Stable Thermally-Modulated Nanodroplet Ultrasound Contrast Agents
Abstract
1. Introduction
2. Materials and Methods
2.1. Nanodroplet Formulation and Characterization
2.1.1. BSA-Shelled Tetradecafluorohexane Nanodroplet Formulation
2.1.2. Nanodroplet Characterization
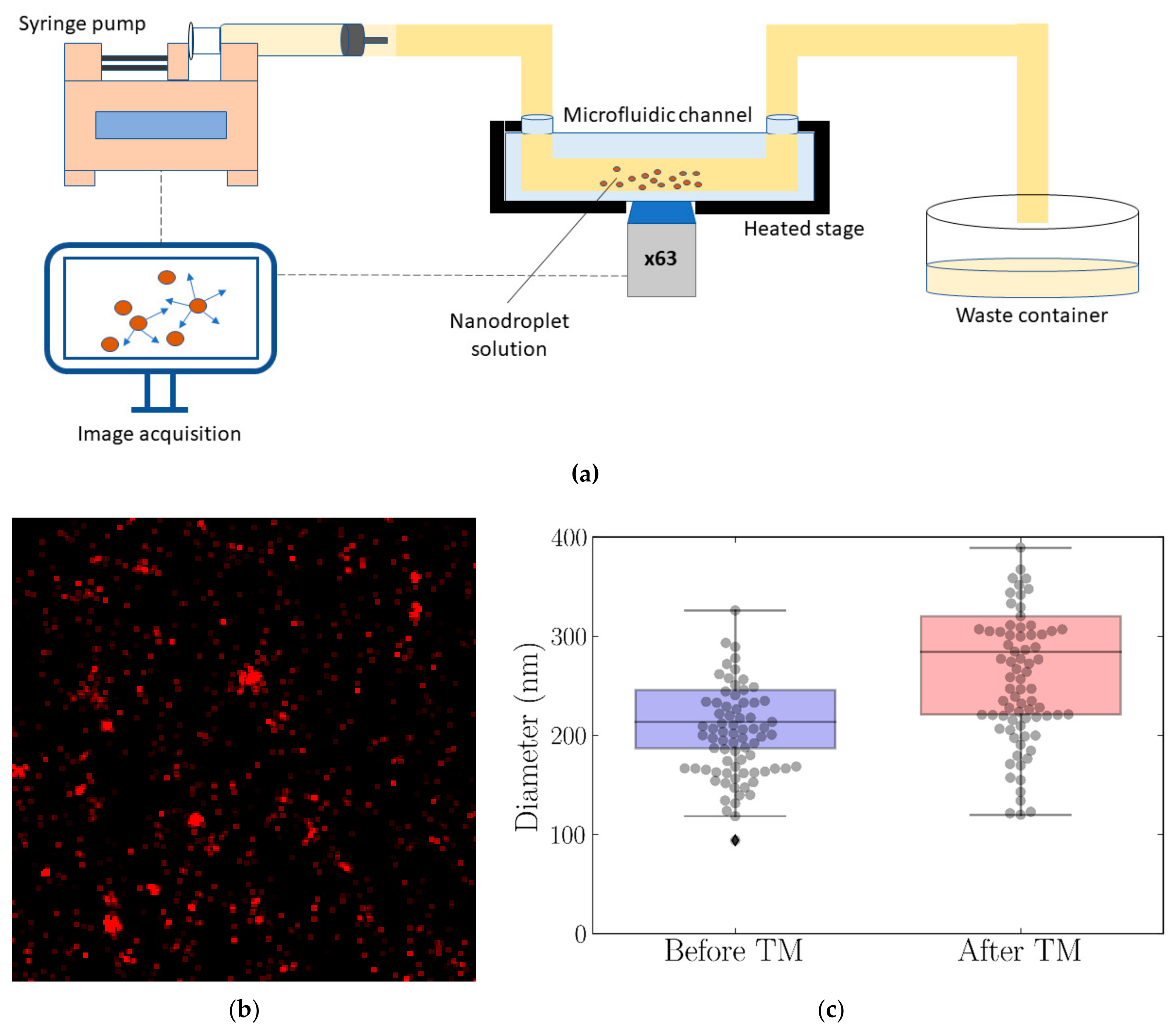
2.2. Echogenicity Characterization
2.2.1. Repeatability of Thermal Modulation
2.2.2. Stability
2.2.3. Temperature Sweep
2.3. Statistical Analysis
3. Results
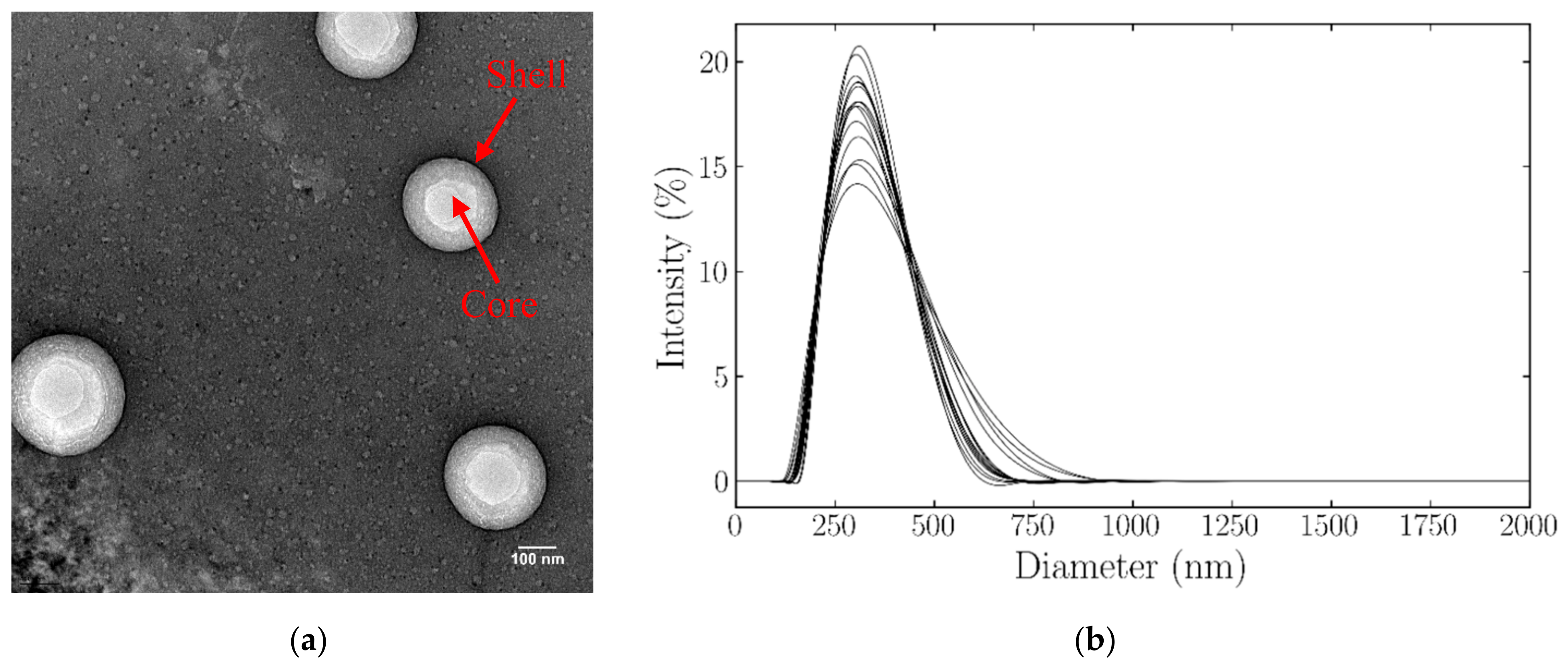
3.1. Nanodroplet Size Distribution and Structural Property
3.2. In Vitro Thermal Modulation: Contrast Enhancement and Repeatability
3.3. Nanodroplet Stability after Heat Modulation
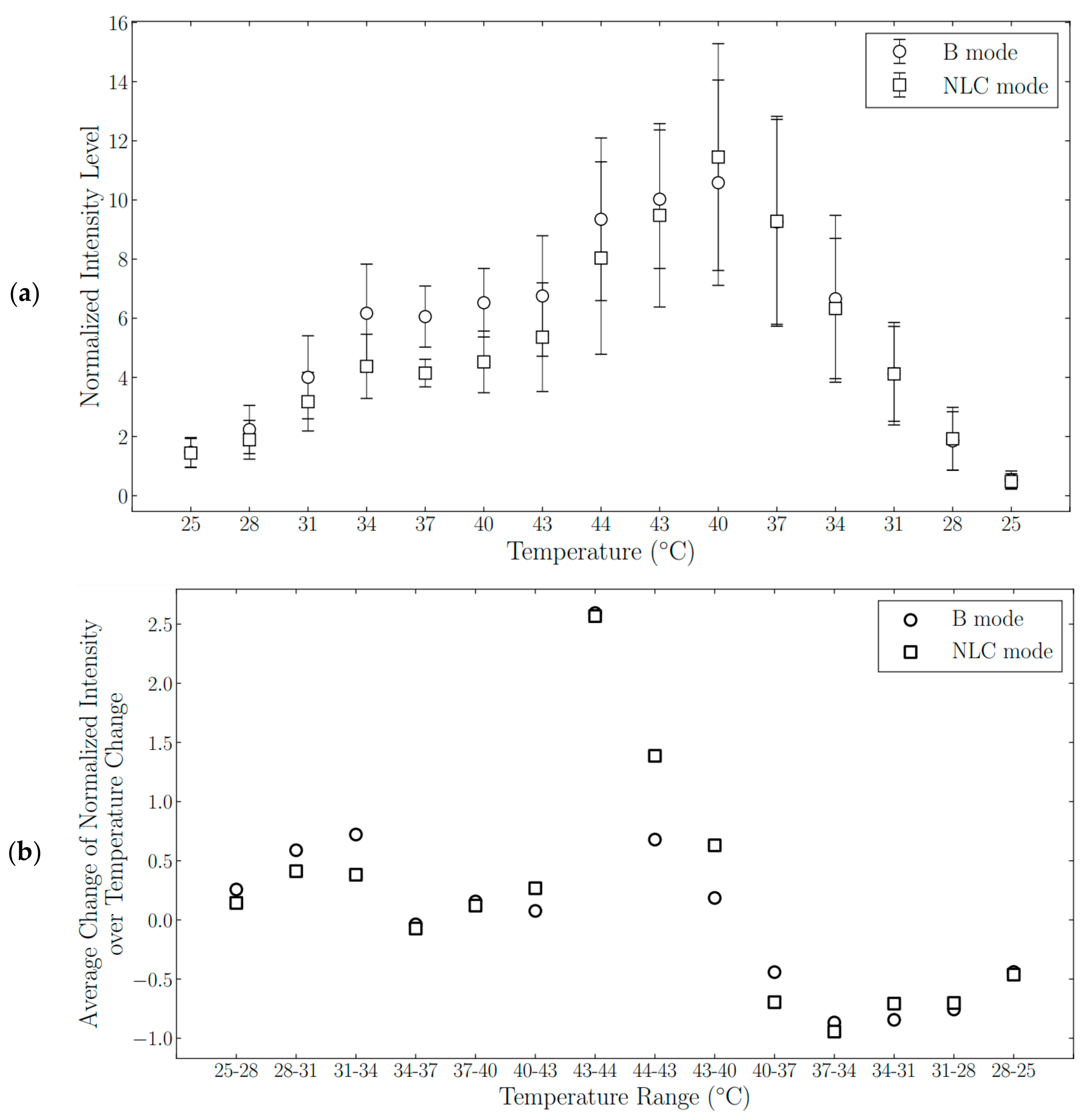
3.4. Nanodroplet Echogenicity as a Function of Temperature
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Contreras-Ortiz, S.H.; Chiu, T.; Fox, M.D. Ultrasound image enhancement: A review. Biomed. Signal Process. Control 2012, 7, 419–428. [Google Scholar] [CrossRef]
- Deshpande, N.; Needles, A.; Willmann, J.K. Molecular ultrasound imaging: Current status and future directions. Clin. Radiol. 2010, 65, 567–581. [Google Scholar] [CrossRef]
- Datta, S.; Coussios, C.C.; Ammi, A.Y.; Mast, T.D.; de Courten-Myers, G.M.; Holland, C.K. Ultrasound-Enhanced Thrombolysis Using Definity® as a Cavitation Nucleation Agent. Ultrasound Med. Biol. 2008, 34, 1421–1433. [Google Scholar] [CrossRef]
- Perera, R.H.; Solorio, L.; Wu, H.; Gangolli, M.; Silverman, E.; Hernandez, C.; Peiris, P.M.; Broome, A.-M.; Exner, A.A. Nanobubble Ultrasound Contrast Agents for Enhanced Delivery of Thermal Sensitizer to Tumors Undergoing Radiofrequency Ablation. Pharm. Res. 2014, 31, 1407–1417. [Google Scholar] [CrossRef]
