Nanostructured PbS-Doped Inorganic Film Synthesized by Sol-Gel Route
Abstract
:1. Introduction
2. Materials and Methods
2.1. Synthesis of the Film
2.2. Measurements
3. Results and Discussion
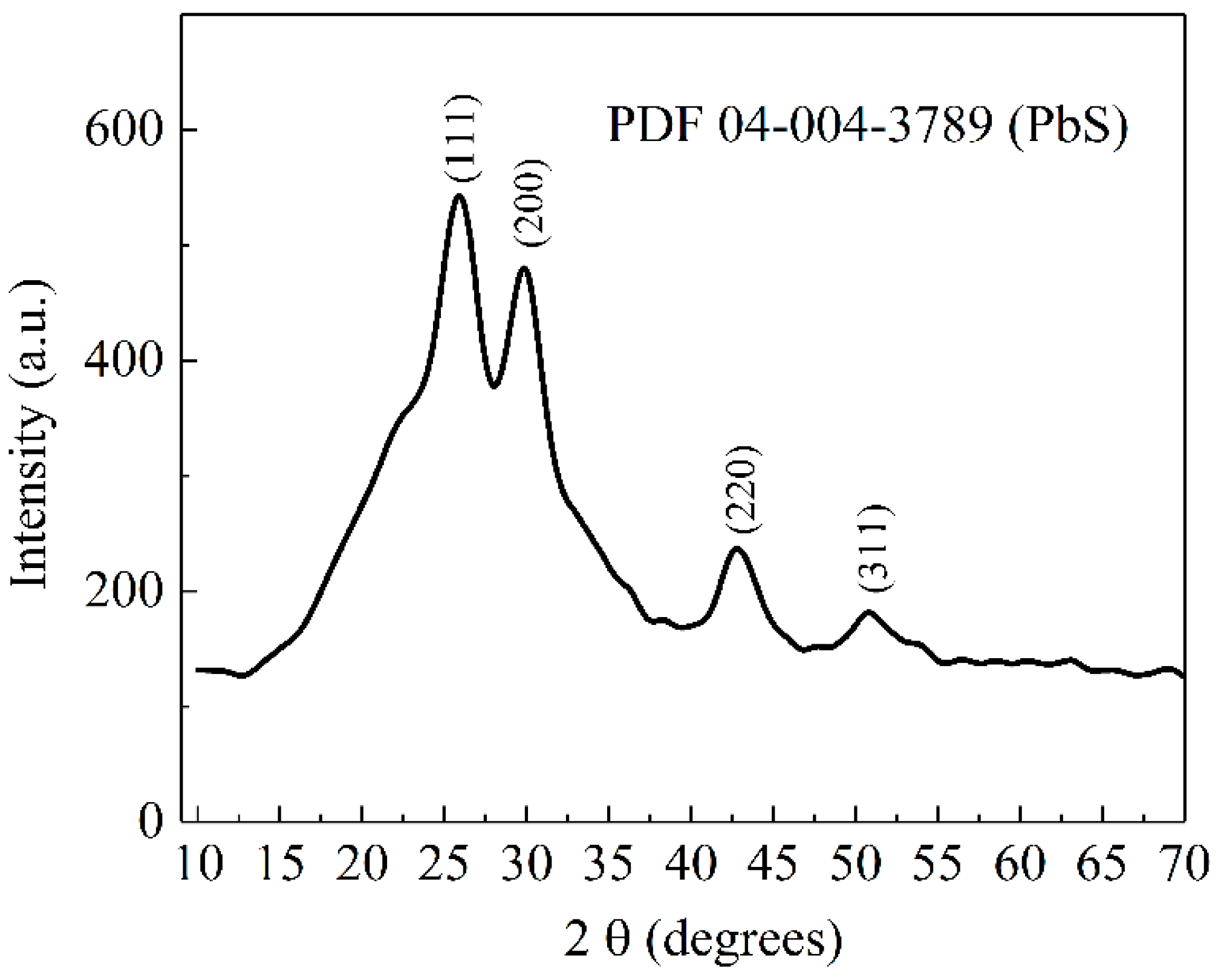
3.1. X-ray Diffraction Analysis (XRD)
3.2. Optical Properties
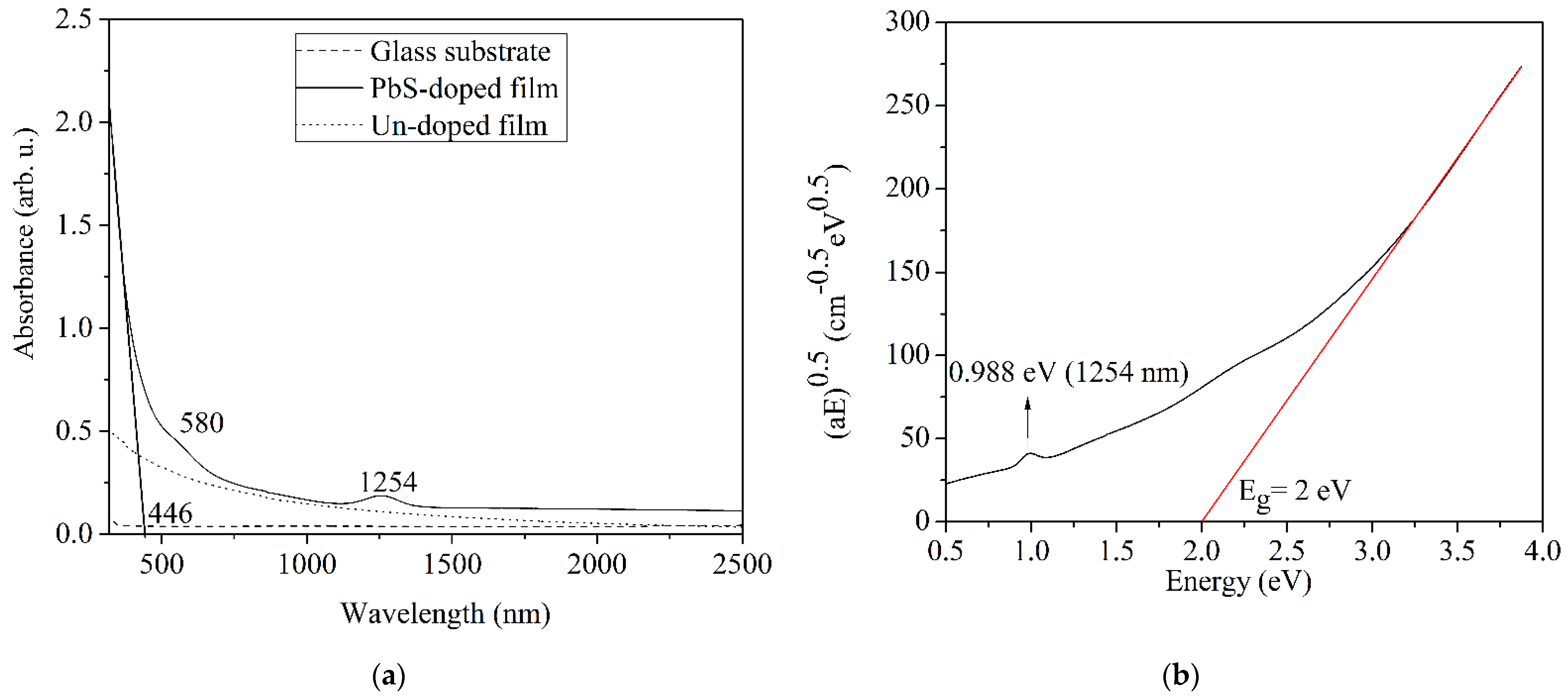
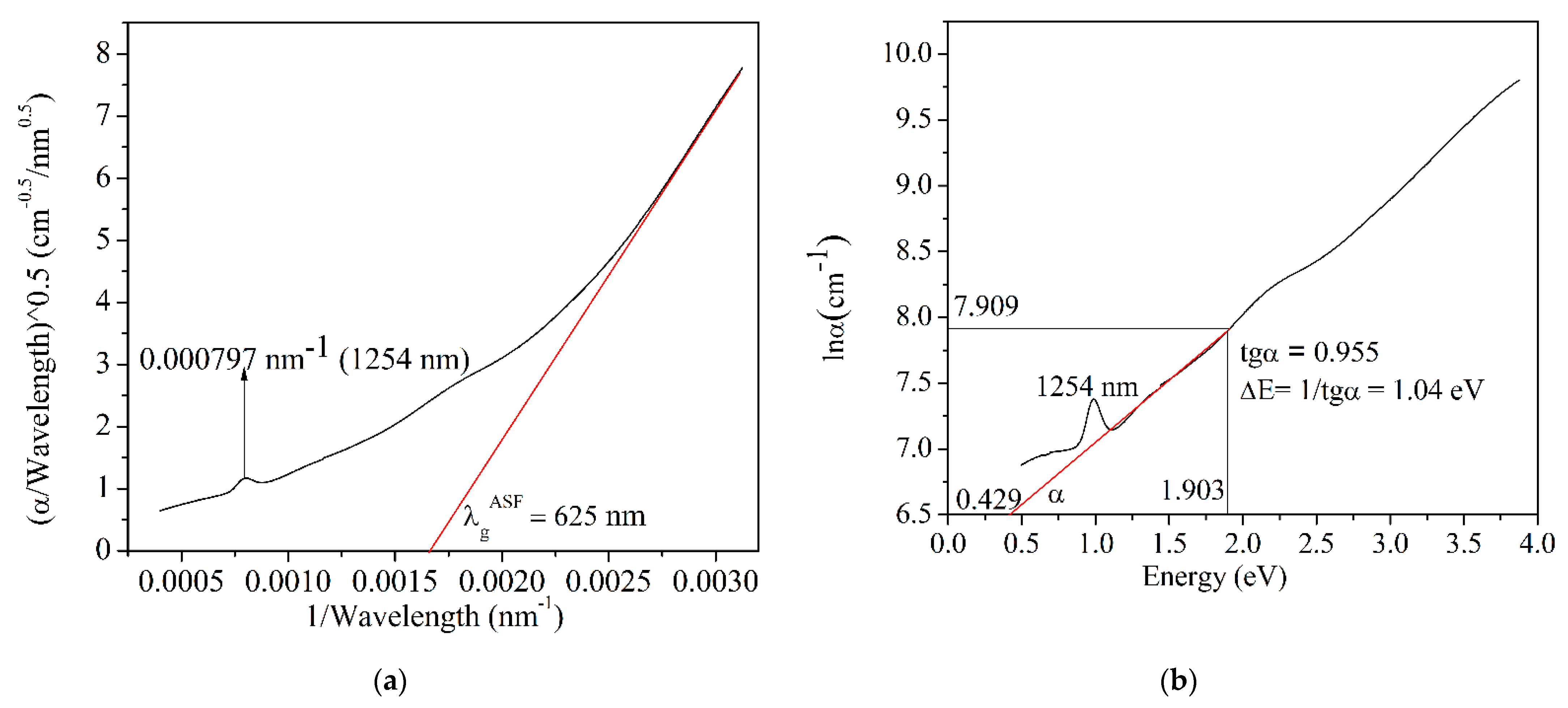
3.2.1. Optical Absorption
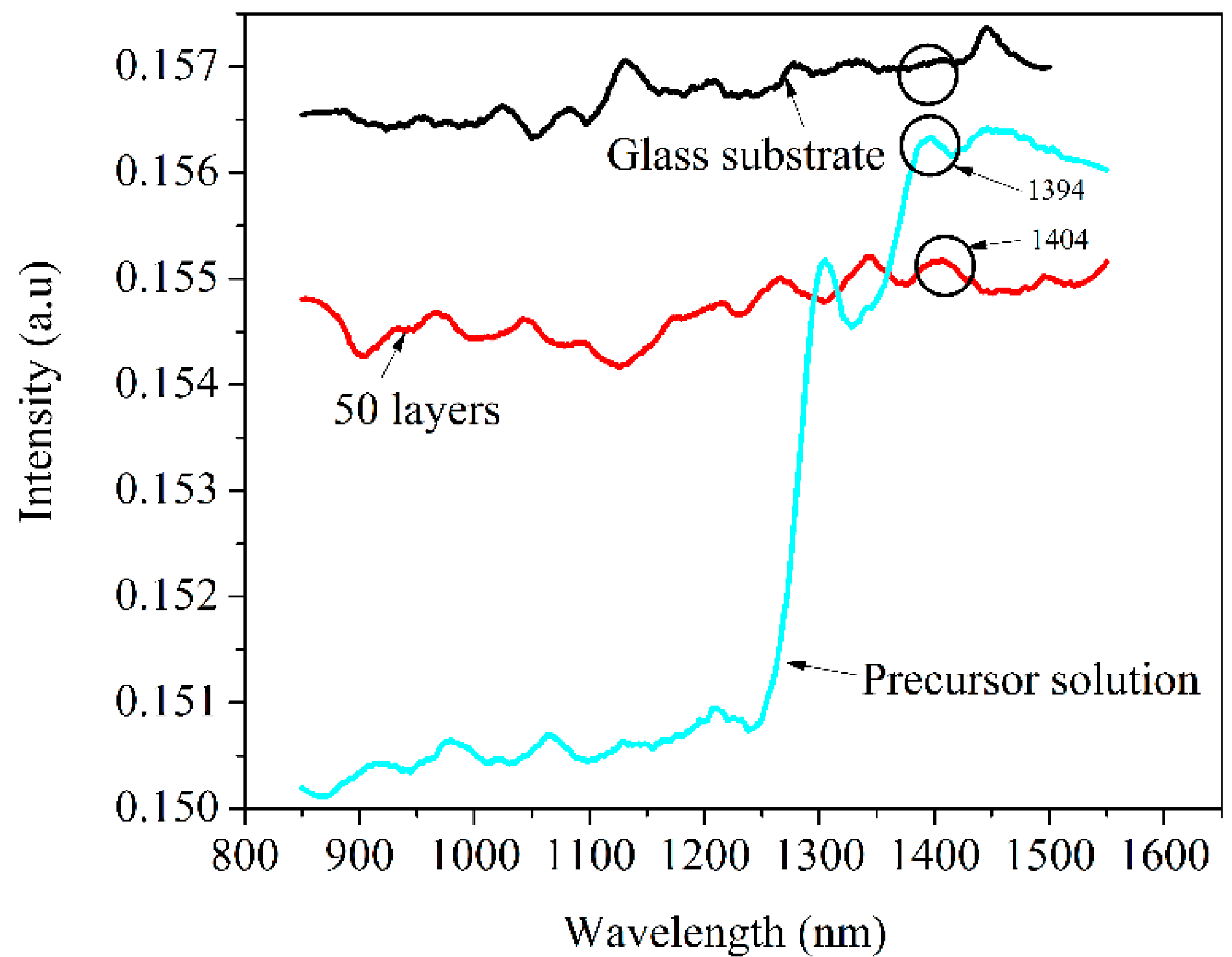
3.2.2. Optical Emission
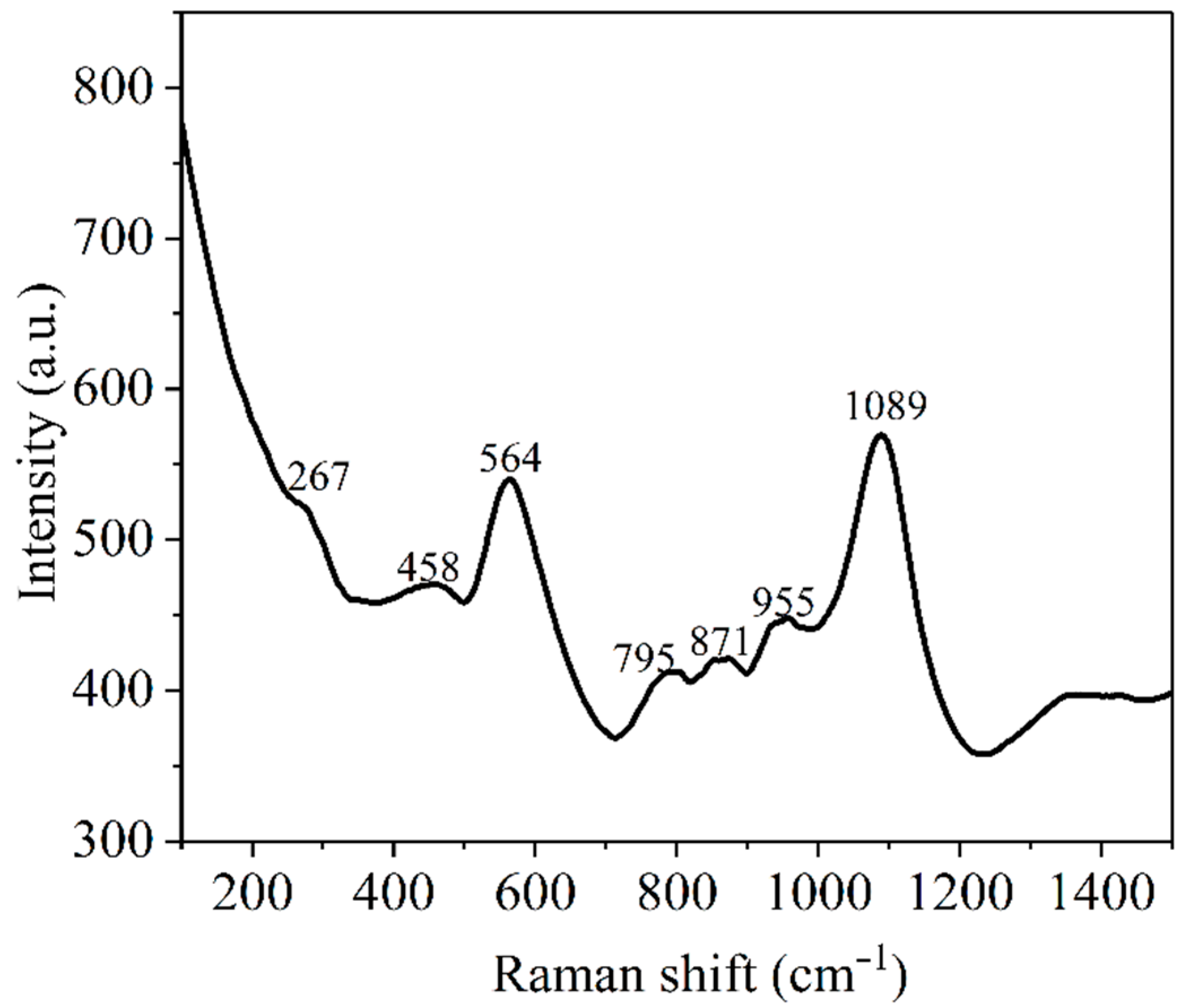
3.3. Raman Spectroscopy
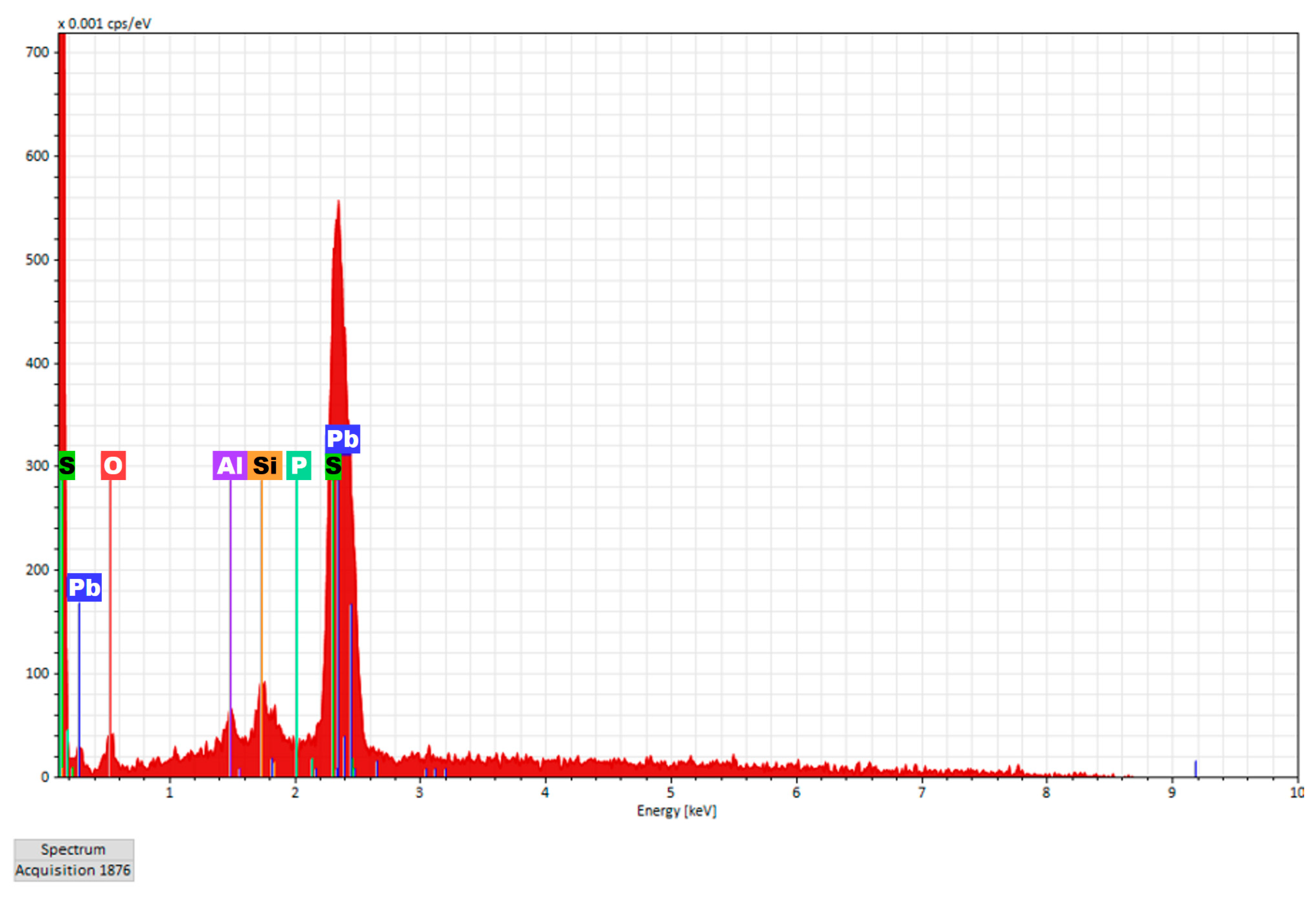
3.4. Scanning Electron Microscopy-Energy Dispersive X-ray (SEM-EDX)
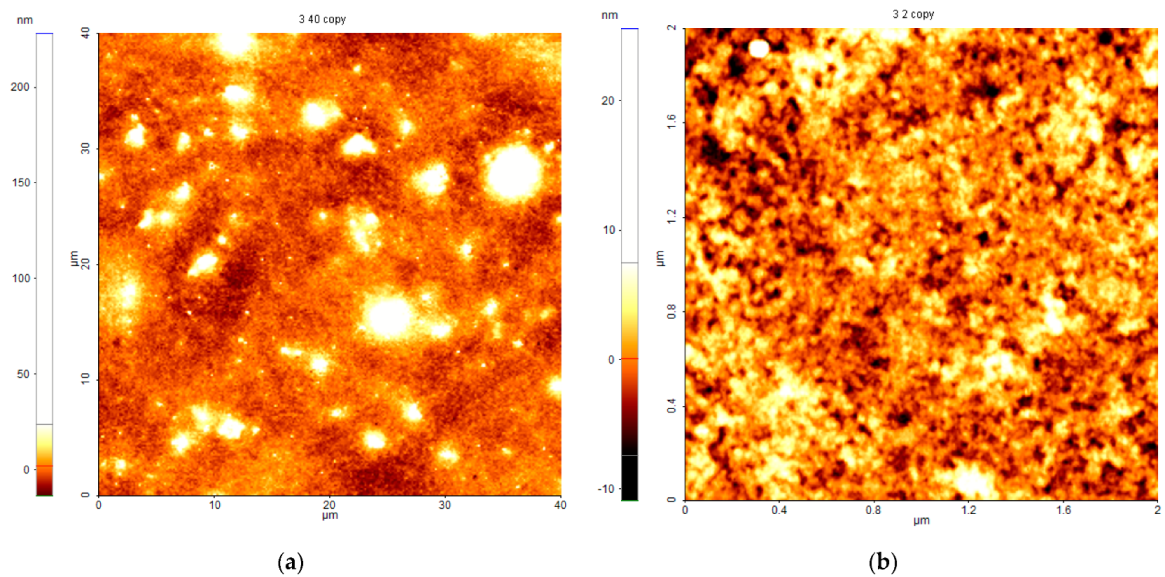
3.5. Atomic Force Microscopy (AFM)
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Jang, Y.; Shapiro, A.; Isarov, M.; Rubin-Brusilovski, A.; Safran, A.; Budniak, A.K.; Horani, F.; Dehnel, J.; Sashchiuk, A.; Lifshitz, E. Interface control of electronic and optical properties in IV–VI and II–VI core/shell colloidal quantum dots: A review. Chem. Commun. 2016, 53, 1002–1024. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Tong, X.; Long, Z.; Channa, A.I.; Zhao, H.; Li, X.; Cai, M.; You, Y.; Sun, X.; Wang, Z. Rational design of eco-friendly Mn-doped nonstoichiometric CuInSe/ZnSe core/shell quantum dots for boosted photoelectrochemical efficiency. Nano Res. 2022, 15, 7614–7621. [Google Scholar] [CrossRef]
- You, Y.; Tong, X.; Channa, A.I.; Li, X.; Liu, C.; Ye, H.; Wang, Z. Tailoring the optoelectronic properties of eco-friendly CuGaS2/ZnSe core/shell quantum dots for boosted photoelectrochemical solar hydrogen production. EcoMat 2022, e12206. [Google Scholar] [CrossRef]
- Zaini, M.S.; Kamarudin, M.A.; Liew, Y.C.J.; Ahmad, S.A.A.; Mohmad, A.R. Temperature and Power Dependence of Photoluminescence in PBS Quantum Dots Nanoparticles. Sains Malays. 2019, 48, 1281–1288. [Google Scholar] [CrossRef]
