Abstract
Graphene nanoribbons are a type of graphene characterized by remarkable electrical and mechanical properties. This review considers the prospects for the application of graphene ribbons in biomedicine, taking into account safety aspects. According to the analysis of the recent studies, the topical areas of using graphene nanoribbons include mechanical, chemical, photo- and acoustic sensors, devices for the direct sequencing of biological macromolecules, including DNA, gene and drug delivery vehicles, and tissue engineering. There is evidence of good biocompatibility of graphene nanoribbons with human cell lines, but a number of researchers have revealed toxic effects, including cytotoxicity and genotoxicity. Moreover, the damaging effects of nanoribbons are often higher than those of chemical analogs, for instance, graphene oxide nanoplates. The possible mechanism of toxicity is the ability of graphene nanoribbons to damage the cell membrane mechanically, stimulate reactive oxidative stress (ROS) production, autophagy, and inhibition of proliferation, as well as apoptosis induction, DNA fragmentation, and the formation of chromosomal aberrations. At the same time, the biodegradability of graphene nanoribbons under the environmental factors has been proven. In general, this review allows us to conclude that graphene nanoribbons, as components of high-precision nanodevices and therapeutic agents, have significant potential for biomedical applications; however, additional studies of their safety are needed. Particular emphasis should be placed on the lack of information about the effect of graphene nanoribbons on the organism as a whole obtained from in vivo experiments, as well as about their ecological toxicity, accumulation, migration, and destruction within ecosystems.
1. Introduction
Graphene nanoribbons (GNRs) are narrow strips of graphene composed of repeating hexagonal carbon cells, up to 50 nm wide and up to several dozens of micrometers long, depending on the synthesis method [1]. Graphene ribbons were theoretically described in 1996 by Fuhita et al. as a model for studying the edge and nanoscale effects of graphene [2,3]. Due to their quasi-one-dimensional nature, GNRs differ significantly from the more widely known two-dimensional graphene sheets [4]. Being almost ideal nanowires or nanotags, GNRs are an extremely accurate tool, promising for nanoelectronic components, ultrasensitive chemical and mechanical sensors, etc. [5,6,7,8,9,10].
Moreover, the structure and physical properties of GNRs vary significantly, depending on the synthesis method. In fact, GNRs obtained via different methods are very different and have little in common. Today, three main approaches to obtaining GNRs exist [11]:
1. Electron-beam lithography and photolithography: This method makes it possible to obtain single-layer GNRs on the substrate surface; however, the fields of application of lithographically obtained GNRs are very limited. In addition, the ribbons obtained by the lithographic method have jagged edges [12,13,14].
2. Bottom-up synthesis from polycyclic molecules: This method includes multistage organic synthesis based on the cyclization of previously synthesized polymer chains. This method provides very narrow ribbons with an atomically precise edge configuration [15,16,17,18].
3. Unwrapping carbon nanotubes (CNTs): The third approach is based on longitudinal opening or cutting of multiwalled carbon nanotubes (MCNTs) [19,20]. This method produces GNRs with controlled width and well-defined edge structures [21].
Typical characteristics of GNRs are determined by transmission electron microscopy (TEM), atomic force microscopy (AFM), Raman-scattering spectroscopy, and X-ray photoelectron spectroscopy (XPS).
As previously noted, GNRs can be considered as quasi-one-dimensional graphene strips. This makes them similar to a large class of conjugated polymers, for which the synthesis conditions and the method of film formation determine the performance characteristics [22,23]. The structural perfection of GNRs is a significant problem, because their electronic and optical properties critically depend on the edge configuration, width, and direction of the crystal [24]. The three most commonly studied types of GNRs edge structures are “armchair” or AGNRs, “zigzag” or GNRs with zigzag edges (ZGNRs), and “cove” (Figure 1) [25,26].
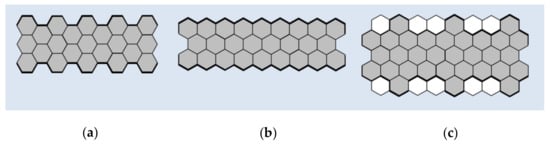
Figure 1.
Types of the edge structures of graphene nanoribbons (GNRs): (a) “armchair”; (b) “zigzag”; (c) “cove”.
GNRs with armchair-type and zigzag-type edges are the most common ones. AGNRs are characterized by a wide band gap that varies with width, while ZGNRs are predicted to have smaller bandgaps with localized edge states that are magnetic and show great potential for spintronic applications [27,28,29,30]. Modifications to the edges of cove-type GNRs are able to smoothly reduce energy bandgaps at the expense of losses in conjugation and increased morphological spreading [31].
Thus, to take full advantage of the exceptional characteristics of GNRs for practical applications, it is necessary to take into account the fabrication method and the type of spatial structure.
2. GNRs in Biomedicine
Remarkable electrical and mechanical properties of GNRs make them a promising material for biological and medical applications [32,33,34,35,36]. However, the widespread use of GNRs requires the careful analysis of the potential toxicity of these materials, both through intentional and accidental exposure.
According to the available information, GNRs in biomedicine are mainly used to create various ultra-small devices such as molecular sensors, photo-, thermo-, and acoustic detectors, sequencers, drug and gene delivery vehicles, and tissue engineering constructions (Figure 2).
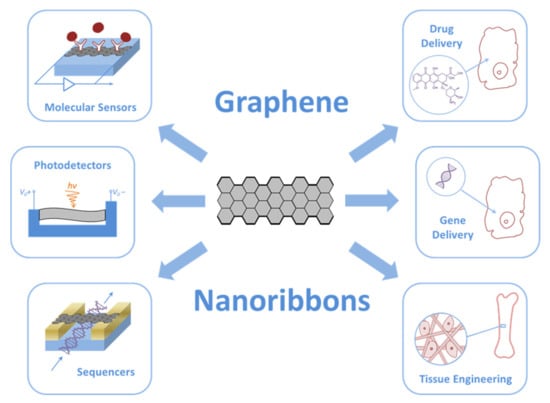
Figure 2.
Biomedical applications of GNRs.
2.1. Electronic and Biomedical Devices
The research works aimed at the integration of GNRs into devices are going in several directions, but all of these devices require connections and heterojunctions [22].
Cai et al. successfully manufactured heterojunctions of GNRs with heterostructures by means of combining pristine hydrocarbon precursors with their nitrogen-substituted equivalents. The obtained heterostructures consisted of seamlessly assembled segments of pristine GNRs (p-GNRs) and deterministically nitrogen-doped GNRs (N-GNRs), which behave similarly to traditional p–n junctions. With a band shift of 0.5 eV and an electric field of 2 × 108 V/m at the heterojunction, these materials are applicable for the photovoltaic industry and electronics [37].
The bottom-up synthesis allows obtaining homogeneous GNRs with an extremely narrow width (less than 1 nm) and an atomically perfect edge structure. GNRs of this type are well suited for a wide variety of electronic devices. Bennett et al. developed a reliable layer transfer process for creating nanoscale GNR field-effect transistors by virtue of chemical synthesis. The researchers proved the high sensitivity and good electrical characteristics of the developed GNRs [38].
The studies [39,40,41,42] were aimed at the devices with integrated GNRs, namely photodetectors and sensors. For instance, Johnson et al. obtained hydrogen sensors from Pd-functionalized multilayer networks based on GNRs [42]. The manufacturing method of these networks is cheap and scalable. The developed networks are characterized by a high specific surface area, so they can be used for functionalization and gas adsorption. According to the research results, the networks had high sensitivity to hydrogen at ppm concentration levels at room temperature with fast response and recovery time.
The basic mechanism in a photodetector is the separation of photo-excited electrons and holes. However, several distinct separation methods have already been discovered, all of which are based on using a longitudinal electric field. Zarei and Sharifi have proposed a novel method based on using a vertical electric field, which induces an asymmetric potential barrier in front of one or both of photo-excited carriers [43]. At the initial stage, the authors used a simple one-dimensional model consisting of a single one-dimensional chain of atoms. The model took into account many aspects, including the influence of the location, height, and width of the potential barrier. For real applications, a new structure based on GNRs and an asymmetric metal gate was developed (Figure 3). The results of the study showed that this structure provides the corresponding separation of carriers.
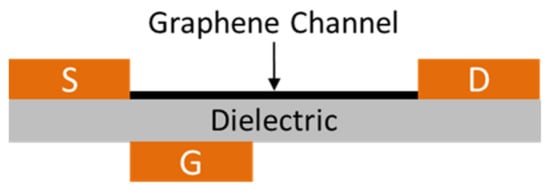
Figure 3.
The scheme of the structure of the novel GNR photodetector with one asymmetric metal gate. The width and length of the nanoribbon are 1.35 and 6 nm, respectively. The width of the metal gate is 1/5 of the length of the GNR channel. Adapted from [43].
In the study [44], it has been revealed that the encapsulation of the deposited HfO2 on an atomic layer of GNRs significantly increases the mobility of charge carriers and decreases their scattering of their nanoribbons, since the dielectric layer weakens the coulomb interactions of the carriers. In addition, a photodetector based on GNRs coated with a HfO2 layer can cover broadband waves from the visible to mid-infrared range at room temperature, demonstrating 10 times higher sensitivity than the one without a HfO2 layer in the visible mode and 8 times higher sensitivity in the mid-infrared mode.
DNA detection has received considerable attention in recent years due to promising applications in diagnostics and treatment, forensic science, food safety assessment, etc. Therefore, molecular diagnostic systems for the detection of DNA with high sensitivity and specificity have an enormous potential. For example, solid-state nanopores can act as single-molecule sensors and can potentially be used for quickly sequencing DNA molecules. However, nanopores are usually made in insulating membranes with the thickness up to 15 bases, which makes it difficult to sequence individual bases by such devices. At the same time, graphene has a thickness of only 0.335 nm (equivalent to the distance between two bases in the DNA chain) that provides a suitable membrane for sequencing [45,46,47,48,49].
