ZnO-Impregnated Polyacrylonitrile Nanofiber Filters against Various Phases of Air Pollutants
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
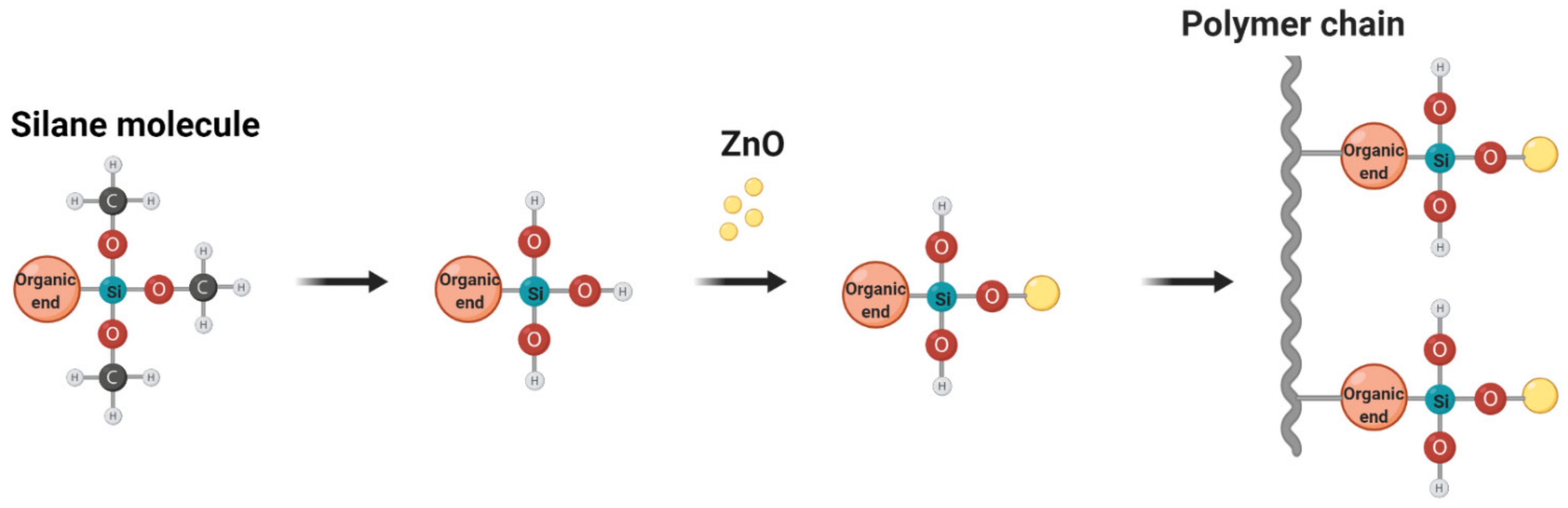
2.2. Modification of ZnO NPs with MPTMS
2.3. Preparation of Electrospinning Solutions
2.4. Nanofiber Fabrication by Electrospinning
2.5. Characterization of Nanofiber Filters
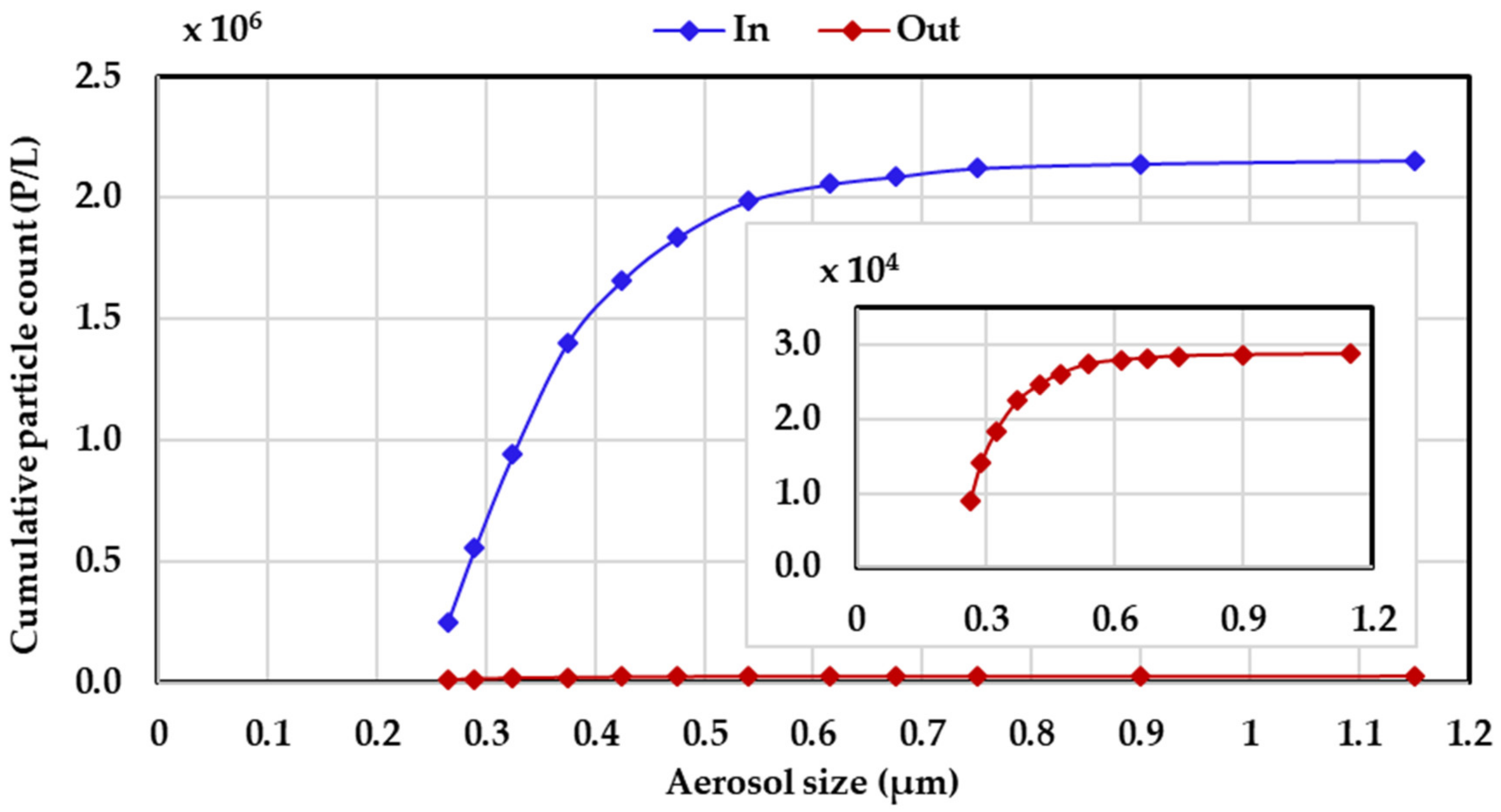
2.6. Filtration Test
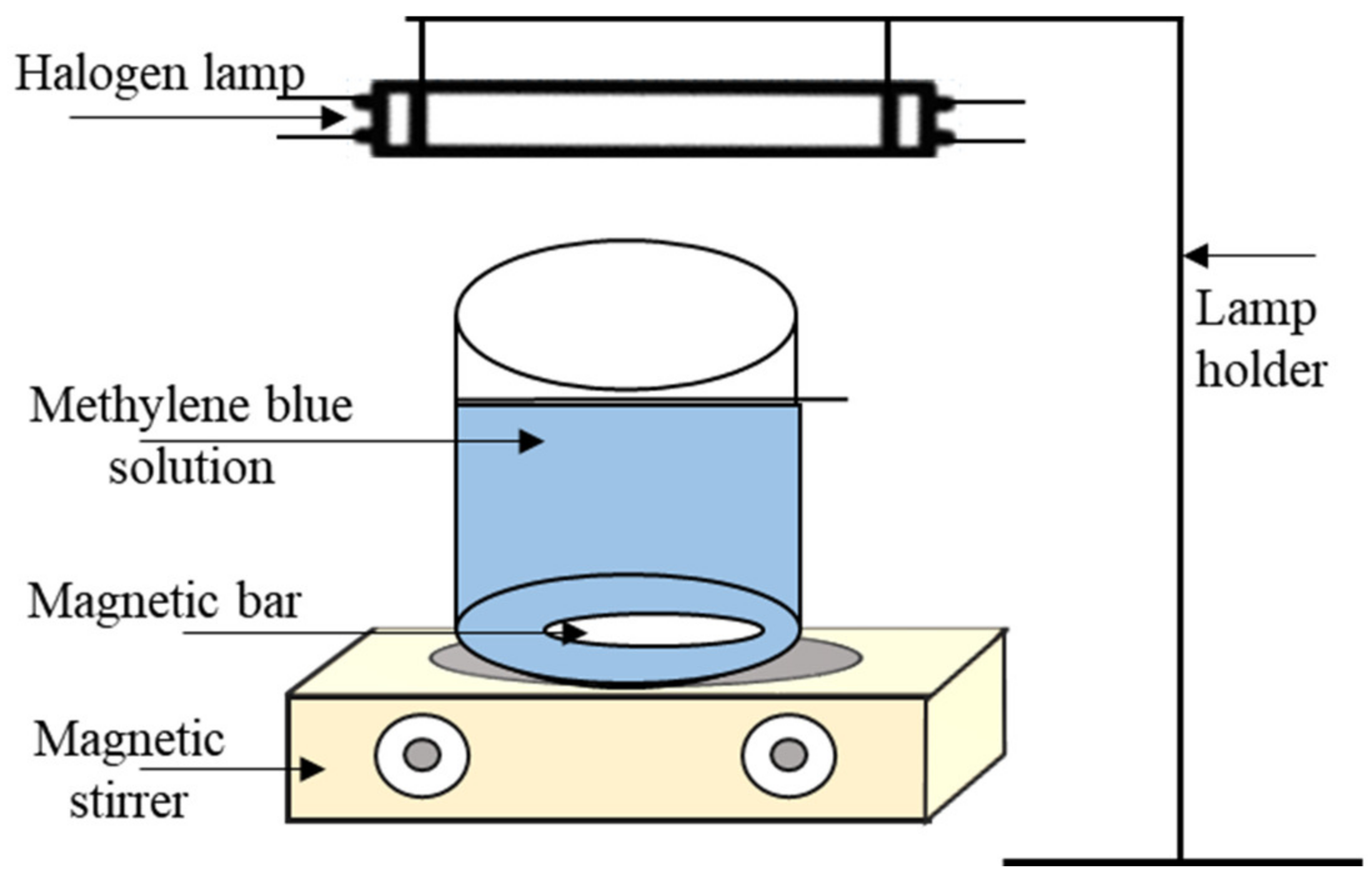
2.7. Photocatalytic Activity Test
2.8. Antibacterial Activity Test
3. Results and Discussion
3.1. Effect of Silane on ZnO Dispersion in Nanofibers
3.1.1. Modified ZnO
3.1.2. Morphological Characterization of PAN/ZnO (Silane) Nanofibers
3.1.3. Chemical Characterization of PAN/ZnO(Silane) Nanofiber
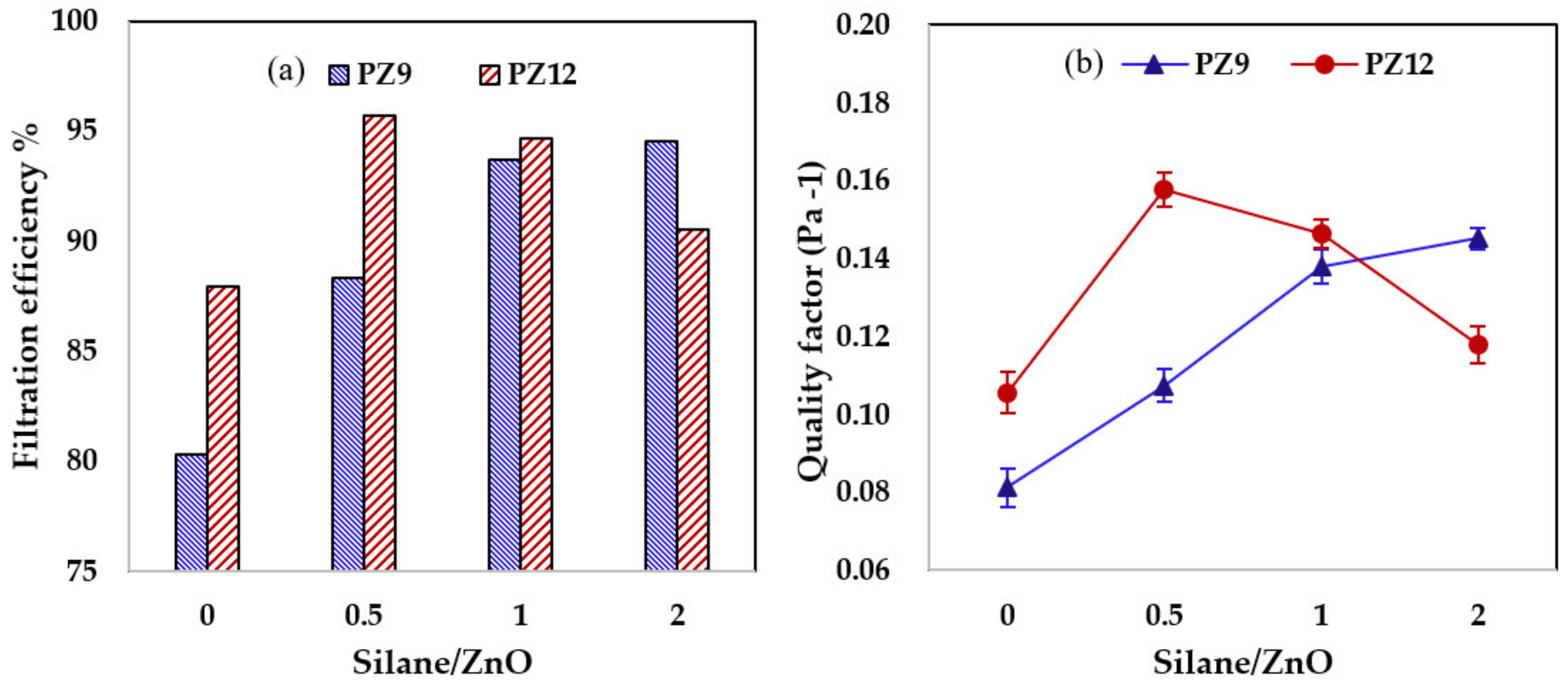
3.2. Performance of PAN/ZnO (Silane) Fiber Filters for Air Pollutants
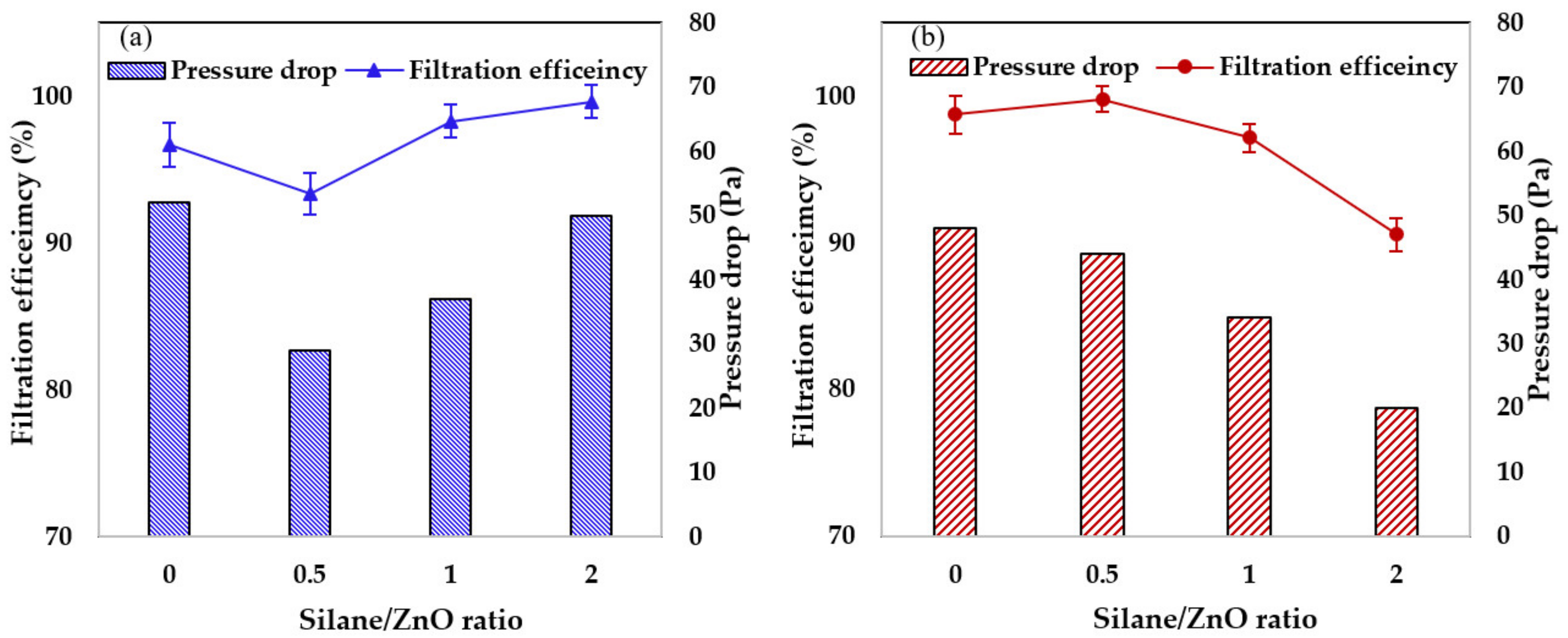
3.2.1. Filtration Performance
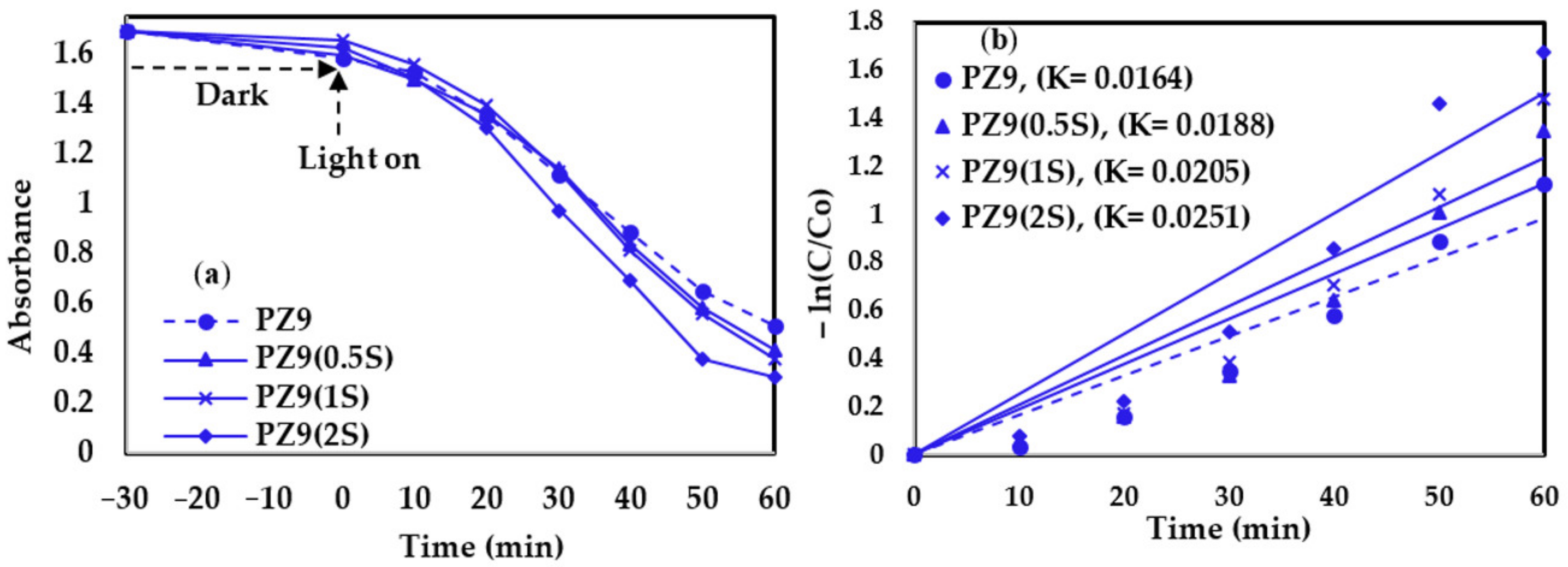
3.2.2. Photocatalytic Activity
3.2.3. Antibacterial Activity
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Bortolassi, A.C.C.; Guerra, V.G.; Aguiar, M.L.; Soussan, L.; Cornu, D.; Miele, P.; Bechelany, M. Composites Based on Nanoparticle and Pan Electrospun Nanofiber Membranes for Air Filtration and Bacterial Removal. Nanomaterials 2019, 9, 1740. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tcharkhtchi, A.; Abbasnezhad, N.; Seydani, M.Z.; Zirak, N.; Farzaneh, S.; Shirinbayan, M. An Overview of Filtration Efficiency through the Masks: Mechanisms of the Aerosols Penetration. Bioact. Mater. 2021, 6, 106–122. [Google Scholar] [CrossRef]
