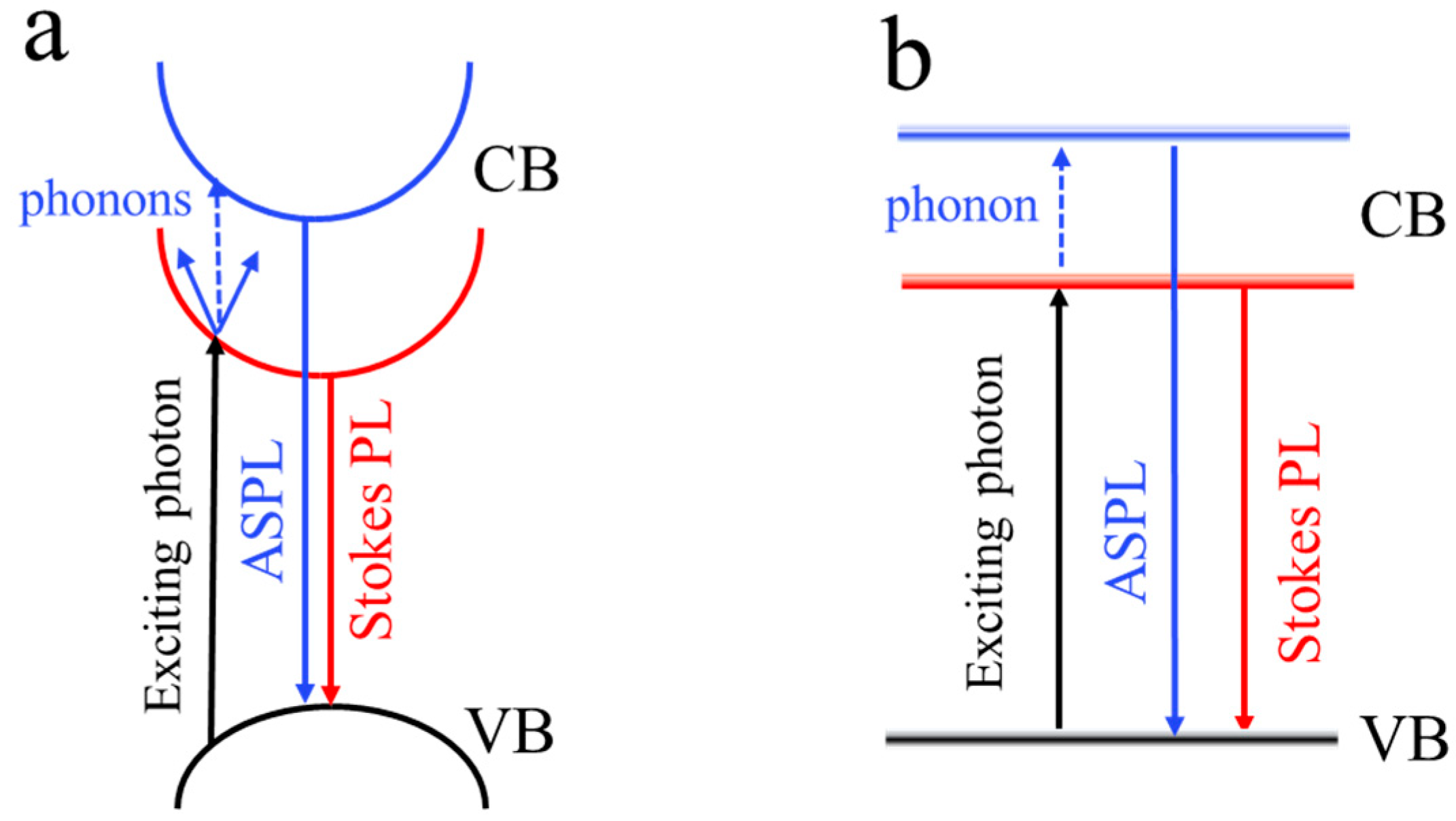
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Shen, Y.R. The Principles of Nonlinear Optics; Wiley-Interscience: Hoboken, NJ, USA, 2003; 576p. [Google Scholar]
- Pope, M.; Swenberg, C.E. Electronic Processes in Organic Crystals and Polymers; Oxford University Press: Oxford, UK, 1999. [Google Scholar]
- Soos, Z.G.; Kepler, R.G. Two-photon-absorption spectrum of poly(di-n-hexylsilane) films. Phys. Rev. B 1991, 43, 11908–11912. [Google Scholar] [CrossRef] [PubMed]
- Lemmer, U.; Rischer, R.; Feldmann, J.; Mahrt, R.F.; Yang, J.; Greiner, A.; Bassler, H.; Gobel, E.O.; Heesel, H.; Kurz, H. Time-resolved studies of two-photon absorption processes in poly(p-phenylenevinylene)s. Chem. Phys. Lett. 1993, 203, 28–32. [Google Scholar] [CrossRef]
- Shalav, A.; Richards, B.S.; Trupke, T.; Krämer, K.W.; Güdel, H.U. Application of NaYF4:Er3+ up-converting phosphors for enhanced nearinfrared silicon solar cell response. Appl. Phys. Lett. 2005, 86, 013505. [Google Scholar] [CrossRef]
- Yamamoto, K.; Fuji, M.; Sowa, S.; Imakita, K.; Aoki, K. Upconversion luminescence of rare-earth-doped Y2O3 nanoparticle with metal nano-cap. J. Phys. Chem. C 2015, 119, 1175–1179. [Google Scholar] [CrossRef]
- Prasad, P.N.; Bhawalkar, J.D.; He, G.S.; Zhao, C.F.; Gvishi, R.; Ruland, G.E.; Zieba, J.; Cheng, P.C.; Pan, S.J. Two-Photon Upconverting Dyes and Applications. U.S. Patent 5,912,257, 15 June 1999. [Google Scholar]