- Chillcce, E.; Ramos-Gonzales, R.; Cordeiro, C.; Gutierrez-Rivera, L.; Fragnito, H.; Cruz, C.D.B.; Bordonalli, A.; Hernández-Figueroa, H.; Braga, R.; Barbosa, L. Luminescence of PbS quantum dots spread on the core surface of a silica microstructured optical fiber. J. Non-Cryst. Solids 2010, 356, 2397–2401. [Google Scholar] [CrossRef]
- Parfenov, P.S.; Litvin, A.P.; Baranov, A.V.; Ushakova, E.V.; Fedorov, A.V.; Prudnikov, A.V.; Artemyev, M.V. Meas-urement of the luminescence decay times of PbS quantum dots in the near-IR spectral range. Opt. Spectrosc. 2012, 112, 868–873. [Google Scholar] [CrossRef]
- Parfenov, P.S.; Litvin, A.P.; Ushakova, E.V.; Fedorov, A.V.; Baranov, A.V.; Berwick, K. Note: Near infrared spectral and transient measurements of PbS quantum dots luminescence. Rev. Sci. Instrum. 2013, 84, 116104. [Google Scholar] [CrossRef]
- Zhao, H.; Vomiero, A.; Rosei, F. Tailoring the Heterostructure of Colloidal Quantum Dots for Ratiometric Optical Nanothermometry. Small 2020, 16, e2000804. [Google Scholar] [CrossRef]
- Litvin, A.P.; Babaev, A.A.; Parfenov, P.S.; Dubavik, A.; Cherevkov, S.A.; Baranov, M.A.; Bogdanov, K.V.; Reznik, I.A.; Ilin, P.O.; Zhang, X.; et al. Ligand-Assisted Formation of Graphene/Quantum Dot Monolayers with Improved Morphological and Electrical Properties. Nanomaterials 2020, 10, 723. [Google Scholar] [CrossRef]
- Turyanska, L.; Patanè, A.; Henini, M.; Hennequin, B.; Thomas, N.R. Temperature dependence of the photoluminescence emission from thiol-capped PbS quantum dots. Appl. Phys. Lett. 2007, 90, 101913. [Google Scholar] [CrossRef] [Green Version]
- Zhao, H.; Liang, H.; Vidal, F.; Rosei, F.; Vomiero, A.; Ma, D. Size Dependence of Temperature-Related Optical Properties of PbS and PbS/CdS Core/Shell Quantum Dots. J. Phys. Chem. C 2014, 118, 20585–20593. [Google Scholar] [CrossRef]
- McSherry, M.; Fitzpatrick, C.; Lewis, E. Review of luminescent based fibre optic temperature sensors. Sens. Rev. 2005, 25, 56–62. [Google Scholar] [CrossRef]
