Magnetic Nanoprobes for Spatio-Mechanical Manipulation in Single Cells
Abstract
:1. Introduction
2. Materials and Methods
3. Results and Discussion
3.1. MNP Characterization
3.2. MNP Cellular Uptake
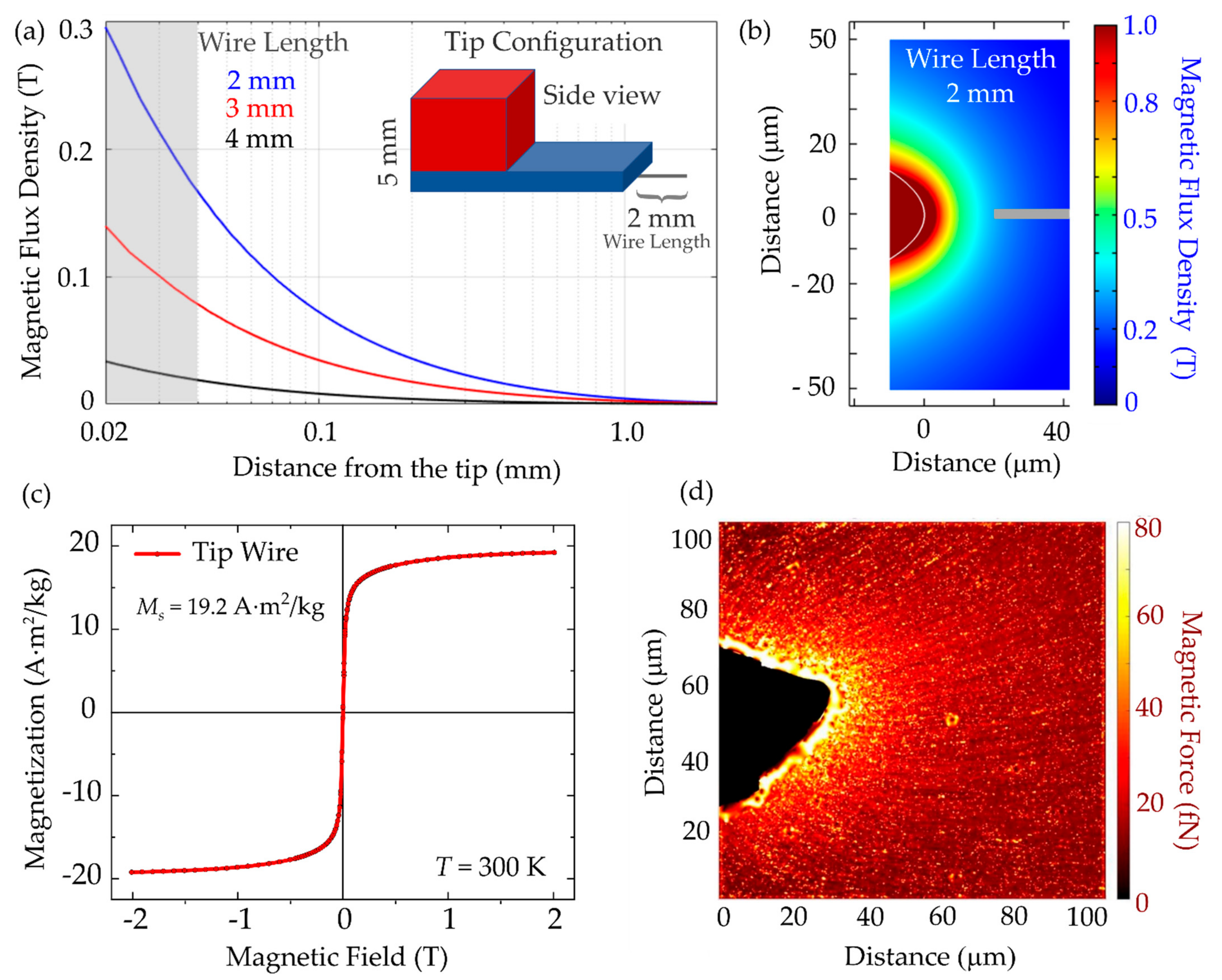
3.3. Magnetic Tip Characterization
3.4. MNP Manipulation in Cells
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Anik, M.I.; Hossain, M.K.; Hossain, I.; Mahfuz, A.M.U.B.; Rahman, M.T.; Ahmed, I. Recent Progress of Magnetic Nanoparticles in Biomedical Applications: A Review. Nano Sel. 2021, 2, 1146–1186. [Google Scholar] [CrossRef]
- Kim, C.; Kim, H.; Park, H.; Lee, K.Y. Controlling the Porous Structure of Alginate Ferrogel for Anticancer Drug Delivery under Magnetic Stimulation. Carbohydr. Polym. 2019, 223, 115045. [Google Scholar] [CrossRef] [PubMed]
- Pavlov, A.M.; Gabriel, S.A.; Sukhorukov, G.B.; Gould, D.J. Improved and Targeted Delivery of Bioactive Molecules to Cells with Magnetic Layer-by-Layer Assembled Microcapsules. Nanoscale 2015, 7, 9686–9693. [Google Scholar] [CrossRef] [PubMed]
- Moghram, W.I.; Kruger, A.; Sander, E.A.; Selby, J.C. Magnetic Tweezers with Magnetic Flux Density Feedback Control. Rev. Sci. Instrum. 2021, 92, 034101. [Google Scholar] [CrossRef] [PubMed]
- Li, J.H.; Santos-Otte, P.; Au, B.; Rentsch, J.; Block, S.; Ewers, H. Directed Manipulation of Membrane Proteins by Fluorescent Magnetic Nanoparticles. Nat. Commun. 2020, 11, 4259. [Google Scholar] [CrossRef]
- Pankhurst, Q.A.; Thanh, N.K.T.; Jones, S.K.; Dobson, J. Progress in Applications of Magnetic Nanoparticles in Biomedicine. J. Phys. D Appl. Phys. 2009, 42, 224001. [Google Scholar] [CrossRef] [Green Version]
- Sangaiya, P.; Jayaprakash, R. A Review on Iron Oxide Nanoparticles and Their Biomedical Applications. J. Supercond. Nov. Magn. 2018, 31, 3397–3413. [Google Scholar] [CrossRef]
- Kurlyandskaya, G.V.; Novoselova, I.P.; Schupletsova, V.V.; Andrade, R.; Dunec, N.A.; Litvinova, L.S.; Safronov, A.P.; Yurova, K.A.; Kulesh, N.A.; Dzyuman, A.N.; et al. Nanoparticles for Magnetic Biosensing Systems. J. Magn. Magn. Mater. 2017, 431, 249–254. [Google Scholar] [CrossRef]