- Yang, H.R.; Han, C.M.; Zhu, X.J.; Liu, Y.; Zhang, K.Y.; Liu, S.J.; Zhao, Q.; Li, F.Y.; Huang, W. Upconversion luminescent chemodosimeter based on NIR organic dye for monitoring methylmercury in vivo. Adv. Funct. Mater. 2016, 26, 1945–1953. [Google Scholar] [CrossRef]
- Akizuki, N.; Aota, S.; Mouri, S.; Matsuda, K.; Miyauchi, Y. Efficient near-infrared up-conversion photoluminescence in carbon nanotubes. Nat. Commun. 2015, 6, 8920. [Google Scholar] [CrossRef]
- Zhang, J.; Li, D.H.; Chen, R.J.; Xiong, Q.H. Laser cooling of a semiconductor by 40 Kelvin. Nature 2013, 493, 504–508. [Google Scholar] [CrossRef]
- Zhang, Q.; Liu, X.F.; Utama, M.I.B.; Xing, G.C.; Sum, T.C.; Xiong, Q.H. Phonon-assisted anti-Stokes lasing in ZnTe nanoribbons. Adv. Mater. 2016, 28, 276–283. [Google Scholar] [CrossRef]
- Wang, X.Y.; Yu, W.W.; Zhang, J.Y.; Aldana, J.; Peng, X.G.; Xiao, M. Photoluminescence upconversion in colloidal CdTe quantum dots. Phys. Rev. B 2003, 68, 125318. [Google Scholar] [CrossRef]
- Chen, W.; Joly, A.G.; McCready, D.E. Upconversion luminescence from CdSe nanoparticles. J. Chem. Phys. 2005, 122, 224708. [Google Scholar] [CrossRef]
- Jakubek, Z.J.; DeVries, J.; Lin, S.Q.; Ripmeester, J.; Yu, K. Exciton recombination and upconverted photoluminescence in colloidal CdSe quantum dots. J. Phys. Chem. C 2008, 112, 8153–8158. [Google Scholar] [CrossRef]
- Downing, E.; Hesselink, L.; Ralston, J.; Macfarlane, R. A three-color, solid-state, three-dimensional display. Science 1993, 273, 1185–1189. [Google Scholar] [CrossRef]