Nelson et al. first studied the DNA translocations using in-plane current signals in GNRs [50]. The authors analyzed the spectra of conductivity and charge density in the presence of different nucleobases inside graphene nanopores and proved that this device can distinguish four different bases. Meanwhile, the conductivity spectrum of nucleotides is little affected by its orientation inside the nanopores. The proposed technique can be extremely useful for real applications in ultrafast and inexpensive DNA sequencing methods.
The first experimental data on DNA translocation through GNR nanopores was reported by Traversi et al. [51]. It was found that a solid-state nanopore could be integrated with a GNR transistor to obtain a sensor for DNA translocation. Figure 4 shows the diagram of the proposed device. As DNA molecules move through the pores, the device can simultaneously measure the ion current drop and local voltage changes in the transistor, which can be used to detect molecules.
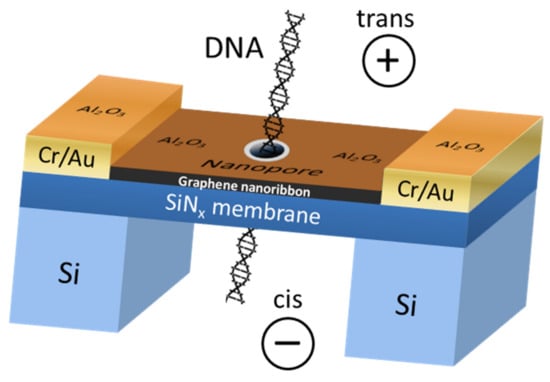
Figure 4.
The scheme of the DNA translocation sensor based on GNRs. Adapted from [51].
When using GNRs based on SiNx membranes, it was shown that the passage of DNA through a nanopore in graphene significantly changes the conductivity of paired nanoribbons. This can be explained by the strong π–π interaction between the atoms of carbon, graphene, and nucleotides in the DNA molecules [52]. Nevertheless, an improved signal-to-noise ratio of graphene electrodes for sequencing single nucleotides is required [53].
Many theoretical studies have predicted that DNA sequencing may be realized by monitoring the transverse current through GNRs while a DNA molecule is translocated through nanopores in ribbons. This type of sequencing benefits from the special transport properties of graphene, provides an ultimate spatial resolution due to the graphene thickness in the monatomic layer and facilitates high-throughput measurements. In the previous experimental attempts to measure such in-plane transverse signals, trivial capacitive response has prevailed. To solve this problem, Heerema et al. [54] used a differential current amplifier, which discriminates between a capacitive current signal and a resistive response in graphene. The authors used short and narrow (30 nm × 30 nm) nanoribbons with 5 nm nanopores obtained using a high-temperature scanning transmission electron microscope to preserve the crystallinity and sensitivity of graphene. It was shown that resistive modulations could be observed in the graphene current due to the DNA translocation through nanopores, thus demonstrating the possibility of measuring DNA using in-plane currents. However, it is worth noting that this approach is challenging due to the low yields in the fabrication of devices associated with a complex multistage device layout.
Puster et al. [55] obtained nanoporous sensors based on graphene nanoribbons (GNR–NP). Pores with a diameter of 2–10 nm were formed at the edge or in the center of a GNR with a width of 20 to 250 nm and a length of 600 nm on a 40 nm silicon nitride substrate. The GNR conductivity was monitored in situ during electron irradiation-induced nanopore formation by a transmission electron microscope operating at 200 kV. It was shown that the GNR resistance increased linearly with increasing electron dose and that conductivity and mobility of GNRs decreased by a factor of 10 or more when GNR images were displayed at a relatively high magnification with a wide beam before creating a nanopore. The TEM analysis in scanning mode (STEM) allows controlling the position of the convergent electron beam with a high spatial accuracy through automatic feedback. Using the STEM mode prevented damage caused by the electron beam that made it possible to create nanopores in the highly conductive GNR sensors. The method used minimizes the effect of GNRs on the beam before and during the formation of nanopores. The resulting GNRs with constant resistances after nanopore formation can withstand microampere currents at low voltages (~50 mV) in a buffered electrolyte solution and exhibit high sensitivity, with a large relative resistance change with gate voltage changes, similar to the original GNRs without nanopores.
In their later work [56], Puster et al. described the scheme (Figure 5) and the technique for measuring ion currents using nanopores, including GNRs up to 50 nm wide and up to 600 nm long, providing a stable ion current linearly dependent on voltage in the open state.
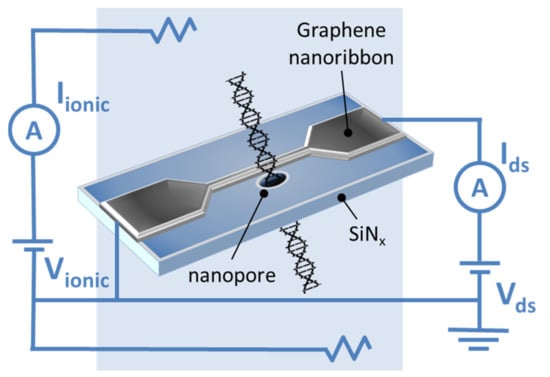
Figure 5.
The scheme of the GNR–nanopore structure during DNA translocation. Reproduced with permission from [56]. Copyright John Wiley and Sons, 2015.
Puster et al. reported that DNA translocation through nanopores occurs when currents from several hundred of nanoamperes to 2 μA at low drain-source voltage (Vds) values (<100 mV) are applied. The DNA translocation modulates both the ionic current through the nanopores and the electronic conductivity of the nanoribbon. Since the ionic signal during DNA translocation usually is a rectangular pulse, the GNR signal can be represented as up- and down-current bursts that occur at the beginning and end of the ion signal. This time-derivative signal is the result of the capacitive coupling between the measurement channels. Crosstalk is not scaled by Vds on the device and is present for the measurements at both high salt (1 M KCl) and low salt (10 mM KCl) concentrations [56].
Saha et al. [57] studied a two-component device for DNA sequencing. The investigated device consists of a metallic ZGNR and a nanopore in its inner part, through which a DNA molecule moves. Using the non-equilibrium Green function method in combination with the density functional theory, it was shown that each of the four DNA nucleic bases inserted into nanopores, in which the edge carbon atoms are passivated by hydrogen or nitrogen, leads to a unique change in the device conductivity. The other recent biosensors based on transverse electron transport through translocated DNA use a small (within the order of picoampere) tunneling current through a nanostructure or nanopore, giving a low signal-to-noise ratio. Contrastingly, the presented device concept is based on the fact that the local-current-density ZGNR reaches a peak at the edges, so drilling the nanopores from the edges will not result in a decrease in conductivity. The insertion of a nucleobase into the nanopore affects the charge density in the surrounding area, thereby modulating the edge conduction currents, the magnitude of which is approximately a microampere at a bias voltage of 0.1 V.
GNRs by functionalized nucleotides are considered as a tool for DNA sequencing in aqueous suspensions. Paulechka et al. [58] proposed a method based on the formation of hydrogen bonds between the nitrogenous bases of the probe and the analyte according to the principle of complementarity. In this case, the selective pairing of nucleotides and the ability of graphene to transform anisotropic deformation of the crystal lattice into changes in the electric current at the nanoscale are combined. Using the methods of modeling atomic and molecular dynamics, the authors estimated the levels of measurable changes in the electrical signal of the nanosensor in response to deformations, taking into account the influence of the environment. From the results, it was concluded that the proposed approach is very promising for the design of DNA sensor devices, allowing obtaining and processing big data with good noise immunity.
GNRs-based electrodes decorated with iron oxide nanoparticles (GNR–Fe3O4) amplify electrochemical signals by more than one order of magnitude compared to bare carbon electrodes and 70% more compared to p-GNRs-based electrodes [59]. The electrochemical currents in immobilized single-stranded DNA and double-stranded DNA are 92 and 49 μA, respectively. These values are indicative of an effective discrimination between the immobilization and hybridization of DNA.
An electrochemical biosensor on the basis of gold nanocages (AuNCs) and GNRs (AuNCs@GNRs) was developed by Feng et al. [60]. The biosensor is a so-called DNA walker that stochastically moves along the surface, depending on the presence of DNA targets. Due to the large surface area and high conductivity of the biosensor components, its sensitivity is improved. The processes of conjugation and the dissociation of the target DNA with the sensor are more efficient, which leads to a significant amplification of the signal. The proposed biosensor based on AuNCs@GNRs demonstrates excellent characteristics for DNA analysis in complex matrices, such as human serum, which opens up prospects for the practical application of a new sensor platform.
Another example of the use of GNRs in biosensors is described in the study [61]. Mehmeti et al. developed an electrochemical biosensor of glucose based on the composition of enzyme glucose oxidase (GOx) with GNRs, which does not require reagents. Nanoribbons are used for the direct transfer of electrons between the coenzyme, flavine adenine dinucleotide (FAD), and the electrode. The method includes the following stages: purification of GOx by separation from the coenzyme; obtaining GNRs by the oxidative cutting of MCNTs; immobilization of GNRs on the surface of the carbon electrode using screen printing; covalent attachment of FAD to nanoribbons; recombination of the enzyme with covalently linked FAD; stabilization of the bilayer using a thin polymer electrolyte membrane. The resulting biosensor shows operability when using a potential of +0.475 V, and the signals are linearly proportional to the glucose concentration in the range from 50 to 2000 mg/L with a detection limit of 20 mg/L. The biosensor was used to determine glucose in human serum, with a high level of repeatability and reproducibility of the results.