- Lv, D.; Wang, R.; Tang, G.; Mou, Z.; Lei, J.; Han, J.; De Smedt, S.; Xiong, R.; Huang, C. Ecofriendly Electrospun Membranes Loaded with Visible-Light-Responding Nanoparticles for Multifunctional Usages: Highly Efficient Air Filtration, Dye Scavenging, and Bactericidal Activity. ACS Appl. Mater. Interfaces 2019, 11, 12880–12889. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dowd, K.O.; Nair, K.M.; Forouzandeh, P.; Mathew, S.; Grant, J.; Moran, R.; Bartlett, J.; Bird, J.; Pillai, S.C. Face Masks and Respirators in the Fight Against the COVID-19 Pandemic: A Review of Current Materials, Advances and Future Perspectives. Materials 2020, 13, 3363. [Google Scholar] [CrossRef]
- Hao, W.; Parasch, A.; Williams, S.; Li, J.; Ma, H.; Burken, J.; Wang, Y. Filtration Performances of Non-Medical Materials as Candidates for Manufacturing Facemasks and Respirators. Int. J. Hyg. Environ. Health 2020, 229, 113582. [Google Scholar] [CrossRef]
- Nam, C.; Lee, S.; Ryu, M.; Lee, J.; Lee, H. Electrospun Nanofiber Filters for Highly Efficient PM2.5 Capture. Korean J. Chem. Eng. 2019, 36, 1565–1574. [Google Scholar] [CrossRef]
- Kadam, V.V.; Wang, L.; Padhye, R. Electrospun Nanofibre Materials to Filter Air Pollutants—A Review. J. Ind. Text. 2018, 47, 2253–2280. [Google Scholar] [CrossRef]
- Feng, J.P.; Wang, J.; Hwang, W.T.; Jo, Y.M. Characterization of Filter Media Prepared from Aligned Nanofibers for Fine Dust Screen. J. Appl. Polym. Sci. 2019, 136, 1–13. [Google Scholar] [CrossRef]