- Fujino, T.; Fujima, T.; Tahara, T. Femtosecond fluorescence dynamics imaging using a fluorescence up-conversion microscope. J. Phys. Chem. B 2005, 109, 15327–15331. [Google Scholar] [CrossRef] [PubMed]
- Kachynski, A.V.; Kuzmin, A.N.; Pudavar, H.E.; Prasad, P.N. Three-dimensional confocal thermal imaging using anti-Stokes luminescence. Appl. Phys. Lett. 2005, 87, 023901. [Google Scholar] [CrossRef]
- Pena, A.-M.; Strupler, M.; Boulesteix, T.; Schanne-Klein, M.-C. Spectroscopic analysis of keratin endogenous signal for skin multiphoton microscopy. Opt. Express 2005, 13, 6268–6274. [Google Scholar] [CrossRef]
- Macfarlane, R.M.; Tong, F.; Silversmith, A.J.; Lenth, W. Violet cw neodymium upconversion laser. Appl. Phys. Lett. 1988, 52, 1300–1302. [Google Scholar] [CrossRef]
- Luo, X.; Eisaman, M.D.; Gosnell, T.R. Laser cooling of a solid by 21 K starting from room temperature. Opt. Lett. 1998, 23, 639–641. [Google Scholar] [CrossRef]
- Thiede, J.; Distel, J.; Greenfield, S.R.; Epstein, R.I. Cooling to 208 K by optical refrigeration. Appl. Phys. Lett. 2005, 86, 154107. [Google Scholar] [CrossRef]
- Kojima, A.; Teshima, K.; Shirai, Y.; Miyasaka, T. Organometal halide perovskites as visible-light sensitizers for photovoltaic cells. J. Am. Chem. Soc. 2009, 131, 6050–6051. [Google Scholar] [CrossRef]