By employing hybrid plasmonic sensing platform with GNR/Ag nanoparticles for the sequential colorimetric detection of dopamine (DA) and glutathione (GSH) in human serum samples was proposed. DA and GSH were successfully detected in low concentrations of 0.04 and 0.23 μM, respectively [62].
Sainz et al. employed chevron-like GNRs to develop a novel electrochemical epinephrine sensor [63]. Pulse voltammetry methods obtain a linear concentration range from 6.4 × 10−6 to 1.0 × 10−4 M and a detection limit of 2.1 × 10−6 M.
Lalwani et al. [64] suggested potential applications of GNRs investigated as contrast agents for photoacoustic and thermoacoustic tomography (TAT). The researchers reported that oxidized single-layer and multiwalled nanoribbons (GO) exhibit a signal amplification for photoacoustic tomography of about 5–10 times compared to lysed bovine blood at a wavelength of 755 nm and ~10–28% signal amplification for thermoacoustic tomography compared to deionized water at a frequency of 3 GHz. Oxidized GNRs are promising in multimodal photoacoustic tomography and TAT contrast agents [65].
Thus, numerous electronic devices based on GNRs have been proposed theoretically and experimentally [46,47,48,66,67,68]. However, the lack of experimental data suggests the need to refine the data on the responses of graphene devices and measure their signals, especially taking into account the previously mentioned differences in the properties of GNRs depending on the methods of fabrication. Table 1 presents potential GNRs applications in biomedical devices.

Table 1.
GNRs in biomedical devices.
2.2. Delivery of Genes and Drugs
The use of drug delivery systems improves the efficacy of many existing drugs and allows the introduction of new therapies. GNRs are an excellent candidate for drug delivery systems [69] and genes delivery [36].
In the study [70], the analysis of electronic and chemical properties of zigzag nanoribbons functionalized with 1-phenylalanine (C9H11NO2) was conducted using the density functional theory. In particular, properties such as the band gap, charge transfer, chemical potential, dipole moment, bond length and energy, and the characteristic of the s-orbital were investigated. Differences depending on the selection of different functionalization sites were determined. The most chemically active and stable variants in the aquatic environment were identified. This study forms the theoretical basis for the effective use of functionalized nanoribbons as drug delivery vehicles.
Chowdhury et al. [71] studied oxidized GNRs (O-GNRs) as non-viral vectors for gene therapy. It was shown that O-GNRs could load large amounts of small single-stranded or double-stranded genetic materials without additional functionalization with positively charged groups or other non-viral vectors. The used O-GNRs doses of 20–60 µg/mL had lower cytotoxicity compared to commercial vectors for the delivery of non-viral genes. The O-GNRs–plasmid-DNA complexes were absorbed by the vesicular structures of dividing HeLa and HUVEC cells, released into the cytoplasm of the cell and entered the nucleus. In these cells, the O-GNRs–plasmid-DNA complexes increased the efficiency of gene delivery and transfection.
Dong et al. [72] proposed a GNR-based nanocarrier grafted by polyethyleneimine (PEI-g-GNR) as an efficient gene vector. The nanoribbons were synthesized by the longitudinal unpacking of MWCNTs treated subsequently with strong acids and ultrasound in order to obtain surface carboxylic acid groups for PEI grafting by electrostatic assembly. It was assumed that PEI-g-GNR protected locked nucleic acid modified by molecular beacon (LNA-m-MB) probes from nuclease digestion or interaction with a single-stranded binding protein. The cytotoxicity and apoptosis caused by PEI-g-GNR were negligible under optimal transfection conditions. In combination with the high specificity of LNA to microRNA, the developed delivery system was proposed for transferring modified LNA into cells for target RNA recognition.
The study [73] is devoted to the use of O-GNRs coated by PEG-1,2-distearoyl-sn-glycero-3-phosphoethanolamine-N-[amino(polyethylene glycol)] (DSPE) (O-GNR–PEG-DSPE) as an agent for the delivery of antitumor drugs into glioblastoma multiformae cells (U251). The antitumor drug, being an APE-1 endonuclease inhibitor, was applied to the O-GNR–PEG-DSPE complex using a simple non-covalent method. The results showed that the absorption of O-GNR–PEG-DSPE by U251 cells exceeded 67% and the increase in APE-1 expression in cells after 24 h was 60%. The MCF-7 (human breast cancer cell line)- and CG-4 (rat glial progenitor cells)-type cells absorbed 38% and 29% of the used oxidized GNRs, respectively. The TEM analysis of U251 showed large aggregates of O-GNR–PEG-DSPE in the vesicles. The O-GNR–PEG-DSPE functionalized by the antitumor drug was highly toxic for U251, but had low or no toxicity for MCF-7 and CG-4 cells [74].
Ligands, such as peptides, antibodies, or other epitopes, bind and activate specific cellular receptors. These substances are used for the targeted cellular delivery of drugs, genes, and imaging agents. Several studies [75,76] are devoted to the activation of epidermal growth factor receptors (EGFRs) by O-GNRs non-covalently functionalized with PEG-DSPE. This activation generates a predominantly dynamin-dependent macropinocytosis-like response and results in the significant absorption of O-GNR–PEG-DSPE by cells with a high EGFR expression. The authors also reported that cells with an integrated human papillomavirus (HPV) genome show the increased internalization of O-GNR–PEG-DSPE due to the modulation of the EFGR-activated viral protein E5 [68].
Sphingolipids [77,78] can be used as possible anticancer agents [79,80,81] due to their powerful proapoptotic effects. However, due to their extreme hydrophobic properties, there is currently no clinically approved in vivo delivery method for these therapeutic agents. Suhrland et al. [82] developed a new method for loading ceramide C6 onto O-GNRs and graphene nanoplates (GNPs). The mass spectrometry analysis showed loading efficiencies of 57% and 51.5%, respectively. The cytotoxicity analysis showed that at 100 μg/mL ceramide-loaded O-GNRs and GNPs, HeLa cell viability was reduced by 93% and 76% compared to the untreated HeLa cells. It should be noted that nanoparticles at the same concentration without C6 ceramide did not significantly affect cell viability. The authors found that the cytotoxicity had an apoptotic nature. The confocal images of live cells with fluorescent NBD–ceramide ((6-((N-(7-nitrobenz-2-oxa-1,3-diazol-4-yl)amino)hexanoyl)sphingosine)) loaded onto the O-GNRs showed sustained uptake over 30 min.
Thus, GNRs are of interest as gene and drug delivery vectors (Table 2) and possibly also in tissue engineering and bioimaging [33]. However, the high hydrophobicity and toxicity of GNRs require further efforts to increase their biocompatibility in case of in vivo applications.

Table 2.
Delivery of genes and drugs.
3. Biocompatibility and Toxicity
3.1. Biocompatibility
Currently, there are several works in which the cytological effects of GNRs have been investigated. At the same time, very few authors have studied the biocompatibility of GNRs. For example, Liu et al. [84] conducted experiments on the cultivation of human medulloblastoma cells (DAOY) with nitrogen-doped GNRs aerogels (N-GNRA) obtained by self-assembly in combination with thermal annealing. Amino groups were grafted to the surface of GNRs using an epoxy ring-opening reaction. A high level of nitrogen doping (7.6 atm.%) was achieved during heat treatment as a result of functionalization and GNRs edge effects. Three-dimensional N-GNRAs had a hierarchical porous structure. The quasi-one-dimensional GNRs acted as the building blocks for making GNRs mesh sheets, which additionally created three-dimensional wireframes with micrometer-scale pores. The GNRs edge effect combined with nitrogen doping and porosity resulted in good electrical conductivity and superhydrophilicity. The study showed the good biocompatibility of the N-GNRA that opens up its opportunities for biomedicine applications.
MCF-7 exposed to PEG-DSPE-coated O-GNRs (O-GNR–PEG-DSPE) and 80 μM lucanthone-loaded O-GNR–PEG-DSPE do not exhibit decreased viability compared to untreated controls after 24 h exposition [73]. In [85], the authors showed the absence of the toxic effect of O-GNRs noncovalently functionalized with DSPE to the components of the blood vascular system at concentrations of 20 μg/mL. In addition, one-hour exposure to O-GNR–PEG-DSPE did not induce histamine release from mast cells, PF4 activation in platelets, and complement activation. However, at every studied concentration, there was a slight decrease (5–10%) in the levels of anti-inflammatory cytokines.
The analysis of the cytotoxicity of films consisting of chitosan, alginate, and 2.5 wt % GNRs (CHI/f–GNR–ALG) obtained by unzipping MWCNTs on the mouse fibroblasts (L929) showed the sample cytocompatibility. In particular, L929 is able to proliferate on the surface of CHI/f–GNR–ALG (200 µL) [86]. On this basis, the authors suggested that biopolymer films containing GNRs have potential applications for wound healing, as well as the engineering of the heart and bone tissue.
Foreman et al. [87] analyzed the viability of adherent HEK 293T epithelial cells and non-adherent A20 cells incubated with O-GNRs. According to the study, there was no change in cell viability after the 24 h or 48 h incubation of the cells at O-GNRs concentrations up to 100 mg/mL.
The authors of reference [88] investigated the biocompatibility of O-GNRs modified with phospholipid-PEG (PL–PEG) in vivo. Doxorubicin-loaded GNRs has 6.7-fold lower IC50 values for chemo-photothermal therapy toward U87 glioma cells than the IC50 values in traditional chemotherapy. They found that PL–PEG–O-GNRs was excreted from the body via the renal pathway in the urine, and the hematological analysis showed that this nanomaterial was not toxic. The authors consider PL-PEG-O-GNRs as potential nanocarriers of drugs to develop an effective cancer treatment strategy that will not only increase the effectiveness of therapy, but also reduce the risk of side effects of the nanocarrier in the body.