- Al-Attabi, R.; Morsi, Y.; Kujawski, W.; Kong, L.; Schütz, J.A.; Dumée, L.F. Wrinkled Silica Doped Electrospun Nano-Fiber Membranes with Engineered Roughness for Advanced Aerosol Air Filtration. Sep. Purif. Technol. 2019, 215, 500–507. [Google Scholar] [CrossRef]
- Mamun, A.; Blachowicz, T. Electrospun Nanofiber Mats for Filtering Applications—Technology, Structure and Materials. Polymers 2021, 13, 1368. [Google Scholar] [CrossRef]
- Lyu, C.; Zhao, P.; Xie, J.; Dong, S.; Liu, J.; Rao, C.; Fu, J. Electrospinning of Nanofibrous Membrane and Its Applications in Air Filtration: A Review. Nanomaterials 2021, 11, 1501. [Google Scholar] [CrossRef] [PubMed]
- Aamer, H.; Heo, S.; Jo, Y. Characterization of Multifunctional PAN/ZnO Nanofibrous Composite Filter for Fine Dust Capture and Photocatalytic Activity. J. Appl. Polym. Sci. 2021, 138, 50607. [Google Scholar] [CrossRef]
- Al-attabi, R.; Kong, L. High Efficiency Poly (Acrylonitrile) Electrospun Nanofiber Membranes for Airborne Nanomaterials Filtration. Adv. Eng. Mater. 2017, 20, 1700572. [Google Scholar] [CrossRef]
- Li, Y.; Yin, X.; Yu, J.; Ding, B. Electrospun Nanofibers for High-Performance Air Filtration. Compos. Commun. 2019, 15, 6–19. [Google Scholar] [CrossRef]
- Liu, K.; Liu, C.; Hsu, P.C.; Xu, J.; Kong, B.; Wu, T.; Zhang, R.; Zhou, G.; Huang, W.; Sun, J.; et al. Core-Shell Nanofibrous Materials with High Particulate Matter Removal Efficiencies and Thermally Triggered Flame Retardant Properties. ACS Cent. Sci. 2018, 4, 894–898. [Google Scholar] [CrossRef]