- Tan, Z.K.; Moghaddam, R.S.; Lai, M.L.; Docampo, P.; Higler, R.; Deschler, F.; Price, M.; Sadhanala, A.; Pazos, L.M.; Credgington, D.; et al. Bright light-emitting diodes based on organometal halide perovskite. Nat. Nanotechnol. 2014, 9, 687–692. [Google Scholar] [CrossRef]
- Ye, J.; Byranvand, M.M.; Martínez, C.O.; Hoye, R.L.; Saliba, M.; Polavarapu, L. Defect passivation in lead-halide perovskite nanocrystals and thin films: Toward efficient LEDs and solar cells. Angew. Chem. Int. Ed. 2021, 60, 21636–21660. [Google Scholar] [CrossRef] [PubMed]
- Kovalenko, M.V.; Protesescu, L.; Bodnarchuk, M.I. Properties and potential optoelectronic applications of lead halide perovskite nanocrystals. Science 2017, 358, 745–750. [Google Scholar] [CrossRef] [PubMed]
- Prochazkova, A.J.; Mayr, F.; Gugujonovic, K.; Hailegnaw, B.; Krajcovic, J.; Salinas, Y.; Scharber, M.C. Anti-Stokes photoluminescence study on a methylammonium lead bromide nanoparticle film. Nanoscale 2020, 12, 16556–16561. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Ye, Y.; Liu, C.; Wang, J.; Ruan, J.; Zhao, X.; Han, J. Two-step Anti-Stokes photoluminescence of CsPbX3 nanocrystals. Adv. Opt. Mater. 2021, 9, 2001885. [Google Scholar] [CrossRef]
- Granados del Águila, A.; Do, T.T.H.; Xing, J.; Jee, W.J.; Khurgin, J.B.; Xiong, Q. Efficient up-conversion photoluminescence in all-inorganic lead halide perovskite nanocrystals. Nano Res. 2020, 13, 1962–1969. [Google Scholar] [CrossRef]