- Wang, M.; Zhang, Y.; Cai, C.; Tu, J.; Guo, X.; Zhang, D. Sonoporation-induced cell membrane permeabilization and cytoskeleton disassembly at varied acoustic and microbubble-cell parameters. Sci. Rep. 2018, 8, 3885. [Google Scholar] [CrossRef]
- Hernot, S.; Klibanov, A.L. Microbubbles in ultrasound-triggered drug and gene delivery. Adv. Drug Deliv. Rev. 2008, 60, 1153–1166. [Google Scholar] [CrossRef]
- Muskula, P.R.; Main, M.L. Safety with Echocardiographic Contrast Agents. Circ. Cardiovasc. Imaging 2017, 10, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Goodwin, A.P.; Nakatsuka, M.A.; Mattrey, R.F. Stimulus-responsive ultrasound contrast agents for clinical imaging: Motivations, demonstrations, and future directions. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnology 2015, 7, 111–123. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wu, S.-K.; Chu, P.-C.; Chai, W.-Y.; Kang, S.-T.; Tsai, C.-H.; Fan, C.-H.; Yeh, C.-K.; Liu, H.-L. Characterization of Different Microbubbles in Assisting Focused Ultrasound-Induced Blood-Brain Barrier Opening. Sci. Rep. 2017, 7, 46689. [Google Scholar] [CrossRef]
- Krupka, T.M.; Solorio, L.; Wilson, R.E.; Wu, H.; Azar, N.; Exner, A.A. Formulation and Characterization of Echogenic Lipid−Pluronic Nanobubbles. Mol. Pharm. 2010, 7, 49–59. [Google Scholar] [CrossRef] [PubMed]
- Bin Cai, W.; Yang, H.L.; Zhang, J.; Yin, J.K.; Yang, Y.L.; Yuan, L.J.; Zhang, L.; Duan, Y.Y. The Optimized Fabrication of Nanobubbles as Ultrasound Contrast Agents for Tumor Imaging. Sci. Rep. 2015, 5, 13725. [Google Scholar] [CrossRef]
- Fan, X.; Wang, L.; Guo, Y.; Tong, H.; Li, L.; Ding, J.; Huang, H. Experimental investigation of the penetration of ultrasound nanobubbles in a gastric cancer xenograft. Nanotechnology 2013, 24, 325102. [Google Scholar] [CrossRef]