- Su, J.; Yang, G.; Cheng, C.; Huang, C.; Xu, H.; Ke, Q. Hierarchically Structured TiO2/PAN Nanofibrous Membranes for High-Efficiency Air Filtration and Toluene Degradation. J. Colloid Interface Sci. 2017, 507, 386–396. [Google Scholar] [CrossRef] [PubMed]
- Shalaby, T.; Hamad, H.; Ibrahim, E.; Mahmoud, O.; Al-oufy, A. Electrospun Nano Fibers Hybrid Composites Membranes for Highly efficient Antibacterial Activity. Ecotoxicol. Environ. Saf. 2018, 162, 354–364. [Google Scholar] [CrossRef]
- Wan, H.; Wang, N.; Yang, J.; Si, Y.; Chen, K.; Ding, B.; Sun, G.; El-newehy, M.; Al-deyab, S.S.; Yu, J. Hierarchically Structured Polysulfone/Titania Fibrous Membranes with Enhanced Air Filtration Performance. J. Colloid Interface Sci. 2014, 417, 18–26. [Google Scholar] [CrossRef]
- Ponnamma, D.; Cabibihan, J.; Rajan, M.; Pethaiah, S.S.; Deshmukh, K.; Prasad, J.; Pasha, S.K.K.; Ahamed, M.B.; Krishnegowda, J.; Chandrashekar, B.N.; et al. Synthesis, Optimization and Applications of ZnO/Polymer Nanocomposites. Mater. Sci. Eng. C 2019, 98, 1210–1240. [Google Scholar] [CrossRef]
- Gold, K.; Slay, B.; Knackstedt, M.; Gaharwar, A.K. Antimicrobial Activity of Metal and Metal-Oxide Based Nanoparticles. Adv. Ther. 2018, 1, 1700033. [Google Scholar] [CrossRef]
- Dizaj, S.M.; Lotfipour, F.; Barzegar-Jalali, M.; Zarrintan, M.H.; Adibkia, K. Antimicrobial Activity of the Metals and Metal Oxide Nanoparticles. Mater. Sci. Eng. C 2014, 44, 278–284. [Google Scholar] [CrossRef]