Thus, there is evidence that the cytotoxicity of GNRs can be significantly reduced by their edge or surface chemical modification. For example, doping with nitrogen, oxidation, coating with biocompatible polymers such as PEG or chitosan give an obvious increase in biocompatibility. The hydrophilization of carbon nanomaterials is a versatile strategy to provide their bioavailability and applicability in biomedicine. This is especially important to take into account when developing bioengineering constructs based on GNR, which should have minimal cytotoxicity.
3.2. Toxicity
According to Mbeh et al. [89], O-GNRs synthesized using the oxidative unpacking of MWCNTs and functionalized with albumin at a concentration of 100 μg/mL have high cytotoxicity towards human epithelial cells, causing the inhibition of proliferation and the induction of apoptosis.
The results of the study [90] showed that O-GNRs with PEG-DSPE have heterogeneous cell-specific cytotoxicity. The screening of the O-GNR–PEG-DSPE cytotoxicity was performed using cervical cancer cells (HeLa), mouse fibroblasts (NIH-3T3), as well as breast cancer cells SKBR3 and MCF7. In general, all the cells showed a dose-dependent (10–400 μg/mL) and time (12–48 h) decrease in viability. However, the toxicity for MCF7 or SKBR3 cells was significantly lower than that for HeLa cells. MCF7 and SKBR3 remained 100% viable for up to 48 h for at the O-GNR–PEG-DSPE concentration of 10 μg/mL, and the viability level dropped to ~78% at the maximum concentration of 400 μg/mL. For HeLa, a significant cell death (5–25%) was observed already at a low concentration of 10 μg/mL, and was also observed with an increase in concentration (CD50 ≥ 100 μg/mL). The toxicity to HeLa has been associated with a higher uptake of O-GNR–PEG-DSPE compared to those to other cell types.
The same authors showed that O-GNR–PEG-DSPE at concentrations of 20, 80, and 160 μg/mL caused a low concentration-dependent deformation of erythrocytes, which did not lead to hemolysis [85]. There was also a significant uptake of O-GNR–PEG-DSPE by endothelial cells and a concentration-dependent decrease in their viability. The cytotoxicity of O-GNRs obtained from single-walled CNTs was analyzed on human neuroblastoma cell lines SK-N-BE(2) and SH-SY5Y by assessing cellular reactive oxidative stress, mitochondrial membrane potential, expression of lysosomal proteins, and cell growth [91]. The results showed that O-GNRs at low concentrations increase reactive oxygen species (ROS) production and induce autophagy in both cell lines within hours of exposure; however, these effects are not accompanied by growth arrest or cell death.
In the study [92], the cytotoxicity of O-GNRs (310 × 5000 nm) and oxidized graphene oxide nanoparticles (O-GNPs; 100 × 100 nm) obtained by oxidative treatment of MWCNTs (100 × 5000 nm) and stacked graphene nanofibers (SGNFs; 100 × 5000 nm) were studied. The evaluation in vitro showed a higher cytotoxicity of O-GNRs compared to that of O-GNPs. The authors suggested that the effect is associated with a large number of carbonyl groups, as well as an increased O-GNRs length, i.e., the strong toxic effect of O-GNRs is the result of a synergistic effect between these two factors. In addition, the carbon source used to prepare oxidized graphene must be considered in biological research.
Chowdhury et al. [93] demonstrated that the ultrasonic bath or probe induces GNR structural disruption on MCF7 and A549 cells (human adenocarcinoma alveolar basal epithelial cells). According to the results, a GNR suspension of 20 μg/L treated with an ultrasonic probe for 1 min could cause a significant decrease in the overall metabolic state of cells compared to bath-treated or untreated suspension. The structural analysis showed that the ultrasonic probe treatment results in the disruption of the GNR structure and the formation of fine carbon “debris”, which may be the cause of toxicity.
A comparative analysis of the toxicity of single-layer reduced O-GNRs (rO-GNRs) and reduced graphene oxide sheets (rGOS) in relation to human mesenchymal stem cells (hMSCs) showed significant cytotoxic effects of rO-GNR at a concentration of 10 μg/mL of after 1 h of exposure, while rOGS showed the same degree of toxicity at a concentration of 100 μg/mL after 96 h. The main mechanism of rGOS action was assumed to be oxidative stress, which causes minor damage to the cell membrane. While the outflow of RNA from hMSCs showed that neither the formation of reactive oxygen nor significant damage of the membranes cells can explain the destruction of cells caused by rO-GNRs. The results also showed that rO-GNRs could penetrate cells and cause DNA fragmentation and chromosomal aberrations even at a low concentration of 1.0 μg/mL, after a short exposure time of 1 h [94,95].
Talukdar et al. [96] investigated the effect of graphene nanostructures with various morphologies, such as O-GNRs, graphene oxide nanoplatelets (O-GNPs), and graphene nano-onions (GNOs)) on the toxicity and differentiation potential of hMSCs. The cells were treated with various concentrations (5–300 μg/mL) of nanomaterials for 24 and 72 h. The results showed the dose-dependent, time-independent cytotoxicity of graphene nanostructures at concentrations above 50 μg/mL. The cellular uptake of GNOs and O-GNPs was shown by TEM and confocal Raman spectroscopy; no such effects were observed for O-GNRs.
For the imaging and phototherapy of human glioblastoma (U87MG), Akhavan et al. functionalized rO-GNRs with polyethylene glycol (rO-GNR–PEG). Cytotoxic and genotoxic effects on the cells depended on the concentration of rO-GNR–PEG. When the cells were incubated in the dark with 100 μg/mL of rO-GNR–PEG for 24 h, more than 72% of cell death and more than 29% of DNA fragmentation were observed. At a lower concentration (1 μg/mL), cell death and DNA fragmentation decreased to about 11% and 7%, respectively [97,98].
According to the considered researches on cyto- and genotoxicity, the possible mechanisms of the toxic action of GNRs can be suggested mechanical damage to cell membranes; ROS production, which leads to the inhibition of proliferation, induction of apoptosis, autophagy, and DNA fragmentation; chromosomal aberrations (Table 3).

Table 3.
Toxicity of GNRs according to various studies.
It should be noted that the studies of the toxicity of GNRs for the whole organism have not yet been carried out, excluding the work of Lu et al. [88]. However, it is known that other carbon nanomaterials have various toxic effects on living organisms, including neurotoxicity, nephrotoxicity, hepatotoxicity, genotoxicity and epigenetic toxicity, and dermatotoxicity [99,100,101,102,103]. This indicates the importance of conducting similar studies on GNRs.
4. GNRs in the Environment
Similar to other carbon nanomaterials, GNRs can enter the environment not only as part of the nanoindustry products, but also during natural processes, for example, during the combustion of organic matter. There is no information on the environmental toxicity of GNRs for ecosystems in the analyzed researches. An exception is the work of Lalwani et al., which investigates the oxidative biodegradation of O-GNRs and rO-GNRs by lignin peroxidase (LiP) [104]. LiP is an enzyme secreted by white rot fungi (Phanerochaete chrysosporium), which are widespread throughout the world in forest soils with dead and decaying organic matter. LiP breaks down lignin contained in the plant cell wall. The TEM and Raman spectroscopic analysis of O-GNRs and rO-GNRs treated with LiP for 4–96 h showed holes formation, confirming the structural degradation of graphene sheets. O-GNRs showed a higher rate of biodegradation compared to rO-GNRs: within 4 h after processing, numerous holes with a diameter of 1–5 nm were found on the O-GNRs sheets, which increased to ~300–350 nm after 48 h. The hole diameter on the rO-GNRs sample was 5–30 nm after 48 h of enzymatic treatment. After 96 h, the O-GNRs appeared to have completely degraded, while numerous holes were observed to extend across the entire width of the rO-GNR sample. These results showed that rO-GNRs can undergo oxidative biodegradation by LiP under environmental conditions [105].
The studies that are concerned with the closest chemical and structural analogues of GNRs–CNTs and graphene indicate their significant effects on various types of living organisms. A large number of studies demonstrate the toxic effects of carbon nanomaterials in relation to bacteria [106,107,108], fungi [109,110], protozoa [111], algae [112,113,114], higher plants [115,116], roundworms [117,118,119], arthropods [120], and mammals [121,122,123,124]. In addition, the possibility of bioaccumulation of carbon nanomaterials in living organisms has been investigated [125,126,127,128].
Based on this, it can be assumed that GNRs can also have a significant impact on components of ecosystems, especially microorganisms, and it can be bioaccumulated and migrate within ecosystems. These issues certainly warrant further study.
5. Conclusions
GNRs demonstrate the greatest prospects in the field of biomedicine, particularly in creating nanodevices for the biomolecules detection and single-molecular techniques. In addition, GNRs are of interest as gene and drug delivery vehicles. There are researches that prove the possibility of using GNRs in tissue engineering.
Practical applications of GNRs in direct contact with the human body are limited by their potential toxicity. A number of researchers have noted the damaging effects of GNRs, including cytotoxicity and genotoxicity. Moreover, the toxicity of nanoribbons is higher than those of such chemical analogs as O-GNPs. According to several studies, the possible mechanisms of toxicity are as follows: the ability to induce ROS production and autophagy; inhibition of proliferation; induction of apoptosis; DNA fragmentation; chromosomal aberrations. However, other authors have not confirmed any toxic effects of GNRs in experiments with human cell lines. The discrepancy in the observed results can be explained by differences in the methods of synthesis and structure of GNRs, their functionalization by different groups, and the use of different concentrations. In addition, different cell lines exhibit individual responses to exposure to nanomaterials. Furthermore, in order to obtain consistent results, the unification of the nanotoxicological experimental conditions is needed.