- Wu, H.; Rognin, N.G.; Krupka, T.M.; Solorio, L.; Yoshiara, H.; Guenette, G.; Sanders, C.; Kamiyama, N.; Exner, A.A. Acoustic Characterization and Pharmacokinetic Analyses of New Nanobubble Ultrasound Contrast Agents. Ultrasound Med. Biol. 2013, 39, 2137–2146. [Google Scholar] [CrossRef]
- Du Toit, L.; Govender, T.; Pillay, V.; Choonara, Y.E.; Kodama, T. Investigating the Effect of Polymeric Approaches on Circulation Time and Physical Properties of Nanobubbles. Pharm. Res. 2011, 28, 494–504. [Google Scholar] [CrossRef] [PubMed]
- Zhou, T.; Cai, W.; Yang, H.; Zhang, H.; Hao, M.; Yuan, L.; Liu, J.; Zhang, L.; Yang, Y.; Liu, X.; et al. Annexin V conjugated nanobubbles: A novel ultrasound contrast agent for in vivo assessment of the apoptotic response in cancer therapy. J. Control. Release 2018, 276, 113–124. [Google Scholar] [CrossRef]
- Yin, T.; Wang, P.; Zheng, R.; Zheng, B.; Cheng, D.; Zhang, X.; Shuai, X. Nanobubbles for enhanced ultrasound imaging of tumors. Int. J. Nanomed. 2012, 7, 895–904. [Google Scholar] [CrossRef]
- Perera, R.H.; Wu, H.; Peiris, P.; Hernandez, C.; Burke, A.; Zhang, H.; Exner, A.A. Improving performance of nanoscale ultrasound contrast agents using N,N-diethylacrylamide stabilization. Nanomed. Nanotechnol. Biol. Med. 2017, 13, 59–67. [Google Scholar] [CrossRef]
- Hernandez, C.; Nieves, L.; de Leon, A.C.; Advincula, R.; Exner, A.A. Role of Surface Tension in Gas Nanobubble Stability Under Ultrasound. ACS Appl. Mater. Interfaces 2018, 10, 9949–9956. [Google Scholar] [CrossRef] [PubMed]
- De Leon, A.; Perera, R.; Hernandez, C.; Cooley, M.; Jung, O.; Jeganathan, S.; Abenojar, E.; Fishbein, G.; Sojahrood, A.J.; Emerson, C.C.; et al. Contrast enhanced ultrasound imaging by nature-inspired ultrastable echogenic nanobubbles. Nanoscale 2019, 11, 15647–15658. [Google Scholar] [CrossRef] [PubMed]