- Ghanbari, F.; Eskandari, M.; Nazari, P.; Gharibzadeh, S.; Kohnehpoushi, S.; Nejand, B.A. Potential Continuous Removal of Toluene by ZnO Nanorods Grown on Permeable Alumina Tube Filters. RSC Adv. 2016, 6, 52360–52371. [Google Scholar] [CrossRef]
- Li, X.W.; Song, R.G.; Jiang, Y.; Wang, C.; Jiang, D. Surface Modification of TiO2 Nanoparticles and Its Effect on the Properties of Fluoropolymer/TiO2 Nanocomposite Coatings. Appl. Surf. Sci. 2013, 276, 761–768. [Google Scholar] [CrossRef]
- Kim, M.; Fang, B.; Chaudhari, N.K.; Song, M.; Bae, T.; Yu, J. A Highly Efficient Synthesis Approach of Supported Pt-Ru Catalyst for Direct Methanol Fuel Cell. Electrochim. Acta 2010, 55, 4543–4550. [Google Scholar] [CrossRef]
- Ding, X.; Li, Y.; Si, Y.; Yin, X.; Yu, J.; Ding, B. Electrospun Polyvinylidene Fluoride/SiO2 Nanofibrous Membranes with Enhanced Electret Property for Efficient Air Filtration. Compos. Commun. 2019, 13, 57–62. [Google Scholar] [CrossRef]
- Xie, Y.; Hill, C.A.S.; Xiao, Z.; Militz, H.; Mai, C. Silane Coupling Agents Used for Natural Fiber/Polymer Composites: A Review. Compos. Part A 2010, 41, 806–819. [Google Scholar] [CrossRef]
- Tang, E. Fabrication of Zinc Oxide/Poly (Styrene) Grafted Nanocomposite Latex and Its Dispersion. Eur. Polym. J. 2007, 43, 4210–4218. [Google Scholar] [CrossRef]
- Cui, L.; Hao, M.; Wang, F.; Fang, B.; Liang, J.; Zhu, M.; Xie, X. Microstructure Optimization of Mos 2/Sepiolite Nanocomposites via a Surfactant-Assisted Hydrothermal Strategy for High Efficiency Photocatalysis. Int. J. Photoenergy 2020, 2020, 8868782. [Google Scholar] [CrossRef]