- Gao, W.; Zhai, G.; Zhang, C.; Shao, Z.; Zheng, L.; Zhang, Y.; Yang, Y.; Li, X.; Liu, X.; Xu, B. Towards understanding the initial performance improvement of PbS quantum dot solar cells upon short-term air exposure. RSC Adv. 2018, 8, 15149–15157. [Google Scholar] [CrossRef]
- Gaponenko, M.S.; Lutich, A.A.; Tolstik, N.A.; Onushchenko, A.A.; Malyarevich, A.; Petrov, E.; Yumashev, K. Temperature-dependent photoluminescence of PbS quantum dots in glass: Evidence of exciton state splitting and carrier trapping. Phys. Rev. B 2010, 82, 125320. [Google Scholar] [CrossRef]
- Yang, P.; Song, C.F.; Lü, M.K.; Yin, X.; Zhou, G.J.; Xu, D.; Yuan, D.R. The luminescence of PbS nanoparticles embedded in sol–gel silica glass. Chem. Phys. Lett. 2001, 345, 429–434. [Google Scholar] [CrossRef]
- Moreels, I.; Lambert, K.; Smeets, D.; Muynck, D.D.; Nollet, T.; Martins, J.C.; Vanhaecke, F.; Vantomme, A.; Delerue, C.; Allan, G.; et al. Size-Dependent Optical Properties of Colloidal PbS Quantum Dots. ACS Nano 2009, 3, 3023–3030. [Google Scholar] [CrossRef] [PubMed]
- Su, G.; Liu, C.; Deng, Z.; Zhao, X.; Zhou, X. Size-dependent photoluminescence of PbS QDs embedded in silicate glasses. Opt. Mater. Express 2017, 7, 2194. [Google Scholar] [CrossRef]
- Mozafari, M.; Moztarzadeh, F. Microstructural and optical properties of spherical lead sulphide quantum dots-based optical sensors. Micro Nano Lett. 2011, 6, 161–164. [Google Scholar] [CrossRef]
- Szendrei, K.; Speirs, M.; Gomulya, W.; Jarzab, D.; Manca, M.; Mikhnenko, O.V.; Yarema, M.; Kooi, B.J.; Heiss, W.; Loi, M.A. Exploring the Origin of the Temperature-Dependent Behavior of PbS Nanocrystal Thin Films and Solar Cells. Adv. Funct. Mater. 2012, 22, 1598–1605. [Google Scholar] [CrossRef]
- De Iacovo, A.; Venettacci, C.; Colace, L.; Scopa, L.; Foglia, S. PbS Colloidal Quantum Dot Photodetectors operating in the near infrared. Sci. Rep. 2016, 6, 37913. [Google Scholar] [CrossRef] [Green Version]
- Lu, K.; Wang, Y.; Liu, Z.; Han, L.; Shi, G.; Fang, H.; Chen, J.; Ye, X.; Chen, S.; Yang, F.; et al. High-Efficiency PbS Quantum-Dot Solar Cells with Greatly Simplified Fabrication Processing via “Solvent-Curing”. Adv. Mater. 2018, 30, e1707572. [Google Scholar] [CrossRef] [PubMed]