- Avugadda, S.K.; Wickramasinghe, S.; Niculaes, D.; Ju, M.; Lak, A.; Silvestri, N.; Nitti, S.; Roy, I.; Samia, A.C.S.; Pellegrino, T. Uncovering the Magnetic Particle Imaging and Magnetic Resonance Imaging Features of Iron Oxide Nanocube Clusters. Nanomaterials 2021, 11, 62. [Google Scholar]
- Etoc, F.; Lisse, D.; Bellaiche, Y.; Piehler, J.; Coppey, M.; Dahan, M. Subcellular Control of Rac-GTPase Signalling by Magnetogenetic Manipulation Inside Living Cells. Nat. Nanotechnol. 2013, 8, 193–198. [Google Scholar] [CrossRef]
- Seo, D.; Southard, K.M.; Kim, J.; Lee, H.J.; Farlow, J.; Lee, J.; Litt, D.B.; Haas, T.; Alivisatos, A.P.; Cheon, J.; et al. A Mechanogenetic Toolkit for Interrogating Cell Signaling in Space and Time. Cell 2016, 165, 1507–1518. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, H.; Delikanli, S.; Zeng, H.; Ferkey, D.M.; Pralle, A. Remote Control of Ion Channels and Neurons through Magnetic-Field Heating of Nanoparticles. Nat. Nanotechnol. 2010, 5, 602–606. [Google Scholar] [CrossRef] [PubMed]
- Liang, Y.; Xie, J.; Yu, J.; Zheng, Z.; Liu, F.; Yang, A. Recent Advances of High Performance Magnetic Iron Oxide Nanoparticles: Controlled Synthesis, Properties Tuning and Cancer Theranostics. Nano Sel. 2021, 2, 216–250. [Google Scholar] [CrossRef]
- Chasteen, N.D.; Harrison, P.M. Mineralization in Ferritin: An Efficient Means of Iron Storage. J. Struct. Biol. 1999, 126, 182–194. [Google Scholar] [CrossRef] [Green Version]
- Aronovitz, N.; Neeman, M.; Zarivach, R. Ferritin Iron Mineralization and Storage: From Structure to Function. In Iron Oxides: From Nature to Applications, 1st ed.; Faivre, D., Ed.; Wiley-VCH: Weinheim, Germany, 2016; pp. 117–141. [Google Scholar]
- Meldrum, F.C.; Heywood, B.R.; Mann, S. Magnetoferritin: In Vitro Synthesis of a Novel Magnetic Protein. Science 1992, 257, 522–523. [Google Scholar] [CrossRef]
- Klem, M.T.; Resnick, D.A.; Gilmore, K.; Young, M.; Idzerda, Y.U.; Douglas, T. Synthetic Control over Magnetic Moment and Exchange Bias in All-Oxide Materials Encapsulated within a Spherical Protein Cage. J. Am. Chem. Soc. 2007, 129, 197–201. [Google Scholar] [CrossRef]
- Yang, C.; Cao, C.; Cai, Y.; Xu, H.; Zhang, T.; Pan, Y. Effects of PEGylation on Biomimetic Synthesis of Magnetoferritin Nanoparticles. J. Nanoparticle Res. 2017, 19, 101. [Google Scholar] [CrossRef]