- Xing, Y.; Wang, Y.; Zhou, C.; Zhang, S.; Fang, B. Simple Synthesis of Mesoporous Carbon Nano Fibers with Hierarchical Nanostructure for Ultrahigh Lithium Storage. ACS Appl. Mater. Interfaces 2014, 6, 2561–2567. [Google Scholar] [CrossRef]
- Chen, K.; Nur, F.; Sari, I.; Ting, J.-M. Multifunctional TiO 2/Polyacrylonitrile Nanofibers for High efficiency PM2. 5 Capture, UV Fi Lter, and Anti-Bacteria Activity. J. Appl. Surf. Sci. 2019, 493, 157–164. [Google Scholar] [CrossRef]
- Kim, S.C.; Kang, S.; Lee, H.; Kwak, D.; Ou, Q.; Pei, C.; Pui, D.Y.H. Nanofiber Filter Performance Improvement: Nanofiber Layer Uniformity and Branched Nanofiber. Aerosol Air Qual. Res. 2020, 1, 80–88. [Google Scholar] [CrossRef] [Green Version]
- Bortolassi, A.C.C.; Nagarajan, S.; de Araújo Lima, B.; Guerra, V.G.; Aguiar, M.L.; Huon, V.; Soussan, L.; Cornu, D.; Miele, P.; Bechelany, M. Efficient Nanoparticles Removal and Bactericidal Action of Electrospun Nanofibers Membranes for Air Filtration. Mater. Sci. Eng. C 2019, 102, 718–729. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, Y.; Qian, X.; Wang, L.; Qian, Y.; Bai, H.; Wang, X. Hierarchical Micro/Nanofibrous Filter for Effective Fine-Particle Capture. Powder Technol. 2020, 360, 1192–1199. [Google Scholar] [CrossRef]







| Sample ID | Silane/ZnO Ratio in Mass | Relative Mass of ZnO to Total Solid Content (%) |
|---|---|---|
| PZ9 | 0 | 9 |
| PZ9(0.5S) | 0.5 | 9 |