- Popescu, M.; Sava, F.; Lorinczi, A.; Savastru, R.; Savastru, D.; Mihailescu, I.N. Optical recording in sulfur-selenium layers. J. Optoelectron. Adv. Mater. 2004, 6, 883–886. [Google Scholar]
- Pop, I.; Nascu, C.; Ionescu, V.; Indrea, E.; Bratu, I. Structural and optical properties of PbS thin films obtained by chemical deposition. Thin Solid Films 1997, 307, 240–244. [Google Scholar] [CrossRef]
- Fedjukin, A.V.; Nituta, A.N.; Lukin, A.N.; Semenov, V.; Ovechkina, N.M. Optical properties and structures of PbS films obtained via pyrolisis of thiourea complexes. Bull. Russ. Acad. Sci. Phys. 2015, 79, 215–217. [Google Scholar] [CrossRef]
- Naşcu, C.; Vomir, V.; Pop, I.; Ionescu, V.; Grecu, R. The study of lead sulphide films. VI. Influence of oxidants on the chemically deposited PbS thin films. Mater. Sci. Eng. B 1996, 41, 235–240. [Google Scholar] [CrossRef]
- Larramendi, E.; Calzadilla, O.; González-Arias, A.; Hernández, E.; Ruiz-Garcia, J. Effect of surface structure on photosensitivity in chemically deposited PbS thin films. Thin Solid Films 2001, 389, 301–306. [Google Scholar] [CrossRef]
- Pentia, E.; Pintilie, L.; Tivarus, C.; Botila, T. Influence of Sb3+ ions on photoconductive properties of chemically deposited PbS films. Mater. Sci. Eng. B 2001, 80, 23–26. [Google Scholar] [CrossRef]
- Joshi, R.K.; Kanjilal, A.; Sehgal, H. Solution grown PbS nanoparticle films. Appl. Surf. Sci. 2004, 221, 43–47. [Google Scholar] [CrossRef]
- Valenzuela-Jáuregui, J.; Ramírez-Bon, R.; Mendoza-Galván, A.; Sotelo-Lerma, M. Optical properties of PbS thin films chemically deposited at different temperatures. Thin Solid Films 2003, 441, 104–110. [Google Scholar] [CrossRef]
- Seghaier, S.; Kamoun, N.; Brini, R.; Amara, A.B. Structural and optical properties of PbS thin films deposited by chemical bath deposition. Mater. Chem. Phys. 2006, 97, 71–80. [Google Scholar] [CrossRef]
