Microstructural Study of MgB2 in the LiBH4-MgH2 Composite by Using TEM
Abstract
:1. Introduction
2. Experimental Section
2.1. Material Preparations
2.2. Kinetic Measurements
2.3. XRD Experiments
2.4. TEM Experiments
3. Results and Discussion
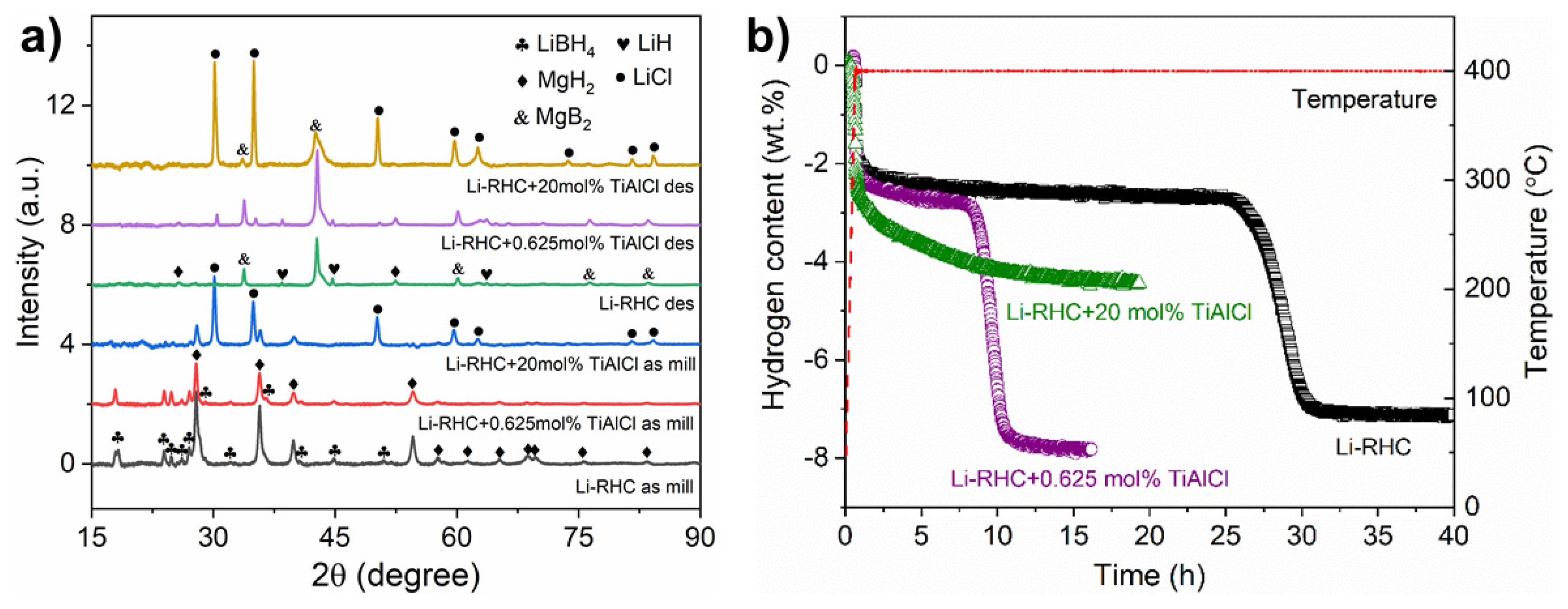
3.1. Material Characterization via XRD and Kinetic Performance
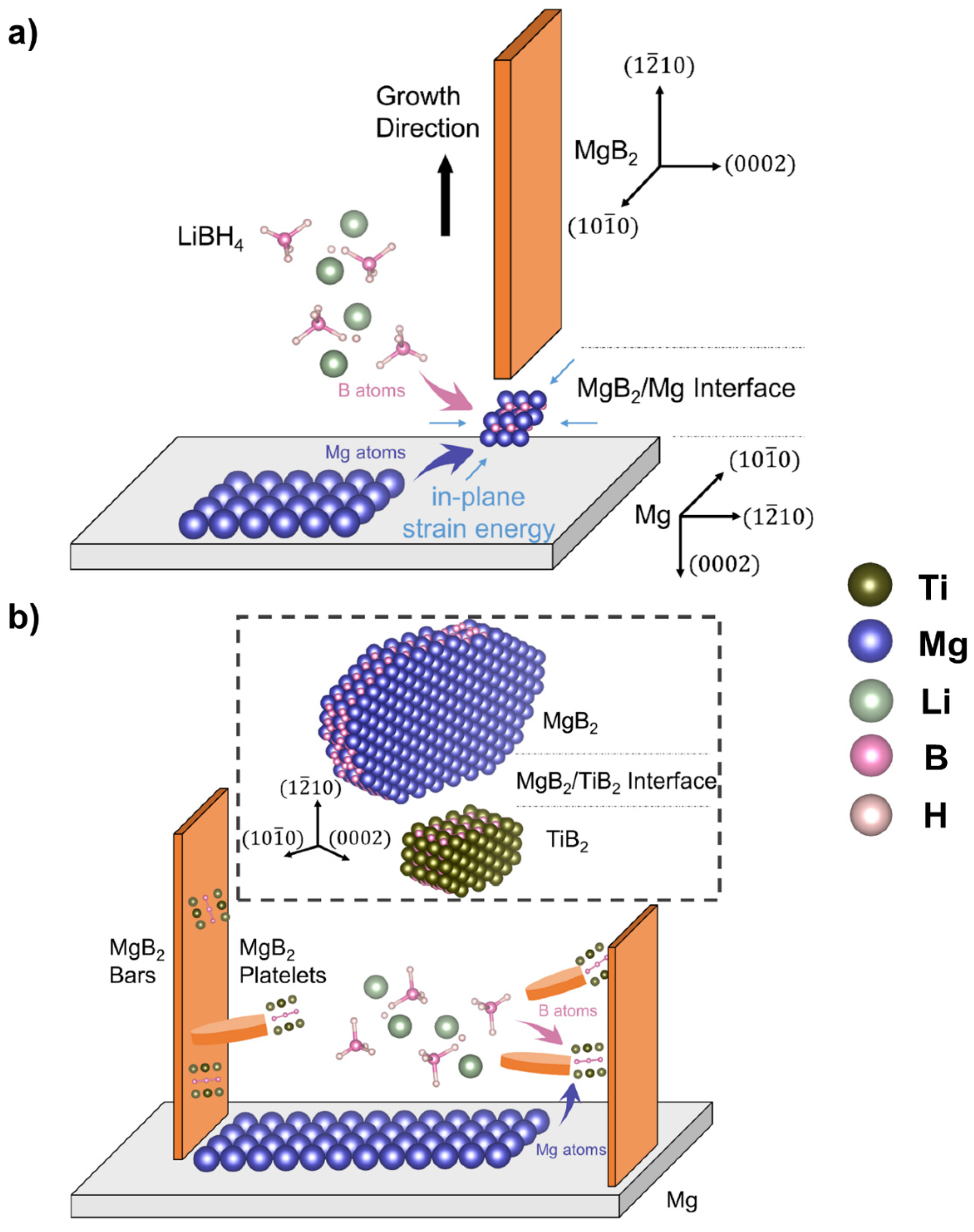
3.2. MgB2 Formation without the Influence of Additives
3.3. MgB2 Formation under the Influence of Additives
3.4. Analysis of Orientation Relationships
3.5. Varied Morphology and Kinetics
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Milanese, C.; Jensen, T.; Hauback, B.; Pistidda, C.; Dornheim, M.; Yang, H.; Lombardo, L.; Zuettel, A.; Filinchuk, Y.; Ngene, P. Complex hydrides for energy storage. Int. J. Hydrog. Energy 2019, 44, 7860–7874. [Google Scholar] [CrossRef] [Green Version]
- Chen, P.; Xiong, Z.; Luo, J.; Lin, J.; Tan, K.L. Interaction of hydrogen with metal nitrides and imides. Nature 2002, 420, 302–304. [Google Scholar] [CrossRef]
- Barkhordarian, G.; Klassen, T.; Dornheim, M.; Bormann, R. Unexpected kinetic effect of MgB2 in reactive hydride composites containing complex borohydrides. J. Alloy. Compd. 2007, 440, L18–L21. [Google Scholar] [CrossRef]
- Vajo, J.J.; Salguero, T.T.; Gross, A.F.; Skeith, S.L.; Olson, G.L. Thermodynamic destabilization and reaction kinetics in light metal hydride systems. J. Alloy. Compd. 2007, 446, 409–414. [Google Scholar] [CrossRef]
- Hauback, B.C. Task 22 of IEA HIA—Fundamental and Applied Hydrogen Storage Materials Development. In 18th World Hydrogen Energy Conference 2010-WHEC 2010; Detlef Stolten, T.G., Ed.; Forschungszentrum Juelich GmbH: Essen, Germany, 2010. [Google Scholar]