- Pankhurst, Q.A.; Connolly, J.; Jones, S.K.; Dobson, J. Applications of Magnetic Nanoparticles in Biomedicine. J. Phys. D Appl. Phys. 2003, 36, R167–R181. [Google Scholar] [CrossRef] [Green Version]
- Chen, R.; Romero, G.; Christiansen, M.G.; Mohr, A.; Anikeeva, P. Wireless Magnetothermal Deep Brain Stimulation. Science 2015, 347, 1477–1480. [Google Scholar] [CrossRef] [Green Version]
- Liße, D.; Monzel, C.; Vicario, C.; Manzi, J.; Maurin, I.; Coppey, M.; Piehler, J.; Dahan, M. Engineered Ferritin for Magnetogenetic Manipulation of Proteins and Organelles Inside Living Cells. Adv. Mater. 2017, 29, 1700189. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Sánchez, M.E.; Barbier, S.; Whitehead, J.; Béalle, G.; Michel, A.; Latorre-Ossa, H.; Rey, C.; Fouassier, L.; Claperon, A.; Brullé, L.; et al. Mechanical Induction of the Tumorigenic β-Catenin Pathway by Tumour Growth Pressure. Nature 2015, 523, 92–95. [Google Scholar] [CrossRef]
- Monzel, C.; Vicario, C.; Piehler, J.; Coppey, M.; Dahan, M. Magnetic Control of Cellular Processes Using Biofunctional Nanoparticles. Chem. Sci. 2017, 8, 7330–7338. [Google Scholar] [CrossRef] [Green Version]
- Laemmli, U.K. Cleavage of Structural Proteins during the Assembly of the Head of Bacteriophage T4. Nature 1970, 227, 680–685. [Google Scholar] [CrossRef]
- Schindelin, J.; Arganda-Carreras, I.; Frise, E.; Kaynig, V.; Longair, M.; Pietzsch, T.; Preibisch, S.; Rueden, C.; Saalfeld, S.; Schmid, B.; et al. Fiji: An Open-Source Platform for Biological-Image Analysis. Nat. Methods 2012, 9, 676–682. [Google Scholar] [CrossRef] [Green Version]
- Gavilán, H.; Kowalski, A.; Heinke, D.; Sugunan, A.; Sommertune, J.; Varón, M.; Bogart, L.K.; Posth, O.; Zeng, L.; González-Alonso, D.; et al. Colloidal Flower-Shaped Iron Oxide Nanoparticles: Synthesis Strategies and Coatings. Part. Part. Syst. Charact. 2017, 34, 1700094. [Google Scholar] [CrossRef] [Green Version]
- Bender, P.; Fock, J.; Frandsen, C.; Hansen, M.F.; Balceris, C.; Ludwig, F.; Posth, O.; Wetterskog, E.; Bogart, L.K.; Southern, P.; et al. Relating Magnetic Properties and High Hyperthermia Performance of Iron Oxide Nanoflowers. J. Phys. Chem. C 2018, 122, 3068–3077. [Google Scholar] [CrossRef] [Green Version]