- Ye, S.; Yu, M.; Zhao, M.; Song, J.; Qu, J. Low temperature synthesis of high-quality all-inorganic cesium lead halide perovskite nanocrystals in open air and their upconversion luminescence. J. Alloy. Compd. 2018, 730, 62–70. [Google Scholar] [CrossRef]
- Sekerbayev, K.S.; Mussabek, G.K.; Pokryshkin, N.S.; Yakunin, V.G.; Taurbayev, Y.T.; Shabdan, Y.; Utegulov, Z.N.; Chirvony, V.S.; Timoshenko, V.Y. Effect of nanocrystal size on Anti-Stokes photoluminescence in lead perovskites. JETP Lett. 2021, 114, 447–450. [Google Scholar] [CrossRef]
- Ha, S.T.; Shen, C.; Zhang, J.; Xiong, Q. Laser cooling of organic–inorganic lead halide perovskites. Nat. Photonics 2016, 10, 115–121. [Google Scholar] [CrossRef]
- Roman, B.J.; Villegas, N.M.; Lytle, K.; Sheldon, M. Optically cooling cesium lead tribromide nanocrystals. Nano Lett. 2020, 20, 8874–8879. [Google Scholar] [CrossRef]
- Tonkaev, P.; Zograf, G.; Makarov, S. Optical cooling of lead halide perovskite nanoparticles enhanced by Mie resonances. Nanoscale 2019, 11, 17800–17806. [Google Scholar] [CrossRef] [Green Version]
- Shamsi, J.; Urban, A.S.; Imran, M.; De Trizio, L.; Manna, L. Metal halide perovskite nanocrystals: Synthesis, post-synthesis modifications, and their optical properties. Chem. Rev. 2019, 119, 3296–3348. [Google Scholar] [CrossRef] [PubMed]
- Prochazkova, A.J.; Scharber, M.C.; Yumusak, C.; Jančík, J.; Másilko, J.; Brüggemann, O.; Weiter, M.; Sariciftci, N.S.; Krajcovic, J.; Salinas, Y.; et al. Synthesis conditions influencing formation of MAPbBr3 perovskite nanoparticles prepared by the ligand-assisted precipitation method. Sci. Rep. 2020, 10, 15720. [Google Scholar] [CrossRef] [PubMed]