- Gode, F.; Yavuz, F.; Kariper, I.A. Preparation and Characterization of Nanocrystalline PbS Thin Films Produced by Chemical Bath Deposition. Acta Phisica Pol. A 2015, 128, B215–B218. [Google Scholar] [CrossRef]
- Sharon, M.; Ramaiah, K.; Kumar, M.; Neumann-Spallart, M.; Levy-Clement, C. Electrodeposition of lead sulphide in acidic medium. J. Electroanal. Chem. 1997, 436, 49–52. [Google Scholar] [CrossRef]
- Nair, P.K.; Garcia, V.M.; Hernandez, A.B.; Nair, M.T.S. Photoaccelerated chemical deposition of PbS thin films: Novel applications in decorative coatings and imaging techniques. J. Phys. D Appl. Phys. 1991, 24, 1466–1472. [Google Scholar] [CrossRef]
- Ni, Y.; Wang, F.; Liu, H.; Yin, G.; Hong, J.; Ma, X.; Xu, Z. A novel aqueous-phase route to prepare flower-shaped PbS micron crystals. J. Cryst. Growth 2004, 262, 399–402. [Google Scholar] [CrossRef]
- Zhao, Y.; Liao, X.-H.; Hong, J.-M.; Zhu, J.-J. Synthesis of lead sulfide nanocrystals via microwave and sonochemical methods. Mater. Chem. Phys. 2004, 87, 149–153. [Google Scholar] [CrossRef]
- Patel, J.; Mighri, F.; Ajji, A.; Tiwari, D.; Chaudhuri, T.K. Spin-coating deposition of PbS and CdS thin films for solar cell application. Appl. Phys. A 2014, 117, 1791–1799. [Google Scholar] [CrossRef]
- Yanover, D.; Čapek, R.K.; Rubin-Brusilovski, A.; Vaxenburg, R.; Grumbach, N.; Maikov, G.I.; Solomeshch, O.; Sashchiuk, A.; Lifshitz, E. Small-Sized PbSe/PbS Core/Shell Colloidal Quantum Dots. Chem. Mater. 2012, 24, 4417–4423. [Google Scholar] [CrossRef]
- Yanover, D.; Vaxenburg, R.; Tilchin, J.; Rubin-Brusilovski, A.; Zaiats, G.; Čapek, R.K.; Sashchiuk, A.; Lifshitz, E. Significance of Small-Sized PbSe/PbS Core/Shell Colloidal Quantum Dots for Optoelectronic Applications. J. Phys. Chem. C 2014, 118, 17001–17009. [Google Scholar] [CrossRef]
- Rubin-Brusilovski, A.; Jang, Y.; Shapiro, A.; Safran, A.; Sashchiuk, A.; Lifshitz, E. Influence of Interfacial Strain on Optical Properties of PbSe/PbS Colloidal Quantum Dots. Chem. Mater. 2016, 28, 9056–9063. [Google Scholar] [CrossRef]