- Riess, J.G. Reassessment of Criteria for the Selection of Perfluorochemicals for Second-Generation Blood Substitutes: Analysis of Structure/Property Relationships. Artif. Organs 1984, 8, 44–56. [Google Scholar] [CrossRef]
- Kaisers, U.; Kelly, K.P.; Busch, T. Liquid ventilation. Br. J. Anaesth. 2003, 91, 143–151. [Google Scholar] [CrossRef]
- Lowe, K.C. Blood substitutes: From chemistry to clinic. J. Mater. Chem. 2006, 16, 4189–4196. [Google Scholar] [CrossRef]
- Strohm, E.; Rui, M.; Gorelikov, I.; Matsuura, N.; Kolios, M. Vaporization of perfluorocarbon droplets using optical irradiation. Biomed. Opt. Express 2011, 2, 1432–1442. [Google Scholar] [CrossRef]
- Matsunaga, T.O.; Sheeran, P.S.; Luois, S.; Streeter, J.E.; Mullin, L.B.; Banerjee, B.; Dayton, P.A. Phase-Change Nanoparticles Using Highly Volatile Perfluorocarbons: Toward a Platform for Extravascular Ultrasound Imaging. Theranostics 2012, 2, 1185–1198. [Google Scholar] [CrossRef]
- Sheeran, P.S.; Luois, S.H.; Mullin, L.B.; Matsunaga, T.O.; Dayton, P.A. Design of ultrasonically-activatable nanoparticles using low boiling point perfluorocarbons. Biomaterials 2012, 33, 3262–3269. [Google Scholar] [CrossRef]
- Sheeran, P.S.; Dayton, P.A. Improving the Performance of Phase-Change Perfluorocarbon Droplets for Medical Ultrasonography: Current Progress, Challenges, and Prospects. Scientifica 2014, 2014, 579684. [Google Scholar] [CrossRef]
- Sheeran, P.S.; Matsunaga, T.O.; Dayton, P.A. Phase-transition thresholds and vaporization phenomena for ultrasound phase-change nanoemulsions assessed via high-speed optical microscopy. Phys. Med. Biol. 2013, 58, 4513–4534. [Google Scholar] [CrossRef]