The chemical modification of the surface of GNRs with hydrophilic groups can significantly increase their bioavailability and biocompatibility.
There is an evidence of the biodegradability of GNRs in the environment. By analogy with other carbon nanomaterials, GNRs can be toxic to living organisms of various species. They can be bioaccumulated and migrate through ecosystems, but there are currently no data on this.
In general, this review allows concluding that GNRs, as components of high-precision nanodevices and therapeutic agents, have good prospects for application in biomedicine, but the limiting factors for the use of GNRs are their high hydrophobicity and insufficiently studied toxicity. There is a lack of information on the effect of GNRs on the whole organism using in vivo experiments, as well as on environmental toxicity, accumulation, migration, and destruction within ecosystems.
Author Contributions
Conceptualization, O.V.Z. and A.A.G.; methodology, O.V.Z. and A.A.G.; investigation, O.V.Z., E.E.M., K.S.G. and A.A.G.; writing—original draft preparation, O.V.Z., E.E.M. and A.A.G. All authors have read and agreed to the published version of the manuscript.
Funding
This study was funded by a grant from the Plekhanov Russian University of Economics.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Hernandez, Y.; Pang, S.; Feng, X.; Müllen, K. 8.16—Graphene and Its Synthesis. In Polymer Science: A Comprehensive Reference; Matyjaszewski, K., Möller, M., Eds.; Elsevier: Amsterdam, The Netherlands, 2012; pp. 415–438. [Google Scholar]
- Nakada, K.; Fujita, M.; Dresselhaus, G.; Dresselhaus, M.S. Edge state in graphene ribbons: Nanometer size effect and edge shape dependence. Phys. Rev. B 1996, 54, 17954–17961. [Google Scholar] [CrossRef] [Green Version]
- Wakabayashi, K.; Fujita, M.; Ajiki, H.; Sigrist, M. Electronic and magnetic properties of nanographite ribbons. Phys. Rev. B 1999, 59, 8271–8282. [Google Scholar] [CrossRef] [Green Version]
- Yang, Y.; Murali, R. Impact of Size Effect on Graphene Nanoribbon Transport. IEEE Electron. Device Lett. 2010, 31, 237–239. [Google Scholar] [CrossRef] [Green Version]
- Kiani, M.J.; Ahmadi, M.T.; Akbari, E.; Karimi, H.; Che Harun, F.K. Graphene Nanoribbon Based Gas Sensor. Key Eng. Mater. 2013, 553, 7–11. [Google Scholar] [CrossRef]
- Berahman, M.; Sheikhi, M.H. Hydrogen sulfide gas sensor based on decorated zigzag graphene nanoribbon with copper. Sens. Actuators B 2015, 219, 338–345. [Google Scholar] [CrossRef]
- Shekhirev, M.; Lipatov, A.; Torres, A.; Vorobeva, N.S.; Harkleroad, A.; Lashkov, A.; Sysoev, V.; Sinitskii, A. Highly Selective Gas Sensors Based on Graphene Nanoribbons Grown by Chemical Vapor Deposition. ACS Appl. Mater. Interfaces 2020, 12, 7392–7402. [Google Scholar] [CrossRef] [PubMed]
- Harada, N.; Sato, S. Electronic properties of NH4-adsorbed graphene nanoribbon as a promising candidate for a gas sensor. AIP Adv. 2016, 6, 055023. [Google Scholar] [CrossRef] [Green Version]
- Han, S.-J.; Jenkins, K.A.; Valdes Garcia, A.; Franklin, A.D.; Bol, A.A.; Haensch, W. High-Frequency Graphene Voltage Amplifier. Nano Lett. 2011, 11, 3690–3693. [Google Scholar] [CrossRef]
- Petrone, N.; Meric, I.; Hone, J.; Shepard, K.L. Graphene Field-Effect Transistors with Gigahertz-Frequency Power Gain on Flexible Substrates. Nano Lett. 2013, 13, 121–125. [Google Scholar] [CrossRef] [Green Version]
- Dimiev, A.M.; Tour, J.M. Graphene Nanoribbons: Production and Applications - Sigma Aldrich. Available online: https://www.sigmaaldrich.com/RU/ru/technical-documents/technical-article/materials-science-and-engineering/microelectronics-and-nanoelectronics/graphene-nanoribbons-production-and-applications (accessed on 4 June 2021).
- Han, M.Y.; Özyilmaz, B.; Zhang, Y.; Kim, P. Energy Band-Gap Engineering of Graphene Nanoribbons. Phys. Rev. Lett. 2007, 98, 206805. [Google Scholar] [CrossRef] [Green Version]
- Wang, X.; Dai, H. Etching and narrowing of graphene from the edges. Nat. Chem. 2010, 2, 661–665. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abramova, V.; Slesarev, A.S.; Tour, J.M. Meniscus-Mask Lithography for Narrow Graphene Nanoribbons. ACS Nano 2013, 7, 6894–6898. [Google Scholar] [CrossRef]
- Chen, L.; Hernandez, Y.; Feng, X.; Müllen, K. From Nanographene and Graphene Nanoribbons to Graphene Sheets: Chemical Synthesis. Angew. Chem. Int. Ed. 2012, 51, 7640–7654. [Google Scholar] [CrossRef] [PubMed]
- Cai, J.; Ruffieux, P.; Jaafar, R.; Bieri, M.; Braun, T.; Blankenburg, S.; Muoth, M.; Seitsonen, A.P.; Saleh, M.; Feng, X.; et al. Atomically precise bottom-up fabrication of graphene nanoribbons. Nature 2010, 466, 470–473. [Google Scholar] [CrossRef]
- Narita, A.; Feng, X.; Hernandez, Y.; Jensen, S.A.; Bonn, M.; Yang, H.; Verzhbitskiy, I.A.; Casiraghi, C.; Hansen, M.R.; Koch, A.H.R.; et al. Synthesis of structurally well-defined and liquid-phase-processable graphene nanoribbons. Nat. Chem. 2014, 6, 126–132. [Google Scholar] [CrossRef] [Green Version]
- Vo, T.H.; Shekhirev, M.; Kunkel, D.A.; Morton, M.D.; Berglund, E.; Kong, L.; Wilson, P.M.; Dowben, P.A.; Enders, A.; Sinitskii, A. Large-scale solution synthesis of narrow graphene nanoribbons. Nat. Commun. 2014, 5, 3189. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kosynkin, D.V.; Lu, W.; Sinitskii, A.; Pera, G.; Sun, Z.; Tour, J.M. Highly Conductive Graphene Nanoribbons by Longitudinal Splitting of Carbon Nanotubes Using Potassium Vapor. ACS Nano 2011, 5, 968–974. [Google Scholar] [CrossRef]
- Dimiev, A.; Lu, W.; Zeller, K.; Crowgey, B.; Kempel, L.C.; Tour, J.M. Low-Loss, High-Permittivity Composites Made from Graphene Nanoribbons. ACS Appl. Mater. Interfaces 2011, 3, 4657–4661. [Google Scholar] [CrossRef] [PubMed]
- Jiao, L.; Zhang, L.; Wang, X.; Diankov, G.; Dai, H. Narrow graphene nanoribbons from carbon nanotubes. Nature 2009, 458, 877–880. [Google Scholar] [CrossRef]
- Celis, A.; Nair, M.N.; Taleb-Ibrahimi, A.; Conrad, E.H.; Berger, C.; de Heer, W.A.; Tejeda, A. Graphene nanoribbons: Fabrication, properties and devices. J. Phys. D: Appl. Phys. 2016, 49, 143001. [Google Scholar] [CrossRef]
- Guo, X.; Baumgarten, M.; Müllen, K. Designing π-conjugated polymers for organic electronics. Prog. Polym. Sci. 2013, 38, 1832–1908. [Google Scholar] [CrossRef]
- Talirz, L.; Ruffieux, P.; Fasel, R. On-Surface Synthesis of Atomically Precise Graphene Nanoribbons. Adv. Mater. 2016, 28, 6222–6231. [Google Scholar] [CrossRef]
- Perkins, W.S. Synthesis and Applications of Graphene Nanoribbons and Heterostructures from Molecular Precursors. Ph.D. Thesis, University of California, Berkeley, CA, USA, 2017. [Google Scholar]
- Narita, A.; Wang, X.-Y.; Feng, X.; Müllen, K. New advances in nanographene chemistry. Chem. Soc. Rev. 2015, 44, 6616–6643. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Magda, G.Z.; Jin, X.; Hagymási, I.; Vancsó, P.; Osváth, Z.; Nemes-Incze, P.; Hwang, C.; Biró, L.P.; Tapasztó, L. Room-temperature magnetic order on zigzag edges of narrow graphene nanoribbons. Nature 2014, 514, 608–611. [Google Scholar] [CrossRef] [Green Version]
- Wang, W.-X.; Zhou, M.; Li, X.; Li, S.-Y.; Wu, X.; Duan, W.; He, L. Energy gaps of atomically precise armchair graphene sidewall nanoribbons. Phys. Rev. B 2016, 93, 241403. [Google Scholar] [CrossRef] [Green Version]
- Merino-Díez, N.; Garcia-Lekue, A.; Carbonell-Sanromà, E.; Li, J.; Corso, M.; Colazzo, L.; Sedona, F.; Sánchez-Portal, D.; Pascual, J.I.; de Oteyza, D.G. Width-Dependent Band Gap in Armchair Graphene Nanoribbons Reveals Fermi Level Pinning on Au(111). ACS Nano 2017, 11, 11661–11668. [Google Scholar] [CrossRef]