- Vogel, P.; Kampf, T.; Rückert, M.; Grüttner, C.; Kowalski, A.; Teller, H.; Behr, V. Synomag®: The New High-Performance Tracer for Magnetic Particle Imaging. Int. J. Magn. Part. Imaging 2021, 7, 1–6. [Google Scholar]
- Griese, F.; Knopp, T.; Gruettner, C.; Thieben, F.; Müller, K.; Loges, S.; Ludewig, P.; Gdaniec, N. Simultaneous Magnetic Particle Imaging and Navigation of Large Superparamagnetic Nanoparticles in Bifurcation Flow Exper-Iments. J. Magn. Magn. Mater. 2020, 498, 166206. [Google Scholar] [CrossRef] [Green Version]
- McNicholas, S.; Potterton, E.; Wilson, K.S.; Noble, M.E.M. Presenting Your Structures: The CCP4mg Molecular-Graphics Software. Acta Crystallogr. Sect. D Biol. Crystallogr. 2011, 67, 386–394. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Etoc, F.; Vicario, C.; Lisse, D.; Siaugue, J.-M.; Piehler, J.; Coppey, M.; Dahan, M. Magnetogenetic Control of Protein Gradients Inside Living Cells with High Spatial and Temporal Resolution. Nano Lett. 2015, 15, 3487–3494. [Google Scholar] [CrossRef]
- Etoc, F.; Balloul, E.; Vicario, C.; Normanno, D.; Liße, D.; Sittner, A.; Piehler, J.; Dahan, M.; Coppey, M. Non-Specific Interactions Govern Cytosolic Diffusion of Nanosized Objects in Mammalian Cells. Nat. Mater. 2018, 17, 740–746. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gupta, A.K.; Gupta, M. Synthesis and Surface Engineering of Iron Oxide Nanoparticles for Biomedical Applications. Biomaterials 2005, 26, 3995–4021. [Google Scholar] [CrossRef]
- Harrison, P.M.; Arosio, P. The Ferritins: Molecular Properties, Iron Storage Function and Cellular Regulation. Biochim. Biophys. Acta-Bioenerg. 1996, 1275, 161–203. [Google Scholar] [CrossRef] [Green Version]
- Fan, K.; Cao, C.; Pan, Y.; Lu, D.; Yang, D.; Feng, J.; Song, L.; Liang, M.; Yan, X. Magnetoferritin Nanoparticles for Targeting and Visualizing Tumour Tissues. Nat. Nanotechnol. 2012, 7, 459–464. [Google Scholar] [CrossRef]
- Kusterle, M.; Jevsevar, S.; Porekar, V.G. Size of Pegylated Protein Conjugates Studied by Various Methods. Acta Chim. Slov. 2008, 55, 594–601. [Google Scholar]