- Wong, Z.Z.; Kripfgans, O.D.; Qamar, A.; Fowlkes, J.; Bull, J.L. Bubble evolution in acoustic droplet vaporization at physiological temperature via ultra-high speed imaging. Soft Matter 2011, 7, 4009–4016. [Google Scholar] [CrossRef]
- Zhang, M.; Fabiilli, M.L.; Haworth, K.J.; Fowlkes, J.B.; Kripfgans, O.D.; Roberts, W.W.; Ives, K.A.; Carson, P.L. Initial Investigation of Acoustic Droplet Vaporization for Occlusion in Canine Kidney. Ultrasound Med. Biol. 2010, 36, 1691–1703. [Google Scholar] [CrossRef] [PubMed]
- Ahmadzadegan, A.; Ardekani, A.M.; Vlachos, P.P. Estimation of the probability density function of random displacements from images. Phys. Rev. E 2020, 102, 033305. [Google Scholar] [CrossRef] [PubMed]
- Einstein, A. The motion of elements suspended in static liquids as claimed in the molecular kinetic theory of heat. Ann. Phys. 1905, 17, 549–565. [Google Scholar] [CrossRef]
- Xue, Z.; Charonko, J.J.; Vlachos, P.P. Particle image patternmutual informationand uncertainty estimation for particle image velocimetry. Meas. Sci. Technol. 2015, 26, 074001. [Google Scholar] [CrossRef]
- Leibe, B.; Leonardis, A.; Schiele, B. Robust Object Detection with Interleaved Categorization and Segmentation. Int. J. Comput. Vis. 2008, 77, 259–289. [Google Scholar] [CrossRef]
- Sheeran, P.S.; Wong, V.P.; Luois, S.; McFarland, R.J.; Ross, W.D.; Feingold, S.; Matsunaga, T.O.; Dayton, P.A. Decafluorobutane as a Phase-Change Contrast Agent for Low-Energy Extravascular Ultrasonic Imaging. Ultrasound Med. Biol. 2011, 37, 1518–1530. [Google Scholar] [CrossRef]
- Rapoport, N.Y.; Kennedy, A.M.; Shea, J.E.; Scaife, C.L.; Nam, K.-H. Controlled and targeted tumor chemotherapy by ultrasound-activated nanoemulsions/microbubbles. J. Control. Release 2009, 138, 268–276. [Google Scholar] [CrossRef]