- Bösenberg, U.; Doppiu, S.; Mosegaard, L.; Barkhordarian, G.; Eigen, N.; Borgschulte, A.; Jensen, T.R.; Cerenius, Y.; Gutfleisch, O.; Klassen, T. Hydrogen sorption properties of MgH2–LiBH4 composites. Acta Mater. 2007, 55, 3951–3958. [Google Scholar] [CrossRef]
- Mauron, P.; Buchter, F.; Friedrichs, O.; Remhof, A.; Bielmann, M.; Zwicky, C.N.; Züttel, A. Stability and reversibility of LiBH4. J. Phys. Chem. B 2008, 112, 906–910. [Google Scholar] [CrossRef] [PubMed]
- Orimo, S.-I.; Nakamori, Y.; Kitahara, G.; Miwa, K.; Ohba, N.; Towata, S.-I.; Züttel, A. Dehydriding and rehydriding reactions of LiBH4. J. Alloy. Compd. 2005, 404, 427–430. [Google Scholar] [CrossRef]
- Vajo, J.J.; Olson, G.L. Hydrogen storage in destabilized chemical systems. Scr. Mater. 2007, 56, 829–834. [Google Scholar] [CrossRef]
- Vajo, J.J.; Skeith, S.L.; Mertens, F. Reversible storage of hydrogen in destabilized LiBH4. J. Phys. Chem. B 2005, 109, 3719–3722. [Google Scholar] [CrossRef]
- Bösenberg, U.; Kim, J.W.; Gosslar, D.; Eigen, N.; Jensen, T.R.; von Colbe, J.B.; Zhou, Y.; Dahms, M.; Kim, D.; Günther, R. Role of additives in LiBH4–MgH2 reactive hydride composites for sorption kinetics. Acta Mater. 2010, 58, 3381–3389. [Google Scholar] [CrossRef] [Green Version]
- Bösenberg, U.; Ravnsbæk, D.B.; Hagemann, H.; D’Anna, V.; Minella, C.B.; Pistidda, C.; van Beek, W.; Jensen, T.R.; Bormann, R.d.; Dornheim, M. Pressure and temperature influence on the desorption pathway of the LiBH4− MgH2 composite system. J. Phys. Chem. C 2010, 114, 15212–15217. [Google Scholar] [CrossRef]
- Gross, A.F.; Vajo, J.J.; van Atta, S.L.; Olson, G.L. Enhanced hydrogen storage kinetics of LiBH4 in nanoporous carbon scaffolds. J. Phys. Chem. C 2008, 112, 5651–5657. [Google Scholar] [CrossRef]