- Novoselova, I.P.; Safronov, A.P.; Samatov, O.M.; Medvedev, A.I.; Kurlyandskaya, G.V. Biocompatible Ferrofluids with Iron Oxide Nanoparticles Fabricated by Laser Target Evaporation. IEEE Magn. Lett. 2015, 6, 6–9. [Google Scholar] [CrossRef]
- Debayle, M.; Balloul, E.; Dembele, F.; Xu, X.; Hanafi, M.; Ribot, F.; Monzel, C.; Coppey, M.; Fragola, A.; Dahan, M.; et al. Zwitterionic Polymer Ligands: An Ideal Surface Coating To Totally Suppress Protein-Nanoparticle Corona Formation? Biomaterials 2019, 219, 119357. [Google Scholar] [CrossRef] [Green Version]
- Lim, J.; Yeap, S.P.; Che, H.X.; Low, S.C. Characterization of Magnetic Nanoparticle by Dynamic Light Scattering. Nanoscale Res. Lett. 2013, 8, 381. [Google Scholar] [CrossRef] [Green Version]
- Forest, V.; Cottier, M.; Pourchez, J. Electrostatic Interactions Favor the Binding of Positive Nanoparticles on Cells: A Reductive Theory. Nano Today 2015, 10, 677–680. [Google Scholar] [CrossRef] [Green Version]
- Schavemaker, P.E.; Śmigiel, W.M.; Poolman, B. Ribosome Surface Properties May Impose Limits on the Nature of the Cytoplasmic Proteome. eLife 2017, 6, e30084. [Google Scholar] [CrossRef] [PubMed]
- Liße, D.; Richter, C.P.; Drees, C.; Birkholz, O.; You, C.; Rampazzo, E.; Piehler, J. Monofunctional Stealth Nanoparticle for Unbiased Single Molecule Tracking Inside Living Cells. Nano Lett. 2014, 14, 2189–2195. [Google Scholar] [CrossRef]
- Coey, J.M.D. Magnetism and Magnetic Materials, 1st ed.; Cambridge University Press: Cambridge, UK, 2010. [Google Scholar]
- Skoropata, E.; Desautels, R.D.; Falvo, E.; Ceci, P.; Kasyutich, O.; Freeland, J.W.; van Lierop, J. Intra–and Interparticle Magnetism of Cobalt-Doped Iron-Oxide Nanoparticles Encapsulated in a Synthetic Ferritin Cage. Phys. Rev. B. 2014, 90, 174424. [Google Scholar] [CrossRef] [Green Version]
- Massner, C.; Sigmund, F.; Pettinger, S.; Seeger, M.; Hartmann, C.; Ivleva, N.P.; Niessner, R.; Fuchs, H.; Hrabě de Angelis, M.; Stelzl, A.; et al. Genetically Controlled Lysosomal Entrapment of Superparamagnetic Ferritin for Multimodal and Multiscale Imaging and Actuation with Low Tissue Attenuation. Adv. Funct. Mater. 2018, 28, 1706793. [Google Scholar] [CrossRef]