- Yadav, S.K.; Jeevanandam, P. Synthesis of PbS–Al2O3 nanocomposites by sol–gel process and studies on their optical properties. Opt. Mater. 2015, 46, 209–215. [Google Scholar] [CrossRef]
- Rusu, M.; Stefan, C.; Elisa, M.; Feraru, I.; Vasiliu, I.C.; Bartha, C.; Trusca, R.; Vasile, E.; Peretz, S. CdS/ZnS-doped silico-phosphate films prepared by sol-gel synthesis. J. Non-Cryst. Solids 2018, 481, 435–440. [Google Scholar] [CrossRef]
- Dinca, M.; Sava, B.; Galca, A.; Kuncser, V.; Iacob, N.; Stan, G.; Boroica, L.; Filip, A.; Elisa, M. Magneto-optical properties of borophosphate glasses co-doped with Tb3+ and Dy3+ions. J. Non-Cryst. Solids 2021, 568, 120967. [Google Scholar] [CrossRef]
- Elisa, M.; Vasiliu, I.C.; Feraru, I.D.; Iordanescu, R.; Rusu, M.I.; Trusca, R.D.; Vasile, E.; Peretz, S. CdSe/ZnS-doped silicophosphate films prepared by sol–gel method. J. Sol-Gel Sci. Technol. 2015, 73, 660–665. [Google Scholar] [CrossRef]
- PDF-ICDD. Powder Diffraction File (PDF-4+ 2018 Software 4.18.0.2); International Centre for Diffraction Data: Newtown Square, PA, USA, 2011. [Google Scholar]
- Zhao, Y.; Li, W. PbS quantum dots band gap tuning via Eu doping. Mater. Res. Express 2019, 6, 115908. [Google Scholar] [CrossRef]
- Joshi, A.; Narsingi, K.Y.; Manasreh, M.O.; Davis, E.A.; Weaver, B.D. Temperature dependence of the band gap of colloidal CdSe/ZnS core/shell nanocrystals embedded into an ultraviolet curable resin. Appl. Phys. Lett. 2006, 89, 131907. [Google Scholar] [CrossRef]
- Bhowmick, M.; Singh, A.K.; Barik, P.; Xi, H.; Ullrich, B. Impact of PbS quantum dots on GaAs photoluminescence. In Proceedings of the SPIE Optical Components and Materials XVIII Conference, 116820Y, San Diego, CA, USA, 5 March 2021. [Google Scholar] [CrossRef]
- Badawi, A.; Al-Hosiny, N.; Merazga, A.; Albaradi, A.M.; Abdallah, S.; Talaat, H. Study of the back recombination processes of PbS quantum dots sensitized solar cells. Superlattices Microstruct. 2016, 100, 694–702. [Google Scholar] [CrossRef]