- Narita, A.; Chen, Z.; Chen, Q.; Müllen, K. Solution and on-surface synthesis of structurally defined graphene nanoribbons as a new family of semiconductors. Chem. Sci. 2019, 10, 964–975. [Google Scholar] [CrossRef] [Green Version]
- de Sousa Araújo Cassiano, T.; Monteiro, F.F.; Evaristo de Sousa, L.; Magela e Silva, G.; de Oliveira Neto, P.H. Smooth gap tuning strategy for cove-type graphene nanoribbons. RSC Adv. 2020, 10, 26937–26943. [Google Scholar] [CrossRef]
- Johnson, A.P.; Sabu, C.; Swamy, N.K.; Anto, A.; Gangadharappa, H.V.; Pramod, K. Graphene nanoribbon: An emerging and efficient flat molecular platform for advanced biosensing. Biosens. Bioelectron. 2021, 184, 113245. [Google Scholar] [CrossRef]
- Shende, P.; Pathan, N. Graphene nanoribbons: A state-of-the-art in health care. Int. J. Pharm. 2021, 595, 120269. [Google Scholar] [CrossRef]
- Johnson, A.P.; Gangadharappa, H.V.; Pramod, K. Graphene nanoribbons: A promising nanomaterial for biomedical applications. J. Controlled Release 2020, 325, 141–162. [Google Scholar] [CrossRef]
- Shende, P.; Augustine, S.; Prabhakar, B. A review on graphene nanoribbons for advanced biomedical applications. Carbon Lett. 2020, 30, 465–475. [Google Scholar] [CrossRef]
- Mousavi, S.M.; Soroshnia, S.; Hashemi, S.A.; Babapoor, A.; Younes, G.; Amani, A. Graphene Nano-ribbon Based high potential and Efficiency for DNA, Cancer therapy and drug delivery applications. Drug Metab. Rev. 2019, 51, 1–35. [Google Scholar] [CrossRef]
- Cai, J.; Pignedoli, C.A.; Talirz, L.; Ruffieux, P.; Söde, H.; Liang, L.; Meunier, V.; Berger, R.; Li, R.; Feng, X.; et al. Graphene nanoribbon heterojunctions. Nat. Nanotech. 2014, 9, 896–900. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bennett, P.B.; Pedramrazi, Z.; Madani, A.; Chen, Y.-C.; Oteyza, D.G.d.; Chen, C.; Fischer, F.R.; Crommie, M.F.; Bokor, J. Bottom-up graphene nanoribbon field-effect transistors. Appl. Phys. Lett. 2013, 103, 253114. [Google Scholar] [CrossRef] [Green Version]
- Wei, D.; Xie, L.; Lee, K.K.; Hu, Z.; Tan, S.; Chen, W.; Sow, C.H.; Chen, K.; Liu, Y.; Wee, A.T.S. Controllable unzipping for intramolecular junctions of graphene nanoribbons and single-walled carbon nanotubes. Nat. Commun. 2013, 4, 1374. [Google Scholar] [CrossRef] [Green Version]
- Freitag, M.; Low, T.; Zhu, W.; Yan, H.; Xia, F.; Avouris, P. Photocurrent in graphene harnessed by tunable intrinsic plasmons. Nat. Commun. 2013, 4, 1951. [Google Scholar] [CrossRef] [Green Version]
- Ryzhii, V.; Ryabova, N.; Ryzhii, M.; Baryshnikov, N.V.; Karasik, V.E.; Mitin, V.; Otsuji, T. Terahertz and infrared photodetectors based on multiple graphene layer and nanoribbon structures. Opto-Electron. Rev. 2012, 20, 15–25. [Google Scholar] [CrossRef]
- Johnson, J.L.; Behnam, A.; Pearton, S.J.; Ural, A. Hydrogen Sensing Using Pd-Functionalized Multi-Layer Graphene Nanoribbon Networks. Adv. Mater. 2010, 22, 4877–4880. [Google Scholar] [CrossRef] [PubMed]
- Zarei, M.; Sharifi, M.J. Graphene nanoribbon photodetectors based on an asymmetric potential barrier: A new concept and a new structure. J. Comput. Electron. 2018, 17, 531–539. [Google Scholar] [CrossRef]
- Yu, X.; Dong, Z.; Liu, Y.; Liu, T.; Tao, J.; Zeng, Y.; Yang, J.K.W.; Wang, Q.J. A high performance, visible to mid-infrared photodetector based on graphene nanoribbons passivated with HfO2. Nanoscale 2016, 8, 327–332. [Google Scholar] [CrossRef]
- Isaeva, O.G.; Katkov, V.L.; Osipov, V.A. DNA sequencing through graphene nanogap: A model of sequential electron transport. Eur. Phys. J. B 2014, 87, 272. [Google Scholar] [CrossRef]
- Chen, W.; Liu, G.-C.; Ouyang, J.; Gao, M.-J.; Liu, B.; Zhao, Y.-D. Graphene nanopores toward DNA sequencing: A review of experimental aspects. Sci. China Chem. 2017, 60, 721–729. [Google Scholar] [CrossRef]
- Wu, X.; Mu, F.; Wang, Y.; Zhao, H. Graphene and Graphene-Based Nanomaterials for DNA Detection: A Review. Molecules 2018, 23, 2050. [Google Scholar] [CrossRef] [Green Version]
- Wu, X.; Fengwen, M.; Zhao, H. Synthesis and potential applications of nanoporous graphene: A review. Proc. Nat. Res. Soc. 2018, 2, 02003. [Google Scholar] [CrossRef]
- Arjmandi-Tash, H.; Belyaeva, L.A.; Schneider, G.F. Single molecule detection with graphene and other two-dimensional materials: Nanopores and beyond. Chem. Soc. Rev. 2016, 45, 476–493. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nelson, T.; Zhang, B.; Prezhdo, O.V. Detection of Nucleic Acids with Graphene Nanopores: Ab Initio Characterization of a Novel Sequencing Device. Nano Lett. 2010, 10, 3237–3242. [Google Scholar] [CrossRef] [PubMed]
- Traversi, F.; Raillon, C.; Benameur, S.M.; Liu, K.; Khlybov, S.; Tosun, M.; Krasnozhon, D.; Kis, A.; Radenovic, A. Detecting the translocation of DNA through a nanopore using graphene nanoribbons. Nat. Nanotech. 2013, 8, 939–945. [Google Scholar] [CrossRef]
- Min, S.K.; Kim, W.Y.; Cho, Y.; Kim, K.S. Fast DNA sequencing with a graphene-based nanochannel device. Nat. Nanotech. 2011, 6, 162–165. [Google Scholar] [CrossRef] [PubMed]
- Agah, S.; Zheng, M.; Pasquali, M.; Kolomeisky, A.B. DNA sequencing by nanopores: Advances and challenges. J. Phys. D: Appl. Phys. 2016, 49, 413001. [Google Scholar] [CrossRef] [Green Version]
- Heerema, S.J.; Vicarelli, L.; Pud, S.; Schouten, R.N.; Zandbergen, H.W.; Dekker, C. Probing DNA Translocations with Inplane Current Signals in a Graphene Nanoribbon with a Nanopore. ACS Nano 2018, 12, 2623–2633. [Google Scholar] [CrossRef] [Green Version]
- Puster, M.; Rodríguez-Manzo, J.A.; Balan, A.; Drndić, M. Toward Sensitive Graphene Nanoribbon–Nanopore Devices by Preventing Electron Beam-Induced Damage. ACS Nano 2013, 7, 11283–11289. [Google Scholar] [CrossRef]
- Puster, M.; Balan, A.; Rodríguez-Manzo, J.A.; Danda, G.; Ahn, J.-H.; Parkin, W.; Drndić, M. Cross-Talk Between Ionic and Nanoribbon Current Signals in Graphene Nanoribbon-Nanopore Sensors for Single-Molecule Detection. Small 2015, 11, 6309–6316. [Google Scholar] [CrossRef] [Green Version]
- Saha, K.K.; Drndić, M.; Nikolić, B.K. DNA Base-Specific Modulation of Microampere Transverse Edge Currents through a Metallic Graphene Nanoribbon with a Nanopore. Nano Lett. 2012, 12, 50–55. [Google Scholar] [CrossRef] [Green Version]
- Paulechka, E.; Wassenaar, T.A.; Kroenlein, K.; Kazakov, A.; Smolyanitsky, A. Nucleobase-functionalized graphene nanoribbons for accurate high-speed DNA sequencing. Nanoscale 2016, 8, 1861–1867. [Google Scholar] [CrossRef] [Green Version]
- Rodríguez, B.A.G.; Pérez-Caro, M.; Alencar, R.S.; Filho, A.G.S.; Aguiar, J.A. Graphene nanoribbons and iron oxide nanoparticles composite as a potential candidate in DNA sensing applications. J. Appl. Phys. 2020, 127, 044901. [Google Scholar] [CrossRef]
- Feng, Q.; Zhao, X.; Guo, Y.; Liu, M.; Wang, P. Stochastic DNA walker for electrochemical biosensing sensitized with gold nanocages@graphene nanoribbons. Biosens. Bioelectron. 2018, 108, 97–102. [Google Scholar] [CrossRef] [PubMed]
- Mehmeti, E.; Stanković, D.M.; Chaiyo, S.; Zavasnik, J.; Žagar, K.; Kalcher, K. Wiring of glucose oxidase with graphene nanoribbons: An electrochemical third generation glucose biosensor. Microchim. Acta 2017, 184, 1127–1134. [Google Scholar] [CrossRef]
- Rostami, S.; Niroumand, R.; Jabbari, A. Enhanced LSPR performance of graphene nanoribbons-silver nanoparticles hybrid as a colorimetric sensor for sequential detection of dopamine and glutathione. Anal. Chim. Acta 2020, 1120, 11–23. [Google Scholar] [CrossRef]
- Sainz, R.; del Pozo, M.; Vilas-Varela, M.; Castro-Esteban, J.; Pérez Corral, M.; Vázquez, L.; Blanco, E.; Peña, D.; Martín-Gago, J.A.; Ellis, G.J.; et al. Chemically synthesized chevron-like graphene nanoribbons for electrochemical sensors development: Determination of epinephrine. Sci. Rep. 2020, 10, 14614. [Google Scholar] [CrossRef] [PubMed]