- Veldhuis, S.A.; Tay, Y.K.E.; Bruno, A.; Dintakurti, S.S.; Bhaumik, S.; Muduli, S.K.; Li, M.; Mathews, N.; Sum, T.C.; Mhaisalkar, S.G. Benzyl alcohol-treated CH3NH3PbBr3 nanocrystals exhibiting high luminescence, stability, and ultralow amplified spontaneous emission thresholds. Nano Lett. 2017, 17, 7424–7432. [Google Scholar] [CrossRef] [PubMed]
- Priante, D.; Dursun, I.; Alias, M.S.; Shi, D.; Melnikov, V.A.; Ng, T.K.; Ooi, B.S. The recombination mechanisms leading to amplified spontaneous emission at the true-green wavelength in CH3NH3PbBr3 perovskites. Appl. Phys. Lett. 2015, 106, 081902. [Google Scholar] [CrossRef]
- Liu, Y.; Yang, Z.; Cui, D.; Ren, X.; Sun, J.; Liu, X.; Zhang, J.; Wei, Q.; Fan, H.; Yu, F.; et al. Two-inch-sized perovskite CH3NH3PbX3 (X = Cl, Br, I) crystals: Growth and characterization. Adv. Mater. 2015, 35, 5176–5183. [Google Scholar] [CrossRef] [PubMed]
- Nakada, K.; Matsumoto, Y.; Shimoi, Y.; Yamada, K.; Furukawa, Y. Temperature-dependent evolution of Raman spectra of methylammonium lead halide perovskites, CH3NH3PbX3 (X=I, Br). Molecules 2019, 24, 626. [Google Scholar] [CrossRef]
- Fan, Y.; Li, S.; Xu, D.P. Raman spectra of oleic acid and linoleic acid. Spectrosc. Spectr. Anal. 2013, 33, 3240–3243. [Google Scholar]