- Nanda, K.K.; Kruis, F.E.; Fissan, H.; Behera, S.N. Effective mass approximation for two extreme semiconductors: Band gap of PbS and CuBr nanoparticles. J. Appl. Phys. 2004, 95, 5035–5041. [Google Scholar] [CrossRef]
- Shkir, M.; Chandekar, K.V.; Hossain, M.; Palanivel, B.; Ahmad, N.; Ashraf, I.; Somaily, H.; Algarni, H.; AlFaify, S. Enhanced dielectric and electrical properties of PbS nanostructures facilely synthesized by low-cost chemical route: An effect of Ce doping concentrations. Mater. Chem. Phys. 2021, 278, 125626. [Google Scholar] [CrossRef]
- Elisa, M.; Iordache, S.-M.; Iordache, A.-M.; Rusu, M.I.; Socol, G.; Filipescu, M.; Bartha, C.; Enculescu, M. Pulsed Laser Deposition Films Based on CdSe-Doped Zinc Aluminophosphate Glass. JOM 2021, 73, 495–503. [Google Scholar] [CrossRef]
- Elisa, M.; Stefan, R.C.; Vasiliu, I.C.; Iordache, S.M.; Iordache, A.-M.; Sava, B.A.; Boroica, L.; Dinca, M.C.; Filip, A.V.; Galca, A.C.; et al. A New Zinc Phosphate-Tellurite Glass for Magneto-Optical Applications. Nanomaterials 2020, 10, 1875. [Google Scholar] [CrossRef]
- Shimakawa, K.; Singh, J.; O’Leary, S.K. Optical Properties of Disordered Condensed Matter. In Optical Properties of Materials and Their Applications, 2nd ed.; Singh, J., Ed.; Wiley: Darwin, Australia, 2020. [Google Scholar]
- Al-Saadi, T.M.; Hussein, B.H.; Hasan, A.B.; Shehab, A. Study the Structural and Optical Properties of Cr doped SnO2 Nanoparticles Synthesized by Sol-Gel Method. Energy Procedia 2019, 157, 457–465. [Google Scholar] [CrossRef]
- Pugh-Thomas, D.; Walsh, B.M.; Gupta, M.C. CdSe(ZnS) nanocomposite luminescent high temperature sensor. Nanotechnology 2011, 22, 185503. [Google Scholar] [CrossRef] [PubMed]
- Suganya, M.; Balu, A. PbS nanopowder–synthesis, characterization and antimicrobial activity. Mater. Sci. 2017, 35, 322–328. [Google Scholar] [CrossRef]
- Shapter, J.G.; Brooker, M.H.; Skinner, W.M. Observation of the oxidation of galena using Raman spectroscopy. Int. J. Miner. Process. 2000, 60, 199–211. [Google Scholar] [CrossRef]