- Lalwani, G.; Cai, X.; Nie, L.; Wang, L.; Sitharaman, B. Graphene-based contrast agents for photoacoustic and thermoacoustic tomography. Photoacoustics 2013, 1, 62–67. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gurunathan, S.; Kim, J.-H. Synthesis, toxicity, biocompatibility, and biomedical applications of graphene and graphene-related materials. Int. J. Nanomed. 2016, 11, 1927–1945. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ouyang, F.-P.; Peng, S.-L.; Zhang, H.; Weng, L.-B.; Xu, H. A biosensor based on graphene nanoribbon with nanopores: A first-principles devices-design. Chin. Phys. B 2011, 20, 058504. [Google Scholar] [CrossRef]
- Avdoshenko, S.M.; Nozaki, D.; Gomes da Rocha, C.; González, J.W.; Lee, M.H.; Gutierrez, R.; Cuniberti, G. Dynamic and Electronic Transport Properties of DNA Translocation through Graphene Nanopores. Nano Lett. 2013, 13, 1969–1976. [Google Scholar] [CrossRef] [PubMed]
- Sadeghi, H.; Algaragholy, L.; Pope, T.; Bailey, S.; Visontai, D.; Manrique, D.; Ferrer, J.; Garcia-Suarez, V.; Sangtarash, S.; Lambert, C.J. Graphene Sculpturene Nanopores for DNA Nucleobase Sensing. J. Phys. Chem. B 2014, 118, 6908–6914. [Google Scholar] [CrossRef]
- Jampilek, J.; Kralova, K. Advances in Drug Delivery Nanosystems Using Graphene-Based Materials and Carbon Nanotubes. Materials 2021, 14, 1059. [Google Scholar] [CrossRef] [PubMed]
- Janani, K.; Thiruvadigal, D.J. Density functional study on covalent functionalization of zigzag graphene nanoribbon through l-Phenylalanine and boron doping: Effective nanocarriers in drug delivery applications. Appl. Surf. Sci. 2018, 449, 815–822. [Google Scholar] [CrossRef]
- Mullick Chowdhury, S.; Zafar, S.; Tellez, V.; Sitharaman, B. Graphene Nanoribbon-Based Platform for Highly Efficacious Nuclear Gene Delivery. ACS Biomater. Sci. Eng. 2016, 2, 798–808. [Google Scholar] [CrossRef]
- Dong, H.; Ding, L.; Yan, F.; Ji, H.; Ju, H. The use of polyethylenimine-grafted graphene nanoribbon for cellular delivery of locked nucleic acid modified molecular beacon for recognition of microRNA. Biomaterials 2011, 32, 3875–3882. [Google Scholar] [CrossRef]
- Chowdhury, S.M.; Surhland, C.; Sanchez, Z.; Chaudhary, P.; Suresh Kumar, M.A.; Lee, S.; Peña, L.A.; Waring, M.; Sitharaman, B.; Naidu, M. Graphene nanoribbons as a drug delivery agent for lucanthone mediated therapy of glioblastoma multiforme. Nanomed. Nanotechnol. Biol. Med. 2015, 11, 109–118. [Google Scholar] [CrossRef] [Green Version]
- Dasari Shareena, T.P.; McShan, D.; Dasmahapatra, A.K.; Tchounwou, P.B. A Review on Graphene-Based Nanomaterials in Biomedical Applications and Risks in Environment and Health. Nano-Micro Lett. 2018, 10, 53. [Google Scholar] [CrossRef]
- Mullick Chowdhury, S.; Manepalli, P.; Sitharaman, B. Graphene nanoribbons elicit cell specific uptake and delivery via activation of epidermal growth factor receptor enhanced by human papillomavirus E5 protein. Acta Biomater. 2014, 10, 4494–4504. [Google Scholar] [CrossRef] [Green Version]
- Wieduwilt, M.J.; Moasser, M.M. The epidermal growth factor receptor family: Biology driving targeted therapeutics. Cell. Mol. Life Sci. 2008, 65, 1566–1584. [Google Scholar] [CrossRef] [Green Version]
- Engelking, L.R. Chapter 59: Sphingolipids. In Textbook of Veterinary Physiological Chemistry (Third Edition); Academic Press: San Diego, CA, USA, 2014; pp. 378–383. [Google Scholar]
- Goñi, F.M.; Alonso, A. Biophysics of sphingolipids I. Membrane properties of sphingosine, ceramides and other simple sphingolipids. Biochim. Biophys. Acta 2006, 1758, 1902–1921. [Google Scholar] [CrossRef] [Green Version]
- Padrón, J.M. Sphingolipids in anticancer therapy. Curr. Med. Chem. 2006, 13, 755–770. [Google Scholar] [CrossRef]
- Saddoughi, S.A.; Song, P.; Ogretmen, B. Roles of bioactive sphingolipids in cancer biology and therapeutics. Subcell. Biochem. 2008, 49, 413–440. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ségui, B.; Andrieu-Abadie, N.; Jaffrézou, J.P.; Benoist, H.; Levade, T. Sphingolipids as modulators of cancer cell death: Potential therapeutic targets. Biochim. Biophys. Acta 2006, 1758, 2104–2120. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Suhrland, C.; Truman, J.P.; Obeid, L.M.; Sitharaman, B. Oxidized graphene nanoparticles as a delivery system for the pro-apoptotic sphingolipid C(6) ceramide. J. Biomed. Mater. Res. Part. A 2019, 107, 25–37. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Suhrland, C.; Truman, J.P.; Obeid, L.M.; Sitharaman, B. Delivery of long chain C(16) and C(24) ceramide in HeLa cells using oxidized graphene nanoribbons. J. Biomed. Mater. Res. Part. B, Appl. Biomater. 2020, 108, 1141–1156. [Google Scholar] [CrossRef]
- Liu, Y.; Wang, X.; Wan, W.; Li, L.; Dong, Y.; Zhao, Z.; Qiu, J. Multifunctional nitrogen-doped graphene nanoribbon aerogels for superior lithium storage and cell culture. Nanoscale 2016, 8, 2159–2167. [Google Scholar] [CrossRef]
- Chowdhury, S.M.; Fang, J.; Sitharaman, B. Interaction of graphene nanoribbons with components of the blood vascular system. Future Sci. OA 2015, 1, 3. [Google Scholar] [CrossRef] [Green Version]
- Silva, M.; Caridade, S.G.; Vale, A.C.; Cunha, E.; Sousa, M.P.; Mano, J.F.; Paiva, M.C.; Alves, N.M. Biomedical films of graphene nanoribbons and nanoflakes with natural polymers. RSC Adv. 2017, 7, 27578–27594. [Google Scholar] [CrossRef] [Green Version]
- Foreman, H.-C.C.; Lalwani, G.; Kalra, J.; Krug, L.T.; Sitharaman, B. Gene delivery to mammalian cells using a graphene nanoribbon platform. J. Mater. Chem. B 2017, 5, 2347–2354. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.-J.; Lin, C.-W.; Yang, H.-W.; Lin, K.-J.; Wey, S.-P.; Sun, C.-L.; Wei, K.-C.; Yen, T.-C.; Lin, C.-I.; Ma, C.-C.M.; et al. Biodistribution of PEGylated graphene oxide nanoribbons and their application in cancer chemo-photothermal therapy. Carbon 2014, 74, 83–95. [Google Scholar] [CrossRef]
- Mbeh, D.A.; Akhavan, O.; Javanbakht, T.; Mahmoudi, M.; Yahia, L.H. Cytotoxicity of protein corona-graphene oxide nanoribbons on human epithelial cells. Appl. Surf. Sci. 2014, 320, 596–601. [Google Scholar] [CrossRef]
- Mullick Chowdhury, S.; Lalwani, G.; Zhang, K.; Yang, J.Y.; Neville, K.; Sitharaman, B. Cell specific cytotoxicity and uptake of graphene nanoribbons. Biomaterials 2013, 34, 283–293. [Google Scholar] [CrossRef] [Green Version]
- Mari, E.; Mardente, S.; Morgante, E.; Tafani, M.; Lococo, E.; Fico, F.; Valentini, F.; Zicari, A. Graphene Oxide Nanoribbons Induce Autophagic Vacuoles in Neuroblastoma Cell Lines. Int. J. Mol. Sci. 2016, 17, 1995. [Google Scholar] [CrossRef] [Green Version]
- Khim Chng, E.L.; Chua, C.K.; Pumera, M. Graphene oxide nanoribbons exhibit significantly greater toxicity than graphene oxide nanoplatelets. Nanoscale 2014, 6, 10792–10797. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mullick Chowdhury, S.; Dasgupta, S.; McElroy, A.E.; Sitharaman, B. Structural disruption increases toxicity of graphene nanoribbons. J. Appl. Toxicol. 2014, 34, 1235–1246. [Google Scholar] [CrossRef]
- Akhavan, O.; Ghaderi, E.; Emamy, H.; Akhavan, F. Genotoxicity of graphene nanoribbons in human mesenchymal stem cells. Carbon 2013, 54, 419–431. [Google Scholar] [CrossRef]
- Ou, L.; Song, B.; Liang, H.; Liu, J.; Feng, X.; Deng, B.; Sun, T.; Shao, L. Toxicity of graphene-family nanoparticles: A general review of the origins and mechanisms. Part. Fibre Toxicol. 2016, 13, 57. [Google Scholar] [CrossRef] [Green Version]