- Tyagi, P.; Arveson, S.M.; Tisdale, W.A. Colloidal organohalide perovskite nanoplatelets exhibiting quantum confinement. J. Phys. Chem. Lett. 2015, 6, 1911–1916. [Google Scholar] [CrossRef]
- Sekerbayev, K.S.; Shabdan, Y.; Mikhailova, S.L.; Pryadun, V.V.; Taurbayev, Y.T.; Taurbayev, T.I.; Timoshenko, V.Y. Bias voltage-dependent photoinduced current and photoluminescence in organometal perovskite layers on silicon substrates. Phys. B Condens. Matter 2020, 582, 412025. [Google Scholar] [CrossRef]
- Takagahara, T. Electron-phonon interactions in semiconductor nanocrystals. J. Lum. 1996, 70, 129–143. [Google Scholar] [CrossRef]
- Xiong, Y.; Liu, C.; Wang, J.; Han, J.; Zhao, X. Near-infrared anti-Stokes photoluminescence of PbS QDs embedded in glasses. Opt. Express 2017, 25, 6874–6882. [Google Scholar] [CrossRef] [PubMed]
- Kovalev, D.; Heckler, H.; Polisski, G.; Koch, F. Optical properties of Si nanocrystals. Phys. Stat. Sol. B 1999, 215, 871–932. [Google Scholar] [CrossRef]
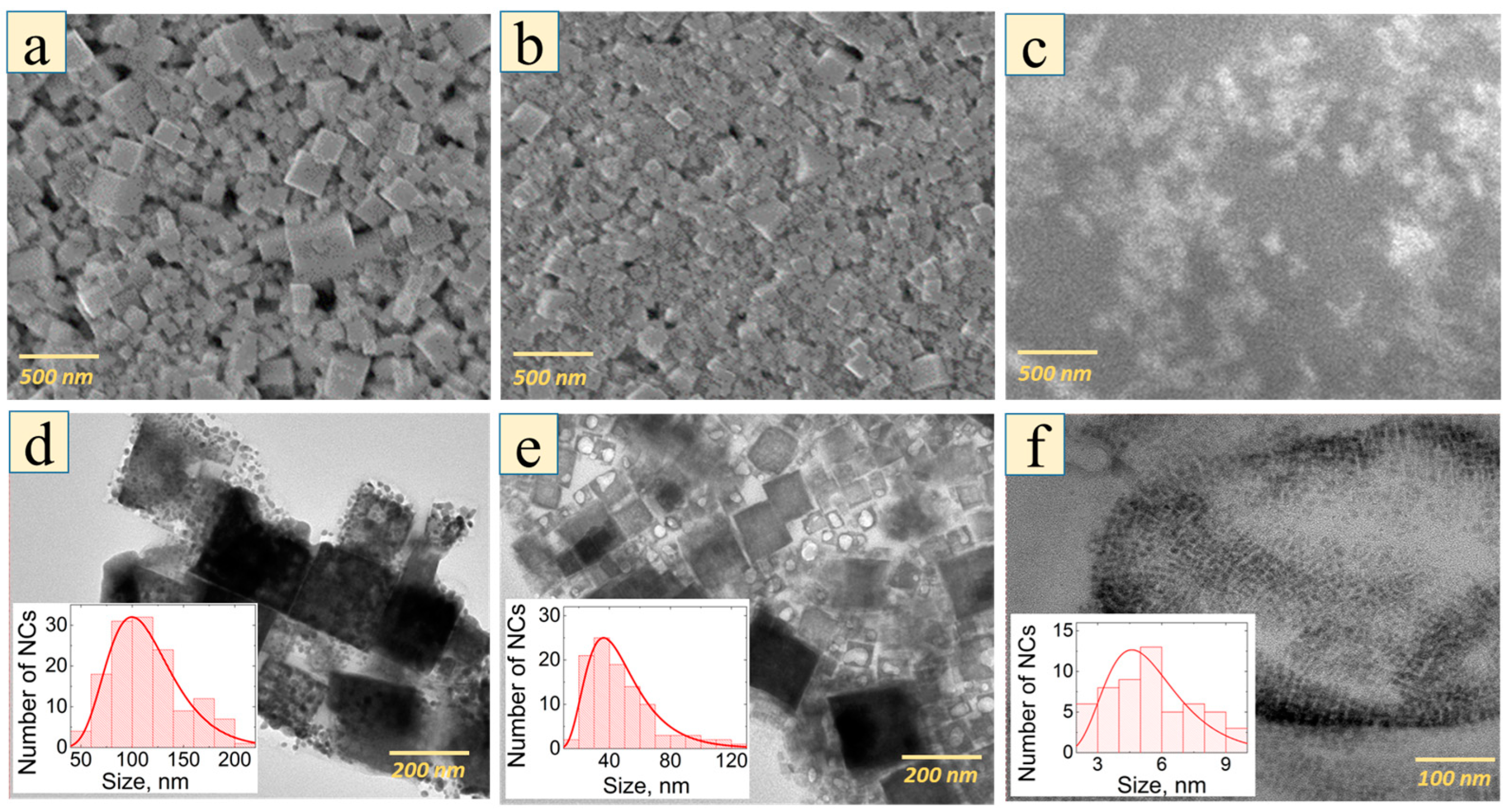

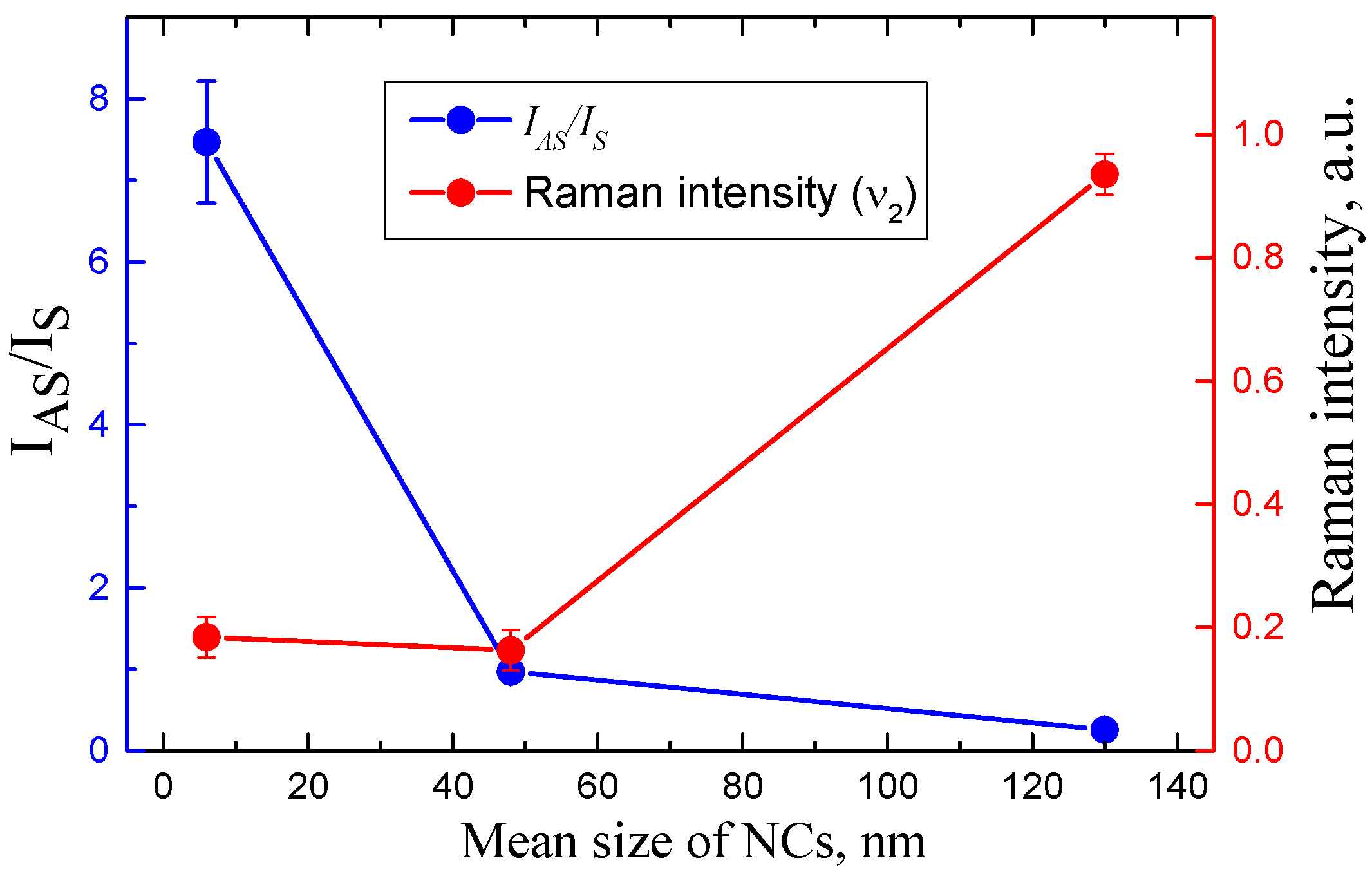
| Series of Samples | Octylamine, % | NCs Mean Size, nm | PL Wavelength, nm |
|---|---|---|---|
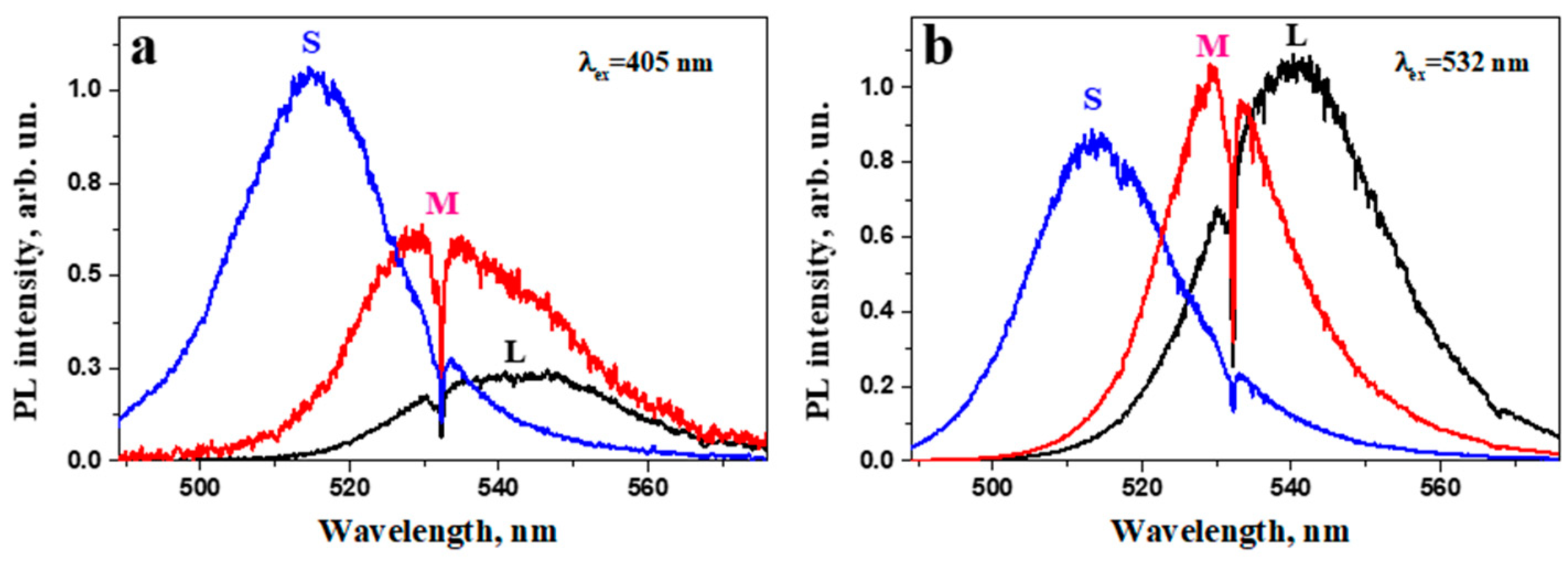
| S (small NCs) | 0.6 | 5.5 ± 1.5 | 515 ± 1 |
| M (medium NCs) | 0.4 | 46.0 ± 4.0 | 534 ± 1 |
| L (large NCs) | 0.2 | 120 ± 24 | 543 ± 1 |
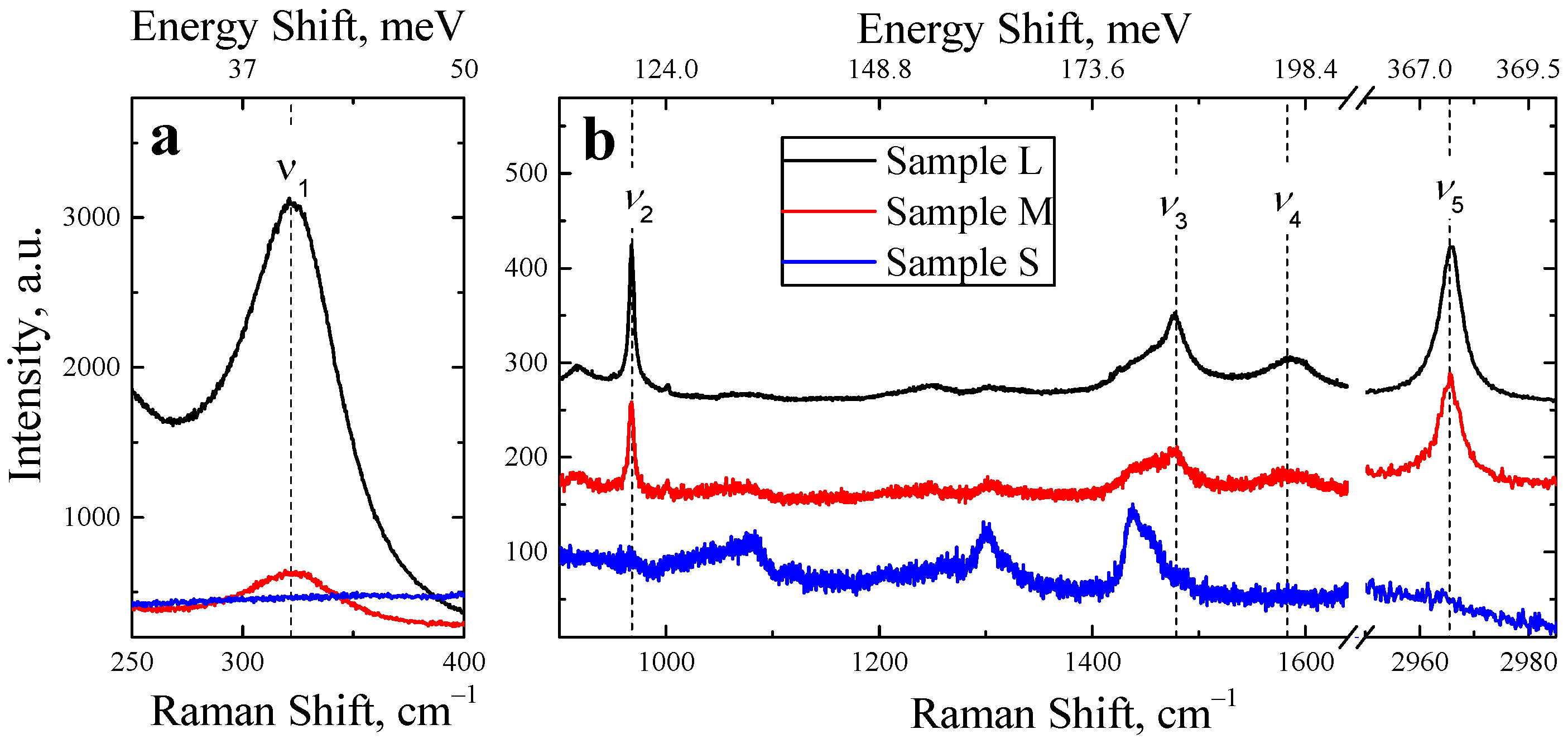
| Mode Notation | Frequency, cm−1 (meV) | Description |
|---|---|---|
| ν1 | 322 (40) | MA rotation |
| ν2 | 967 (120) | C–N stretching |