- Gosalawit–Utke, R.; Thiangviriya, S.; Javadian, P.; Laipple, D.; Pistidda, C.; Bergemann, N.; Horstmann, C.; Jensen, T.R.; Klassen, T.; Dornheim, M. Effective nanoconfinement of 2LiBH4–MgH2 via simply MgH2 premilling for reversible hydrogen storages. Int. J. Hydrog. Energy 2014, 39, 15614–15626. [Google Scholar] [CrossRef]
- Huang, X.; Xiao, X.; Shao, J.; Zhai, B.; Fan, X.; Cheng, C.; Li, S.; Ge, H.; Wang, Q.; Chen, L. Building robust architectures of carbon-wrapped transition metal nanoparticles for high catalytic enhancement of the 2LiBH4-MgH2 system for hydrogen storage cycling performance. Nanoscale 2016, 8, 14898–14908. [Google Scholar] [CrossRef]
- Deprez, E.; Justo, A.; Rojas, T.; López-Cartés, C.; Minella, C.B.; Bösenberg, U.; Dornheim, M.; Bormann, R.; Fernández, A. Microstructural study of the LiBH4–MgH2 reactive hydride composite with and without Ti-isopropoxide additive. Acta Mater. 2010, 58, 5683–5694. [Google Scholar] [CrossRef] [Green Version]
- Fan, M.-Q.; Sun, L.-X.; Zhang, Y.; Xu, F.; Zhang, J.; Chu, H.-l. The catalytic effect of additive Nb2O5 on the reversible hydrogen storage performances of LiBH4–MgH2 composite. Int. J. Hydrog. Energy 2008, 33, 74–80. [Google Scholar] [CrossRef]
- Le, T.-T.; Pistidda, C.; Puszkiel, J.n.; Riglos, M.a.V.C.; Karimi, F.; Skibsted, J.; GharibDoust, S.P.; Richter, B.; Emmler, T.; Milanese, C. Design of a nanometric AlTi additive for MgB2-based reactive hydride composites with superior kinetic properties. J. Phys. Chem. C 2018, 122, 7642–7655. [Google Scholar] [CrossRef] [Green Version]
- Kremer, J.R.; Mastronarde, D.N.; McIntosh, J.R. Computer visualization of three-dimensional image data using IMOD. J. Struct. Biol. 1996, 116, 71–76. [Google Scholar] [CrossRef] [Green Version]
- Gilbert, P. Iterative methods for the three-dimensional reconstruction of an object from projections. J. Theor. Biol. 1972, 36, 105–117. [Google Scholar] [CrossRef]