- Aragón, R. Cubic Magnetic Anisotropy of Nonstoichiometric Magnetite. Phys. Rev. B. 1992, 46, 5334–5338. [Google Scholar] [CrossRef]
- Efremova, M.V.; Nalench, Y.A.; Myrovali, E.; Garanina, A.S.; Grebennikov, I.S.; Gifer, P.K.; Abakumov, M.A.; Spasova, M.; Angelakeris, M.; Savchenko, A.G.; et al. SIze-Selected Fe3O4–Au Hybrid Nanoparticles for Improved Magnetism-Based Theranostics. Beilstein J. Nanotechnol. 2018, 9, 2684–2699. [Google Scholar] [CrossRef] [Green Version]
- Phong, P.T.; Oanh, V.T.K.; Lam, T.D.; Phuc, N.X.; Tung, L.D.; Thanh, N.T.K.; Manh, D.H. Iron Oxide Nanoparticles: Tunable Size Synthesis and Analysis in Terms of the Core–Shell Structure and Mixed Coercive Model. J. Electron. Mater. 2017, 46, 2533–2539. [Google Scholar] [CrossRef]
- Nayek, C.; Manna, K.; Imam, A.A.; Alqasrawi, A.Y.; Obaidat, I.M. Size-Dependent Magnetic Anisotropy of PEG Coated Fe3O4 Nanoparticles; Comparing Two Magnetization Methods. IOP Conf. Ser. Mater. Sci. Eng. 2018, 305, 012012. [Google Scholar] [CrossRef]
- Jiang, Z.; Shan, K.; Song, J.; Liu, J.; Rajendran, S.; Pugazhendhi, A.; Jacob, J.A.; Chen, B. Toxic Effects of Magnetic Nanoparticles on Normal Cells and Organs. Life Sci. 2019, 220, 156–161. [Google Scholar] [CrossRef]
- Lartigue, L.; Hugounenq, P.; Alloyeau, D.; Clarke, S.P.; Lévy, M.; Bacri, J.-C.; Bazzi, R.; Brougham, D.F.; Wilhelm, C.; Gazeau, F. Cooperative Organization in Iron Oxide Multi-Core Nanoparticles Potentiates Their Efficiency as Heating Mediators and MRI Contrast Agents. ACS Nano 2012, 6, 10935–10949. [Google Scholar] [CrossRef]
- Guardia, P.; Di Corato, R.; Lartigue, L.; Wilhelm, C.; Espinosa, A.; Garcia-Hernandez, M.; Gazeau, F.; Manna, L.; Pellegrino, T. Water-Soluble Iron Oxide Nanocubes with High Values of Specific Absorption Rate for Cancer Cell Hyperthermia Treatment. ACS Nano 2012, 6, 3080–3091. [Google Scholar] [CrossRef]
- Curcio, A.; Silva, A.K.A.; Cabana, S.; Espinosa, A.; Baptiste, B.; Menguy, N.; Wilhelm, C.; Abou-Hassan, A. Iron Oxide Nanoflowers @ CuS Hybrids for Cancer Tri-Therapy: Interplay of Photothermal Therapy, Magnetic Hyperthermia and PhotoDynamic Therapy. Theranostics 2019, 9, 1288–1302. [Google Scholar] [CrossRef]