- Talukdar, Y.; Rashkow, J.; Lalwani, G.; Kanakia, S.; Sitharaman, B. The effects of graphene nanostructures on mesenchymal stem cells. Biomaterials 2014, 35, 4863–4877. [Google Scholar] [CrossRef] [Green Version]
- Akhavan, O.; Ghaderi, E.; Emamy, H. Nontoxic concentrations of PEGylated graphene nanoribbons for selective cancer cell imaging and photothermal therapy. J. Mater. Chem. 2012, 22, 20626–20633. [Google Scholar] [CrossRef]
- Gurcan, C.; Taheri, H.; Bianco, A.; Delogu, L.G.; Yilmazer, A. A closer look at the genotoxicity of graphene based materials. J. Phys. Mater. 2019, 3, 014007. [Google Scholar] [CrossRef]
- Madannejad, R.; Shoaie, N.; Jahanpeyma, F.; Darvishi, M.H.; Azimzadeh, M.; Javadi, H. Toxicity of carbon-based nanomaterials: Reviewing recent reports in medical and biological systems. Chem. Biol. Interact. 2019, 307, 206–222. [Google Scholar] [CrossRef] [PubMed]
- Ema, M.; Gamo, M.; Honda, K. A review of toxicity studies of single-walled carbon nanotubes in laboratory animals. Regul. Toxicol. Pharm. 2016, 74, 42–63. [Google Scholar] [CrossRef] [PubMed]
- Morimoto, Y.; Horie, M.; Kobayashi, N.; Shinohara, N.; Shimada, M. Inhalation Toxicity Assessment of Carbon-Based Nanoparticles. Acc. Chem. Res. 2013, 46, 770–781. [Google Scholar] [CrossRef] [PubMed]
- De Maio, F.; Palmieri, V.; De Spirito, M.; Delogu, G.; Papi, M. Carbon nanomaterials: A new way against tuberculosis. Expert Rev. Med. Devices 2019, 16, 863–875. [Google Scholar] [CrossRef]
- Raja, I.S.; Song, S.J.; Kang, M.S.; Lee, Y.B.; Kim, B. Toxicity of Zero- and One-Dimensional Carbon. Nanomaterials 2019, 9, 1214. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lalwani, G.; Xing, W.; Sitharaman, B. Enzymatic degradation of oxidized and reduced graphene nanoribbons by lignin peroxidase. J. Mater. Chem. B 2014, 2, 6354–6362. [Google Scholar] [CrossRef] [Green Version]
- Lalwani, G.; D’Agati, M.; Khan, A.M.; Sitharaman, B. Toxicology of graphene-based nanomaterials. Adv. Drug Delivery Rev. 2016, 105, 109–144. [Google Scholar] [CrossRef] [Green Version]
- Al-Jumaili, A.; Alancherry, S.; Bazaka, K.; Jacob, M.V. Review on the Antimicrobial Properties of Carbon Nanostructures. Materials 2017, 10, 1066. [Google Scholar] [CrossRef] [PubMed]
- Maas, M. Carbon Nanomaterials as Antibacterial Colloids. Materials 2016, 9, 617. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maleki Dizaj, S.; Mennati, A.; Jafari, S.; Khezri, K.; Adibkia, K. Antimicrobial activity of carbon-based nanoparticles. Adv. Pharm Bull. 2015, 5, 19–23. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Liu, X.; Chen, J.; Han, H.; Yuan, Z. Evaluation and mechanism of antifungal effects of carbon nanomaterials in controlling plant fungal pathogen. Carbon 2014, 68, 798–806. [Google Scholar] [CrossRef]
- Hao, Y.; Cao, X.; Ma, C.; Zhang, Z.; Zhao, N.; Ali, A.; Hou, T.; Xiang, Z.; Zhuang, J.; Wu, S.; et al. Potential Applications and Antifungal Activities of Engineered Nanomaterials against Gray Mold Disease Agent Botrytis cinerea on Rose Petals. Front. Plant Sci. 2017, 8, 1332. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ghafari, P.; St-Denis, C.H.; Power, M.E.; Jin, X.; Tsou, V.; Mandal, H.S.; Bols, N.C.; Tang, X.S. Impact of carbon nanotubes on the ingestion and digestion of bacteria by ciliated protozoa. Nat. Nanotech. 2008, 3, 347–351. [Google Scholar] [CrossRef] [PubMed]
- Basiuk, E.V.; Ochoa-Olmos, O.E.; De la Mora-Estrada, L.F. Ecotoxicological effects of carbon nanomaterials on algae, fungi and plants. J. Nanosci. Nanotech. 2011, 11, 3016–3038. [Google Scholar] [CrossRef]
- Freixa, A.; Acuña, V.; Sanchís, J.; Farré, M.; Barceló, D.; Sabater, S. Ecotoxicological effects of carbon based nanomaterials in aquatic organisms. Sci. Total Environ. 2018, 619–620, 328–337. [Google Scholar] [CrossRef]
- Saxena, P.; Sangela, V.; Ranjan, S.; Dutta, V.; Dasgupta, N.; Phulwaria, M.; Rathore, D.S.; Harish. Aquatic nanotoxicology: Impact of carbon nanomaterials on algal flora. Energy Ecol. Environ. 2020, 5, 240–252. [Google Scholar] [CrossRef]
- Zaytseva, O.; Neumann, G. Carbon nanomaterials: Production, impact on plant development, agricultural and environmental applications. Chem. Biol. Technol. Agric. 2016, 3, 1–26. [Google Scholar] [CrossRef] [Green Version]
- Zhang, P.; Zhang, R.; Fang, X.; Song, T.; Cai, X.; Liu, H.; Du, S. Toxic effects of graphene on the growth and nutritional levels of wheat (Triticum aestivum L.): Short- and long-term exposure studies. J. Hazard. Mater. 2016, 317, 543–551. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Jin, L.; Wang, Y.; Kong, Y.; Wang, D. Prolonged exposure to multi-walled carbon nanotubes dysregulates intestinal mir-35 and its direct target MAB-3 in nematode Caenorhabditis elegans. Sci. Rep. 2019, 9, 12144. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Yang, J.; Wang, D. A MicroRNA-Mediated Insulin Signaling Pathway Regulates the Toxicity of Multi-Walled Carbon Nanotubes in Nematode Caenorhabditis elegans. Sci. Rep. 2016, 6, 23234. [Google Scholar] [CrossRef] [Green Version]
- Walczynska, M.; Jakubowski, W.; Wasiak, T.; Kadziola, K.; Bartoszek, N.; Kotarba, S.; Siatkowska, M.; Komorowski, P.; Walkowiak, B. Toxicity of silver nanoparticles, multiwalled carbon nanotubes, and dendrimers assessed with multicellular organism Caenorhabditis elegans. Toxicol. Mech. Meth. 2018, 28, 432–439. [Google Scholar] [CrossRef]
- Bai, X.; Zhao, S.; Duo, L. Impacts of carbon nanomaterials on the diversity of microarthropods in turfgrass soil. Sci. Rep. 2017, 7, 1–6. [Google Scholar] [CrossRef] [Green Version]
- Hassan, A. Toxic and benefit effects on human and animal health Book 2. In Carbon Nanomaterials; Elsevier: Amsterdam, The Netherlands, 2019; p. 25. [Google Scholar]
- Liang, G.; Yin, L.; Zhang, J.; Liu, R.; Zhang, T.; Ye, B.; Pu, Y. Effects of subchronic exposure to multi-walled carbon nanotubes on mice. J. Toxicol. Environ. Health. Part A 2010, 73, 463–470. [Google Scholar] [CrossRef]
- Mohammadi, E.; Zeinali, M.; Mohammadi-Sardoo, M.; Iranpour, M.; Behnam, B.; Mandegary, A. The effects of functionalization of carbon nanotubes on toxicological parameters in mice. Hum. Exp. Toxicol. 2020, 39, 1147–1167. [Google Scholar] [CrossRef]
- Porter, D.W.; Hubbs, A.F.; Mercer, R.R.; Wu, N.; Wolfarth, M.G.; Sriram, K.; Leonard, S.; Battelli, L.; Schwegler-Berry, D.; Friend, S.; et al. Mouse pulmonary dose- and time course-responses induced by exposure to multi-walled carbon nanotubes. Toxicology 2010, 269, 136–147. [Google Scholar] [CrossRef]
- Jackson, P.; Jacobsen, N.R.; Baun, A.; Birkedal, R.; Kühnel, D.; Jensen, K.A.; Vogel, U.; Wallin, H. Bioaccumulation and ecotoxicity of carbon nanotubes. Chem. Cent. J. 2013, 7, 154. [Google Scholar] [CrossRef] [Green Version]
- Xie, P.; Yang, S.-T.; He, T.; Yang, S.; Tang, X.-H. Bioaccumulation and Toxicity of Carbon Nanoparticles Suspension Injection in Intravenously Exposed Mice. Int. J. Mol. Sci. 2017, 18, 2562. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sarma, S.; Bhattacharya, I.; Brar, S.; Tyagi, R.; Surampalli, R. Carbon Nanotube-Bioaccumulation and Recent Advances in Environmental Monitoring. Crit. Rev. Environ. Sci.Technol. 2015, 45, 905–938. [Google Scholar] [CrossRef]
- Smirnova, E.; Gusev, A.; Zaytseva, O.; Sheina, O.; Tkachev, A.; Kuznetsova, E.; Lazareva, E.; Onishchenko, G.; Feofanov, A.; Kirpichnikov, M. Uptake and accumulation of multiwalled carbon nanotubes change the morphometric and biochemical characteristics of Onobrychis arenaria seedlings. Front. Chem. Sci. Eng. 2012, 6, 132–138. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).