- Wold, S.; Esbensen, K.; Geladi, P. Principal component analysis. Chemom. Intell. Lab. Syst. 1987, 2, 37–52. [Google Scholar] [CrossRef]
- Abdi, H.; Williams, L.J. Principal component analysis. Wiley Interdiscip. Rev. Comput. Stat. 2010, 2, 433–459. [Google Scholar] [CrossRef]
- Lee, S. Crystal growth of MgB2. Phys. C Supercond. 2003, 385, 31–41. [Google Scholar] [CrossRef]
- Kong, X.; Wang, Y.; Li, H.; Duan, X.; Yu, R.; Li, S.; Li, F.; Jin, C. Electron energy-loss spectroscopy characterization of the boron p-like density of states in MgB2. Appl. Phys. Lett. 2002, 80, 778–780. [Google Scholar] [CrossRef]
- Deprez, E.; Munoz-Márquez, M.A.; Roldán, M.A.; Prestipino, C.; Palomares, F.J.; Minella, C.B.; Bosenberg, U.; Dornheim, M.; Bormann, R.; Fernández, A. Oxidation state and local structure of Ti-based additives in the reactive hydride composite 2LiBH4+ MgH2. J. Phys. Chem. C 2010, 114, 3309–3317. [Google Scholar] [CrossRef] [Green Version]
- Fan, X.; Xiao, X.; Chen, L.; Wang, X.; Li, S.; Ge, H.; Wang, Q. High catalytic efficiency of amorphous TiB2 and NbB2 nanoparticles for hydrogen storage using the 2LiBH4–MgH2 system. J. Mater. Chem. A 2013, 1, 11368–11375. [Google Scholar] [CrossRef]
- Zhang, M.-X.; Kelly, P. Edge-to-edge matching model for predicting orientation relationships and habit planes—the improvements. Scr. Mater. 2005, 52, 963–968. [Google Scholar] [CrossRef]
- Zhang, M.-X.; Kelly, P. Edge-to-edge matching and its applications: Part II. Application to Mg–Al, Mg–Y and Mg–Mn alloys. Acta Mater. 2005, 53, 1085–1096. [Google Scholar] [CrossRef]
- Zhang, M.-X.; Kelly, P.M.; Easton, M.A.; Taylor, J.A. Crystallographic study of grain refinement in aluminum alloys using the edge-to-edge matching model. Acta Mater. 2005, 53, 1427–1438. [Google Scholar] [CrossRef]