| PZ9(1S) | 1 | 9 |
| PZ9(2S) | 2 | 9 |
| PZ12 | 0 | 12 |
| PZ12(0.5S) | 0.5 | 12 |
| PZ12(1S) | 1 | 12 |
| PZ12(2S) | 2 | 12 |
| Sample | Particle Size (nm) | Standard Deviation (SD) |
|---|---|---|
| Z | 54.65 | 28.29 |
| Z(0.5S) | 40.82 | 15.91 |
| Z(1S) | 39.26 | 23.75 |
| Z(2S) | 41.20 | 19.54 |
| Sample | A (m2/g) | Vp (Total) (cm3/g) | Vmeso (cm3/g) | davg (nm) |
|---|---|---|---|---|
| PZ9 | 21 | 0.058 | 0.056 | 8.42 |
| PZ9(0.5S) | 22 | 0.061 | 0.060 | 8.55 |
| PZ9(1S) | 25 | 0.070 | 0.069 | 10.79 |
| PZ9(2S) | 29 | 0.081 | 0.079 | 12.32 |
| PZ12 | 24 | 0.064 | 0.062 | 11.53 |
| PZ12(0.5S) | 36 | 0.099 | 0.098 | 19.52 |
| PZ12(1S) | 33 | 0.092 | 0.090 | 18.01 |
| PZ12(2S) | 27 | 0.075 | 0.072 | 17.18 |
| Sample | Zn wt% |
|---|---|
| PZ9 | 4.81 |
| PZ9(0.5S) | 6.02 |
| PZ9(1S) | 6.41 |
| PZ9(2S) | 6.70 |
| PZ12 | 6.97 |
| PZ12(0.5S) | 9.76 |
| PZ12(1S) | 8.62 |
| PZ12(2S) | 7.76 |
| Polymer or Polymer Composite | Test Aerosol (Size in nm) | Face Velocity (cm/s) | Filtration Efficiency (%) | Pressure Drop (Pa) | Quality Factor (Pa−1) | References |
|---|---|---|---|---|---|---|
| ZnO@PVA/KGM | NaCl (100) | 5.3 | 99.99 | 130 | 0.0708 | [3] |
| PAN/TiO2 beads/P25 | NaCl (30–500) | NA | 96.75 | 88 | 0.039 | [30] |
| PEO | KCl (100) | 5 | 94.1 | 90.42 | 0.031 | [31] |
| Ag/PAN | NaCl (9–300) | 5 | 98.65 | 68 | 0.05 | [32] |
| PU/PET | NaCl (260) | 5.3 | 76.63 | 17.3 | 0.084 | [33] |
| PZ9 | KCl (300) | 5.3 | 96.64 | 52 | 0.065 | This work |
| PZ9(2S) | KCl (300) | 5.3 | 98.80 | 48 | 0.092 | This work |