- Sheeran, P.S.; Luois, S.; Dayton, P.A.; Matsunaga, T.O. Formulation and Acoustic Studies of a New Phase-Shift Agent for Diagnostic and Therapeutic Ultrasound. Langmuir 2011, 27, 10412–10420. [Google Scholar] [CrossRef]
- Paefgen, V.; Doleschel, D.; Kiessling, F. Evolution of contrast agents for ultrasound imaging and ultrasound-mediated drug delivery. Front. Pharmacol. 2015, 6, 197. [Google Scholar] [CrossRef]
- Langeveld, S.; Beekers, I.; Collado-Lara, G.; van der Steen, A.; de Jong, N.; Kooiman, K. The Impact of Lipid Handling and Phase Distribution on the Acoustic Behavior of Microbubbles. Pharmaceutics 2021, 13, 119. [Google Scholar] [CrossRef] [PubMed]
- Gregorčič, P.; Zupančič, M.; Golobič, I. Scalable Surface Microstructuring by a Fiber Laser for Controlled Nucleate Boiling Performance of High- and Low-Surface-Tension Fluids. Sci. Rep. 2018, 8, 7461. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vasiukhina, A.; Eshraghi, J.; Ahmadzadegan, A.; Goergen, C.J.; Vlachos, P.P.; Solorio, L. Stable Thermally-Modulated Nanodroplet Ultrasound Contrast Agents. Nanomaterials 2021, 11, 2225. https://doi.org/10.3390/nano11092225
Vasiukhina A, Eshraghi J, Ahmadzadegan A, Goergen CJ, Vlachos PP, Solorio L. Stable Thermally-Modulated Nanodroplet Ultrasound Contrast Agents. Nanomaterials. 2021; 11(9):2225. https://doi.org/10.3390/nano11092225
Chicago/Turabian StyleVasiukhina, Anastasiia, Javad Eshraghi, Adib Ahmadzadegan, Craig J. Goergen, Pavlos P. Vlachos, and Luis Solorio. 2021. "Stable Thermally-Modulated Nanodroplet Ultrasound Contrast Agents" Nanomaterials 11, no. 9: 2225. https://doi.org/10.3390/nano11092225
APA StyleVasiukhina, A., Eshraghi, J., Ahmadzadegan, A., Goergen, C. J., Vlachos, P. P., & Solorio, L. (2021). Stable Thermally-Modulated Nanodroplet Ultrasound Contrast Agents. Nanomaterials, 11(9), 2225. https://doi.org/10.3390/nano11092225