- Kelly, P.; Zhang, M.-X. Edge-to-edge matching—The fundamentals. Metall. Mater. Trans. A 2006, 37, 833–839. [Google Scholar] [CrossRef]
- Yang, J.; Wang, J.; Wu, Y.; Wang, L.; Zhang, H. Extended application of edge-to-edge matching model to HCP/HCP (α-Mg/MgZn2) system in magnesium alloys. Mater. Sci. Eng. A 2007, 460, 296–300. [Google Scholar] [CrossRef]




| MgB2/Mg | ||||
|---|---|---|---|---|
| 39.8 | 48.6 | 58.4 | 8.5 |
| MgB2/Mg | ||||
|---|---|---|---|---|
| −4.2 | 2.1 | −80.8 | −69.5 |
| MgB2/MB2 | ||||
|---|---|---|---|---|
| TiB2 | −15.8 | 8.2 | −49.2 | 13.9 |
| AlB2 | −15.5 | 7.6 | −47.9 | 14.6 |
| MgB2/MB2 | ||||
|---|---|---|---|---|
| TiB2 | 1.7 | −70.3 | 43.3 | 1.7 |
| AlB2 | 2.6 | −68.7 | 43.8 | 2.6 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jin, O.; Shang, Y.; Huang, X.; Mu, X.; Szabó, D.V.; Le, T.T.; Wagner, S.; Kübel, C.; Pistidda, C.; Pundt, A. Microstructural Study of MgB2 in the LiBH4-MgH2 Composite by Using TEM. Nanomaterials 2022, 12, 1893. https://doi.org/10.3390/nano12111893
Jin O, Shang Y, Huang X, Mu X, Szabó DV, Le TT, Wagner S, Kübel C, Pistidda C, Pundt A. Microstructural Study of MgB2 in the LiBH4-MgH2 Composite by Using TEM. Nanomaterials. 2022; 12(11):1893. https://doi.org/10.3390/nano12111893
Chicago/Turabian StyleJin, Ou, Yuanyuan Shang, Xiaohui Huang, Xiaoke Mu, Dorothée Vinga Szabó, Thi Thu Le, Stefan Wagner, Christian Kübel, Claudio Pistidda, and Astrid Pundt. 2022. "Microstructural Study of MgB2 in the LiBH4-MgH2 Composite by Using TEM" Nanomaterials 12, no. 11: 1893. https://doi.org/10.3390/nano12111893
APA StyleJin, O., Shang, Y., Huang, X., Mu, X., Szabó, D. V., Le, T. T., Wagner, S., Kübel, C., Pistidda, C., & Pundt, A. (2022). Microstructural Study of MgB2 in the LiBH4-MgH2 Composite by Using TEM. Nanomaterials, 12(11), 1893. https://doi.org/10.3390/nano12111893