| PZ12 | KCl (300) | 5.3 | 99.64 | 50 | 0.112 | This work |
| PZ(0.5S) | KCl (300) | 5.3 | 99.76 | 44 | 0.137 | This work |
| Sample | Photocatalytic Degradation Efficiency (%) |
|---|---|
| PZ9 | 67.65 |
| PZ9(0.5S) | 73.98 |
| PZ9(1S) | 77.21 |
| PZ9(2S) | 81.30 |
| PZ12 | 70.12 |
| PZ12(0.5S) | 85.11 |
| PZ12(1S) | 80.99 |
| PZ12(2S) | 75.99 |
| Sample | Zone of Inhibition (mm) |
|---|---|
| PZ0 | 0 |
| PZ9 | 18 |
| PZ9(0.5S) | 14 |
| PZ9(1S) | 18 |
| PZ9(2S) | 20 |
| PZ12 | 20 |
| PZ12(0.5S) | 23 |
| PZ12(1S) | 20 |
| PZ12(2S) | 17 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aamer, H.; Kim, S.-B.; Oh, J.-M.; Park, H.; Jo, Y.-M. ZnO-Impregnated Polyacrylonitrile Nanofiber Filters against Various Phases of Air Pollutants. Nanomaterials 2021, 11, 2313. https://doi.org/10.3390/nano11092313
Aamer H, Kim S-B, Oh J-M, Park H, Jo Y-M. ZnO-Impregnated Polyacrylonitrile Nanofiber Filters against Various Phases of Air Pollutants. Nanomaterials. 2021; 11(9):2313. https://doi.org/10.3390/nano11092313
Chicago/Turabian StyleAamer, Hanaa, Sang-Bum Kim, Jong-Min Oh, Hyeokjin Park, and Young-Min Jo. 2021. "ZnO-Impregnated Polyacrylonitrile Nanofiber Filters against Various Phases of Air Pollutants" Nanomaterials 11, no. 9: 2313. https://doi.org/10.3390/nano11092313
APA StyleAamer, H., Kim, S.-B., Oh, J.-M., Park, H., & Jo, Y.-M. (2021). ZnO-Impregnated Polyacrylonitrile Nanofiber Filters against Various Phases of Air Pollutants. Nanomaterials, 11(9), 2313. https://doi.org/10.3390/nano11092313