- White, W.B.; Minser, D.G. Raman spectra and structure of natural glasses. J. Non-Cryst. Solids 1984, 67, 45–59. [Google Scholar] [CrossRef]
- González, P.; Serra, J.; Liste, S.; Chiussi, S.; León, B.; Pérez-Amor, M. Raman spectroscopic study of bioactive silica based glasses. J. Non-Cryst. Solids 2003, 320, 92–99. [Google Scholar] [CrossRef]
- Yadav, A.K.; Singh, P. A review of the structures of oxide glasses by Raman spectroscopy. RSC Adv. 2015, 5, 67583–67609. [Google Scholar] [CrossRef]
- Tung, S. High proton conductive glass electrolyte synthesized by an accelerated sol–gel process with water/vapor management. J. Membr. Sci. 2004, 241, 315–323. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nicoara, A.I.; Eftimie, M.; Elisa, M.; Vasiliu, I.C.; Bartha, C.; Enculescu, M.; Filipescu, M.; Aguado, C.E.; Lopez, D.; Sava, B.A.; et al. Nanostructured PbS-Doped Inorganic Film Synthesized by Sol-Gel Route. Nanomaterials 2022, 12, 3006. https://doi.org/10.3390/nano12173006
Nicoara AI, Eftimie M, Elisa M, Vasiliu IC, Bartha C, Enculescu M, Filipescu M, Aguado CE, Lopez D, Sava BA, et al. Nanostructured PbS-Doped Inorganic Film Synthesized by Sol-Gel Route. Nanomaterials. 2022; 12(17):3006. https://doi.org/10.3390/nano12173006
Chicago/Turabian StyleNicoara, Adrian Ionut, Mihai Eftimie, Mihail Elisa, Ileana Cristina Vasiliu, Cristina Bartha, Monica Enculescu, Mihaela Filipescu, César Elosúa Aguado, Diego Lopez, Bogdan Alexandru Sava, and et al. 2022. "Nanostructured PbS-Doped Inorganic Film Synthesized by Sol-Gel Route" Nanomaterials 12, no. 17: 3006. https://doi.org/10.3390/nano12173006
APA StyleNicoara, A. I., Eftimie, M., Elisa, M., Vasiliu, I. C., Bartha, C., Enculescu, M., Filipescu, M., Aguado, C. E., Lopez, D., Sava, B. A., & Oane, M. (2022). Nanostructured PbS-Doped Inorganic Film Synthesized by Sol-Gel Route. Nanomaterials, 12(17), 3006. https://doi.org/10.3390/nano12173006