- Salatin, S.; Dizaj, S.M.; Khosroushahi, A.Y. Effect of the Surface Modification, Size, and Shape on Cellular Uptake of Nanoparticles. Cell Biol. Int. 2015, 39, 881–890. [Google Scholar] [CrossRef] [PubMed]
- Tinevez, J.Y.; Perry, N.; Schindelin, J.; Hoopes, G.M.; Reynolds, G.D.; Laplantine, E.; Bednarek, S.Y.; Shorte, S.L.; Eliceiri, K.W. TrackMate: An Open and Extensible Platform for Single-Particle Tracking. Methods 2017, 115, 80–90. [Google Scholar] [CrossRef]
- Tseng, P.; Judy, J.W.; Di Carlo, D. Magnetic Nanoparticle-Mediated Massively Parallel Mechanical Modulation of Single-Cell Behavior. Nat. Methods 2012, 9, 1113–1119. [Google Scholar] [CrossRef] [Green Version]
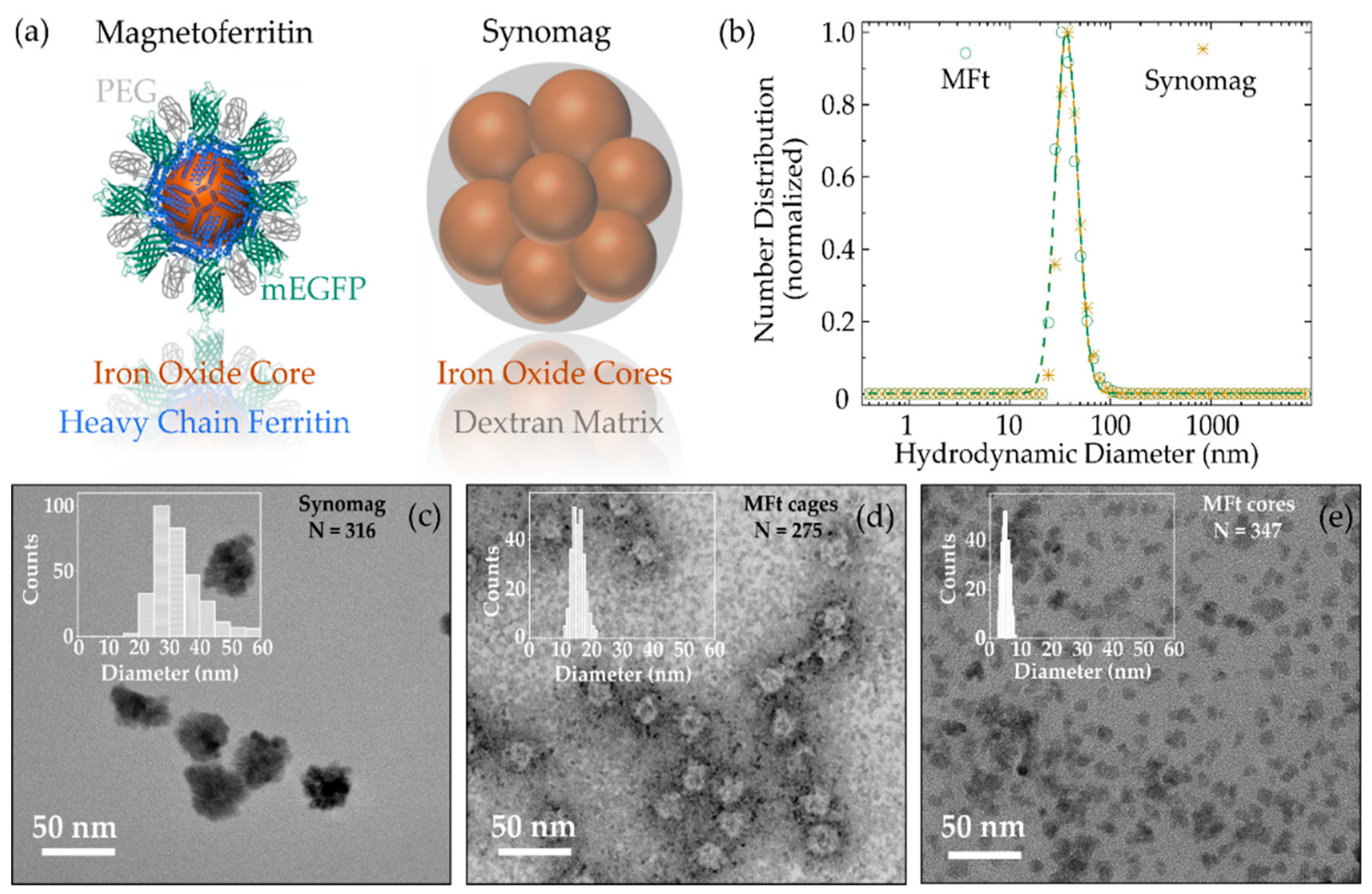





| Sample | PdI | DH, nm | DTEM, nm | ζ-Potential, mV |
|---|---|---|---|---|
| PEGylated magnetoferritin | 0.11 ± 0.01 | 39 ± 3 | Cages: 16 ± 2 Cores: 5 ± 1 | −3.7 |
| PEGylated Synomag | 0.17 ± 0.03 | 39 ± 2 | 34 ± 8 | −2.0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Novoselova, I.P.; Neusch, A.; Brand, J.-S.; Otten, M.; Safari, M.R.; Bartels, N.; Karg, M.; Farle, M.; Wiedwald, U.; Monzel, C. Magnetic Nanoprobes for Spatio-Mechanical Manipulation in Single Cells. Nanomaterials 2021, 11, 2267. https://doi.org/10.3390/nano11092267
Novoselova IP, Neusch A, Brand J-S, Otten M, Safari MR, Bartels N, Karg M, Farle M, Wiedwald U, Monzel C. Magnetic Nanoprobes for Spatio-Mechanical Manipulation in Single Cells. Nanomaterials. 2021; 11(9):2267. https://doi.org/10.3390/nano11092267
Chicago/Turabian StyleNovoselova, Iuliia P., Andreas Neusch, Julia-Sarita Brand, Marius Otten, Mohammad Reza Safari, Nina Bartels, Matthias Karg, Michael Farle, Ulf Wiedwald, and Cornelia Monzel. 2021. "Magnetic Nanoprobes for Spatio-Mechanical Manipulation in Single Cells" Nanomaterials 11, no. 9: 2267. https://doi.org/10.3390/nano11092267
APA StyleNovoselova, I. P., Neusch, A., Brand, J.-S., Otten, M., Safari, M. R., Bartels, N., Karg, M., Farle, M., Wiedwald, U., & Monzel, C. (2021). Magnetic Nanoprobes for Spatio-Mechanical Manipulation in Single Cells. Nanomaterials, 11(9), 2267. https://doi.org/10.3390/nano11092267