| ν3 | 1478 (183) | NH3+ symmetric deformation |
| ν4 | 1581 (196) | C–N twisting |
| ν5 | 2965 (368) | CH3 symmetric stretching |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sekerbayev, K.; Taurbayev, Y.; Mussabek, G.; Baktygerey, S.; Pokryshkin, N.S.; Yakunin, V.G.; Utegulov, Z.; Timoshenko, V.Y. Size-Dependent Phonon-Assisted Anti-Stokes Photoluminescence in Nanocrystals of Organometal Perovskites. Nanomaterials 2022, 12, 3184. https://doi.org/10.3390/nano12183184
Sekerbayev K, Taurbayev Y, Mussabek G, Baktygerey S, Pokryshkin NS, Yakunin VG, Utegulov Z, Timoshenko VY. Size-Dependent Phonon-Assisted Anti-Stokes Photoluminescence in Nanocrystals of Organometal Perovskites. Nanomaterials. 2022; 12(18):3184. https://doi.org/10.3390/nano12183184
Chicago/Turabian StyleSekerbayev, Kairolla, Yerzhan Taurbayev, Gauhar Mussabek, Saule Baktygerey, Nikolay S. Pokryshkin, Valery G. Yakunin, Zhandos Utegulov, and Victor Yu. Timoshenko. 2022. "Size-Dependent Phonon-Assisted Anti-Stokes Photoluminescence in Nanocrystals of Organometal Perovskites" Nanomaterials 12, no. 18: 3184. https://doi.org/10.3390/nano12183184
APA StyleSekerbayev, K., Taurbayev, Y., Mussabek, G., Baktygerey, S., Pokryshkin, N. S., Yakunin, V. G., Utegulov, Z., & Timoshenko, V. Y. (2022). Size-Dependent Phonon-Assisted Anti-Stokes Photoluminescence in Nanocrystals of Organometal Perovskites. Nanomaterials, 12(18), 3184. https://doi.org/10.3390/nano12183184





